Abstract
The amino acid esters ethyl glycinate (EG),
Introduction
Osmolytes are interesting candidates for clinical and cosmetic dermal indications since they may compensate for osmotic disturbances in human skin as possibly induced by UV radiation, atopic eczema, or as present in aged skin. In a previous study, several lipophilic prodrugs were shown to be promising candidates for such applications based on in vitro studies in human keratinocytes, comprising ethyl glycinate (EG) and 4 esters composed of α-tocopherol (vitamin E) and different amino acids,
In this study, we report on the investigation of the skin sensitizing potential of these 5 esters. Although the parent compounds are not considered to be relevant skin sensitizing agents, the properties of the esters need further assessment since a rapid and complete cleavage by esterases in the skin is not necessarily taking place. 2 For this purpose, a Local Lymph Node Assay (LLNA) 3,4 was conducted for EG and 2 of the 4 vitamin E esters. Over the last decade, the LLNA that has become a standard method for the evaluation of chemicals with improved predictive capacity of the sensitizing potency as an alternative to traditional guinea pig test.
In addition, for all esters, a mechanistic analysis of structural alerts for protein binding, an important step in the induction of a skin allergy, was performed assisted by an in silico program. 5 Covalent binding of electrophilic skin allergens to nucleophilic moieties of proteins is considered a mechanism by which a majority of skin sensitizers (haptens) exert their adverse effects. The concept of haptenation already dates back to the first half of the last century. 6 It implies that especially small organic molecules must react covalently with skin proteins in order to form complete antigens.
The combined information from the in vivo studies and the in silico analyses was used in a weight-of-evidence (WoE) expert judgment including a read across analysis for the 2 esters that were not tested in the LLNA.
Materials and Methods
Materials
All esters, EG, TMB, TMG, TMP, and TMS, were obtained from Phenion GmbH & Co KG (Frankfurt, Germany) and were synthesized as described previously. 1 Chemical structures of the vitamin E esters are displayed in Figure 1.
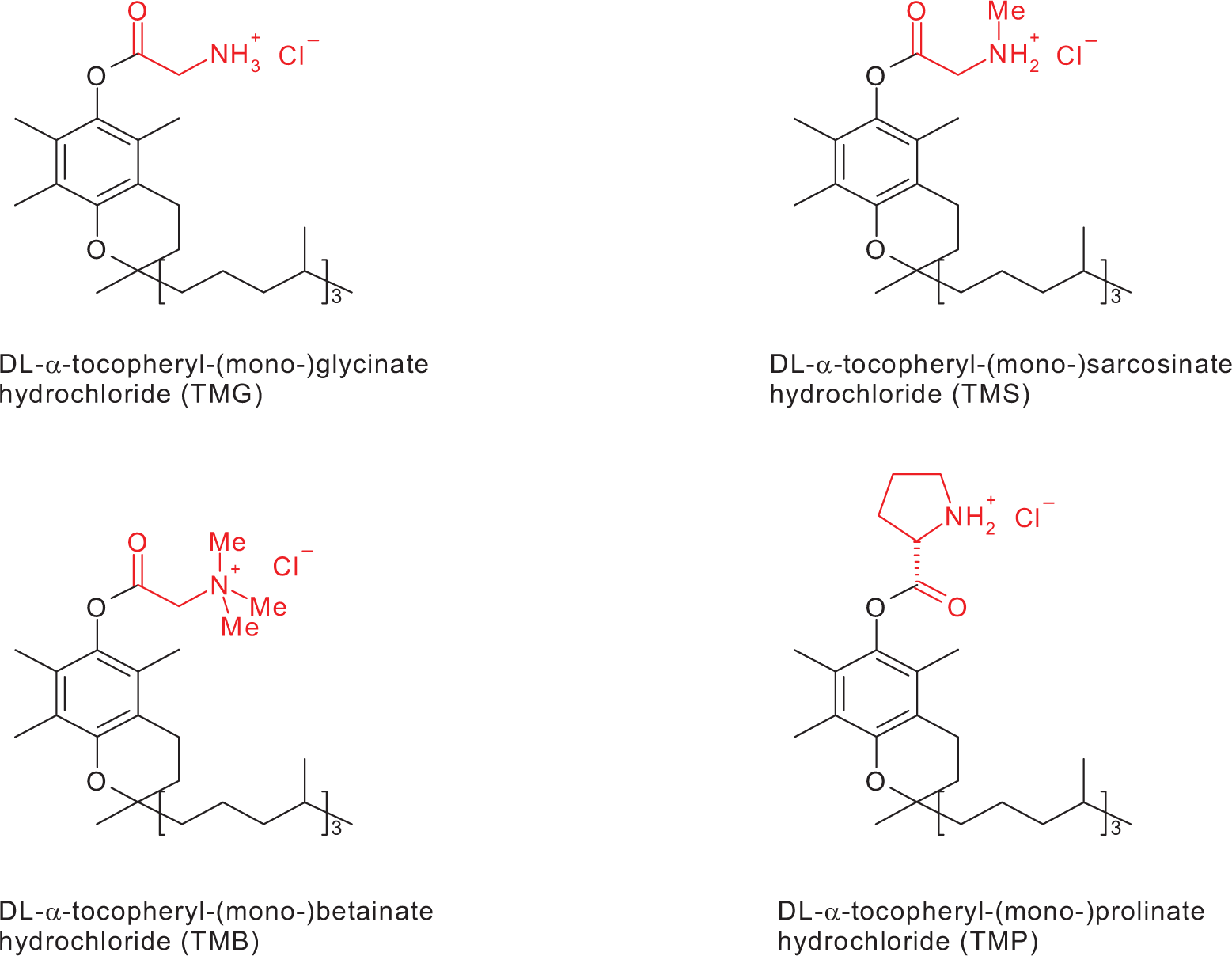
Chemical structures of selected vitamin E esters.
Local Lymph Node Assay
The LLNA was conducted according to OECD TG 429 standard procedures 3 and in compliance with Good Laboratory Practice (GLP). In brief, 3 groups, each composed of 4 female mice (TMP and EG: CBA/CaOlaHsd, TMG: CBA/Jlbm), were treated with 3 different concentrations of the test items by topical application at the dorsum of each ear lobe (left and right) on 3 consecutive days. A control group of 4 mice was treated with the vehicle only. To determine the highest nonirritant and technically applicable test concentration, a non-GLP pretest was performed with 2 mice. Criteria for the choice of vehicle (Table 1) were its suitability to dissolve the test item, the stability of the test item in the solution, and the applicability in the context of the LLNA protocol. Dilutions were prepared freshly before dosing. Five days after the first topical application, mice were intravenously injected into a tail vein with radio-labeled 3 H-methyl thymidine. Approximately 5 hours after intravenous injection, mice were sacrificed and the draining auricular lymph nodes excised and pooled per group. Single-cell suspensions of lymph node cells were prepared from pooled lymph nodes, which were subsequently washed and incubated with trichloroacetic acid overnight. The proliferative capacity of pooled lymph node cells was determined by the incorporation of 3 H-methyl thymidine measured in a β-scintillation counter.
Results of Local Lymph Node Assay
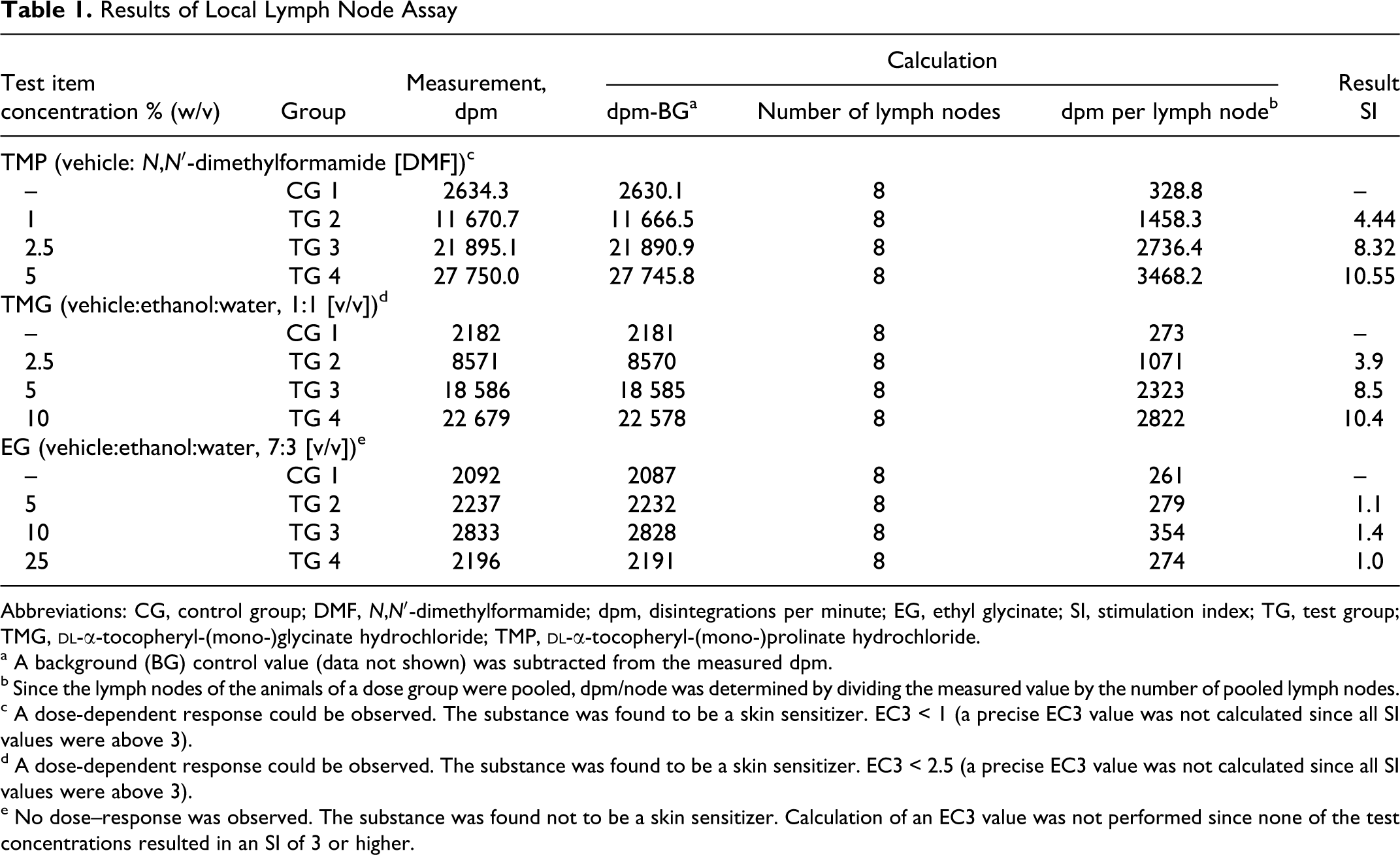
Abbreviations: CG, control group; DMF,
a A background (BG) control value (data not shown) was subtracted from the measured dpm.
b Since the lymph nodes of the animals of a dose group were pooled, dpm/node was determined by dividing the measured value by the number of pooled lymph nodes.
c A dose-dependent response could be observed. The substance was found to be a skin sensitizer. EC3 < 1 (a precise EC3 value was not calculated since all SI values were above 3).
d A dose-dependent response could be observed. The substance was found to be a skin sensitizer. EC3 < 2.5 (a precise EC3 value was not calculated since all SI values were above 3).
e No dose–response was observed. The substance was found not to be a skin sensitizer. Calculation of an EC3 value was not performed since none of the test concentrations resulted in an SI of 3 or higher.
In silico Analysis
Mechanistic in silico analysis was performed with ToxTree in the version 2.5.0. ToxTree is a free of charge, open source application (http://toxtree.sourceforge.net/), which is intended to estimate toxic hazard. The current version ToxTree 2.5.0 is distributed with 14 components for different end points. In particular, structural alerts for skin sensitization are identified using a SMARTS (Smiles Arbitrary Target Specification) pattern-based approach 5 which is used in this study. The relevant structural alerts for protein-binding reactions and indications of the possible mode of action are represented within the profiler included in the program.
Weight-of-Evidence and Read Across
Both WoE and read across are considered as general principles to be employed in regulatory risk assessment, for example, referred to in European cosmetics and chemicals regulations. 7 –9 The term WoE has been used in different ways in the literature. 10 Here WoE is referred to as expert-judgment based on all available information which is relevant for the question under investigation.
Results
Local Lymph Node Assay
The results of LLNA experiments are presented in Table 1. Both TMG and TMP were found to be the sensitizers in this test system, whereas EG was not a sensitizer. Although an exact EC 3 value (estimated substance concentration that produces a stimulation index of 3; threshold for sensitization) could not be determined, the sensitizing potency of TMG and TMP was unambiguous.
In Silico Analysis
As shown in Table 2, all vitamin E esters can act as acyl transfer agents, that is, direct acylation involving a leaving group is the critical electrophilic mechanistic alert. The protein NH2– or SH– groups are good nucleophils that can attack the electrophilic carbonyl carbon of the acetate group (Figure 2). Additionally, TMP, TMB, and TMS share the possibility of a second mechanism, Schiff base formation, due to their substitution pattern around the reactive carbonyl fragment (Figure 3).
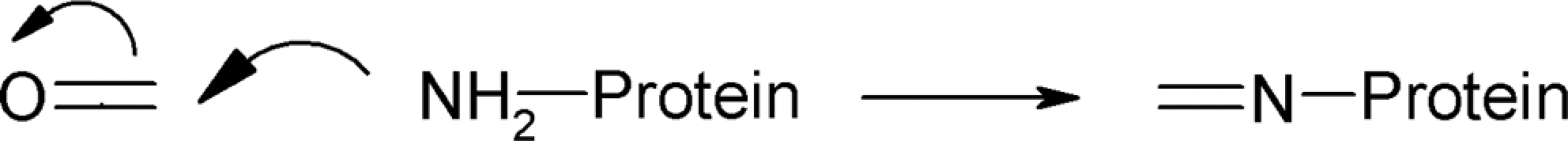
Mechanism of protein acylation.
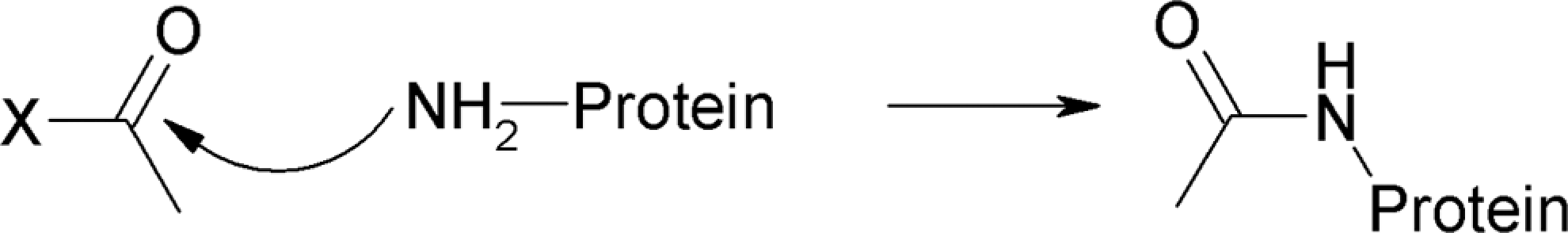
Mechanism of Schiff base formation.
Results From the ToxTree Mechanistic Analysis

Abbreviations: EG, ethyl glycinate; TMB,
Weight-of-Evidence and Read Across
The results of WoE and read across are summarized in Table 3. The skin sensitization potential of TMB and TMS was assessed to be positive based on (
Results From WoE and Read Across
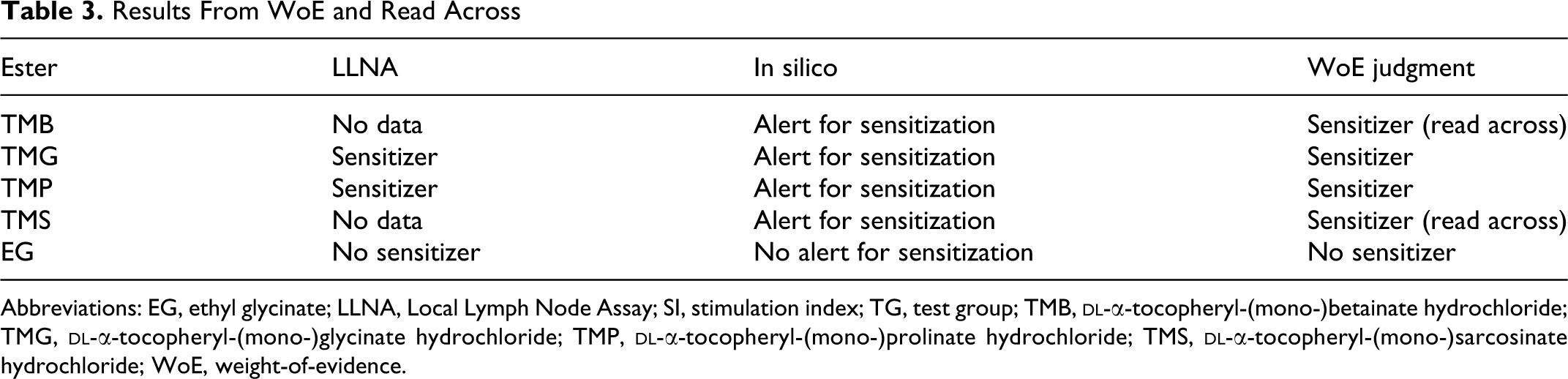
Abbreviations: EG, ethyl glycinate; LLNA, Local Lymph Node Assay; SI, stimulation index; TG, test group; TMB,
Discussion
In this study, we investigated the skin sensitizing properties of a series of esters with osmoprotective properties. We demonstrated how read-across can be used to assess a group of related esters. Although more work is needed to reliably translate chemistry to biological responses via (quantitative) structure–activity relationships, computer-assisted inclusion of mechanistic considerations can now be used to analyze links from chemistry to toxicology. Tools like ToxTree, the program used in this study, or the OECD Toolbox provide an efficient way to perform a screening of potential sensitizing properties. However, a potency estimate similar to the LLNA is not feasible so far, with a major reason being that protein binding reflects only an initial step in the process of sensitization, also termed an “adverse outcome pathway” as currently discussed in the context of chemical grouping at OECD. 11 This limitation is true for the Toxtree program used in this study but also for other in silico tools, for example, the commercially available expert system DEREK or TOPKAT and M-Case.
When experimental data on similar compounds are not directly available to perform a read-across analysis, programs like ChemIDPlus (http://chem.sis.nlm.nih.gov/chemidplus) or the OECD toolbox (www.oecd.org) in the public domain may allow a user to find a set of compounds that share a common substructure or to look for other compounds with structural features that are similar to the target molecule. However, in many cases, retrieved compounds may share features but arrange these structural features differently than the parent and thus give different biological activity. To improve the results of a similarity search, the mode of action should also be taken into account to allow for read across.
A main weakness of available mechanistic models is that they do not contain structural alerts for metabolites which may lead to (de)activation and that they provide limited or no information on potential oxidization products and metabolites. It is believed that the amino acid esters are more or less rapidly split by hydrolase enzymes into an acid and α-tocopherol once they are absorbed into the skin. Furthermore, under use conditions, dermal absorption of the substances will strongly depend on the lipophilicity of the medium, whereby the choice of vehicles in the LLNA tests in addition has to reflect the applicability in the context of the testing protocol.
It has been reported that different vehicles can not only lead to differences in dpm/LN levels but also lead to different classification, for example, the positive control α-hexylcinnamaldehyde, which was a stronger sensitizer when tested in dimethylsulfoxide or acetone/olive oil (4:1 [v/v]) compared with other vehicles including ethanol:water (7:3 [v/v]) and
Besides the in silico tools, there are other alternative methods to animal testing, in vitro and in chemico, that aim to address the very question of the reactivity of potential skin sensitizers. Similar to the in silico programs that predict protein-binding capacity, the concept of in chemico methods, as recently reviewed, is based on the idea that “the rate determining step in the skin sensitisation process is likely to be the reaction of the sensitizer with skin nucleophiles has led to initiatives to develop methods and to generate data on the reactivity of chemicals toward model nucleophiles representing peptide and protein nucleophiles in the skin.” 14 In the same review, ongoing research and validation activities of in vitro methods, addressing various steps in the process, are described.
Interestingly, vitamin E, although generally acknowledged not to be a relevant sensitizer to humans, occasionally produces positive results 15 including the LLNA where it turned out to be a moderate sensitizer with an EC3 (estimated substance concentration that produces a stimulation index of 3, threshold for sensitization) of 7.4. 16 In a previous assessment, other tocopheryl esters were considered not to be sensitizing but LLNA data were not presented. 15 In the light of exonerative results from guinea pig tests, the question remains whether the α-tocopherol and possibly related compounds may be prone to yield “false positive” results in the LLNA. The 2 vitamin E esters tested in this study, however, had a considerably higher potency than vitamin E alone.
As discussed in a recent workshop of the European Partnership for Alternatives to Animal Testing, a future assessment of skin sensitization is thought to combine information from different sources in a WoE approach, including in vitro methods which are currently under development (http://ec.europa.eu/enterprise/epaa/index_en.htm).
Conclusions
Based on the presented information, TMB, TMG, TMP, and TMS are predicted to possess a skin sensitizing potential, and EG not to be a sensitizer. Results from in silico analysis and LLNA were in concordance for all tested esters in this study. It could be shown that read across can be a useful approach to reduce or even replace animal testing with regard to the assessment of specific substances or products. In general, all available information has to be considered within a WoE approach to enable most valid decisions and to minimize testing requirements. Further investigations and experience would be needed to better characterize the application domain of individual methods and combinations thereof.
Footnotes
Acknowledgments
The authors would like to thank Andreas Bock (previously Phenion GmbH & Co KG, now Henkel AG & Co KGaA) for helpful advice, Michael Kock (previously Phenion GmbH & Co KG) for providing test samples for the LLNA, and Elke Lehringer (Henkel AG & Co KGaA) for editorial assistance.
The authors are employees of Henkel AG & Co KGaA.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Henkel AG & Co KGaA and Phenion GmbH & Co. KG.
