Abstract
Historically, formaldehyde was used as a preservative in personal care products to extend product shelf-life; however, given its skin sensitization potential it has been phased out of use and replaced with formaldehyde-releasing preservatives, such as Dimethyloldimethyl hydantoin (DMDMH). A relationship has been established between positive patch test results following exposure to DMDMH and previous sensitization to formaldehyde. Upon direct contact with the skin, formaldehyde can react with skin proteins and cause an acute inflammatory reaction, which may progress to skin sensitization following repeated exposure. This quantitative risk assessment (QRA) aimed to assess the risk of skin sensitization induction following use of shampoo products containing the maximum allowable concentrations of DMDMH in formulation (1% w/v), translating to a free formaldehyde concentration of 0.02%. To determine a margin of safety (MOS) for exposure to DMDMH from use of shampoo products, consumer exposure levels (CEL) were estimated based on typical use scenarios and then benchmarked against an acceptable exposure level (AEL). The AEL was derived using a weight of evidence approach where a range of no expected sensitization induction levels (NESILs) was utilized. The MOS values for a shampoo product containing 1% DMDMH (.02% formaldehyde) was above 1 for the typical use scenario indicating a low likelihood of skin sensitization induction among healthy individuals. Thus, it can be concluded that shampoo products containing DMDMH at or below current allowable concentrations are not expected to increase the risk of skin sensitization induction.
Introduction
Personal care products, such as hair care products and cosmetics, are comprised of numerous chemical ingredients including preservatives to prolong shelf-life and maintain product stability. 1 Formaldehyde, a known contact allergen, has been phased out of use in personal care and cosmetic products in favor of alternative preservatives, some of which release formaldehyde. 2 Dimethyloldimethyl hydantoin (DMDMH) is one such formaldehyde-releasing preservative that is effective as a broad-spectrum antimicrobial agent against fungi, yeast, and both Gram positive and Gram-negative bacteria. 1 Each DMDMH molecule can release 2 equivalents of formaldehyde through a retro-aldol reaction, thereby releasing formaldehyde until equilibrium is reached.1,3,4 Formaldehyde release from DMDMH is dependent on temperature, pH, storage time, and product matrix (eg, aqueous vs organic).1,4 Specifically, DMDMH releases more formaldehyde under alkaline and neutral pH conditions and higher temperatures. Additionally, enhanced release was generally noted in the first hour, although formaldehyde continues to be released up to the maximum of 2% at equilibrium more slowly over time when present in cosmetic matrices.1,4 However, reports indicate that the maximum amount of free formaldehyde can be found in equilibrium with DMDMH is 2% of the total concentration of the ingredient contained in the product (ie, if 1% of DMDMH was utilized a maximum concentration of 0.02% formaldehyde is possible). 1
As of 2002, DMDMH was reportedly present in approximately 963 cosmetic product formulations on file with the United States (U.S.) Food and Drug Administration (FDA) and was considered one of the most frequently used preservatives in the U.S. 5 Aside from DMDMH, other common formaldehyde releasing preservatives include quaternium-15, 2-bromo-2-nitropropane-1,3 diol, diazolidinyl urea, and imidazolidinyl urea.6,7 Reportedly, nearly 25% of cosmetic products in the U.S. and Europe contain at least one the aforementioned preservatives. 7 DMDMH was most frequently found in shampoos, followed by skin care products, hair conditioners, make-up foundations and bases, hair rinses, and personal cleanliness products. 8 Currently, DMDMH is regulated by the European Union (EU) regulation EC No 1223/2009, and is permitted up to a maximum concentration in ready for use preparations of 0.6%. 9 Further, in 2021 the EU proposed reducing the total free formaldehyde in a product to 0.001% (10 ppm) to “protect the vast majority of consumers sensitized to formaldehyde.” 10 In the U.S., DMDMH has been reported to be utilized in cosmetic and personal care products in concentrations of up to 1%. 1 Free formaldehyde can be found in various non-oral cosmetic and household products, however, is allowed to be present up to only 0.2% (2000 ppm).2,9 Any finished cosmetic products with formaldehyde concentrations exceeding 0.05% must be labelled with the warning “contains formaldehyde.” 9 Although the U.S. FDA lists DMDMH as a common allergen in cosmetic products, it does not regulate the use or allowable concentration in cosmetic products. 11 Similarly, Canada does not regulate DMDMH directly, but regulates the amount of formaldehyde that may be present in the final cosmetic product.12,13 Specifically, according to Canadian regulation, any non-oral (or non-aerosol) cosmetic product may not contain concentrations of formaldehyde greater than 0.2%.12,13
According to an evaluation conducted by the Cosmetic Ingredient Review (CIR) Expert Panel, DMDMH as a cosmetic ingredient was determined to be “safe as used,” although no maximum safe concentration was specified.1,5,14 It was reported that DMDMH was safe in the practices of use and that there is indication that current concentrations found in various personal care products, range from 0.00001 to 0.8% DMDMH, and specifically in shampoos at 0.0005 to 0.6%. 5 This concentration is slightly lower than what was reported in the 1980s, however, it cannot be confirmed as most product labels do not explicitly provide ingredient concentrations. On the contrary, formaldehyde is classified by the CIR Expert Panel as unsafe for use in cosmetics if the concentration exceeds 0.074% (w/w).7,15 Specifically, it was noted that formaldehyde is unsafe in present practices in hair smoothing or straightening products due to the application of heat (eg, blow-drying or flat ironing), which may cause the release of formaldehyde gas from the product solution.4,15 The FDA and Occupational Safety and Health Administration (OSHA) have also issued warnings regarding formaldehyde or formaldehyde-releasing ingredients found in several hair smoothing products where heat is applied following application.
Free formaldehyde concentrations as low as 0.2% are reportedly sufficient to provoke allergic contact dermatitis. 2 Contact allergy caused by formaldehyde is a common occurrence, with prevalence rates of sensitization to formaldehyde reported to be approximately 2%-3% in Europe and 8%-9% the U.S.16-18 A few studies have demonstrated a cross-reactivity between DMDMH and formaldehyde allergies.8,19 Pre-sensitization to formaldehyde may explain adverse reactions to DMDMH. 8 Patch testing results indicate that aqueous solutions of DMDMH at concentrations similar to those in cosmetic products, contained enough free formaldehyde to elicit dermatitis in those allergic to formaldehyde. 8
Formaldehyde acts as an effective preservative through the alkylation of proteins associated with microorganisms resulting in irreversible chemical modification inhibiting cell division and proliferation.20,21 When skin is exposed to formaldehyde, such as through cosmetics products containing formaldehyde-releasing preservatives, proteins in the skin can react with formaldehyde to create modified proteins that can cause an acute inflammatory response; this response may ultimately result in skin sensitization if repeated exposure occurs. 22 Thus, as the number of personal care products containing DMDMH and other formaldehyde-releasing preservatives increases, an understanding of potential health effects associated with its use is needed.
A quantitative risk assessment (QRA) was conducted to assess the risk of skin sensitization induction following exposure to formaldehyde from the use of shampoo products containing the formaldehyde-releasing preservative DMDMH at the maximum allowable concentration. Given that up to 2% free formaldehyde is present in equilibrium with DMDMH, this QRA assesses shampoo products expected to contain 0.02% formaldehyde to determine if current use scenarios result in an increased risk of skin sensitization induction in consumers.
Methods
Identification of DMDM Hydantoin Quantities in Shampoo Products
A survey of various personal care product formulations was conducted to determine the concentration of DMDMH, which was then used to calculate the maximum concentration of resulting free formaldehyde. Since DMDMH was most frequently found in shampoos, the scope of this analysis was limited to shampoo products. A literature search was conducted primarily using PubMed (https://pubmed.ncbi.nlm.nih.gov/) and Google Scholar (https://scholar.google.com/) with search terms including “DMDM hydantoin,” “1,3-dimethylol-5,5-dimethyl hydantoin,” “1,3-Bis(hydroxymethyl)-5,5-dimethylimidazolidine-2,4-dione,” “dimethyloldimethyl hydantoin” in combination with “shampoo,” “personal care products,” “personal hygiene products,” or “cosmetics.” Relevant references cited in primary references identified using these search criteria were also reviewed where applicable. Additionally, available governmental agency use restrictions were reviewed to determine permitted maximum use concentrations.
Consumer Exposure Assessment
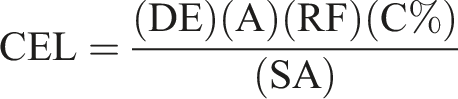
Sensitization Benchmark Identification
The no expected sensitization induction level (NESIL), expressed in dose per unit area of exposed skin (μg/cm2), is a benchmark value unique to skin sensitization. 23 It is defined as the quantitative threshold exposure level (ie, dose) that does not induce skin sensitization in humans. 24 In an effort to select the most appropriate NESIL for formaldehyde, a weight of evidence approach was used to assess the available data for formaldehyde sensitization, which included data from a local lymph node assay (LLNA) in mice and human repeat insult patch test (HRIPT) data.25,26
The HRIPT study by Marzulli and Maibach 25 measured the frequency of sensitization responses following induction with solutions ranging from 0.1 to 10% formalin (corresponding to 0.037 to 1.9% formaldehyde). Although Marzulli and Maibach 25 did not report a statistically significant increase in sensitization responses with increasing concentrations of formalin (P = .12), sensitization responses were reported in animals induced with 1% (0.37% formaldehyde, corresponding to 370 μg formaldehyde/m2), but not 0.1% (0.037% formaldehyde, corresponding to 37 μg formaldehyde/m2) solutions of formalin. Benchmark dose (BMD) analysis was used to further refine the NESIL between these concentrations. Specifically, the U.S. Environmental Protection Agency’s (EPA’s) Benchmark Dose Software (BMDS) 3.2 (U.S. EPA Benchmark Dose Software 2020, Version 3.2) was utilized to model dose-response relationships between chemical exposure or dose and the sensitization response to identify the BMD and its 95% lower confidence limit (BMDL), or concentration associated with a specified benchmark response (BMR). A BMR of 1% extra risk was used for this analysis to account for the sensitive nature of the effect. Dose-response models were selected based on the U.S. EPA BMDS guidance. 27
No Expected Sensitization Induction Levels (NESILS).

Abbreviations: HRIPT, human repeat-insult patch test; NOAEC, no expected adverse effect concentration; BMDL, benchmark dose low.
Risk Characterization
Acceptable exposure limits (AELs) were calculated by applying sensitization assessment factors (SAFs) to the most appropriate NESILs identified. The SAFs are product-specific and account for variations and uncertainty when extrapolating from an experimental NESIL to a real-life consumer exposure scenario.23,24,29 For example, SAFs may account for interspecies extrapolation (animal to human), variability between individual consumers, differences in product matrix or composition, and differences in frequency and duration of product use, among others.24,29,30
A margin of safety (MOS) was determined by comparing the calculated CEL to the AEL as follows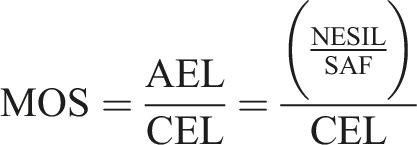
The MOS is indicative of likelihood of sensitization induction resulting from exposure to the ingredient of interest under a specific use scenario. An MOS above 1, which results when the CEL is less than the benchmark AEL, suggests a low likelihood of increased risk of skin sensitization following exposure to formaldehyde released from shampoo containing DMDMH.
Results
Given the limited documentation in the literature regarding actual concentrations of DMDMH used in personal care and cosmetic products, the maximum allowable concentration of 1% DMDMH was selected for calculation of the CEL. This concentration of DMDMH corresponds to a maximum formaldehyde concentration (C%) of 0.02%, assuming that up to 2% free formaldehyde is in equilibrium with the DMDMH in the product solution. 8 It is important to note that this is a worst case-scenario in which the maximum DMDMH was used and it is likely that a much lower concentration of DMDMH is needed to work as an effective preservative within the product. Additionally, this assumed that the ingredient was not affected by the medium (shampoo) in which it is in. It is likely that a lower concentration is used within products, as illustrated by a few studies.31,32 Taken together with the selected values for each parameter shown in Table 1, a mean CEL of 0.0072 μg/cm2 was calculated.
To account for variability in susceptibility between individuals, multiple NESILs from both human and animal testing was selected for calculation of the AEL (Table 2). A NESILhigh of 37 μg/cm2 was selected based on the NOAEC derived from the HRIPT performed by Marzulli and Maibach. 25 Due to limitations in the HRIPT conducted by Marzulli and Maibach, 25 such as the relatively small sample size and lack of an observed dose-response relationship following formaldehyde exposure, BMD modeling was conducted to further elucidate the most appropriate NESIL for formaldehyde. Specifically, a BMDL01, or NESILlow, of 10 μg/cm2 was derived from a Dichotomous Hill model; this model was selected as it was the only model that fit the full range of the dose-response data (see Supplemental Material). The model illustrates that the responses to doses utilized in the HRIPT may be attributable to background risk, and accounts for the plateaued responses at concentrations above 0.1% formalin (corresponding to 37 μg formaldehyde/m2).
Safety Assessment Factors (SAFs).

The parameters utilized in the calculation of the CEL are based on a typical use scenario and are outlined in Table 1. Given that the mean amount of product per application based on the surface area of the scalp (14.7 mg/cm2) exceeded the maximum skin adherence amount, the amount of product per application area was limited to 10 mg/cm2 (IH SkinPerm v2.0 Reference Manual). The calculated CEL based on the maximum allowable DMDMH concentration used in shampoo products was found to be 0.22 μg/cm2.
Risk Characterization.

Discussion
This study sought to evaluate the risk of skin sensitization induction following use of a shampoo product containing approximately 1% DMDMH, which corresponds to approximately 0.02% formaldehyde. The maximum concentrations of DMDMH used in personal care products are 1%, with reported concentrations in shampoo products specifically ranging from 0.0005 to 0.6%.1,5 This study utilized a typical use scenario for a rinse-off shampoo product that contained up to the maximum of 2% free formaldehyde released from DMDMH at equilibrium. Benchmarking the CEL against both the low and high AELs resulted in MOS values greater than 1. These results suggest that there is a low likelihood of skin sensitization induction under the specific DMDMH use scenario evaluated.
It should be noted that some of the MOS values are slightly above 1 for worst-case scenarios in which the rinse-off product evaluated contained the maximum amount of free formaldehyde (2%) released from the maximum allowable DMDMH concentration (1%) in comparison to a conservative NESIL (ie, NESILlow) derived from BMD methodology. While it is unlikely that products will contain the maximum of 0.02% free formaldehyde (1% DMDMH × 2% free formaldehyde at equilibrium), this situation was evaluated to assess the risk of skin sensitization under a worst-case consumer exposure scenario. The resulting MOS value of 1.5 for a worst-case consumer use scenario suggests that at the maximum concentration of use, the exposure is approaching a level where there may be an increase in risk of skin sensitization induction. However, MOS values for more realistic consumer exposure scenarios are above 1 and suggest a lack of concern for increased skin sensitization induction to DMDMH and the released formaldehyde.
Additionally, the selected BMD modeling approaches were based on conservative assumptions and used a sensitive BMR of 1% response. Because only one model (the Dichotomous Hill model) adequately fit the data, additional modeling was considered in which the highest tested concentration of 10% formalin (corresponding to 1900 μg/cm2 formaldehyde), as reported by Marzulli and Maibach, 25 was removed from the quantitative analysis. This approach is in accordance with US EPA’s BMDS guidance. 27 In these sensitivity analyses, linearized models (such as the log-logistic, multistage, and quantal linear models) better described the underlying data and estimated a BMDL01, or NESILlow, of approximately 110 μg/cm2 (see Supplemental Material). This NESILlow is approximately 10-fold higher than that derived from the full range of dose-response data, and would correspond to a further increased MOS.
One of the most crucial pieces of information in a QRA is the NESIL, as it functions as a point of departure for the risk assessment of a potential allergen. It is important to employ a weight of the evidence approach, which includes consideration of all available data (eg, in silico, in vitro, nonclinical animal models, and human data), when deriving or determining the most appropriate NESIL for use in the AEL calculation. 29 In general, EC3 values obtained from nonclinical LLNAs 30 correlate with non-sensitizing levels of exposure to a potential allergen obtained from HRIPTs. However, LLNA EC3 values may also over- or under-estimate the true NESIL in humans if analyzed in a vacuum. 24 Conversely, human sensitization testing (eg, HRIPT) is generally intended to confirm the lack of sensitization following an exposure level (eg, a level that consumers may be exposed to via product use) that has been identified as highly unlikely to induce sensitization. 24 As such, a scientifically sound NESIL is one that considers all lines of available evidence, with more weight placed on exposures in the most relevant species to the risk assessment (eg, humans) and on effect levels obtained from high quality assays. Specific to formaldehyde, EC3 values of 29 and 45 μg/cm2 were conservatively estimated from data gathered in short-term LLNA studies in mice.26,28,33 These LLNA EC3 values were in alignment with the NOAEC of 37 μg/cm2 estimated from the HRIPT results presented by Marzulli and Maibach, 25 which was selected as one of the NESILs in this QRA. However, analysis of formaldehyde patch testing results is considered challenging due to the high number of both false positive and false negative reactions. 16 Given limitations in the patch test study design of Marzulli and Maibach 25 and the available human patch test data for formaldehyde, a BMD approach was utilized to more accurately estimate the true NESIL for formaldehyde based on patch test data from humans. The estimated BMDL01 (ie, NESIL) of 10 μg/cm2, which is derived using a conservative BMR of 1%, provides a conservative estimate for a NESIL, thus allowing for a conservative risk assessment approach.
The DMDMH QRA included additional assumptions that align with a conservative risk assessment approach. First, the CEL calculation included an assumption that the surface area of the scalp in contact with the product was the maximum skin adherence value of 10 mg/cm2 (IH SkinPerm v2.0 Reference Manual). Additionally, calculation of the AEL involved the application of an SAF to the selected NESIL to account for various uncertainties when extrapolating from a controlled exposure scenario to a real-life consumer use scenario. Application of a SAF of 30, which includes a standard AFH of 10 to account for individuals with increased susceptibility (Table 3), 30 was included based on suggestions presented in regulatory documents. While the SAF of 30 was deemed appropriate for products applied to the hair with some hand contact by multiple sources, application of a higher SAF to the AEL could result in MOS values of <1 for some DMDMH use scenarios.
It is important to note that this analysis applies only to individuals who have not been previously sensitized to formaldehyde. Because formaldehyde is a common and ubiquitous allergen, the threshold that is not associated with adverse dermal reactions is unclear, particularly in previously sensitized individuals.16,17 Thus, it is often difficult to establish a true causal relationship between one particular formaldehyde-releasing preservative and a positive sensitization reaction, such as acute contact dermatitis (ACD). 16 The NESIL for formaldehyde in previously sensitized individuals may be lower than that selected for this QRA, although there are inherent uncertainties and limitations when attempting to quantitate the true NESIL for previously sensitized individuals. The level at which previously sensitized individuals experience a reaction to a contact allergen (sensitizer) is the skin sensitization elicitation threshold. Individuals that have already undergone the induction phase of skin sensitization may experience redness, swelling, itching, and blistering of the affected skin upon subsequent exposure to the sensitizer.34,35 While it has been suggested that the threshold for skin sensitization elicitation is lower than that of induction, a study performed in mice suggests that the exact concentration that elicitation occurs cannot be easily extrapolated from known induction levels. 36 It has been recommended that case by case scenarios and studies be explored as all sensitizers have unique properties that may cause differences in the relationships between elicitation and induction. 37 Thus, the current analysis is limited to induction of skin sensitization following exposure to formaldehyde released from DMDMH. Additionally, it should be noted that exposure to a sensitizer does not guarantee that an individual will be sensitized. Further, every individual that is sensitized does not develop allergic contact dermatitis or experience sensitization elicitation upon subsequent exposure to the sensitizer. 38
Previous studies have shown that patients with positive patch test reactions to DMDMH were often also allergic to formaldehyde.8,19,39,40 For example, de Groot et al 8 reported that patients with stronger allergies to formaldehyde were more likely to have positive reactions to lower concentrations of DMDMH, but did not reaction to DM hydantoin, which does not release free formaldehyde. However, the authors concluded that there was not convincing evidence to support the allegation that DMDMH allergies were solely based on formaldehyde sensitivity. 8 Other studies have demonstrated that cross-reactivity to formaldehyde was limited in patch tests that included both DMDMH and formaldehyde solutions, such that patients had an isolated reaction to DMDMH without co-reactivity to formaldehyde. 41 These discrepancies further demonstrate the difficulties and challenges in analyzing patch test results and determining the true source of sensitization reactions. 16
A potential limitation of this study is the focus on exposure to formaldehyde from a single DMDM-containing product. In reality, consumers are likely to be exposed to formaldehyde through the use multiple personal care products that contain formaldehyde from one or more formaldehyde-releasing preservatives. These products may be used at different times throughout the day or concurrently on different skin contact surface sites. The present study does not consider potential increased risk of skin sensitization induction due to aggregate exposures. Given that skin sensitization involves a systemic immune response, it is possible that use of a product containing DMDMH, another formaldehyde-releasing preservative, or formaldehyde itself on a location of the body remote from the scalp could induce sensitization that may lead to future scalp reactions to products containing these ingredients. Additionally, the QRA only evaluated rinse-off products containing DMDMH and did not include leave-on products, such as lotions, oils, makeup, and fragrances, which have been reported to contain the formaldehyde-releasing preservative. 5 Evaluation of leave-on products was outside the scope of the current analysis, as these products may result in longer exposure durations and potential absorption of the preservative and/or formaldehyde that would require a unique risk assessment focused on potential systemic effects following dermal exposure.
Given the current gap in understanding regarding the concentration of formaldehyde releasers and formaldehyde that is found in cosmetic products, future studies could aim to quantify ingredients like DMDMH in a variety of products. Quantification of formaldehyde releasers in products could assist in better understanding the inherent risk to consumers as the current study assumes an unlikely worst-case scenario. Additionally, when specifically considering exposure to formaldehyde-releasing preservatives via the scalp, the role that hair, or the lack thereof, plays in the interactions that take place between applied products and the scalp is largely not understood. Understanding these interactions could aid in understanding if some individuals are more susceptible to certain ingredient exposures.
Conclusions
Given that the calculated MOS value for the typical use scenario was above 1, it can be concluded that shampoo products containing DMDMH at or below current allowable concentrations are not expected to increase the risk of skin sensitization induction in healthy individuals not previously sensitized to formaldehyde.
Acknowledgments
Footnotes
Acknowledgements
The authors would like to thank Kenneth Unice for his review, comments, and assistance in preparing this manuscript.
Author Contributions
Authorship Statement Stewart, C. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Parker, J. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Hwang, R. contributed to conception, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Vincent, M. contributed to analysis and interpretation, drafted manuscript, critically revised manuscript; Fung, E. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
All the authors are or were employed by Stantec ChemRisk, a consulting firm that provides scientific advice to the government, corporations, law firms, and various scientific and professional organizations. One of the authors (EF) has served as an expert witness in legal matters involving DMDM Hydantoin.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

