Abstract
In this study, an in vivo toxicological safety assessment of
Introduction
Probiotics are “beneficial live microorganisms which, when administered in adequate amounts, confer a health benefit to the host.” 1 Within the last decade, the supplementation of probiotics to various food products and as therapeutic agents against several infectious diseases has increased tremendously. 2 The health benefits of probiotics include inhibition of gastrointestinal pathogens; aid in digestion and nutrient absorption; prevention of infectious diarrhea, irritable bowel syndrome, constipation, gas, or lactose intolerance; and contribution to immune function. Furthermore, other clinical investigations document further benefits such as lowering cholesterol levels, preventing dental caries, delaying of allergies in children, and treatment and prevention of vaginal and urinary infections in women. 3 Probiotics can also serve as an alternative to commonly used therapeutic and prophylactic avenues for increasing incidence of antibiotic resistance and a growing prevalence of cancer and allergic conditions in an aging population.
The majority of the probiotics studied and commercialized are lactic acid bacteria (LAB) and bifidobacteria.
4,5
In contrast, only very few species of the spore-forming
Some of the
Only limited information is available about the in vivo as well as in vitro safety evaluation of
Materials and Methods
Chemicals and Reagent Kits
Assay kits for determination of urea, creatinine, bilirubin, aspartate aminotransferase (SGOT), alanine amino transferase (SGPT), lactate dehydrogenase (LDH), creatinine kinase (CK Nac), and alkaline phosphatase (ALP) were obtained from Aspen Laboratories Pvt Ltd, India. Assay kit for analysis of glucose, cholesterol, and triglycerides (TG) were from Span Diagnostics Ltd, India. Kits for determination of sodium, potassium, and chloride were obtained from Coreal Clinical Systems, India. All other chemicals and solvents used for this study were of the highest purity obtained from Merck Pvt Ltd, India.
Preparation of Test Article and Diet
The culture
Experimental Rats
Animal care and handling conformed to the guidelines of the Committee for the purpose of control and supervision of experiments on animals (CPCSEA), Government of India, and the protocols were approved by the Institutional Animal Ethical Committee (IAEC). The albino Wistar rats bred in the Central Food Technological Research Institute (CFTRI) animal house facility were housed in stainless steel cages (1 rat/cage) with a 12-hour light/dark cycle in a controlled atmosphere viz temperature 22°C ± 3°C and a relative humidity of 60% to 70%. The animals were fed ad libitum and had free access to drinking water during the experimental period. The drinking water was periodically analyzed and was determined to be free of contaminants. The animals were acclimatized to above experimental conditions for 5 days prior to the start of dosing for any toxicity study.
Acute Toxicity Study
An acute toxicity study was performed in accordance with Organization for Economic Cooperation and Development (OECD) Guideline for the Testing of Chemicals No. 423; Acute Oral Toxicity—Acute Toxic Class Method, adopted December 17, 2001. Twelve healthy adult male albino Wistar rats (220 ± 10 g) were randomly assigned into 2 groups (6 per group), namely the control group which received the vehicle alone and the probiotic-treated group which received test article of 1000 mg/kg BW of rat, as a single dose. This dose level corresponds to 1.1 × 1011 CFU/kg BW of rat and was selected based on previous references that have evaluated the in vivo safety of probiotic bacteria. 25,26,29 No female rats were used for acute toxicity study. Following the initial dose, the animals were observed with respect to general behavior, signs of toxicity, and mortality for 4 continuous hours and then twice per day over a period of 14 consecutive days. The general behavior, signs of toxicity include changes in the skin and fur, eyes and mucous membranes, and also respiratory, circulatory, digestive, autonomic, and central nervous systems. Observations were also made on behavior patterns, such as tremors, convulsions, salivation, stool consistency, lethargy, sleep and changes in gait, posture, and response to handling. Daily feed and water intake was recorded throughout the experimental period and the BW of the rats were documented on days 0, 4, 7, 10, and 14. On the 15th day of the experiment, following an overnight fast, the animals were weighed and sacrificed with ether anesthesia. The blood samples were collected in sterile conditions by cardiac puncture in ethylenediaminetetraacetic acid (EDTA-2K) containing tubes for hematological analysis and in nonanticoagulant tubes for serum biochemical investigations. The vital organs were weighed and relative organ weights (g/100 g BW) were calculated. A gross pathological organ examination was performed. Histopatholological analysis (if necessary) was done for any treatment-related abnormalities.
Subchronic Toxicity Study
Male and female albino Wistar rats bred in the CFTRI animal house facility were selected and randomly split into 3 groups, each comprising 6 males and 6 females, weighing 36 ± 1 g. The first group was kept as control and fed only the basal diet. Experimental diet consisting of basal diet supplemented with test article at 2 dose levels 100 and 1000 mg/kg BW/d (which corresponds to 1.1 × 1010 and 1.1 × 1011 CFU) was fed to groups 2 and 3, respectively, for 13 weeks. Test diet and uncontaminated water were available ad libitum throughout the experimental period. At the end of the experiment, all surviving animals were fasted overnight before anesthetization with diethyl ether and sacrificed. The results were analyzed according to the OECD Guideline for the Testing of Chemicals No. 408; Repeated Dose 90-Day Oral Toxicity Study in Rodents, adopted September 21, 1998.
Feed and water intake, clinical observations, and BW
The feed intake was recorded daily and from this, the mean daily feed intake for each week was calculated. The water intake by the rats was also noted. All the animals were examined for general behavior, signs of toxicity, and mortality, twice daily, as in the case of acute toxicity study. Body weights were measured at the initiation of the experiment and then at weekly intervals. Before sacrifice, the final BW of the rats was also recorded following overnight fasting.
Viability of B licheniformis Me1
Fresh fecal samples were collected from each test group along with the control group on randomly selected days (10, 30, 50, and 80). This was done to confirm that the administered test article survived the stress within the GIT. The feces samples were homogenized in normal saline (0.85% NaCl) and serially diluted. The diluted homogenates (0.1 mL) were spread plated on LB agar plates for the enumeration of
Hematology and biochemical studies
At the end of the experiment, all the surviving animals were fasted overnight before anesthetization and necropsy. Blood samples were collected in 2 centrifuge tubes: one pre-filled with EDTA-2K as an anticoagulant and other without any supplement. The hematological parameters determined with an automated hematology analyzer, K-4500 (Sysmex Corp, Japan), included white blood cells (WBCs), red blood cells (RBCs), hemoglobin (HGB), mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), MCH concentration (MCHC), platelet count (PLT), and differential leukocyte count. For measuring the differential leukocyte count, blood samples were mixed with 1/4 volume of 5.0% EDTA-2K and analyzed with a Microx HEG-120A (Omron Tateishi Electronics Co, Ltd, Tokyo, Japan). The non-EDTA-2K-treated blood samples were used for a biochemical examination of the serum. These samples were used to determine the levels of glucose, triglycerides, cholesterol, urea, creatinine, and for the activity of ALP, SGPT, SGOT, and LDH by using standard kits. Blood biochemistry determinations were performed manually with a spectrophotometer (Shimadzu, Japan).
Relative organ weights and histopathological analysis
Gross observations were made at necropsy and recorded. Before further histopathological examinations, the organ weights of liver, lungs, kidneys, heart, spleen, testis/ovary, epididymis/uterus, brain, and adrenals were weighed. The relative organ weights were calculated based on the final BW of the rats. At necropsy the vital organs were surgically removed from the rats, washed with normal saline, fixed, and preserved in 10% neutral phosphate-buffered formalin. Collected tissues were grossly and microscopically examined during histopathological examination according to OECD guidelines. All the major tissues were further processed and trimmed, embedded in paraffin, sectioned to a thickness of 4 µm and stained with hematoxylin and eosin, 30 and analyzed by light microscopy.
Micronucleus Assay in Mice
Adult male and female mice (Swiss Albino,
Acute Eye Irritation Study in Rabbits
Male albino rabbits weighing 2500 ± 250 g from our animal house facility were used for the study. Animals were housed individually in metal cages at 20°C ± 3°C, relative humidity of 40% to 70% and at a 12-h light/dark cycle. Animals were fed ad libitum with conventional laboratory diet. Tap water was routinely analyzed for contaminants and was also available ad libitum. Three healthy male albino rabbits were used for acute eye irritation study. Both eyes of the animals were examined 24 hours prior to the start of the study. Only animals not showing any ocular defects or preexisting corneal injury were used in the experiment. The dosage of test article (0.1 g of the undiluted
Acute Skin Irritation Study in Rabbits
The conditions for housing, feeding, and drinking of the rabbits were the same as described above. For the acute skin irritation experiment, 3 male albino rabbits were used. The fur was removed from a small area of the animal’s trunk, approximately 24 hours before the test. An undiluted dose of 0.5 g of the test article (corresponding to a concentration of 1.1 × 1013 CFU/g) was moistened sufficiently with water to ensure good contact with the skin and applied to a small skin area (approximately 6 cm2) of 3 animals and covered with sterile gauze patches, held in place with nonirritating tape. Untreated skin areas of each animal served as the control. After 4 hours of exposure, the test article was removed from the animal’s skin by washing with water. Animals were examined for erythema and edema at 1, 24, 48, and 72 hours after the removal of the test article. The test article was evaluated according to the Draize (1959) method (OECD 404, 2002) for any skin irritant effect. The study was performed in accordance with the OECD Guidelines for Testing of Chemicals No. 404; Acute dermal irritation/Corrosion, adopted April 24, 2002.
Statistical Analysis
Statistical analysis of the data was performed with SPSS Software (version 16.0). Comparison of results between control and treatment groups of male and female groups were separately carried out by 1-way analysis of variance (ANOVA), and a post hoc analysis of individual pair difference was performed by Duncan multiple range tests. All the data are presented as mean ± standard error of the mean (SEM). A
Results
Acute Oral Toxicity Study
A 14-day oral acute toxicity study in adult male albino Wistar rats was performed to investigate the short-term effect of the test article,
Subchronic Oral Toxicity in Rats
Mortality and clinical symptoms
A 13-week repeated-dose toxicity study was performed in rats for determination of a no-observed-adverse-effect level (NOAEL) and used to establish a safe chronic oral dose for humans. During this experimental period, all the animals survived the test article administration. Animals of both treated and control groups appeared and behaved normal in their cages throughout the experimental period, with no clinical signs of toxicity or allergic reactions. Neither treatment-related incidence of diarrhea, colonic effects, or stomach irritation, constipation, or other gastrointestinal disorders nor changes in locomotors activity, respiratory, circulatory, autonomic and central nervous system were observed.
Feed and water intake and BW
Table 1 shows the daily feed intake of male and female rats over 13 weeks. Male rat group fed with a test dose of 1.1 × 1011 CFU/kg BW showed a significantly higher (
Daily Feed Intake of Male and Female Wistar Rats Fed With Test Article for 13 Weeksa,b
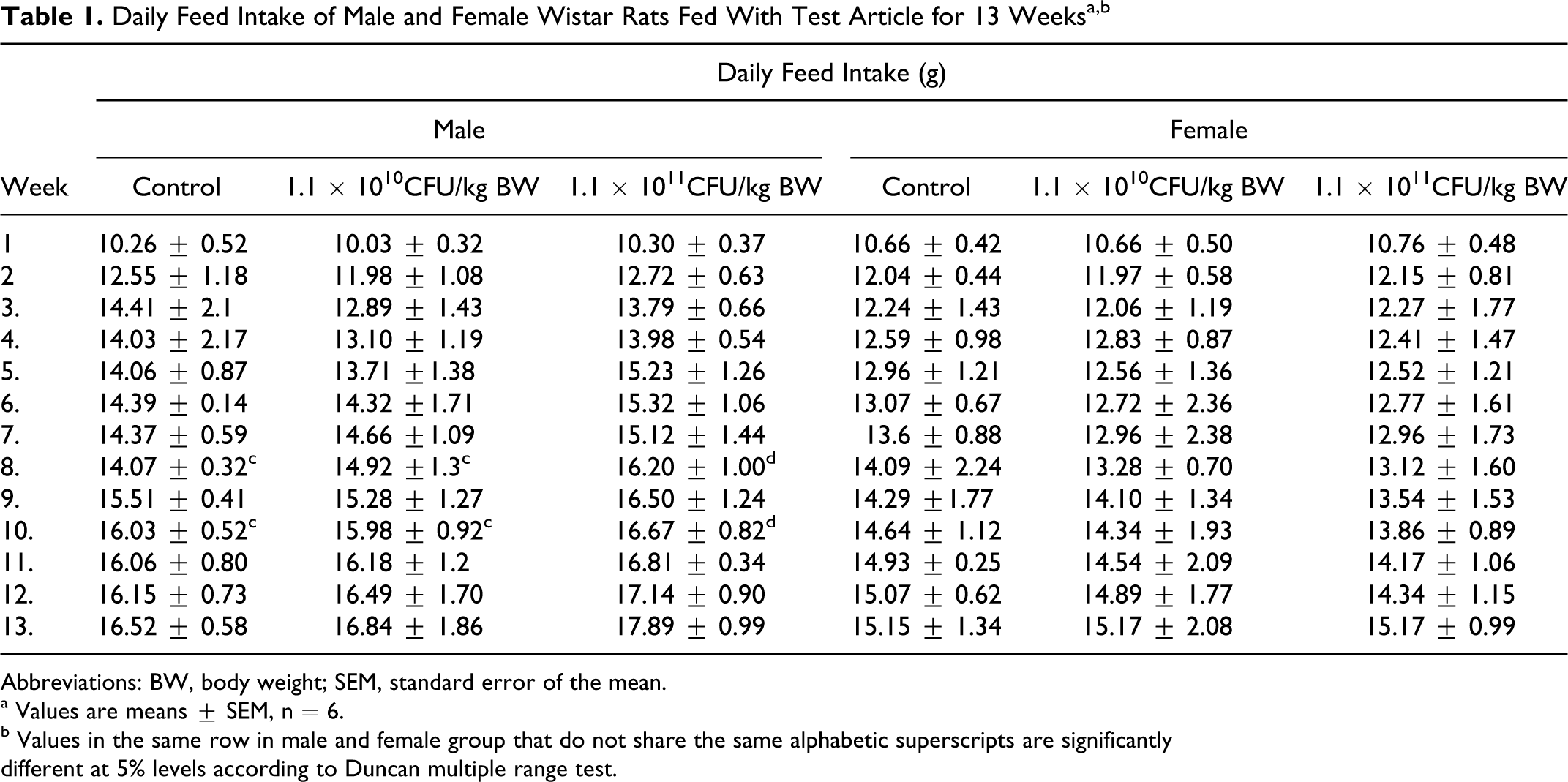
Abbreviations: BW, body weight; SEM, standard error of the mean.
a Values are means ± SEM, n = 6.
b Values in the same row in male and female group that do not share the same alphabetic superscripts are significantly
different at 5% levels according to Duncan multiple range test.
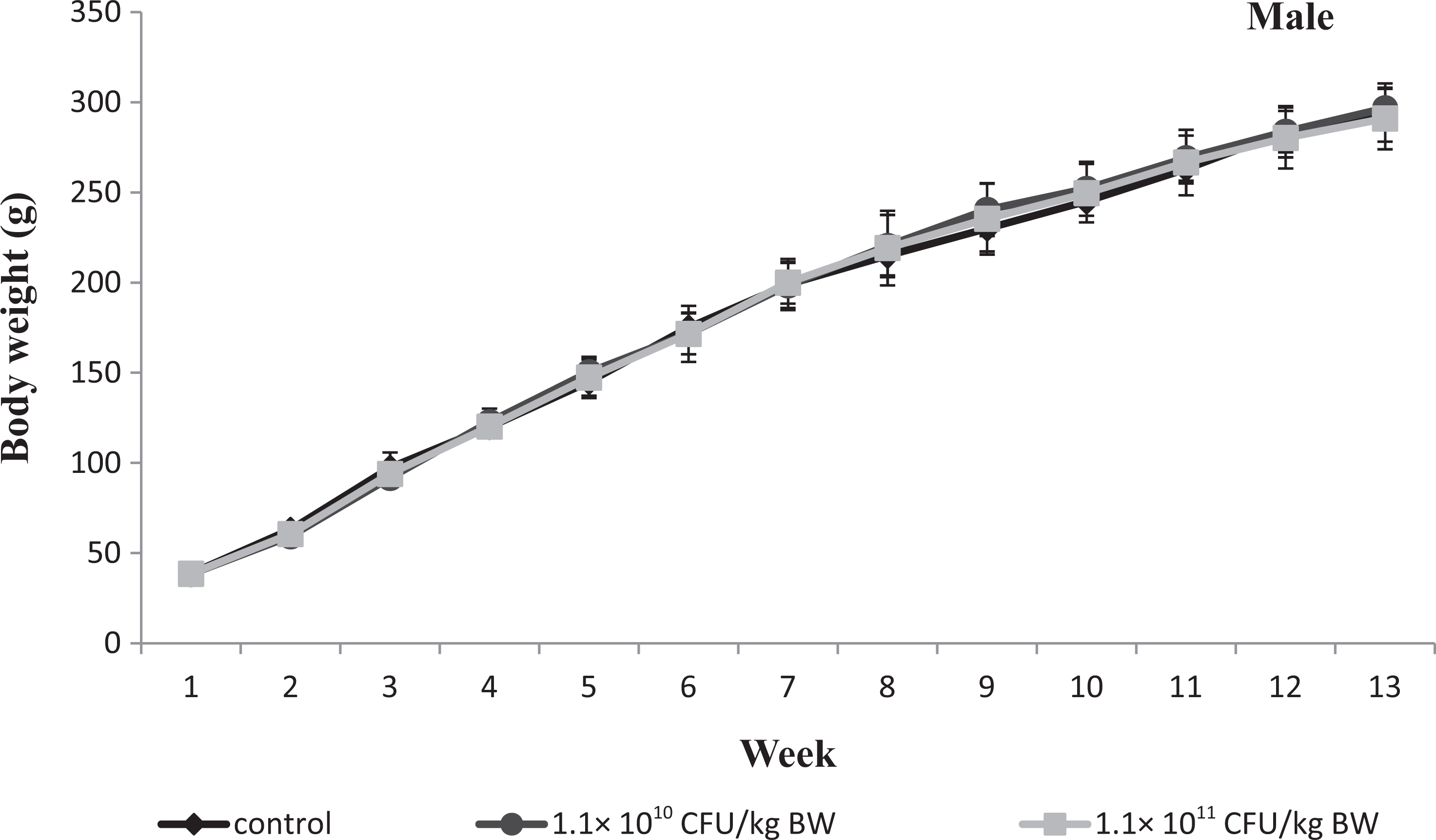
Graph showing body weight of male Wistar rats given test article for 13 weeks. Values are means ± standard error of the mean (SEM) of 6 rats.
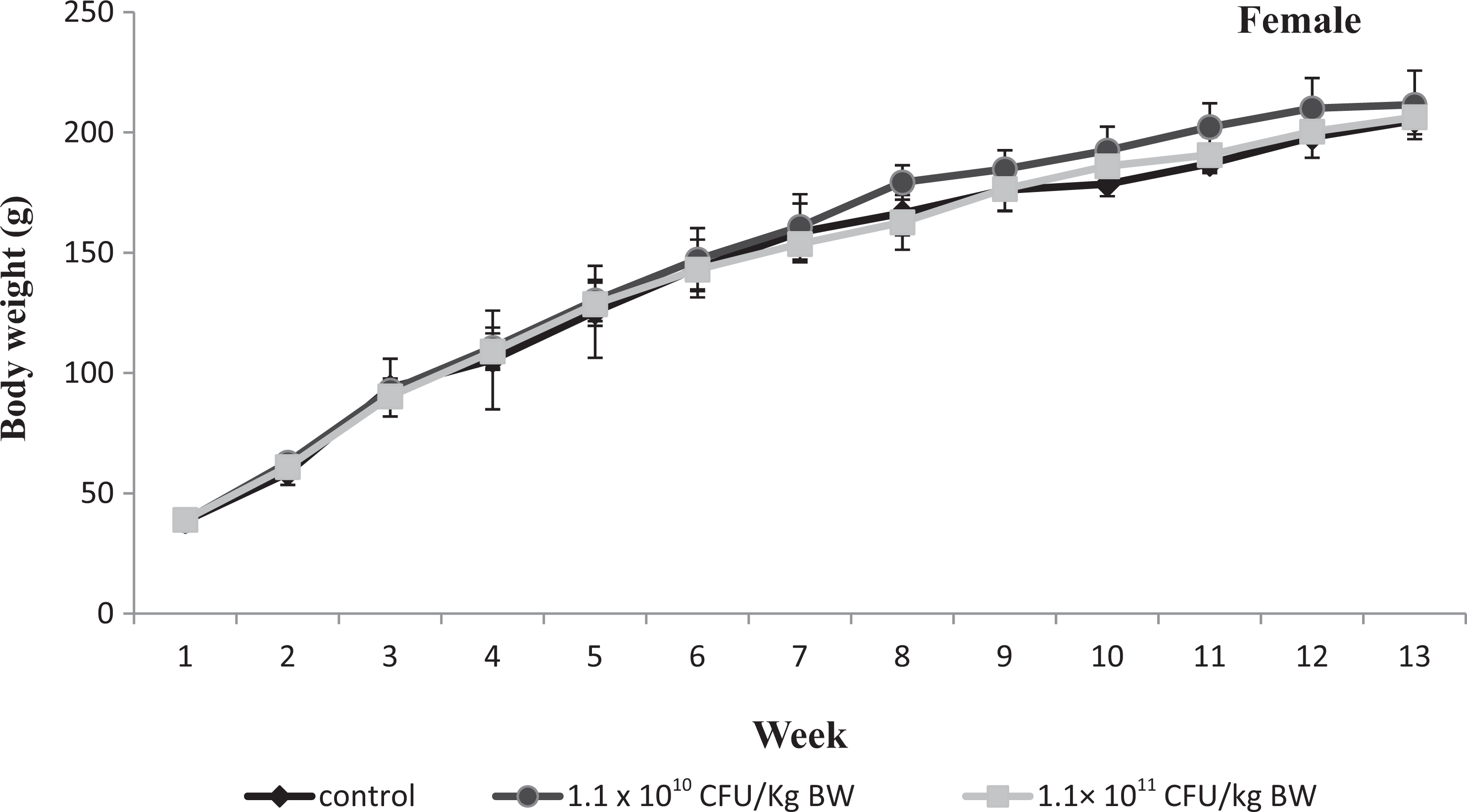
Graph showing body weight of female Wistar rats given test article for 13 weeks. Values are means ± standard error of the mean (SEM) of 6 rats.
Viability of B. licheniformis Me1
Viability of the test article in the rat GIT was determined by monitoring the presence of
Relative organ weights and histopathology
At necropsy, macroscopic observation of the organs revealed no treatment-related damages or differences. The relative organ weights of adrenals, spleen, kidney, and heart did not show any significant deviation from that of control in any of the treated animal groups (Supplemental Tables 1 and 2). However, a marginal, but statistically nonsignificant decrease in relative organ weight of lungs in females was noticed (Supplemental Table 2).
Histopathological examination of the tissues of the control and treated groups revealed no treatment-related abnormalities in morphology or toxicity of the organs. No swelling of epithelium and occlusion of intestinal lumen were noticed even in the highest dose group. The villi pattern of the small intestine was well preserved in all the rats treated with
Hematology and serum biochemical studies
No significant differences were found in any of the analyzed aspects in hematology for both male and female test groups when compared to that of the control groups (Supplemental Table 3). On serum biochemical analysis, no statistically significant dose-dependent alterations were detected either in the levels of glucose, cholesterol, triglyceride, urea, or in the activity of ALP, LDH, AST, and ALT in both the sexes in comparison to the control groups (Supplemental Table 4).
Micronucleus Assay in Mice
The micronucleus test was conducted to investigate the formation of micronuclei containing chromosome fragments or whole chromosomes and is considered the most reliable assay for cytogenetic damage. In the micronucleus assay, neither any differences in BW between the treatment groups compared to the control group nor any signs of toxicity were noted in clinical observations following administration of the test article at doses of 1.1 × 1011 and 1.1 × 1010 CFU/kg BW. The ratio of reticulocytes to total erythrocytes was used an indicator for the evaluation of bone marrow toxicity. The results clearly demonstrate that the number of immature erythrocytes in each dose did not significantly increase above the concurrent negative (water) control frequencies. Additionally, it was always within the historical negative control range. As expected, animals in the EMS-treated positive control group showed a significant increase in the frequency of micronuclei compared to the negative controls. In contrast, no statistically significant induction of micronuclei in the test article fed groups was observed. The ratio of reticulocytes to total erythrocytes in the treated groups did not show any significant decrease as compared to the negative control group. For female mice, the average reticulocyte to total erythrocyte ratio in the negative control group was 2.17%. Also, the treated groups 1.1 × 1011 and 1.1 × 1010 CFU/kg BW per day showed 2.07% and 1.93% of reticulocyte to total erythrocyte ratio, respectively, a reduction of 25.9% from the positive control group. In male mice, the negative control group showed a ratio of 1.85% and the 2 dose groups 1.1 × 1011 and 1.1 × 1010 CFU/kg BW/d exhibited 1.51% and 1.10% reticulocytes to erythrocytes ratio. In the male group, the positive control showed 27.12% decrease in the ratio. These results clearly show that Me1 did not cause any signs of bone marrow cytotoxicity of mice in the range of the doses tested.
In the negative control groups, the incidence of micronucleated reticulocytes in the peripheral blood per 1000 reticulocytes was 1.18 ± 0.5 in males and 0.98 ± 1.1 in females. These results were within the historical reference range.
27
The positive control group had a statistically increased mean frequency of 26.14 ± 3 in males and 23.2 ± 5 in females as compared to the negative control group. The micronucleated reticulocytes per 1000 reticulocytes were found to be 1.03 ± 0.7 and 0.89 ± 1.2 in males and 1.2 ± 1 and 1.03 ± 0.9 in females at the test article dose levels of 1.1 × 1011 and 1.1 × 1010 CFU/kg BW per day, respectively. Since the mice peripheral blood micronucleus assay did not show any statistically significant changes according to the OECD Guideline for the Testing of Chemicals No. 474, there is no indication that
Acute Eye Irritation Study in Rabbits
Skin Irritation Study in Rabbits
Discussion
As the demand for probiotic foods among consumers continues to grow, several new foods are likely to include probiotics in the future. It is of obvious importance that novel probiotic cultures for human and animal consumption are evaluated carefully and precisely for safety and efficacy before commercialization.
26
Many reports have shown that selected strains of
The ability of
To assess the safety of
Organ weight changes have long been accepted as a sensitive indicator of chemically induced changes to organs. Therefore, in toxicological experiments, the comparison of organ weights between control and treated groups have conventionally been used to predict the toxic effect of a test article.
39
The absence of significant changes in the vital organs of the treated groups during the subchronic oral toxicity study shows that the ingestion of
Hematological values were not significantly affected by the administration of
The viability test result indicates the survivability of the test article
The in vivo micronucleus test conducted in mice to assay the cytogenotoxic effect of
The concentration of
Conclusion
The test article
Footnotes
Acknowledgments
The authors are grateful to the Director, CFTRI, Mysore and Dr S Umesh Kumar, Head, Food Microbiology Department, CFTRI, and the staff of Animal house, B&N Department for providing all facilities to carry out this experiment successfully. V.N. acknowledges CSIR for the Fellowships. Work was carried out under the Institute project MLP83.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article
