Abstract
Dimethoate (DM) is an organophosphate insecticide with numerous uses on field and agricultural crops and ornamentals. Data concerning DM-acute genotoxicity are controversial and knowledge on its delayed effect is limited. For this reason, we aimed to further explore DM genotoxicity resulting from subchronic intoxication of experimental mice. Thus, DM was administered to mice at doses ranging from 1 to 30 mg/kg body weight for a period of 30 consecutive days. There was a significant increase (P < .05) in the frequency of micronucleated bone marrow cells following DM administration. Furthermore, the chromosome aberration assay revealed a significant increase in the percentage of chromosome abnormalities in a dose-dependent manner. Dimethoate was also found to induce significant DNA damage in mouse bone marrow cells as assessed by the comet assay. Altogether, our results showed that, after a subchronic exposure, DM was a genotoxic compound in experimental mice.
Keywords
Introduction
Pesticides play an important role in modern agriculture, and their use has increased steadily. Because many of them are suspected to have mutagenic and carcinogenic activities, 1 concern for their adverse effects on human health has increased. Several studies have reported the genotoxic potential of organophosphorous (OP) pesticide, 2 but the results reported so far are not conclusive. Organophosphorous compounds are among the most widely used pesticides, either as insecticides or as herbicides. In acute exposure, the main mechanism of toxicity of OP pesticides is irreversibly binding to the enzyme acetylcholinesterase and inhibiting its activity that results in accumulation and prolonged effect of acetylcholine and consequently follows with acute muscarinic and nicotinic effects. 3 In chronic and subchronic exposures, added to cholinesterase inhibition, induction of oxidative stress has been reported as the main mechanism of OP pesticides’ toxicity. 4 –8 In addition, other mechanisms are responsible for their genotoxicity, such as those related to their alkylating 9,10 and phosphorylating properties. 11
Dimethoate ([DM], O,O-dimethyl S-methyl carbamoylphosphoro-dithioate) is one of the most important OP insecticides used extensively on a large number of crops against several pests. Dimethoate exerts toxic effects on many tissues and organs including pancreas, 12 - 14 liver, 15– 20 kidney, 21 and brain. 16 - 19 Dimethoate was also revealed to be immunotoxic 22 24 and causes several adverse effects in the reproductive system of male and female mice. 25–28 Interestingly, DM was reported to cause benign and malignant neoplasms of the liver, endocrine organs, and lymphatic system 29 ; however, the International Agency for Research on Cancer (IARC) was unable to classify DM with regard to its potential carcinogenicity due to the inadequacies of existing studies. 30
The information on the genotoxic properties of DM is limited and inconsistent. It is reported that DM was mutagenic in Escherichia coli, Saccharomyces cerevisiae, and Salmonella typhimurium TA 100 but nonmutagenic in S typhimurium strains 31 –36 TA 1535, TA 1537, TA 1538, TA 98, and TA 102. In vitro exposure to DM induced a concentration-dependent increase in sister chromatid exchange (SCE) frequency in toadfish lymphocytes 37 and in mammalian cell cultures. 38 Dimethoate gave positive responses in the Salmonella reversion assay and the SOS chromotest. 39 Dimethoate induced a statistically significant elevation of micronuclei (MNs) frequency in human lymphocytes in a nondose-related manner 40 and in mouse bone marrow after acute intoxication. 41 This pesticide was also found to increase the incidence of numerical but not structural chromosomal aberrations in male Wistar rats. 24 On the other hand, it was also reported that DM was nonmutagenic in some other genotoxicity tests. 42 Recent study of Undeger and Basaran 43 showed that DM significantly increased DNA damage in human lymphocytes using the comet assay.
Since data on the DM acute genotoxicity are controversial and knowledge on its delayed effect is much more limited, we aimed in the present study to further explore DM genotoxicity after subchronic intoxication of experimental mice. For this, we utilized the MN and chromosome aberrations tests and the comet assay in mouse bone marrow cells.
Material and Methods
Chemicals
Dimethoate (analytical standard), supplied by Sigma Aldrich Chemie GmbH (France), was used for the study. Corn oil was used for preparing suspensions of DM (volume of injection was 100 µL per animal). All other chemicals used were of analytical grade.
Animals
This study was conducted on female mice BALBL/c (average body weight 20 ± 0.3 g, age: 6 weeks) purchased from the Central Pharmacy (SIPHAT, Tunis, Tunisia). Before any experience, all animals were maintained 7 days under the same laboratory conditions of temperature (22°C ± 3°C), relative humidity (55% ± 5%), and a 12:12 hours light/dark cycle and received a nutritionally standard diet (SICO, Sfax, Tunisia) and tap water ad libitum. The experimental procedures were carried out according to the American College of Toxicology Statement on the Use of Animals in Toxicology and approved by the local Ethics Committee.
Experimental Design
After an acclimation period, mice were randomly divided into 6 groups (15 mice per group). Animals of groups 2, 3, 4, 5, and 6 were administered intraperitoneally (ip) DM (dissolved in corn oil) at doses of 1, 5, 10, 15, and 30 mg/kg body weight for 30 consecutive days. These doses 44 correspond, respectively, to 2%, 10%, 20%, 30%, and 60% of the LD50. Group 1, receiving equivalent amount of vehicle alone, served as the control. Animals within different treatment groups were divided into 3 subgroups: A (for the MN assay), B (for the comet assay), and C (for the chromosome aberration assay; 5 animals per subgroup) and received their respective treatment. All animals were sacrificed by cervical dislocation on the 31st day of the study.
Body Weight
Animals were examined daily throughout the experimental period for signs of toxicity. Body weight was recorded in the beginning and at the end of the treatment period.
In Vivo MN Assay
Immediately after the animals were sacrificed, femur and tibia of mice in the subgroup A were freed from adherent tissues and were dissected out. The bone marrow was sampled by injection of filtered fetal calf serum using a syringe. The collected cells were centrifuged at 390g for 5 minutes, a little volume of supernatant was discarded and the cells were resuspended in the remaining fluid. A small drop of the resuspended cell pellet was spread on a glass slide, fixed in absolute methanol for 5 minutes, and air-dried for conservation at room temperature. Air-dried slides were stained for 15 minutes in phosphate-buffered saline ([PBS], 0.15 mol/L, pH 7.4) containing 10 µg/mL of acridine orange (freshly prepared), rinsed in the same buffer for 15 minutes, and allowed to dry in the dark at room temperature.
Scoring Criteria
As a result of acridine orange staining, red fluorescence is observed in polychromatic erythrocytes (PCEs); normochromatic erythrocytes (NCEs) appear slightly green or opaque and polychromatic erythrocyte micronucleated (PCEMN) appear red with one or more yellow fluorescent corpuscles which are MN. Scoring of MN was performed according to the criteria described by Hayashi et al. 45 These criteria are based essentially on the diameter and the shape of the MN.
Slide Analysis
The slides, randomly coded, were observed under ×1000 magnification using a fluorescence microscope (Nikon Eclipse E 400, New York). The number of PCE among 1000 (NCE + PCE) was determined in order to evaluate any cytotoxic effect of DM. The number of PCEMN among 2000 PCE per mouse was calculated to determine the induction of MN.
In Vivo Comet Assay
After sacrificing the animals of the subgroup B, both the femurs and tibias were removed and the content was directly flushed out with the help of a 24-gauge needle into a microcentrifuge tube. The cell suspension was prepared in PBS. From the suspension, 5 µL of sample containing approximately 2 to 5 × 104 cells/mL was added to 95 µL of 0.5% low melting point agarose (LMA in Ca2+- and Mg2+-free PBS) to prepare the final cell–agarose suspension. The comet assay was performed as described 46 with some modifications. From the final cell–agarose suspension, 80 µL was spread over the microscope slide which was precoated with 1% normal melting point agarose (NMA). The cells were then lysed in a buffer containing 2.5 mol/L NaCl, 100 mmol/L EDTA, and 10 mmol/L Tris (pH 10.0) with freshly prepared 1% Triton X-100 and 10% dimethyl sulfoxide (DMSO) for 24 hours at 4°C. After lysis, slides were rinsed 3 times in deionized water to remove salt and detergent. Slides were placed in a horizontal electrophoresis unit and DNA was allowed to unwind for 20 minutes in alkaline solution containing 300 mmol/L NaOH and 1 mmol/L EDTA, pH > 13. The DNA was electrophoresed for 15 minutes at 300 mA and 25 V (0.90 V/cm). The slides were neutralized with 0.4 mol/L Tris (pH 7.5), stained with ethidium bromide (20 µg/mL) before examination with a Nikon Eclipse TE 300 fluorescence microscope (Nikon, Tokyo, Japan). A total of 100 comets on each randomly coded slide were visually scored according to the relative intensity of fluorescence in the tail and classified as belonging to 1 of 5 classes. Each comet class was given a value of 0, 1, 2, 3, or 4 (from undamaged, 0, to maximally damaged, 4) as described previously by Collins et al. 47 The total score was calculated by the following equation: (percentage of cells in class 0 × 0) + (percentage of cells in class 1 × 1) + (percentage of cells in class 2 × 2) + (percentage of cells in class 3 × 3) + (percentage of cells in class 4 × 4). Consequently, the total score ranges from 0 to 400.
In Vivo Chromosome Aberration Assay
Twenty-four hours before sacrifice, the animals of subgroup C were given a suspension of yeast powder (100 mg/500 µL) to accelerate mitosis of bone marrow cells. Vinblastine (200 µL; 250 µg/mL) was injected to animals 45 minutes before sacrifice in order to block dividing cells in metaphase. 48 Bone marrow cells from femurs and tibia were collected, subjected to hypotonic shock (KCl 0.075 mol/L), and fixed 3 times using methanol–acetic acid, according to the technique of Evans et al. 49 The cells were spread on glass slides that were blazed on a flame for 5 seconds, then air-dried for conservation at room temperature and finally stained with a 4% dilution of Giemsa reagent in water for 15 minutes. After randomly coding the slides, the chromosomes of 100 cells in metaphase were examined for abnormalities at a magnification of ×1000 using an optical microscope (Carl Zeiss, Germany). This was done for each 1 of 5 replicates (500 metaphases per dose level) for the negative control and the treated groups. Chromosome aberrations were identified according to criteria described by Savage. 50 Metaphases with chromosome breaks, gaps, rings, and centric fusions (Robertsonian translocation) were recorded and expressed as percentage of total metaphases per group.
Statistical Analysis
All values were expressed as mean ± standard deviation (SD) of 5 animals per group. Data were analyzed using 1-way analysis of variance (ANOVA) followed by Fisher post hoc analysis. All statements of significance were based on a probability (P) ≤ .05.
Results
Body Weight
There were no deaths reported during the course of the study in any of the treated groups. There was a significant reduction in body weight and weight gain in the treated groups of 15 and 30 mg/kg per d at the end of the treatment compared with the control and other treated groups (Table 1 ).
Effect of Dimethoate (DM) Treatment on Body Weight (g) of Experimental Mice After Subchronic Exposure of 30 Days a
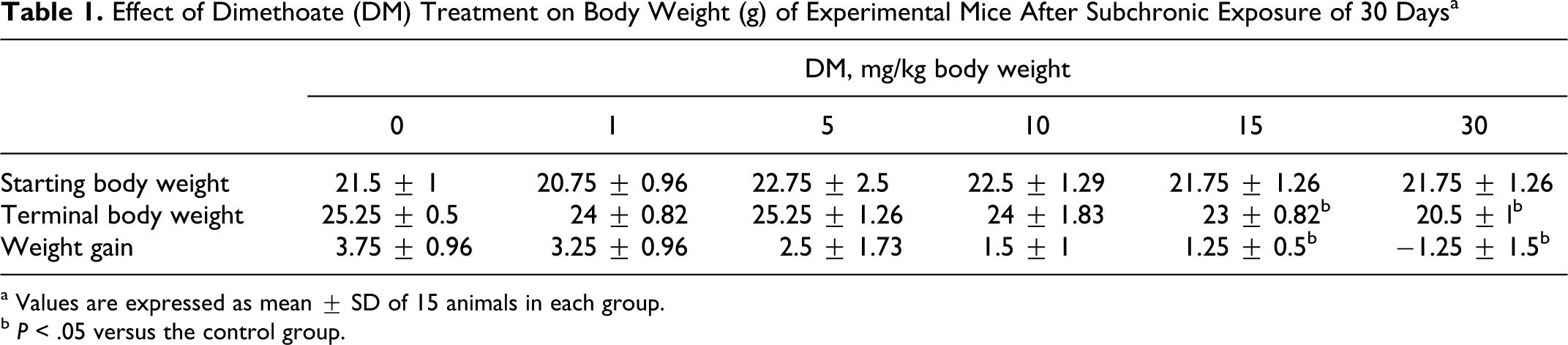
a Values are expressed as mean ± SD of 15 animals in each group.
b P < .05 versus the control group.
In Vivo MN Assay
The cytotoxic potential of DM was evaluated by counting the number of PCE among 1000 cells (PCE + NCE). This number showed a mean value of 378.3 ± 7.6 in the control group and decreased first slightly in mice treated with DM at 1, 5, 10 mg/kg body weight to, respectively, 373.67 ± 8.5, 367.67 ± 8.5, and 357.00 ± 9.5 and then significantly in mice treated with DM at 15 and 30 mg/kg body weight to, respectively, 295.0 ± 22.3 and 275.6 ± 24.1 (P ≤ .05; Figure 1 ).
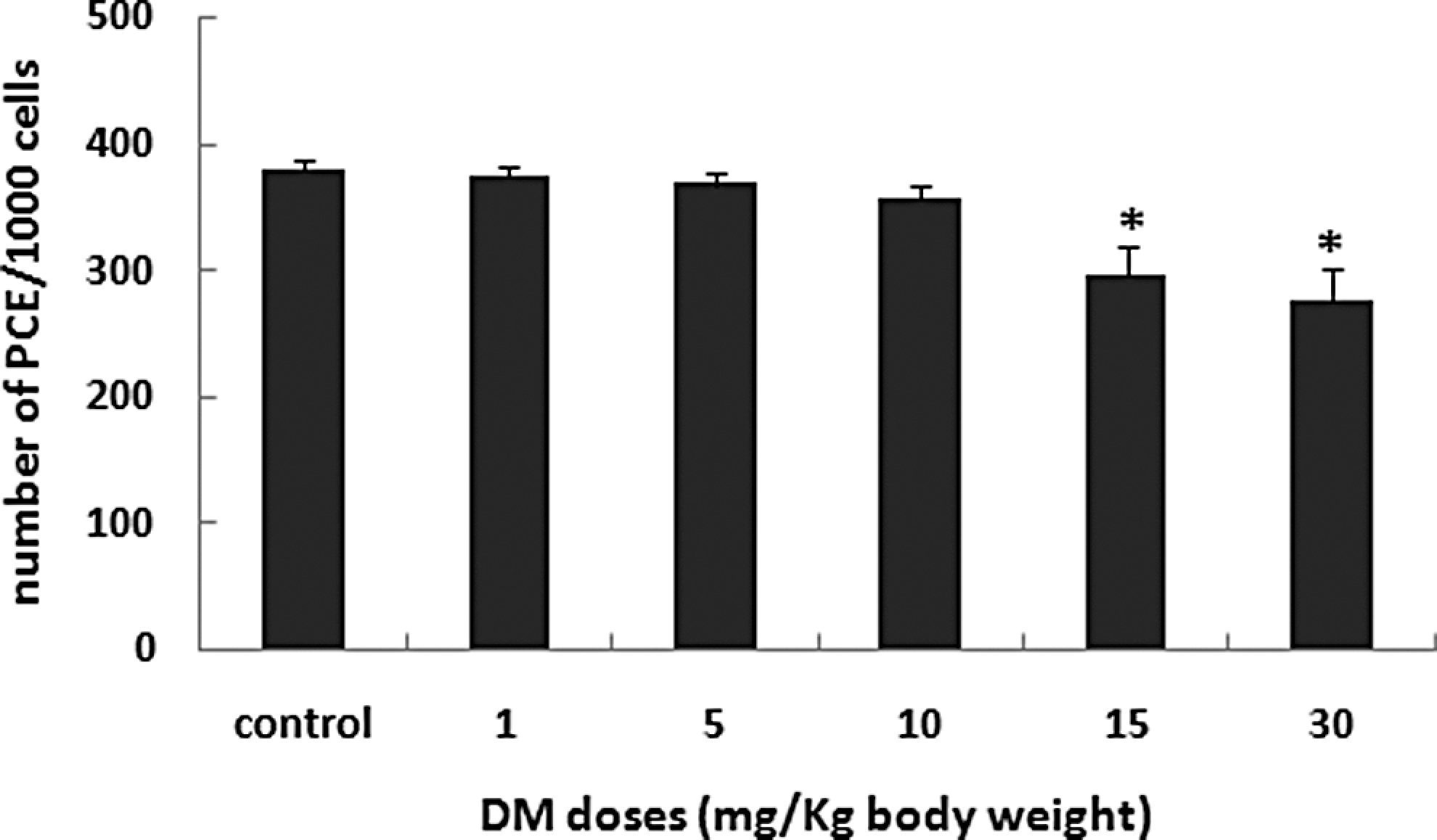
Number of polychromatic erythrocytes (PCEs) in 1000 PCEs and normochromatic erythrocytes (PCE + NCE) in animals treated with DM at the indicated doses for 30 consecutive days. Values are expressed as mean ± SD, n = 5. *Significant difference from the control group (*P < .05). DM indicates dimethoate; SD, standard deviation.
The number of PCEMN among 2000 PCE, indicative of genotoxicity, is presented in Figure 2. Dimethoate induced a significant (P ≤ .05) increase in PCEMN number in a dose-dependent manner. In fact, this number passed from a basal value of 10 ± 1 in the control group to 18 ± 1, 29.6 ± 3, and 49.6 ± 9.8 in groups treated with DM at, respectively, 5, 10, and 15 mg/kg body weight. At the highest dose of DM (30 mg/kg body weight), the PCEMN number significantly increased to reach 63.3 ± 4.6.
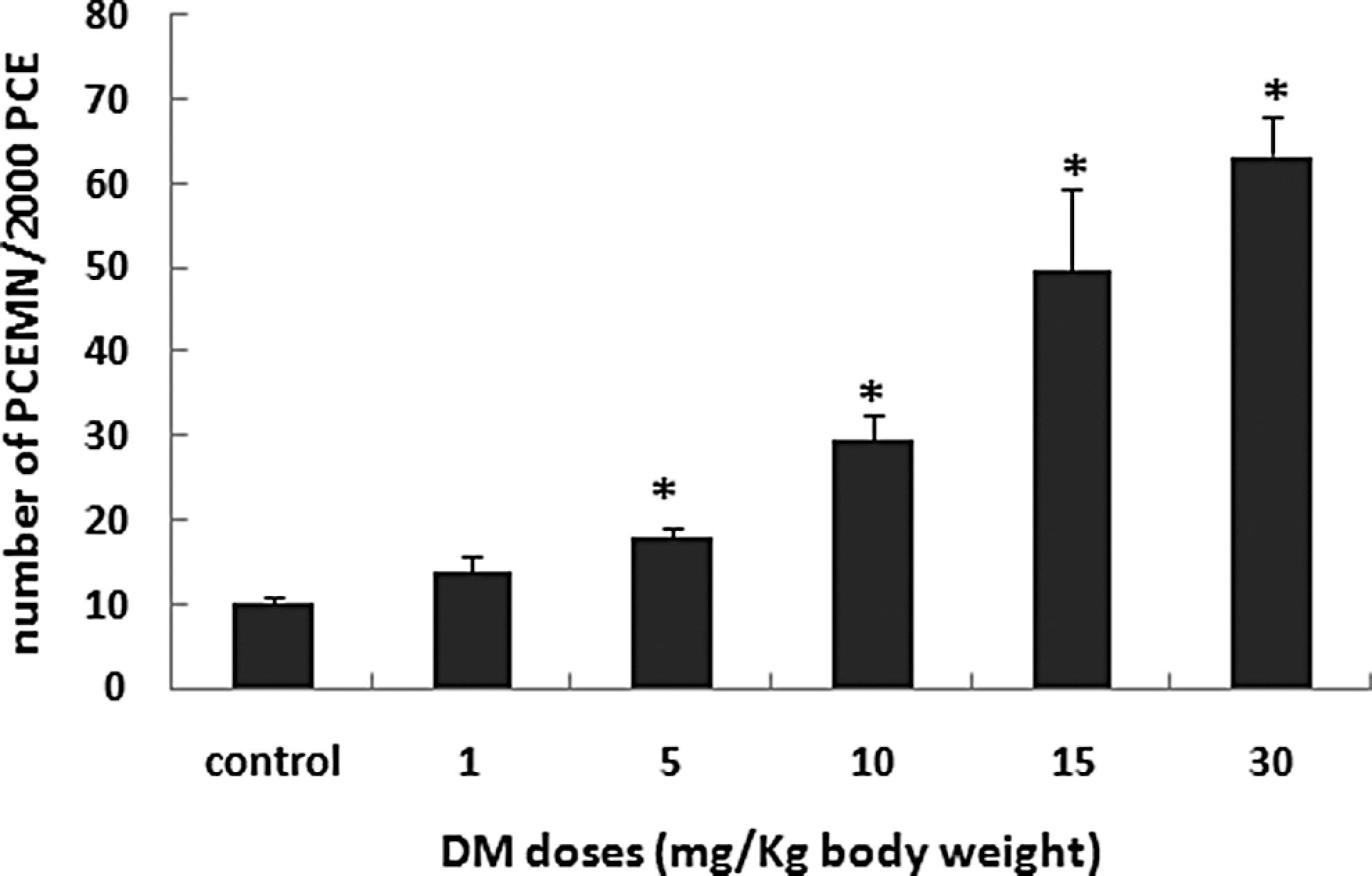
Number of polychromatic erythrocytes micronucleated (PCEMN) in 2000 polychromatic erythrocytes (PCEs) in animals treated with increasing doses of DM for 30 consecutive days. Values are expressed as mean ± SD, n = 5. *Significant difference from the control group (*P < .05). DM indicates dimethoate; SD, standard deviation.
In Vivo Comet Assay
Bone marrow cells were used to assess genotoxicity of DM using the comet assay which is considered a sensitive, rapid, and relatively simple method for detecting DNA damage at the level of individual cells. 46 Results of the visual scoring of total DNA damage observed in bone marrow cells are illustrated in Figure 3. Significantly higher levels of DNA damage were detected in mice exposed to DM compared with the negative control. In summary, DM induced significantly (P < .05) a dose-dependent increase in DNA damage of exposed mice (Figure 3).
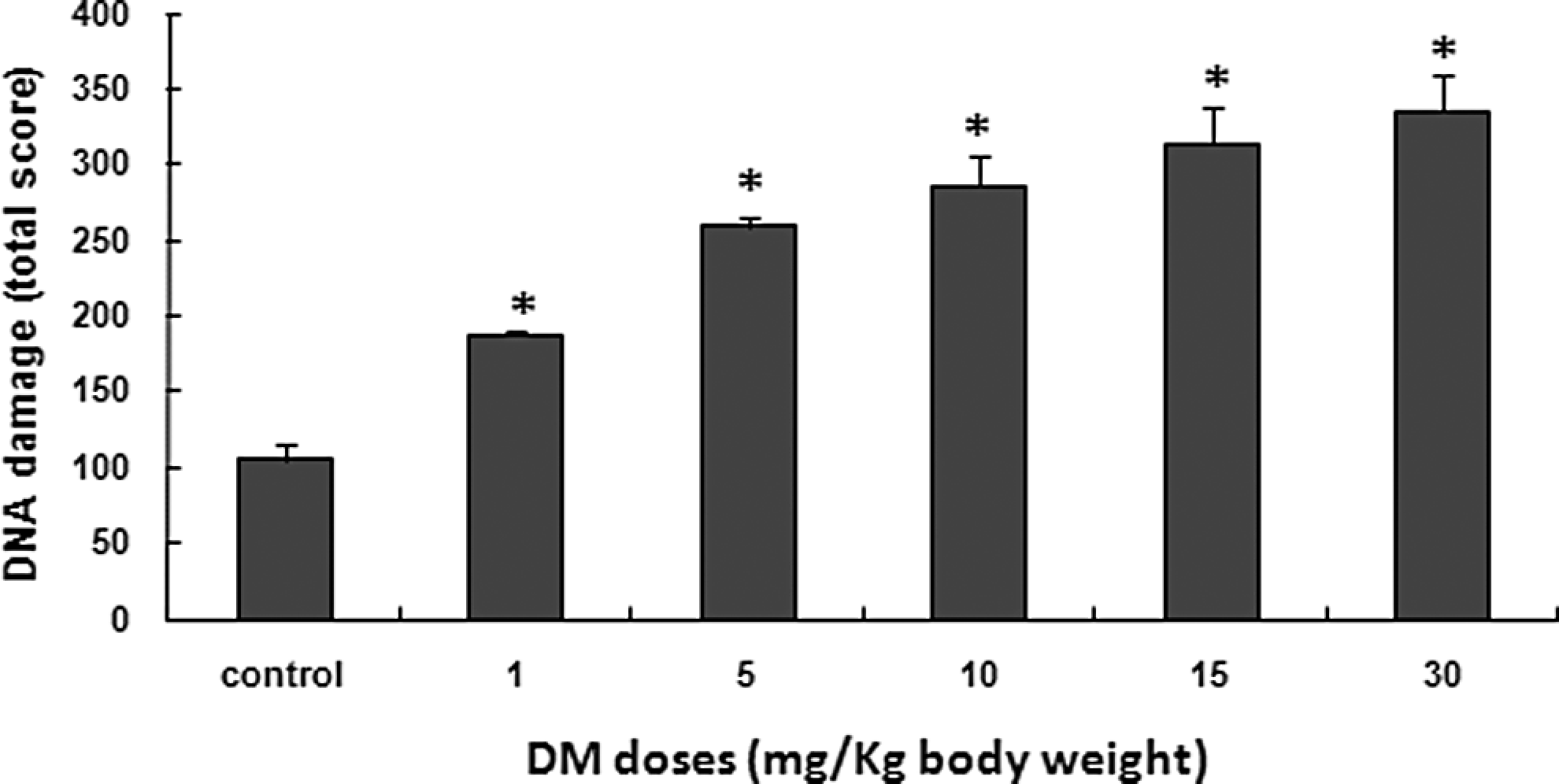
DNA damage (as evaluated by the comet assay) induced by DM in bone marrow cells after treatment with increasing doses of DM for 30 consecutive days. Five hundred comets were counted in each group (100 comets per animal). Values are expressed as mean ± SD of 5 animals in each group. *Significant difference from the control group (*P < .05). DM indicates dimethoate; SD, standard deviation.
In Vivo Chromosome Aberration Assay
Only structural aberrations induced by the different treatments were enumerated in the present study, with special emphasis on gaps, rings, breaks, and centric fusions. All these types of structural abnormalities and their frequencies for both control and treated groups are presented in Table 2. Numerical chromosome abnormalities such as polyploidy and aneuploidy were not evaluated. Chromosome aberrations induced in mouse bone marrow cells after 30 days of injections of increasing doses of DM are presented in Figure 4 together with the negative control. Chromosome aberrations whatever the type, especially centric fusions and breaks were statistically significantly (P ≤ .05) increased in a dose-dependent manner in mice treated with DM (Table 2, Figure 4).
Different Types of Chromosome Aberrations and Their Percentage in Bone Marrow Cells After Treatment With Increasing Doses of DM for 30 Consecutive Days a
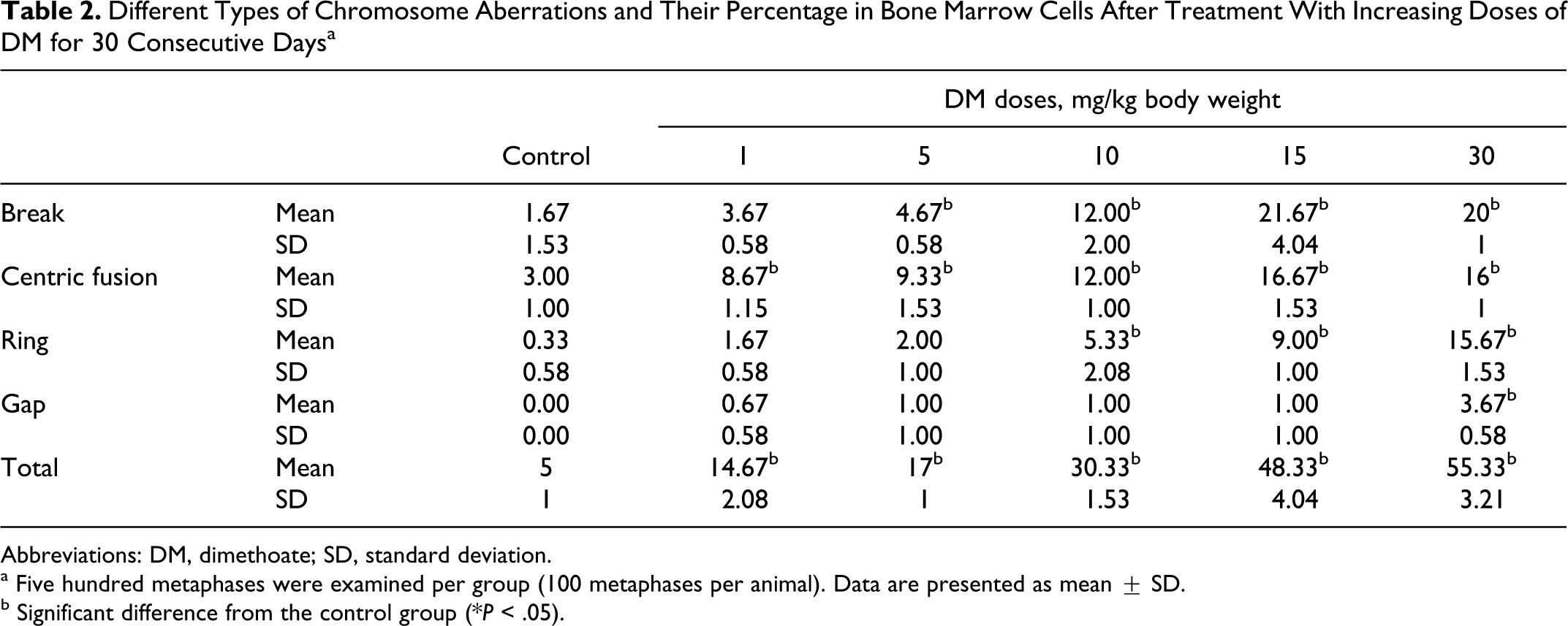
Abbreviations: DM, dimethoate; SD, standard deviation.
a Five hundred metaphases were examined per group (100 metaphases per animal). Data are presented as mean ± SD.
b Significant difference from the control group (*P < .05).
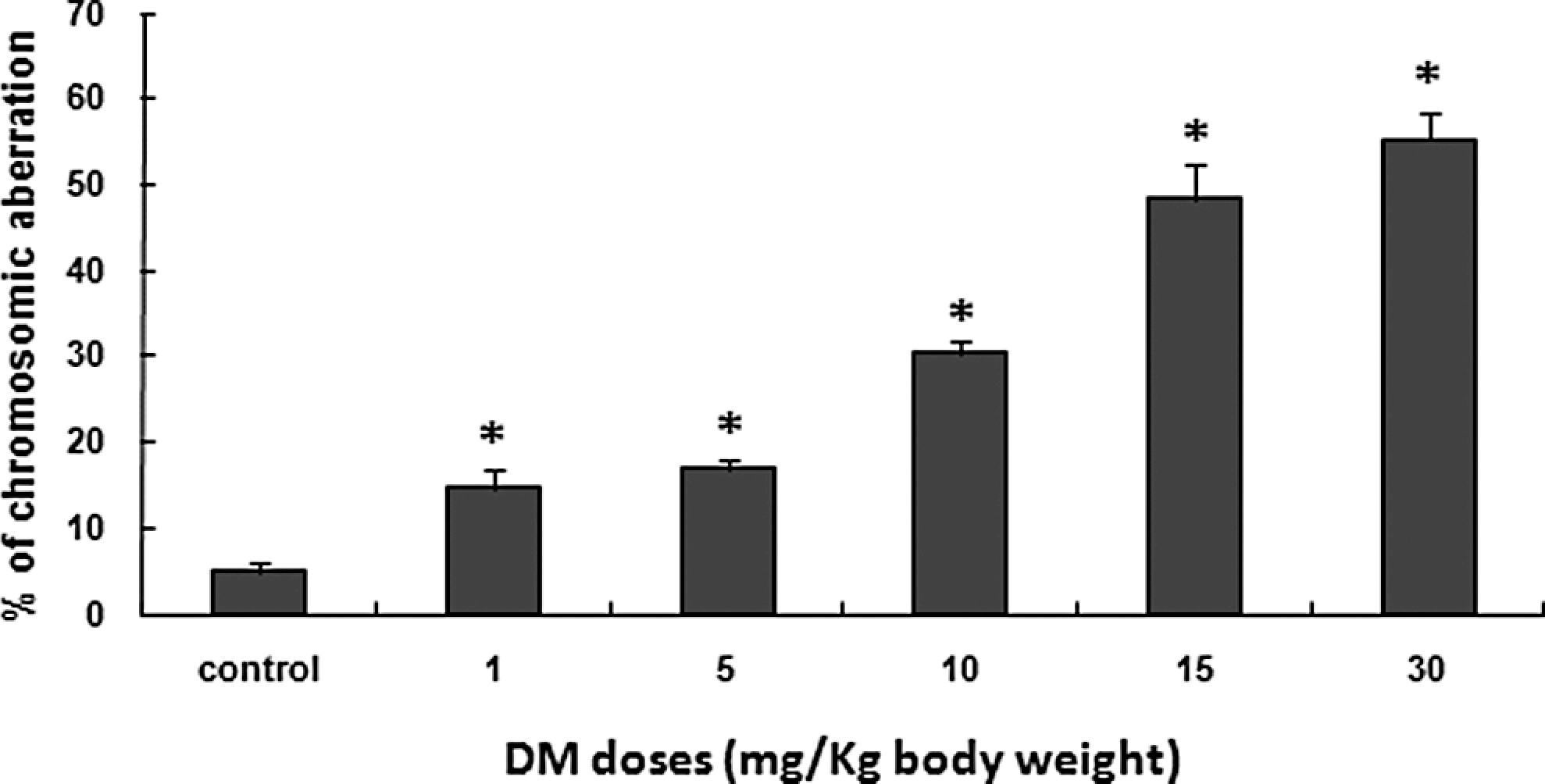
Percentage of bone marrow cells containing chromosome aberrations (total of all types of chromosome aberrations) after treatment with increasing doses of DM for 30 consecutive days. Five hundred metaphases were examined per group (100 metaphases per animal). Data are presented as mean ± SD. *Significant difference from the control group (*P < .05). DM indicates dimethoate; SD, standard deviation.
Discussion
In spite of several attempts that have been made to establish the genotoxicity of DM, this matter remains somehow controversial. Furthermore, IARC was unable to classify DM with regard to its potential carcinogenicity. 30 Indeed, the mutagenicity of DM was not sufficiently proven in S typhimurium strains TA 1535, TA 1537, TA 1538, TA 98, and TA 102. 31 –36 Dimethoate gave negative results in a mutation assay in Schizosaccharomyces pombe 42 and in CHO cells. 51 Despite these findings, DM has been shown to cause an increase in SCE frequency in toadfish lymphocytes. 37 Besides, DM induced DNA damage and an increase in the frequency of MN in human lymphocytes at doses ranging from 0.1 to 200 µg/mL. 43 , 40 ,52
Because of these controversial reports on the genotoxicity and mutagenicity of DM and to elucidate the mechanism underlying the genotoxicity of this pesticide, we evaluated, in the present investigation, its ability to induce MN, chromosome aberrations, and DNA damage in bone marrow cells of mice treated with DM for 30 consecutive days.
The MN assay is a test widely used to assess genotoxicity of chemicals. 53,54 Furthermore, it gives indication concerning the cytotoxic potential of the tested compound. Our results showed that the number of PCE per 1000 cells was significantly reduced for DM doses of 15 and 30 mg/kg body weight (Figure 1). Several mechanisms could explain this reduction, among them, either direct cytotoxicity or MN formation and heavy DNA damage leading to cell death or apoptosis. The data obtained show clearly that DM induced MN in bone marrow cells of mice treated in a dose-dependent manner (Figure 2). From these results, we can deduce, on one hand that DM may interact directly with DNA causing DNA damage. On the other hand, DM may cause dysfunction of the mitotic spindle. In this latter case, it might be considered as an aneugenic compound. However, staining bone marrow cells with acridine orange does not discriminate between MN-containing fragments or whole chromosomes. That is why we performed the comet assay and the chromosome aberration test.
The comet assay is increasingly used in industrial genotoxicity testing in vitro 55 –58 and is also becoming an important tool for evaluating the genotoxic potential of compounds in vivo 59 –61 because of its rapidity, simplicity to perform, 59 and its higher sensitivity and specificity. 59 Our results clearly showed that DM induced an increase in DNA damage score in a dose-dependent manner (Figure 3).
The chromosome aberration test, recommended by regulatory authorities for the assessment of genotoxicity and mutagenicity of many chemicals and natural compounds, has provided positive data. In fact, DM increased the number of chromosome aberrations in a dose-dependent manner with relatively high frequencies of DNA breaks compared with other types of chromosome abnormalities (Figure 4, Table 2). Centric fusions (Robertsonian translocations) are also frequent, which may be due to the fact that almost all mouse chromosomes are acrocentric. These types of chromosomes have the exceptional facility to merge with each other. Only the structural chromosome aberrations are presented here, the reason being that only very little polyploidy or aneuploidy were detected. This specific aspect needs further investigation.
The design of the present experiments does not allow formation of direct stable mutations since cells were arrested at the first stage of mitosis. It is strongly believed that cells with heavy chromosome abnormalities cannot survive and will rapidly undergo apoptosis. In fact, DM induced apoptosis in spiders from industrially polluted areas. 62 One may assume that when cells with a small number of chromosome aberrations are allowed to survive (when not arrested at mitosis), they will divide with possible occurrence of mutations that could be transmitted to the next generation through clonal expansion of somatic or even stem cells. This will then give rise to tumor formation. However, DM was not classified by IARC with regard to its potential carcinogenicity. 30
Genotoxicity induced by DM could be attributed to the parent compound itself as well as to its oxygen analog omethoate, which was considerably more toxic than DM. 63 These effects can also be attributed to active oxygen species generated by DM. 13 - 20 , 64 Thus, further investigations are needed to elucidate the mechanism/mechanisms by which DM induced DNA damage.
In conclusion, our results showed that DM induced, in a dose-dependent manner, MN formation, DNA damage revealed by the comet assay, and increased chromosome aberration levels in bone marrow cells of mice treated with DM for 30 consecutive days. These results confirmed the genotoxic potential of DM in vivo in subchronic condition and indicated a need for further investigations to evaluate the carcinogenic potency of DM and the potential risks associated with the exposure of humans and wildlife to this pesticide.
Footnotes
Guidelines for Ethical Publications
Authors declare that this manuscript was consistent with the guidelines and principles of American College of Toxicology Statement on the Use of Animals in Toxicology.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Ministère Tunisien de l’Enseignement Supérieur et de la Recherche Scientifique et de la Technologie (Laboratoire de Recherche sur les Substances Biologiquement Compatibles: LRSBC).
