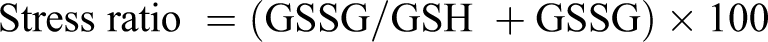Abstract
To examine the hepatoprotective activities of Nigella sativa (Ns) and thymoquinone (TQ) against carbon tetrachloride (CCl4)-induced hepatotoxicity, the effects of water extract of Ns seeds (50 mg/kg) or TQ (5 mg/kg in corn oil) by gavage for 5 days on detoxifying enzymes and glutathione were compared in healthy and CCl4-challenged (1 mL/kg in corn oil, intraperitoneally [ip], a single dose) rats. Both Ns and TQ countered the elevations in serum alanine aminotransferase activity, oxidized glutathione level, and stress ratio caused by CCl4. Both Ns and TQ ameliorated the reductions in the activities and messenger RNA (mRNA) levels of glutathione S-transferase, NAD(P)H-quinone oxidoreductase, and microsomal epoxide hydrolase, as well as the reductions in reduced glutathione and cysteine levels produced by CCl4. In many instances, Ns was much superior to TQ in providing protection against the damaging effects caused by CCl4. This protection could be attributed to the induction of chemoprotective enzymes probably through increasing transcription.
Keywords
Introduction
For decades, scientists have been exploring nature searching for substances that could be used for treatment of human disorders. The discovery of novel drugs usually starts with a search for active compounds from plant or animal sources. Nigella sativa ([Ns] family Ranunculaceae) or black seed has been extensively used as a spice and as a natural remedy in folk medicine for many disorders such as ulcers, diabetes, asthma, and hypertension 1 and has recently proved efficacious against cancer in animals. 2,3 The various applications reported for Ns as a remedy make it a potential candidate for drug discovery. Nigella sativa was shown to protect against hepatotoxicity, nephrotoxicity, and cardiotoxicity. 4,5 Thymoquinone (TQ) is one of the major active constituents in Ns. 6 Similarly, TQ was also shown to protect against nephrotoxicity and hepatotoxicity. 7 –9 Like Ns, TQ was also reported to chemosensitize cancer cells to chemotherapy 10 and protect against cancer as well. 11 The safety and toxicity is a major concern too. Thymoquinone was found to be safe with LD50 of ∼800 mg/kg in rats when ingested orally. 12 However, 50 mg/kg of TQ was found to cause oxidative stress and hepatic damage in mice and the LD50 reported was 90.3 mg/kg. 8
The therapeutic effects of Ns and TQ were attributed to their antioxidant activities and ability to induce detoxifying enzymes. 5,13,14 However, few chemoprotective enzymes were investigated and the exact mechanism of action is still ambiguous. The antioxidant and scavenging activity against free radicals are suggested mechanisms. The enzymes often considered included catalase, glutathione peroxidase, superoxide dismutase (SOD), and glutathione reductase. 5,9,13 To a lesser extent, other enzymes such as NAD(P)H-quinone oxidoreductase (NQO) and glutathione S-transferase (GST) were also documented. Thymoquinone elevated NQO and GST activities in mice. 14,15 Although extensive data are accumulating about the protective and potential therapeutic effects of both Ns and TQ, however, it was reported that the aqueous extract of Ns not only failed to protect against cancer induced by N-methyl-N'-nitro-N-nitrosoguandine but also was itself mutagenic. 16 It was also reported that TQ depleted hepatic glutathione (GSH), increasing oxidative stress and causing liver damage. 17 This discrepancy needed further investigation.
In the present study, the ability of Ns and TQ for inducing the messenger RNA (mRNA) levels and/or the activities of a wide range of chemoprotective and drug-metabolizing enzymes (DMEs) in healthy and carbon tetrachloride (CCl4)-challenged rats was investigated. The enzymes considered were GST, NQO, uridine diphosphate-glucuronosyltransferase (UDPGT), and microsomal epoxide hydrolase (mEH), γ-glutamylcysteine synthetase (γ-GCS), and GSH. Elevation of these chemoprotective enzymes is always favored as these enzymes protect the cells against the oxidative stress, which is implicated in the pathogenesis of a variety of diseases. Assuming that Ns and TQ resulted in a similar induction pattern of these enzymes, this would mean that TQ is the main active constituent of the seed responsible for its antioxidant effects. Given that the accumulated information known about the mechanisms involved in the control of the studied enzymes, and the broad range of enzymes under investigation, a conclusion could be drawn about the possible mechanism of action of Ns and TQ, which is currently not clearly understood.
To achieve the goals of the study, there was a need for a well-studied and well-understood model of chemically induced hepatotoxicity. CCl4 is widely used in industry because of its excellent solvent, nonflammable, and nonexplosive properties. It is used as a vermifuge and in fire extinguishers. Drinking water is a source of human exposure to CCl4. CCl4 is of concern in many countries as a drinking water contaminant, and despite the limited solubility of CCl4 in water, a sufficient amount could be administered orally to produce toxicity. 18 CCl4 is toxic in itself but, in addition, it generates reactive oxygen species or free radicals that overwhelm the antioxidant capacity of the biological system leading to oxidative stress. CCl4 is known to reduce GSH levels and activities of antioxidant enzymes such as GST, catalase, and SOD in the liver. 19 –21 Despite the fact that CCl4 has been comprehensively studied and reported extensively in the literature, it was surprising that the effects on key enzymes such as UDPGT, γ-GCS, NQO, and mEH were not reported after CCl4 intoxication; and out of almost 6500 articles on PubMed.gov, very few investigated the effects of CCl4 on these enzymes and with many discrepancies as well. CCl4 decreased UDPGT and NQO activities. 22,23 Contradictory reports showed that CCl4 elevated Nqo1 mRNA levels 24 and elevated UDPGT activity 25 in rats. CCl4 also caused reduction in mEH activity in rats. 25,26 Therefore, one goal of the present study was an attempt to rectify these discrepancies.
Materials and Methods
Chemicals
All chemicals were from Sigma (St Louis, Missouri) except where indicated in the specified methods. Dithymoquinone (DTQ) and thymohydroquinone (THQ) were prepared from TQ, 27 and the purified synthetic components were analyzed by thin layer chromatography (TLC) and nuclear magnetic resonance (NMR) spectroscopy, in addition to measuring the melting point to confirm their identity and purity. Nuclear magnetic resonance spectra were obtained using a Varian Unity 400 MHz Fourier transform-NMR spectrometer. TLC was carried out using Whatman (Clifton, New Jersey) flexible backed 60 Å silica gel plates (20 × 20 cm2 and 0.25 mm thickness) with visualization under ultraviolet (UV). C18 PrepSep solid phase extraction cartridges were obtained from Waters Chromatography Division (Milford, Massachusetts). The commercial Ns seeds were purchased from a large public market in Cairo, Egypt.
Extraction and Analysis of the Seeds
Extraction of the seeds was performed using a continuous technique in which the seeds were dried and milled. The powder was added to distilled water, boiled for 5 minutes, and then left at room temperature for 3 days and filtered. The water was then evaporated under vacuum using rotary evaporator. The quinone content of the seed extracts was then analyzed.
Preparation of DTQ and THQ
Thymohydroquinone (THQ) was prepared by dissolving sodium bisulfite (2.5 g) in a minimal volume of distilled water. Absolute ethanol was then added until a slight opalescence appeared, which was removed by the addition of a few drops of water. Thymoquinone (1.98 g, 0.01 mol) was dissolved in a minimum volume of absolute ethanol and added to the sodium bisulfite; the mixture was then left for 24 hours at 4°C. The separated colorless solid was filtered and washed with a small volume of water and alcohol. On acidification of a suspension of this derivative in water with 0.1 N HCl, extraction of the liberated compound in ether and crystallization from hot chloroform produced colorless crystals of THQ, with 89% yield. The crystals were collected by vacuum filtration and dried overnight under vacuum. The melting point of 143°C confirmed the identity and purity of the prepared compound. The TLC was performed using water/methanol/2-propanol (50:45:5) and the obtained Rf value was 0.88. 1 H NMR details were as follows: (d6-acetone, 400 MHz) δ 1.14, 1.16 (2d, J = 7 Hz, 6H, CH 3), 2.08 (s, 3H, CH 3), 3.20 to 3.23 (m, J = 7Hz, 1H, (CH3)2-CH), 6.55 (s, 1H, C4-OH), 6.63 (s, 1H, C1-OH), 7.29, 7.37 (s, 1H, C3-H and C6-H).
Dithymoquinone (DTQ) was prepared from the dimerization of TQ. A flask containing an ether solution of TQ (0.99 g, 0.01 mol) in a minimum volume of ether was whirled continuously until the ether completely evaporated leaving a thin layer of TQ. This was left in indirect sunlight for 1 week. Recrystallization of the product from 80% ethanol gave very pale yellow needles of DTQ, with 83% yield. The crystals were collected by vacuum filtration and dried overnight under vacuum. The measured melting point of 197°C confirmed the identity and purity of the prepared compound. The TLC was performed using water/methanol/2-propanol (50:45:5), and the obtained R f value was 0.50. 1 H NMR details were as follows; (d6-acetone, 400 MHz) δ 1.11, 1.12 (2d, J = 6 Hz, 12H, CH 3), 1.99 (s, 6H, CH 3), 2.96 to 3.00 (m, J = 6Hz, 2H, (CH3)2-CH), 6.52 (s, 2H, 3 and 3′ CH), 6.63 (s, 2H, 6 and 6′ CH).
Purification of the Extract
The extract was purified by passing through a C18 PrepSep solid phase extraction column (pre-eluted with methanol) prior to high-performance liquid chromatography (HPLC) analysis. A sample of seed extract (20 μL) followed by 800 μL (400 μL × 2) of methanol was passed through the column to afford an eluate free from greasy materials.
High-Performance Liquid Chromatography Instrumentation and Conditions
A C18 reversed-phase μBondaPak analytical column (300 × 3.9 mm2, 10 μm particle size) connected to a μBondaPak C18 guard cartridge (3.9 × 20 mm2; Waters Chromatography Division, Milford, Massachusetts) was used. The chromatographic system consisted of a Hitachi Model L-6200A pump equipped with a 4250 UV-Visible detector and an AS-2000 autosampler with a Rheodyne injection valve and a 100-μL sample loop (Hitachi, San Jose, California). Hitachi Model D-6000 version 2, revision 06 software was used to control system operation and facilitate data collection.
The isocratic mobile phase was composed of water:methanol:2-propanol (50:45:5 v/v) and was filtered through a 0.45-μm Millipore filter and deaerated before use. Analyses were performed at room temperature, and UV detection was carried out at 254 nm for TQ, DTQ, and thymol (THY). Thymohydroquinone was detected at 294 nm because of its low absorptivity at 254 nm. A flow rate of 1 mL/min was used. 28
Animal Treatment
Adult male Wistar albino rats (Rattus norvegicus, 130-180 g) were maintained in a humidity- and temperature-controlled environment on a 12-hour light/dark cycle with free access to standard rodent pellets and water. Animals were randomly divided into 6 groups of 6 animals each in clear suitable cages. Group 1 was the control group and received a single injection intraperitoneally (ip) of corn oil 1 mL/kg. Groups 2 and 3 were given by gavage either 50 mg/kg (w/v) water extract of Ns seeds (the same way people in Europe and the Middle East consume the seeds) or 5 mg/kg of pure TQ in corn oil (based on HPLC analytical data of almost 10% content) once daily for 5 consecutive days. Animals in group 4 were administered (ip) a single dose of CCl4 at 1 mL/kg in corn oil. Groups 5 and 6 were treated with Ns or TQ for 2 days before and 3 days after administration of CCl4 using the same dosing regimens described previously. The body weight gain percentage was determined from the following equation: ([final weight − initial weight]/initial weight) × 100.
Biological Sample Preparation
Animals were sacrificed 24 hours after the final dose, blood samples were collected for immediate serum preparation, and the livers were quickly perfused in situ (via the hepatic portal vein) with normal ice-cold saline. A 100-mg sample of liver was removed, homogenized in 2 mL of TRIzol solution (Invitrogen; Carlsbad, California), and frozen at −80°C for later RNA isolation. Another 100 mg sample was homogenized in 3 mL of 10% w/v of perchloric acid/1 mmol/L bathophenanthrolinedisulfonic acid, and the supernatant was used for measuring reduced GSH, oxidized glutathione (GSSG), and cysteine. The remaining portion of liver was homogenized in 0.25 mol/L sucrose (20% w/v) and subjected to a 3-stage differential centrifugation (9000g for 15 minutes, 19 000g for 15 minutes, and 105 000g for 60 minutes) to prepare the cytosolic and microsomal fractions. Protein content of both fractions was determined using Folin–Ciocalteu phenol reagent, 29 and the fractions were stored at −80°C until assayed for enzyme activity. All animal procedures were conducted in concordance with National Institutes of Health (NIH) guidelines for the humane care of laboratory animals.
Enzyme Activity
Serum alanine aminotransferase (sALT) activity was determined using a coupled reaction by monitoring the serum-dependent absorbance change of NADH oxidation at 340 nm in the presence of optimized concentrations of
Measurements of Thiolamines (GSH, GSSG, and Cysteine)
Thiolamines were measured by HPLC. 35 Stress ratio was calculated using the following formula.
Messenger RNA Determination
Hepatic mRNA levels were determined by Northern blotting of 20 μg of total RNA isolated by TRIzol extraction. Gel electrophoresis, nucleic acid transfer to membranes and 32 P probe labeling, washing stringency, and autoradiographic film development were all performed as described previously. 36
For GST-alpha (Gsta), the probe spanned the regions 4 to 755 of Gsta2 (K00136), giving 77% homology with both Gsta1 (NM001024361) and Gsta3 (NM031509). The Gstm probe spanned the regions 349 to 713 (NM017014), giving 82% homology with Gstm2 (NM177426), 90% homology with Gstm3 (NM020540), and 84% homology with Gstm6 (NM001109192). NAD(P)H-quinone oxidoreductase (Nqo1), microsomal epoxide hydrolase (Ephx1), and γ-Gcs probes spanned the regions 107 to 1495 of Nqo1 (J02608), 107 to 1531 of Ephx1 (M26125), and 251 to 820 of γ-Gcs (J05181), respectively. Nqo1 has no similarity to the other Nqo2 (BC079157) in rat. UDP-glucuronosyltransferase probes spanned the region 152 to 934 of Ugt1a6 (J02612), 243 to 1000 of Ugt1a9 (NM201425), and 8 to 710 of Ugt2b1 (M13506). Ugt1a9 probe gives ∼85% homology with Ugt1a7 (U75903) and Ugt1a8 (NM175846). All mRNA bands were normalized to the same-sample cyclophilin mRNA band to control for gel loading and transfer variations. Results are expressed as fold change from corn oil to control animals.
Statistical Analysis of Data
Results are expressed as the mean ± standard error of the mean (SEM). Treated group size was 6 animals. Statistical analyses were performed using analysis of variance (ANOVA), followed by Fisher protected least significant difference multiple range test. The data for Ns, TQ, and CCl4 were compared against those treated with corn oil, which in turn was not statistically different from naive animals. The data for CCl4-intoxicated animals treated with Ns or TQ were compared against those treated with CCl4 only. Differences were considered significant at P < .05.
Results
The HPLC analysis of the quinone content of the seed extract showed that TQ is the main quinone present in the seed extract (almost 10% w/w), while THQ content was only 1%. The thymol and DTQ content were 0.5% and 0.2%, respectively.
Treating rats with TQ significantly increased the body weight gain, while Ns did not significantly elevate the weight gain compared with control animals (Table 1). The CCl4 intoxication resulted in a significant reduction (∼24%) in weight gain. The reduction seen in the body weight gain after treatment with CCl4 was alleviated by either Ns or TQ treatment on comparison with CCl4-treated animals (Table 1). Similar scenario was reported for sALT, a serum marker of hepatotoxicity, where Ns and TQ did not significantly increase the enzyme activity in normal animals. The increase (∼40%) in sALT activity caused by TQ was not statistically significant (Table 1). Treating animals with CCl4 resulted in a 50-fold increase in sALT activity compared with control animals. On treating the CCl4-intoxicated animals with either Ns or TQ, significant reductions in enzyme activity (∼31-folds) were produced when compared with CCl4-treated animals (Table 1).
Effect of Nigella sativa and Thymoquinone on Serum Alanine Aminotransferase (a Serum Marker of Hepatotoxicity) and Body Weight Gain in CCl4-Intoxicated Rats
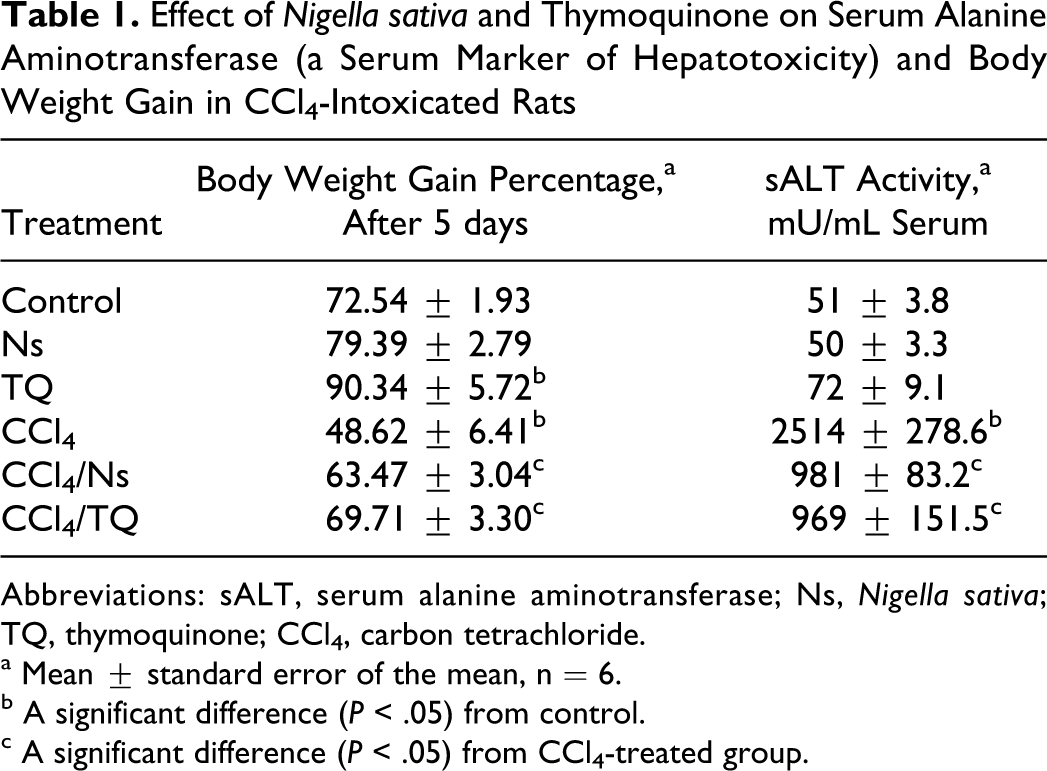
Abbreviations: sALT, serum alanine aminotransferase; Ns, Nigella sativa; TQ, thymoquinone; CCl4, carbon tetrachloride.
a Mean ± standard error of the mean, n = 6.
b A significant difference (P < .05) from control.
c A significant difference (P < .05) from CCl4-treated group.
Nigella sativa was more efficacious than TQ in elevating the GST activity (Table 2) and Gsta transcript (Figure 1). The elevations in activity and mRNA transcript caused by TQ did not achieve statistical significance. Neither Ns nor TQ affected Gstm transcript level in healthy animals (Figure 1). CCl4 reduced both GST activity and mRNA levels of both α and μ forms (Table 2 and Figure 1). Both Ns and TQ modulated the effects of CCl4; both significantly increased the enzyme activity and the transcript levels of Gsta and Gstm (Table 2 and Figure 1). Both Ns and TQ were also similar in their effects on another phase II drug-metabolizing enzyme UDPGT. Both elevated the enzyme activity (Table 2) and mRNA levels of Ugt −1a6, −1a9, and −2b1 in normal animals (Figure 2). Although CCl4 produced modest reductions in the enzyme activity and mRNA transcripts, neither the decline in the enzyme activity nor the reductions in any of the Ugt transcripts studied was statistically significant when compared with control rats. Approximately 50% reduction in Ugt2b1 level was not significant either. Both Ns and TQ elevated the transcripts of Ugt1a family in CCl4-intoxicated rats but without manifesting an increase in enzyme activity toward p-nitrophenol (PNP). Nigella sativa significantly elevated both Ugt −1a6 and −1a9, and TQ elevated only Ugt1a6 transcript level (Figure 2).
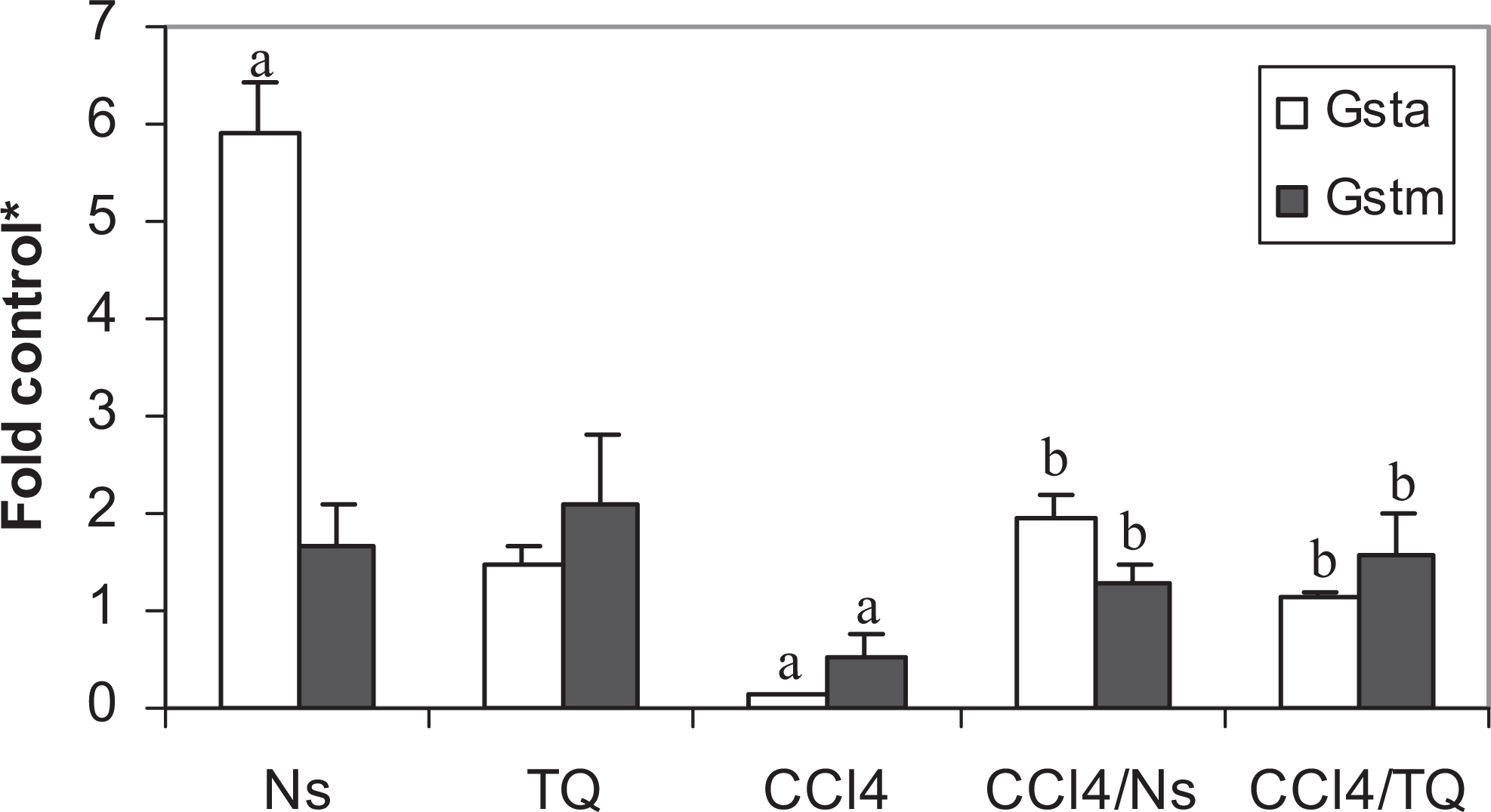
Effect of Nigella sativa (Ns) and thymoquinone (TQ) on glutathione S-transferase messenger RNAs ([mRNAs] Gsta and Gstm) in carbon tetrachloride (CCl4)-intoxicated rats. a Significant difference (P < .05) from control; b significant difference (P < .05) from CCl4-treated group; *fold control (mean ± standard error of the mean, n = 6) from the control value.
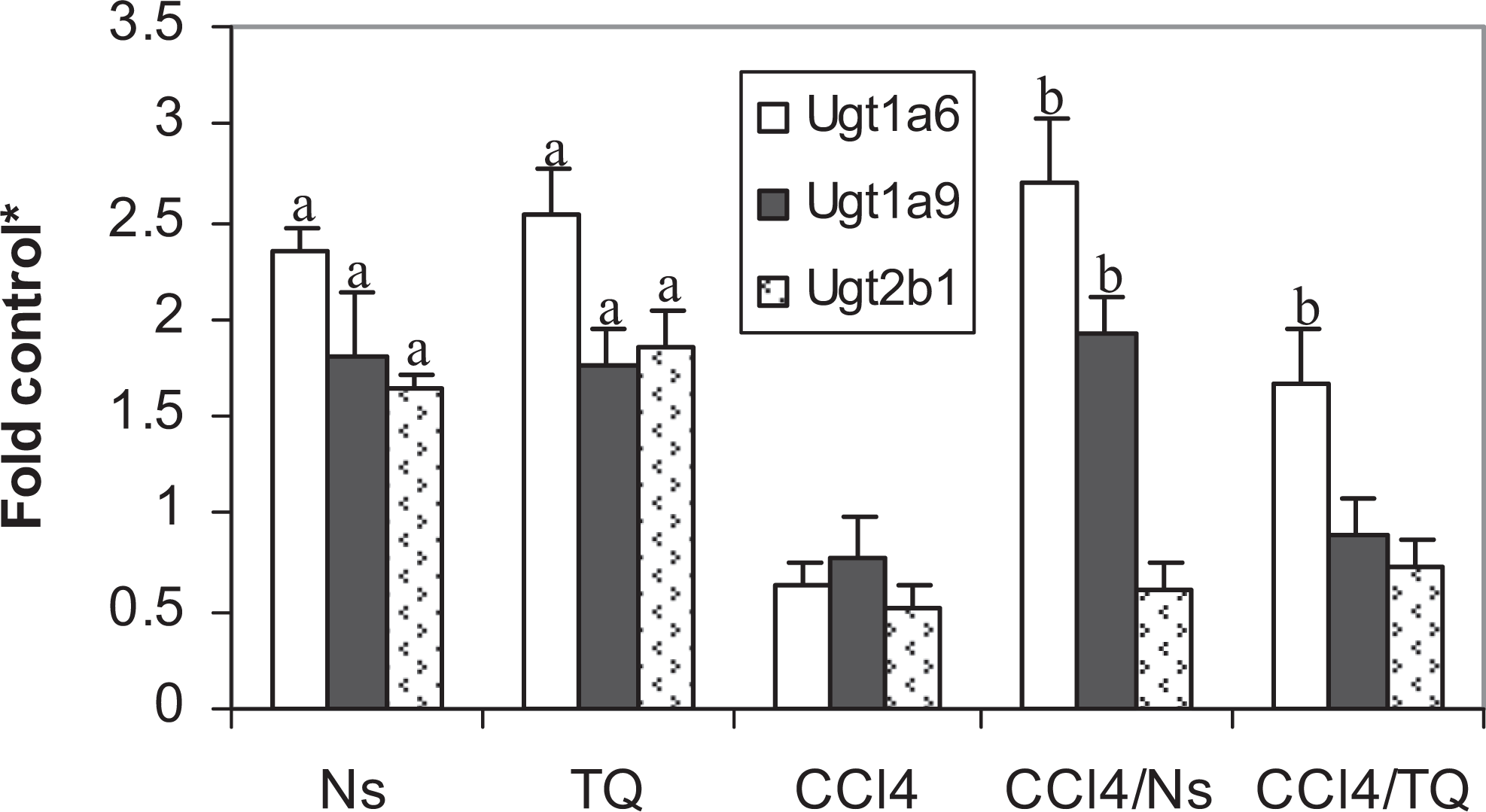
Effect of Nigella sativa (Ns) and thymoquinone (TQ) on UDP-glucuronosyltransferase messenger RNAs ([mRNAs] Ugt1a6, Ugt1a9, and Ugt2b1) in carbon tetrachloride (CCl4)-intoxicated rats. a Significant difference (P < .05) from control; b significant difference (P < .05) from CCl4-treated group; *fold control (mean ± standard error of the mean, n = 6) from the control value.
Effect of Nigella sativa and Thymoquinone on the Activities of Glutathione S-Transferase, Glucuronosyltransferase, Quinone Oxidoreductase, and Microsomal Epoxide Hydrolase in CCl4-Intoxicated Rats
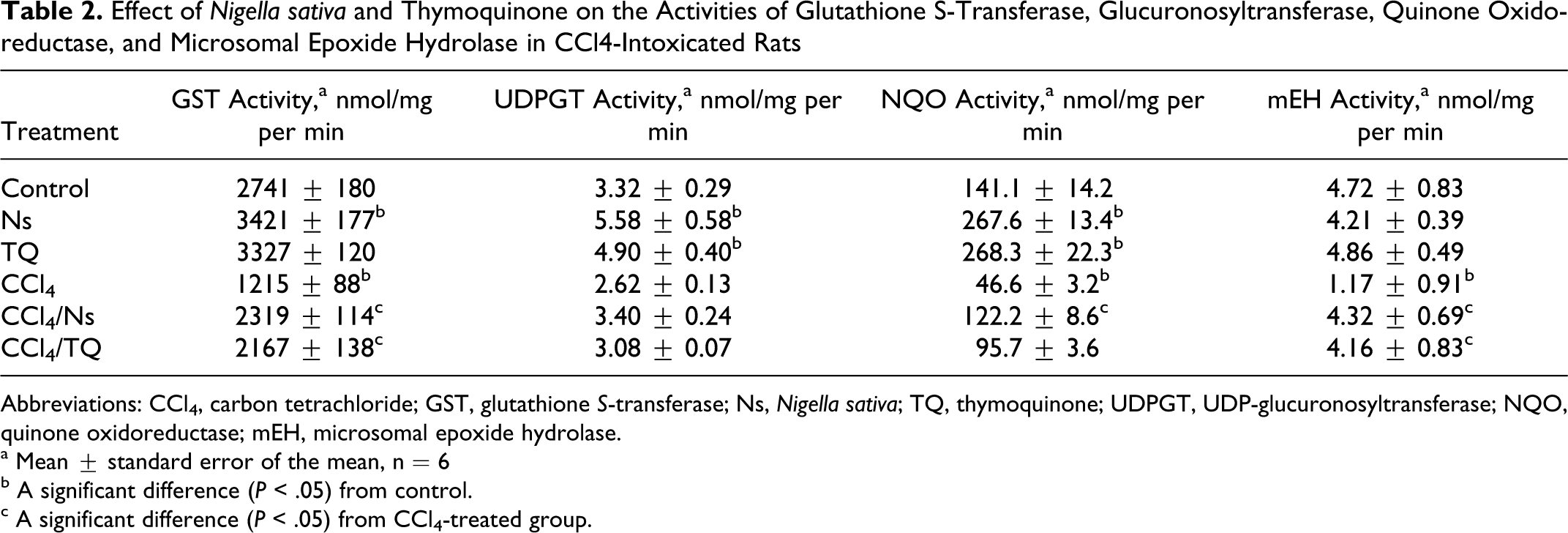
Abbreviations: CCl4, carbon tetrachloride; GST, glutathione S-transferase; Ns, Nigella sativa; TQ, thymoquinone; UDPGT, UDP-glucuronosyltransferase; NQO, quinone oxidoreductase; mEH, microsomal epoxide hydrolase.
a Mean ± standard error of the mean, n = 6
b A significant difference (P < .05) from control.
c A significant difference (P < .05) from CCl4-treated group.
Both Ns and TQ elevated the activity and mRNA level of NQO in control animals (Table 2 and Figure 3). Intoxication with CCl4 resulted in significant reductions in both the enzyme activity and the transcript level. Nigella sativa completely ameliorated the destructive effects of CCl4 and restored the enzyme activity and mRNA level, while TQ elevated only the transcript level to a lower extent than Ns, with no reflection on the enzyme activity. The 2-fold increase in enzyme activity caused by TQ compared with control was not enough to gain the statistical significance (Table 2). Neither Ns nor TQ affected mEH activity in healthy rats (Table 2), and only Ns elevated the enzyme transcript (Ephx1) level (Figure 3). CCl4 reduced both enzyme activity and mRNA level compared with control. Both Ns and TQ were able to counteract against these harmful effects of CCl4, and both the treatments caused significant increases in enzyme activity and transcript levels (Table 2 and Figure 3) when compared with CCl4-intoxicated animals.
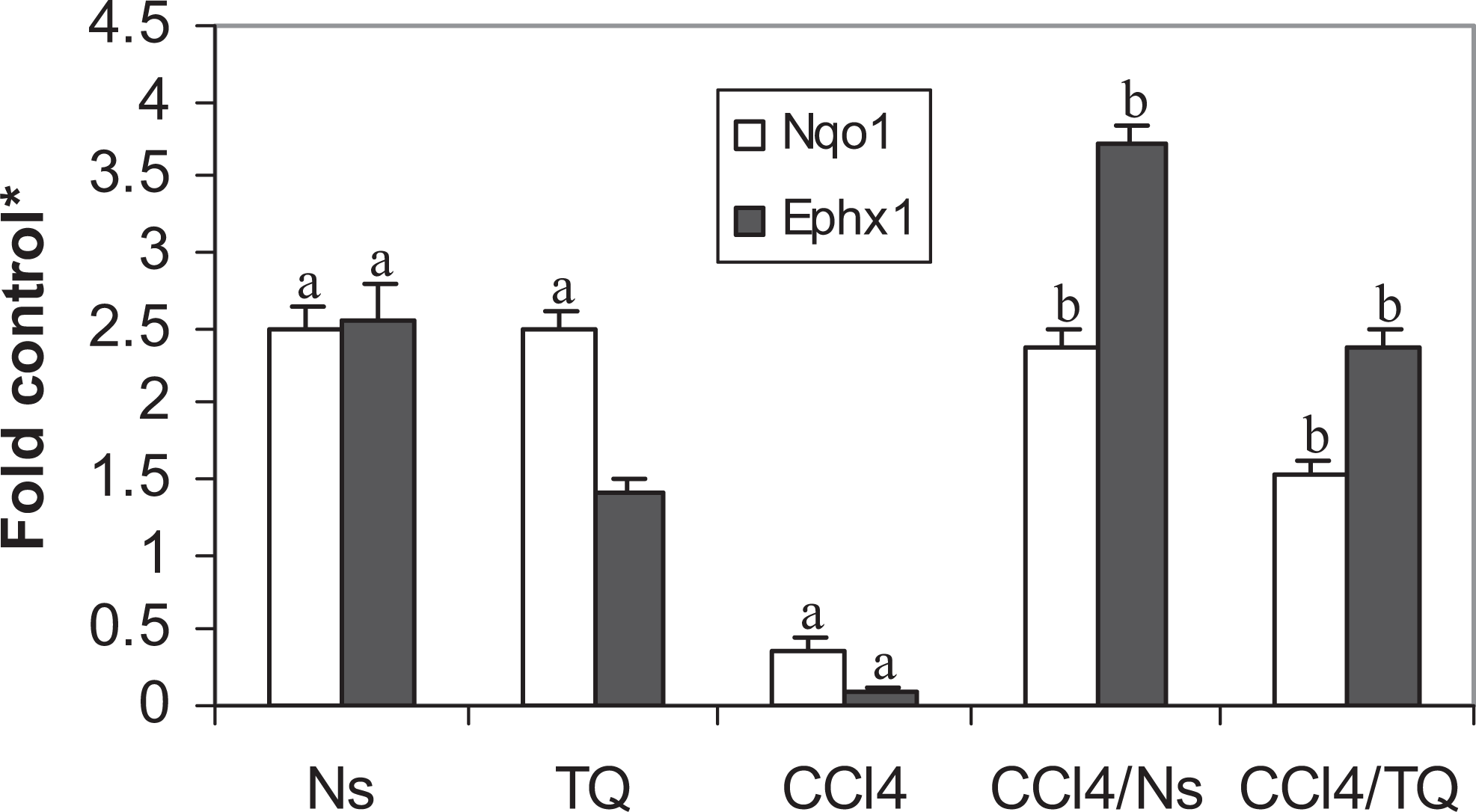
Effect of Nigella sativa (Ns) and thymoquinone (TQ) on NAD(P)H-quinone oxidoreductase (Nqo1) and microsomal epoxide hydrolase (Ephx1) messenger RNAs (mRNAs) in carbon tetrachloride (CCl4)-intoxicated rats. a Significant difference (P < .05) from control; b significant difference (P < .05) from CCl4-treated group; *fold control (mean ± standard error of the mean, n = 6) from the control value.
Responses of reduced GSH, disulfide form of glutathione (GSSG), stress ratio, and cysteine were very much similar. Treatment with either Ns or TQ did not result in a significant change in any of the aforementioned parameters compared with control animals (Table 3), while CCl4 intoxication caused a significant decline in GSH and cysteine and a significant increase in GSSG level and stress ratio. Treatment of CCl4-intoxicated animals with either Ns or TQ reversed the situation as demonstrated by the elevation in GSH and cysteine levels and the reduction in GSSG level and stress ratio on comparison with CCl4-treated animals (Table 3).
Effect of Nigella sativa and Thymoquinone on Glutathione Levels and γ-Glutamylcysteine Synthetase mRNA in CCl4-Intoxicated Rats
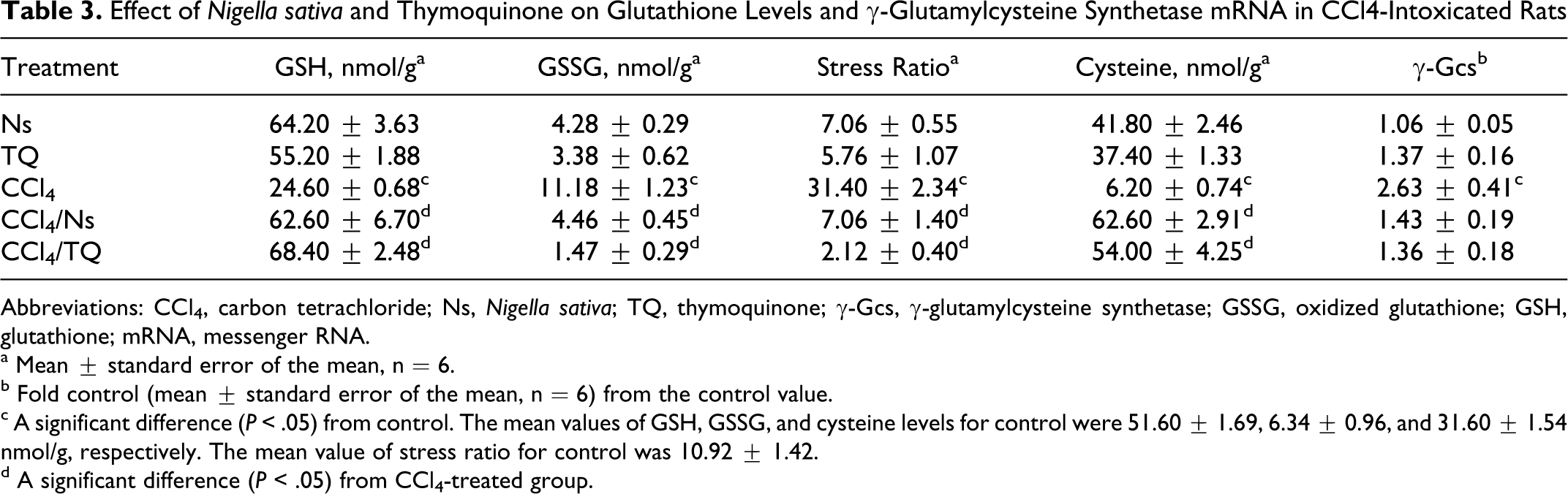
Abbreviations: CCl4, carbon tetrachloride; Ns, Nigella sativa; TQ, thymoquinone; γ-Gcs, γ-glutamylcysteine synthetase; GSSG, oxidized glutathione; GSH, glutathione; mRNA, messenger RNA.
a Mean ± standard error of the mean, n = 6.
b Fold control (mean ± standard error of the mean, n = 6) from the control value.
c A significant difference (P < .05) from control. The mean values of GSH, GSSG, and cysteine levels for control were 51.60 ± 1.69, 6.34 ± 0.96, and 31.60 ± 1.54 nmol/g, respectively. The mean value of stress ratio for control was 10.92 ± 1.42.
d A significant difference (P < .05) from CCl4-treated group.
CCl4 was the only treatment that significantly elevated γ-Gcs mRNA level. Both Ns and TQ had no significant effects on γ-Gcs levels in healthy or in CCl4-treated animals (Table 3).
Discussion
Searching for natural remedies for human ailments is an ongoing process. Thousands of indigenous herbs are used throughout the world. Many of these herbs have successfully served as a source of various drugs in use today and will continue to provide cheap, safe, and effective drugs. In the present study, the effects of Ns and TQ (one of the its major constituents) on chemoprotective and drug-metabolizing enzymes in healthy and CCl4-intoxicated rats were investigated. Although Ns and TQ are used extensively to treat many disorders, the identification of the exact mechanism of action has been elusive. This study was undertaken in an attempt to (i) understand the mechanism of action of Ns and TQ, (ii) test whether there are other active constituents in the seeds or whether TQ is the only active constituent responsible for antioxidant activity, and (iii) explore the effects of the environmental hepatotoxin CCl4 on drug-metabolizing enzymes, a goal that was not originally planned for.
Body weight loss is a typical feature of CCl4-induced hepatotoxicity. 13 In the present study, both Ns and TQ alleviated the reductions seen in body weight gain after treatment with CCl4 (Table 1). TQ significantly increased weight gain in healthy rats (Table 1). Neither Ns nor TQ elevated sALT, a serum biomarker of hepatotoxicity, in healthy rats but TQ caused an insignificant (∼40%) increase in the enzyme activity. High doses of TQ (∼50 mg/kg) were reported to deplete the hepatic GSH, 14,17 cause oxidative stress and hepatic injury in mice, 8 and have genotoxic and cytotoxic activities in hepatocytes. 17 However, TQ at 12.5 mg/kg was reported to be very safe 8 and the LD50 in rats after oral ingestion was 794.3 mg/kg. 12 In the current study, the dose used was much less, but the reported elevation in alanine aminotransferase activity caused by TQ treatment could be an early indication of toxicity. Further investigation is needed for the toxicological effect of TQ.
As expected, CCl4 resulted in a 50-fold elevation in sALT, indicating severe damage of hepatic parenchymal cells and leakage of the enzyme. This is a well-known effect of CCl4. 21,37 Both Ns and TQ caused more than 30-fold reductions in sALT in CCl4-challenged animals (Table 1), probably because of their abilities to scavenge the free radicals produced by the hepatotoxin or by elevating the cellular antioxidant enzymes and stabilizing the cell membranes. This could be best explained by the induction pattern of Ns and TQ on GST, NQO, and mEH. These enzymes are all implicated in the removal of reactive oxygen species and detoxification of xenobiotics making the cell resistant to the debilitating assaults of the CCl4. These enzymes can also detoxify a diversity of electrophilic species arising from CCl4 metabolism. Both Ns and TQ modulated the reductions caused by CCl4 in these enzymes. CCl4 decreased the activities of GST, NQO, and mEH by ∼56%, 67%, and 75%, respectively (Table 2). This is in accordance with the previous reports. 19,22,25,26 Treating the intoxicated animals with Ns resulted in ∼200%, 262%, and 369% increase in the activities of these enzymes, respectively, affording protection against CCl4-induced reductions (Table 2). Thymoquinone was somewhat less efficacious than Ns, elevating the activities of these enzymes by 178%, 205%, and 355%, respectively. This is in concordance with previous reports. 14,15 These elevations in the activities of enzymes were escorted by upregulation in the mRNA levels. Nigella sativa was more efficacious than TQ, probably because of the many active components of the seeds that might act synergistically, amplifying the induction effect. In CCl4-challenged animals, Ns caused 16-, 7-, and 37-fold increases in Gsta, Nqo1, and Ephx1 transcript levels and TQ resulted in 10-, 4-, and 24-fold increases in the mRNA levels of these enzymes (Figures 1 and 3), respectively. Both Ns and TQ were similar in their effect on mRNA level of Gstm causing a 3-fold induction (Figure 1). Thus, the increases seen in activities could be related, but not restricted, to the ability of Ns and TQ of inducing the transcripts of these enzymes. However, the methodology used in the present study does not differentiate between increasing the induction rate, stabilizing the mRNA, or any translational or posttranslational effects caused by either Ns or TQ. This is typified by the induction of Ephx1, by Ns without exhibiting any effect on the mEH activity in control rats (Table 2 and Figure 3), and by the significant elevations seen in Ugt1a6 after treatment with Ns and TQ, and Ugt1a9 after treatment with Ns that were all not accompanied by significant elevations in enzyme activity either (Table 2 and Figure 3). Although TQ was devoid of any significant effect on either activities or mRNA levels of GST and mEH in healthy rats, Ns elevated both GST activity and Gsta and Ephx1 transcript levels (Table 2 and Figures 1 and 3). Both Ns and TQ elevated NQO activity and Nqo1 level in normal animals (Tables 2 and Figure 3). Elevated levels of Gsta, Gstm, and Ephx1 were reported to protect against CCl4 hepatotoxicity in previous studies. 38,39 The parallel responses of mEH, NQO, and GST were previously reported, 40 –42 suggesting a common pathway of regulation. The 3 enzymes, GST, NQO, and mEH, are all upregulated by nuclear-related factor (Nrf2)/antioxidant response element (ARE), indicating that Ns and TQ might be potential activators of the Nrf2-/ARE-dependent detoxification pathway. Induction of Nrf2 protected against hepatotoxicity in rats. 43
Both TQ and Ns were similar in their effects on UDPGT; both caused ∼150% elevation in the enzyme activity and ∼2-fold induction in Ugt −1a6, −1a9, and −2b1 mRNA levels in normal animals (Table 2 and Figure 2). Elevations of UDPGT activity and/or mRNAs were found to protect against hepatotoxicity in rats. 43 Thymoquinone was suggested to elevate the UDPGT activity through aryl hydrocarbon receptor (AhR). 44 CCl4 did not result in a significant decrease in either UDPGT activity or any of the Ugt transcripts investigated, although it was shown to decrease the activity of the enzyme. 23
Neither Ns nor TQ affected reduced GSH, disulfide form of glutathione or GSSG, stress ratio, or cysteine level in normal rats. The deleterious effects of CCl4 on GSH were all ameliorated by either Ns or TQ; both neutralized the reductions in GSH and cysteine, and the elevations in GSSG and stress ratio caused by CCl4 (Table 3). The reduction in cysteine could provide an explanation to the decrease in GSH caused by CCl4 since the availability of cysteine is known to be a rate-limiting factor in the first reaction involved in GSH synthesis. The negative effects of CCl4 on GSH levels are well documented and can be reviewed elsewhere, 19,21 while the elevations in GSH by either Ns or TQ in animals challenged by various toxins including CCl4 are reviewed elsewhere. 9,13,15 γ-Glutamylcysteine synthetase (γ-GCS) is one of the enzymes responsible for GSH synthesis, the response of the enzyme transcript was unique throughout the study, being elevated only by CCl4 (Table 3). The decrease in GSH caused by CCl4 resulted in the induction of γ-Gcs probably through a feedback mechanism to overcome the drastic reduction in GSH, and this may be a late defense response.
Conclusions
In many instances, Ns was more efficacious than TQ in elevating the cellular antioxidant milieu and hence the protection against hepatotoxicity indicating that there are other synergistic active constituents in the seeds responsible for the protection against the hepatotoxic effects induced by CCl4. The protection against CCl4 is most probably exerted through improving the antioxidant status of the cell by upregulation or induction of many detoxifying enzymes and GSH. Therefore, Ns could be safely used against chemically induced hepatotoxicity.
Footnotes
Acknowledgment
The author thanks Dr Yang Xie (School of Pharmacy, University of Utah) for assistance with 1 H NMR analyses.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.