Abstract
Paeonol, a natural phenolic compound, possesses diverse beneficial effects including antioxidant and anti-inflammatory effects. Gastric ulcer is still the most prevalent irritant illness among the gastrointestinal diseases. The present study explored the protective effect of paeonol at two dose levels in indomethacin (IND)-induced gastric ulcer in rats. Forty-eight male Wistar rats were arranged into six groups: control, paeonol-treated, IND-treated, IND/paeonol (low and high doses)-treated, and ranitidine-treated groups. The oxidative status was evaluated by determining malondialdehyde level, superoxide dismutase activity, reduced glutathione content as well as hemoxygenase-1 (HO-1) gene expressions, and the antioxidant protein; NAD(P)H quinone oxidoreductase 1 (NQO1) immunostaining. The pro-inflammatory genes nuclear factor κB (NF-κB) and interleukin 1β (IL-1β) were estimated together with the proapoptotic gene of caspase 3. IND caused multiple gastric ulcers with evident oxidative damage and elevated pro-inflammatory and proapoptotic markers. Paeonol protected significantly, in a dose-dependent manner, the gastric mucosa from ulcerative lesion of IND similar to the reference drug ranitidine. Paeonol pretreatment diminished gastric oxidative stress and restored the gastric antioxidant capacity by elevating gastric gene expression of HO-1 and protein expression of NQO1. Paeonol also reduced NF-κB, IL-1β, and caspase 3 gene expressions. In conclusion, paeonol offered a gastroprotection dependent on its antioxidant, anti-inflammatory, and antiapoptotic effects.
Keywords
Introduction
Gastric ulcer is an irritating disease with terrible complications. It affects seriously the quality of life. 1 The underlying causes of its prevalence include increase usage of nonsteroidal anti-inflammatory drugs (NSAIDs), smoking, emotional stress as well as the infective burden of Helicobacter pylori. 2 The current effective therapeutic strategies for ulcer embrace firstly antisecretory drugs, 3 which tend to decrease hydrochloric acid sufficiently to permit ulcer healing. The second intervening limb for therapy is to construct and retain the gastroprotective wall. 4 The pathogenesis of gastric ulcer is due to imbalance between the two previous causes. This imbalance changes gastric cellular redox status and favors oxidative stress and inflammatory damage of the mucosa. 5 The disastrous complications of ulcer, as perforation, together with its recurrence mandate continuous searching for new anti-ulcer modalities. 6
The integration of plants in medicine is an ancient successful modality for the treatment and forms the basis of much of modern therapy. Numerous natural substances with antioxidant and anti-inflammatory prospective were examined for anti-ulcerative effect with potential safety profile. 7,8 Paeonol, a phenolic compound isolated from paeonies, has been shown to have beneficial properties including antioxidant and anti-inflammatory properties. 9 These properties of paeonol offered the basis for its function as an organ protective agent in various disease models. 10 –12 Despite that the role of paeonol in the gastrointestinal diseases was examined, 13 the mechanisms underlying its gastroprotective effect need to be clarified. The present study aimed at elucidating possible mechanisms underlying paeonol protective effect on gastric ulcer. Taking into consideration the pivotal role of NSAIDs in ulcer formation, indomethacin (IND) was used as a reliable method of induction of experimental ulcer in different reports. 14,15
Materials and methods
Drugs and chemicals
IND and paeonol were procured from Sigma Chemical Co. (St Louis, Missouri, USA). Ranitidine was obtained from Medical Union Pharmaceuticals (MUP; Ismailia, Egypt). Rabbit polyclonal NAD(P)H quinone oxidoreductase 1 (NQO1) antibody was purchased from Invitrogen/Thermo Fisher Scientific (Fremont, California, USA). Other chemicals or reagents were of analytical grade.
Animals and experimental procedures
Forty-eight male Wistar rats weighing 200 ± 20 g were randomly set in six groups of eight rats each. The rats were acclimatized to the animal house conditions (12-h lighting cycle and 25 ± 2°C temperature) for 1 week and then arranged as normal control, paeonol control (200 mg/kg orally), 16 ulcer model IND (50 mg/kg orally) 15 -treated, IND/paeonol (50 mg/kg orally) 12 -treated, IND/paeonol (200 mg/kg orally)-treated, and positive control ranitidine (50 mg/kg orally) 17 -treated groups. Both paeonol and ranitidine were administered once daily for 3 days before ulcer induction. The study was done in accordance with the internationally accepted principles for laboratory animal use and care that followed the Ethics Committee for the Care and Use of Laboratory Animals in Faculty of Medicine, Minia University, Egypt. Five hours after IND administration, rats were anesthetized and killed. The stomach was dissected, opened, and washed with saline to calculate the ulcer index for the visible lesions grossly according to Harusato et al. 18 Part of the stomach was immersed in 10% neutral-buffered formalin, other parts were snipped freeze in liquid nitrogen, and then transferred to −80°C. These parts were subsequently processed for analysis of oxidative parameters and RNA extraction.
Biochemical analysis in gastric mucosa
To assess the oxidative damage induced by IND, a lipid peroxidation end product, malondialdehyde (MDA) was determined in the scraped gastric mucosa as described by Buege and Aust. 19 Reduced glutathione (GSH) level and superoxide dismutase (SOD) enzyme activity were assessed chemically depending on the methods described by Moron et al. 20 and Marklund and Marklund, 21 respectively.
Real-time polymerase chain reaction
Gene quantifications for hemoxygenase 1 (HO-1), nuclear factor κB (NF-κB), interleukin 1β (IL-1β), and caspase 3 were assessed by real-time polymerase chain reaction (PCR). Total RNA was taken out from gastric mucosa using RiboZol RNA extraction reagent (Amresco, Solon, Ohio, USA) succeeding the manufacturer’s instructions. Complementary DNAs (cDNAs) were produced using RevertAid™ First Strand cDNA Synthesis kit (Fermentas/Thermo Fisher Scientific), and then, they were reversely transcribed. Real-time PCR was done with 50-ng cDNA per reaction using Verso SYBR Green 1-Step qRT-PCR Kit (Thermo Fisher Scientific) containing 20 μM of specific primers in the PCR detection system. The SYBR green data were examined and quantified relative to glyceraldehyde-3-phosphate dehydrogenase as the reference gene. Table 1 enumerates the sets of primers used. The relative level of expression of each gene was calculated according to VanGuilder et al. 27 They were scaled relative to controls where control samples were set at a value of 1.
Primers used for real-time PCR analysis.

PCR: polymerase chain reaction; HO-1: hemoxygenase-1; IL-1β: interleukin-1β; NF-κB: nuclear factor-κB; GAPDH: glyceraldehyde-3-phosphate dehydrogenase.
Histological studies and immunohistochemical localization
Parts of the stomach fixed in 10% neutral-buffered formalin were prepared in paraffin and stained with hematoxylin and eosin stain for histopathological examination. Immunostaining was performed following instructions of kits strictly. Briefly, slides were deparaffinized and prepared for antigen retrieval and blockade of the endogenous peroxidase activity. Incubation with the primary antibody against NQO1 diluted in 1:100 overnight at 4°C was followed by the secondary antibody and diaminobenzidine applications. Scoring was performed according to Cui et al. 28
Statistical analysis
The results of the study are articulated as the mean ± SEM. One-way analysis of variance followed by the Tukey–Kramer posttest were performed for analysis of data. The value of p < 0.05 was considered significant.
Results
Effect of paeonol on ulcer index and gastric injury
Administration of IND produced extensive ulcerations in the gastric mucosa. Ulcerative indices were improved significantly by ranitidine and paeonol at both doses (Figure 1(a) and (b)). Microscopically, the gastric lining of IND-treated rats showed multiple areas of shedded mucosa and underlying inflammatory cells recruitment. While both drugs protected the mucosa from the ulcerative effect of IND, paeonol at 200 mg/kg offered extreme protection (Figure 2).
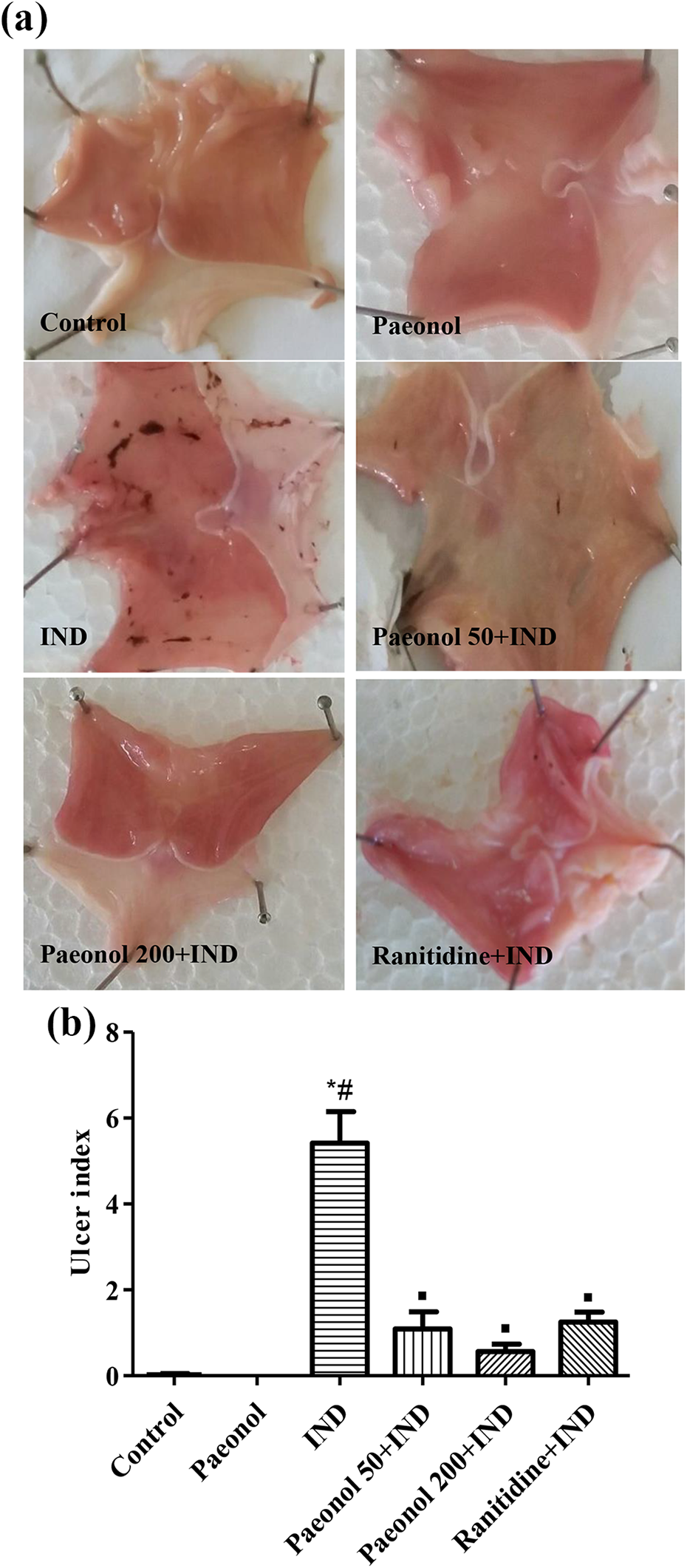
(a) Macroscopic picture of gastric mucosa showed the changes induced by indomethacin (IND) as red hemorrhagic bands of different sizes and the effect of paeonol and ranitidine on these gastric lesions. (b) Ulcer index. *,#,▪Significantly different from control, paeonol, and IND groups, respectively, at P < 0.05.

A photomicrograph of gastric mucosa stained with hematoxylin and eosin at ×100 magnification showing control rats with normal fundic glands (curved arrows). IND-injected rat shows severe ulceration of the mucosa (long arrow). While both paeonol 50 mg/kg and ranitidine-treated rats showed superficial erosion (short arrow), mucosa of rat pretreated with paeonol 200 mg/kg appears nearly normal. IND: indomethacin.
Effect of paeonol on oxidant status parameters
The data in Table 2 showed that IND administration elevated mucosal gastric content of MDA consistent with significant drop of cellular GSH content and SOD activitiy compared to control rats. Pretreatment with paeonol (50 and 200 mg/kg) improved the oxidant status significantly by augmenting SOD activity and GSH level and reducing MDA level.
Effect of paeonol on gastric mucosal MDA and GSH levels as well as SOD activity in IND-induced ulcer in rats.a

MDA: malondialdehyde; GSH: reduced glutathione; SOD: superoxide dismutase; IND: indomethacin.
a All parameters are expressed as means ± SEM (n = 8).
b Significantly different from control group at p < 0.05.
c Significantly different from paeonol group at p < 0.05.
d Significantly different from IND group at p < 0.05.
e Significantly different from paeonol 50 + IND group at p < 0.05.
f Significantly different from paeonol 200 + IND group at p < 0.05.
Effect of paeonol on gene expressions of HO-1, NF-κB, IL-1β, and caspase 3
Quantification of HO-1 gene in gastric mucosa indicated increased expression in all treated groups and its decrease in the IND-induced ulcer group (Figure 3(a)). High dose of paeonol showed significant induction of HO-1 as compared to the low dose. The pro-inflammatory genes for NF-κB and IL-1β were upregulated by IND administration; however, paeonol dose dependently disallowed their elevation. The high dose of paeonol significantly reduced their levels as compared to the low paeonol dose and ranitidine. Caspase 3 gene, a marker of apoptosis, is significantly elevated in the ulcer group; all treatments decreased its level significantly compared to the ulcer group. Paeonol 200 mg/kg showed a significant drop in caspase 3 level compared to the paeonol 50 mg/kg.

Real-time PCR for quantification of mRNA in gastric mucosa. Relative expression of (a) HO-1, (b) NF-κB, (c) IL-1β, and
Effect of paeonol on immunoexpression of NQO1
Control rats expressed normal gastric mucosal immunostaining for NQO1. Little or no staining was observed in the IND-induced ulcer group. Treatment with paeonol at the two doses significantly induced NQO1 expression in the gastric mucosa as compared to ulcer group. Ranitidine failed to significantly alter NQO1 expression (Figure 4).

(a) A photomicrograph of gastric mucosa of rats showing NQO1 expression immunohistochemically stained, ×400. Control rats showing positive NQO1 expression in the glandular tissue as well as the interstitium, while rat of indomethacin (IND)-induced ulcer showed nearly negative NQO1 expression. Paeonol pretreatment upregulated NQO1 expression at both doses, especially in the glandular tissue. The reference drug, ranitidine, failed to restore the NAQ1 expression after its decrease in ulcer group. (b) NQO1 semiquantative scoring. *,#,▪,°,ΔSignificantly different from control, paeonol, IND, paeonol 50 + IND, and paeonol 200 + IND groups, respectively, at P < 0.05.
Discussion
The present study revealed gastroprotective influence of paeonol at both low and high doses that was mediated through its antioxidant, anti-inflammatory, and antiapoptotic effects. Paeonol ability to reduce the ulcer index was comparable to the reference drug, ranitidine, and in accordance with the gastroprotection reported by Zhang et al. 13
Oxidative damage observed in the gastric mucosa of IND group was well explained by Maity et al. 29 Paeonol ameliorated IND-induced lipid peroxidation in the gastric mucosa may be owed to its previously reported antioxidant property. 10,11,13 The current study shows that paeonol replenished GSH content, restored SOD activity, and increased HO-1 gene expression in the gastric mucosa. Therefore, these antioxidant parameters could partly be involved in the observed decrease in lipid peroxidation.
The beneficial role of HO-1 was demonstrated in the gastrointestinal diseases collectively 30 and in NSAIDs-induced gastric ulcer particularly. 31 HO-1 is a crucial cytoprotective enzyme responsible for heme degradation that dampens different oxidant radicals and enhances the cellular antioxidant capacity. In the present study, the increment in the important nonenzymatic antioxidant GSH may be attributed to increase in HO-1 expression. 32 Moreover, carbon monoxide derived from HO-1 can increase gastric ulcer healing process by vasodilatation and regulation of gastric blood flow at ulcer margin. 33
Other antioxidant enzyme that works side by side to HO-1 to guard against oxidative damage is NQO1. 34 NQO1 is a reductase enzyme that uses NADH or NADPH as reducing cofactors to catalyze the reduction of different reactive species to less toxic compounds. 35 NQO1 is prominently involved in the pathogenesis of gastric ulceration. 36 Paeonol increased NQO1 protein expression in the gastric tissues of IND-treated rats as a part of its antioxidant effect as demonstrated in myocardial tissue by Li et al. 37 The former study reported that both HO-1 and NQO1 antioxidant enzyme genes were induced under oxidative stress condition by the same nuclear factor, Nrf2. However, HO-1 broadens its cytoprotective effect on the cell to include anti-inflammatory properties. 38
In the present study, inflammation in the ulcer group was evident by increased gene expressions of both NF-κB and IL-1β. NF-κB not only mediates the inflammatory response but also acts as a link connecting inflammation and cancer development in gastric ulcer. This transcriptional factor is encoded by a set of genes containing homologous domains as p65 that regulate NF-κB activity and function. 39 Implication of the inflammatory cytokine IL-1β in either pathogenesis or outcome of peptic ulcer was documented by Garcia-Gonzalez et al. 40 In the current study, paeonol showed dose dependent decrease in their gene expressions in line with its anti-inflammatory effect reported by He et al. 41 Nevertheless, the decreased NF-κB gene expression following paeonol treatment may be secondary to its antioxidant activity as oxidative stress can stimulate transcription factors, including NF-κB. 42,43
IND-induced caspase-3 activation is an important pathological event during the process of gastric epithelial cells apoptosis. 29,44 In the present study, rats in the ulcer group showed a significant increase of caspase 3 gene expression that was regressed by paeonol treatment dose dependently. In line with our findings, paeonol significantly reduced epirubicin-induced 45 and lipopolysaccharides-induced 46 activation of caspase 3.
Alternatively, NF-κB induces expression of target genes that result in apoptosis through activation of caspases including caspase 3. 47 In the current study, the effect of paeonol on NF-κB and caspase 3 is consistent with the results of Bao et al. 10 who reported that paeonol suppresses the activation of NF-κB in endothelial cell therefore might inhibit cell apoptosis via decreasing of caspase 3 expression.
In conclusion, paeonol has a valued anti-ulcer potential. The gastric protective property of paeonol is due, at least partly, to inhibition of oxidative stress, inflammation, and apoptosis in the gastric ulcer via modulating their gene and/or protein expressions.
Footnotes
Acknowledgements
The authors would like to thank Dr Hanaa H. Mohamed (Assistant Professor of Histology, Histology Department, Minia University, Egypt) for assistance in histopatholgy and immunohistochemistry.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
