Abstract
In the process of drug design, it is important to consider potential structural alerts that may lead to toxicosis. This work illustrates how using trifluoroethane as a part of a novel chemical entity led to cytochrome P450 – mediated N-dealkylation and the formation of trifluoroacetaldehyde, a known testicular toxicant, in exploratory safety studies in rats. Testicular toxicosis was noted microscopically in a dose-dependent manner as measured by testicular spermatocytic degeneration and necrosis and excessive intratubular cellular debris in the epididymis. This apparent toxic effect correlated well with the dose-dependent formation of trifluoroacetaldehyde, identified from in vitro rat liver microsome metabolism studies. A similar safety study performed with an N-tetrazole substitution in place of the N-trifluoroethane showed no evidence of testicular injury, implicating further the role of trifluoroacetaldehyde in the testicular lesion observed. These results highlight the relevance of early metabolic and safety testing in assessing potential structural alerts in drug design.
Introduction
The avoidance of structural alerts in drug design reduces the probability that a new drug will lead to toxicosis through a bioactivation mechanism.1,2 At the same time, it is appreciated that simply having a structural alert within a new chemical entity does not by default lead to injury as other factors such as species differences, dose, duration of use, quantity, and detoxification of reactive metabolites, and multiple metabolic pathways play an important role in its ultimate adverse reaction profile. The purpose of this work was to highlight the need to consider the formation of a structural alert due to metabolism in the process of drug design. In particular, the use of trifluoroethane conjugated to a nitrogen, oxygen, or sulfur in a molecule can lead to the formation of trifluoracetaldehyde (TFALD) through cytochrome P450 (CYP450) N-, O-, or S-dealkylation. Trifluoracetaldehyde has been reported in the literature to cause testicular injury in the rat.3–5 These effects were characterized by reduced testis weight and morphological changes including damage to late pachytene and dividing spermatocytes with effects increasing in severity with dose.3–5 Similar morphological effects have been reported in the literature for other toxicants (eg, Ethylene Glycol Monomethyl Ether [EGME]) that resulted in a reduction in fertility in rats.
6
Being an alkyl aldehyde, TFALD is capable of covalently binding to biological macromolecules, which is generally recognized as an initial event in the etiology of many chemical-induced toxicoses. The parent compound (
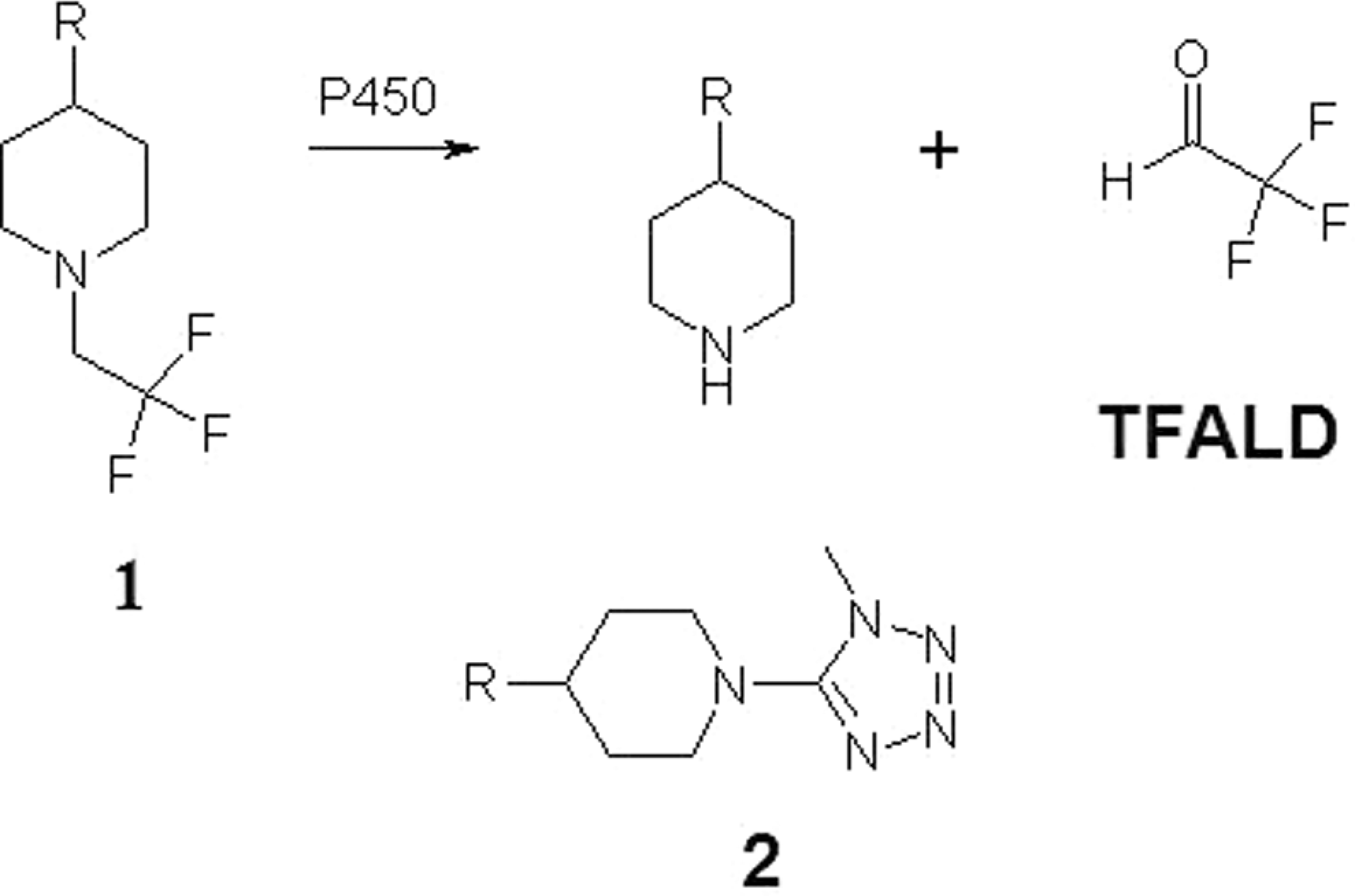
Formation of trifluoroacetaldehyde (TFALD) through cytochrome P450 – mediated N-dealkylation of parent compound
Experimental Procedures
Materials
General materials used were of analytical grade or better unless indicated. Compounds
In Vitro Metabolism Studies
Compound
In Vivo Safety Testing
The Pfizer Institutional Animal Care and Use Committee reviewed and approved the animal use in these studies. The Association for Assessment and Accreditation of Laboratory Animal Care International fully accredits the Pfizer animal care and use program.
The safety and exposure of compound
At necropsy, the lungs, liver, kidney, mesentery, and mesenteric lymph nodes, heart, and spleen were immersed in 10% neutral-buffered formalin (NBF). The testes and epididymis from both sides were fixed in modified Davidson’s solution. All tissues were processed whole on a Sakura VIP 5 series by dehydrating through a series of graded ethanol solutions, cleared with xylene, and impregnated with paraffin. Sections of 4 μm thickness were cut to expose the microstructures of each organ. Sectioned tissues were heated in a 60°C oven for a minimum of 1 hour, stained via automated linear stainer with hematoxylin-eosin, and coverslipped. The processed glass slides were evaluated under an Olympus light microscope and the images were captured by a SPOT Insight Firewire Camera and analyzed by SPOT software Advanced (Diagnostic Instruments, Inc, Sterling Heights, Michigan).
Results
The in vitro intrinsic liver metabolic clearance of compound
From in vivo safety studies with compound
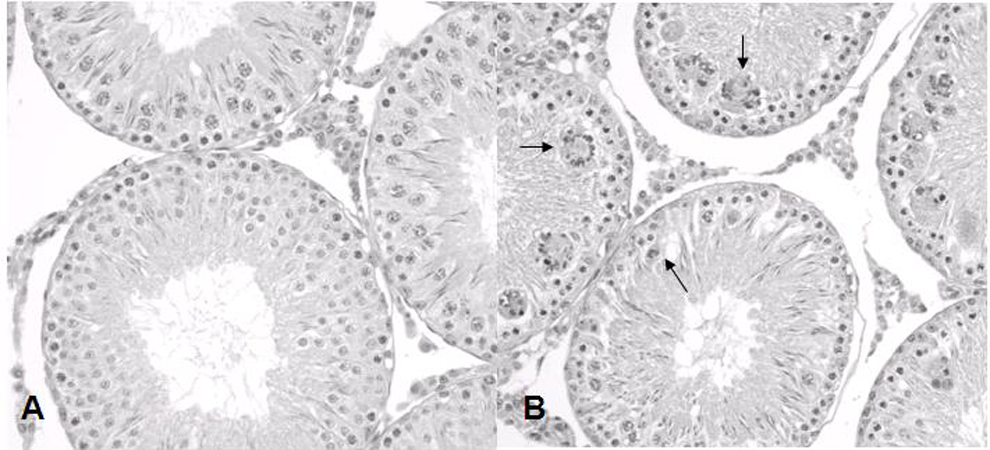
Representative histology of the rat testes in vehicle control (A) and compound
Systemic Exposure Data and Testicular Scoring in the Rat Following 7 Days of Dosing of Compound
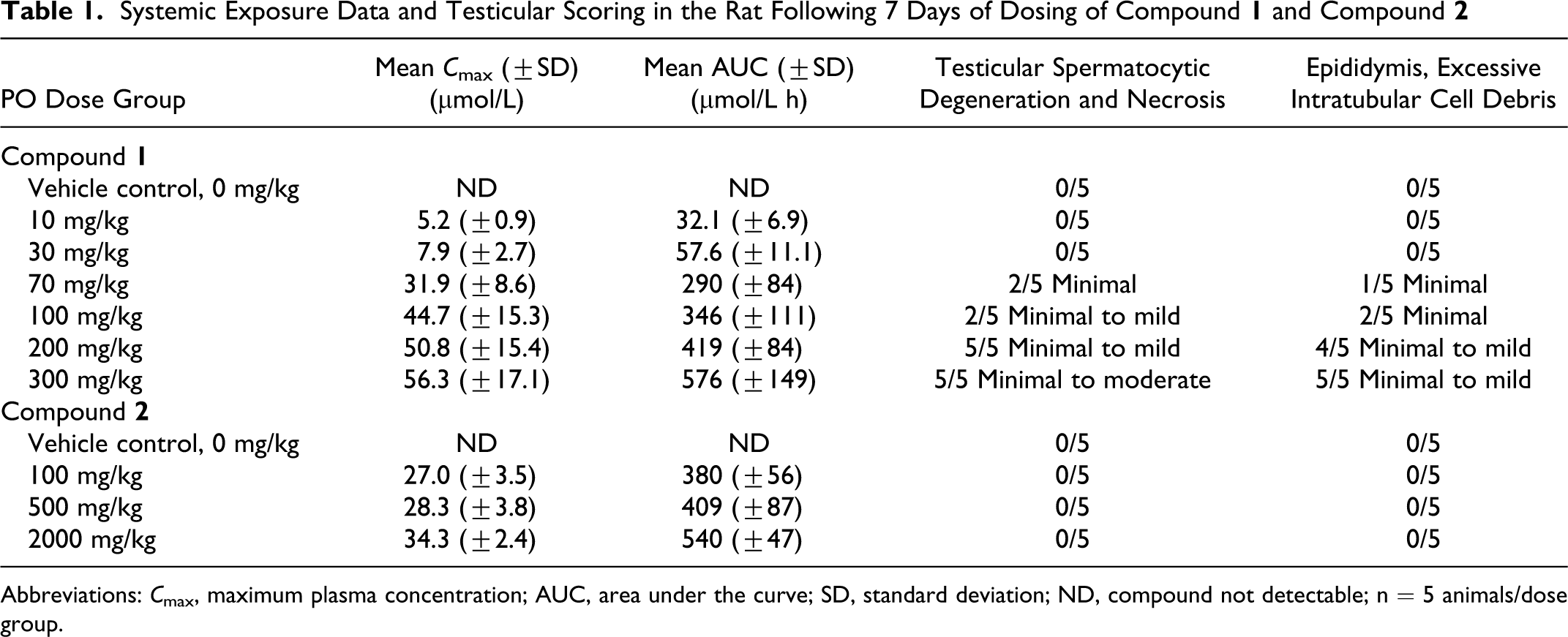
Abbreviations: C max, maximum plasma concentration; AUC, area under the curve; SD, standard deviation; ND, compound not detectable; n = 5 animals/dose group.
In most of the rats with testicular lesions, the ductuli efferentes of the epididymis contained excessive multinucleated giant cells, degenerated cellular debris, and sperm debris (Figure 3). The level of cellular debris was consistent with the severity of the testicular lesion. In some cases, the body of the ductus epididymis had cellular debris but rarely did the tail of the epididymis have degenerative cells and cellular debris. All of the lining epithelial cells and the interstitium remained intact. The degenerated germ cells and cellular debris in the epididymis occurred in the rats dosed at both 200 and 300 mg/kg levels, which correlated to the severity of their testicular lesions.
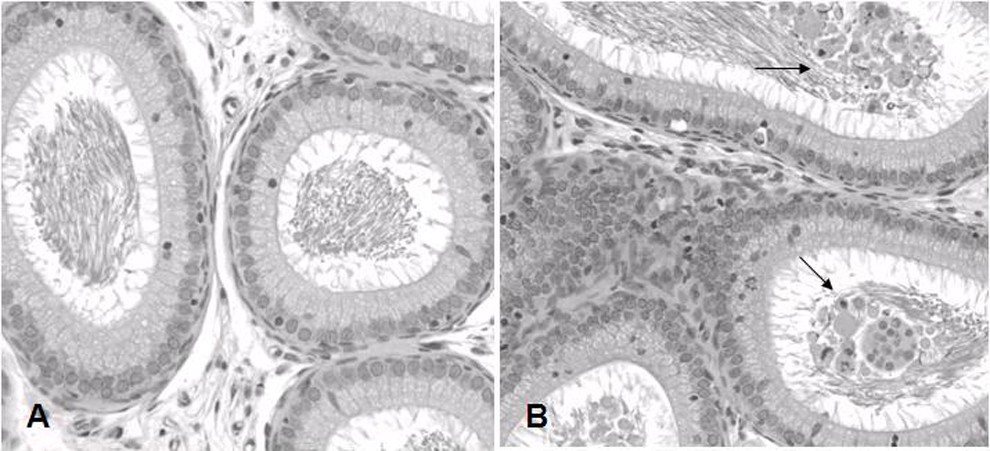
Representative histology of the rat epididymis of vehicle control (A; normal) and compound
There were no significant differences in body weights, hematology, blood chemistry, organ weights, and terminal body weights among the dosed and control groups. The clinical observations and gross pathology had no abnormal findings. Microscopic findings were limited to the testes and epididymis. All other organs evaluated were within normal limits.
Plasma exposures, in terms of maximum concentrations and area under the curve of the parent compound
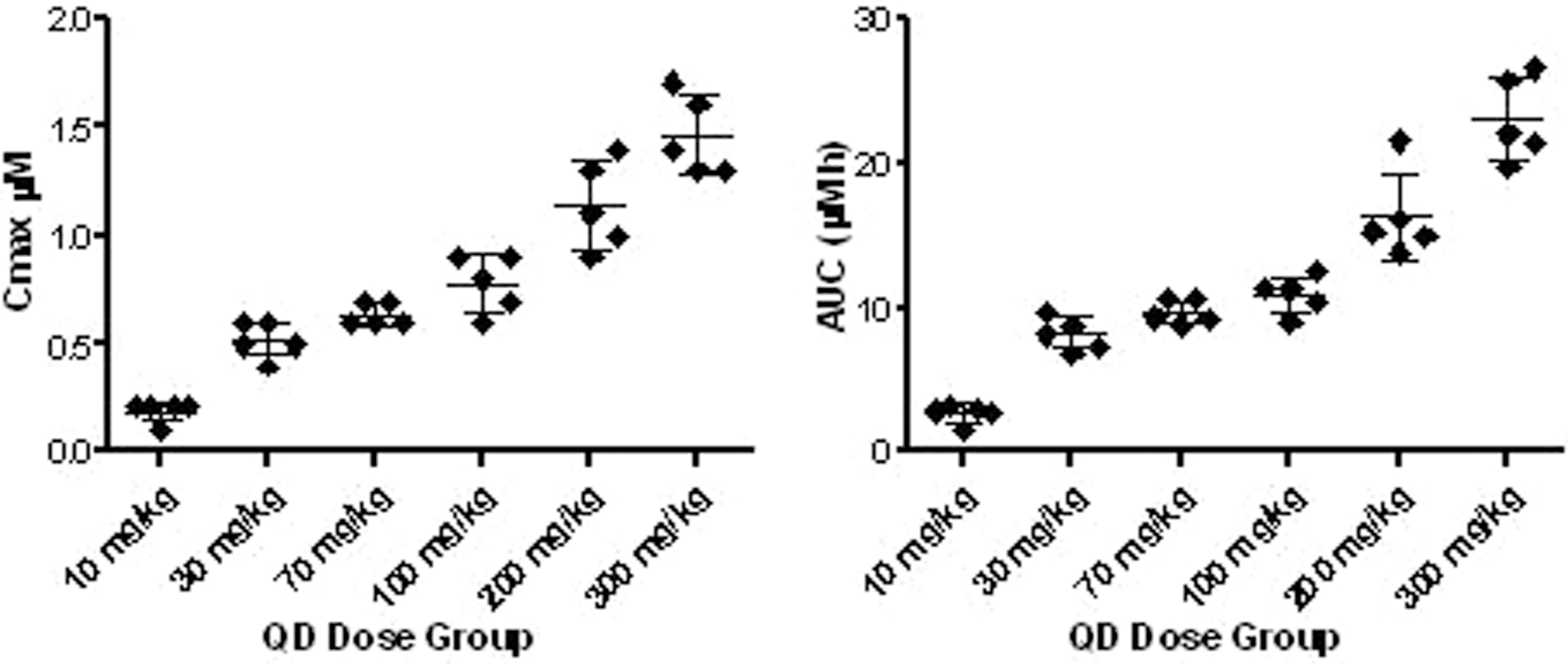
Relative exposure levels in the plasma from individual animals (C
max indicates maximal concentration; AUC, area under the curve 0-24 hours) of trifluoroacetaldehyde based on the assessment of the N-dealkylated fragment of the parent compound
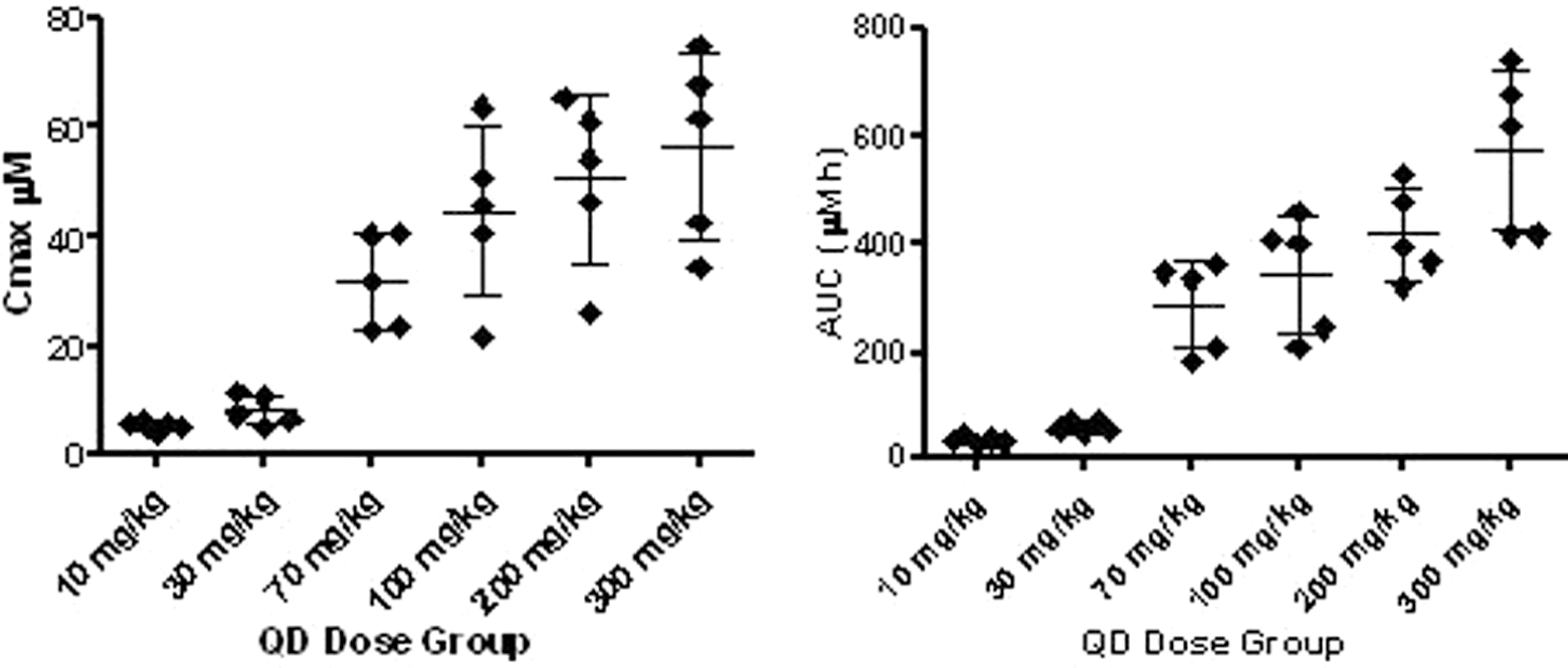
Parent compound
Discussion
Correlation of microscopic findings to parent compound
The toxicokinetic/toxicodynamic correlation of compound
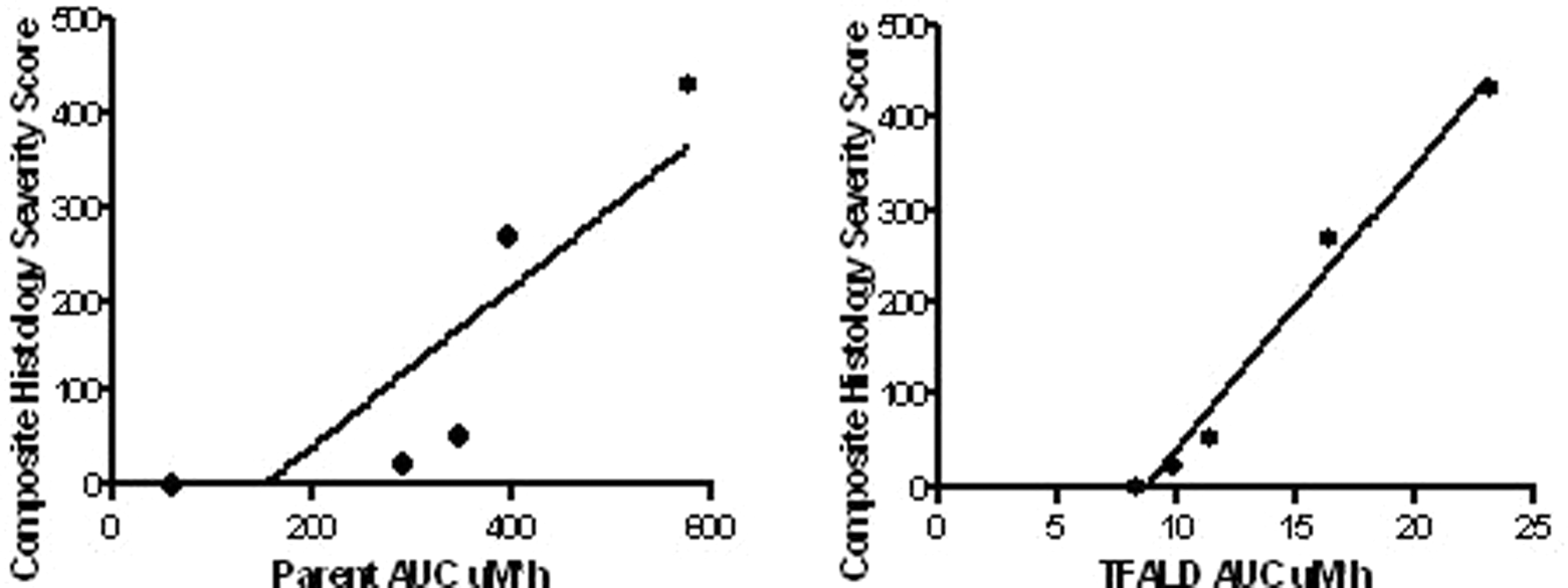
Toxicokinetic/toxicodynamic correlations of the composite histological severity score and area under the curve (AUC) of compound
Considering the literature precedence for the connection between TFALD and testicular lesions, the initial assessment of the toxicology findings of compound
It is apparent that the there are (previously) marketed drugs which contain the N-, O-, or S-trifluorethane fragment as a part of their chemical structure such as halazepam, quazepam, lansoprazole, epitizid, and flecainide. However, only 1 of these compounds has been reported to result in testicular effects in rodents (lansoprazole). Presumably, these findings emphasize that the overall safety profile of a molecule is determined not only by the presence of a structural alert but also with consideration of species differences, dose, duration of use, detoxification of reactive metabolites, and the presence of multiple metabolic pathways.
In summary, this work emphasizes the importance of understanding overall risks as early as is feasible in drug discovery and development, in particular as it relates to the use of trifluoroethane linked to N, O, or S within novel chemical entities. Early metabolic and safety testing can be useful in this regard, as was illustrated by this work. While it is appreciated that a number of considerations go into an overall risk assessment, the authors recommend that the use of this fragment be carefully considered in a risk-benefit paradigm and/or avoided when located in a region of a molecule that may be subject to dealkylation.
Footnotes
Acknowledgments
The authors thank Sheri Cox and Bridget Weber for coordinating the in vivo toxicology studies, and Kimberly Shevlin for managing the necropsy and histology processing.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: supported entirely by Pfizer Global Research & Development, Pfizer, Inc.
