Abstract
There is an increasing concern that chemicals in the environment are contributing to the global rise in the prevalence of type 2 diabetes (T2D). However, there is limited evidence for direct effects of these chemicals on beta cell function. Therefore, the goals of this study were (1) to test the hypothesis that environmental contaminants can directly affect beta cell function and (2) examine mechanistic pathways by which these contaminants could affect beta cell function. Using mouse beta TC-6 cells, we examined the acute effects of 6 substances (benzo[a]pyrene, bisphenol A [BPA], propylparaben, methylparaben, perfluorooctanoic acid, and perfluorooctyl sulfone) on insulin secretion. Only BPA treatment directly affected insulin secretion. Furthermore, chronic exposure to BPA altered the expression of key proteins in the cellular and endoplasmic reticulum stress response. These data suggest that long-term BPA exposure may be detrimental to beta cell function and ultimately be an important contributor to the etiology of T2D.
Keywords
Introduction
Type 2 diabetes (T2D) has become a major public health concern 1 worldwide and is a disease that is rapidly increasing in prevalence in adults, 2 –5 children, and adolescents. 6 Indeed, the total number of people with diabetes worldwide was estimated to be approximately 170 million in 2000, with that number expected to climb to over 300 million by 2030. 5 Furthermore, in 2006, T2D was ranked as the seventh leading cause of death in the United States. 7
Type 2 diabetes develops due to a progressive reduction in the ability of the pancreas to make enough insulin to maintain normal glucose homeostasis and compensate for any underlying resistance to the action of insulin. 8 –11 Major characteristics of T2D include deficient insulin secretion from pancreatic beta cells (ie, reduced beta cell function), impaired insulin-stimulated glucose uptake by the peripheral tissues (ie, insulin resistance), and uncontrolled hepatic glucose production. 8 –11 In the early stages of T2D, the beta cells compensate through hypersecretion of insulin in order to maintain normal blood glucose levels. 10 However, beta cell compensation often fails due to mitochondrial dysfunction 12 and/or endoplasmic reticulum (ER) stress, 13 which in turn leads to beta cell apoptosis. 14 The end result is a reduction in beta cell mass as has been observed in patients with T2D. 8,15,16
Because the beta cell is central to the regulation of glucose homeostasis, the development of T2D is influenced by factors that initiate, facilitate, and accelerate beta cell loss and dysfunction. In 2001, Longnecker and Daniels put forward the hypothesis that chemical contaminants in the environment may have a role in the etiology of T2D. 17 Support for this hypothesis comes from epidemiological studies which have demonstrated that populations with high exposure to arsenic, persistent organochlorine pollutants (including dioxins, polychlorinated biphenyls [PCBs], and organochlorine pesticides), phthalate esters, and air pollution have an increased incidence of insulin resistance and/or T2D. 18 –26 However, although these studies suggest that chemicals in the environment are associated with T2D, support for a causal relationship between exposure to environmental contaminants and T2D in human populations is limited. Therefore, the goals of this study were (1) to test the hypothesis that environmental contaminants can directly affect beta cell function, thus providing a plausible mechanism by which environmental contaminant exposure can lead to the development of T2D and (2) examine mechanistic pathways by which these contaminants could affect beta cell function.
Materials and Methods
Cell Culture
Beta TC-6 cells, a mouse beta cell line, were purchased from ATCC (Manassas, Virginia). Cells (passages 1-13) were cultured at 37°C, 5% CO2, and 95% humidity in Dulbecco Modified Eagle Medium (DMEM) supplemented with 15% fetal bovine serum ([FBS] HyClone, Logan, Utah).
Chemical Screen
The effects of 6 common environmental contaminants on insulin release were examined. These were benzo[a]pyrene (BaP), a representative polyaromatic hydrocarbon; bisphenol A (BPA), an ubiquitous plastic monomer and component of thermal paper; propylparaben and methylparaben, components in various personal care products; perfluorooctanoic acid (PFOA), an antiwetting agent; and perfluorooctyl sulfone (PFOS), a stain-resistant additive. The chemicals for the screen were selected because there is widespread human exposure 27 –29 and/or evidence of an association between exposure and an increased risk of T2D in human populations. 30,31 All substances were purchased from Sigma Chemical Co (St Louis, Missouri) and had a purity of 95% or higher. The effects of these compounds were examined under both low (3.3 mmol/L) and high (16.7) glucose conditions to determine whether the glucose environment affected the cellular response.
Cells were seeded in 6-well plates (BD Biosciences, Mississauga, Ontario) at a density of 250 000 cells per well. Forty-eight hours after seeding, the cells were washed twice with glucose-free Krebs-Ringer buffer ([KRBH] 135 mmol/L NaCl, 3.6 mmol/L KCl, 5 mmol/L NaHCO3, 0.5 mmol/L NaH2PO4·2H2O, 0.5 mmol/L MgCl·6H2O 1.5 mmol/L CaCl2·2H2O, 10 mmol/L Hepes, and 0.1% bovine serum albumin [BSA], pH 7.4 [assay buffer]). Cells were then incubated for 2 hours in 1 mL/well of glucose-free KRBH and then for 30 minutes in 1 mL/well of KRBH buffer with 2.8 mmol/L glucose. The KRBH buffer was then removed and replaced with 1 mL/well of assay buffer containing vehicle (ethanol, 0.1%) or one of the test compounds at increasing log concentrations (0.1-1000 ng/mL) in a KRBH containing either low (3.3 mmol/L) or high (16.7 mmol/L) glucose concentrations for 1 hour. All test chemicals were evaluated in duplicate within an experiment (N = 5 independent experiments per compound). Following the 1-hour incubation, media was collected, centrifuged (5000 rpm for 2 minutes), and the supernatant was collected and stored at –20°C until analysis. Insulin secretion was measured using a radioimmunoassay (RIA), according to the manufacturer’s instructions (Millipore, St Charles, Missouri). Total protein content was determined using a bicinchoninic acid (BCA) protein assay kit according to the manufacturer’s instructions (ThermoScientific, Rockford, Illinois). Insulin release (ng/mL) was normalized to the protein content (mg/mL) and expressed as ng insulin per mg protein.
Because BPA was the only chemical from our initial screen that was found to affect insulin secretion from beta TC-6 cells, the following experiments focused on more fully examining the effects of BPA on beta cell function.
Bisphenol A effects on beta cell function
The effects of BPA on beta cell function were evaluated using 100 ng/mL BPA, as this was the lowest concentration at which we observed an effect on insulin secretion (Figure 1). Because BPA stimulated insulin secretion under both low- (ie, 3.3 mmol/L) and high- (ie, 16.7 mmol/L) glucose conditions, investigations of the mechanism of BPA’s action were also conducted under both glucose conditions.
Mitochondrial Function
To determine whether BPA can directly affect mitochondrial function in pancreatic beta cells, beta TC-6 cells were treated with 100 ng/mL BPA or vehicle in glucose-free DMEM (Invitrogen, Burlington, Ontario) supplemented with 15% FBS and either low- (3.3 mmol/L) or high- (16.7 mmol/L) glucose for 1, 24, or 48 hours. Treatments were done in triplicate in a minimum of 4 independent experiments. Following the incubation period, cells were washed in Dulbeccos-modified PBS, removed from the plate using cell scrapers (BD Bioscience), pelleted by centrifugation (1000g for 10 minutes at 4°C), and resuspended in homogenization buffer (5 mmol/L HEPES pH 7.4, 100 mmol/L KCl, 70 mmol/L sucrose, 220 mmol/L mannitol, and 1 mmol/L EGTA) supplemented with a protease inhibitor (Roche Applied Science, Laval, Quebec, Canada) and fatty acid-free BSA (2 mg/mL). The cell suspension was sonicated (Microsonix 200) at a low-power setting (7 Hz) for three 10-second bursts. The cell lysate was clarified by centrifugation (16 000g for 20 minutes at 4°C) and the pellet was resuspended in BSA-free Hepes buffer. The mitochondrial suspension was flash frozen in liquid nitrogen and stored at −80°C until analysis. Citrate synthase activity, an indicator of total mitochondrial mass, 32 was measured using the thiol reagent 5,5'-dithio-bis-(2-nitrobenzoic acid [DTNB], Sigma Chemical Co). Complex IV (cytochrome c oxidase) activity was assessed by measuring the rate of cytochrome c (from equine heart; Sigma Chemical Co) oxidation. Both activity assays were performed using UV-spectrophotometry (Varian Inc, California) as previously described. 33,34 Data are expressed as the mean enzyme activity relative to the protein content in the sample.
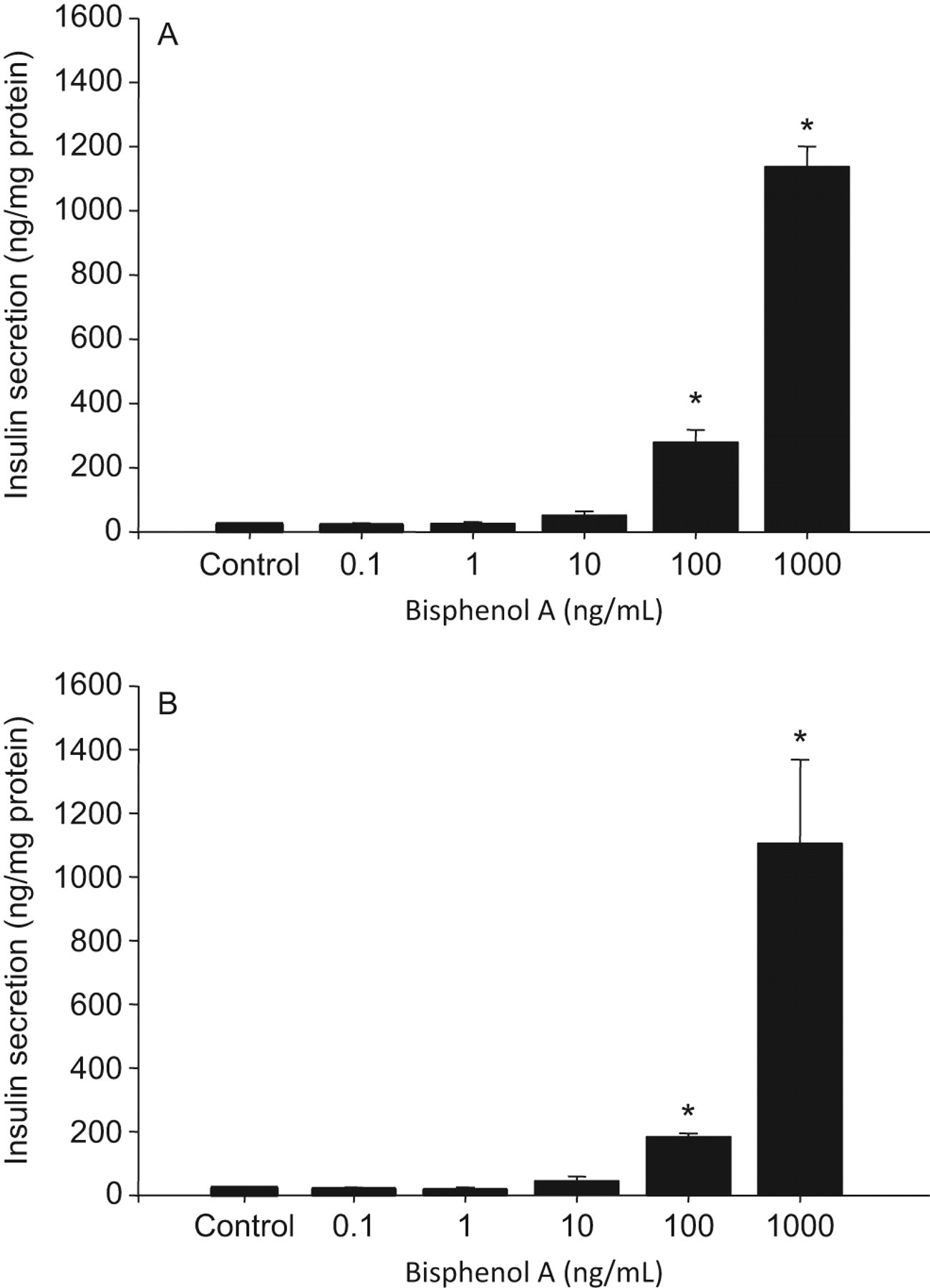
Effect of bisphenol A (BPA) treatment on insulin secretion by beta TC-6 cells under (A) low- (3.3 mmol/L) and (B) high- (16.7 mmol/L) glucose conditions. Data are presented as mean ± SEM. Values marked with an asterisk (*) are significantly different (P < .05) from the vehicle controls.
Cellular and ER Stress Markers: Western Blotting
Protein expression of markers of cellular and ER stress, Hsp70, glucose-regulated protein 78 (GRP 78), glucose-regulated protein 94 (GRP94), and CHOP/GADD153 were measured in cells treated with BPA (100 ng/mL) for 1, 24, and 48 hours under both low- and high-glucose conditions (N = 3 independent experiments per group). In all, 30 μg of total protein was separated on an 8% polyacrylamide gel and transferred to polyvinylidene fluoride (PVDF) membrane (BioRad Laboratories, Hercules, California). Membranes were blocked overnight with 5% (wt/vol) skim milk in Tris-buffered saline Tween 20 (TBST) at 4°C and then incubated for 1 hour at room temperature with the following primary antibodies: rabbit polyclonal Hsp70 (1:2000; 72 kDa; Cell Signaling Technology, Danvers, Massachusetts); rabbit polyclonal CHOP/GADD 153 (1:250; 19 kDa; Santa Cruz Biotechnology, Inc, Santa Cruz, California); rabbit polyclonal beta actin (1:8000, 43 kDa, AbCam, Cambridge, Massachusetts), and rabbit polyclonal KDEL (1:4000; AbCam). The KDEL antibody recognizes proteins containing the ER retention sequence SEKDEL including GRP 78 and GRP94. Membranes were cut horizontally and one half was probed for Hsp70, KDEL, and CHOP/GADD153, and the other half of each blot for beta actin as a loading control. After washing, blots were then incubated in peroxidase-conjugated secondary anti-rabbit antibody (1:2000; SantaCruz Biotechnology). Reactive protein was detected with ECL Plus chemiluminescence (GE Healthcare Canada Ltd, Baie d’Urfe, Quebec, Canada) and Amersham Hyperfilm ECL (GE Healthcare Canada Ltd). Densitometric analysis of immunoblots was performed using ImageJ 1.37v 160 software; all proteins were normalized to the beta actin loading control. For blots using KDEL, both bands from the sample (78 and 94 kDa) were quantified relative to the loading control from that lane. All samples (ie, low-glucose control, low-glucose BPA, high-glucose control, and high-glucose BPA) from a single time point (ie, 1, 24, or 48 hours) were analyzed on the same blot.
Statistical Analysis
All statistical analyses were performed using SigmaStat (v.3.5, SYSTAT, Chicago, Illinois). Data were tested for normality using the Kolmogorov-Smirnov test as well as equal variance using Spearman rank correlations. Where normality or variance tests failed, data were analyzed using Kruskal-Wallis analysis of variance (ANOVA) on ranks.
Chemical screen
Insulin secretion data (ng insulin/mg protein) for each test compound was analyzed by 2-way ANOVA (α = .05) with glucose level and dose of the test compound as the main variables. When there was a significant main effect of treatment, data within each glucose level were reanalyzed by 1-way ANOVA (α = .05) with appropriate post hoc tests to determine dose-related effects of treatment
Bisphenol A effects on beta cell function
The effect of BPA on mitochondrial electron transport chain (ETC) activity at each time point was analyzed by 2-way ANOVA (a α = .05) with glucose level and treatment as the main variables. When a significant (α = .05) interaction effect was identified, data were compared by appropriate post hoc tests
For the measures of cellular stress (ie, Hsp70) and ER stress (ie, CHOP/GADD153, GRP78, and GRP94), protein expression was analyzed by 2-way ANOVA (α = .05) with glucose level and treatment as the main variables. When a significant (α= .05) interaction effect was identified, data were compared by appropriate post hoc tests.
Results
Chemical Screen
There was no main effect of BaP, methylparaben, propylparaben, PFOA, or PFOS to affect insulin secretion and no interaction effect between the treatment with these test compounds and glucose level to alter insulin secretion (all Ps > .05; data not shown). There was a main effect of BPA treatment to increase insulin secretion (P < .001); however, this response was not affected by the glucose level (interaction effect; P = .901). Under both low- and high-glucose conditions, BPA at concentrations of 100 and 1000 ng/mL significantly stimulated insulin secretion (Figure 1).
Mitochondrial Function
There was no effect of BPA treatment on either complex IV or citrate synthase activity under any condition tested (data not shown).
Cellular Stress
Bisphenol A treatment significantly (P = .024) increased the expression of Hsp70 in beta cells; an effect that was only observed after 24 hours of treatment (Figure 2). There was no interaction between BPA treatment and glucose level at any time examined.
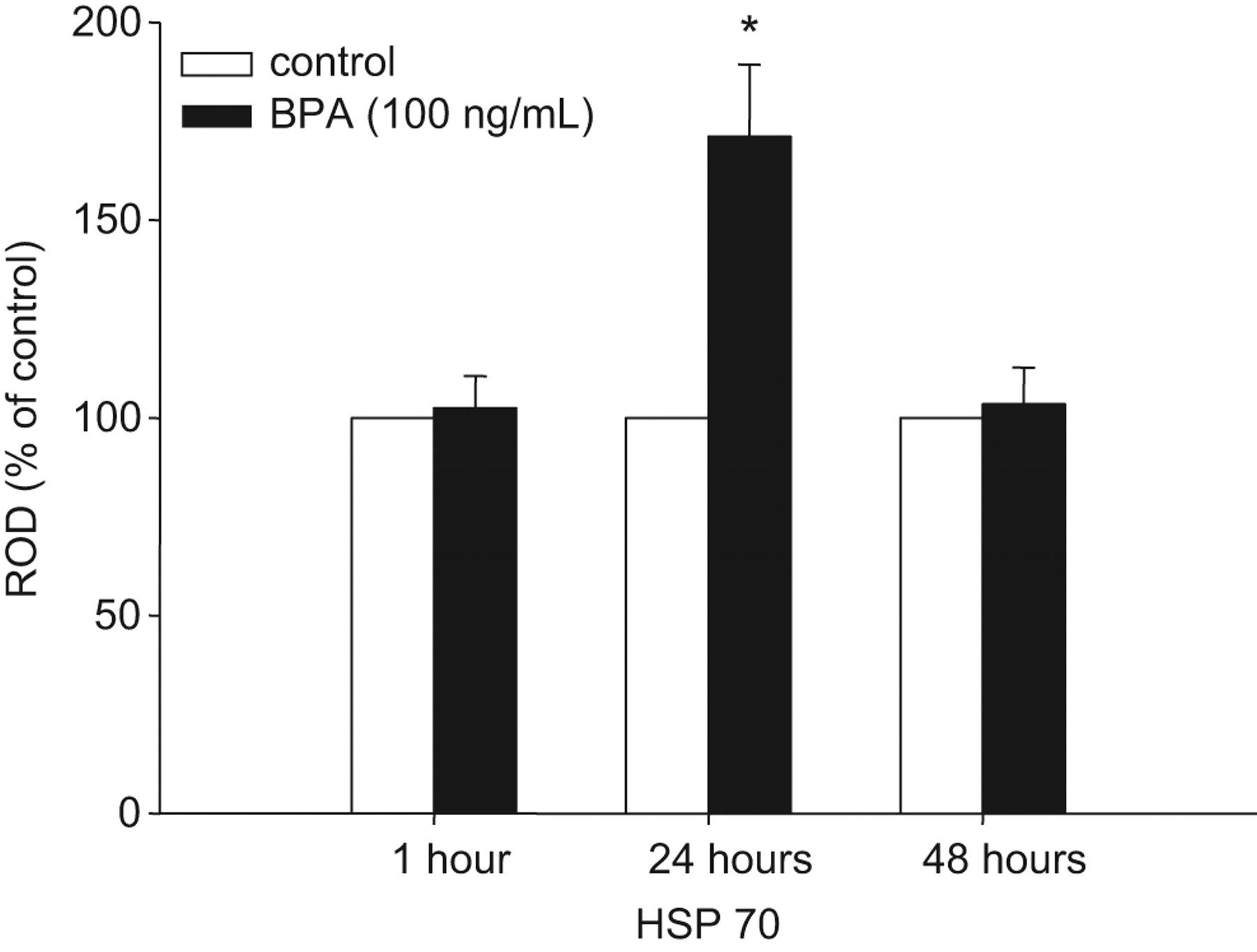
Main effect of bisphenol A (BPA) treatment on the expression of HSP70 in beta TC-6 cells. Least square mean optical densities (relative to beta actin; ROD) are presented as percentage of vehicle control ± SEM at each time point. There was a significant main effect (*P < .05) of BPA treatment to increase HSP70 expression relative to the vehicle controls at 24 hours.
Endoplasmic Reticulum Stress
Bisphenol A treatment significantly reduced the expression of both GRP78 (Figure 3) and GRP94 (Figure 4). The expression of GRP78 was significantly reduced by BPA treatment after 24 hours. There was a significant interaction between BPA treatment and glucose level (interaction effect P = .010) such that the effect of BPA to reduce GRP78 expression was only significant under low-glucose conditions. Bisphenol A treatment significantly reduced GRP94 expression after 48 hours of treatment; however, there was no interaction between BPA treatment and glucose level on this outcome (interaction effect, P = .319). There was no effect of BPA on the expression of CHOP/GADD153 at any time examined (all Ps > .05, data not shown).
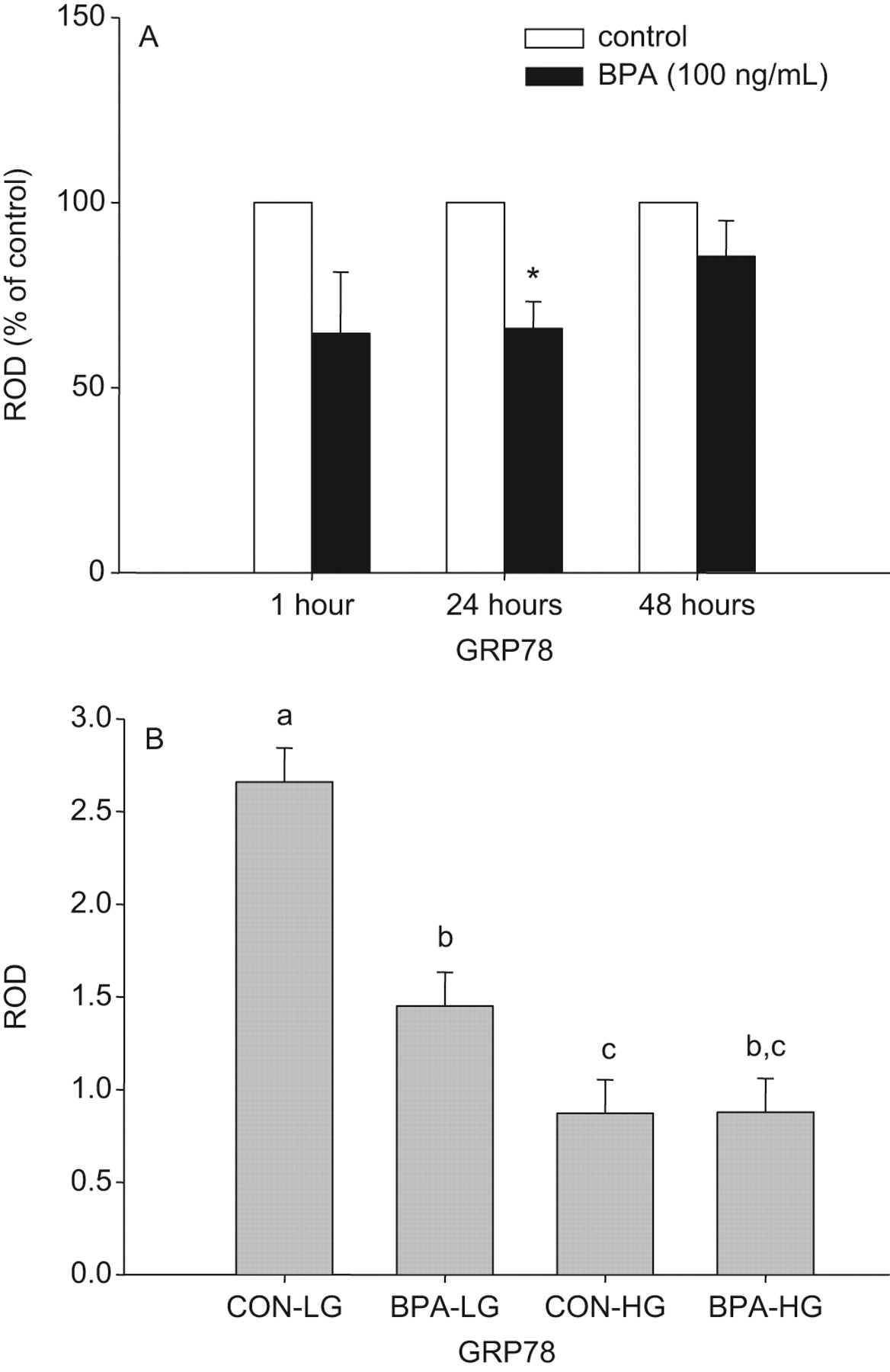
Effect of bisphenol A (BPA) treatment on the expression of GRP78 in beta TC-6 cells. Panel A, Least square mean optical densities (relative to beta actin; ROD) for the main effect of BPA treatment are presented as percentage of vehicle control ± SE at each time point. There was a significant main effect (*P < .05) of BPA treatment to decrease GRP78 expression relative to the vehicle controls at 24 hours. Panel B, Least square means of optical densities (relative to beta actin; ROD) for glucose × treatment interaction (CON indicates vehicle control; BPA, 100 ng/mL BPA; LG, 3.3 mmol/L low glucose, and HG = 16.7 mmol/L high glucose) after 24 hours of BPA treatment. Data are presented as mean ± SE. Values with different superscript letters are significantly (P < .05) different from each other.
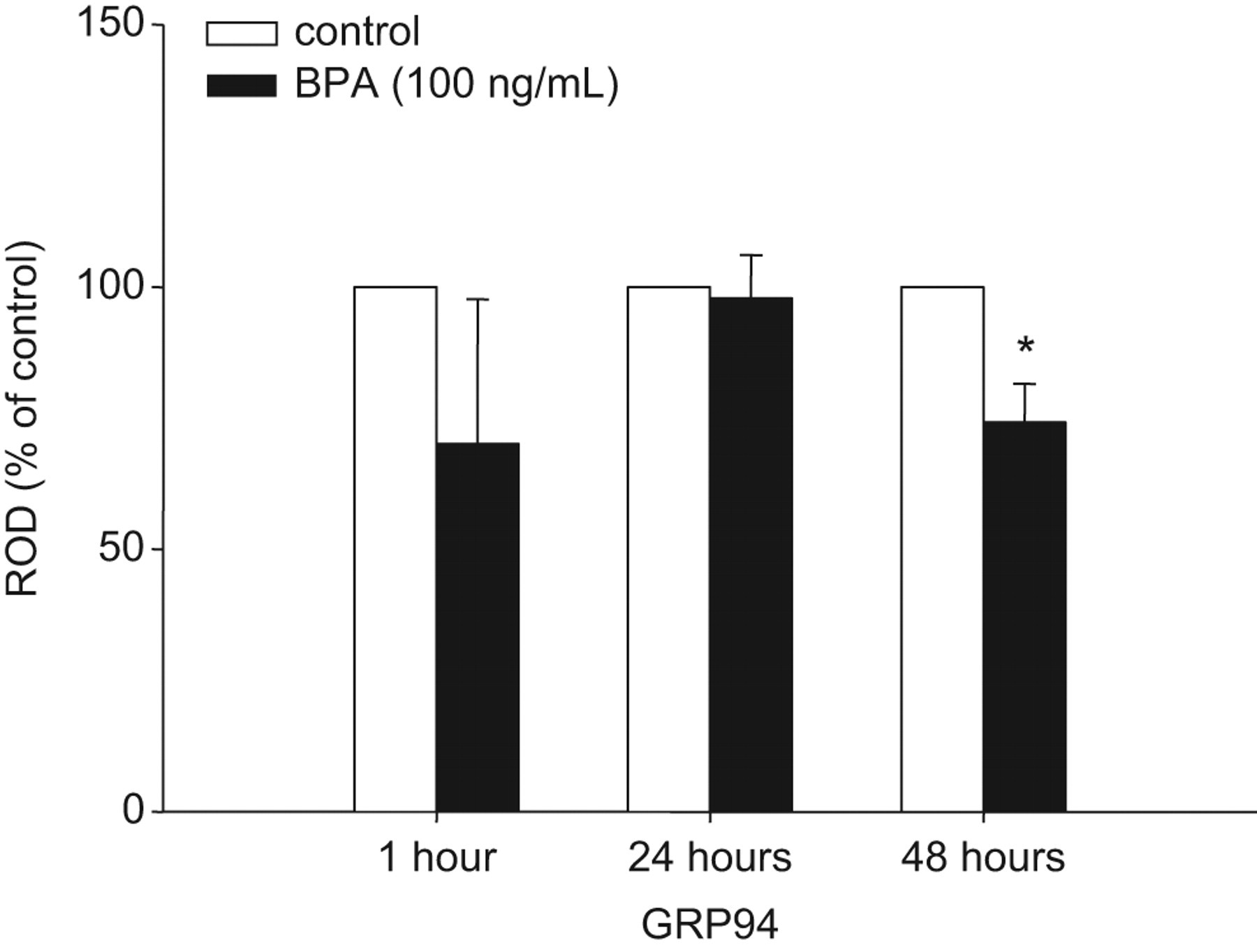
Main effect of bisphenol A (BPA) treatment on the expression of GRP94 in beta TC-6 cells. Least square mean optical densities (relative to beta actin; ROD) are presented as percentage of vehicle control ± SEM at each time point. There was a significant main effect (*P < .05) of BPA treatment to decrease GRP94 expression relative to the vehicle controls at 48 hours.
Discussion
Type 2 diabetes mellitus is rapidly increasing in prevalence worldwide. 2 –6 This increase in T2D globally has been linked, in part, to lifestyle changes associated with industrialization, rapid economic development, and modernization. Indeed, the prevalence of T2D is rising most rapidly in “developing” countries as they adopt more Westernized lifestyles and diets. 35,36 However, increased industrialization also coincides with a dramatic increase in exposure to synthetic chemicals associated with consumer goods and industrial and agricultural practices. 37 Accumulating evidence that exposure to ubiquitous pollutants may be contributing to the increasing incidence of T2D in the developed world 18 -26 has led us to investigate the hypothesis that some ubiquitous contaminants induce diabetes by altering insulin release from pancreatic beta cells. We screened 6 chemicals for their ability to interfere with normal beta cell function (ie, insulin secretion) using mouse pancreatic beta TC-6 cells. Of the 6 chemicals screened, only BPA significantly affected beta cell function.
Benzo[a]pyrene
Numerous studies have suggested an association between air pollution and T2D. 26,38 Benzo[a]pyrene is a major component of air pollution 39 and has been shown to cause oxidative damage to cellular lipids, proteins, and DNA 40 –42 and alter ER function in a hepatoma cell line. 43 Since oxidative and ER stress can negatively affect the maintenance of beta cell mass and beta cell function, 13,44 –46 we predicted that BaP treatment would directly impair insulin secretion from pancreatic beta cells. However, in this model system, there was no effect of BaP on insulin secretion, suggesting that if BaP exposure leads to T2D, it may be via an alternative mechanism and not directly via effects on beta cell function.
Perfluoroalkyl Compounds
Perfluorinated compounds (PFCs) are widely used as surfactants, paper, and textile coatings and in food packaging. Two PFCs, PFOS and PFOA are consistently detected in serum from children and adults 29 as well as cord blood and breast milk samples. 47 It has been reported that there is an association between serum perfluoroalkyl chemicals (PFCs), glucose homeostasis, and metabolic syndrome in adolescents and adults. 31 However, other human and animal studies have failed to find consistent associations between exposure to polyfluoroalkyl chemicals and measures of impaired glucose homeostasis (ie, insulin resistance and hyperglycemia). 48,49 In this study, neither PFOS nor PFOA significantly affected insulin secretion from beta TC-6 cells. Many of the effects of PFOS and PFOA are mediated via the peroxisome proliferator-activated receptors (PPARs) PPARα, PPARβ/δ and PPARγ. 50 –52 Although beta TC-6 cells do express all of these PPARs, their expression is relatively low, 53 which may explain the lack of effect of PFOS and PFOA on insulin secretion in this study. Moreover, as PPARs are nuclear receptors which exert their effects as ligand-activated transcription factors by altering the expression of target genes, 54 the cellular responses to PPAR agonists/antagonists may require a longer period of preincubation than was tested in the current study for their full effects to be expressed. Our results indicate that very short-term exposure to PFOS and PFOA did not influence the release of insulin from beta TC-6 cells.
Parabens
Parabens are widely used as antimicrobial preservatives in cosmetics, pharmaceutical, and food products and are widely detected in humans. Indeed, results from NHANES (2005-2006) reported that methylparaben and propylparaben were detected in 99.1% and 92.7% of the samples tested, respectively. 55 To date, there are no epidemiological studies showing a link between parabens and diabetes. However, parabens have been reported to cause impaired mitochondrial function and mitochondrial-mediated cell death 56 ; pathways that are known to be important for beta cell function. 12 Furthermore, parabens may affect insulin secretion and beta cell function via their interactions with estrogen receptors. 56,57 However, results from this study failed to find an effect of parabens on insulin secretion from beta TC-6 cells. These results are consistent with those of Boberg et al 58 who reported that maternal exposure to parabens did not affect fetal insulin levels, suggesting that there was no beta cell defect as a result of this exposure.
Bisphenol A
The use of BPA, a component of polycarbonate plastic, in a large variety of consumer products has resulted in widespread human exposure. 59 Human epidemiological data suggest an association between urinary BPA concentrations with diabetes in the adult US population. 30 However, whether this association is causal remains highly controversial. Results from this study have shown that acute BPA exposure results in a profound dose-dependent stimulation of insulin secretion from isolated pancreatic beta cells (Figure 1); an effect which is consistent with the majority of reports in the literature, in which BPA treatment in vivo results in hyperinsulinemia. 60 In isolated beta TC-6 cells, BPA-stimulated insulin secretion is evident after only 1 hour of exposure, in contrast to isolated rat islets where a longer (ie, 24 hours) exposure was required to observe BPA-stimulated insulin secretion. 61
Secretion of insulin from the beta cell requires folding, export, and processing of newly synthesized insulin by the ER and the production of energy, in the form of adenosine triphosphate (ATP), by the mitochondria. 13,62 Dysregulation of the normal function of the ER and the mitochondrial electron transport chain have both been shown to be associated with impaired beta cell function and increased beta cell death. 13,45,46,62 Since BPA treatment has been shown to cause ER stress and mitochondrial dysfunction in other tissues, 63 –65 we hypothesized that exposure of pancreatic beta cells to BPA would also lead to mitochondrial dysfunction and/or ER stress.
In our study, treatment of beta TC-6 cells with BPA for up to 48 hours did not have any effect on mitochondrial ETC activity but did affect components of the cellular and ER stress response. The lack of effect of BPA on mitochondrial function was surprising, given that BPA has previously been shown to increase reactive oxygen species production in other cells 63,64 and mitochondrial electron transport chain enzyme activity is profoundly impaired by oxidative stress. 46 However, a recent review by Kumar et al 66 has highlighted the fact that the effects of estrogenic compounds on redox balance (ie, whether a compound has pro-oxidant or anti-oxidant effects) are largely dependent on the cell type and estrogen receptors involved in the cellular response of interest. In pancreatic beta cells, estrogen-stimulated insulin secretion appears to be mediated via the nonclassical G-protein-coupled receptor GPR30, 67 whereas estrogen-induced increases in insulin content are mediated via the estrogen receptor ERα 68 Bisphenol A has been shown to initiate signaling via GPR30 in breast cancer cells, 69 and in beta cells the effect of BPA to increase insulin content is via the ERα 68 Importantly, activation of both ERα and GPR30 have been shown to protect against oxidative stress 70,71 ; observations that are consistent with a lack of evidence for BPA-induced oxidative stress in this study. However, the effects of BPA on redox balance in the beta cell remain to be more fully explored.
At the highest concentration of BPA (ie, 1000 ng/mL), insulin secretion was approximately 50-fold higher in the treated cells versus controls. Chronic overstimulation of secretory cells often means that proteins destined for secretion enter the ER faster than they can be folded, which leads to cellular stress and more specifically ER stress. 13 In the presence of cellular and/or ER stress, the expression of chaperone proteins, including HSP70, GRP78, and GRP94, is upregulated to alleviate cellular and ER stress, maintain ER function (ie, normal protein folding), and protect the cell from apoptosis. 72,73 We hypothesized that the hypersecretion of insulin in response to BPA would lead to both cellular and ER stress and the induction of chaperone proteins. After 24 hours of treatment, cells treated with BPA, at a dose that caused profound stimulation of insulin secretion, had significantly higher levels of HSP70. This finding is consistent with an in vivo study that reported increased pancreatic HSP70 expression in the presence of hyperinsulinemia. 74 However, contrary to our hypothesis, in beta TC-6 cells, BPA treatment significantly reduced the expression of the ER chaperone proteins GRP78 and GRP94 at 24 and 48 hours, respectively. Under conditions of ER stress, the induction of GRP78 and GRP94 promotes increased protein-folding activity, decreased protein-aggregation, and thus the prevention of further cell injury and death. 75 Indeed, reduced expression of both GRP94 and GRP78 is associated with increased apoptosis in pancreatic cells. 76 –78 Furthermore, in the NIT-1 insulinoma cell line, downregulation of GRP78 was associated with both increased expression of CHOP/GAD153, the proapoptotic transcription factor induced by ER stress, and apoptosis. 78 Although we did not observe an upregulation of CHOP/GADD153 in association with the reduction in GRP78 and GRP94, we cannot rule out the possibility that longer exposure to BPA would lead to ER stress-mediated beta cell death, especially since the reduction in GRP94 is not evident until after 48 hours of treatment.
The effect of BPA to stimulate insulin secretion in this study is observable under both hypoglycemic and hyperglycemic conditions, suggesting that in the general population, the effects of BPA to stimulate insulin secretion will be robust. The minimum concentration of BPA which showed an effect on insulin secretion was 100 ng/mL. This concentration is higher than mean serum and urine concentrations reported in most biomonitoring studies but within the reported ranges for some populations. 59
Although 5 of the 6 chemicals tested did not have any impact on beta cell function as measured by insulin secretion, we do provide proof of principle that in some cases epidemiological associations between exposure to ubiquitous environmental contaminants and the risk of T2D may involve direct effects of these chemicals on beta cell function. Specifically, we have demonstrated that BPA directly affects beta cell function and that in vitro exposure to BPA results in ER stress in the pancreatic beta cell; a pathway that is thought to be an important contributor to the etiology of T2D. 13 Therefore, exposure to BPA in human populations may in fact be causally related to the development of T2D and warrants careful investigation.
Footnotes
Acknowledgment
We thank Bernice Tsoi and Jillian Hyslop for their help with cell culture work.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a Natural Sciences and Engineering Research Council Individual Discovery grant to ACH.
