Abstract
AVI-4658 is a phosphorodiamidate morpholino oligomer (PMO) drug designed to restore dystrophin expression in a subset of patients with Duchenne muscular dystrophy (DMD). Previous reports demonstrated this clinical proof-of-principle in patients with DMD following intramuscular injection of AVI-4658. This preclinical study evaluated the toxicity and toxicokinetic profile of AVI-4658 when administered either intravenously (IV) or subcutaneously (SC) to cynomolgus monkeys once weekly over 12 weeks, at doses up to the maximum feasible dose of 320 mg/kg per injection. No drug-related effects were noted on survival, clinical observations, body weight, food consumption, opthalmoscopic or electrocardiographic evaluations, hematology, clinical chemistry, urinalysis, organ weights, and macroscopic evaluations. Drug-related microscopic renal effects were dose-dependent, apparently reversible, and included basophilic granules (minimal), basophilic tubules (minimal to moderate), and tubular vacuolation (minimal to mild). These data establish the tolerability of AVI-4658 at doses up to and including the maximum feasible dose of 320 mg/kg by IV bolus or SC injection.
Keywords
Introduction
AVI-4658 is a member of the phosphorodiamidate morpholino oligomer (PMO) drug class. Phosphorodiamidate morpholino oligomers have an oligomeric structure that generally follows that of DNA and RNA, but with several key chemical modifications designed to improve their drug-like character. Stability against a large number of nucleases and proteases 1 is imparted by the replacement of the ribose or deoxyribose moiety with a morpholine residue, and substitution of the natural negatively charged phosphodiester backbone linkage by a phosphorodiamidate linkage, which is uncharged. AVI-4658 is a 30-mer with the base sequence: CTC CAA CAT CAA GGA AGA TGG CAT TTC TAG (Figure 1 ), which binds dystrophin pre-mRNA at exon 51 by Watson-Crick base pairing.
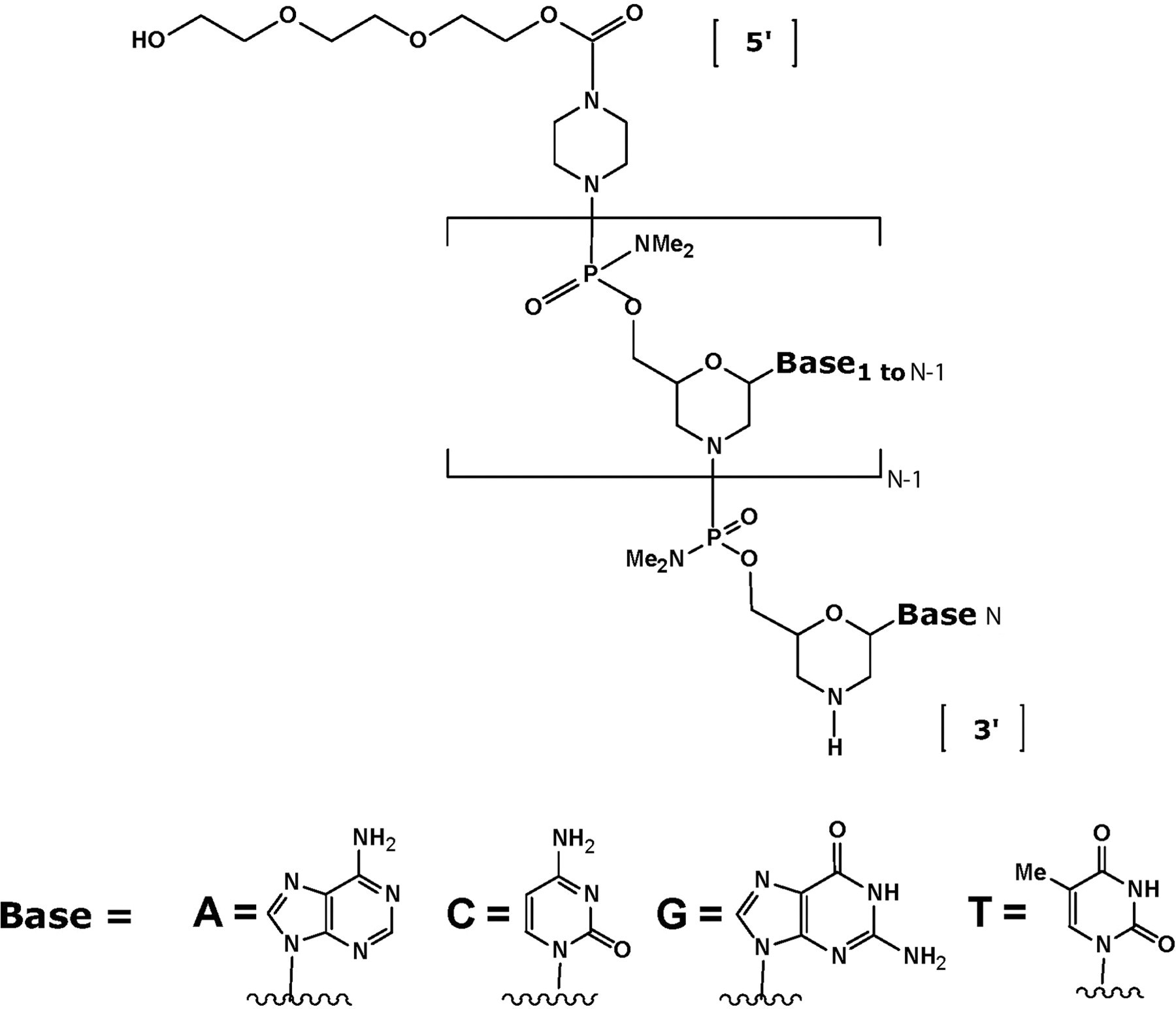
Chemical structure of the phosphorodiamidate morpholino oligomer (PMO), AVI-4658.
The lack of negative charge on PMO drugs is in contrast to most antisense oligonucleotides, the majority of which are based on the negatively charged phosphorothioate backbone. 2 The phosphorothioate moiety in those compounds is thought to improve their stability, serum protein binding, and pharmacokinetics; however, dose-limiting toxicities were identified in preclinical and clinical evaluations. 3 Iversen and colleagues describe mortality in nonhuman primates following doses of a first generation phosphorothioate oligonucleotides at 10 mg/kg administered as an IV bolus. 4 This effect has been mitigated largely through the use of administration routes that blunt the serum Cmax, such as IV infusions or subcutaneous (SC) dosing. Other, less effects such as lethargy, central hypotension, reduced cardiac output, and complement activation 5 may also be reduced in this way. Complement activation, along with hepatotoxicity, are considered to be a class effect of oligonucleotides that contain the phosphorothioate moiety. 6,7
AVI-4658 is a splice switching oligomer (SSO) designed to induce skipping of exon 51 of human dystrophin. Thus, it has the capability in patients with dystrophin deletions of exons 50, 52, 52-63, 45-50, 48-50, or 49-50, which disrupt the protein production, to restore the translation reading frame and enable the synthesis of an internally truncated but almost fully functional dystrophin protein. Proteins of similar composition are found naturally in patients with a much milder form of the disease called Becker muscular dystrophy (BMD), in which the mutation reduces but does not completely abrogate the overall level and function of dystrophin.
The ability of AVI-4658 to restore dystrophin expression in the muscle cells of patients was first demonstrated in cell culture. 8 Genotoxicity and safety pharmacology testing of AVI-4658 showed no mutagenic potential, and no cardiovascular, pulmonary, or neurological adverse effects in cynomolgus monkeys at doses up to the maximum feasible dose of 320 mg/kg by IV infusion or SC. 9 These data were sufficient to initiate and complete a “proof of concept” clinical trial of AVI-4658 in the United Kingdom (UK), in which intramuscular injection in patients with DMD was evaluated for safety and efficacy. 10 Doses of 0.9 mg of AVI-4658 injected locally (intramuscular) into the extensor digitorum brevis (EDB) resulted in increased dystrophin production, as measured by a 44% to 79% increase in dystrophin expression in myofibers, which was localized to the sarcolemma. In contrast, the contralateral EDB muscle, which received vehicle-only, showed no evidence of dystrophin restoration, confirming that AVI-4658 was functioning by the expected mechanism of action. No local, drug-related adverse effects were noted. In particular, no local immune reaction was noted in response to newly made dystrophin protein in the EDB.
Clinical evaluation of the safety and efficacy of AVI-4658 following systemic administration is now underway with a dose-ranging systemic Phase 1b/2 clinical study in ambulant patients in the United Kingdom recently completed. In this study, patients were dosed by IV infusion once weekly at 0.5, 1.0, 2.0, 4.0, 10.0, or 20.0 mg/kg for 12 weeks. To support this study, and to enable the initiation of similar studies in the United States, we recently completed a repeat-dose toxicology study of AVI-4658 in cynomolgus monkeys in which animals were given 12 once-weekly injections. Here, we report the results of this study, designed to evaluate the toxicological consequences of repeated IV bolus administrations of AVI-4658 at doses up to and including the maximum feasible dose of 320 mg/kg per injection; subcutaneous administration was also tested at the high dose of 320 mg/kg per injection. The results show that AVI-4658 was well-tolerated at all doses tested, with no adverse effects reported, with only minimal and reversing histopathological changes occurring in the kidney, without any adverse renal functional changes.
Materials and Methods
Regulatory Compliance and Animal Use
This nonclinical laboratory study was conducted in accordance with the United States Food and Drug Administration (FDA) Good Laboratory Practice (GLP) Regulations, 21 Code of Federal Regulations (CFR) Part 58. This repeat dose toxicological evaluation of AVI-4658 in cynomolgus monkeys (Chinese origin) was performed at MPI Research (Mattawan, Michigan). The Testing Facility is AAALAC accredited, and the study plan was reviewed and approved by the Institutional Animal Care and Use Committee (IACUC), according to the existing animal health and welfare guidelines.
AVI-4658 Test Material
AVI-4658 was manufactured at AVI BioPharma (Corvallis, Oregon). Fresh vehicle, Dulbecco’s Phosphate Buffered Saline (PBS) without Calcium and Magnesium, 1X, was dispensed for use on study weekly and was stored refrigerated (approximately 2 to 8°C). The AVI-4658 test article was mixed with the appropriate amount of vehicle to achieve a bulk (stock) formulation, which targeted the highest concentration required for dosing. Three bulk (stock) formulations were prepared at concentrations of approximately 100 mg/ml, filtered through a 0.2 µm filter unit, and diluted aseptically to the required concentrations to achieve nominal dosing concentrations of 2, 20, or 100 mg/mL. Dosing formulations were prepared weekly, as needed, and were stored refrigerated (approximately 2 to 8°C) where it remained stable.
AVI-4658 Administration
Sixty animals (approximately 2.7 to 3 years of age and weighing 2.19 to 2.80 kg [30 males] and 2.15 to 2.69 kg [30 females], at randomization) were assigned to the control and treatment groups identified in Table 1 . The age of the animals was reviewed following randomization to avoid an age bias within the groups. Animals assigned to study had body weights within ~20% of the mean body weight for each sex.
Experimental Design of the Repeat-Dose Toxicological Evaluation of AVI-4658
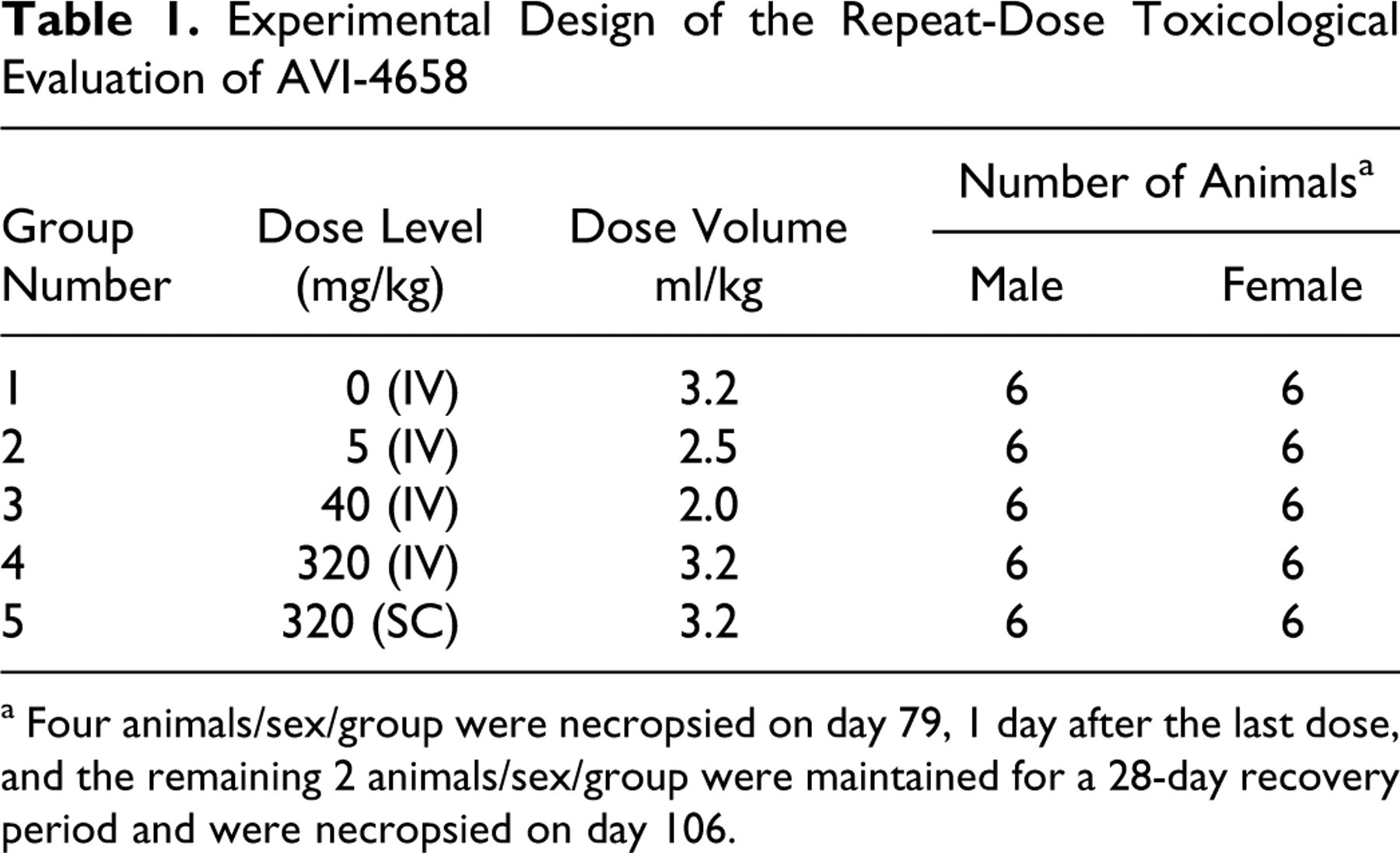
a Four animals/sex/group were necropsied on day 79, 1 day after the last dose, and the remaining 2 animals/sex/group were maintained for a 28-day recovery period and were necropsied on day 106.
Fluorescent lighting was provided for approximately 12 hours per day. The dark cycle was interrupted intermittently due to study-related activities. Temperature and humidity were continuously monitored and recorded. The protocol-designated ranges were 64 to 84°F and 30% to 70%, respectively. Lab Diet (Certified Primate Diet #5048, PMI Nutrition International, Inc) was available to the monkeys on a feeding regimen of 12 biscuits of food per day, except during designated periods. PrimaTreats were offered twice a day and other enrichment foods were provided on a regular basis. Tap water was available ad libitum via an automatic watering system.
Animals were dosed once weekly with AVI-4658 or vehicle for 12 weeks by IV bolus or SC injection to support these regimens clinically. For IV bolus administration, the range of dose levels was from 5 to 320 mg/kg; SC administration was evaluated at the high-dose level only. The high-dose level (320 mg/kg) is the maximum feasible dose based on the solubility of the test article and the maximum allowable volume of an IV bolus injection. Following the dosing period, 2 animals/sex per group were maintained for a 28-day recovery period. The control group received the vehicle in the same dose volume (3.2 mL/kg) as the treated groups.
In-Life Examinations
All animals were observed for morbidity, mortality, injury, and the availability of food and water twice daily throughout the duration of the study. A detailed clinical examination of each animal was performed weekly during the study. Body weights for all animals were measured and recorded prior to randomization (day −1) and weekly during the study. Food consumption was estimated daily by biscuit count. Opthalmoscopic examinations were conducted pretest and prior to the terminal and recovery necropsies.
All animals received an electrocardiographic examination pretest, predose and at approximately 1 to 2 hours (±15 minutes) postdose during the last week of dosing, and once during the week prior to the recovery necropsy. Insofar as possible, care was taken to avoid causing undue excitement of the animals before the recording of electrocardiograms (ECGs) in order to minimize extreme fluctuations or artifacts in these measurements. Standard ECGs (10 Lead) were recorded at 50 mm/sec. Using Lead II (or another appropriate lead) recorded at 50 mm/s, the RR, PR, and QT intervals, and QRS duration were measured and heart rate was determined. Corrected QT (QTc) interval was calculated using a procedure based on the method described by Bazett. 11 All tracings were evaluated and reported by a consulting veterinary cardiologist.
Clinical pathology evaluations on blood samples were conducted on all animals pretest, during week 4, and prior to the terminal and recovery necropsies. Urine samples for clinical pathology evaluations were collected from all animals pretest and prior to the terminal and recovery necropsies. Blood samples (approximately 4.8 to 5.8 mL) were collected from the femoral vein. Samples were collected into tubes containing K3EDTA for evaluation of hematology parameters and sodium citrate for evaluation of coagulation parameters. A serum separator was used for the clinical chemistry samples. Urine samples were collected using steel pans placed under the cages for approximately 16 hours.
Bioanalytical and Toxicokinetic Analysis
Blood samples (approximately 1.0 mL) were collected from all animals via the femoral vein for determination of the plasma concentrations of the test article. Samples were collected predose and at 0.25, 0.5, 1, 2, 4, 12, and 36 hours postdose on days 1 and 71. The samples were initially placed on dry ice as needed, stored frozen at approximately −70°C, and analyzed at BASi (McMinnville, Oregon). The toxicokinetic analysis and interpretation of the results was performed from mean concentration-time data in the test species. A noncompartmental module of WinNonlin Enterprise was used to calculate toxicokinetic parameters.
Nominal sample collection times and doses were used in the data analysis. Plasma concentrations of AVI-4658 were used to construct semi-logarithmic plasma concentration versus time curves. Toxicokinetic parameters were determined by noncompartmental methods using WinNonlin, version 5.0.1 (Pharsight Corporation, Mountain View, California). Values below the limit of quantitation (10.0 ng/mL) were treated as 0. Bioavailability was calculated by dividing mean AUC0-tlast for the SC dose on days 1 and 71 by the mean AUC0-tlast for the intravenous 320 mg/kg dose on days 1 and 71, respectively. Plasma concentrations and toxicokinetic parameters were rounded to 3 significant figures for presentation. All calculations were done with unrounded numbers. Toxicokinetic parameters are defined in Table 2 .
Summary of TK Parameters: Combined Male (n = 5) and Female (n = 5) Data
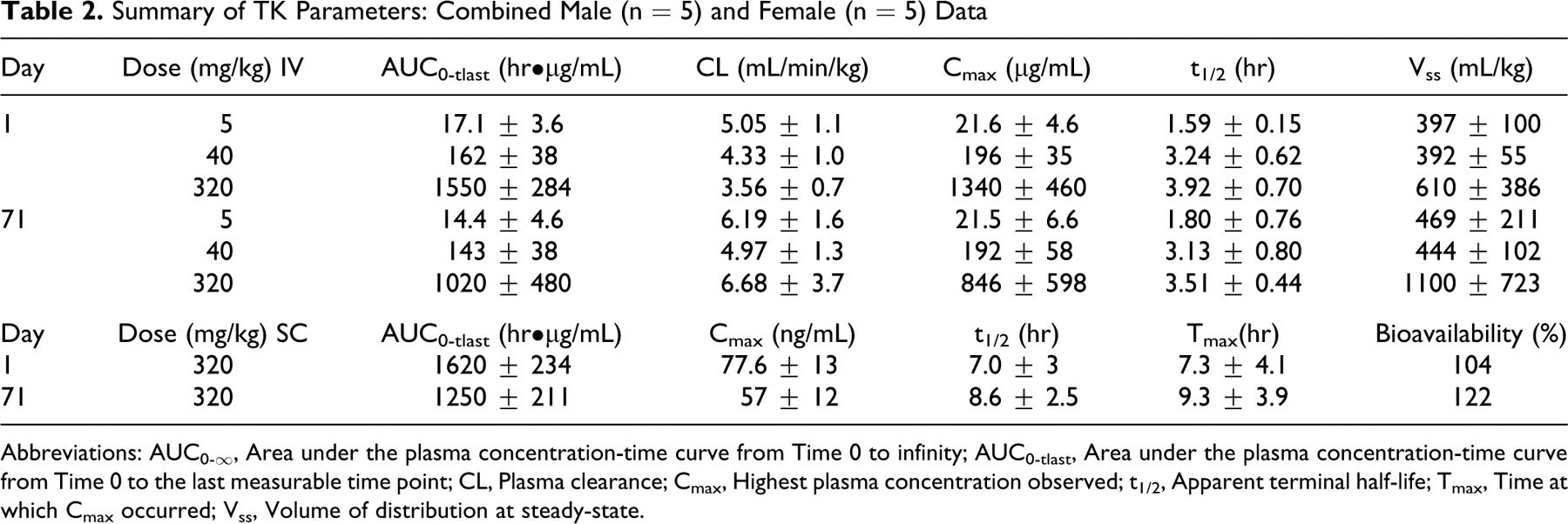
Abbreviations: AUC0-∞, Area under the plasma concentration-time curve from Time 0 to infinity; AUC0-tlast, Area under the plasma concentration-time curve from Time 0 to the last measurable time point; CL, Plasma clearance; Cmax, Highest plasma concentration observed; t1/2, Apparent terminal half-life; Tmax, Time at which Cmax occurred; Vss, Volume of distribution at steady-state.
Postmortem Study Evaluations
Macroscopic
Necropsy examinations were performed under procedures approved by a veterinary pathologist on any animal euthanized in extremis and at the scheduled terminal and recovery necropsies (days 79 and 106, respectively). The animals were euthanized by sedation with ketamine, followed by an IV overdose of sodium pentobarbital solution via the saphenous vein/artery or the cephalic vein and exsanguination by severing the femoral vessels. The animals were examined carefully for external abnormalities including masses. The skin was reflected from a ventral midline incision and any abnormalities were identified and correlated with antemortem findings. The abdominal, thoracic, and cranial cavities were examined for abnormalities and the organs removed, examined, and, where required, placed in fixative. A comprehensive panel of 59 different tissues were excised and fixed in neutral buffered formalin, except for the eyes (including the optic nerve) and testes, which were fixed using a modified Davidson’s fixative. The tissues examined were adrenal gland, aorta, bone with bone marrow (femur, sternum), brain (cerebrum, midbrain, cerebellum, medulla/pons), diaphragm, epididymis, esophagus, eye (with optic nerve), gallbladder, heart, injection sites(s), kidney, large intestine (cecum, colon), rectum, larynx, liver, lung with bronchi, lymph node (mandibular, mesenteric), mammary gland, ovary, oviducts, pancreas, Peyer’s patch, pituitary, prostate, salivary glands (mandibular, parotid, sublingual), sciatic nerve, seminal vesicles, skeletal muscle (biceps femoris, quadriceps, rectus femoris), skin, small intestine (duodenum, ileum, jejunum), spinal cord (cervical, lumbar, thoracic), spleen, stomach (cardia, fundus, pylorus), testis, thymus, thyroid gland (with parathyroid), tibiofemoral joint, tongue, trachea, ureters, urinary bladder, uterus with cervix, and vagina.
Organ weights
Body weights and protocol-designated organ weights were recorded for all animals at the terminal and recovery necropsies and appropriate organ weight ratios were calculated (relative to body and brain weights).
Microscopic
Microscopic examination of fixed hematoxylin and eosin-stained (H&E) paraffin sections was performed on protocol-designated sections of tissues. The slides were examined by a board certified veterinary pathologist. A 4-step grading system was utilized to define gradable lesions for comparison between dose groups (0 = normal, 1 = minimal, 2 = mild, 3 = moderate, 4 = severe).
Statistics
Group pairwise comparisons were made using Levene test to assess homogeneity of group variances for each endpoint. If Levene test was not significant (P > .01), a pooled estimate of the variance (Mean Square Error or MSE) was computed from a 1-way analysis of variance (ANOVA) and utilized by a Dunnett comparison of each treatment group with the control group. If Levene test was significant (P< .01), comparisons with the control group was made using Welch t-test with a Bonferroni correction. Log transformation was performed on the leukocyte count data and group pairwise comparisons analyses were performed as described above. Rank transformation was performed on the creatine kinase and urinalysis data and analyzed using Dunnett test.
Results
AVI-4658 was dosed once weekly for 12 weeks in groups of experimentally naïve cynomolgus monkeys (see Table 1), at doses up to and including the maximum feasible dose of 320 mg/kg per injection. No effects related to treatment with AVI-4658 were observed with respect to clinical observations, mean body weight, organ weights, mean food consumption, urinalysis values, macroscopic observations, or opthalmoscopic examinations at any dose level, either at termination or following the 28-day recovery period. In addition, no abnormal findings were observed during the electrocardiographic examinations taken at 1 to 2 hours postdose after the last dose and at the end of the recovery period. All animals were in sinus rhythm or sinus arrhythmia, both of which are normal in cynomolgus monkeys. Sinus tachycardia (ie, >270 beats per minute for a single interval) was observed on occasion across both treated and vehicle control groups, and is a normal variant in these animals. Otherwise, there was no effect of the IV bolus or SC injection of AVI-4658 on quantitative ECG parameters (Table 3 ). At week 4, there was statistically significant (P < .01) prolongation of activated partial thromboplastin time (APPT) of approximately 5 to 6 seconds relative to the control group (Table 3) in males and females at 320 mg/kg SC that by termination and recovery had completely resolved. Otherwise, APTT and prothrombin times were not meaningfully affected in any group or individual animal at any time or dose. No changes were noted in the above parameters at 5 or 40 mg/kg (data not shown).
Summary of Electrocardiographic, Coagulation and Clinical Chemistry Evaluation in Male (M) and Female (F) Monkeys Following Intravenous (IV) or Subcutaneous (SC) Administration
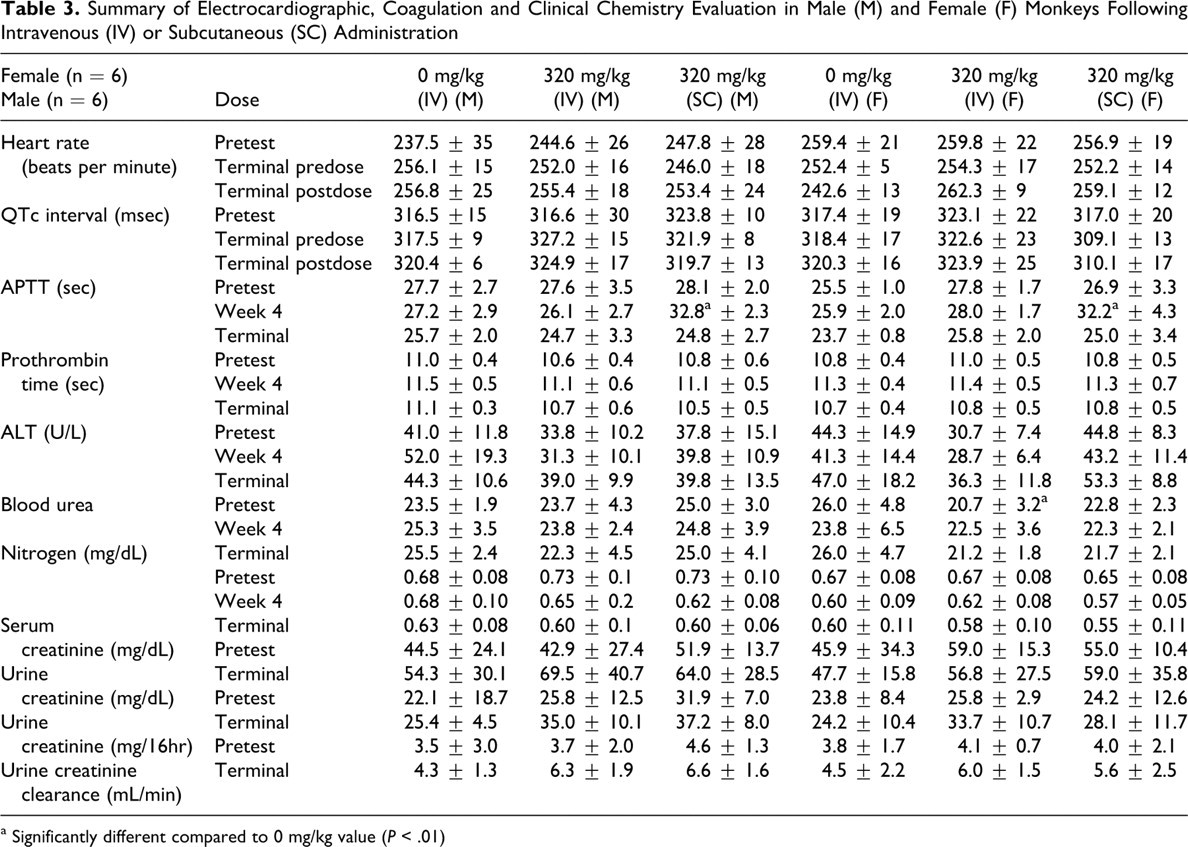
a Significantly different compared to 0 mg/kg value (P < .01)
Administration of AVI-4658 resulted in dose-dependent histopathological findings in the kidney that were present in both males and females, and included basophilic granules, basophilic tubules, and tubular vacuolation (Table 4 ). All of the changes were restricted to cortical tubules and were especially prominent in the tubules of the medullary ray. Findings in kidneys of monkeys given AVI-4658 at 320 mg/kg by SC route were similar to those in monkeys given AVI-4658 at 320 mg/kg by IV route. Mild basophilia within the cytoplasm of cortical tubules and minimal tubular vacuolation were present in all 8 animals administered 320 mg/kg IV (Figure 2A) while mild (5 of 8 animals) to moderate (3 of 8 animals) basophilic tubules with minimal (7 of 8 animals) to mild (1 of 8 animals) tubular vacuolation were observed in animals dosed 320 mg/kg SC (Figure 2B). Monkeys at 40 mg/kg (IV) had minimal basophilic tubules in the cortex (5 of 8 animals) with no associated vacuolation. Basophilic granules were present in 2 of 4 males at 5 mg/kg (IV) and at 40 mg/kg (IV). Following the recovery period, only some of the aforementioned microscopic observations remained present in the kidney (Table 4) and were either reduced in severity or present only focally or multifocally in clusters of 2 to 4 tubules. For example, 3 of 4 animals administered 320 mg/kg IV had minimal basophilic tubules in the cortex and 1 of 4 animals with minimal tubular vacuolation at the end of recovery while all 8 animals of this group had mild tubular basophilia in the cortex and minimal tubular vacuolation at termination. In the lower dose groups at recovery, there were no renal histopathology findings at 5 mg/kg and only 1 of 4 animals had minimal basophilia in the tubules at 40 mg/kg. Although the findings did not completely resolve following the 28-day recovery period, the above data suggest that they have the capability of reversing. Furthermore, the findings did not correlate with any clinical chemistry changes (see Table 3). In particular, no elevations in blood urea nitrogen (BUN), serum creatinine, electrolytes, or changes in urine creatinine or creatinine clearance were detected at week 4 of the study, or at the end of the 12 weekly doses (Table 3, Terminal), as compared to pretest values.
Microscopic Evaluation Summary of AVI-4658 in Monkeys
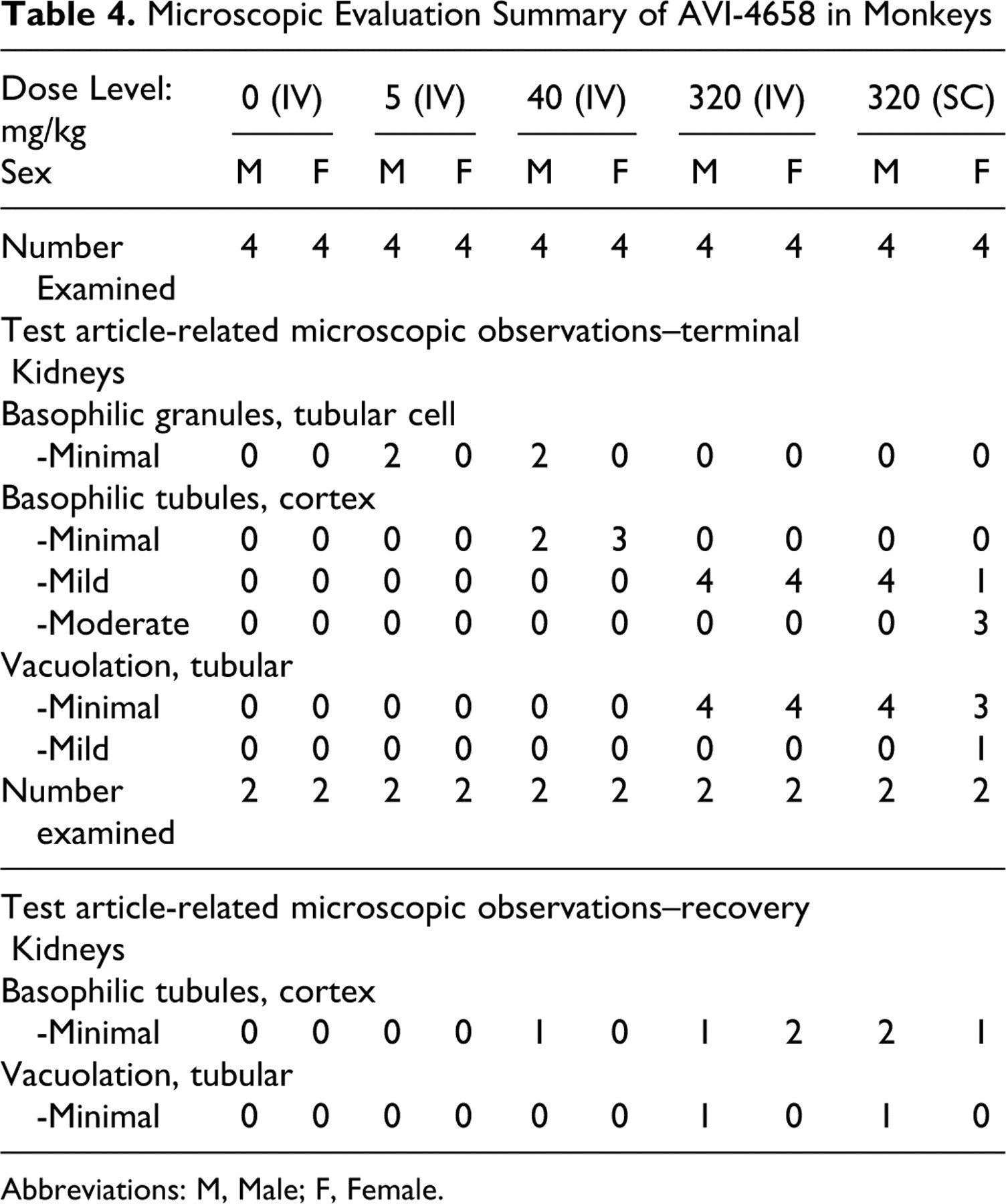
Abbreviations: M, Male; F, Female.
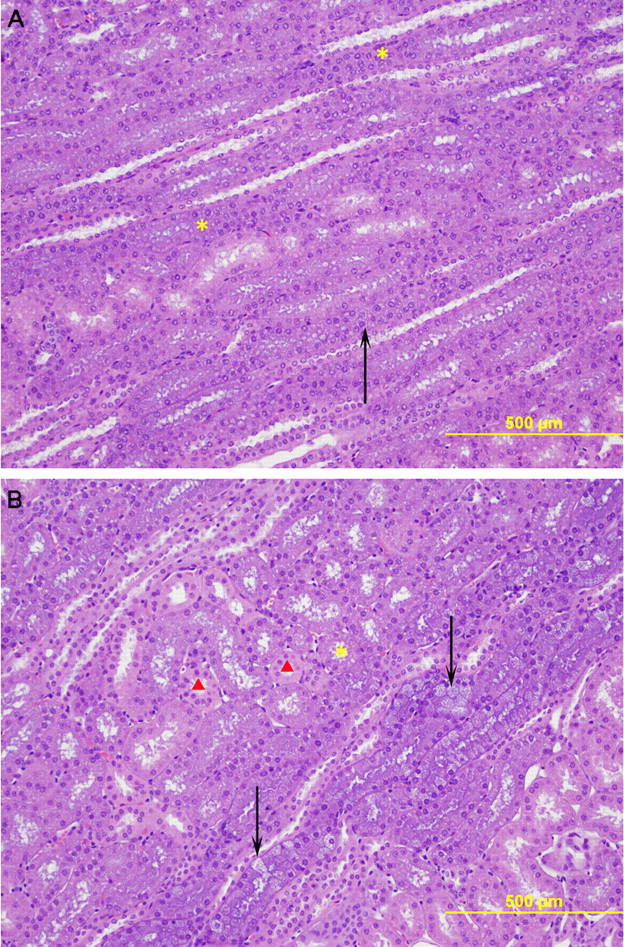
Representative photomicrographs of kidney tissue obtained at the end of dosing stained with hematoxylin and eosin. A, Photomicrograph from a male monkey treated with 320 mg/kg (IV) showing mild basophilic tubules (*) and minimal tubular vacuolation (arrow). B, Photomicrograph from a male monkey treated with 320 mg/kg (SC) of AVI-4658 showing moderate basophilic tubules (*) and mild tubular vacuolation (arrows). A small number of renal tubules are unaffected (red triangles). There was no evidence of degenerative changes in the tubular epithelium for either route of administration (Original objective lens magnification: ×10).
Basophilic macrophages were present (minimal to moderate) at the injection sites in most monkeys given 320 mg/kg SC and in 1 animal at 320 mg/kg IV (data not shown). At recovery, there was a reduction in the incidence and severity of this finding as roughly half the animals had minimal basophilic granules. Other findings at the injection sites included hemorrhage, inflammation, vascular degeneration/regeneration, and myofiber degeneration/regeneration, which were considered to be secondary to the injection procedure rather than AVI-4658 as many of these findings were also found in animals from the vehicle control group. At recovery, these findings were either absent or reduced in severity and/or incidence (data not shown).
Toxicokinetic analysis was performed following the first (day 1) and 11th (day 71) once-weekly dose of AVI-4658 for all animals in all groups, at 0.25, 0.5, 1, 2, 4, 12, and 36 hours postdose (Figure 3A and B , respectively). Bioavailability was 104% on day 1 and 122% on day 71. There were no significant differences in the toxicokinetic parameters calculated for male and female monkeys for the IV or SC administrations. There were also no differences in the toxicokinetic parameters calculated for days 1 and 71 (Table 2), thus showing no evidence for accumulation of AVI-4658 following administration by either route.
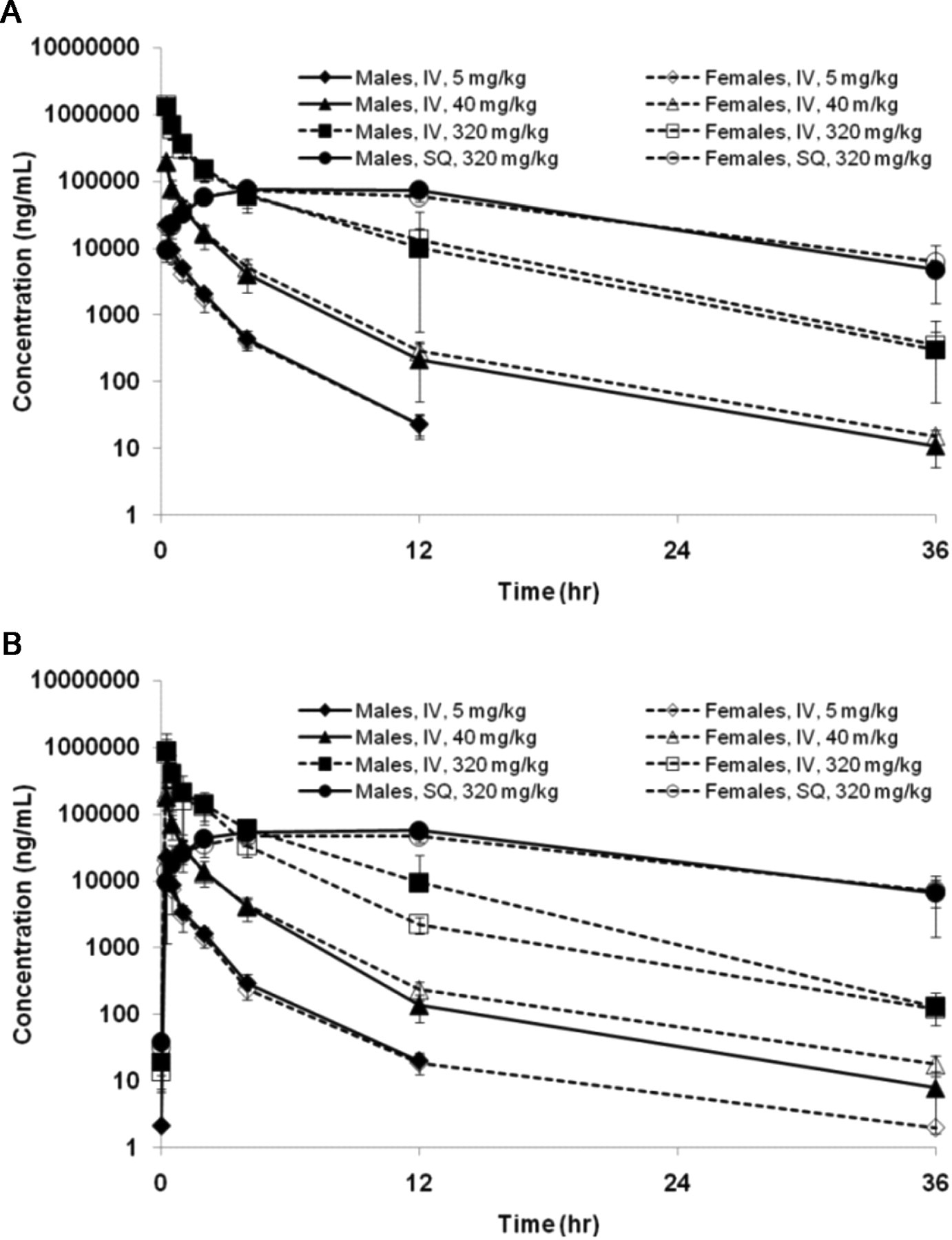
Mean (±SD) Plasma concentrations of AVI-4658 in male and female monkeys after a single (A) or the 11th (B) intravenous (IV) or subcutaneous (SQ) dose.
The above data are in agreement with the short half-life of AVI-4658 of between 1.6 and 3.5 hours (IV bolus) and 7 and 9 hours (SC), which combined with a 1-week dosing interval would provide sufficient time for AVI-4658 to decline to nonmeasurable levels prior to the next dose, with no accumulation expected at either the 11th or 12th dose. Note that the plasma half-life was generally dose independent, and that the slightly lower half-lives observed at the 5 mg/kg dose are likely due to a drop below the lower limit of quantification of the values at later time points. Area under the curve increased in approximately linear proportion to dose between 5 and 320 mg/kg (Table 2). As expected, the CL and Vss were similar across dose levels. Thus, the toxicokinetic profile of AVI-4658 is fairly standard, with the intensity of histological findings in the kidney increasing in proportion to total exposure to the drug over 12 weekly doses.
Discussion
Duchenne muscular dystrophy results from mutations of the dystrophin gene, and affects 1 in every 3500 male newborns worldwide. 12 The lack of this vital structural protein reduces the stability of the sarcolemma of the muscle and ultimately leads to muscle fiber degeneration. 12,13 The clinical consequences of the disease are mild early in life, although often seen in the toddler years as ambulation even then may become impaired, but become significantly more severe as the boys progress from age 7 to 11. By this age, the accumulation of fibrosis that replaces the damaged muscle and the invasion of adipose tissue reduces muscle function, and loss of ambulation typically occurs before age 12. 14 To date, there are no approved drugs that directly address the root cause of the disease, dystrophin deficiency. The current standard of care includes physical therapy and administration of glucocorticoids, 15 such as prednisolone and deflazocort, that have been shown to delay loss-of-ambulation by 2 to 3 years. 12,16 However, these steroids are associated with significant side effects, including obesity, spine deformities, bone loss, and growth retardation. 17,18 Additional support to patients with DMD include assisted ventilation with portable ventilators, which can significantly prolong life to around age 24. The above underscores the need for a safe, effective treatment for DMD that can restore dystrophin and maintain and/or restore muscle function.
This study was conducted to evaluate the toxicity and toxicokinetic profile of AVI-4658 when administered via IV bolus or SC injection to cynomolgus monkeys once weekly over 12 weeks, to support the same regimen clinically. The reversibility, progression, or delayed appearance of any observed changes was evaluated following a 28-day postdose observation period. The results confirm the tolerability of AVI-4658 at doses up to and including the maximum feasible dose of 320 mg/kg per injection previously observed in cynomolgus monkeys. 9 While there were variations in the dose volumes across dose groups, we believe that this had no impact on the interpretation of this study, as control monkeys were dosed at the same volume as the high-dose monkeys and the differences in dose volumes were relatively small. No drug-related effects were noted on survival, clinical observations, body weight, food consumption, opthalmoscopic and electrocardiographic evaluations, hematology, clinical chemistry, urinalysis, organ weights, and macroscopic evaluations. The only drug-related effects in these animals were dose-dependent and apparent reversible microscopic kidney findings in both male and female animals, which included basophilic granules and tubules, and tubular vacuolation without any evidence of degeneration. In particular, at 5 and 40 mg/kg, a relatively low incidence of basophilic granules was observed in males at the end of dosing and was completely absent in all groups at recovery. Similarly, the basophilic tubules and vacuolization, which were observed to some degree in all animals in the 320 mg/kg IV group, was reduced in intensity and limited to 3 of 4 animals for basophilic tubules, and to 1 of 4 animals for vacuolization after 28 days of recovery, suggesting that the resolution is in progress. There was no evidence of associated inflammation, tubular degeneration, necrosis, and casts in the kidney. Furthermore, there were no concomitant changes in renal serum or urine markers that would suggest significantly (eg, >60%) reduced kidney function.
The presence of basophilic granules and tubules without changes in cell morphology, such as degeneration, is thought not to reflect an adverse event or imply reduced renal function but as an observation of the sequestered accumulation of oligo in endosomes or phagolysosomes. 19 –21 To determine whether the basophilic granules observed in the kidneys of treated monkeys contains AVI-4658, we are currently developing methods to demonstrate that we have local exposure at a target tissue.
Recently, the Predictive Safety Testing Consortium along with industry and regulatory investigators have identified and recommended the use of 7 sensitive and specific renal urinary biomarkers (KIM-1, albumin, total protein, β2-Microglobulin, cystatin C, clusterin, and trefoil factor-3) to use in preclinical studies as biomarkers of potential renal injury to supplement the traditional but relatively insensitive biomarkers BUN and creatinine. 22 In future preclinical studies, we are planning to incorporate some of these biomarkers to detect potential proximal tubule damage. Taken together, the above data agree with the interpretation that no adverse effects were observed following 12 weekly IV bolus or SC administrations of AVI-4658 in cynomolgus monkeys at up to and including 320 mg/kg.
Toxicokinetic evaluation revealed that there were no differences in the toxicokinetic parameters calculated for days 1 and 71, establishing that there was no accumulation of drug in the plasma upon weekly dosing. This is most likely due to the relatively short plasma half-life of the drug, allowing complete clearance prior to subsequent dosing. Accumulation of AVI-4658 in renal cells is thought to be a result of high renal clearance rates and uptake by nonhybridization-specific mechanisms, as there is no renal dystrophin expression. Based on the absence of adverse findings, the No-Observed-Adverse-Effect-Level for AVI-4658 in male and female cynomolgus monkeys administered 12 weekly IV bolus doses was 320 mg/kg (IV and SC) corresponding to an average AUC of 1020 hr•µg/mL (IV) and 1250 hr•µg/mL (SC) at day 71 of the study.
Footnotes
Acknowledgments
We would like to thank J. Schuh, R. Kole, and P. Medeiros for their critical evaluation of this manuscript. This work was supported by a research grant W81XWH-09-1-0215 (PI: Eric P Hoffman) from the DOD/USAMRAA.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
