Abstract
AVI-4658 is a phosphorodiamidate morpholino oligomer (PMO) designed to induce skipping of dystrophin exon 51 and restore its expression in patients with Duchenne muscular dystrophy (DMD). Preclinically, restoration of dystrophin in the dystrophic mdx mouse model requires skipping of exon 23, achieved with the mouse-specific PMO, AVI-4225. Herein, we report the potential toxicological consequences of exon skipping and dystrophin restoration in mdx mice using AVI-4225. We also evaluated the toxicological effects of AVI-4658 in both mdx and wild-type mice. In both studies, animals were dosed once weekly for 12 weeks up to the maximum feasible dose of 960 mg/kg per injection. Both AVI-4658 and AVI-4225 were well-tolerated at all doses. Findings in AVI-4225-treated animals were generally limited to mild renal tubular basophilia/vacuolation, without any significant changes in renal function and with evidence of reversing. No toxicity associated with the mechanism of action of AVI-4225 in a dystrophic animal was observed.
Introduction
Since first described in detail by Duchenne de Boulogne in 1861, it was noted that DMD predominantly affects males, suggesting that genetic variations were responsible for the disease, and that the trait was recessive and linked to the X chromosome. 1 Upon the discovery of the dystrophin gene approximately 25 years ago, various causal genetic mutation types were identified, including exon deletions and nonsense mutations. 2 The absence of dystrophin protein expression has pathogenic and physiological implications in both patients and animals. 3 The best characterized of the naturally occurring dystrophic animal strains is the mdx mouse, which harbors a spontaneous nonsense mutation in exon 23 of dystrophin. 4 The nonsense mutation completely abrogates dystrophin expression, which manifests in a clear dystrophic phenotype. Although the severity of the phenotype is less than that of patients with DMD, there are several important similarities. Patients with DMD typically show signs of muscle hypertrophy early in life, which is the manifestation of rapid muscle degeneration / regeneration that leads to damage and fibrosis. 1 As the disease progresses, degeneration becomes more dominant, leading to loss of mobility by age 11, and death due to cardiac or respiratory failure in their early 20s. The mdx mice show a similar pattern of muscle degeneration/regeneration, hypertrophy, and loss of strength although, in the skeletal muscle, the rate of muscle wasting is much slower. 5,6 Like patients with DMD, mdx mice also exhibit reduced cardiac 7 and respiratory capacity, 8 which worsens with age. The lifespan of mdx mice is reduced by approximately 20% (22 vs. 27 months) compared to C57 wild-type (C57) mice. 9 The mdx mouse exhibits muscle pathology, including a marked increase in serum levels of creatine kinase (CK), alanine aminotransferase (ALT), and aspartate aminotransferase (AST), due to release from degenerating muscle fibers. All other serum chemistries (eg, blood urea nitrogen [BUN], creatinine, etc) are normal compared to age-matched C57 control animals. 10 This typical serum chemistry profile is similar to that of patients with DMD.
In most cases, lack of dystrophin expression in patients with DMD is caused by internal deletions that result in the disruption of the reading frame of the resulting messenger RNA (mRNA). 1,11 Complete lack of dystrophin is linked with the most severe cases of the disease. A milder form of the disease, Becker Muscular Dystrophy (BMD), can be caused by internal deletions that do not disrupt the reading frame, leading to internally truncated but still at least partially functional protein. Patients with BMD usually have a near-normal life-expectancy and may remain ambulant throughout their lives.
Splice switching oligomers (SSO) are a new class of antisense compound that can modulate dystrophin mRNA splicing and convert out-of-frame DMD mutations into the nearest in-frame BMD-like mutation. 12 –15 Some of the most striking data in this regard have been produced using phosphorodiamidate morpholino oligomers (PMO, see Figure 1) as SSOs. Phosphorodiamidate morpholino oligomers are chemically modified to impart stability against a large number of nucleases and proteases, 16 compared to native DNA or RNA, and are uncharged at physiological pH. Skipping exon 23 using PMO removes the premature stop codon from the spliced mRNA, restoring the reading frame and enabling expression of internally truncated but functional dystrophin protein. In the mdx mouse, AVI-4225, a PMO targeted to exon 23 of mouse pre-mRNA, was administered by systemic injection. 17 Once weekly intravenous (IV) doses at 100 mg/kg induced expression of functional levels of dystrophin in body-wide skeletal muscles of the dystrophic mdx mouse. 17 Physiological examination of tibialis anterior (TA) muscles 2 weeks after 3 once weekly IV injections of AVI-4225 (100 mg/kg per dose) revealed a significantly higher normalized maximum isometric tetanic force in all TA muscles from the treated versus untreated mdx mice. Importantly, once produced, newly made dystrophin protein has been shown to persist in muscles for up to 26 weeks. 18
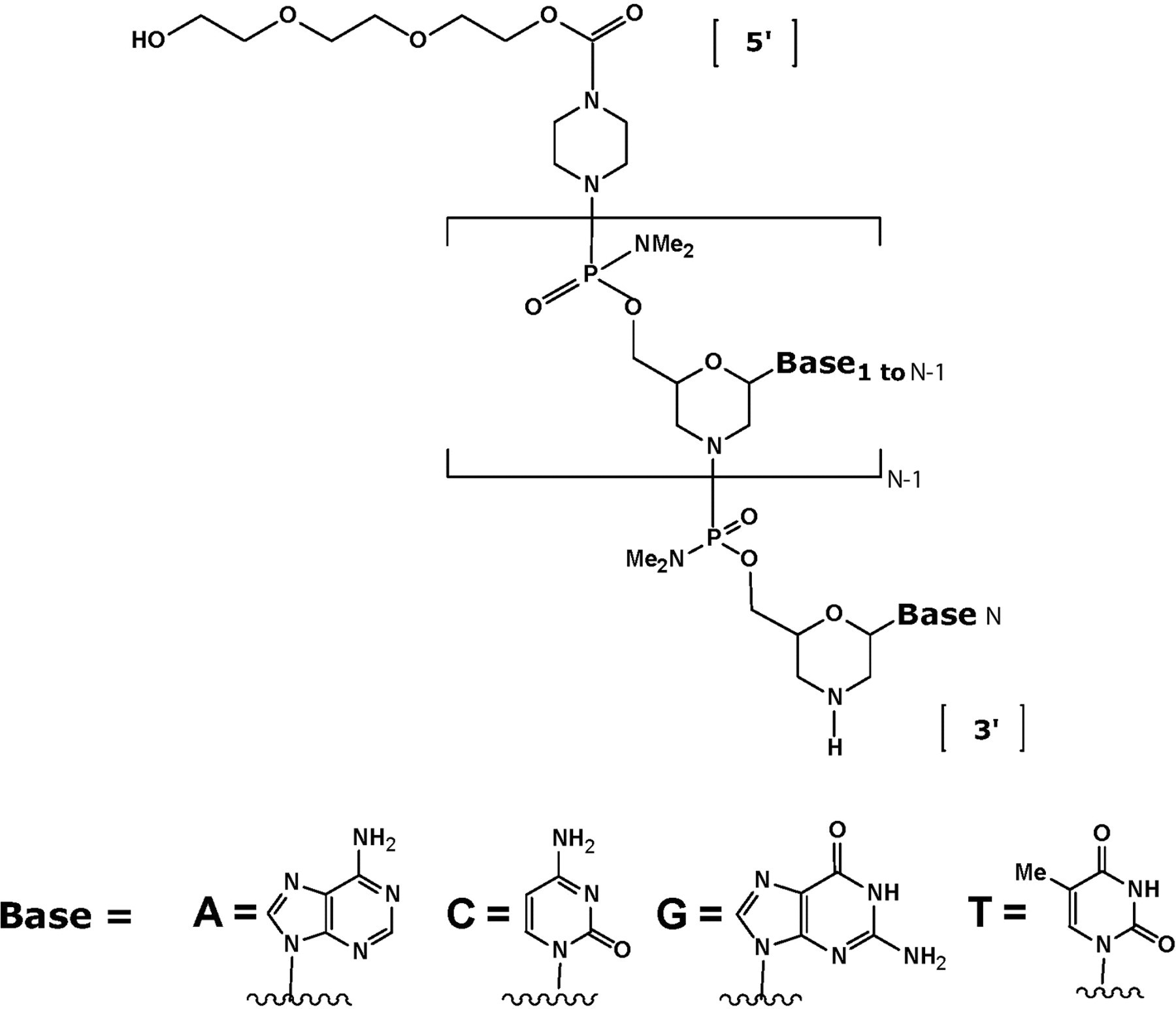
Chemical structure of phosphorodiamidate morpholino oligomer (PMO). The base sequence of AVI-4658 is CTC CAA CAT CAA GGA AGA TGG CAT TTC TAG; the base sequence of AVI-4225 is GGC CAA ACC TCG GCT TAC CTG AAA T.
AVI-4658 is a PMO currently in clinical development for the treatment of DMD in patients and induces skipping of human dystrophin exon 51. In patients with reading frame-disrupting mutations in the dystrophin gene, including deletions of exons 50, 52, 52-63, 45-50, 48-50, or 49-50, AVI-4658 is predicted to restore the translation reading frame and thus enable the synthesis of an internally truncated dystrophin protein which is anticipated to be almost fully functional. 19 Recently, a proof of concept clinical trial was conducted, in which AVI-4658 was delivered locally to a single muscle by intramuscular injection, confirming the ability of the drug to induce dystrophin protein in patients with DMD. 20 Clinical evaluation of the safety and efficacy of AVI-4658 following systemic administration is now underway. In a recently completed study in the United Kingdom (UK), AVI-4658 was administered once weekly at 6 different dose levels, intravenously for up to 12 weeks, while clinical observations were continued for 26 weeks from the first dose.
We previously reported the data from studies of AVI-4658 genotoxicity along with safety pharmacology in nonhuman primates. 21 In the current 12-week preclinical study, our goal was to evaluate the potential toxicological consequences of exon skipping and dystrophin restoration in the dystrophic mdx mice. As AVI-4658 is targeted to exon 51, which is downstream of the exon 23 genetic lesion in the mdx mouse, it will not repair or restore dystrophin expression in the mdx mouse. We therefore used AVI-4225 as a surrogate for this study and evaluated the toxicity of this compound in both dystrophic mdx mice and C57 mice. Further, we evaluated AVI-4658, which does not affect dystrophin expression in mice, in both strains in a separate, but similarly designed, 12-week study.
Materials and Methods
Regulatory Compliance and Animal Use
The mdx mice (C57BL/10ScSn-Dmdmdx/J) were acquired from The Jackson Laboratory (Bar Harbor, Maine); the C57 wild-type mice (C57BL/6NCrl) were acquired from Charles River Laboratories (Portage, Michigan). All studies were conducted in accordance with the United States Food and Drug Administration (FDA) Good Laboratory Practice (GLP) Regulations, 21 Code of Federal Regulations (CFR) Part 58 at MPI Research (Mattawan, Michigan). All study plans were reviewed and approved by the Institutional Animal Care and Use Committee (IACUC), according to the existing animal health and welfare guidelines; the MPI Research testing facility is AAALAC accredited.
Test Article and Administration
AVI-4658 and AVI-4225 (Figure 1) were manufactured at AVI BioPharma (Corvallis, Oregon). Using aseptic techniques and sterile equipment, the required volumes of vehicle, Dulbecco’s Phosphate Buffered Saline without calcium and magnesium, were used to suspend test article. The stock solution was subsequently filtered through an appropriate number of 0.22 μm polyethersulfone (PES) filters. Using aseptic techniques and sterile equipment, dosing formulations were prepared weekly at nominal concentrations of 2, 20, and 100 mg/mL by adding an appropriate amount of stock solution into amber glass serum bottles containing a measured amount of vehicle. The bottles were capped, gently swirled, or stirred using a magnetic stir bar and stir plate until uniform in appearance and stored refrigerated (2° to 8°C) when not in use.
Using standard randomization procedures, 150 male and 150 female mdx mice (weighing 20.2 to 25.7 g and 15.6 to 22.6 g, respectively) and 60 male and 60 female C57 mice (weighing 19.2 to 23.3 g and 17.3 to 19.9 g, respectively) were divided into 2 equal groups for 2 studies and assigned to the control or treatment groups dosed either AVI-4225 or AVI-4658 depending on the study; each with the identical study design outlined in Table 1. Animals assigned to study had body weights within ±20% of the mean body weight for each sex. Each animal was implanted with a microchip bearing a unique identification number, and animal identification was verified during the course of the study.
Experimental Design for both of the Repeat Dose Toxicological Evaluations of AVI-4658 and AVI-4225 in Mice Given Weekly IV or SC Bolus Injections for 12 Weeks a
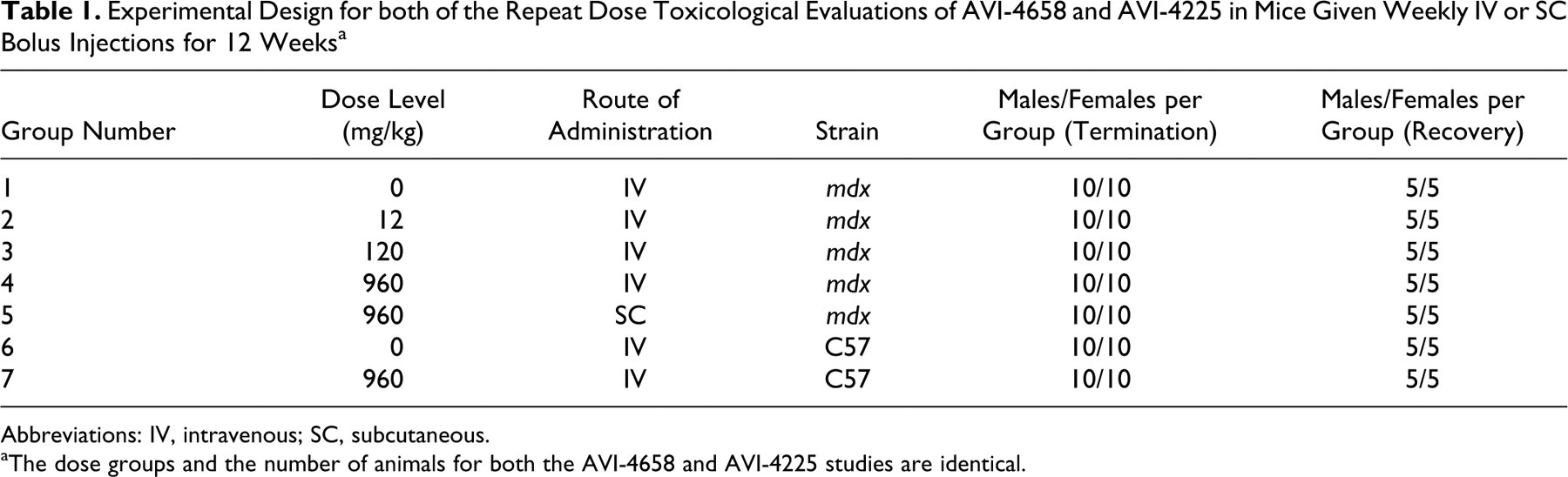
Abbreviations: IV, intravenous; SC, subcutaneous.
aThe dose groups and the number of animals for both the AVI-4658 and AVI-4225 studies are identical.
The animals were individually housed in suspended, stainless steel, wire-mesh type cages. Fluorescent lighting was provided for approximately 12 hours per day. Temperature and humidity were continuously monitored and recorded. The protocol-designated ranges were 64 to 79°F and 30% to 70%, respectively. Meal Lab Diet (Certified Rodent Diet #5002, PMI Nutrition International, Inc) was available ad libitum, except during designated periods. The lot number from each diet lot used for this study was recorded. Certification analysis of each diet lot was performed by the manufacturer. Tap water was available ad libitum via an automatic watering system. The water supply was monitored for specified contaminants at periodic intervals according to standard operating procedures.
All animals were dosed once weekly for 12 weeks. For intravenous injections, the vehicle or test article was administered via a 30-second bolus injection into the tail vein of restrained animals using 26 gauge × 3/8 inch beveled needles attached to appropriately sized syringes. The dose levels were 12, 120, and 960 mg/kg per injection (IV; mdx strain) and 960 mg/kg per injection (IV; non-mdx strain) and administered at respective dose volumes of 6, 6, 9.6, and 9.6 mL/kg. The control groups [0 mg/kg (IV; mdx strain) and 0 mg/kg (IV; non-mdx strain)] received the vehicle in the same manner as the treated groups at a dose volume of 9.6 mL/kg. Individual doses were based on the most recent body weights. These dose levels represent escalation up to and including the maximum feasible dose, based on the solubility of the compounds. For subcutaneous (SC) administration, at dose of 960 mg/kg, the test article formulation was injected between the skin and underlying layers of tissue at 4 alternating dose sites on the dorsal region of restrained animals using a 26-gauge needle attached to a 1 cc syringe. Dose sites were rotated weekly.
In-Life Examinations
All animals were observed for morbidity, mortality, injury, and the availability of food and water twice daily throughout the duration of the study. A detailed clinical examination of each animal was performed prior to randomization and weekly during the study. The observations included, but were not limited to, evaluation of the skin, fur, eyes, ears, nose, oral cavity, thorax, abdomen, external genitalia, limbs and feet, respiratory and circulatory effects, autonomic effects such as salivation, and nervous system effects including tremors, convulsions, reactivity to handling, and unusual behavior. Body weights for all animals were measured and recorded at receipt, prior to randomization, and weekly during the study. The body weights recorded at receipt are not reported but are maintained in the study file. Food consumption was measured and recorded weekly during the study.
Clinical pathology evaluations were conducted on all animals designated for each necropsy (10 animals/sex per group at the terminal necropsy and 5 animals/sex per group at the recovery necropsy). The first 5 animals/sex per group were used for hematology parameters (leukocyte count, erythrocyte count, hemoglobin, hematocrit, and blood cell morphology) and the last 5 animals/sex per group were used for clinical chemistry (CK, creatinine, AST, ALT, urea nitrogen, total bilirubin, total protein, albumin, glucose, calcium, phosphorus, and alkaline phosphatase [AP]) parameters at termination, while the first 2 animals/sex per group were used for hematology parameters and the last 3 animals/sex per group were used for clinical chemistry parameters at recovery. The animals had access to drinking water but were fasted overnight prior to sample collection. Blood samples (maximum amount possible) were collected via the vena cava following carbon dioxide inhalation. Samples were collected into tubes containing K3EDTA for evaluation of hematology parameters. No anticoagulant was used for the clinical chemistry samples. The order of bleeding was by alternating 1 animal from each dose group, then repeating to reduce handling and time biases. The animals were housed in wire mesh cages as above and urine was collected for at least 12 hours.
Postmortem Study Evaluations
Macroscopic.
Necropsy examinations were performed under procedures approved by a veterinary pathologist on all animals. The animals were euthanized by carbon dioxide inhalation followed by exsanguination of the abdominal vena cava, and were examined carefully for external abnormalities including palpable masses. The skin was reflected from a ventral midline incision and any abnormalities were identified and correlated with antemortem findings. The abdominal, thoracic, and cranial cavities were examined for abnormalities and the organs removed, examined, and, where required, placed in fixative. Tissues were fixed in neutral buffered formalin, except for the eye (including the optic nerve) and testes, which were fixed using a modified Davidson’s fixative. Formalin was infused into the lung via the trachea and into the urinary bladder.
Organ weights and microscopic evaluations.
Body weights and protocol-designated organ weights were recorded for all surviving animals at the scheduled necropsy and appropriate organ weight ratios were calculated (relative to body and brain weights). Paired organs were weighed together. The thyroid/parathyroid gland and pituitary gland were weighed following fixation. Microscopic examination of fixed hematoxylin and eosin-stained paraffin sections was performed on a panel of tissues, and slides were examined by a board-certified veterinary pathologist. A 4-step grading system was utilized to define gradable lesions for comparison between dose groups. Microscopic evaluation was performed on the following tissues: adrenal gland, aorta, bone with bone marrow (femur, sternum), brain (cerebrum, midbrain, cerebellum, medulla/pons), diaphragm, epididymis, esophagus, eye (with optic nerve), gallbladder, heart, injection sites(s), kidney, large intestine (cecum, colon, rectum), larynx, liver, lung with bronchi, lymph node (mandibular, mesenteric), mammary gland, ovary, oviducts, pancreas, Peyer’s patch, pituitary, prostate, salivary glands (mandibular, parotid, sublingual), sciatic nerve, seminal vesicles, skeletal muscle (biceps femoris and quadriceps), skin, small intestine (duodenum, ileum, jejunum), spinal cord (cervical, lumbar, thoracic), spleen, stomach (glandular, nonglandular), testis, thymus, thyroid gland (with parathyroid), tibiofemoral joint, tongue, trachea, ureters, urinary bladder, uterus with cervix, and vagina
Results
AVI-4225 or AVI-4658 were dosed once weekly (IV and SC) for 12 weeks in groups of dystrophic mdx and C57 mice (see Table 1), at doses up to and including the maximum feasible dose of 960 mg/kg per injection. Both PMOs were well tolerated, as no effects related to treatment with AVI-4225 or AVI-4658 were observed with respect to clinical observations, mean body weight, organ weights, mean food consumption, macroscopic observations, or opthalmoscopic examinations at any dose level, either at termination or following the 28-day recovery period.
As is typically observed with the dystrophic mdx mouse strain, serum values of ALT, AST, CK, and AP (Table 2) were generally elevated in both control and treatment groups of mdx mice, relative to the C57 mice. 10 In mdx mice, there were treatment-related reductions in serum ALT and AP in mdx mice with AVI-4225 administration at the end of dosing, consistent with the restoration of functional dystrophin expression of AVI-4225 in these animals (see Discussion). As expected, there were no treatment-related changes in these markers with C57 mice. Note that for CK, a widely used marker for muscle damage and pathology, large intergroup variability, and/or lack of complete data sets due to low serum volumes made meaningful interpretation of these data difficult.
Selected Clinical Chemistry Summary at Termination and Recovery for AVI-4225 (Mean ± Standard Deviation)
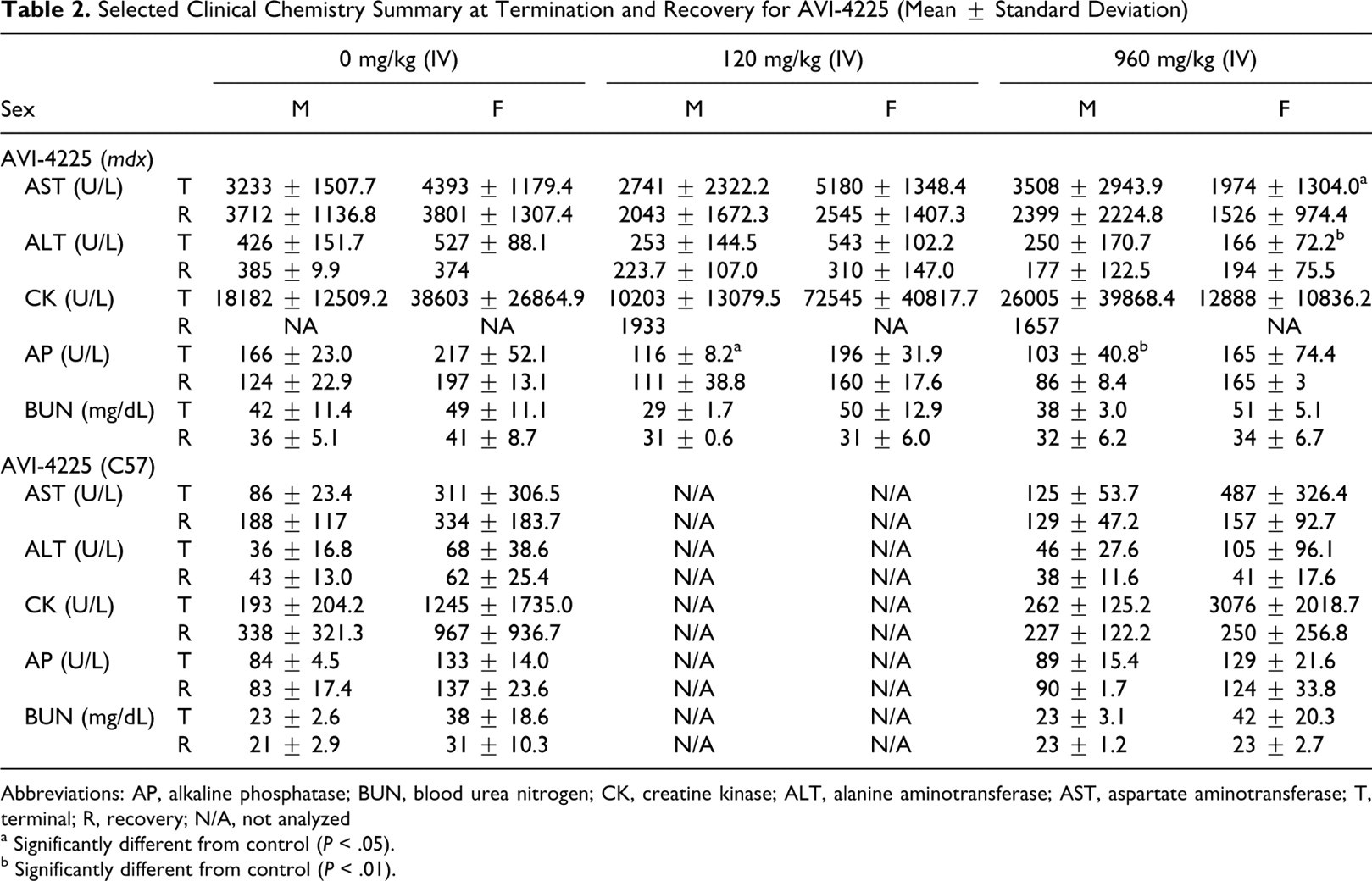
Abbreviations: AP, alkaline phosphatase; BUN, blood urea nitrogen; CK, creatine kinase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; T, terminal; R, recovery; N/A, not analyzed
a Significantly different from control (P < .05).
b Significantly different from control (P < .01).
With AVI-4225, histopathological findings were restricted to the kidney, and included minimal-to-mild tubular vacuolization, basophilic granules and tubules in the cortex of both mdx and C57 mice at 960 mg/kg per dose (IV and SC) by the end of dosing (Table 3). Basophilic granules consisted of amorphous, granular (<1 μm to >5 μm) accumulations of basophilic material with staining characteristics consistent with nucleic acids within the cytoplasm of affected tubular epithelial cells; representative images of kidney changes in mdx mice following AVI-4225 treatment are shown in Figure 2. At the end of recovery, 28 days after the last dose, animals dosed with AVI-4225 exhibited signs of reversibility of the kidney findings observed at termination. The incidence of basophilic granules, which occurred minimally at termination in 8/20 mdx mice at 960 mg/kg (IV), was present minimally in 3 of 10 animals at recovery (Table 3). In C57 mice, the relative severity of the basophilic tubules in the cortex was reduced from 7/20 and 9/20 scoring minimal and mild, respectively, to 9 out of 10 animals scoring minimal and no animals scoring mild at recovery.
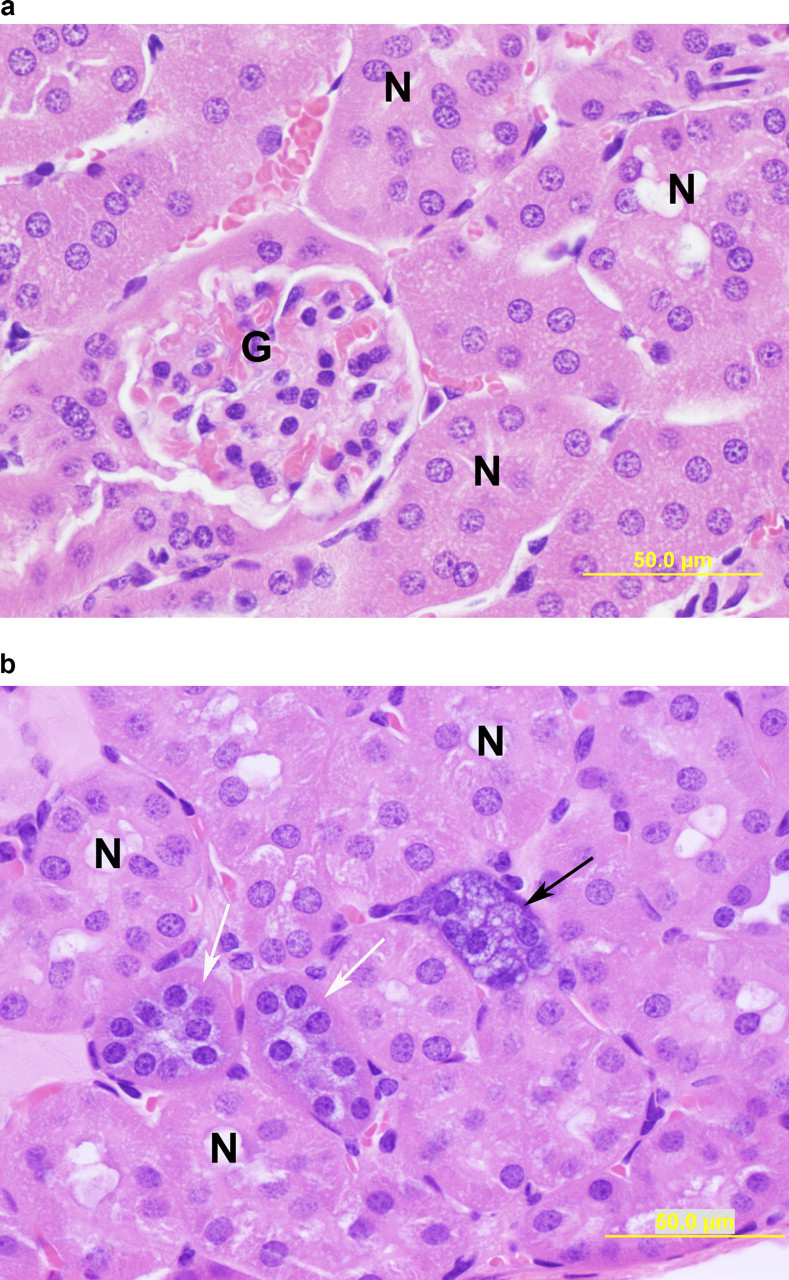
Kidney section at termination from mdx mouse treated with (A) vehicle control (B) 960 mg/kg IV AVI-4225 stained with hematoxylin and eosin stain. Presented here are tubules with basophilic epithelium (white arrows) and tubules with basophilic epithelium and variably vacuolated epithelial cells (black arrows). Tubules undergoing degeneration are not seen. Normal tubules (N) and glomeruli (G) are also present. (Original objective lens magnification: ×40.)
Microscopic Evaluation Summary of AVI-4225 in Mice at Termination
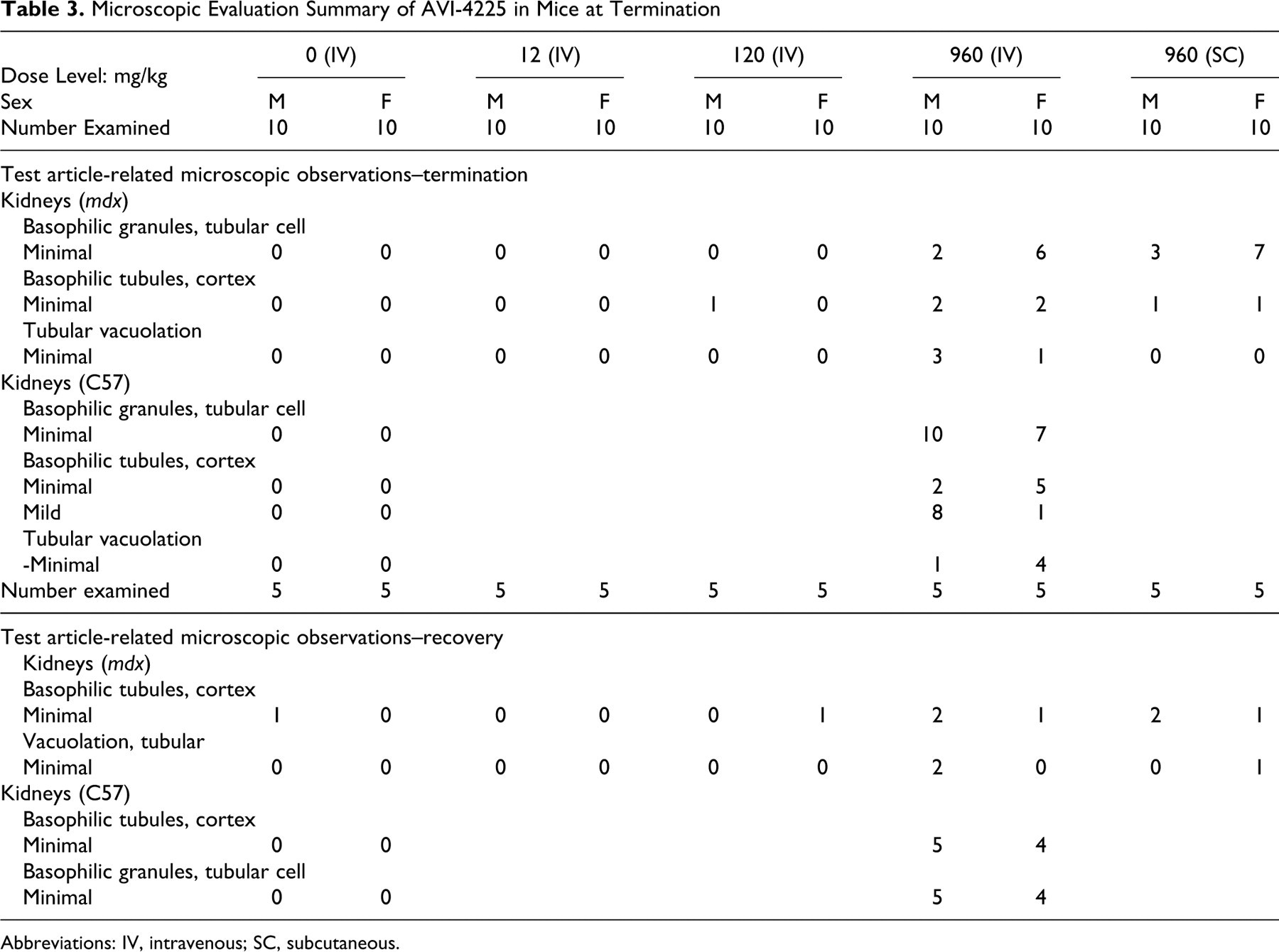
Abbreviations: IV, intravenous; SC, subcutaneous.
The mdx mice presented with their expected phenotype that included myofiber degeneration in the larynx, biceps femoris, diaphragm, quadriceps, and tongue, as shown by the high instances of these findings in the vehicle control group (Table 4). Following AVI-4225 treatment, these findings were reduced in frequency and severity in all muscles in a dose-dependent manner. These improvements in muscle pathology, consistent with the mechanism of action of AVI-4225 in mdx mice, were still present in the animals in the 28 day recovery group.
Improvement in Muscle Pathology Following AVI-4225 in mdx Mice
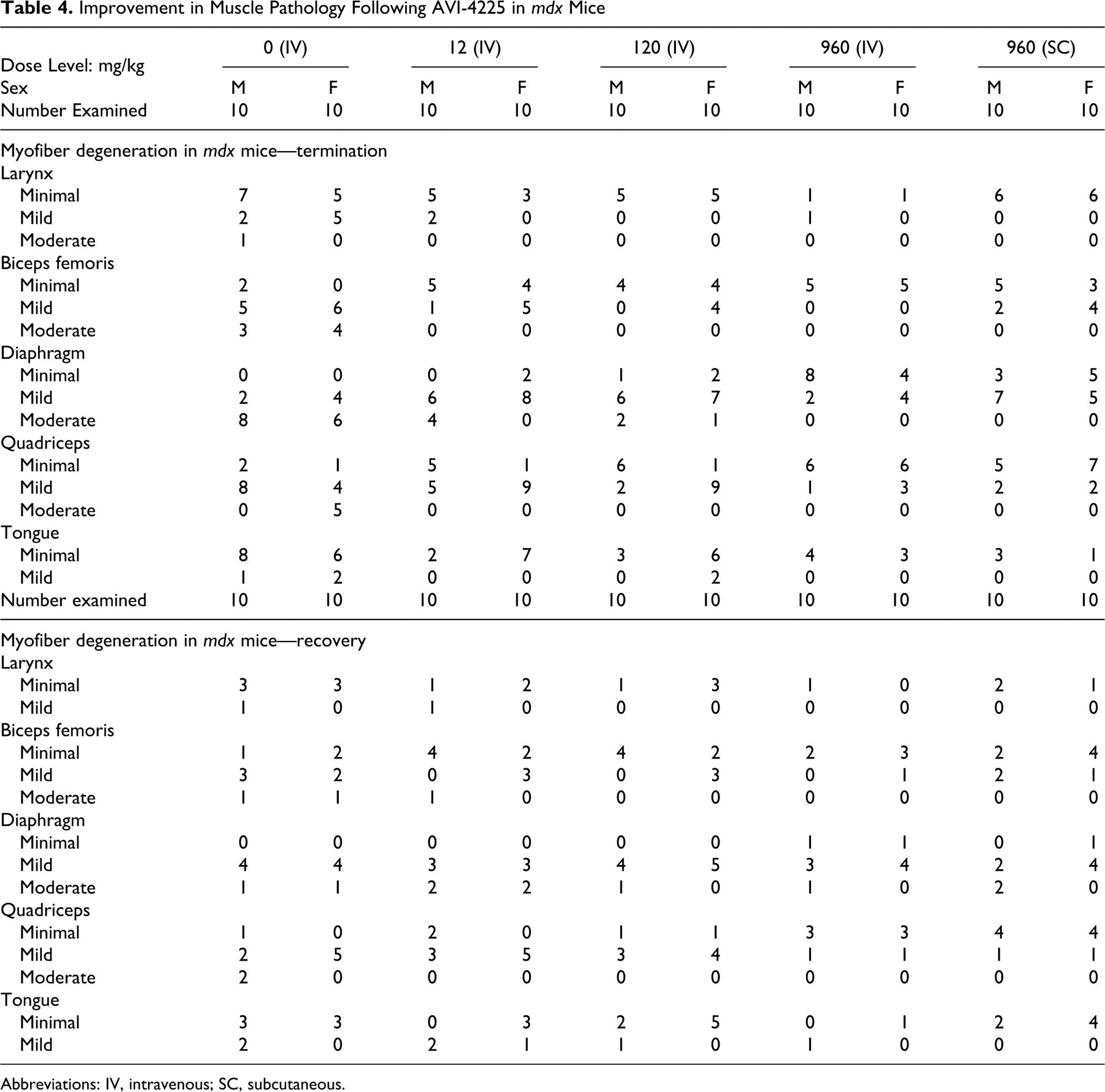
Abbreviations: IV, intravenous; SC, subcutaneous.
For mdx and C57 mice treated with AVI-4658 intravenously, treatment-related renal histopathological changes were similar to those for AVI-4225 and limited to minimal tubular cytoplasmic basophilia and tubular vacuolation at 120 and 960 mg/kg after 12 once-weekly doses (Table 5). Renal tubular basophilia consisted of tubules with epithelial cells having a bluish cast compared to controls, shading upward to tubules with epithelial cells having a distinctly blue cytoplasm, rather than pink. Often, affected cells had a granular appearance to the cytoplasm. Renal tubular vacuolization was seen in some of the basophilic tubules and consisted of variable levels of clear to blue-tinged vacuoles expanding the cytoplasm of affected cells. Vacuoles varied in size from 2-3 μm up to 50 μm or more. Occasionally, vacuoles expanded the cells to the point where the tubule lumen was obscured by large bluish vacuoles. Reversibility of these findings was noted in the 28-day recovery animals, with the complete absence of kidney findings at the 120 mg/kg dose level. In mdx mice administered 960 mg/kg per dose SC, the incidence of basophilic tubules and vacuolation had decreased; in mdx mice the incidence of these findings was reduced from 2/10 and 1/10, to 0/4 and 0/4, respectively. Only in C57 mice dosed at 960 mg/kg per dose (IV), minimal renal tubular degeneration/regeneration at the terminal necropsy was observed (Figure 3) that was fully resolved by the end of the recovery period. Tubular degeneration/regeneration was characterized by tubules having epithelial cells ranging from swollen and hypereosinophilic to shrunken and attenuated. Nuclei in affected cells were oriented toward the luminal side of the cells and often pyknotic or fragmented. Affected tubules had rare dissociated necrotic cells in the lumen. Degenerate tubules often had cytoplasmic basophilia.
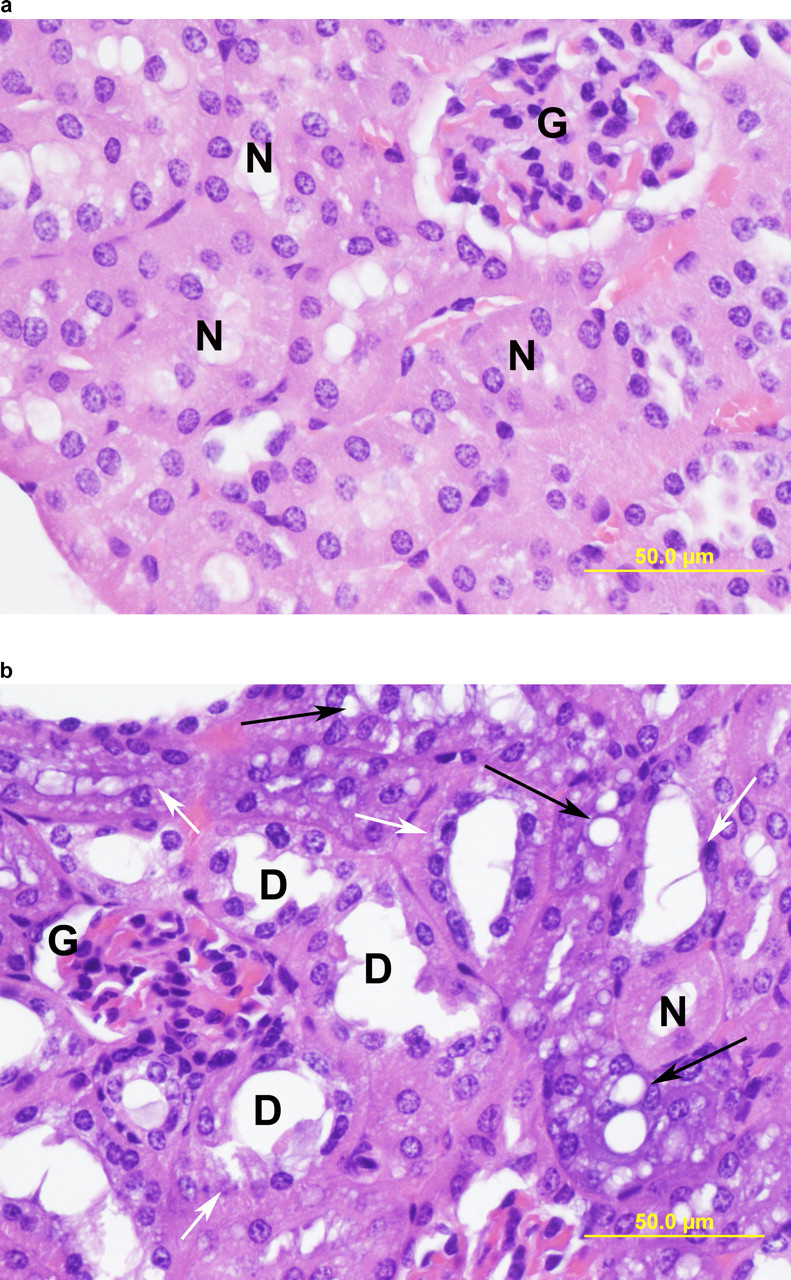
Kidney section at termination from wild-type mouse treated with (A) vehicle control (B) 960 mg/kg intravenous (IV) AVI-4658 stained with hematoxylin and eosin stain. Visible here are tubules with epithelial cells that are attenuated and shrunken, with small, condensed nuclei close to the luminal edge of the cells, consistent with tubular epithelial degeneration. Seen in this image are basophilic tubules, which have abundant cytoplasm that is slightly to deeply basophilic, often with a more granular appearance and having variably sized intracytoplasmic vacuoles (D, degenerate tubule; arrows, basophilic (white) and vacuolated (black) tubule; N, tubule with normal epithelium; G, glomerulus; Original objective lens magnification: ×40).
Microscopic Evaluation Summary of AVI-4658 in Mice at Termination and Recovery
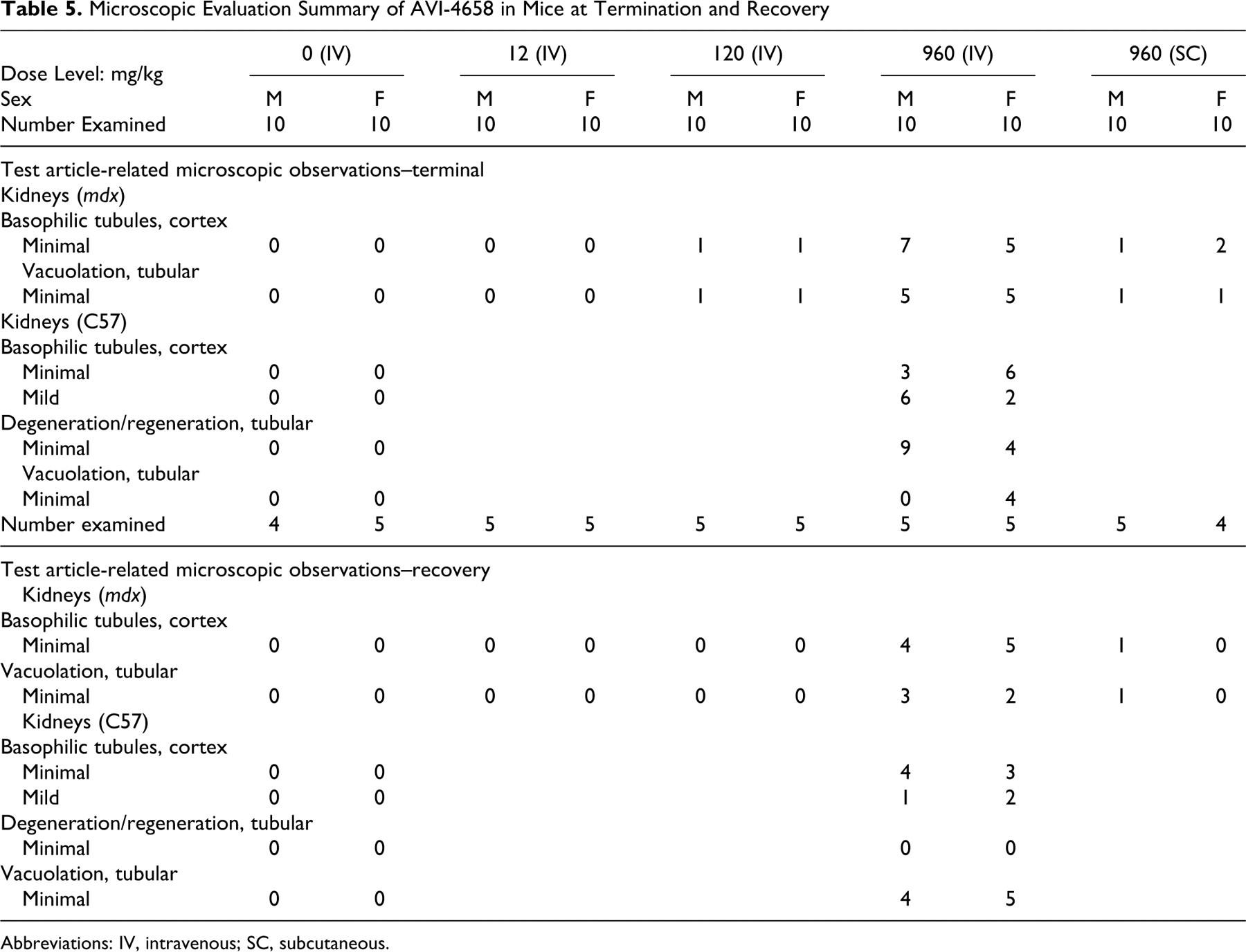
Abbreviations: IV, intravenous; SC, subcutaneous.
There were no treatment-related serum chemistry changes associated with impaired renal function following treatment with AVI-4225 or AVI-4658, as mdx or C57 mice had normal BUN levels at the end of dosing and following the recovery period (see Table 2 and Discussion). Similarly, the specific gravity of urine from female mdx and C57 mice treated with AVI-4658 at termination and recovery showed no drug-related changes (data not shown), suggesting a lack of significant renal impairment.
Discussion
In the current study, AVI-4225 and AVI-4658 were evaluated in dystrophic mdx or C57 mice with weekly IV or SC injections for 12 weeks, followed by a 4-week recovery period, at doses up to 960 mg/kg per injection. These studies were designed to support the conduct of clinical trials similar to the above-mentioned Phase 1b/2 clinical trial, which also employ a once-weekly for 12 weeks induction regimen.
Following 12 once-weekly IV or SC doses of up to 960 mg/kg, there were no test article-related effects on survival, clinical observations, body weight changes, food consumption, ophthalmoscopy, hematology, clinical chemistry, urinalysis, organ weights, or macroscopic evaluations. Dystrophic mdx and C57 mice administered AVI-4225 displayed similar responses, that is, no significant changes, in renal serum markers as with the human-sequence specific PMO AVI-4658. Basophilic tubules and granules were reduced or not present, respectively, in mdx mice at the end of recovery period while in C57 mice there was no reduction over the 4-week recovery period in the frequency of these observations. The reduced severity and incidence of basophilic tubules and granules in mdx mice, compared to C57 mice, at both the end of the dosing period and the end of the recovery period suggests that both AVI-4225 and AVI-4658 have decreased renal effects in the mdx mouse, compared to C57 mice. The lack of associated renal clinical pathology findings, coupled with the evidence of a trend toward reversibility and the relatively low level of severity of the findings, suggests that these changes should not be considered adverse within the scope of this study (see below). 22
Drug-related effects in mdx or C57 mice were limited to minimal-to-mild dose-dependent but reversing microscopic kidney findings which included basophilic granules in proximal tubule epithelium, and minimal and reversible or reversing tubular degeneration/regeneration and tubular vacuolation, respectively. For AVI-4225 and AVI-4658, the basophilic granules are likely a consequence of accumulation of the PMOs in the kidney through uptake by the proximal tubular epithelium, as renal clearance is the primary elimination route for these drugs. The appearance of basophilic granules is a typical histologic change reported with RNA-based oligomers as they are evidence of endosomal or phagolysosomal accumulation and compartmentalization of oligos that is not considered to be toxicologically significant. 23 –25 For phosphorothioate oligonucleotides, an older, polyanionic class of oligonucleotide drug, Henry et al described the appearance of basophilic granules in renal proximal tubular epithelium and liver-associated Kupffer cells. 24 It was found that for phosphorothioates, accumulation in the liver was also substantial, and the material in the basophilic granules was confirmed to be oligonucleotide by sequence-specific fluorescence in situ hybridization techniques. 26,27 We are currently developing similar methods to determine whether the basophilic granules observed in the kidneys of treated mice consists of the PMO compounds.
The histological findings in the kidney, including the minimal, fully reversible tubular degeneration/regeneration noted following the high dose AVI-4658 of 960 mg/kg per week in C57 wild-type mice, were not accompanied by changes in BUN or serum creatinine levels in any case. These data suggest that the degree and nature of the renal pathology was not severe enough to change these relatively insensitive markers of renal injury. Generally, a 50% to 70% decrease in the glomerular filtration rate (GFR) must occur before elevations in serum creatinine and BUN are observed, thus a substantial amount of renal damage may occur without detection by these means. 28 There were no treatment-related changes in urinary specific gravity in mdx and C57 mice administered AVI-4658 at termination and recovery suggesting that the animal’s capacity for concentrating urine has not been compromised. Additionally, there were no dose-related trends in urinary creatinine concentrations in mdx and C57 mice treated with AVI-4658 at both recovery and at termination. While comprehensive urinalyses were scheduled for these studies, limited urine volumes restricted the use of this invaluable source for biomarkers of renal functionality.
The need for a more robust set of renal biomarkers, that may be more reflective of the kidney changes seen in this study, that are both sensitive and specific, has been recognized by the Critical Path Institute’s Predictive Safety Testing Consortium initiative, and has led to the recommendation for 7 new urinary protein biomarkers. Both the FDA and EMA will now accept data generated with these new biomarkers as additional and complementary information to the currently available standards. 29 Seven urinary biomarkers: KIM-1, albumin, total protein, β2-microglobulin, cystatin C, clusterin, and trefoil factor-3 were selected based on their sensitivity and their ability to phenotypically anchor renal damage to specific regions of the kidney. While well characterized in rats, and gaining acceptance in preclinical studies using other species, their usefulness in predicting renal injury in mouse studies may be limited due to the inherent low volumes of urine produced by these animals.
No toxicity associated with the mechanism of action of AVI-4225, that is, skipping murine dystrophin exon 23, was observed in these studies as there were no observed toxicities specific to muscles in mdx mice that had restored dystrophin protein. While confirmatory molecular markers of exon skipping and dystrophin restoration were not performed in these studies, histopathology observations trending toward improvements in the dystrophic state of muscle tissue in mdx mice following only AVI-4225, but not AVI-4658, treatment, is suggestive of myelopathy reversal of the dystrophic-specific lesion. Compared to C57 mice, mdx mice normally have relatively high serum concentrations for AST, AP, and ALT as this phenotype reflects, in part, ongoing myofiber degeneration. 30
Dose-related decreases in ALT and AP following AVI-4225 treatment at termination reflects improvement in muscle pathology. Note that elevated serum ALT and AP are typically associated with liver dysfunction and in the case of AP, contributions to circulating serum levels can be from isozymes that originate from the intestine, bone, heart, kidney, and skeletal muscles. 31,32 These serum enzymes are elevated for cases of severe muscle damage, without accompanying hepatic pathology, such as DMD patients and mdx mice and have been observed in regenerating muscle fibers. 33 –35 For example, elevated AP-staining muscle fibers containing few AP-staining cells were identified in muscle biopsy samples from patients with DMD when contrasted to comparable samples from normal controls. 35 The degree of myofiber degeneration reversal was slightly greater following IV versus SC administration of AVI-4225, in the larynx and the biceps femoris while in the diaphragm, quadriceps, and tongue, the myofiber degeneration was similar for both routes. These data suggest that IV bolus administration may be more efficacious than SC; however, further study is required to determine the specific reason. Additionally, the overall incidence of basophilic granules and vacuolization in the proximal tubular epithelia of mdx mice at recovery (Table 3) was less than was observed in the C57 mice at the same dose and time point, suggesting that AVI-4225 treatment in mdx mice may have less effect on the kidney. Therefore, dystrophin expression restoration does not interfere with processes involved with renal lesion reversal produced after dosing.
Footnotes
Acknowledgments
We would like to thank Study Director W. Maier of MPI Research, Inc. and staff for their efforts in performance of these repeat dose studies of AVI-4225 and AVI-4658 in mice. We would also like to thank R Kole and J Schuh for critical evaluation of this manuscript.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: A research grant W81XWH-09-1-0215 (PI: Eric P Hoffman) from the DOD/USAMRAA and by grants from Cure Duchenne and Federation to Eradicate Duchenne.
