Abstract
Cisplatin (Cisp) is one of the most widely used chemotherapeutic agents for the treatment of several human malignancies. The efficacy of Cisp is dose dependent and at higher doses serious kidney injury may occur. Recombinant human erythropoietin (rhEPO) has recently been shown to exert an important cytoprotective effect in experimental brain injury and ischemic acute renal failure. The aim of the present study was to explore whether rhEPO administration is protective against Cisp-induced oxidative damage and renal injury. Our results showed that Cisp induced a marked oxidative stress and renal failure. Administration of rhEPO (pre-, co- or postadministration with regard to Cisp) decreased oxidative damage induced by Cisp. Recombinant human EPO reduced malondialdehyde and protein carbonyl levels. Recombinant human EPO also prevented glutathione depletion and ameliorated the increased catalase activity induced by Cisp treatment. Furthermore, rhEPO restored creatinine and blood urea nitrogen levels increased by Cisp. We concluded that rhEPO administration especially in pretreatment condition protected rats against Cisp-induced renal oxidative stress and nephrotoxicity.
Introduction
While Cisplatin (Cisp) is one of the most widely used anticancer drugs effective in treating a range of neoplastic diseases, several toxic effects of this drug are known in humans and in animals. Cisplatin is associated with adverse drug reactions including renal damage and gastrointestinal dysfunction. 1 Major side effects of Cisp include nephrotoxicity, leading to acute kidney injury and renal failure.2,3 However, it is unclear how the signals are integrated to determine tubular cell injury and death.2,3 Several lines of evidence have demonstrated that Cisp is able to cause oxidative damage to macromolecules such as lipids and proteins, probably resulting in normal cell death.4–6 Furthermore, it has been suggested that reactive oxygen species (ROS) are implicated in the pathogenesis of Cisp-induced renal tubular injury.7,8 Many compounds, such as a variety of ROS scavengers, have been suggested to protect against Cisp-induced oxidative damage in kidney.9–11
Erythropoietin (EPO) is a glycoprotein hormone primarily, but not exclusively, synthesized by renal cortical interstitial fibroblasts in response to tissue hypoxia. 12 Erythropoietin, which is used clinically as recombinant human EPO (rhEPO), has been used as a successful treatment of human anemia associated with end-stage renal failure and cancer chemotherapy.13–15 More recently, the biological effects of rhEPO are not limited to the hematopoietic system; many studies have shown that rhEPO is a pleiotropic cytokine that exerts broad tissue-protective effects in diverse nonhematopoietic organs. 16 Several investigations have shown that rhEPO can reduce the renal dysfunction and injury caused by oxidative stress.17,18
In this context, to highlight the protective effect of rhEPO, we evaluated the possible antioxidant properties of rhEPO against Cisp-induced oxidative damage and the eventual nephroprotective effect of rhEPO against Cisp nephrotoxicity. For this purpose, we measured the renal tissue levels of malondialdehyde (MDA), protein carbonyl, reduced glutathione (GSH), and catalase activity in rats treated with Cisp and rhEPO in different treatment conditions. Nephrotoxicity was evaluated by the measure of creatinine and blood urea nitrogen (BUN) levels.
Materials and Methods
Chemicals
Cisplatin (cis-diamminedichloroplatinum II) was purchased from Sigma-Aldrich, France. Experiments were performed with a commercially available preparation of rhEPO (Hemax, Bio SIDUS SA, Argentina). Triobarbituric acid (TBA); trichloroacetic acid (TCA), and butul hydoxyl toluene (BHT) were obtained from Sigma Chemical Co. (St. Louis, Missouri); 2,4-dinitro-phenylhydrazine (2,4-DNPH) and guanidine were from Prolabo (France). All other chemicals used were of analytical grade.
Animal Treatment
Experiments were performed on male Wistar rats in the weight range of 120 to 140 g, which were kept at controlled environmental conditions at room temperature 22°C ± 2°C and 12-hour light/dark cycles and allowed free access to food and water but fasted overnight before treatment. For the time course experiment, rats were divided at random into 6 groups, 6 animals in each group. All injections were administered by the intraperitoneal (ip) route. The control group received a single injection of saline solution 0.9%. In each type of treatment, rhEPO and Cisp were used, respectively, at 3000 IU/kg body weight (bw) and 6 mg/kg bw. The rhEPO group was given only rhEPO and the Cisp group was given only a single injection of Cisp. To test the effect of rhEPO on Cisp-induced oxidative damage and nephrotoxicity, 3 treatment conditions were performed. In the cotreatment group, a single dose of rhEPO was administered simultaneously with Cisp. In pretreatment group, a single dose of rhEPO was given 1 day before Cisp. In the posttreatment group, a single dose of rhEPO was given 5 days after Cisp. Experimental design was detailed in Table 1. After animal sacrifice, kidneys were immediately removed and serum was collected for subsequent experiments.
Animal Groups and Treatments in the Experimental Design of This Study a
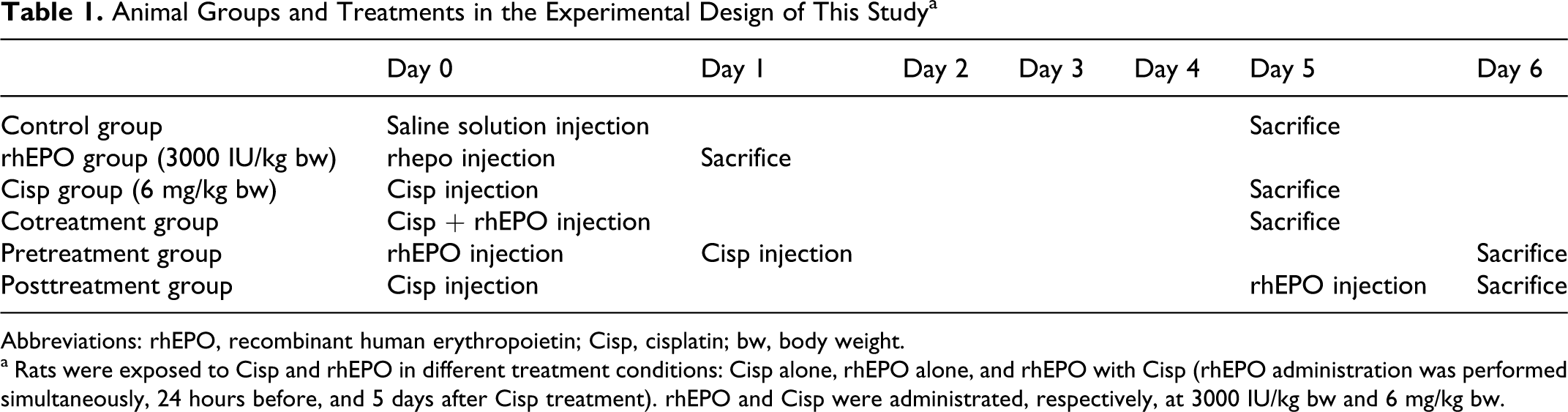
Abbreviations: rhEPO, recombinant human erythropoietin; Cisp, cisplatin; bw, body weight.
a Rats were exposed to Cisp and rhEPO in different treatment conditions: Cisp alone, rhEPO alone, and rhEPO with Cisp (rhEPO administration was performed simultaneously, 24 hours before, and 5 days after Cisp treatment). rhEPO and Cisp were administrated, respectively, at 3000 IU/kg bw and 6 mg/kg bw.
Preparation of Kidney Extracts
Kidneys were with a homogenizer in the presence of 10 mmol/L Tris-HCl, pH 7.4 at 4°C and centrifuged at 4000 rpm for 30 minutes at 4°C. The supernatant was collected for analysis and the protein concentrations were determined in kidney extract using Protein BioRad assay. 19
Evaluation of Lipid Peroxidation Level
The renal MDA levels in the extract were quantified according to the method of Aust et al. 20 Briefly, 200 µL of kidney extract were mixed with 150 µL of Tris-buffered saline ([TBS] Tris 50 mmol/L and NaCl 150 mmol/L, pH 7.4) and 250 µL TCA-BHT (20% TCA and 1% BHT). The mixture was vigorously vortexed and centrifuged at 1500g for 10 minutes; 400 µL of the supernatant were added with HCl 0.6 N and 320 µL Tris-TBA (Tris 26 mmol/L and TBA 120 mmol/L), the content was mixed and incubated for 10 minutes at 80°C. The absorbance was measured at 535 nm. The optic density corresponding to the complex formed with the TBA-MDA is proportional to the concentration of MDA and to the lipid peroxide. The concentration of µmol of MDA/mg of proteins is calculated from the absorbance at 530 nm using the molar extinction coefficient of MDA 1.56 × 10 5 /mol/L per cm. The results were expressed as µmol of MDA/mg of proteins.
Protein Carbonyl Assay
Protein carbonyl content was determined as described by Mercier et al 21 in kidney homogenates by measuring the reactivity of carbonyl groups with 2,4-DNPH. Thus, 200 µL of supernatant of kidney was placed in 2 glass tubes; 800 µL of 10 mmol/L DNPH in 2.5 mol/L HCl was added. Tubes were left for 1 hour incubation at room temperature in the dark. Samples were vortexed every 15 minutes. Then 1 mL of 20% TCA was added to samples, and the tubes were left on ice for 10 minutes and centrifuged for 5 minutes at 4000 rpm to collect the protein precipitates and the supernatants were discarded. Next, another wash was performed using 1 mL of 10% TCA, and protein pellets were broken mechanically with the aid of a glass rod. Finally, the pellets were washed with 1 mL of ethanolethyl acetate (1:1, v/v) to remove the free DNPH. The final pellets were dissolved in 500 µL of guanidine hydrochloride 6 mol/L and were left for 10 minutes at 37°C with general vortex mixing. Any insoluble materials were removed by additional centrifugation. Protein carbonyls concentration was determined from the absorbance at 370 nm, applying the molar extinction coefficient of 22.0/mmol/Lper cm. A range of nmoles of carbonyls per milliliter was usually obtained for most proteins and was related to the protein content in the pellets. The results were expressed as nmol of protein carbonyl/mg of proteins.
Glutathione Assay
The renal GSH level was measured using colorimetric assay kit according to the recommendations of the manufacturer (Sigma). The method was based on the reduction of 5,5-dithiobis-2-nitrobenzoic acid with GSH to produce a yellow colored product. The reduced compound directly proportional to GSH concentration and its absorbance22,23 was measured at 412 nm. The kidney tissue of different groups was flash frozen and grounded in liquid nitrogen immediately after excision. A total of 0.3 g of the kidney powder was taken and mixed with the 5% 5-sulfosalicylic acid solution for renal protein removal. Aliquots of kidney extract were submitted to photometric GSH determination; DTNB formation rate was monitored at 412 nm and compared with GSH standards (1-20 µmol/L). Each experiment was performed in triplicate. The results were expressed as nmol of GSH/g of samples.
Determination of Catalase Activity
Catalase activity was measured in the kidney extract at 240 nm, 25°C according to Clairbone. 24 Briefly, 20 µL of the extracts was added in the quartz cuvette containing 780 µL phosphate buffer (pH = 7) and 200 µL of H2O2 0.5 mol/L. The activity of catalase was calculated using the molar extinction coefficient (0.04/mmol/L per cm). The results were expressed as µmol of H2O2/min per mg of proteins.
Estimation of Plasma Creatinine
After sacrifice, blood samples were collected for determination of creatinine. Plasma creatinine concentration was measured by the picric acid colorimetric method. 25 A picric acid solution was made by a 10× dilution of saturated picric acid with 1% NaOH. Plasma (50 µL) was added to picric acid-NaOH solution (200 µL) in a microtiter plate. The samples were incubated 15 minutes before being read at 490 nm in a microplate reader. A standard curve was prepared on the same assay plate to derive the creatinine concentration. The results were expressed as µmol of creatinine/L of serum.
Determination of BUN
Blood urea nitrogen was measured using colorimetric assay kit according to the manufacturer’s instructions (Stanbio Laboratory, Boerne, USA). Urea in the sample was hydrolyzed by the enzyme urease to yield ammonia and carbon dioxide. Then the ammonium ions react with a mixture of salicylate, sodium nitroprusside, and hypochlorite to yield a blue-green chromophore. The absorbance of this chromophore at 600 nm is proportional to urea concentration in the sample. The results were expressed as mmol of BUN/L of serum.
Statistical Analysis
Each experiment was carried out at least 3 times separately. All data were expressed as mean ± SD. Statistical significance of differences among different groups was evaluated by 1-way analysis of variance (ANOVA) followed by Fisher multiple comparisons test as a post hoc test. Value of P < .05 was considered significant.
Results
Induction of Lipid Peroxidation
To evaluate lipid peroxidation status, MDA level was measured and the results are shown in Figure 1. Malondialdehyde is the end product of the major reactions, leading to significant oxidation of polyunsaturated fatty acids in cellular membranes and thus serves as a reliable marker of oxidative stress.26–28 When compared to control groups, MDA level in the kidney was significantly higher (P < .05) in the group treated with Cisp alone at 6 mg/kg bw. Therefore, the MDA level increased from a basal level of 15.5 ± 0.5 µmol/mg of protein to 38.46 ± 0.5 µmol/mg of protein, the increase in MDA levels was about 2.5-fold as compared to the untreated group (P < .05). On the other hand, rhEPO administration at 3000 IU/kg bw, whether simultaneously, before, or after Cisp, was associated with a fall in the MDA levels to reach the control group. Malondialdehyde level was significantly decreased as compared to the Cisp group: 24.14 ± 0.2, 19.10 ± 0.03, and 27.85 ± 0.04 µmol/mg of protein, respectively, in co-, pre-, and posttreatment as compared to 38.46 ± 0.05 µmol/mg of protein, for the Cisp-treated group. Moreover, as shown in Figure 1, the optimum preventive action was shown in groups 5, where the rats were treated with rhEPO 24 hours before Cisp exposure.
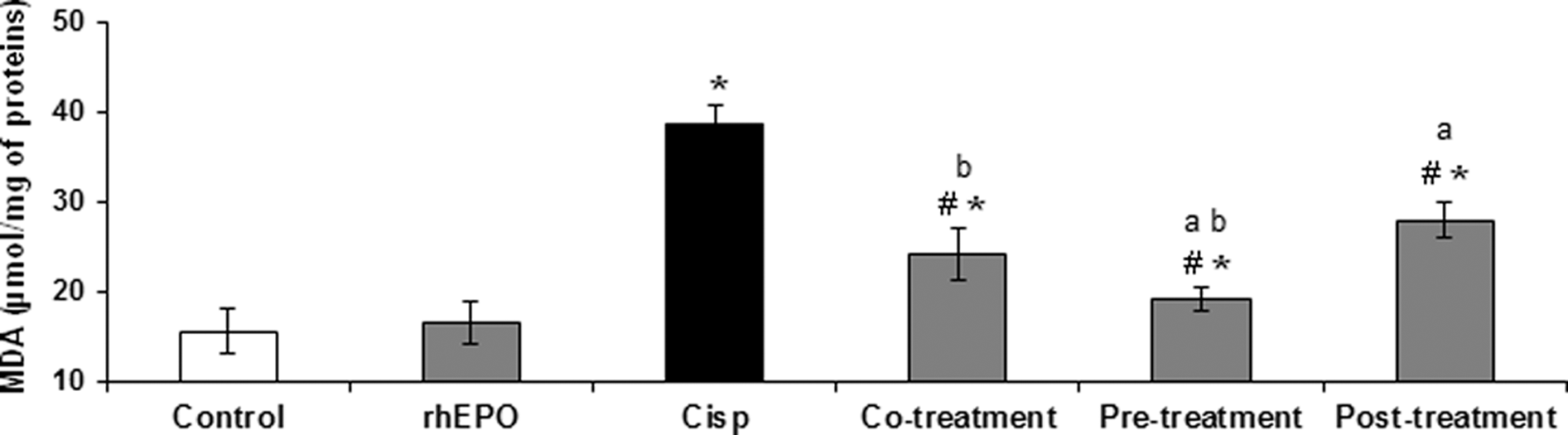
Lipid peroxydation as determined by MDA level in Wistar rat kidney. The MDA level was estimated after a single exposure to Cisp and rhEPO in different treatment conditions: Cisp alone, rhEPO alone, and rhEPO with Cisp (rhEPO administration was performed simultaneously, 24 hours before and 5 days after Cisp treatment). rhEPO (3000 IU/kg body weight, ip), and Cisp (6 mg/kg body weight, ip). Values are expressed as mean ± SD. Superscript characters indicate a significant difference at P ≤ .05 using 1-way ANOVA followed by the Fisher test for multiple comparisons. (*) indicates significant difference from control; (#), significant difference from Cisp-treated rats; (a), significant difference from cotreatment group; (b), significant difference from cotreatment group; MDA, malondialdehyde; Cisp, cisplatin; rhEPO, recombinant human erythropoietin; ip, intraperitoneally; ANOVA, analysis of variance; SD, standard deviation.
Protein Carbonyl Assay
The formation of protein carbonyls, the most widely used marker of severe protein oxidation, was assayed in kidney homogenates and results are illustrated in Figure 2. We showed that Cisp alone generated a significant increase in protein carbonyls formation in kidney as compared to the control group. The protein carbonyl level increased from the basal value of 1.49 ± 0.08 nmol/mg of protein in the control group to 8.67 ± 0.54 nmol/mg of protein in the Cisp-treated group. Recombinant human EPO administration in any treatment conditions (simultaneously, 24 hours before, or 5 days after Cisp exposure) decreased significantly the protein carbonyl formation induced by a fixed dose of Cisp (6 mg/kg bw) as compared to the Cisp-treated group. Thus, protein carbonyl levels decreased from the value of 8.67 ± 0.54 nmol/mg of protein in the Cisp group to 4.29 ± 0.51, 2.73 ± 0.14, and 4.92 ± 0.73 nmol/mg of protein in groups treated with rhEPO, respectively, in co-, pre- and posttreatment. Furthermore, our results clearly demonstrated that pretreatment with rhEPO decreased significantly the protein carbonyl formation as compared to co- and posttreatment conditions. We can deduce that rhEPO was more efficient when it was administrated 24 hours before Cisp exposure.
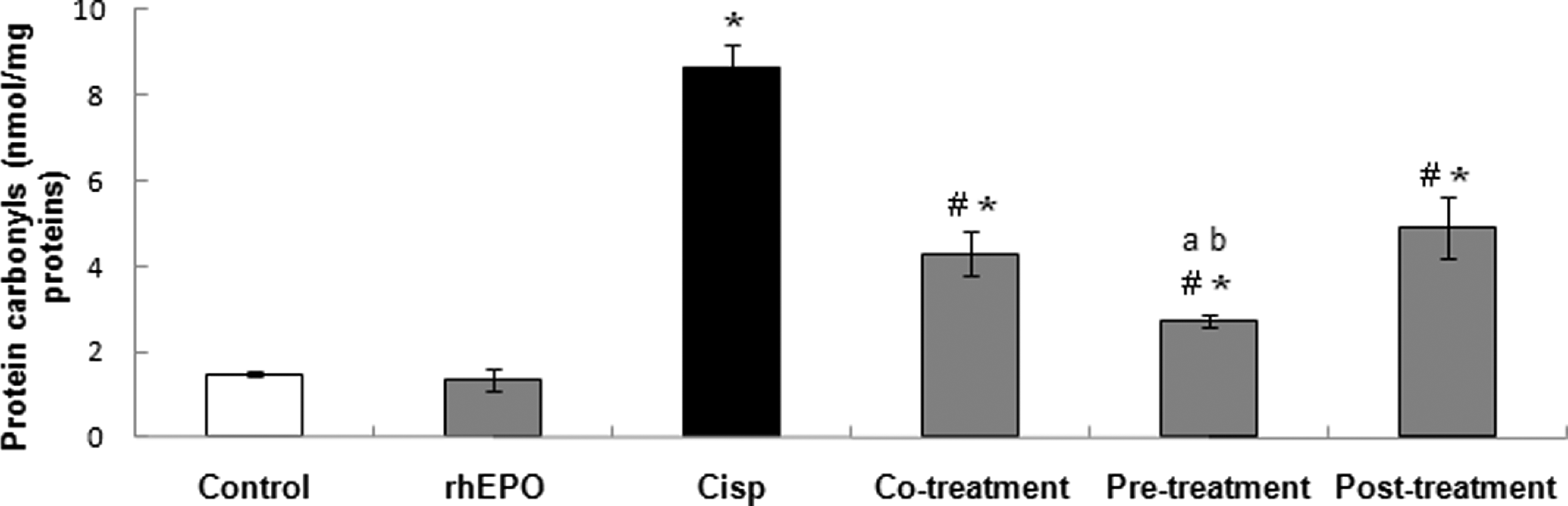
Protein carbonyl concentrations in rat kidney treated with Cisp and rhEPO. rhEPO was administered simultaneously, 24 hours before and 5 days after Cisp treatment. rhEPO (3000 IU/kg body weight, ip) and Cisp (6 mg/kg body weight, ip). Values are expressed as means ± SD. Superscript characters indicate a significant difference at P ≤ .05 using 1-way ANOVA followed by the Fisher test for multiple comparisons. (*) indicates significant difference from control; (#), significant difference from Cisp-treated rats; (a), significant difference from cotreatment group; (b), significant difference from cotreatment group; Cisp, cisplatin; rhEPO, recombinant human erythropoietin; ip, intraperitoneally; ANOVA, analysis of variance; SD, standard deviation.
Glutathione Depletion
The effect of Cisp and rhEPO on GSH modulation in kidney tissue is illustrated in Figure 3. Our data demonstrated that Wistar rats exposed to Cisp alone at 6 mg/kg showed a noticeable depletion of GSH level (P < .05) as compared to the untreated group (18.02 ± 1.95 vs 26.27 ± 2.88 nmoles GSH/g of sample). In group treated with Cisp + rhEPO 24 hours before Cisp exposure, GSH level was equivalent to normal kidney GSH content. This level reached 25.7 ± 1.86 nmoles GSH/g of sample as compared to the control group (26.27 ± 2.88 nmoles GSH/g of sample). However, rhEPO administration in co- and posttreatment conditions does not change significantly the GSH level as compared to the Cisp-treated group.
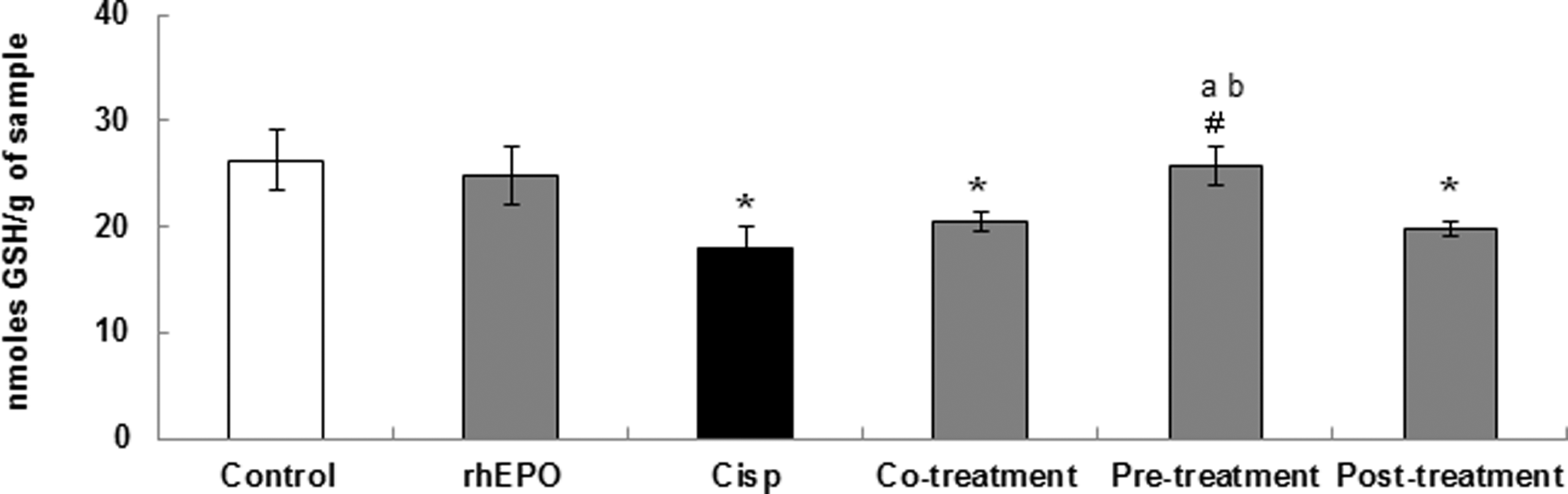
Effect of rhEPO administration on reduced glutathione level in Wistar rat kidney. Different treatment conditions were performed: rhEPO (3000 IU/kg body weight, ip) was administered simultaneously, 24 hours before and 5 days after Cisp (6 mg/kg body weight, ip) exposure. Values are expressed as means ± SD. Superscript characters indicate a significant difference at P ≤ .05 using 1-way ANOVA followed by the Fisher test for multiple comparisons. (*) indicates significant difference from control; (#), significant difference from Cisp-treated rats; (a), significant difference from cotreatment group; (b), significant difference from cotreatment group; Cisp, cisplatin; rhEPO, recombinant human erythropoietin; ip, intraperitoneally; ANOVA, analysis of variance; SD, standard deviation.
Catalase Activity
Catalase is an endogenous antioxidant enzyme that protects the cells from detrimental effects of ROS. The levels of catalase can indicate the magnitude of oxidative stress that occurs during several injuries. The effect of Cisp and rhEPO on catalase activity was illustrated in Figure 4. Our results showed that Cisp alone induced a marked increase in catalase activity in kidney extracts. Catalase activity increased from the basal value of 111.18 ± 12 nmol/min per mg proteins in control group to 650 ± 34.15 nmol/min per mg in the Cisp-treated group. Recombinant human EPO administration simultaneously, 24 hours prior, and 5 days after Cisp treatment prevented the increase in catalase activity. This activity passed from 650 ± 34.15 nmol/min per mg in the Cisp group to 208.20 ± 21.95, 121.88 ± 17.13, and 162.5 ± 15.45 nmol/min/mg in groups treated with Cisp and rhEPO, respectively, in co-, pre- and posttreatment. Thus, the pretreatment condition provided the best protection against Cisp-induced oxidative stress.
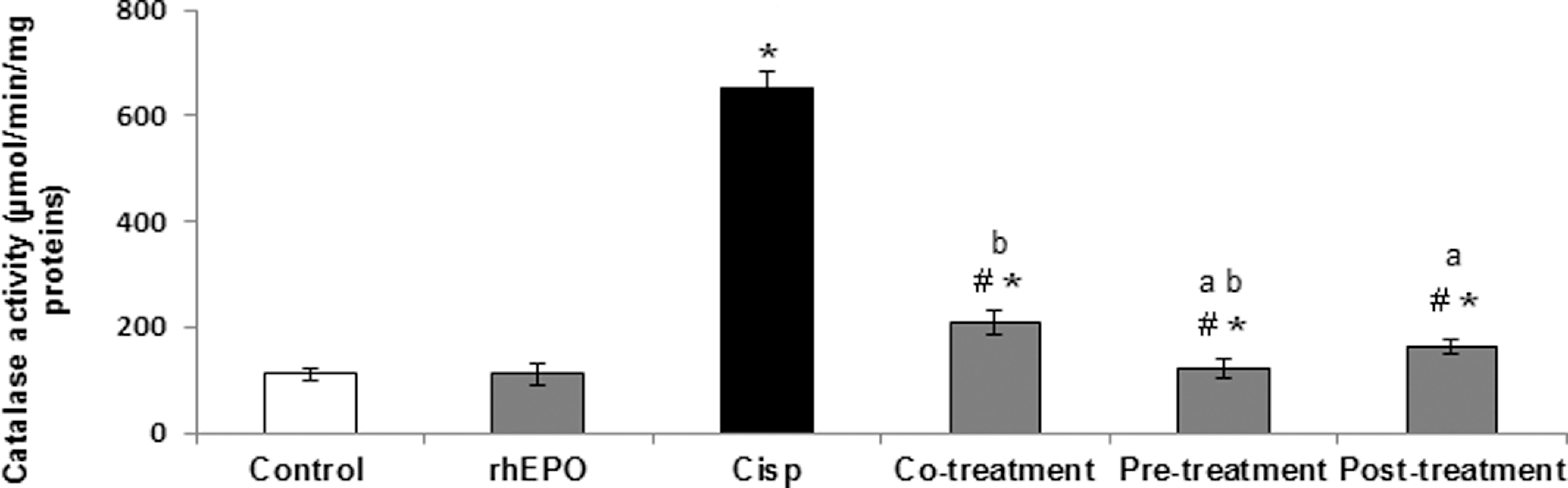
Protective effect of rhEPO on Cisp-induced catalase enzyme activity in rat kidney. rhEPO (3000 IU/kg body weight, ip) was added simultaneously, 24 hours prior, and 5 days after Cisp administration (6 mg/kg body weight, ip). Values are expressed as mean ± SD. Superscript characters indicate a significant difference at P ≤ .05 using 1-way ANOVA followed by the Fisher test for multiple comparisons. (*) indicates significant difference from control; (#), significant difference from Cisp-treated rats; (a), significant difference from cotreatment group; (b), significant difference from cotreatment group; Cisp, cisplatin; rhEPO, recombinant human erythropoietin; ip, intraperitoneally; ANOVA, analysis of variance; SD, standard deviation.
Changes in BUN and Serum Creatinine Levels
To evaluate the nephroprotective effect of rhEPO against Cisp-induced acute renal injury in rat, we evaluated BUN and serum creatinine levels, common indicators of acute renal failure. Our results clearly demonstrated that at posttreatment condition, rats showed a marked deterioration in renal function as reflected by increased concentrations of BUN and serum creatinine as compared to the control group. As shown in Figure 5, BUN level increased from 6.58 ± 1.01 to 154.1 ± 7.53 mmol/L in the Cisp group, and serum creatinine increased from 25.5 ± 2.88 to 376 ± 35.13 µmol/L. Recombinant human EPO treatments (co-, pre-, and posttreatment) protected the increase in BUN and serum creatinine levels caused by Cisp treatment. Our data showed that rhEPO administration 24 hours before Cisp presented the best protection against Cisp nephrotoxicity. In fact, BUN level passed from 154.1 ± 7.53 in the Cisp treatment group to 43.28 ± 10.5 mmol/L in the pretreatment group. Serum creatinine levels decreased from 376 ± 35.13 to 113.6 ± 8.2 µmol/L. Hence, rhEPO exhibited a nephroprotective effect against Cisp nephrotoxicity.
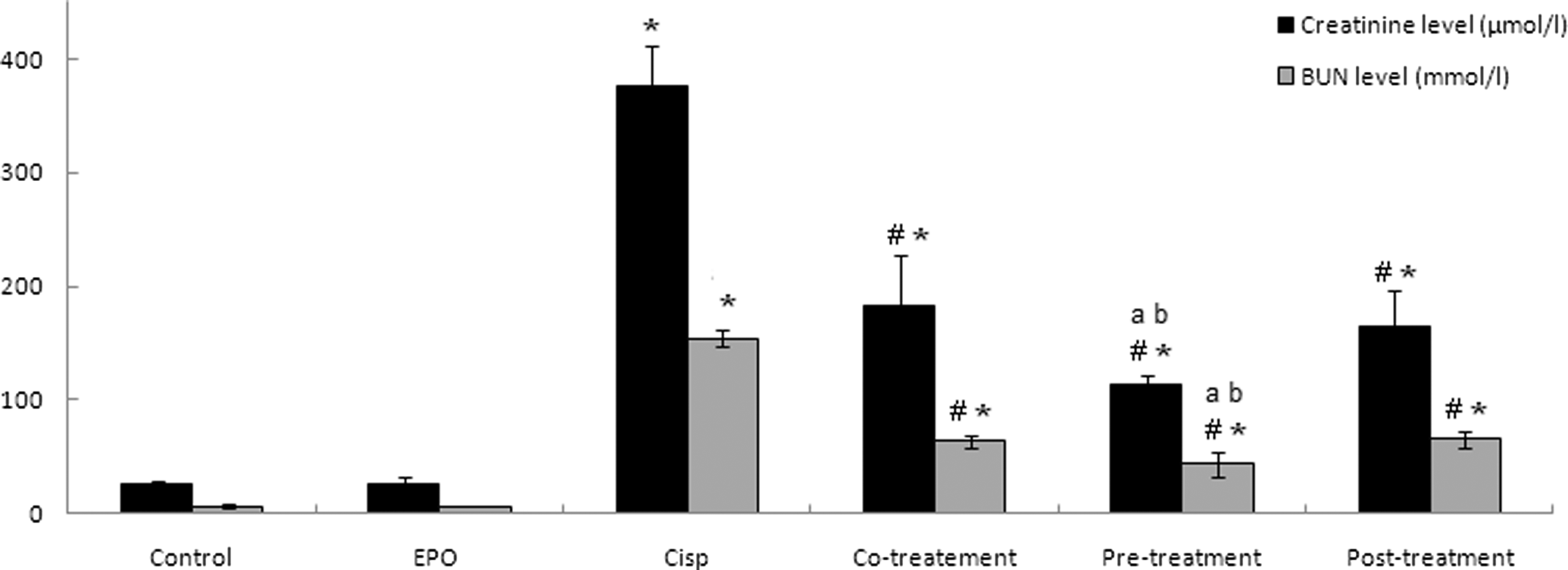
rhEPO ameliorates renal functional impairment following Cisp injury. Wistar rats were treated with rhEPO (3000 IU/kg body weight, ip) simultaneously, 24 hours before and 5 days after Cisp administration (6 mg/kg body weight, ip). Animals were scarified and serum samples were collected for determination of serum creatinine and BUN concentrations. Values are expressed as mean ± SD. Superscript characters indicate a significant difference at P ≤ .05 using 1-way ANOVA followed by the Fisher test for multiple comparisons. (*) indicates significant difference from control; (#), significant difference from Cisp-treated rats; (a), significant difference from cotreatment group; (b), significant difference from cotreatment group; Cisp, cisplatin; rhEPO, recombinant human erythropoietin; ip, intraperitoneally; BUN, blood urea nitrogen; ANOVA, analysis of variance; SD, standard deviation.
Discussion
Cisp is one of the most active cytotoxic agents in the treatment of many kinds of cancer. However, clinical use of this drug usually demolishes the physiological homoeostasis in various organs causing many harmful effects, especially nephrotoxicity. The mechanisms by which Cisp induced nephrotoxicity remain unclear. However, a role for Cisp-induced oxidative stress may provide an attractive hypothesis. 29 In fact, lipid peroxidation and free radical generation in the renal tubular cells have been suggested to be responsible for Cisp-induced renal failure.30–32 In the same way, many antioxidants have been shown to be protective against Cisp-induced nephrotoxicity. In addition, various free radical scavengers have been shown to be effective in protection against Cisp-induced nephrotoxicity, and treatment with such agents provides significant protection against Cisp-induced acute renal failure.30,33,10
Erythropoietin is essential for the survival, proliferation, and differentiation of the erythrocytic progenitors in the bone marrow. Besides, another key physiological role of EPO on general tissue protection was previously confirmed.34–36 Many studies showed that EPO is directly involved in the prevention of oxidative stress with activation of antioxidant enzymes, inhibition of nitric oxide production, and decrease of lipid peroxidation.37,38 Thus, the aim of the present study was to evaluate the involvement of oxidative stress in Cisp-induced nephrotoxicity and to demonstrate a possible protective effect of rhEPO against Cisp deleterious oxidative damage in rat kidney.
To evaluate the oxidative damage, we measured the lipid peroxidation and protein carbonyl content as 2 biomarkers of oxidative stress as well as GSH content and catalase activity, which are considered 2 biomarkers of antioxidant defense. In the present study, while exposure to Cisp (6 mg/kg bw) induced a noticeable increase in MDA formation in kidney rat, rhEPO administration either simultaneously, before, or after Cisp exposure provided a significant reduction of this induction which dropped to the control level (Figure 1). Furthermore, our results showed that rhEPO treatment 24 hours before rat intoxication with Cisp promoted the best protection against the oxidative stress induced by this drug. Cisp induced a marked increase in protein carbonyl generation in kidney extracts that was significantly reduced with rhEPO in the different experimental conditions (co-, pre- and posttreatment; Figure 2). The protective action of rhEPO was significantly better in the pretreatment condition. Glutathione is the most abundant intracellular thiol that plays an important role in the detoxification of ROS and xenobiotics. 39 Our results obviously showed that Cisp induced a significant GSH depletion in rat kidney. The amount of GSH was clearly enhanced in the presence of rhEPO only in the pretreatment condition (Figure 3). Moreover, Cisp enhanced catalase activity and this activation was significantly reduced when animals were treated with rhEPO in the different treatment conditions. We clearly demonstrated that the optimum prevention of rhEPO against Cisp-induced oxidative lesion was observed in the pretreatment condition. Our findings are in accordance with those of Naziroglu et al, 33 who demonstrated that Cisp enhanced MDA level and decreased the amount of GSH in liver and lens of rats. Results from Öztürk et al 40 demonstrated that EPO reduced lipid peroxidation in rat brain injured tissue, but it had no effect on catalase level in rats subjected to closed head injury.
Oxidative stress via ROS has been implicated in renal injury under various pathological conditions.7,8 To highlight the effect of rhEPO on Cisp-induced acute kidney failure, creatinine level and BUN were monitored. Our results clearly demonstrated that the administration of Cisp to rats caused an increase in BUN and in serum creatinine as compared to the control group (Figure 5). These results were in agreement with several studies suggesting that a single injection of Cisp at doses of 5 to 10 mg/kg bw in rats caused a marked reduction in the glomerular filtration rate, accompanied by an increase in the serum creatinine level, thus indicating induction of acute renal failure.41–43 Recombinant human EPO treatment simultaneously, 24 hours prior, or 5 days after Cisp injection to rats resulted in a significant improvement of nephrotoxicity indices as indicated by the significant reduction in BUN and serum creatinine. Furthermore, our results showed that the pretreatment with rhEPO was the most effective in preventing damages caused by Cisp. This is in accord with Esposito et al, 44 who demonstrated that EPO preconditioning was effective to protect kidney against ischemia/reperfusion injury by reducing both tubular cell injury and interstitial infiltration.
According to these data, we can suggest that oxidative stress is the major contributor to Cisp nephrotoxicity and rhEPO treatments can provide a good protective strategy against oxidative stress and renal failure caused by this anticancer drug.
In conclusion, the present study demonstrated that rhEPO provided a significant protective effect against Cisp-induced acute kidney failure by an antioxidant mechanism. It is important to support rhEPO administration for Cisp-treated cancer patients to minimize its nephrotoxicity, which is a major and dose-limiting clinical problem.
Footnotes
Acknowledgment
This research was supported by the Ministère Tunisien de l’Enseignement Supérieur et de la Recherche Scientifique et de la Technologie (Laboratoire de Recherche sur les Substances Biologiquement Compatibles: LRSBC).
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
This research was supported by the Ministère Tunisien de l'Enseignement Supérieur et de la Recherche Scientifique et de la Technologie [Laboratoire de Recherche sur les Substances Biologiquement Compatibles: LRSBC].
Authors declare that this manuscript was consistent with the guidelines and principles of the American College of Toxicology Statement on the Use of Animals in Toxicology.
