Abstract
Although cisplatin is one of the most efficient chemotherapeutic agents for the treatment of solid tumors, frequently observed nephrotoxicity has limited its use in several patients. Materials and methods: The protective effect of Glycine max (GM) and Chrysanthemum indicum (CM) extracts on cisplatin-induced apoptosis in human proximal tubular HK-2 cells was evaluated by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), Hoechst 33342, and propidium iodide assays. Reactive oxygen species (ROS) were determined by flow cytometry with 2,7-dichlorofluorescein diacetate (DCFH2-DA). Results: Cisplatin-induced renal cell toxicity through the induction of hydrogen peroxide (H2O2•−) and hydroxyl radical (OH). CM extract protected cisplatin-induced apoptosis by its anti-oxidant activity against H2O2 and OH•−, while GM extract scavenged only H2O2. Furthermore, GM and CM extracts protect renal cells without significant interfering effect on cisplatin toxicity in lung cancer H460 and melanoma G361 cells. Conclusion: GM and CM extracts exhibited a promising protective effect on cisplatin-induced nephrotoxicity which could benefit the development for nephroprotective approaches.
Introduction
Despite novel effective strategies in protection of chemotherapy-induced normal cell damage, chemotherapeutic toxicity is a major limiting factor in the usage of many anti-cancer agents. Among various anti-cancer agents, cis-diamminedichloroplatinum (II) (cisplatin) is one of the most frequently perscribed drugs and has been reported for its high efficiency against various solid tumors including ovarian, head and neck, testicular, and lung cancers. 1 Nephrotoxicity due to cisplatin-induced epithelial cell apoptosis in an area of S3 segment of proximal tubule has limits in the dose and usage of this agent.2–6 Many strategies have been proposed for protection of cisplatin-mediated renal damage including forced diuresis and additive drugs; however, the protective efficiency is still far from satisfactory. Due to the fact that cisplatin is able to elevate the intracellular reactive oxygen species (ROS) level in many cells,7,8 such ROS up-regulation has been shown to be a vital event participating in cisplatin-induced various cell damages.9–12 This concept led to introducing of cellular anti-oxidant likes reduced glutathione (GSH) together with cisplatin treatment in order to reduce nephrotoxicity.13,14 However, the results that GSH protected both normal and cancerous cells from cisplatin-induced cell death had attenuated the further development of this agent.15,16 So far, no substance has been found to possess a renoprotective effect, well-tolerated by the patients and not interfering with anti-cancer action of cisplatin.
A large number of natural products possessing anti-oxidant property are the promising sources of additive treatment that may improve cisplatin intolerance.17–19 Among them, extract of Glycine max (GM) contains high protein and isoflavone content which have been shown to benefit patients with cardiovascular diseases, hyperlipidemia, osteoporosis, and cancers.20–23 Likewise, Chrysanthemum extract, an extract from Chrysanthemum indicum (CM), has been long used as a traditional medicine. Its pharmacological properties have been continuously reported such as anti-oxidant, anti-tumor, anti-inflammation, and neuroprotective effects.24–27 Since both plant extracts not only have anti-oxidant effects but also benefit cancer patients in many ways, and the evidence regarding renoprotective property of these extracts on cisplatin-induced renal cell damage is still unclear, the present study evaluated GM and CM extracts for their potential to be developed for further use as a nephroprotective agent.
Materials and methods
Cells culture
Human proximal tubular epithelial HK-2, human lung cancer epithelial H460, and human melanoma G361 cells were obtained from the American Type Culture Collection (ATCC, Manassas, VA, USA). HK-2 cells were cultured in DMEM medium, while H460 and G361 cells were cultured in RPMI 1640 medium in a 5% CO2 environment at 37°C. All media were supplemented with 10% fetal bovine serum (FBS), 2 mM
Chemicals
GM extract was obtained from Xi’an Huarui Bio-Engineering Co., Ltd. (Shaanxi, China). CM extract was obtained from Shanghai Leasun Chemical Co., Ltd. (Shanghai, China). N-acetylcystein (NAC), reduced glutathione, cisplatin, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), 2,7-dichlorofluorescein diacetate (DCFH2-DA), Hoechst 33342, propidium iodide (PI), deferoxamine (DFO), and catalase (CAT) were obtained from Sigma Chemical, Inc. (St. Louis, MO, USA). Mn(III)tetrakis(4-benzoic acid) porphyrin chloride (MnTBAP) was obtained from Calbiochem (San Diego, CA, USA).
Cytotoxicity assay
HK-2, H460, and G361 cells were treated with cisplatin in the presence or absence of NAC, GSH, GM, or CM as previously described,12,28 and cell viability was determined by MTT assay. Briefly, cells in 96-well plates were incubated with 500 μg/ml of MTT for 4 h at 37°C. The intensity of formazan product was measured at 570 nm using a microplate reader. All analyses were performed in at least three independent replicate cultures. Absorbance ratio of treated to non-treated control cells was calculated and presented as relative cell viability.
Apoptosis and necrosis assay
HK-2, H460, and G361 cells were treated with cisplatin in the presence or absence of NAC, GSH, GM, or CM, and apoptotic and necrosis cell death was determined by Hoechst 33342 and propidium iodide (PI) co-staining. After specific treatments, cells were incubated with 10 μM of the Hoechst and 5 μg/ml PI dye for 30 min at 37°C. The apoptotic cells having condensed chromatin and/or fragmented nuclei and PI-positive necrotic cells were visualized and scored under a fluorescence microscope (Olympus IX51 with DP70).
ROS detection
Cells were pretreated with NAC, GSH, GM, CM, or PBS and treated with 50 μM of cisplatin. Cellular ROS was then determined by flow cytometry and fluorescence microscope using DCFH2-DA as a fluorescent probe. Cells were incubated with DCFH2-DA (15 μM) for 30 min at 37°C, after which they were washed and re-suspended in phosphate-buffered saline (PBS) and analyzed for fluorescence intensity using a 485 nm excitation beam and a 538-nm band-pass filter (FACSort, Becton Dickinson, Rutherford, NJ, USA). The mean fluorescence intensity was quantified by CellQuest software (Becton Dickinson) analysis of the recorded histograms.
Statistical analysis
Data were expressed as the means ± SD from three or more independent experiments. Statistical analysis was performed by Student’s t test at a significance level of p < 0.05.
Results
Cisplatin induces human renal epithelial cell apoptosis
Because a number of studies have found that cisplatin-induced nephrotoxicity was mainly observed to cause significant damage of proximal tubular cells,4–6 we used human kidney proximal tubular HK-2 cells as a cell model. Cisplatin-induced cell death was characterized by leaving the cells untreated or treated with various concentrations of cisplatin (5–100 μM) for 24 h and cell survival was analyzed by MTT assay. Figure 1a shows that cisplatin treatment caused a dose-dependent decrease in cell viability with approximately 60% and 40% of the cells remaining viable at cisplatin concentrations of 50 µM and 100 µM, respectively. Since apoptosis and necrosis have been shown to be two major mechanisms of cell death, we further identified modes of cell death in response to cisplatin treatments. Hoechst 33342 assay was used for apoptosis detection, and propidium iodide staining assay was performed for necrosis detection. Analysis of cell death in Figure 1b further shows that the decrease in cell survival presenting in Figure 1a was mainly due to apoptosis, as indicated by the increase of cells with intense nuclear fluorescence and DNA condensation in response to cisplatin treatment, whereas cisplatin treatment resulted in necrotic cell death in very less quantity as indicated by the cells exhibiting red fluorescence staining.

Cytotoxic effect of cisplatin on HK-2 cells. (a) Cells were treated with various concentrations of cisplatin (0–100 µM) for 24 h and cell viability was determined by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. Values are means on triplicate samples ± S.D. *p < 0.01 versus non-treated control. (b) 50 µM cisplatin-induced apoptosis in HK-2 cells. Apoptotic cells were detected by Hoechst 33342 and propidium iodide co-staining. Representative photographs are shown from three independent experiments.
Cisplatin induces cell death via reactive oxygen species-dependent mechanism
The current study and others have shown that ROS induced by cisplatin are responsible for cisplatin-induced cell death in many cell systems.7–11 However, whether or not cisplatin-induced ROS could play a key role in renal cell damage remains unclear. The present study evaluated the effect of known anti-oxidants NAC and GSH on cisplatin-induced renal cell damage. Cells were incubated with NAC, GSH, or PBS for 1 h and treated with 50 µM of cisplatin. Then cell viability, cell apoptosis, and cell necrosis were analyzed after 24-h incubation period. Figure 2a shows that cisplatin treatment alone caused approximately 40% reduction of cell survival, and pretreatment of the cells with either NAC or GSH dramatically abolished the toxic effect of cisplatin. Hoechst 33342 and PI assays in Figure 2b further revealed that both apoptotic and necrotic cell deaths induced by cisplatin treatment were significantly decreased in anti-oxidants pretreated cells. To provide supporting evidence for the correlation of ROS and cisplatin-induced cytotoxicity, intracellular ROS level in response to cisplatin treatment was evaluated by ROS-specific fluorescent dye, DCFH2-DA. After treating the cells with 50 µM of cisplatin in the presence or absence of anti-oxidants, cells were collected, incubated with DCFH2-DA, and cellular ROS levels were determined by flow cytometry and fluorescence microscopy. Figure 3 shows that cisplatin significantly increased intracellular ROS level in HK-2 cells. NAC and GSH were able to decrease intracellular ROS level. These results indicate the role of ROS as a key regulator of cisplatin-induced renal cell damage.
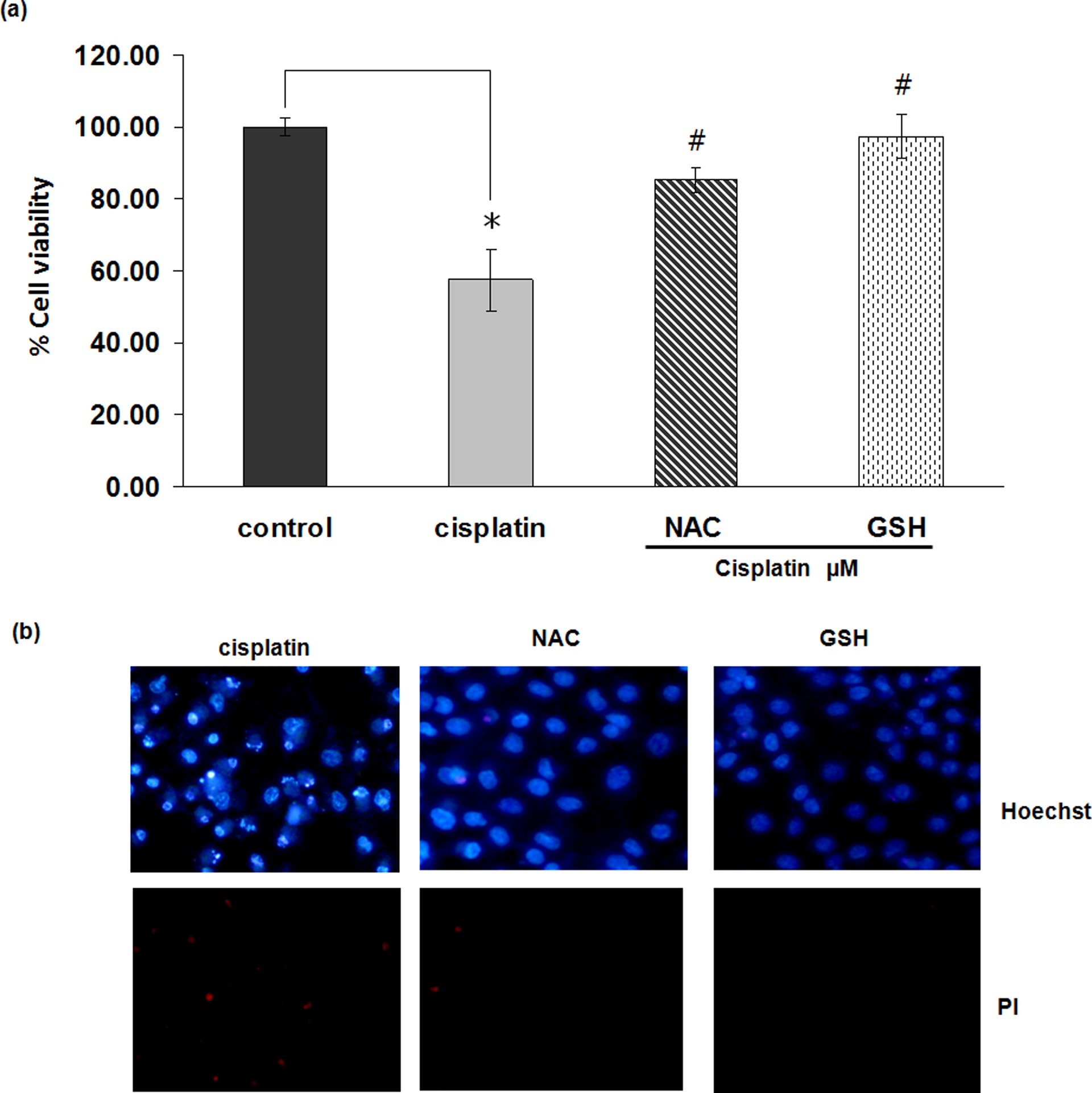
Anti-oxidants decrease cisplatin-induced cell death. (a) Cells were pretreated with N-acetylcystein (NAC; 5 mM) or reduced glutathione (GSH; 5 mM) for 1 h, and treated with cisplatin (50 µM). After 24 h, cell viability was determined by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. Values are means on triplicate samples ± S.D. *p < 0.01 versus non-treated control and #p < 0.01 versus cisplatin-treated control. (b) Apoptosis and necrosis cells were investigated by Hoechst 33342 and propidium iodide co-staining and detected by fluorescence microscope. Representative photographs are shown from three replicated experiments.
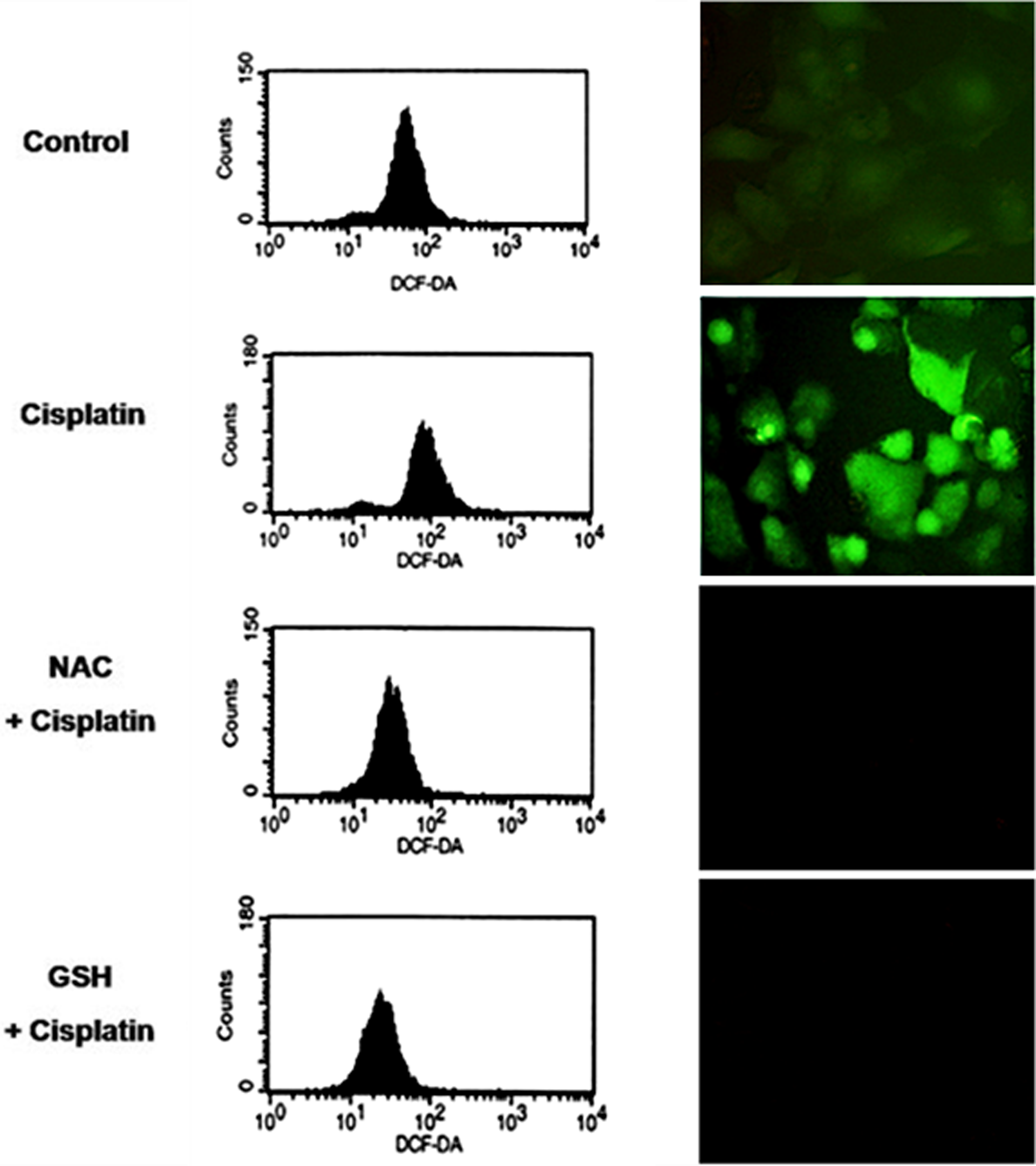
Cisplatin induces reactive oxygen species (ROS) generation in HK-2 cells. The left panel, cells were pretreated with N-acetylcystein (NAC; 5 mM) or reduced glutathione (GSH; 5 mM) for 1 h and exposed to cisplatin (50 µM). Cellular ROS levels were determined after 1 h by flow cytometry with 2,7-dichlorofluorescein diacetate (DCFH2-DA) (15 µM). In the right panel, cells were similarly treated and intracellular ROS signal was detected under fluorescence microscope.
GM and CM extracts protect HK-2 cells from cisplatin-induced cytotoxicity
Cells were incubated with various concentrations of GM (0.025–0.2 %v/v) or CM (0.025–0.2 %v/v) for 1 h and treated with 50 µM of cisplatin. After 24 h, cell viability and cell death were analyzed by MTT, Hoechst 33342, and PI staining assays. Figure 4a indicates that GM and CM, at the concentrations of 0.1 and 0.2 % v/v, was able to prevent the loss of cell viability induced by cisplatin in a dose-dependent manner. The results of Hoechst 33342 and PI assays indicate that GM and CM were able to decrease both apoptotic and necrotic cell deaths induced by cisplatin (Figure 4b). Consequently, both of the natural extracts markedly reduce cisplatin-induced apoptotic cells death. In addition to the protective effect of GM and CM on cisplatin-exposing HK-2 cells, the present study also determined the direct effect of GM and CM on HK-2 cells. The results showed that either GM or CM had no significant effect on the HK-2 cell viability, suggesting that both GM and CM have neither proliferative nor toxic effects on HK-2 cells (Figure 4c). To clarify whether the protective effect of these extracts was due to their anti-oxidant properties, cells were incubated with GM and CM or left untreated prior to cisplatin treatment and cellular ROS levels were evaluated after 1 h by flow cytometry and fluorescence microscopy using DCFH2-DA as an oxidative probe. Figure 5 shows that treatment of the cells with GM and CM inhibited cellular ROS induced by cisplatin, suggesting that the anti-oxidant property of GM and CM played a role in attenuating cisplatin toxicity.

Protective effect of Glycine max (GM) and Chrysanthemum indicum (CM) on cisplatin-treated HK-2 cells. (a) Cells were pretreated with various concentrations of GM and CM (0.025–0.2 %v/v) for 1 h prior to 24-h cisplatin exposure. Cell viability was determined by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. (b) Apoptosis and necrosis were detected after staining with Hoechst 33342 and propidium iodide, and examined under a fluorescence microscope. Representative photographs are shown from three replicated experiments. (c) Effect of GM and CM on HK-2 cell proliferation. After treatment with various concentrations of GM and CM for 24 h, cell viability was determined by MTT assay. Values are means on triplicate samples ± S.D. *p < 0.05 versus non-treated control and #p < 0.05 versus cisplatin-treated control.
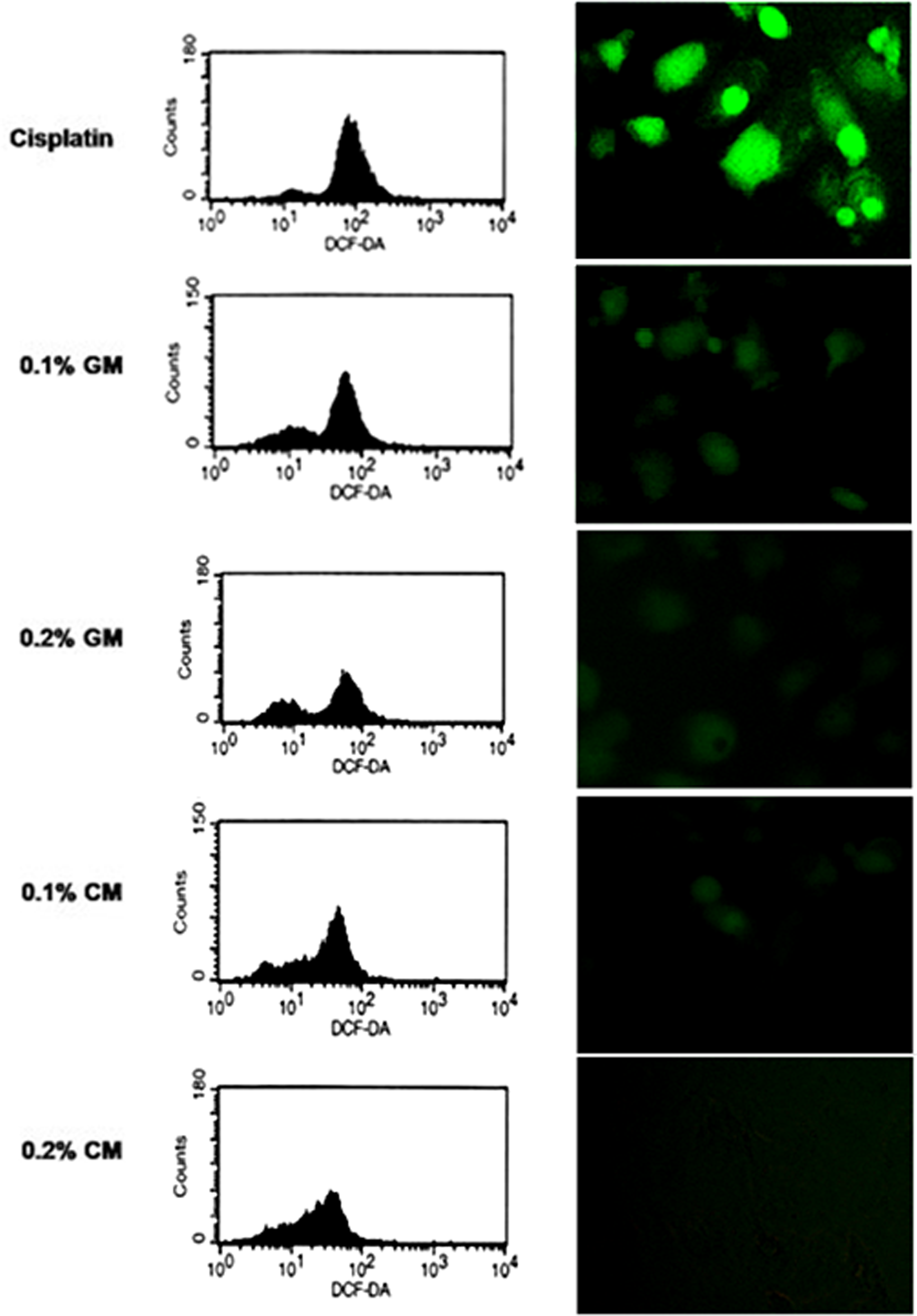
Effect of Glycine max (GM) and Chrysanthemum indicum (CM) on intracellular reactive oxygen species (ROS) induced by cisplatin. Cells were pretreated with 0.1 and 0.2 %v/v of GM or CM for 1 h followed by cisplatin (50 µM) treatment. Intracellular ROS levels were evaluated after 1 h by flow cytometer and fluorescence microscope.
Hydrogen peroxide and hydroxyl radical induced by cisplatin are key ROS in cisplatin-induced renal cell damage
Having shown that ROS mediated by cisplatin plays an important part in cisplatin-induced renal cell death, the present work further identified the specific ROS which were responsible for this cytotoxicity. Cells were pre-incubated with various specific ROS scavengers such as MnTBAP (superoxide anion scavenger), catalase (hydrogen peroxide scavenger), and deferoxamine (hydroxyl radical inhibitor) for 1 h prior to cisplatin (50 µM) treatment. After cisplatin treatment for 24 h, cell viability was evaluated by MTT assay. Moreover, the present study also investigated the role of specific ROS scavengers in cisplatin-induced apoptotic and necrotic cell death by Hoecht 33342 and PI assay. The data of this result indicate that only catalase and deferoxamine pretreatment were able to protect renal HK-2 cells from cisplatin-induced cell death, suggesting that hydrogen peroxide and hydroxyl radical were key mediators in cisplatin-induced renal cell damage (Figure 6a and b).

Effect of reactive oxygen species (ROS) scavengers on cisplatin-induced cell death. (a) HK-2 cells were pretreated with various specific ROS scavengers; Mn(III)tetrakis(4-benzoic acid) porphyrin chloride (MnTBAP) (50 µM), deferoxamine (DFO; 1 mM), and catalase (5000 U/ml) for 1 h before 24 h cisplatin treatment and cell viability was determined by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. Values are means on triplicate samples ± S.D. *p < 0.05 versus non-treated control and #p < 0.05 versus cisplatin-treated control. (b) Nuclear morphology of apoptotic and necrotic cells after pretreated with various specific ROS scavengers and treated with cisplatin followed by Hoechst 33342 and propidium iodide staining. Representative photographs are shown from three independent experiments.
GM and CM extracts protect cells from cisplatin-induced cell death by their hydrogen peroxide and hydroxyl radical scavenging activities
To test whether GM and CM could protect renal cells by the anti-oxidant activity against hydrogen peroxide and hydroxyl radicals, cells were either left untreated or pretreated with GM and CM for 1 h and followed by specific ROS generators which are hydrogen peroxide and ferrous sulphate (hydroxyl radical generator). After 1-h incubation, the intracellular ROS level was determined by DCFH2-DA and flow cytometry. Figure 7 indicates that treatment with either hydrogen peroxide or ferrous sulphate significantly increased cellular ROS in these cells. The results of the present study showed that CM exhibited a strong anti-oxidant effect against both hydrogen peroxide and hydroxyl radical (Figures 7a and b), whereas GM could only prevent hydrogen peroxide up-regulation in hydrogen peroxide-treated cells (Figure 7b). These results suggest that GM and CM could, at least in part, protect cisplatin-induced renal cell damage by their anti-oxidant activities against hydrogen peroxide and hydroxyl radicals.

Anti-oxidant activity of Glycine max (GM) and Chrysanthemum indicum (CM) against hydrogen peroxide and hydroxyl radical in HK-2 cells. (a) Cells were pretreated with 0.1 and 0.2 %v/v of GM or CM for 1 h, then cell were exposed to 100 µM ferrous sulphate for 1 h. Cellular reactive oxygen species (ROS) levels were evaluated by flow cytometry with 2,7-dichlorofluorescein diacetate (DCFH2-DA). (b) Cells were pretreated with 0.1 and 0.2 %v/v of GM or CM for 1 h, and exposed to 200 µM hydrogen peroxide for 1 h. Intracellular ROS levels were evaluated by flow cytometry.
GM and CM extracts cause minimal interfering effect on cisplatin-induced lung and melanoma cancer cell apoptosis
A major limitation of using anti-oxidants for the prevention of renal toxicity in cisplatin-based chemotherapy is the ameliorating effect of anti-oxidant on tumor cells. To test whether these extracts could reduce efficacy of cisplatin in induction of cancer cell death, human lung cancer H460 and human melanoma G361 cells were used. Cells were left untreated or pretreated with 0.1 and 0.2 % v/v of GM or CM for 1 h and treated with 100 µM cisplatin. After 24-h incubation, cell viability, apoptosis, and necrosis were evaluated as described. Figure 8a shows that treatment with 100 µM cisplatin caused approximately 50% reduction of cell viability in H460 and G631 cells. Importantly, co-treating the cells with either GM or CM and cisplatin had no significant effect on cisplatin-induced cell death as compared to the cisplatin-treated control. Besides, our Hoechst 33342 and PI staining assays confirm that pretreatment of these extracts caused no significant change in mode and quantity of cell death in response to cisplatin (Figure 8b).

Effect of Glycine max (GM) and Chrysanthemum indicum (CM) on cisplatin-treated melanoma (G361) and lung (H460) cancer cells. (a) Cells were pretreated with 0.1 and 0.2 %v/v GM or CM for 1 h and cisplatin (100 µM) was added. After 24-h incubation, cell viability was determined by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. Values are means on triplicate samples ± S.D. *p < 0.05 versus non-treated control and (b and c) Apoptotic and necrotic cell death of melanoma and lung cancer cells, respectively, were detected after staining with Hoechst 33342 and propidium iodide, under a fluorescence microscope. Representative photographs are shown from three replicated experiments.
Discussion
Cisplatin-induced nephrotoxicity is a major side effect that limits the use of cisplatin in many patients. Since the amount of evidence shows that oxidative stress is the primary factor that contributes to cisplatin nephrotoxicity, much effort has been directed toward the search for natural products that are capable of attenuating renal damage. Previous studies have reported that soy bean extract was able to relieve signs and symptoms in chronic renal disease, 29 and their phenolic compositions were reported to reduce gentamicin-induced renal injury. 30 Likewise, Chrysanthemum indicum, the herbal Chinese medicine, is composed of flavonoids which exhibit anti-oxidant activity.24,31,32 However, their roles on cisplatin-induced renal cell apoptosis are largely unknown. We reported herein for the first time that GM and CM extracts could be able to protect human renal tubular cell from cisplatin-induced apoptosis via the mechanism involved in their ability to inhibit cisplatin-mediated cellular ROS formation.
In accordance with the evidence indicating that ROS generation in response to cisplatin treatment plays a prominent role in cisplatin-induced cell death as well as acute renal failure in vivo,10,11,33–35 the results of the current study indicate that the treatment with cisplatin caused a significant induction of intracellular ROS, and that an inhibition of that by anti-oxidants (NAC and GSH) could be able to block renal cell apoptosis induced by cisplatin. Furthermore, the results showed that only the blockage of either hydrogen peroxide or hydroxyl radical could be able to protect renal HK-2 cells from cisplatin-induced injury. We determined the role of specific ROS generated by cisplatin to induce cell death by pretreating the cells with various specific ROS scavengers prior to cisplatin treatment. Deferoxamine inhibited hydroxyl radical formation by chelating cellular iron ion (Fe2+), which subsequently attenuated hydroxyl radical formation via Fenton’s reaction.36,37 MnTBAP are cell-permeable superoxide dismutase mimetic functioning in transformation of superoxide anion to hydrogen peroxide,36,38 and catalase will catalyse the transformation of hydrogen peroxide to water and oxygen.36,37 The results of the present study indicate that pretreatment with catalase and deferoxamine could be able to both reduce ROS generation (data not shown) and cell death induced by cisplatin, whereas treatment with MnTBAP had only non-significant alteration. These results imply that some specific ROS like superoxide anion may not play a role in killing renal cells which is consistent with the previous finding indicating that cisplatin-induced cell apoptosis in human lung cancer cells was mainly due to cisplatin-mediated hydrogen peroxide generation. 39 In addition, we observed that CM and GM exhibited different anti-oxidant characteristics. CM possessed both hydrogen peroxide and hydroxyl radical scavenging activities whereas GM could only inhibit cellular hydrogen peroxide up-regulation. However, ability of both extracts in inhibition of cisplatin-induced hydrogen peroxide (data not shown) as well as cisplatin-induced cell damage was quite similar quantitatively, suggesting that GM and CM extracts protect renal cells mainly through the anti-hydrogen peroxide mechanism. It is generally accepted that cisplatin mediates cell death by 2 major pathways which are ROS and DNA-adduct formation pathways. Because renal cells are more susceptible to oxidative stress-induced damage,40–43 while frequently proliferated cancer cells are more sensitive to DNA-adduct induced cell apoptosis,44–46 an introduction of these extracts which focus on specific ROS inhibition may allow the selectivity in protecting only renal cells but not interfering with the cisplatin sensitivity of lung cancer and melanoma cells.
In summary, the present study demonstrates that cisplatin induced human proximal cell apoptosis via a ROS-dependent mechanism. GM and CM extracts showed strong anti-oxidant activity against cisplatin-induced ROS production and consequently protected renal cell damage. Importantly, we report for the first time that in renal epithelial cells hydrogen peroxide and hydroxyl radicals are the principle ROS playing a key role in cisplatin-induced cytotoxicity. Unlike other anti-oxidants, GM and CM extracts selectively protected renal cells while causing no significant interfering effect on cisplatin-induced cancer cell death demonstrated in lung carcinoma H460 and melanoma G361 cells. These findings may at least provide the initial evidence necessary for the further development of these extracts to be used as renoprotective agents.
Footnotes
Faculty of Pharmaceutical Sciences, Chulalongkorn University is gratefully acknowledged for a research grant.
