Abstract
Barometric whole-body plethysmography (WBP) was used to examine pulmonary functions at 4 and 24 hours postexposure to soman (GD) in guinea pigs without therapeutics to improve survival. Endotracheal aerosolization by microinstillation was used to administer GD (280, 561, and 841 mg/m3) or saline to anesthetized guinea pigs. Significant increases in respiratory frequency (RF), tidal volume (TV), and minute volume (MV) were observed with 841 mg/m3 GD at 4 hours and that were reduced at 24 hours postexposure. A dose-dependent increase in peak inspiration flow and peak expiration flow was present at 4-hour post-GD exposure that was reduced at 24 hours. Time of inspiration and expiration were decreased in all doses of GD exposure at 4 and 24 hours, with significant inhibition at 841 mg/m3. End-expiratory pause (EEP) increased at 280 and 561 mg/m3, but decreased in animals exposed 841 mg/m3 at 24 hours postexposure. Pseudo-lung resistance (Penh) and pause followed similar patterns and increased at 4 hours, but decreased at 24 hours postexposure to 841 mg/m3 of GD compared to control. These studies indicate GD exposure induces dose-dependent changes in pulmonary function that are significant at 841 mg/m3 at 4 hours and remains 24 hours postexposure. Furthermore, at 4 hours, GD induces bronchoconstriction possibly due to copious airway secretion and ongoing lung injury in addition to cholinergic effects, while at 24 hours GD induces bronchodilation a possible consequence of initial compensatory mechanisms.
Keywords
Soman (pinacolyl methylphosphonofluoridate or GD) is one of the most toxic chemical warfare nerve agents (CWNA), because it permanently inactivates AChE within minutes after exposure and the aged AChE is resistant to reactivation by oximes. 1 Although respiratory failure is the main cause of death following lethal exposure to GD, the exact mechanisms involved are not well understood. Attempts are being made to delineate the contribution of central nervous system respiratory disturbance, peripheral nervous system effects, or direct pulmonary effects of GD in respiratory failure. 2 –5 Diaphragm muscles are still functional at doses of GD that cause respiratory failure, suggesting that CNS respiratory depression is the main cause of respiratory toxicity. 3,4,6 Low levels of GD induce respiratory depression mainly due to CNS respiratory effects and exposure to higher doses cause peripheral nervous system effects and respiratory depression. 2
Majority of the studies on the respiratory toxicity of GD exposure are the outcome of noninhalation administration. The effects of nerve agents on respiratory failure are affected by the route of administration of the agent. 7 Infusion of GD (30 μg/kg per 10 minutes) by routes of intravenous, subcutaneous, intraperitoneal, intratracheal, upper airway showed different times to respiratory failure. 7 Inhalation exposure studies are required to assess the immediate effects of GD on the respiratory system and the amount that contributes to the overall respiratory failure. Vaporized GD exposure in the upper airway of baboons showed increased lung resistance, decreased dynamic compliance, and slow expirations following exposure to GD. 6,8 We have demonstrated that endotracheal aerosolization of GD causes respiratory toxicity and lung injury. 9 The nature of the toxic effects of GD on respiratory physiology and function following inhalation exposure and contribution of the peripheral nervous system versus CNS respiratory center effects for respiratory failure is not well understood.
We have developed a microinstillation methodology of inhalation exposure to CWNAs for the assessment of respiratory toxicity, lung injury, and potential therapeutic evaluation. 10 –14 The method involves endotracheal aerosolization of the nerve agent precisely above the bifurcation of the trachea using a microcatheter with 5 peripheral holes to aerosolize the agent that is delivered through the central hole. Endotracheal aerosolization bypasses the detoxification of the nerve agents by high levels of carboxylesterases and mucous present in the upper airways of rodents. 15 –17 Microinstillation technology requires small amounts of CWNA compared to nose-only or whole-body exposure systems and is more practical.
Treatment with oximes or atropine sulfate is generally used to increase survival of animals following exposure to nerve agents. Atropenized animals survive longer and have been used in the past to study GD toxicokinetics. 18 –20 Treatment with atropine sulfate may affect the respiratory physiology. It has been reported that atropine treatment protects and reverses respiratory toxicity induced by GD at different doses. 2,21 –23 Endotracheal atropine methyl bromide blocks profuse airway secretions and protects against nerve agent-induced respiratory toxicity. 24 Atropine methyl bromide does not cross the blood-brain barrier, suggesting that the protective effect is due to its local effects on the respiratory system or peripheral nervous system. 25,26 Therefore, it is important to investigate the effects of GD on the respiratory system in animals without any pre or posttreatments to provide an accurate evaluation of respiratory toxicity.
In this study, we investigated the effects of different doses of GD on pulmonary function at 4 hours and 24 hours postexposure by WBP in the absence of any treatments and demonstrate that exposure to GD dose-dependently alters respiratory function. We also reveal that only at early time points GD exposure induces bronchoconstriction, but at later time points GD induces bronchodilation.
Methods
Animals
Research was conducted in compliance with the Animal Welfare Act and other federal statutes and regulations relating to animals and experiments involving animals. It adheres to principles stated in the Guide for the Care and Use of Laboratory Animals, National Research Council, published by the National Academy Press, 1996, and the Animal welfare Act of 1966, as amended. Adult male Hartley guinea pigs (250-300 g, Charles River Laboratories, Wilmington, Massachusetts) were housed individually under standard conditions with 12 hours light/dark cycle and food and water were available ad libitum. Animals were quarantined for 1 week prior to experiments. The study protocol was approved by the Institution Animal Care and Use Committee, US Army Medical Research Institute of Chemical Defense, Aberdeen Proving Ground, Maryland.
Chemicals
Soman (pinacolyl methylphosphonofluoridate or GD, 98.8% pure) was obtained as a stock of 1.88 to 1.90 mg/mL from Edgewood Chemical and Biological Center, Edgewood, Maryland, and diluted in saline and kept frozen at −80°C until use. Telazol was purchased from Wyeth Pharmaceuticals (Madison, New Jersy). Medetomidine was obtained from Pfizer Pharmaceuticals (New York, New York). Sterile saline solution was purchased from Pierce Chemical Co (Rockford, Illinois). The microinstillation equipment was obtained from Trudell Medical International, Inc (Ontario, Canada). Exposure to GD was performed in an approved fume hood with the appropriate personal protective equipment for the handling of CWNAs as set forth by the US Army. At the end of all agent exposures, any remaining diluted GD and materials contaminated with GD were decontaminated with 10% sodium hydroxide prior to disposal.
General Experimental Design
The primary objective of this study was to assess the effects of GD microinstillation inhalation exposure on respiratory function in guinea pigs that are not treated with atropine or oxime and delineate the potential mechanism of respiratory toxicity. Age-matched male Hartley guinea pigs were anesthetized, intubated, and exposed to various doses of GD or saline by microinstillation inhalation exposure. The animals were allowed to stay in the agent exposure hood for 15-minute postexposure before being placed in new polycarbonate cage. Development of cholinergic signs, such as increased salivation, lacrimation, defecation, urination and mild muscular fasciculation, convulsions, and breathing variations were monitored for 2 hours. Food and water were available ad libitum prior to being placed in the whole-body barometric plethysmography chamber. The respiratory dynamics were measured in unrestrained, conscious guinea pigs at 4 and 24 hours after GD exposure and compared to saline or baseline values obtained before the exposure. Typically, the animals wake up from anesthesia at 4 hours after the exposure. A total of 8 saline controls and 24 GD-exposed animals (8 = 280 mg/m3, 8 = 561 mg/m3, and 8 = 841 mg/m3) were used in this study. Animals were given no other postexposure therapeutics. A total of 32 guinea pigs were used for this study.
Microinstillation Inhalation Exposure to GD
Microinstillation exposure to GD was performed essentially as described earlier. 27 Briefly, guinea pigs (saline or GD exposed) were anesthetized using a combination of Telazol (40 mg/kg, im) and Medetomidine (0.125 mg/kg, sc) 15 minutes prior to the start of intubation procedure (Buchanan et al, 1998). Animals were intubated with a 10-cm translucent polystyrene tube inserted with a metal stylet and the tip of the tube was placed 2.5 cm above the bifurcation of the trachea 28 based on the typical tracheal length in guinea pigs of similar size. Intubation was verified by condensation of vapor at the external end of the intubation tube. The microinstillation catheter was inserted through the intubation tube and the tip was placed 0.5 cm past the intubation tube. Exposure to GD was performed in an approved fume hood with the appropriate personal protective equipment for the handling of CWNAs as set forth by the US Army. The guinea pigs were exposed to different doses of GD (280, 561, and 841 mg/m3), 40 pulses/min for 4-minute duration. Each pulse aerosolized 1.4 μL of GD in a volume of 1.1 mL air. 27 After exposure, the intubation tube and microcatheter were removed, decontaminated, and the animals were allowed to stay in the hood for 15 minutes and then transferred to a clean cage and kept for 24 hours.
Body Weight Loss
Percentage body weight loss of the animals was calculated by weighing the guinea pigs prior to the start of the experiment and again just before euthanasia at 24 hours postexposure.
Evaluation of Lung Edema by Wet/Dry Weight Ratio
Following necropsy, the accessory lung lobe was tied off using surgical suture before conducting bronchoalveolar lavage (BAL) to preserve the native postexposure lung conditions. By this method, samples from a single animal can be used for multiple analysis including the determination of both wet/dry ratio and biochemical analysis of BAL fluid and cells. The accessory lung lobe was removed and weighed immediately using an analytical balance. The lobe was dried at 100°C dry heat for 7 days and weighed again. The wet/dry weight ratio was calculated to determine the degree of lung edema.
Barometric Plethysmography
Conscious, unrestrained guinea pigs were placed in a whole-body barometric plethysmography (WBP; Buxco Electronics, Inc, Sharon, Connecticut) before GD exposure or following 4 and 24 hours postexposure to determine the respiratory function using Bio System XE software. 29,30 The barometric WBP was calibrated according to the method described by Drorbaugh and Fenn 31 and Bargeton and Gauge. 32 The total volume of the WBP chamber is 6.28 mL, and the airflow rate was set at 2.5 L/min. 10,33,34 A very small rejection index was used to exclude external noise. For the measurement of respiratory dynamics, following initial 30-minute stabilization in the barometric WBP chamber to allow the guinea pigs to acclimate to the WBP chamber, baselines for each guinea pig were recorded for 75 minutes. The animals were monitored again in the barometric WBP at 4 and 24 hours after GD exposure. The interval between data point measurements was 10 seconds. The definitions of respiratory dynamics parameters that were measured by barometric WBP are as follows: RF: Instantaneous breath-by-breath rate of breathing. Tidal volume (TV): the volume of air inspired. Minute volume (MV): the product of TV and respiratory rate calculated on a breath-by-breath basis. Inspiratory (Ti) and expiratory (Te) time: the time spent inhaling and exhaling during each breath, respectively. Peak inspiratory flow (PIF) and peak expiratory flow (PEF): the maximum inspiratory and maximum expiratory flow that occurs in 1 breath. End-inspiratory pause (EIP): the time of pause after the end of inspiration before the start of next expiration. End expiratory pause: the time of pause after the end of expiration before the start of next inspiration. Penh (enhanced pause): An estimate of lung resistance, and is calculated from pause (P) using the ratio of Te and RT: P = (Te/RT) − 1, so that Penh = P × PEF/PIF. A detailed evaluation of the diagnosis of Penh has been described. 35,36
Statistics
Statistical analyses were performed using the Graph Pad Prism V4.03 software (Graph Pad Software Inc, San Diego, California). The Mann-Whitney test was used to calculate the P values. A P value <.05 was considered significant.
Results
General Effects of GD Microinstillation Inhalation Exposure
All guinea pigs exposed to 280 and 561 mg/m3 of GD or saline survived 24 hours. A total of 3 animals out of the 8 that were exposed to 841 mg/m3 of GD survived. All 3 doses of GD-exposed animals exhibited signs of cholinergic toxicity, such as increased salivation, lacrimation, defecation, urination, and mild muscular fasciculation. A number of animals exposed to 841 mg/m3 of GD experienced upper and lower limb convulsions and died 5 to 10 minutes post-GD exposure. Copious secretions coming out of the mouth and nostrils were observed and were exacerbated in animals, exposed to 841 mg/m3 GD. Guinea pigs breathed normally during the first 3 minutes of GD exposure. Blood O2 saturation and pulse rates decreased at 4 to 5 minutes post-GD exposure as reported previously. 9
Analysis of Body Weight and Lung Edema
There was 7% to 8% body weight loss in animals exposed to 280, 561, and 841 mg/m3 of GD, whereas a body weight loss of 4.1% was observed in saline controls at 24 hours postexposure. The loss in the percentage of body weight was significant (P = .006) in animals exposed to 841 mg/m3 GD in comparison to saline-treated controls (Figure 1A). Animals exposed to 561 mg/m3 of GD also resulted in a significant (P = .008) increase in body weight loss, while exposure to 280 mg/m3 did not produce a significant increase.
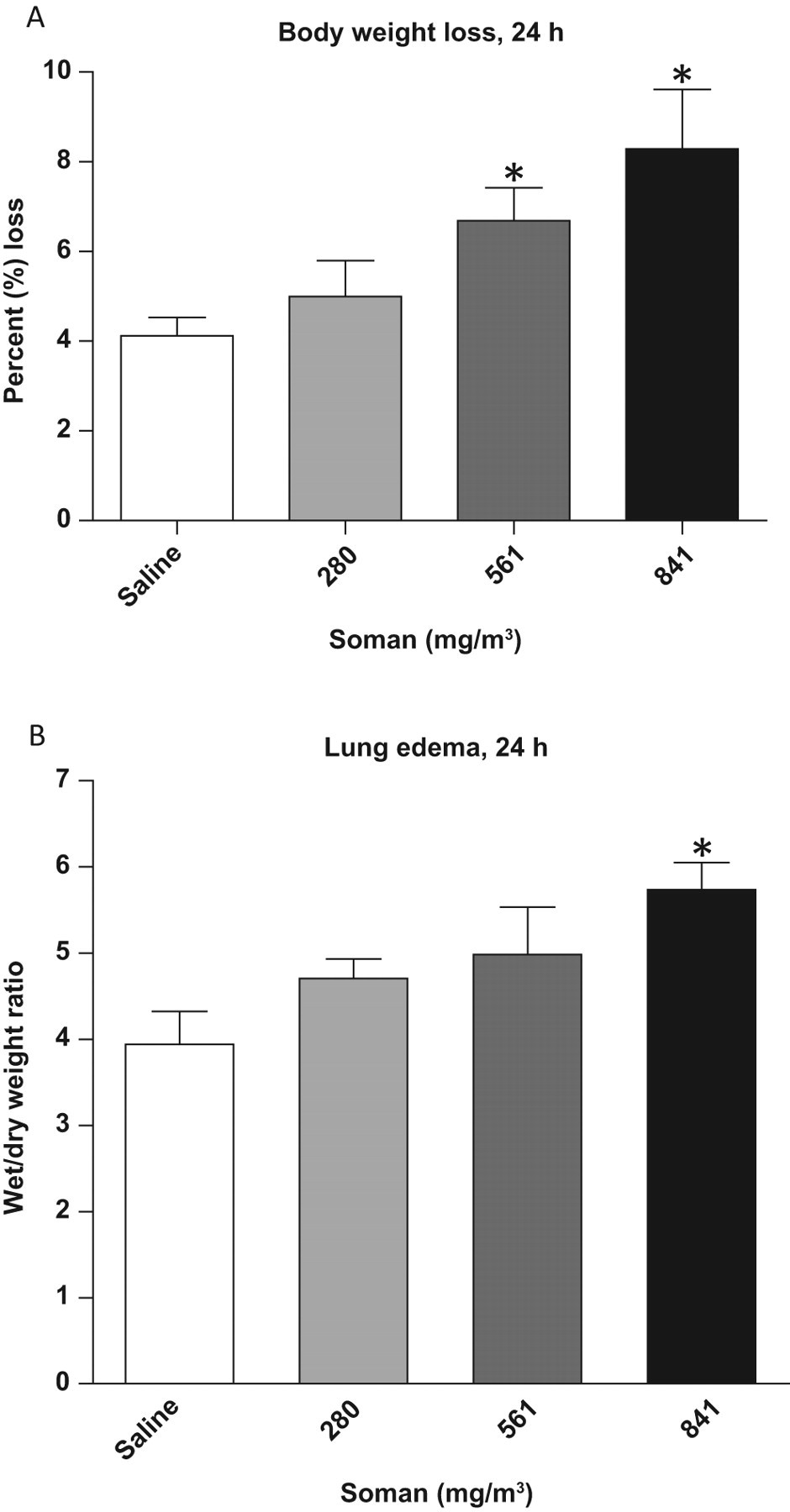
Percentage body weight loss, accessory lung lobe edema. Animals were weighed before administration of anesthesia and prior to euthanasia, 24 hours postexposure, to calculate the percentage body weight loss. A, Percentage body loss. There was a significant (P = .008 and .006) increase in the percentage weight loss in animals exposed to 561 and 841 mg/m3 of soman (GD) in comparison to saline-treated controls (n = 6-8). B, Accessory lobe edema. There was a significant increase (P = .008) in accessory lung lobe edema in all 841 mg/m3 GD-exposed animals in comparison to saline-treated controls (n = 3-8). Asterisk indicates statistical significance.
Lung edema measured as wet/dry weight ratio of the accessory lung lobe was significantly increased (P = .008) in animals exposed to 841 mg/m3 of GD (Figure 1B). Lower concentrations of GD (280 and 561 mg/m3) did not significantly alter accessory lung lobe edema.
Barometric Plethysmography Signal Following GD Exposure
A representative segment of barometric WBP recording of animals at 4 hours after 841 mg/m3 GD exposure is shown in Figure 2 . Breathing pattern of the saline control animals was similar to the baseline, while GD-exposed animals showed an irregular pattern of respiration and increased respiratory difficulty at 4 hours. The breathing peaks were more random and closer following GD exposure as a consequence of rapid breathing.
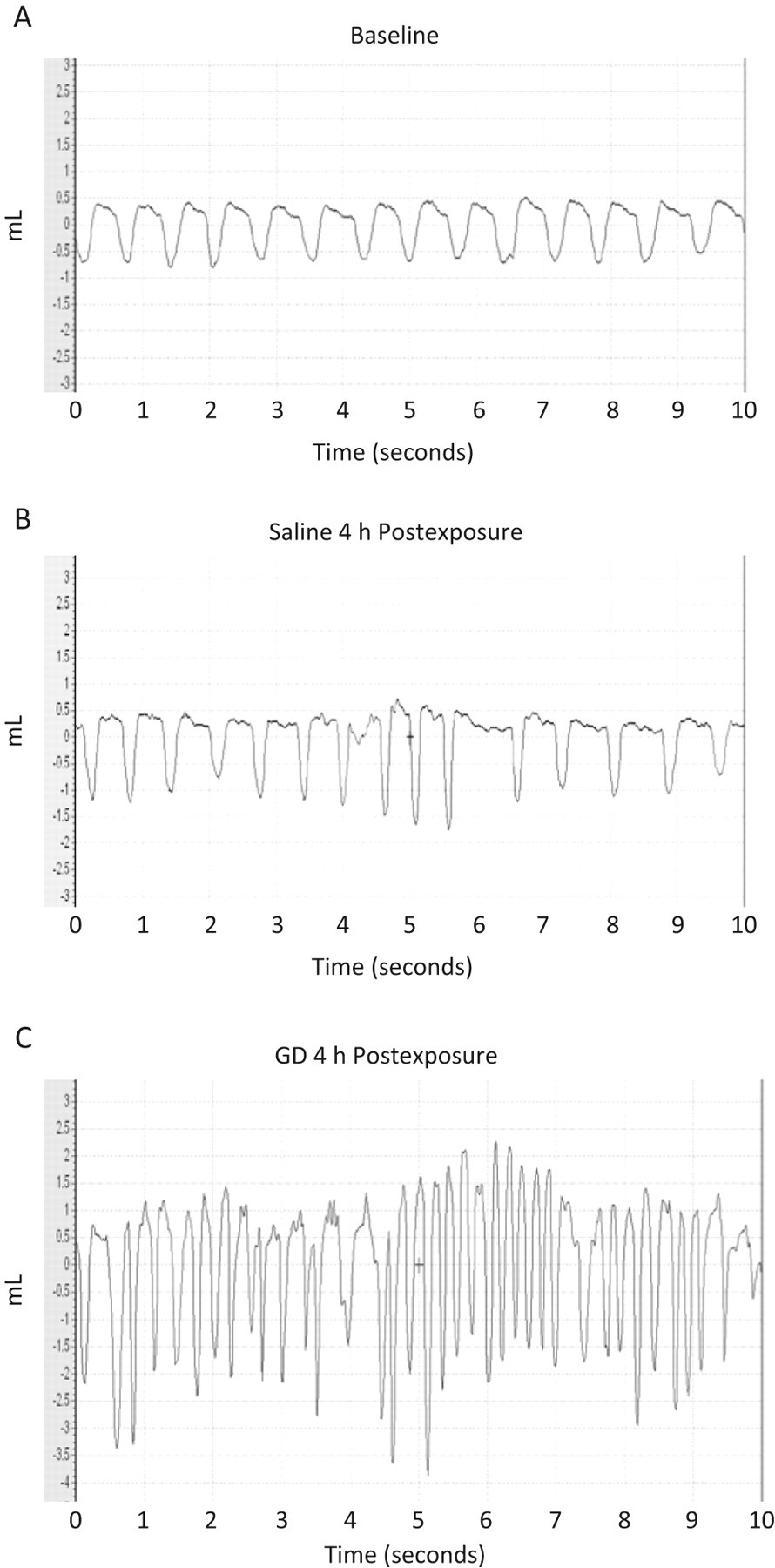
Barometric plethysmography recordings of soman (GD)-exposed guinea pigs: Whole-body barometric plethysmography measured in animals at baseline, and 4 hours after exposure to 841 mg/m3 GD or saline exposure. Animals were exposed to GD or saline and allowed to recover in room air. Awaken, unrestrained animals were placed in the barometric whole-body plethysmography and the respiratory dynamics measured. A, Representative segment (10 seconds) of baseline (BL) recordings. B, A representative segment of saline-exposed animals 4 hours postexposure. C, A representative segment of animals exposed to 841 mg/m3 of GD 4 hours postexposure. The x-axis is defined as time (seconds) and the y-axis as volume (mL).
Effects of GD on RF, TV, and MV
The average RF, TV, and MV following GD exposure from surviving animals is shown in Figure 3 . Animals that survived following 841 mg/m3 GD exposure showed a significant increase in RF at 4 and 24 hours, respectively (P = .018 and .028). A significant increase in TV was observed at 4 hours following GD exposure (P = .024). Minute volume, a product of TV and RF, increased in all GD-exposed groups at 4 hours, with a significant increase in animals exposed to 841 mg/m3 at 4 and 24 hours in comparison to saline controls (P = .018 and .038). The increases in RF, MV, and TV postexposure to 841 mg/m3 GD lasted for 24 hours, whereas these parameters returned to saline control levels in animals exposed to 280 and 541 mg/m3.
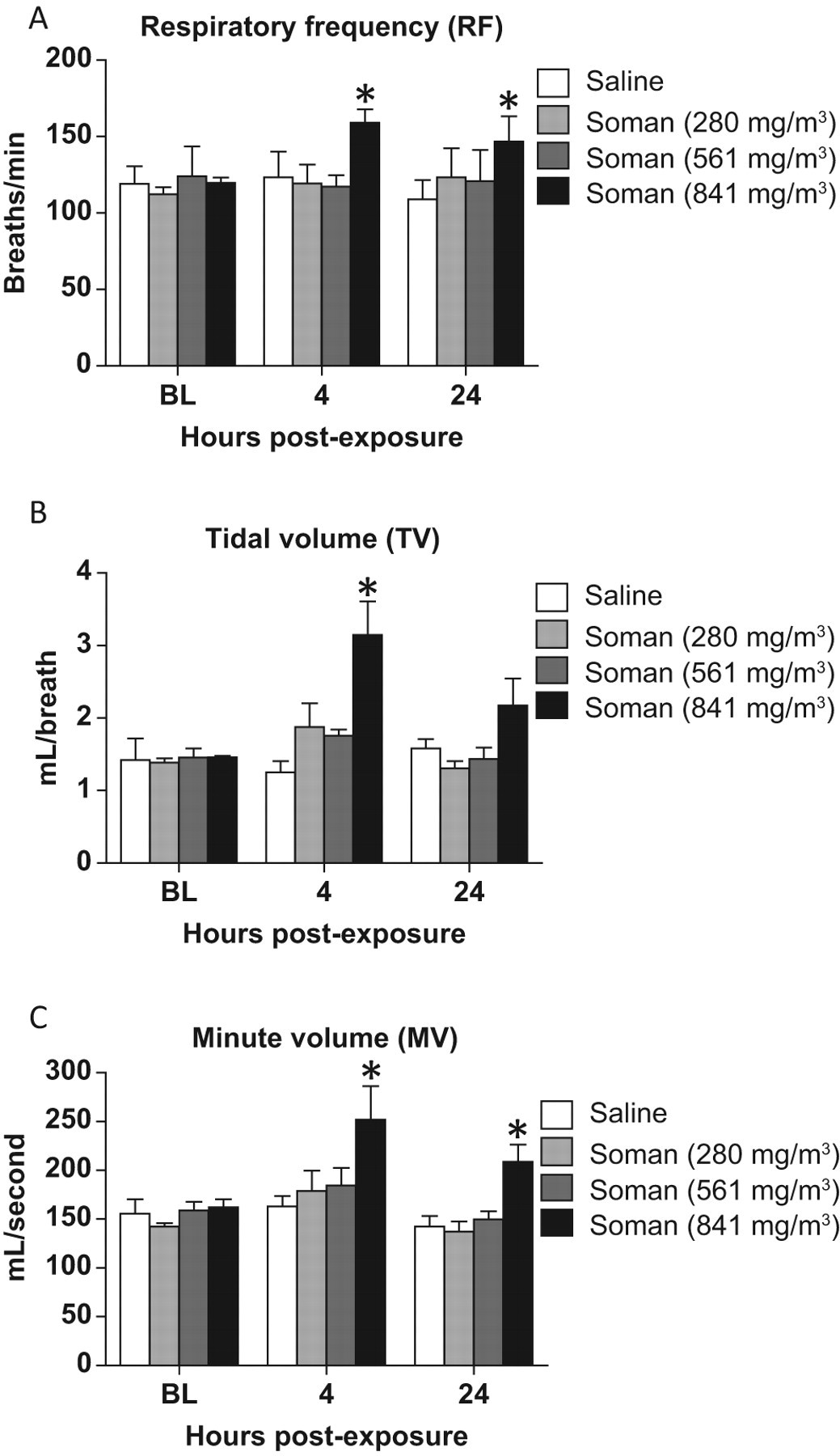
Respiratory frequency (RF), tidal volume (TV), and minute volume (MV) following soman (GD) exposure: Respiratory frequency, MV, and TV were determined by whole-body barometric plethysmography measured in animals at 4 and 24 hours after GD exposure (n = 3-8). At 4 hours postexposure, animals exposed to 841 mg/m3 GD resulted in significant increase in RF (P = .018), TV (P = .024), and MV (P = .038). At 24 hours, significant increases in MV (P = .038) and RF (P = .028) were observed following exposure to 841 mg/m3 GD. Asterisk indicates statistical significance.
Effects of GD on PIF and PEF
A dose-dependent increase in PIF (Figure 4A) and PEF (Figure 4B) was observed at 4 hours following GD exposure. A significant increase in PIF and PEF was observed in animals exposed to 841 mg/m3 at 4 hours (P = .048 and .024, respectively). Animals exposed to 841 mg/m3 GD resulted in a significant increase in PIF 24 hours post-GD exposure (P = .048). No significant changes in PEF were observed in GD-exposed animals at 24 hours postexposure.
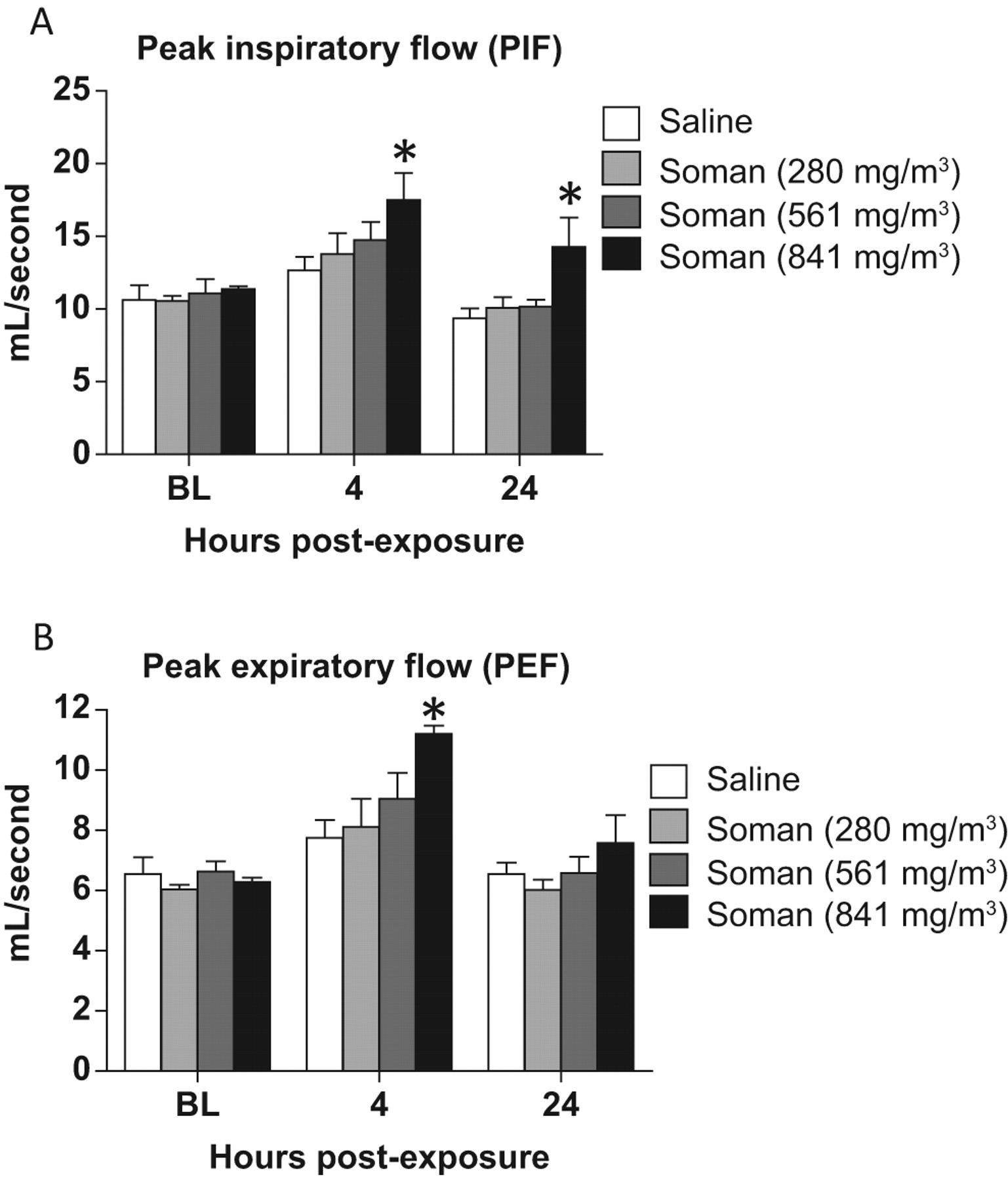
Peak inspiratory and expiratory flow (PIF and PEF) following GD exposure: PIF and PEF, the maximum inspiratory and expiratory flow that occurs in a breath was determined by whole-body barometric plethysmography measured in animals at 4 and 24 hours after soman (GD) exposure (n = 3-8). Animals exposed to 841 mg/m3 of GD resulted in a significant increase in PIF (P = .048) and PEF (P = .024), 4 hours postexposure. Asterisk indicates statistical significance.
Effects of GD on Ti and Te
A dose-dependent decrease in Ti (Figure 5A) and Te (Figure 5B) was observed at 4 hours post-GD exposure in comparison to saline controls. A similar result was observed at 24 hours post-GD exposure. Significant changes in Ti were observed at 841 mg/m3 GD at 4 and 24 hours (P = .012 and .038, respectively). Similarly, significant changes in Te were also observed at 4 and 24 hours following 841 mg/m3 GD exposure (P = .012 and .038, respectively) but were dose dependent for all GD-exposure groups at both intervals.
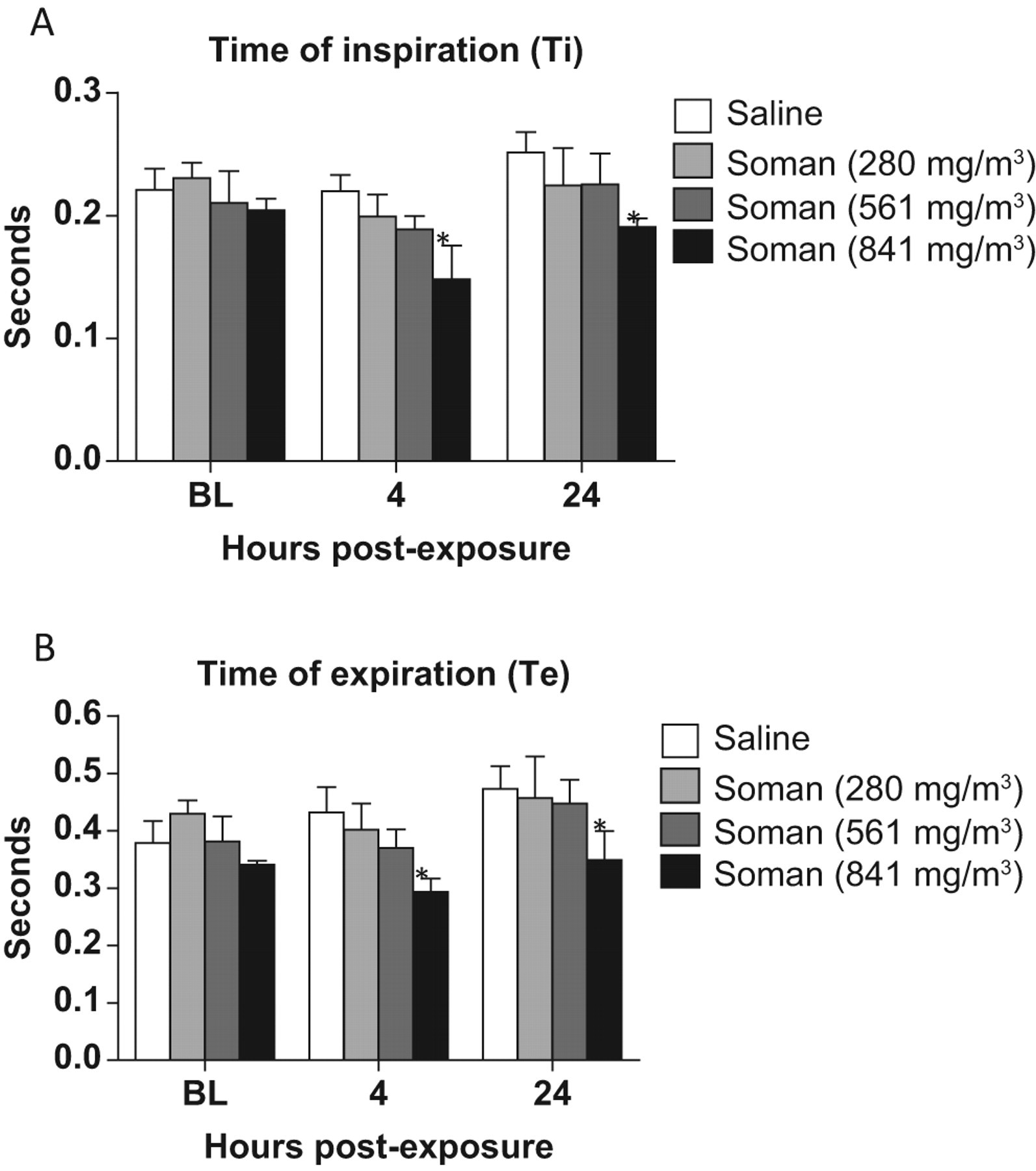
The effects of soman microinstillation inhalation exposure on time of inspiration, and expiration (Ti, Te): Animals exposed to soman (GD) resulted in a dose-dependent decrease in Ti, with a significant decrease in animals exposed to 841 mg/m3 GD at 4 and 24 hours postexposure. A significant decrease in Te was observed 4 and 24 hours postexposure in animals exposed to 841 mg/m3 GD in comparison to saline controls (n = 3-8). Asterisk indicates statistical significance.
Effects of GD on EIP and EEP
There was no significant difference in EIP at 4 or 24 hours after exposure for all GD doses. A dose-dependent decrease in EIP was observed in animals exposed to 841 mg/m3 GD at 24 hours (Figure 6A). The EEP was not significantly altered at 4 hours postexposure and there was a dose-dependent increase in animals exposed to 280 and 561 mg/m3. However, at 24 hours postexposure, the EEP was decreased significantly in animals exposed to 841 mg/m3 GD in comparison to saline controls (P = .035; Figure 6B). There was a dose-dependent increase in EEP at 24 hour postexposure in animals exposed to 280 and 561 mg/m3 in comparison to saline controls but was nonsignificant.
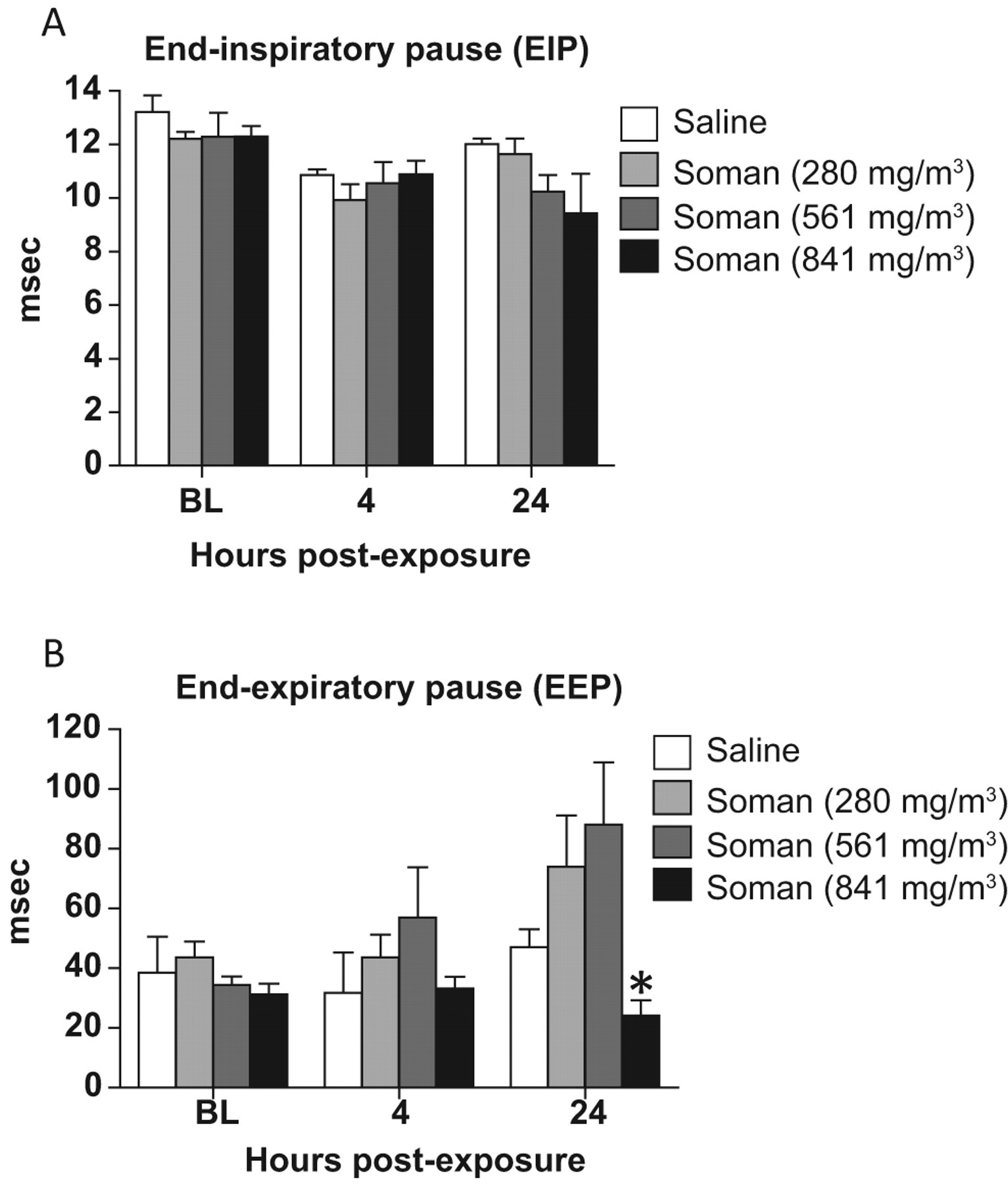
End-inspiratory and -expiratory pause (EIP and EEP) following soman (GD) exposure: EIP and EEP were evaluated 4 and 24 hours postexposure to GD (n = 3-8). Animals exposed to 841 mg/m3 GD at 4 hours resulted in a significant (P = .036) decrease in EEP at 24 hours postexposure. Asterisk indicates statistical significance.
GD Induced Pseudo Lung Resistance (Penh) and Pause
At 4 hours post-GD exposure, mean Penh, a measure of bronchoconstriction, was increased dose dependently. Significant increase in Penh (P = .024) was observed at 841 mg/m3 dose of GD at 4 hours. On the other hand, a significant (P = .023) decrease in Penh was observed in animals exposed to 841 mg/m3 of GD at 24 hours in comparison to saline controls (Figure 7A). Penh was similar to the control levels at 280 and 561 mg/m3 GD at 24 hours. Pause, another measure of bronchoconstriction, was similar to Penh, with a significant increase at 4 hours and decrease at 24 hours (P = .047 and .024, respectively) following 841 mg/m3 GD exposure (Figure 7B). There was also a significant (P = 0.046) increase in Pause at 4 hours following exposure to 561 mg/m3 GD.
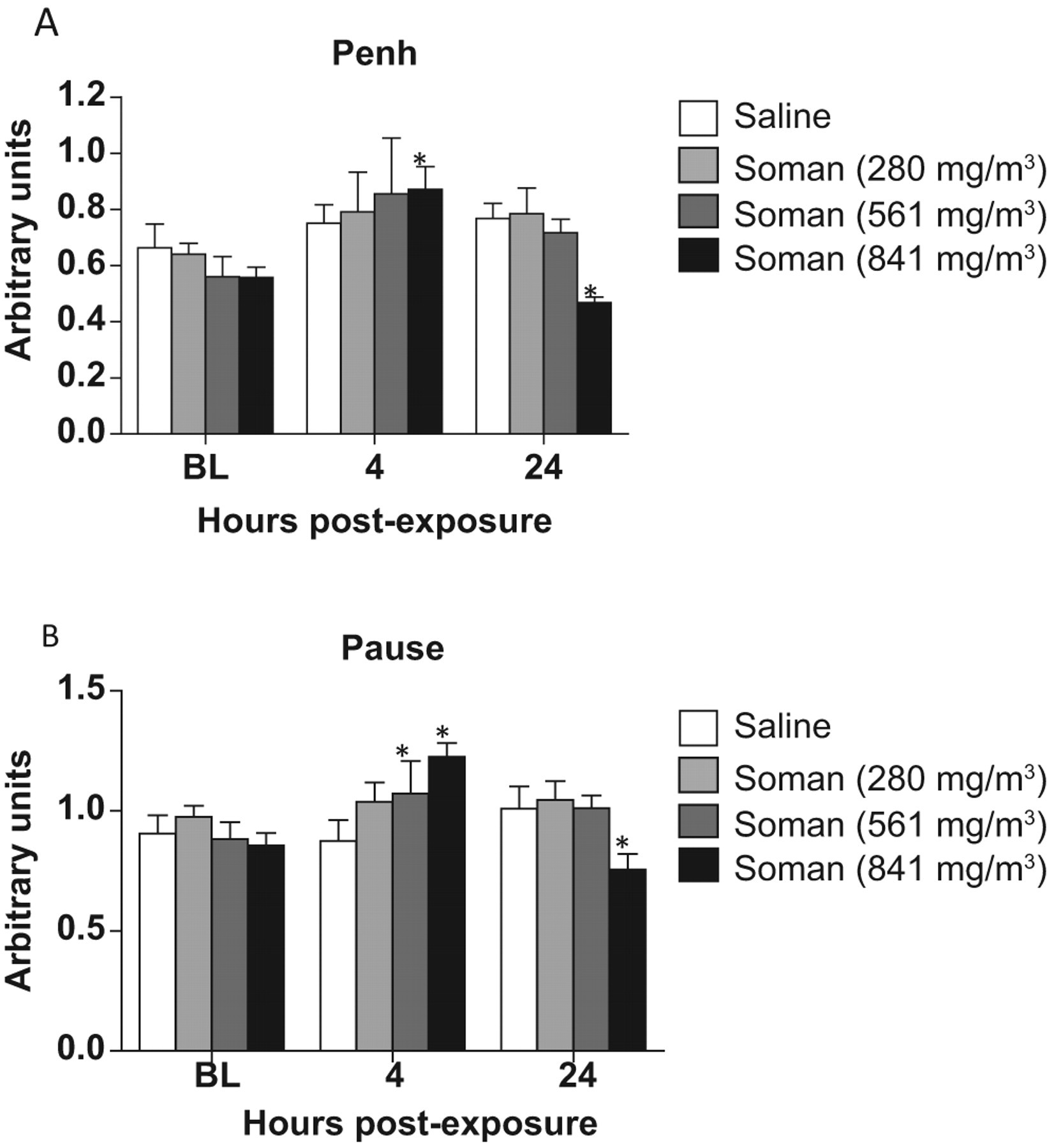
The effects of microinstillation inhalation exposure to soman (GD) on enhanced pause (Penh) and pause: Penh and pause were measured by whole-body barometric plethysmography in animals at 4 and 24 hours after GD exposure (n = 3-8). A significant increase at 4 hours and a significant decrease in Penh was observed in animals exposed to 841 mg/m3 of GD 24 hours postexposure. Similarly, results indicated a significant increase in pause 4 hours postexposure to 841 mg/m3 GD and a decrease 4 hours postexposure. Pause also significantly increased 4 hours postexposure to 561 mg/m3 of GD. Asterisk indicates statistical significance.
Discussion
In this study, our results show that acute microinstillation exposure to GD causes significant changes in various respiratory parameters that were dependent on the dose and time after exposure. No postexposure treatments were administered in this study in order to provide a more realistic view of the effects on pulmonary function following microinstillation inhalation exposure to GD. In addition, the respiratory dynamics were measured at an early time point, 4 hours post-GD exposure in conscious animals to evaluate early changes in respiratory function. A lethal dose and 2 lower doses of GD were used to determine the effects of GD on respiratory function. The results of this study should be valuable in the evaluation of therapeutics to counteract the respiratory toxicity as a result of inhalation exposure to GD.
We have previously reported the effects of GD exposure on respiratory dynamics in the presence of 2.5 mg/kg nasal atropine methyl bromide. 33 Microinstillation administration of nasal atropine methyl bromide 30 seconds after 841 mg/m3 GD exposure resulted in 70% survival in contrast to a survival rate of 38% observed in this study. 33 Atropine methyl bromide counteracts peripheral cholinergic effects and reduces CWNA-induced copious oronasal secretions in addition to bronchodilation that may prevent blockage of the airways and thereby increase survival. Exposure to 841 mg/m3 GD also changed several parameters of respiratory dynamics and function in atropine methyl bromide-treated guinea pigs at 24 and 48 hours postexposure in guinea pigs. 33 Atropine methyl bromide does not cross the blood-brain barrier and probably protects by reducing the peripheral nervous system effects, bronchodilation, and abundant airway secretion, indicating that local effects on the respiratory system also contribute to nerve agent-induced respiratory failure. 24,37 Atropine treatment has been shown to reduce respiratory toxicity induced by GD. 2,22,23,38 –40 Atropine is reported to protect against lower doses of GD by providing protection against CNS respiratory center effects. 2 We reported that endotracheal microinstillation administration of methyl atropine protects against lethal doses of CWNA exposure. 33 Many of the studies with GD were performed by different routes of administration and studies with inhalation exposure are limited. Route of administration has been shown to produce different effects on the respiratory toxicity. 7
Guinea pigs showed significant body weight loss at 841 mg/m3 with less variability between animals unlike atropenized animals. 33 Postexposure treatment with atropine methyl bromide seems to reduce body weight loss, while untreated animals had a significant increase in body weight loss 24 hours postexposure. Wet/dry weight ratio of the accessory lung lobe showed significant edema in untreated guinea pigs unlike insignificant changes in atropenized guinea pigs, suggesting that nasal atropine methyl bromide affords significant protective effects to the respiratory system without the involvement of central respiratory effects. Barometric WBP recordings showed more severe Cheyne-stokes breathing pattern at 4 hours compared to breathing of atropenized guinea pigs at 24 hours after 841 mg/m3 GD exposure. Soman exposure appears to cause more severe effects on respiratory physiology at 4 hours after exposure. Whole-body plethysmography data also showed that the magnitude of respiratory changes is stronger at 4 hours compared to 24 hours. Multiple factors, including airway secretions, peripheral cholinergic effects, seizure-related effects, and local effects on the nonneuronal cholinergic system present in the respiratory system, in addition to the central respiratory disturbance seems to contribute to initial respiratory toxicity. 41,42 Chemosensory effects also proposed to contribute to CWNA-induced respiratory toxicity and may contribute to the acute effect following GD exposure. 43
With regard to the dose of GD, the most notable changes were observed in animals exposed to 841 mg/m3 compared to 280 or 561 mg/m3 GD exposures. Although not significant, mean MV, PIF, PEF, Ti, Te, EEP, pause, and Penh values showed dose-dependent changes with 280 or 561 mg/m3 GD at 4 hours post-GD exposure that was reduced at 24 hours postexposure. Animals exposed to 280 and 580 mg/m3 recover quickly compared to the animals exposed to 841 mg/m3, suggesting that the effect of lower doses of GD on the CNS respiratory center is reversible. Moreover, it indicates that a lethal dose of GD is required to produce significant long-term changes in respiratory function and animals exposed to lower doses retain the ability to recover from GD-induced respiratory toxicity. It has been reported that lower doses of organophosphate, paraoxon, lead to respiratory disturbances, and ventilator effects of paraoxon are mediated by central effects rather than peripheral effects. 44
Exposure to 841 mg/m3 GD resulted in significant increases in RF, TV, and MV at 4 hours, which were reduced at 24 hours, but still significant. Atropenized guinea pigs also showed similar results. 33 Increased minute ventilation, TV and RF has been reported at early time points following subcutaneous administration of 2 x LD50 GD. 23 Although PIF and PEF increased in animals after 841 mg/m3 GD exposure at 24 hours, in atropenized animals PEF was reduced at 24 hours after GD exposure, indicating that atropine treatment reduces the recoiling capacity of the lungs after GD exposure. Ti and Te were reduced in both untreated and nonatropenized animals exposed to 841 mg/m3 GD at 24 hours postexposure. There was no significant change in EEP at 4 hours, but there was a decrease at 24 hours in the 841 mg/m3 GD-exposed group. End-expiratory pause increased at 280 and 561 mg/m3 GD at both 4 and 24 hours but was decreased at 841 mg/m3 GD exposure. The increase in EEP at lower doses of GD suggests that at this concentration GD may be stimulating the central respiratory center and may not be inducing central respiratory depression. GD at 841 mg/m3 leads to central respiratory depression that may in turn lead to rapid breathing initially, a compensatory effect for the metabolic demand and decreased EEP. On the other hand, it has been reported that lower doses of GD act primarily through CNS respiratory center effects and higher doses involve both CNS respiratory center and peripheral effects. 2 The opposite effects of low and higher doses of GD on EEP need to be further investigated. End-expiratory pause was increased following VX exposure that produces less central nervous system seizures following exposure. 10
Penh and pause, assumed measures of bronchoconstriction, significantly increased at 4 hours after 841 mg/m3 GD exposure. Conversely, at 24 hours, there was a significant reduction in Penh and Pause in 841 mg/m3 GD-exposed animals. A very similar reduction was observed in atropenized guinea pigs. 33 These results show that GD induces bronchoconstriction at earlier time points and bronchodilation at 24 hours. It should be noted that pulmonary function was investigated by Penh as a measure of “pseudo-lung resistance” a derived measurement of pulmonary function testing. Other effects on the CNS could also alter derived Penh measurements. Increased airway secretions immediately after GD exposure in addition to cholinergic effects may contribute to increased lung resistance and airway obstruction. In addition, ensuing lung edema may lead to bronchoconstriction at earlier time points. Clearly, there is a dichotomy of GD effects on bronchoconstriction at early and later time points. It seems that multiple mechanisms are contributing to the bronchoconstriction at early time points after GD exposure. Prevention of airways hypersecretions and other toxic responses should reduce the bronchoconstriction and aid in reducing respiratory toxicity and respiratory failure following nerve agent exposure.
Bronchodilatory effects at 24 hours may be a consequence of the initial respiratory toxicity following GD exposure. Increased RF and TV at early time points due to compensatory effect and possible hyperinflation of the lung immediately following GD exposure may reduce the recoiling capacity of the lungs. Accumulated volume increased in a dose-dependent manner at 4 hours following exposure to 841 mg/m3 GD and was reduced at 24 hours (data not shown). A reduction in copious airway secretions, fluid accumulation, edema, the weakening consequences of GD on the respiratory system as well as long-term effects of GD might be contributing to the observed bronchodilation. Accumulation of acetylcholine due to GD exposure may lead to stronger chest muscle contraction, expansion of thoracic cavity, and increased TV at early and late time points. We have reported the bronchodilatory effects following sarin exposure at 24 hours. 45
Higher doses of GD are required for the induction of significant respiratory changes. Similar to this study, it has been reported that subcutaneous paraoxon exposure results in significant respiratory disturbance at higher doses compared to lower doses. 46 However, respiratory changes such as RF are different between paraoxon and GD and also paraoxon-induced respiratory toxicity is not protected by methyl atropine. On the other hand, treatment with atropine that goes to the brain reversed paraoxon induced respiratory changes indicating that central respiratory effects are the main cause of toxicity. Pathophysiology of respiratory failure following acute dichlorvos exposure involves sequential CNS respiratory disturbance-induced apnea and subsequent pulmonary impairments with prominent airway secretions. 47 The central respiratory depressant diazepam protected against dichlorvos-induced respiratory depression and combination of diazepam and a peripheral anticholinergic showed more profound protection. 48 Surgical vagotomy results in more rapid effects on ventilation feedback from the vagus nerve, which may be an important aspect of respiratory toxicity. 49
In summary, these studies indicate that GD exposure induces dose-dependent changes in pulmonary function that are significant at 841 mg/m3 GD at 4 hours and remains up to 24 hours postexposure. This is the first study in which respiratory toxicity following GD microinstillation inhalation exposure was evaluated without atropine or oxime treatment. Overall, some of the effects of GD on respiratory dynamics were similar between untreated and previously evaluated atropenized animals, but there are many differences. Administration of lower doses of methyl atropine bromide seems to have a protective effect and reduces the magnitude of respiratory functional changes. Higher doses of GD are required for the induction of significant respiratory changes. Furthermore, we report that only at 4 hours or earlier time points, GD induces bronchoconstriction, while at 24 hours after exposure, GD induces bronchodilation. The mechanism of bronchoconstriction possibly involves copious airway secretion, lung edema, and ongoing lung injury in addition to cholinergic effects. On the other hand, the mechanism of bronchodilation may be the consequence of initial compensatory mechanisms, reduction in edema, lung injury, and gradual recovery from the toxic effects. A reduction in respiratory toxicity and bronchoconstriction at early time points after GD or organophosphate exposure by nasal therapeutics may serve as a potential countermeasure to increase survival, reduce acute toxicity, bronchodilatory effect and long-term pulmonary toxicity.
Footnotes
Acknowledgements
The project was supported by Defense Threat Reduction Agency. Its contents, opinions, and assertions contained herein are private views of the authors are not to be construed as official or reflecting the views of the Department of the Army or the Department of Defense.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: Defense Threat Reduction Agency, proposal # 3.F0006_08_WR_C.
