Abstract
The protective efficacy of the antimuscarinic agent scopolamine was evaluated against soman (o-pinacolyl methylphosphonofluoridate [GD])-induced respiratory toxicity in guinea pigs. Anesthetized animals were exposed to GD (841 mg/m3) by microinstillation inhalation exposure and treated 30 seconds later with endotracheally aerosolized scopolamine (0.25 mg/kg) and allowed to recover for 24 hours. Treatment with scopolamine significantly increased survival and reduced clinical signs of toxicity and body weight loss in GD-exposed animals. Analysis of bronchoalveolar lavage (BAL) fluid showed normalization of GD-induced increased cell death, total cell count, and protein following scopolamine treatment. The BAL fluid acetylcholinesterase and butyrylcholinesterase levels were also increased by scopolamine treatment. Respiratory dynamics parameters were normalized at 4 and 24 hours post–GD exposure in scopolamine-treated animals. Lung histology showed that scopolamine treatment reduced bronchial epithelial and subepithelial inflammation and multifocal alveolar septal edema. These results suggest that aerosolized scopolamine considerably protects against GD-induced respiratory toxicity.
Keywords
Soman (pinacolyl methylphosphonofluoridate [GD]), a chemical warfare nerve agent (CWNA), exerts toxicity by irreversibly inhibiting the enzyme acetylcholinesterase (AChE) that degrades the neurotransmitter acetylcholine. Increased accumulation of acetylcholine at the synapse produces cholinergic effects including central nervous system (CNS) seizure/status epilepticus, hypersecretions, bronchoconstriction, respiratory depression, and muscular tremors. 1 Inhalation is predicted to be the most likely route of exposure if CWNAs are used in war or terrorism. 2 Respiratory failure is the most common cause of death following CWNA exposure. A combination of CNS respiratory center depression and peripheral effects play roles in CWNA-induced toxicity. The extent of CNS respiratory depression versus the peripheral effects contributing to the development of respiratory failure is ambiguous. Most of the published data suggest that CNS effects are predominant. 1,3 –8 Furthermore, the extent of contribution of CNS respiratory depression and peripheral effects varies with the species studied, CWNAs used, and the dose of the agent involved. 8
Peripheral effects of CWNA mainly involve peripheral neuromuscular effects including effects on the phrenic nerve and diaphragm. It has been suggested that peripheral neuromuscular effects are developed only at higher doses of CWNA exposure unlike CNS respiratory depression. 1,5 Effects of CWNAs on respiratory muscles also contribute to the respiratory toxicity. However, activity of the diaphragm is retained after the termination of spontaneous respiration following CWNA exposure, indicating that respiratory failure may not be the consequence of diaphragm inactivation. 1,3,9 After CWNA exposure, spontaneous respiration is restored in surviving animals with low AChE activity, suggesting that AChE inhibition is not solely responsible for the CNS respiratory depression. 10
The route of exposure to CWNA contributes to the difference in toxicity probably due to variations in agent absorption distribution, and binding to other nonspecific sites. 11 For instance, studies have shown discrete differences in lung function following intravenous administration of GD in comparison to inhalation exposure. 3,12 Nevertheless, respiratory toxicity has been observed in animals exposed to GD by different routes. Intratracheal administration of GD caused respiratory changes. 11 Inhalation exposure to GD has been reported to cause significant respiratory toxicity and respiratory failure in different species. 4,12 –14 Intravenous infusion of GD that has been described as similar to inhalation exposure also induces acute respiratory toxicity. 1,3,6,11 The mechanisms of GD-induced respiratory toxicity are not very clear and need further investigation. We reported that CWNA directly affects the respiratory system and that affects partly contribute to respiratory failure. 13,15,16 Copious airway secretion induced by CWNA may block the bronchiolar tree, reduce oxygen exchange, and promote respiratory failure. 17 In addition, acetylcholine is reported to play diverse functional roles in nonneuronal cells of the lungs. 18,19 The existence of the nonneuronal cholinergic structure in the respiratory system hints that inhalation exposure to CWNA may directly affect the pulmonary system and its function. 18,19
Considering the complexity in whole-body and nose-only inhalation exposure systems, we developed a microinstillation inhalation exposure technology to study the mechanisms of CWNA-induced respiratory toxicity and develop targeted therapies for protection against CWNA exposure. 20 Microinstillation inhalation exposure involves endotracheal aerosolization of the CWNA by a microcatheter which has 5 peripheral holes to pump air that aerosolizes the agent delivered through a central hole at the tip. 21 The technology requires minimal amount of CWNA and has advantages of accurate dosing and simple operation. Aerosolizing the agent endotracheally prevents the potential detoxification of the CWNAs by carboxylesterases present in the upper airways of rodents and minimizes external aerosol release. 22 –24 Microinstillation inhalation exposure showed that CWNA causes acute lung injury in guinea pigs. 13,25,26 Guinea pigs have low levels of carboxylesterases and sensitive airways that undergo bronchoconstriction and are considered one of the best nonhuman primate models for studying CWNA toxicity and therapeutic development. 24,27 –31
Scopolamine is an anticholinergic/muscarinic drug that crosses the blood–brain barrier better than atropine sulfate and has been evaluated for protection against organophosphate exposure involving CNS toxicity. 32 It is a potent antisecretory agent being used to prevent motion sickness. 33,34 Scopolamine has been investigated as an antidotal treatment for CWNA exposure and is reported to protect better than atropine. 35 Prophylactic treatment with scopolamine combined with physostigmine protected against 2× LD50 sarin exposure. 36 Intramuscular administration of scopolamine provides protection following GD exposure and is more effective in reducing seizures and neuropathology than other anticonvulsants. 37 –45 Efficacy studies of aerosolized scopolamine against CWNA exposure are limited. Bioavailability studies suggest that the intranasal route may provide a noninvasive, rapid, and effective way for administering scopolamine. 46 –48 Considering the superior ability of scopolamine to cross the blood–brain barrier, aerosolized scopolamine may provide enhanced protection by counteracting both central respiratory disturbance and peripheral nervous system effects including respiratory toxicity induced by GD.
Traditionally, the regimen of pyridostigmine bromide to shield AChE in the periphery from irreversible inhibition with the anticholinergic drug atropine sulfate, which antagonizes the effects of the excess ACh and an oxime, 2-pralidoxime (2-PAM), for reactivation of unaged AChE is not sufficient to protect against nerve agents. 41,49 Anticonvulsant diazepam is also administered to control CWNA-induced seizure activity. 50 The short half-life of aging makes it hard to successfully treat GD toxicity with the standard therapy of antimuscarinic antidotes and AChE reactivator at later time points after exposure. 51 –53 Since aged AChE cannot be reactivated by oximes, immediate treatment is critical for seizure control and effective medical countermeasure against GD exposure. 54,55
The purpose of this study is to evaluate the efficacy of postexposure treatment by aerosolized scopolamine to protect against GD inhalation exposure using the microinstillation inhalation exposure system in guinea pigs. Survival, pulse rate, blood O2 saturation, lung and tracheal edema, body weight loss, biochemical analysis of blood, bronchoalveolar lavage (BAL) cells and fluid, respiratory dynamics, and lung histopathology were used for efficacy evaluation. The data demonstrate that postexposure treatment with scopolamine protects against respiratory toxicity, normalizes respiratory function, and dramatically increases survival of GD-exposed animals.
Methods
Animals
Adult male Hartley guinea pigs (250-300 g; Charles River Laboratories, Wilmington, Massachusetts) were housed individually under standard conditions with 12-hour light/dark cycle and food and water available ad libitum. Research was conducted in compliance with the Animal Welfare Act and other federal statutes and regulations relating to animals and experiments involving animals. It adheres to principles stated in the Guide for the Care and Use of Laboratory Animals, National Research Council, published by the National Academy Press, 1996, and the Animal Welfare Act of 1966, as amended. The study protocol was approved by the Institution Animal Care and Use Committee, USAMRICD, Edgewood, Maryland. A total of 34 guinea pigs were used for this study.
Chemicals
Soman (pinacolyl methylphosphonofluoridate or GD) was obtained from Edgewood Chemical and Biological Center, Edgewood, Maryland, and diluted in fresh sterile saline and kept frozen at −80°C until use. The GD dilutions were periodically verified for the amount of agent. Telazol was purchased from Wyeth Pharmaceuticals (Madison, New Jersey) and Meditomidine was from Pfizer Pharmaceuticals (New York, New York). Scopolamine, heparin, acetylthiocholine iodide (ATCh), tetraisopropylamidopyrophosphate (Iso-OMPA), butyrylthiocholine iodide (BTCh), and huperzine A were obtained from Sigma-Aldrich (St Louis, Missouri). Microinstillation equipment and microcatheter were obtained from Trudell Medical (Ontario, Canada).
O- Pinacolyl Methylphosphonofluoridate Microinstillation Inhalation Exposure
Animals were exposed to GD by microinstillation inhalation exposure as described earlier. 13 Anesthetized animals (telazol, 40 mg/kg, intramuscularly [im], and medetomidine, 0.125 mg/kg, subcutaneously [sc]) were intubated with a 10-cm piece of translucent polystyrene tube. 21 GD (841 mg/m3) was aerosolized using a microinstillation catheter passed through the intubation tube and placed 2 cm above the bifurcation of the trachea. Each pulse aerosolized 1.4 μL of GD in a volume of 1.1 mL air, and 40 pulses/min for 2 minutes were used.
Scopolamine Treatment
After GD exposure, a different microcatheter prepulsed with scopolamine was inserted through the intubation tube 30 seconds later and scopolamine was delivered within 1 minute at 40 pulses. Each pulse was equivalent to 1.4 µL of the therapeutic agents in 1.1 mL air to obtain a final dose of 0.25 mg/kg. The animals were kept in the hood for 15 minutes before taking them out and placing them in new polycarbonate cages for 24-hour evaluation. Development of cholinergic signs, convulsions, and breathing variations were monitored for 2 hours.
Blood O2 Saturation and Pulse Rate
Blood O2 saturation and pulse rate of guinea pigs were monitored using a pulse oximeter (Noni Medical IN, Minneapolis, Minnesota). Readings were taken at pre-/postintubation and every 30 seconds during GD exposure/treatment for a total of 15 minutes and plotted against time.
Body Weight Loss
To calculate the percentage of body weight loss, the guinea pigs were weighed before administering anesthesia and just prior to euthanasia at 24-hour postexposure.
Trachea and Lung Edema
The lung accessory lobe was used to determine the lung wet/dry weight ratio with and without scopolamine treatment. Survived animals were deeply anesthetized and euthanized by exsanguination. Since, lavage, tissues, and edema data from each animal were used to evaluate scopolamine efficacy, the accessory lung lobe was tied off using surgical string before lavage to prevent fluid entry. The accessory lobe and a 1 cm piece of trachea were removed, weighed using an analytical balance, and placed in 100°C dry heat for 7 days. The wet/dry weight ratio was determined to evaluate the degree of lung and trachea edema.
Bronchoalveolar Lavage
Bronchoalveolar lavage was collected by lavaging the lungs 3 times in and out with 7 mL of oxygen-free saline and then placed on ice. The average BAL volume recovered was 5.0 mL ± 8%. Bronchoalveolar lavage cells were separated from BAL fluid by centrifugation at 2000 rpm for 10 minutes using a Sorvall tabletop centrifuge. Total protein, AChE, and BChE activities were determined immediately.
Bronchoalveolar Lavage Cell Counts
Bronchoalveolar lavage cells were diluted 20-fold with trypan blue dye and cells were counted on a Nebauer cell counting slide at ×10 magnification using a phase-contrast microscope (Olympus IX51, Melville, New York). The total numbers of viable and dead cells were counted in triplicates.
Blood and BAL Fluid AChE and BChE Activity
Activities of both AChE and BChE in the blood (diluted 50-fold using deionized water) and in BAL fluid were determined using Ellman's assay with acetylthiocholine iodide (ATCh) and butyrylthiocholine iodide (BTCh) as the substrates. For the determination of BChE activity using BTCh substrate, samples were incubated at room temperature for 30 minutes in the presence of AChE inhibitor huperzine A (400 nmol/L). Similarly, AChE activity was determined using substrate ATCh after incubating the samples for 30 minutes with BChE inhibitor iso-OMPA (4 μmol/L). Activities of AChE and BChE were expressed as Vmax normalized to optical density (OD) of hemoglobin measured at 412 nm. All spectrophotometric readings were taken on a microtiter plate reader by Spectra Max and Softmax Plus 4.3 LS software (Molecular Devices, Sunnyvale, California). The bronchoalveolar lavage fluid (BALF) AChE and BChE activities were determined using 20 µL fluid and expressed as Vmax.
Protein Analysis
Total protein levels in the BAL fluid were determined by a bicinchoninic acid (BCA) protein assay (Pierce Chemical Company, Rockford, Illinois) in triplicates. The average volume of BAL recovered was 5.0 mL ± 8%. Twenty microliters of BAL fluid were used for protein assay.
Respiratory Dynamics
Barometric whole-body plethysmography (Buxco Electronics, Inc, Sharon, Connecticut) was used to measure respiratory dynamics in conscious, unrestrained guinea pigs as described previously. 56,57
Lung Histopathology
Different groups of animals were used for histopathology. The lungs and trachea were infused and fixed in 4% formaldehyde solution after necropsy. Tissues were embedded in paraffin wax, sectioned at 5 µm thickness, placed on glass slides, and stained using hematoxylin and eosin. Two cross-sections of each of the 7 lung lobes (right cranial, right medial, right caudal, right accessory, left cranial, left medial, and left caudal) were examined and an average score for each lobe was determined. The sections were taken at approximately one third and two thirds the length of each lobe. Each lobe was evaluated for the amount and distribution of inflammatory cells in the bronchointerstitial areas, and if present, whether the inflammation was centered on bronchi/bronchioles (bronchial to peribronchial), or more randomly distributed throughout the alveoli (bronchoalveolar). The number of inflammatory cells within the bronchial epithelium and lumen, edema within the alveolar septae, fibrin and/or mucin within alveolar spaces, and alveolar hemorrhage was also evaluated. Lesions were graded on the following scale: 0 = no change, 1 = minimal (less than 10% of section affected), 2 = mild (11%-24%), 3 = moderate (25%-49%), 4 = marked 50%-74%), 5 = severe (75%-100%).
Statistical Analysis
Statistical analyses were performed using the Graph Pad Prism V4.03 software (Graph Pad Software Inc, San Diego, California). The t test was used to calculate the P values, and a P value less than .05 was considered significant.
Results
Survival After GD Microinstillation Inhalation Exposure and Scopolamine Treatment
The survival rate of guinea pigs following microinstillation inhalation exposure to saline, 841 mg/m3 GD, and GD treated with scopolamine is shown in Table 1 . Animals exposed to saline (n = 10) showed no signs of toxicity and all of them survived. A total of 31% of the animals exposed to GD (n = 16) survived. A total of 16 animals were exposed to 841 mg/m3 GD, to obtain adequate number of surviving animals that would provide statistical significance of the various toxicological parameters analyzed. Postexposure treatment with scopolamine (n = 8) resulted in 100% survival of guinea pigs exposed to GD.
Survival (24 Hours) of Guinea Pigs Following Postexposure Treatment With Scopolamine
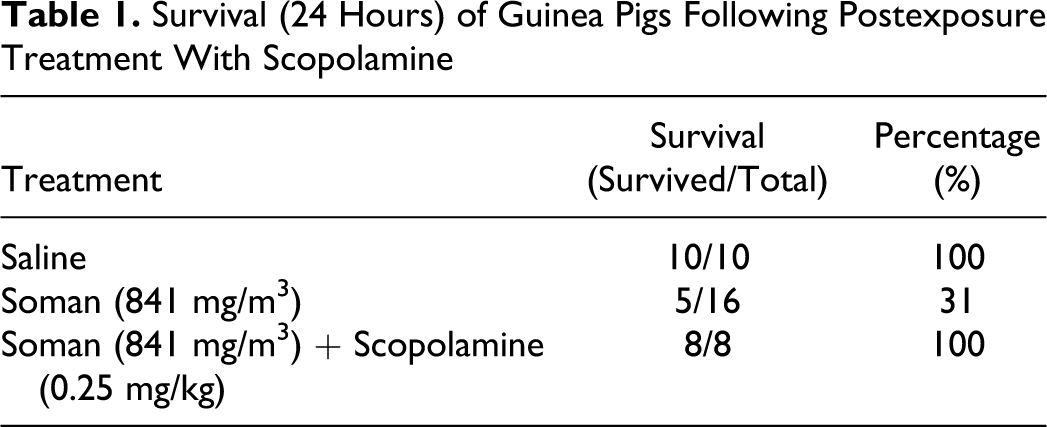
Of the 16 guinea pigs exposed to GD, 11 developed convulsions immediately after the 2-minute exposure and died 15 to 30 minutes post-GD exposure. Convulsions were not observed in many of the scopolamine-treated animals. The animals that produced convulsions at the end of the exposure and were treated with scopolamine survived 24 hours, indicating that treatment with endotracheal scopolamine increases the length of survival time in GD-exposed animals that exhibit convulsions. Reduction in saliva and lacrimal secretion were visible in animals exposed to GD and treated with scopolamine compared to GD controls. Scopolamine-treated animals woke up quickly from anesthesia and showed better recovery than control GD-exposed animals.
Blood O2 Saturation and Pulse Rate
The blood oxygen saturation (SpO2) for saline, GD-exposed, or GD-exposed and scopolamine-treated animals is shown in Figure 1A. Exposure to GD resulted in 50% reduction in SpO2 from 4 to 5.5 minutes after GD exposure that did not return to normal levels within the 15-minute observational period. Animals exposed to GD and treated with scopolamine showed a 35% reduction in SpO2 from 4.0 to 4.5 minutes that returned to values similar to saline control groups. Scopolamine-treated animals maintained nearly 100% SpO2 during the 15-minute observational period in comparison to untreated GD-exposed animals.
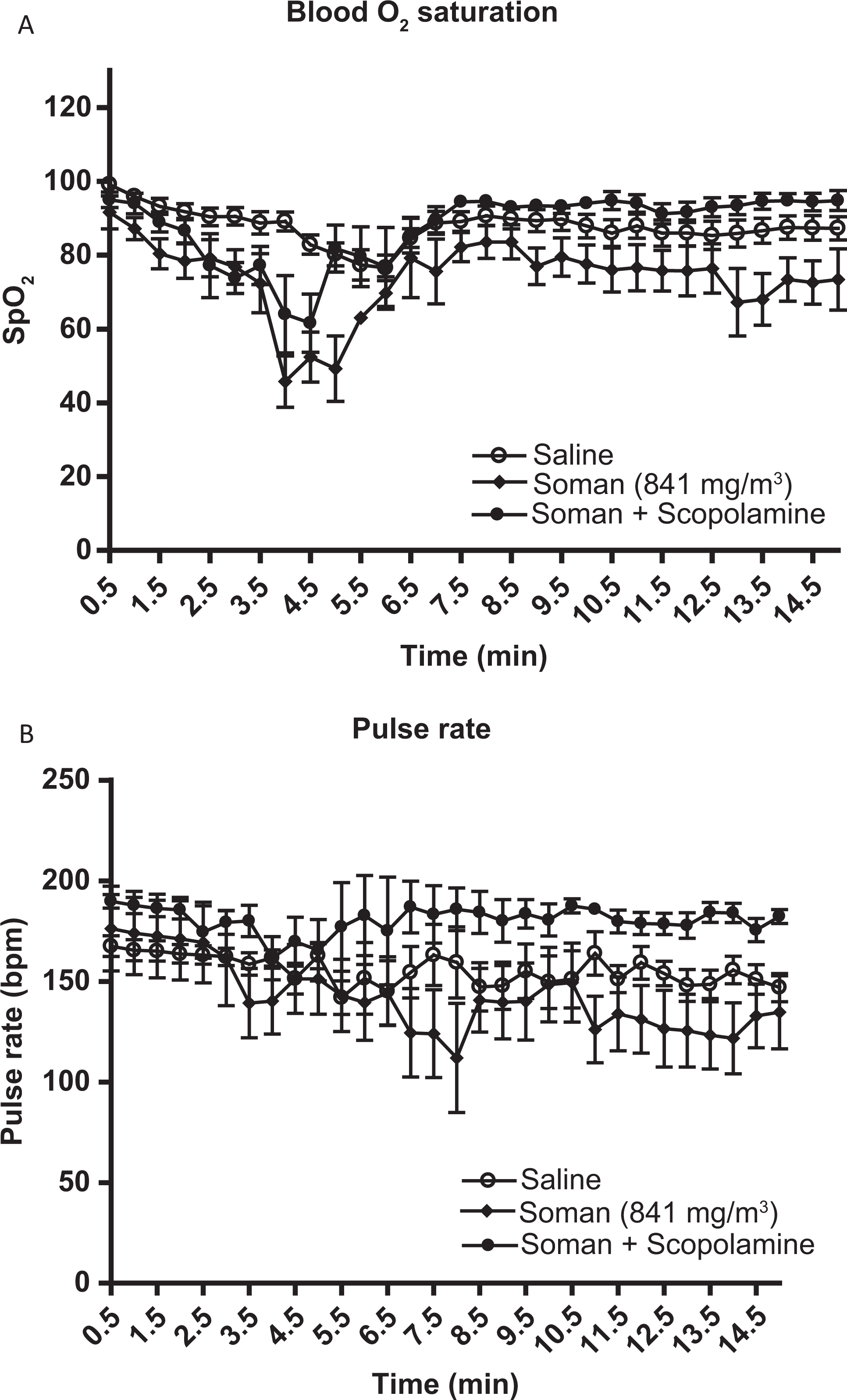
Blood oxygen saturation and pulse rate. The blood oxygen saturation (SpO2) and pulse rate (A; B) were recorded continuously every 30 seconds for saline, scopolamine treated and soman (841 mg/m3)-exposed animals for a total of 15 minutes. A, Blood O2 saturation was plotted against time; saline (n = 10), 841 mg/m3 GD (n = 5), and 841 mg/m3 GD treated with scopolamine (n = 8). B, Pulse rate (bpm) was plotted against time; saline (n = 10), 841 mg/m3 GD (n = 5), and 841 mg/m3 GD treated with scopolamine (n = 8). Asterisk indicates statistical significance.
The pulse rate of saline-treated, GD-exposed, and GD-exposed animals treated with scopolamine is shown in Figure 1B. Saline controls showed a stable pulse rate throughout the 15-minute examination period. There was a decrease in pulse rate that fluctuated but was prominent at 3.5 to 8.5 minutes in animals exposed to GD, which did not return to normal values during the 15-minute recording. Animals exposed to GD and treated with scopolamine maintained baseline levels of pulse rate and did not show any decrease in pulse rate. Conversely, the pulse rate was increased slightly above saline controls over the 15-minute observational period in scopolamine-treated animals.
Body Weight Loss
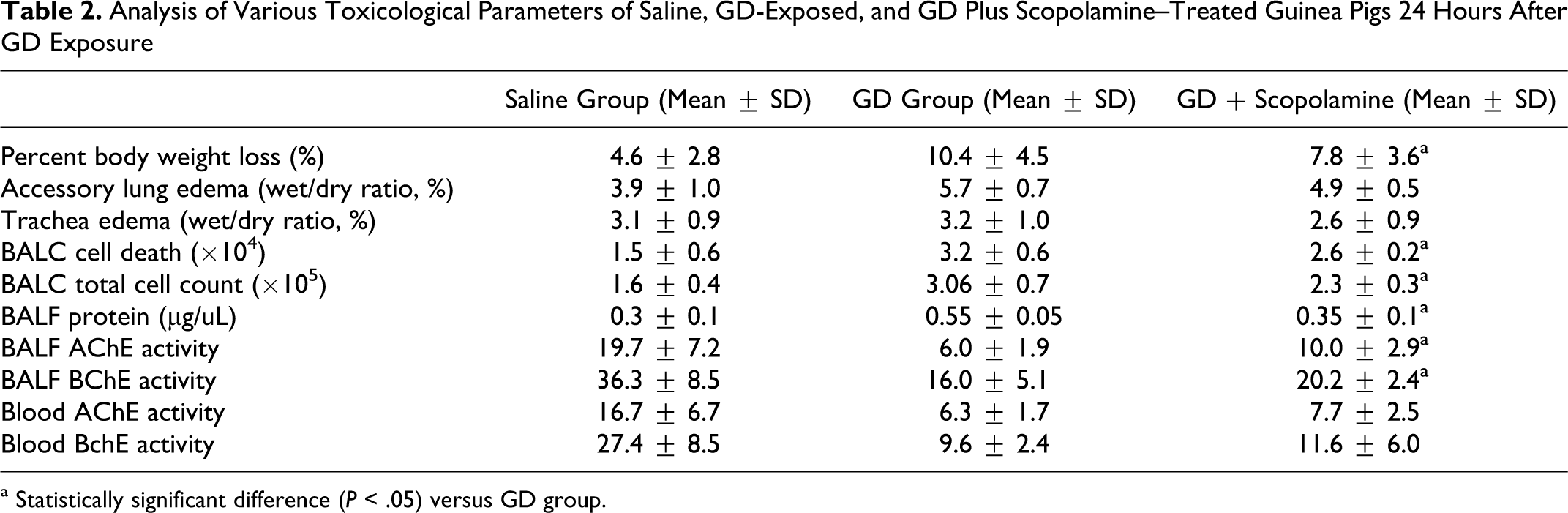
Animals exposed to GD showed an 8.5% decrease in percentage body weight loss, while saline controls lost 3.8% at 24 hours. There was a significant (P = .034) reduction in the percentage body weight loss in animals exposed to GD and treated with scopolamine compared to GD-exposed animals (Table 2 ). Food and water consumptions were not recorded in this study.
Analysis of Various Toxicological Parameters of Saline, GD-Exposed, and GD Plus Scopolamine–Treated Guinea Pigs 24 Hours After GD Exposure
a Statistically significant difference (P < .05) versus GD group.
Trachea and Lung Edema
The accessory lung lobe and trachea edema represented as a ratio of wet/dry weight is shown in Table 2. A consistent increase in accessory lung lobe edema observed in animals exposed to GD compared to controls that was slightly reduced by scopolamine treatment. Treatment with scopolamine resulted in minimal changes in trachea edema in animals exposed to GD at 24-hour postexposure.
Bronchoalveolar Lavage Cell Counts
Increased BAL cell death was observed in animals exposed to 841 mg/m3 GD (Table 2) and that was significantly (P = .048) decreased in animals exposed to GD and treated with scopolamine. A significant (P = .015) decrease in total BAL cell count (viable and dead) was also observed in animals exposed to GD and treated with scopolamine compared to GD-exposed animals.
Blood and BAL Fluid AChE and BChE Activity
Bronchoalveolar lavage fluid total protein in saline, GD-exposed, and GD-exposed animals treated with scopolamine are shown in (Table 2). A significant (P = .027) decrease in total BAL fluid protein was observed in animals exposed to GD and treated with scopolamine compared to GD controls. There was significant (P = .038 and .033) increase in BAL fluid AChE and BChE activities in animals exposed to GD and treated with scopolamine compared to GD-exposed animals (Table 2).
Cardiac blood AChE and BChE activities at 24-hour post–GD exposure is shown in Table 2. Significant inhibition of AChE and BChE activities was observed in untreated animals exposed to 841 mg/m3 GD. An insignificant increase in AChE and BChE activities was observed in animals exposed to 841 mg/m3 of GD and treated with scopolamine.
Respiratory Dynamics
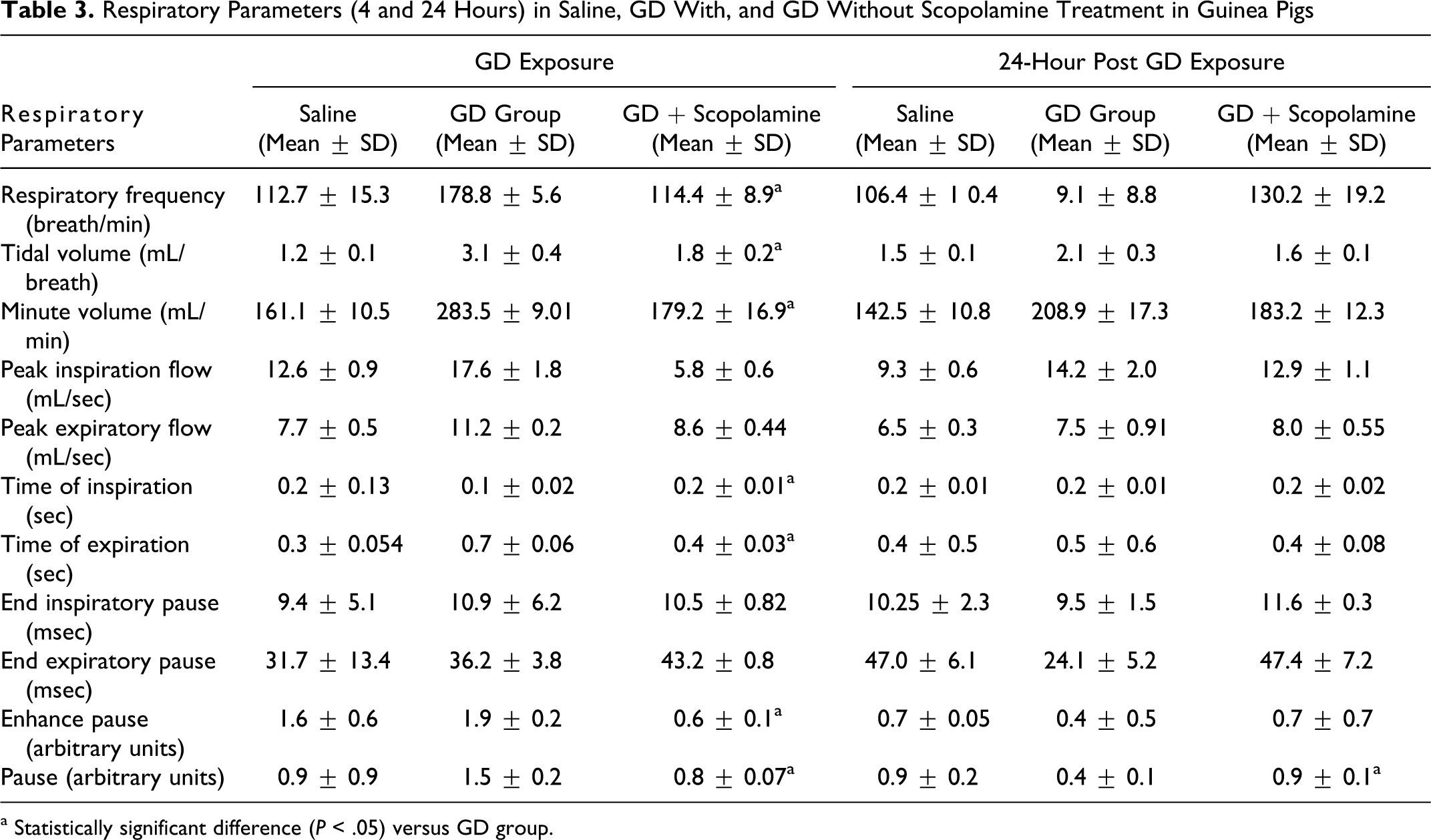
Respiratory dynamic parameters measured by whole-body plethysmography at 4 and 24 hours in guinea pigs exposed to saline, GD with, and GD without scopolamine treatment are listed in Table 3 . At 4-hour postexposure, animals exposed to GD and treated with scopolamine resulted in a significant decrease and normalization in respiratory frequency [RF] (P = .018), tidal volume [TV] (P = .006), and Minute Volume [MV] (P = .005). At 24 hours, the RF and TV also returned to normal levels in GD-exposed animals treated with scopolamine compared to GD controls. The maximum inspiratory (peak inspiratory flow [PIF]) and expiratory flow (peak expiratory flow [PEF]) that occurs in a breath were decreased at 4 and 24 hours in animals exposed to GD and treated with scopolamine compared to GD controls. Animals exposed to GD and treated with scopolamine showed a significant increase (P = .004) and normalization of inspiratory time (Ti) at 4-hour postexposure. A significant increase (P = .002) in the expiratory time (Te) was also observed at 4-hour postexposure in animals exposed to GD and treated with scopolamine compared to untreated GD-exposed animals.
Respiratory Parameters (4 and 24 Hours) in Saline, GD With, and GD Without Scopolamine Treatment in Guinea Pigs
a Statistically significant difference (P < .05) versus GD group.
Guinea pigs exposed to GD and treated with scopolamine resulted in a significant (P = .033) increase and normalization of end expiratory pause (EEP) at 24-hour postexposure compared to untreated GD-exposed animals. Results of Penh and pause showed a significant decrease in enhanced pause (Penh; P = .027) and pause (P = .006) at 4 hours in animals exposed to GD and treated with scopolamine in comparison to untreated GD-exposed animals. At 24 hours, scopolamine treatment resulted in significant increase in pause (P = .012) in GD-exposed animals compared to untreated GD-exposed animals.
Lung Histopathology
The majority of histopathological lesions in animals averaged minimal (score 1) or mild (score 2) with a few individual lung lobes in various animals exhibiting moderate (score 3) changes. The majority of inflammation was peribronchial and composed of heterophils and fewer macrophages, and many admixed eosinophils (Figure 2 ). Some animals had additional heterophils within the bronchial epithelium and within the lumen. The majority of animals in all groups had minimal hemorrhage and minimal-to-mild alveolar septal edema.
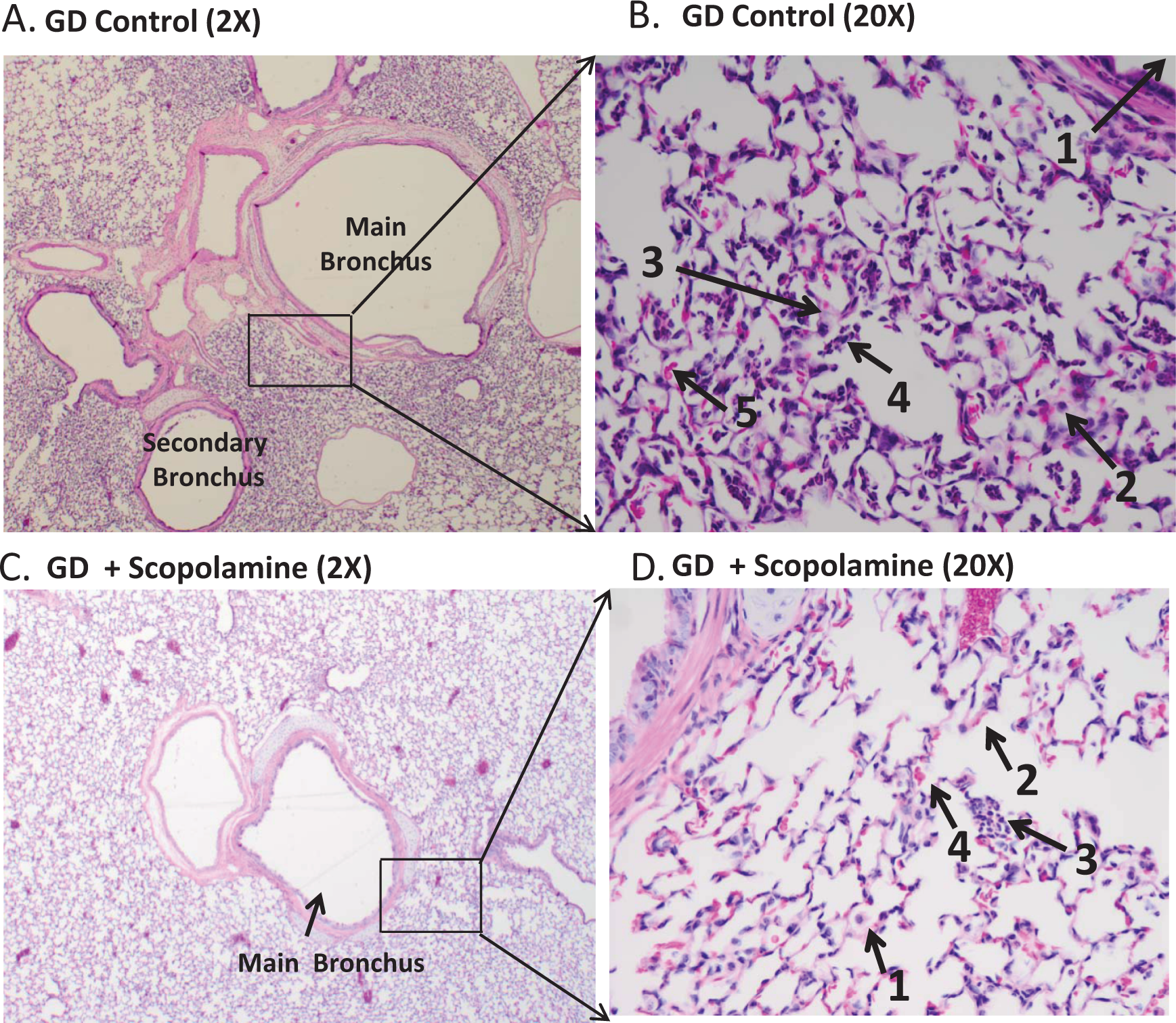
Scopolamine treatment reduces GD-induced lung histopathology. A, GD exposed, left caudal lung lobe (×2). Mild peribronchial heterophilic and histiocytic inflammation with scattered eosinophils, septal edema, and alveolar hemorrhage affecting 10% to 25% of the section. B, GD exposed, left caudal lung lobe (×20). Close-up view of Figure A. (1) main bronchus, (2) macrophage, (3) septal edema, (4) cluster of heterophils, and (5) erythrocytes. C, GD + scopolamine, left cranial lung lobe (×2). Minimal peribronchial heterophilic and histiocytic inflammation with scattered eosinophils, septal edema, and alveolar hemorrhage affecting <10% of the section. D, GD + scopolamine, left cranial lung lobe (×20). Close-up view of Figure C. (1) macrophage, (2) septal edema, (3) heterophils, and (4) erythrocytes.
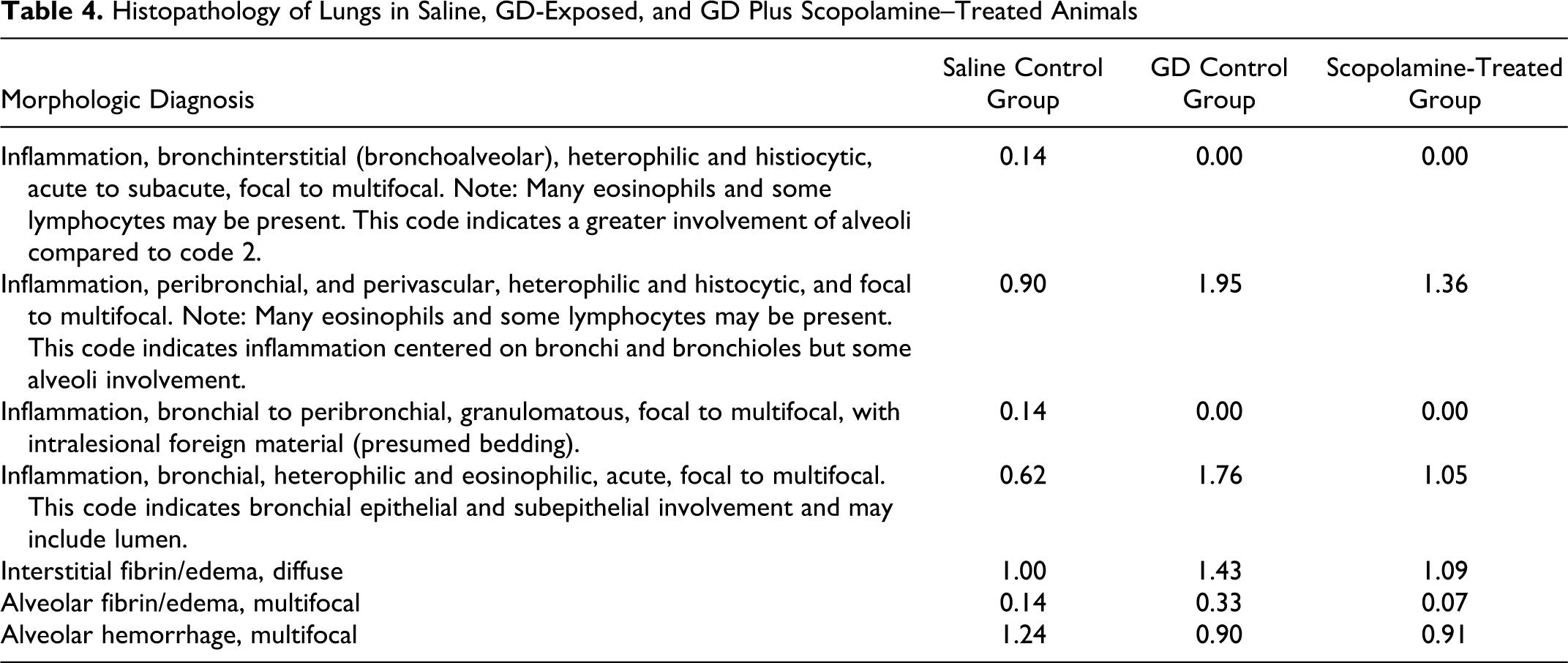
The scores for all lung lobes for all animals in a group were averaged together for a group score for each parameter evaluated and are listed in Table 4 . The scores for multifocal peribronchial inflammation averaged 0.90 for the saline control group (n = 3), 1.95 for the GD control group (n = 3), and 1.36 for the scopolamine-treated group (n = 4). Bronchial inflammation with/without luminal involvement averaged 0.62 for the saline group, 1.76 for the GD group, and 1.05 for the scopolamine group. Interstitial alveolar edema averaged 1.00 for the saline group, 1.43 for the GD group, and 1.09 for the scopolamine group. Alveolar hemorrhage averaged 1.24 in the saline group, 0.90 in the GD group, and 0.91 in the scopolamine group. Two animals in the saline group, 1 animal in the GD control group and 1 animal in the scopolamine-treated group had evidence of inhaled foreign bodies, assumed to be bedding material. The bedding induced inflammation was minimal in only the right accessory lobe and mild in the right cranial lobe in 2 saline-exposed animals. Mild-to-moderate inflammation in all lung lobes was observed in 1 of the GD controls, and minimal inflammation in the right cranial lung lobe of 1 of the scopolamine-treated animals. In this context, it is reasonable to speculate that the presence of foreign bodies increased the amount of inflammatory cell numbers and distribution, as well as alveolar septal edema, alveolar fibrin, and/or mucin in these animals. This artificially increased the average scores in the control and treated groups but the degree was minimal overall. A separate code for granulomatous inflammation was noted for those lobes with foreign material in the lumens, but the foreign bodies also affected the amount and distribution of inflammation and edema included in the other parameters, as well.
Histopathology of Lungs in Saline, GD-Exposed, and GD Plus Scopolamine–Treated Animals
Discussion
The results presented here demonstrate that postexposure treatment with endotracheally aerosolized scopolamine, an anticholinergic agent protects against lethal GD exposure and normalizes several end points of respiratory toxicity, dynamics, and lung injury. Scopolamine protected against respiratory toxicity when administered 30 seconds after 2-minute GD exposure. Administration of scopolamine at 1 minute after GD exposure may protect and increase survival but further delays in time may not be effective due to the rapid “aging” of GD-bound AChE and irreversible toxicity. 55,58 It is well known that short half-life of aging makes it difficult to successfully treat GD exposure with the available combination treatments including antimuscarinic antidotes. 59 Pretreatment with 1-2-hydroxyiminomethyl-1-pyridino-3-(4-carbamoyl-1-pyridino-2-oxapropane dichloride (HI-6) that prolongs survival will allow evaluating the efficacy of scopolamine at later time points after soman exposure. Protection by scopolamine was observed in the absence of any additional therapeutic agents. Also, the guinea pig model used in this study was not treated with atropine or oxime, suggesting that aerosolized scopolamine alone was effective against GD exposure. Endotracheal administration of an anticholinergic treatment alone, such as atropine or scopolamine, provides protection against CWNA exposure when administered immediately following nerve agent inhalation exposure. 15,17,73 Anesthesia has been reported to influence soman toxicity and the use of anesthesia might affect the results. 60 In this study, all the animals including untreated and scopolamine treated were anesthetized for comparison. It has been previously shown that ketamine protects against GD-induced neuropathology, while anesthetic doses of barbiturate increase GD toxicity. 60 –64 It also has been reported that intramuscular treatment of scopolamine protects against CWNA exposure. 35 , 36 , 41–45 , 65
In addition to the CNS anticholinergic effects, the mechanism of protection by aerosolized scopolamine may involve antisecretory effects and reduction in copious airway secretion and respiratory toxicity and peripheral nervous system effects. Previously, we demonstrated that endotracheal aerosolization of atropine methyl bromide that does not cross the blood–brain barrier protects against GD exposure, indicating that protection against peripheral nervous system effects and respiratory toxicity of GD is sufficient to provide significant increases in survival. 26 Delivery of therapeutic agents to the lungs and rapid prevention of copious airway secretion seems to improve breathing and reduce respiratory toxicity and thereby enhances survival. Furthermore, muscarinic receptor antagonism by systemically absorbed scopolamine could block the effects of increased ACh on muscarinic receptors in the CNS and reduce GD-induced hyperdepolarization, seizure development, and neuropathology. 41,44,45 Direct microinjection of drugs to various areas of the brain showed that scopolamine protects at different areas unlike anticonvulsants and N-methyl-D-aspartate receptor antagonists, suggesting that scopolamine is effective in various brain regions 66 –68 against CWNA exposure. Thus, scopolamine that can protect against respiratory toxicity, peripheral effects, and CNS effects will be a better protectant against GD toxicity than that which protects against respiratory toxicity alone.
The dose of scopolamine used in this study was 0.25 mg/kg. A range of 0.06 to 0.13 mg/kg delivered intravenously was required to stop nerve-agent-induced seizures. 39 Preliminary studies with increasing dose of scopolamine (0.5 mg/kg) did not show any improvements in the protective efficacy, rather the animals were found to be more toxic. Similarly, scopolamine at 0.25 mg/kg did not protect from higher doses of GD exposure. Thus, the protective effect of endotracheally aerosolized scopolamine is limited to the dose of the drug and concentration of the agent. Additional studies with more doses of the drug may be required for further optimization of the efficacy of endotracheal aerosolized scopolamine. Scopolamine has been reported to have toxic effects at higher dose. 46,69,70 For antisecretory effect and protection against respiratory toxicity, a smaller dose of scopolamine may be adequate. 46
Scopolamine treatment is not only effective in increasing the survival due to anti-secretory effects, but also reduces lung injury and improves respiratory functions. Treatment with scopolamine normalized the increase in enhanced pause at 4 hours after GD exposure, indicating a reduction in GD-induced bronchoconstriction. Scopolamine treatment prevented the reduction in enhanced pause or bronchodilation observed at 24-hour post–GD exposure, suggesting that it possibly prevents air trapping and inflation of the lungs early due to GD exposure. Although the number of animals was limited, scopolamine treatment also reduces bronchial epithelial and subepithelial inflammation and multifocal alveolar septal edema. The scopolamine-treated animal group showed a decline in peribronchial and bronchial inflammation, interstitial alveolar edema, and alveolar fibrin/edema compared to the GD control group. The cause of the increased multifocal alveolar hemorrhage in the saline group was not apparent, but was very slight, and not considered a significant change. The effect of inhaled bedding was minimal in the saline and scopolamine groups.
The activity of BALF AChE in animals treated with scopolamine after GD exposure was increased compared to GD controls. More studies are required to explain this phenomenon. It might be due to enhanced degradation and elimination of GD in scopolamine-treated animals compared to controls. Restoration of normal breathing and improved blood O2 saturation in animals treated with scopolamine may promote the degradation and elimination of GD and its metabolites from the body compared to soman-exposed animals that are untreated. Muscular contraction leads to increased AChE synthesis and that could contribute to the increased BALF AChE levels following treatment with nasal therapeutic agents post GD exposure. 71 Other possible reasons include increased synthesis or recruitment of AChE from extracellular fluid to the BALF. However, de novo synthesis of AChE is slow and may not solely account for the observed increase in the activity in scopolamine-treated animals. We have reported earlier an increase in BALF ChE after exposure to nonlethal doses of VX possibly due to air–blood barrier damage and leakage from the plasma. 72 Unpublished data from the laboratory also show higher ChE activity in the BALF following treatment with other nasal therapeutic agents. Blood levels of AChE and BChE consistently increased in scopolamine-treated animals, indicating a better recovery from GD exposure.
The exact mechanism of respiratory depression following GD exposure is still not well understood, although it is known that a combination of central and peripheral effects are major contributors. 5 The nonneuronal cholinergic system (cholinergic system in nonneuronal cells such as epithelial cells) in the respiratory system plays many roles in pulmonary function, and scopolamine may counteract those nonneuronal cholinergic pathways in the lungs. 9,18 The nonneuronal cholinergic system is involved in air–blood barrier function, and endotracheal administration of aerosolized scopolamine might be reversing the cholinergic effects produced by GD on the respiratory system. Reduction in lavage protein, cell death, and total cell counts also suppors that scopolamine prevents local respiratory toxicity induced by GD.
Intranasal administration of scopolamine has some advantages. It has been reported that intranasal administration results in rapid absorption and the bioavailability is similar to that of intravenous delivery. 46 Drowsiness due to intravenous administration of scopolamine is reduced by intranasal administration. Blurred vision was absent after intranasal administration unlike intravascular administration of scopolamine. 46 Intranasal administration is noninvasive and can be used under nonsterile conditions in the field and the dose can be titrated based on the symptoms and for children or populations with predisposed medical conditions. Direct aerosolization of antimuscarinic agents to the respiratory system seems to be more effective than intramuscular administration in reducing copious airway secretion and increasing the survival in the absence of any other treatments. Rapid onset of scopolamine action on the respiratory system and subsequent prevention of airway secretions and reduction in bronchoconstriction immediately after exposure seem to be critical for survival in addition to systemic absorption and CNS protection.
Comparing the protective effects of endotracheally aerosolized scopolamine versus atropine sulfate (2.5 mg/kg) did not show any significant difference between the 2 drugs. 73 Most of the evaluated parameters including recovery from body weight loss, lung and trachea edema, increased BAL protein, decreased cell death, and respiratory dynamics were similar between scopolamine and atropine sulfate treatment. However, lung histopathology data showed that scopolamine was more effective with respect to inflammation compared to atropine sulfate. Scopolamine was reported to be a better anticonvulsant than midazolam and diazepam in guinea pigs. 41 Comparison of the protective efficacy of scopolamine and atropine sulfate against neuropathology is currently in progress.
Conclusions
In conclusion, the aerosolized antimuscarinic agent scopolamine protects against respiratory toxicity induced by inhalation exposure to soman and significantly increases survival in guinea pigs. The protective efficacy of scopolamine against soman was also observed at the level of body weight loss, lung injury, and respiratory function. The mechanism of protection mainly involves antisecretory effects of scopolamine on the airways in addition to antimuscarinic effects on the peripheral nervous system and CNS.
Footnotes
Acknowledgments
The project was supported by Defense Threat Reduction Agency. Its contents, opinions, and assertions contained herein are private views of the authors and are not to be construed as official or reflecting the views of the Department of the Army or the Department of Defense.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Defense Threat Reduction Agency, proposal #3.F0006_08_WR_C.
