Abstract
This investigation reports the effects of various terminal procedures, and how they modified the responses to a toxicant (polychlorinated biphenyls [A1254], 130 mg/kg/day × 5 days) administered by gavage to Sprague-Dawley male rats. Terminal procedures included exsanguination via the abdominal aorta under anesthesia (isoflurane inhalation or Equithesin injection), decapitation with or without anesthesia, or narcosis induced by carbon dioxide inhalation. Effects of repeated anesthesia were also tested. Terminal procedures induced confounding stress responses, particularly when Equithesin was used. The terminal procedures modified the conclusions about effects of A1254 on the concentrations of corticosterone, insulin, glucagon, glucose, alkaline phosphatase, lactate dehydrogenase, uric acid, and blood urea nitrogen, from nonstatistically significant to significant changes, and in the case of luteinizing hormone from a statistically significant increase to a significant decrease. Investigations of effects of toxicants should be designed and interpreted considering potential changes induced by the selection of a terminal procedure.
Introduction
The ability to consistently detect subtle treatment effects can be compromised by the selection of inappropriate terminal procedures. This can increase the variability of the results and thus the ability to observe statistically significant effects. Terminal procedures in rodents can involve a number of anesthetics, immobilization, and blood collection procedures. 1,2 Isoflurane is regularly used in surgical procedures requiring light anesthesia or in terminal procedures. It is the preferred anesthetic for all rodents when equipment for gas anesthesia is available. 1 CO2-induced narcosis is a classical method used to sedate rats during minor surgical procedure, or after longer exposure to induce asphyxiation and euthanasia. It is considered acceptable for short-term procedures in rodents. 1,3 Equithesin is a mixture of magnesium sulphate, chloral hydrate, sodium pentobarbital, ethanol, and propylene glycol. Magnesium sulphate is a central depressant. 4 Chloral hydrate and sodium pentobarbital are both sedative hypnotics, the former metabolised by alcohol dehydrogenase and the latter by cytochrome P450 (CYP) 2A6 and 3A4. 5 This mixture is no longer available commercially but is easily prepared in the laboratories. 6 Equithesin was originally formulated for horses and cattle 7 and is currently recommended for use in birds. 8,9 However, Equithesin is still suggested in rat neurobiology protocols, 10 –12 but it is less tolerated in mice. 10 Many neurobiology laboratories use Equithesin based on the facts that: (1) it is difficult to use nose inhalation apparatus while performing brain surgery, (2) it is a medium duration anesthetic with a better safety margin than pentobarbital alone, (3) it was reported to have less effects on neuroreceptors than ketamine-based anesthetics, 7 and (4) it has no effects on mitochondrial activities. 12 However, due to its chloral hydrate content, Equithesin could be responsible for causing unpredictable adynamic ileus as a long-term effect. 7
The commercial mixture of polychlorinated biphenyls (PCB) known as Aroclor 1254 (A1254) is a broad toxicant. It is neurotoxic, 13 –17 immunotoxic, 18 thyrotoxic, 19,20 and antiandrogenic, 21 and affects reproductive functions. 22,23 It is the most potent Aroclor mixture for inducing liver cancer in female rats. 24 The A1254 mixture include non-ortho-, mono-ortho-, and di-ortho-chlorinated PCBs, as previously described. 25 The wide biological activity of A1254 is attributed to its chemical content interacting with various receptor systems. Non-ortho-PCBs (also referred to as dioxin-like or coplanar PCBs) have the highest affinity for the aryl-hydrocarbon receptor (AhR), and induce the expression of CYP1A enzymes. Di-ortho-chlorinated PCBs (phenobarbital-like PCBs) have the least affinity for the AhR but are most efficient at inducing the expression of CYP2B and 3A enzymes through other nuclear receptor pathways (constitutive androstane receptor [CAR], pregnane X receptor [PXR] 26 ;). The mono-ortho-chlorinated PCBs act on the AhR and CAR/PXR receptor systems and are mixed CYP inducers. 27 In humans, PCBs are classified as probable carcinogens, and are associated with neurobehavioral and immunological changes in children, 28 acne-like skin conditions, and ophthalmologic symptoms. 29 Prevalence of metabolic syndrome also appears to be affected by PCB exposure. 30 Polychlorinated biphenyl concentration, more abundant in obese individuals, is associated with hypertension in the general population. 31 Increases in blood sugar and cholesterol were also reported in the Yusho population accidentally exposed 40 years ago to a mixture including PCBs and polychlorinated dibenzofurans. 29
The samples in the current investigation were collected from a previous study performed in our laboratory to identify the terminal procedures that would have the least impact on brain neurochemistry. 14 This experiment compared effects of terminal procedures in naïve rats and in A1254-treated rats. A1254 was selected because it is a PCB mixture known to be neurotoxic (see previous paragraph). Immobilization was achieved using either isoflurane inhalation, Equithesin injection, or CO2 inhalation. These were associated with 2 blood sampling methods, either decapitation and trunk blood collection or exsanguinations through the abdominal aorta under anesthesia. Nakai et al 14 reported that all terminal procedures had some effects on the neurochemistry. In the present study, given the broad effects of A1254 and using samples from the same animals as Nakai et al, 14 the effects of the various terminal procedures were monitored by measuring the concentrations of 7 hormones, 4 hepatic microsomal enzyme activities, and 10 serum biochemistry endpoints in Sprague-Dawley male rats. This species was selected because of its historical and recurrent use in our laboratory. Females were not selected to avoid possible confounding effects of the estrous cycle. The investigated hormones were corticosterone (CS; the active glucocorticoid in the rat), insulin, glucagon, prolactin (PRL), luteinizing hormone (LH), thyroid-stimulating hormone (TSH), and thyroxine (T4). Prolactin and CS were selected because they are known to be increased in stressed rats, 32 –34 and changes in CS affects glycaemia, which is subjected to feedback regulation by insulin and glucagon. Reduction in the serum thyroxine level is a landmark of PCB exposure 35 and was monitored here in association with TSH.
Polychlorinated biphenyl exposure increases the activities of xenobiotic inducible CYP enzyme families, and some members have been shown to be sensitive to anesthetic neurodepressants. 36 Therefore, changes in CYP activities have been monitored by measuring hepatic microsomal ethoxy- (EROD), pentoxy- (PROD), and benzyloxyresorufin O-dealkylase (BROD) activities and aniline hydroxylase (AH) activities. While PROD and BROD activities have been considered indicators of CYP2B1/2 and 3A activities, respectively, they have been shown to predominantly reflect CYP2B activities. 37 EROD and AH activities were used here as indicator of CYP1A1 and CYP2E activities, 38,39 respectively. Finally, routine clinical biochemistry endpoints were investigated including lactate dehydrogenase (LD), alanine transaminase (ALT), alkaline phosphatise (AP), glucose, total cholesterol, blood urea nitrogen, uric acid, phosphate, and calcium.
Materials and Methods
Anesthetic Agents
The Equithesin mixture (EQ) 6 was prepared in our laboratory and each 100 mL of aqueous solution contained 0.97 g sodium pentobarbital (CDMV, Ste. Hyacinthe, QC, Canada), 4.04 g chloral hydrate, 2.13 g magnesium sulfate, 40 g propylene glycol, and 14.8 g of 95% ethanol (SigmaAldrich, Oakville, ON, Canada). Equithesin mixture was delivered at a dose of 3 mL/kg body weight ip. 6 Isoflurane was delivered by inhalation at 3% to 5% (AErrane, 100% 1-chloro-2,2,2-trifluroethyl difluromethyl ether; Janssen Cie., North York, ON, Canada). Animals treated with isoflurane were initially anesthetized by placing them into a bell-shaped chamber infused with the anesthetic and were kept under anesthesia using a nose-cone while being exsanguinated. CO2 was administered to induce narcosis in a 1-minute cycle with oxygen using an automated inhalation system. The animals treated with carbon dioxide were placed into a small chamber which was then infused with a CO2/O2 mixture and applied just long enough to produce a narcosis prior to decapitating the animals. The chamber was cleaned and allowed to equilibrate with the room air prior to inserting each subject.
Animal Treatment
All animal procedures were approved by the Health Canada Animal Care Committee in accordance with the principles and guidelines of the Canadian Council on Animal Care (CCAC). 40 Both the CCAC and the American Veterinary Medical Association (AVMA) indicate that pre-sedation is not necessary before decapitation with guillotine as euthanasia method in rodents when scientifically defended and approved by the Institutional Animal Care and Use Committee. 41
Male Sprague-Dawley rats (7-8 weeks old, 225-275 g, Charles River Canada, St Constant, Quebec) were divided randomly into 11 treatment groups of 10 animals each and housed in individual Health Guard cages (Research Equipment Co., Bryan, TX, USA). The animals were allowed to acclimatize for 1 week prior to initiating the study. Lighting in the animal facility was on a 12-h light/dark cycle and the room was maintained at 21 ± 3°C with 30% to 70% humidity. All animals were allowed free access to food (Purina Lab Chow 5002; PMI Feed, St Louis, Missouri) and water. The rats were subjected to the treatments as described in Table 1. Group D was terminated by decapitation (D) on day 8 and is considered as the control group. Group (Iso-D) was subjected to isoflurane (Iso) anesthesia on day 8 just prior to decapitation. Group Iso-E received the same Iso treatment but was terminated by complete exsanguination (E) via the abdominal aorta. Group 3xIso-E and 3xEQ-E were used to assess the effects of repeated exposures to the anesthetics, and were anesthetized on days 6, 7, and 8 using either Iso or EQ. They were exsanguinated via the abdominal aorta while under anesthesia on day 8. Investigating the effects of repeated exposures to an anesthetic is useful for the interpretation of data generated by experiments requiring surgical interventions prior to the day of euthanasia. Group EQ-E was anesthetized once with EQ on day 8 prior to exsanguination. Group CO2-D was exposed to carbon dioxide to produce narcosis (suppression of function) prior to decapitation on day 8. Groups A1254-D, A1254-Iso-E, A1254-EQ-E, and A1254-CO2-D, were dosed by gavage on each of day 1 through day 5 (the course of one work week) with A1254 (AccuStandard Inc, New Haven, Connecticut. Lot number 124-191) at 130 mg/kg body weight per day. Therefore, the A1254-treated groups were subjected to additional manipulations and to corn oil compared to the non-A1254-treated groups. The selected dose of A1254 was 10-fold lower than the LD50 (1300 mg/kg) in weanling rat (the most sensitive age period). 42 Lower environmentally relevant doses would have induced subtle effects difficult to measure with certainty, and the demonstration of confounding effects due to terminal procedures might have been ambiguous. The rats were then euthanized on day 8. These A1254-treated groups were subjected to the same terminal procedures as their respective non-A1254-treated groups (D, Iso-E, EQ-E, CO2-D). The brains were collected for neurochemical analysis 14 and the liver for measuring CYP activities. Blood was collected in SST Vacutainer tubes (Becton-Dickinson, Franklin Lakes, New Jersey) and allowed to clot at room temperature. Serum was separated by centrifugation at 300 g and stored at −80°C until analysis. Samples were separated in 3 aliquots for analysis by the endocrinology, neurochemical, and clinical biochemistry laboratory.
Description of the Experimental Design a
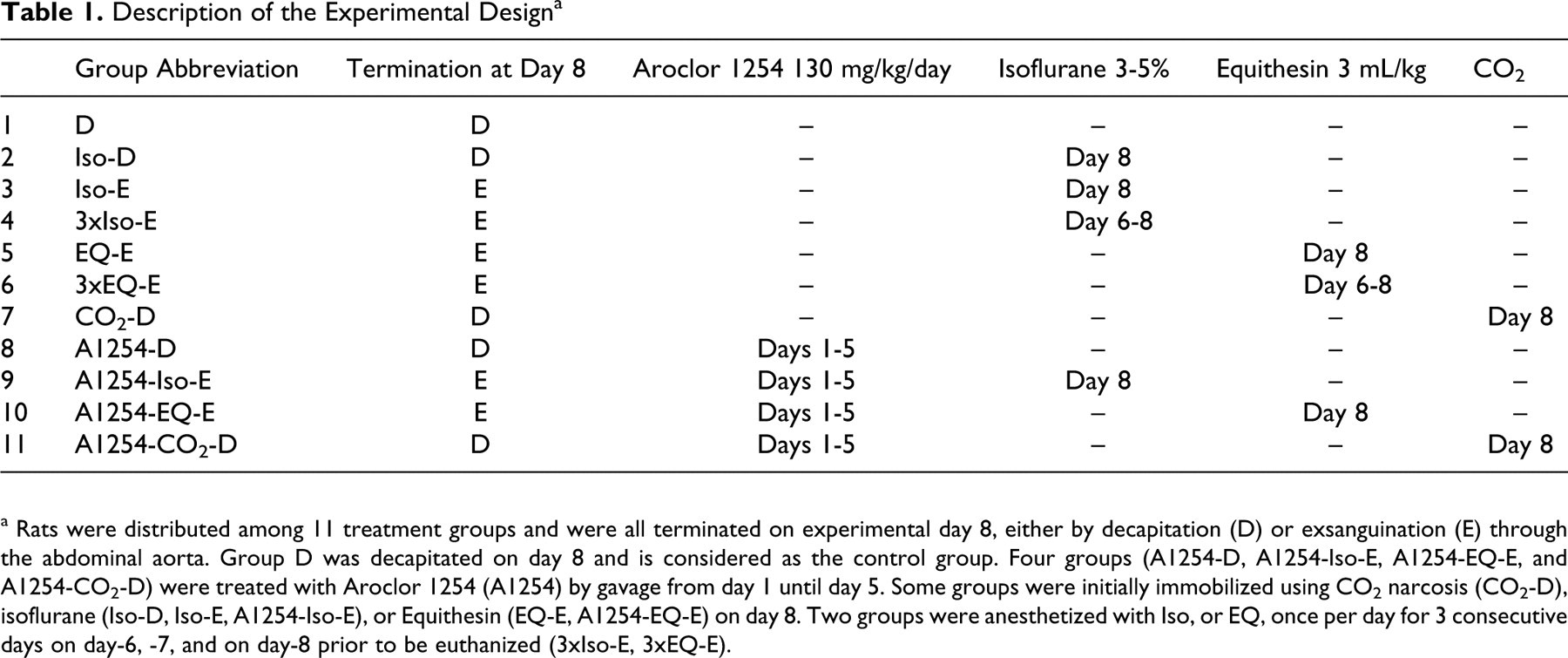
a Rats were distributed among 11 treatment groups and were all terminated on experimental day 8, either by decapitation (D) or exsanguination (E) through the abdominal aorta. Group D was decapitated on day 8 and is considered as the control group. Four groups (A1254-D, A1254-Iso-E, A1254-EQ-E, and A1254-CO2-D) were treated with Aroclor 1254 (A1254) by gavage from day 1 until day 5. Some groups were initially immobilized using CO2 narcosis (CO2-D), isoflurane (Iso-D, Iso-E, A1254-Iso-E), or Equithesin (EQ-E, A1254-EQ-E) on day 8. Two groups were anesthetized with Iso, or EQ, once per day for 3 consecutive days on day-6, -7, and on day-8 prior to be euthanized (3xIso-E, 3xEQ-E).
Radioimmunoassays
The peptide hormone radioimmunoassays and the method of iodination were performed as previously described. 43,44 All reagents were kindly provided by the National Hormone and Pituitary Program, the National Institute of Diabetes and Digestive and Kidney Diseases. The hormones used for 125I-iodination and for the standard curve, and the primary antiserum for the LH, TSH, and PRL assays were rLH-I-9 AFP-10250C, rLH-RP-3 AFP-7187B, anti-rLH-S-11, rTSH-I-9 AFP-11542B, rTSH-RP3 AFP-5512B, anti-rTSH-RIA-6 AFP329691Rb, rPRL-I-6 AFP-10505B, rPRL-RP-3 AFP-4459B, and anti-rPRL-S-9 AFP-131581570, respectively. The secondary antibody was an anti-rabbit immunoglobulin G (IgG) developed in goat (Sigma Immunochemicals, St-Louis, Missouri). All samples were analyzed in a single assay to avoid interassay variations. Commercial I125 RIA kits were used according to manufacturers' instructions for the measurement of thyroxine and corticosterone (ICN Biomedicals, Costa Mesa, California), as well as for rat insulin and glucagon (LINCO Research Inc, St Charles, Missouri).
Hepatic Cytochrome P450 Enzyme Assays
At the time of necropsy, a 2- to 3-g piece of liver was homogenized in 2.5 volumes of ice-cold 0.05 mol/L Tris/1.15% KCI buffer, pH 7.4, and centrifuged at 10 000 g. The supernatant was separated and frozen, and later assayed for EROD, PROD, and BROD activities using the method of Lubet et al. 45 Aniline hydroxylase activity was also measured as previously published. 39
Clinical Enzymology and Chemistry
Serum inorganic phosphate, total protein, alkaline phosphatase (AP), alanine transaminase (ALT), lactate dehydrogenase (LD), calcium, cholesterol, glucose, blood urea nitrogen (BUN), and uric acid were quantified using an Express Plus clinical analyzer (Bayer Inc, Tarrytown, New York) according to the manufacturer’s instructions.
Data Analysis
All statistical analyses were performed using the software JMP. 46 Prior to conducting the analysis, the normality of the data was verified using the Shapiro-Wilk Goodness-of-Fit test. Normally distributed data were subjected to analysis of variance (ANOVA) followed by the Student-t all comparison procedure. In all figures, the vertical bar separates the non-A1254-treated from the A1254-treated groups. The data to the left of the vertical bar were analysed first and were used to detect effects of terminal procedures. The data to the right of the vertical bar represent A1254-treated groups and were analysed independently. Two-way ANOVA were also applied, using the data shown with hatched bars in all figures (the A1254-treated groups and respective non-A1254-treated groups), to test for effects of terminal procedures, effects of the A1254 treatment, and their interactions. If the data were not normally distributed they were log transformed and retested for normality. If the transformed data were still not normally distributed then the original untransformed data were analyzed using the nonparametric Wilcoxon test for pair-wise comparison or the Kruskal-Wallis procedures for multiple comparisons. In addition to show means by histograms, the median values are presented by circles when the data failed the normality test. Values of P ≤ .05 indicated significant differences.
Results
Figure 1 summarizes endpoints indicative of stress responses (corticosterone, glucose) and regulation of glycaemia (insulin, glucagon). The corticosterone concentrations were significantly higher in both the EQ-E and 3xEQ-E treated groups, than in the D group (the control group). Among A1254-treated groups, the corticosterone concentrations were the highest in the A1254-EQ-E group and the lowest in the A1254-CO2-D group. The A1254-EQ-E group had significantly higher corticosterone concentrations than the EQ-E group. The insulin concentrations were significantly higher in the Iso-D and EQ-E group than in the D group, and they were significantly lower in the 3xEQ-E than in the EQ-E group. The concentrations of insulin were significantly lower in the 3xIso-E and Iso-E groups than in the Iso-D group. Two A1254-treated groups (A1254-EQ-E and A1254-CO2-D) had insulin levels significantly lower than in their non-A1254-treated counterparts. The concentrations of glucagon were significantly higher in the 3xEQ-E group than in the EQ-E group, but they were not different from the D group. Three A1254-treated groups (A1254-Iso-E, A1254-EQ-E and A1254-CO2-D) had glucagon levels significantly higher than in their non-A1254-treated groups. The glucose concentrations were significantly higher in the Iso-D, EQ-E, and 3xEQ-E groups than in group D, and glucose levels were significantly lower in the 3xEQ-E than in the EQ-E group. The A1254-EQ-E group had the highest concentrations of glucose among A1254-treated groups. Two A1254-treated groups (A1254-Iso-E and A1254-EQ-E) had glucose concentrations significantly lower than their non-A1254-treated counterparts.
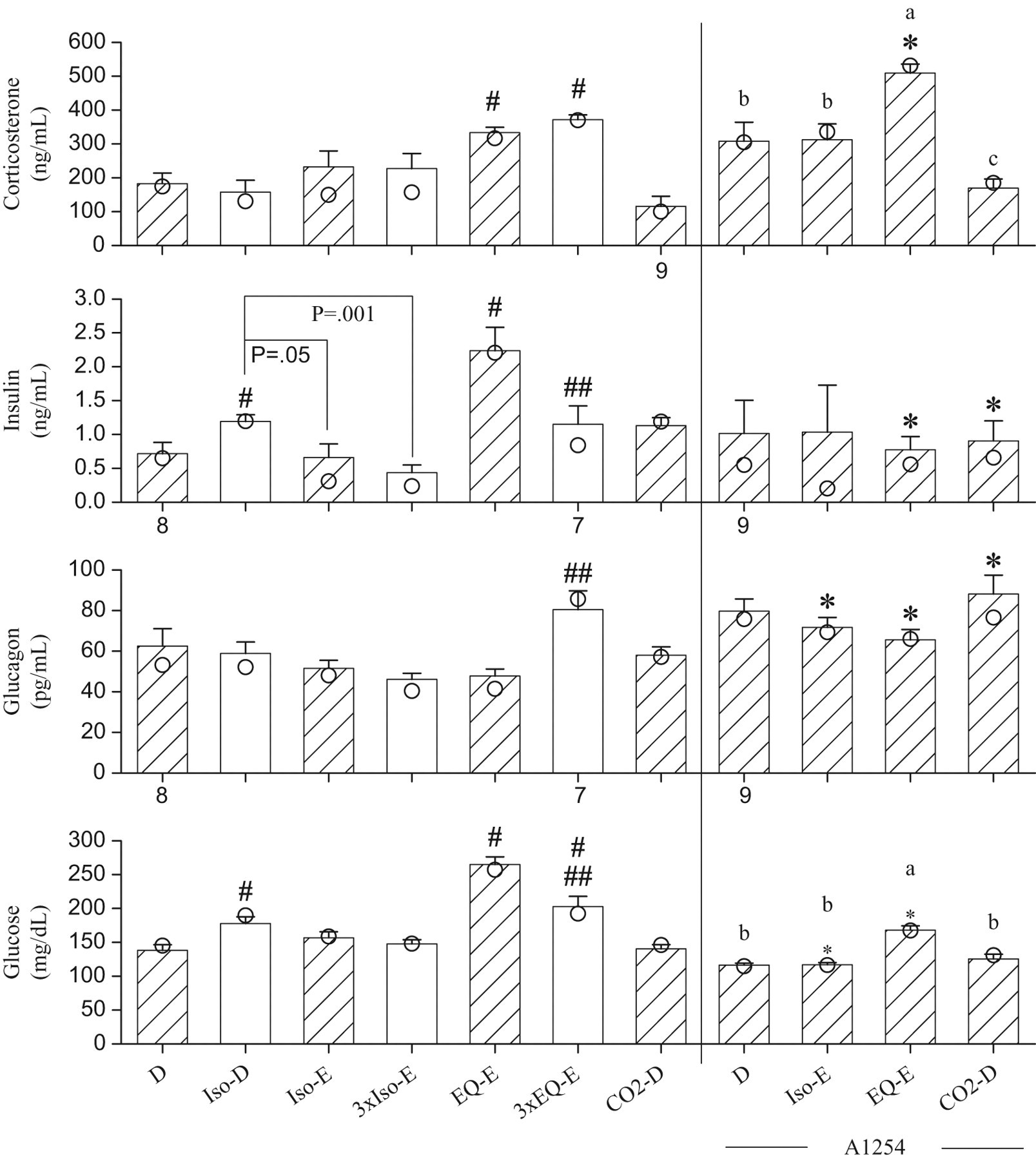
Serum concentrations of corticosterone, insulin, glucagon, and glucose, measured from blood samples collected from the trunk after decapitation (D), or following exsanguination (E) through the abdominal aorta, from rats anesthetized with isoflurane (Iso), or Equithesin (EQ), or subjected to CO2 narcosis. The bars represent the means ± SE of 10 rats unless indicated otherwise by the numbers shown below the bars. Median values shown as circles are presented when data not normally distributed are involved in non-parametric analysis. The hatched bars show groups that are compared to test the effects of A1254. The non-A1254-treated and A1254-treated groups are separated by the vertical bar. #, significantly different from group D (P < .05); ##, significant difference between rats anesthetized once or thrice (P < .05).*, this A1254-treated group differs from its respective non-A1254-treated group (P < .05). a, b, c: data normally distributed and anlayzed by ANOVA followed by the Student-t all comparison procedure; means sharing the same letters are not significantly different. The letter “a” is assigned to the highest mean.
Figure 2 summarizes the analyses of the pituitary hormones and thyroxine measured in serum. The changes in PRL concentrations were similar to those observed for corticosterone. The concentrations of PRL were significantly higher in the 3xIso-E, EQ-E, and 3xEQ-E groups than in group D. A 2-way ANOVA, comparing non-A1254-treated and A1254-treated groups, revealed that A1254 had no effects on the PRL concentrations. Among A1254-treated groups, the highest PRL concentrations were in the group A1254-EQ-E. Among groups not exposed to A1254, non-parametric analyses of LH concentrations revealed significant effects of terminal procedures (P = .0002), with significantly higher levels in Iso-D, Iso-E, 3xIso-E, EQ-E, and CO2-D groups than in group D (P < .05). Effects of A1254 could not be analysed by a 2-way ANOVA. Luteinizing hormone concentrations were significantly higher in the group A1254-D than in group D (P = .002), but in contrast they were significantly lower in the A1254-Iso-E than in the Iso-E group (P = .001). The later 2 comparisons show that a reverse effect of A1254 was detected depending on the selected terminal procedure (D vs A1254-D or Iso-E vs A1254-Iso-E). Among A1254 treated groups (1-way ANOVA, log, P = .015), LH concentrations were significantly higher in the A1254-D than in the A1254-Iso-E group. TSH concentrations (log) were lower in the EQ-E and 3xEQ-E groups than in the 3xIso-E group. A 2-way ANOVA indicated no effects of A1254 and no interactions, but an effect of terminal procedures (log, P = .0005). Thyroid-stimulating hormone concentrations are significantly higher in the A1254-CO2-D group than in the A1254-Iso-E or A1254-EQ-E group. Thyroxine concentrations were decreased in all groups, except 3xIso-E, which had concentrations similar to group D. Thyroid-stimulating hormone stimulates the production of thyroxine and coincidently, the concentrations of TSH in that group (3xIso-E) were high, but not significantly different from group D. Repeated exposures to EQ significantly reduced thyroxine concentrations (EQ-E vs 3xEQ-E). There is a similarity in the patterns of TSH and thyroxine concentrations (low concentrations of TSH are associated with low thyroxine levels) except for the A1254-treated groups, which showed suppressed thyroxine concentrations.
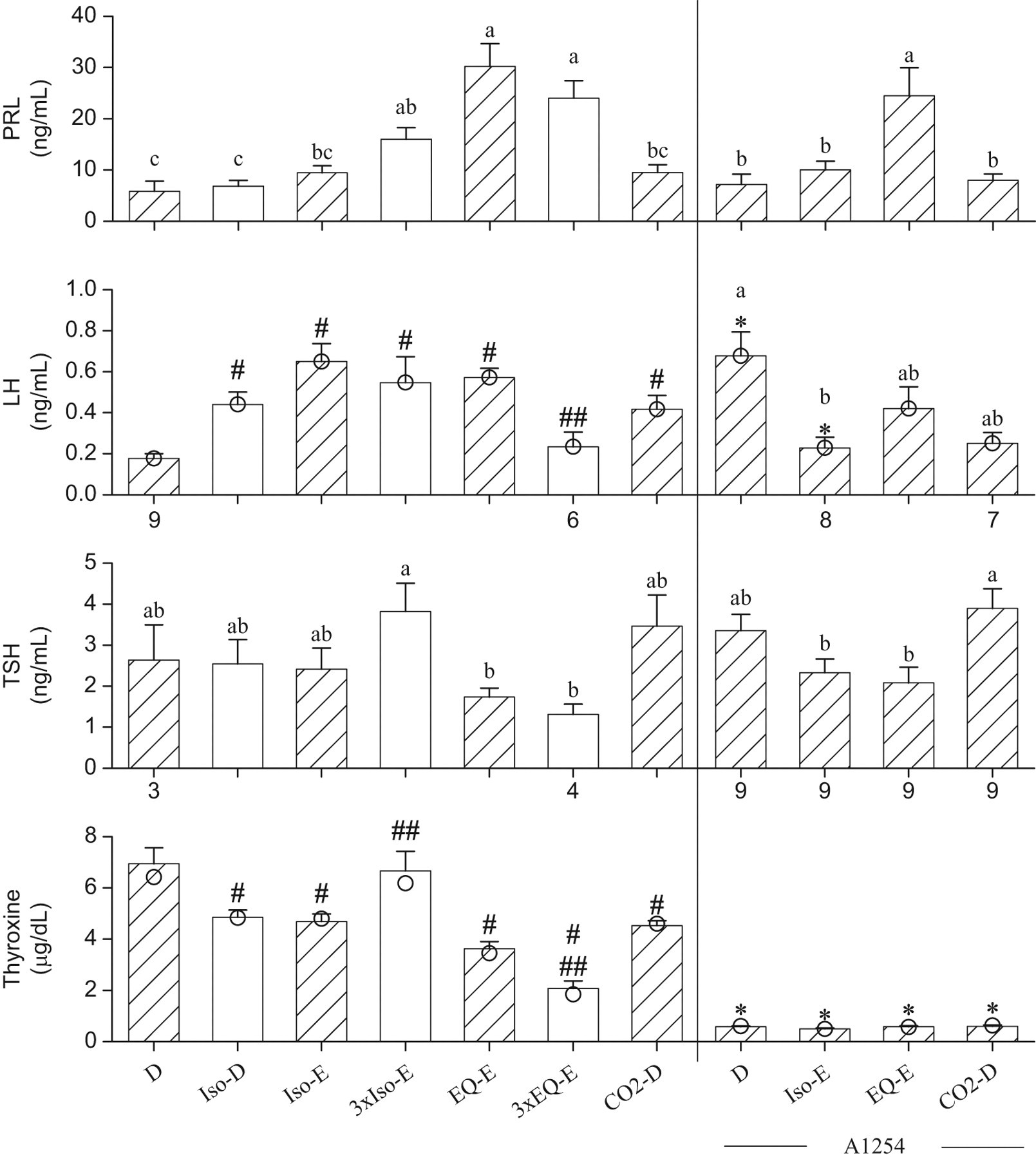
Serum concentrations of prolactin (PRL), luteinizing hormone (LH), thyroid-stimulating hormone (TSH), and thyroxine, measured from blood samples collected from the trunk after decapitation (D), or following exsanguinations (E) through the abdominal aorta, from rats anesthetized with isoflurane (Iso), or Equithesin (EQ), or subjected to CO2 narcosis. The bars represent the means ± SE of 10 rats unless indicated otherwise by the numbers shown below the bars. Median values shown as circles are presented when data not normally distributed are involved in non-parametric analysis. The hatched bars show groups that are compared to test the effects of A1254. The non-A1254-treated and A1254-treated groups are separated by the vertical bar. #, significantly different from group D (P < .05); ##, significant difference between rats anesthetized once or thrice (P < .05).*, this A1254-treated group differs from its respective non-A1254-treated group (P < .05). a, b, c: data normally distributed and analyzed by ANOVA followed by the Student-t all comparison procedure; means sharing the same letters are not significantly different. The letter “a” is assigned to the highest mean.
In Figure 3, relative to group D, the blood content of LD was increased by the Iso-E treatment but reduced by the Iso-D and CO2-D treatments. Among A1254-treated groups, the group exposed to CO2 also had the lowest LD levels. Lactate dehydrogenase activity was significantly higher in the group A1254-EQ-E than in group EQ-E. Alanine transaminase activity was lower in the groups 3xIso-E and EQ-E than in group D, but it was increased by CO2 and all A1254 treatments. Alkaline phosphatise was reduced by the 3xEQ-E treatment compared with group D. Although A1254 had generally no effects on AP, the A1254-Iso-E group had significantly lower AP activity than in group Iso-E.
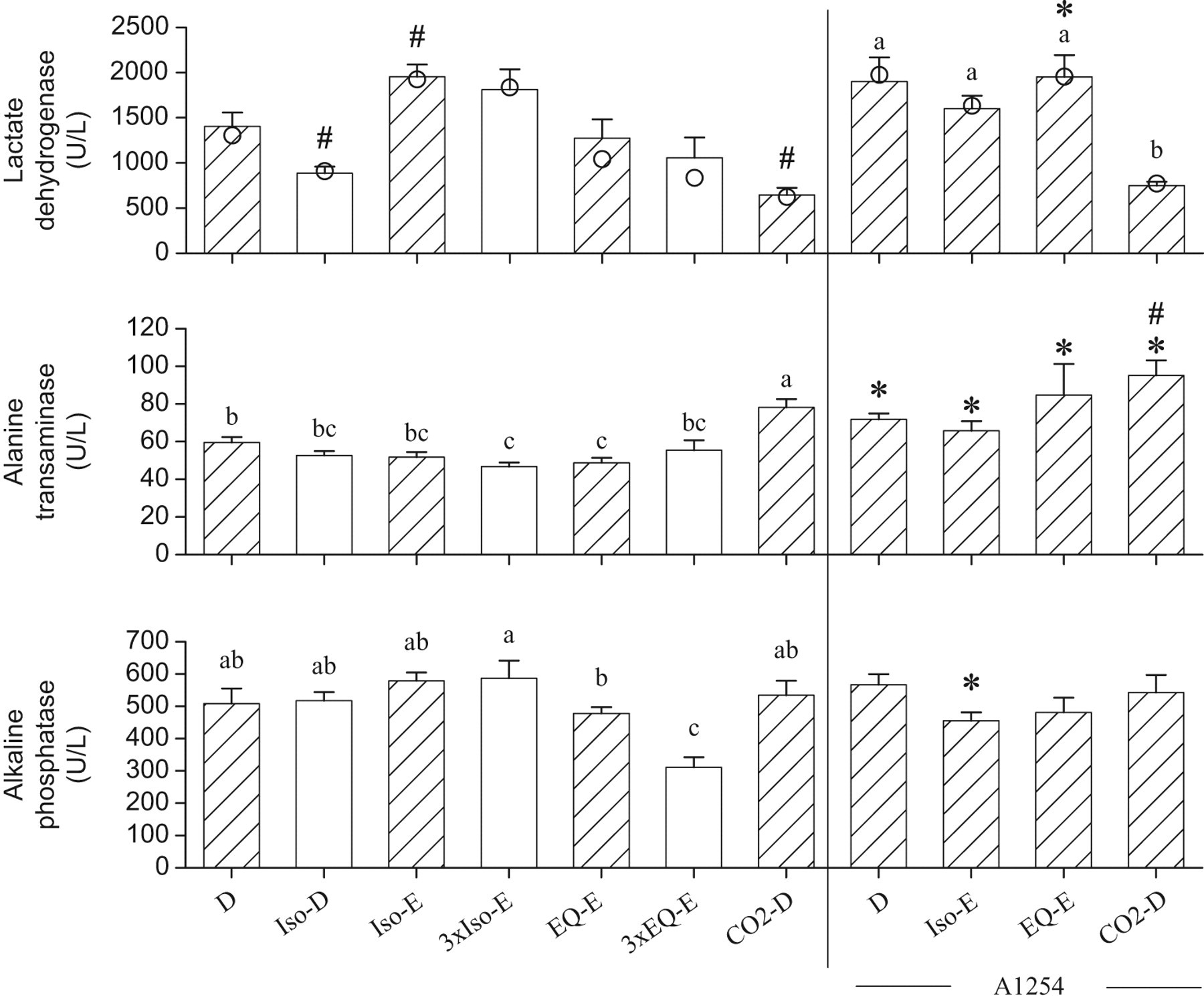
Serum concentrations of lactate dehydrogenase, alanine transaminase, and alkaline phosphatase, measured from blood samples collected from the trunk after decapitation (D), or following exsanguinations (E) through the abdominal aorta, from rats anesthetized with isoflurane (Iso), or Equithesin (EQ), or subjected to CO2 narcosis. The bars represent the means ± SE of 10 rats the bars. Median values shown as circles are presented when data not normally distributed are involved in non-parametric analysis. The hatched bars show groups that are compared to test the effects of A1254. The non-A1254-treated and A1254-treated groups are separated by the vertical bar. #, lactate dehydrogenase significantly different from group D, or alanine transaminase different from A1254-D (P<.05); *, this A1254-treated group differs from its respective non-A1254-treated group (P < .05). a, b, c: data normally distributed and analyzed by ANOVA followed by the Student-t all comparison procedure; means sharing the same letters are not significantly different. The letter “a” is assigned to the highest mean.
Total protein concentrations were significantly lower in the group 3xEQ-E than in group D (Figure 4). A 2-way ANOVA comparing non-A1254-treated and A1254-treated groups revealed significant effects of terminal procedures (P = .01) and of A1254 (P = .04). The latter effect was small and not associated with significant differences between A1254-treated and respective non-A1254-treated groups. Total protein concentrations were significantly lower in the A1254-Iso-E group compared with the A1254-D and A1254-CO2-D treated groups. The concentrations of BUN were higher in group Iso-E and EQ-E than in group D. The group A1254-D had significantly higher BUN concentrations than group D. None of the A1254-treated groups were significantly different from group A1254-D. The concentrations of uric acid were significantly reduced in groups Iso-D, EQ-E, 3xEQ-E, and CO2-D, compared with group D. A 2-way ANOVA revealed effects of terminal procedures and A1254 (both with P < .0001, log). The concentrations of uric acid were significantly lower in the A1254-Iso-E and A1254-EQ-E groups than in their respective non-A1254-treated groups. The concentrations of cholesterol were significantly higher in group 3xEQ-E than in group D, and were significantly increased in all A1254-treated groups compared with non-A1254-treated groups. Concentrations of phosphate were significantly reduced by all terminal procedures involving anesthesia but were not affected by CO2 narcosis. A1254 had no effect on phosphate concentrations. Finally, calcium concentrations in the group 3xEQ-E were significantly lower than in group D. There was no effect of A1254, but among A1254-treated rats, calcium concentrations were significantly lower in the group A1254-EQ-E than in group A1254-D and A1254-CO2-D.
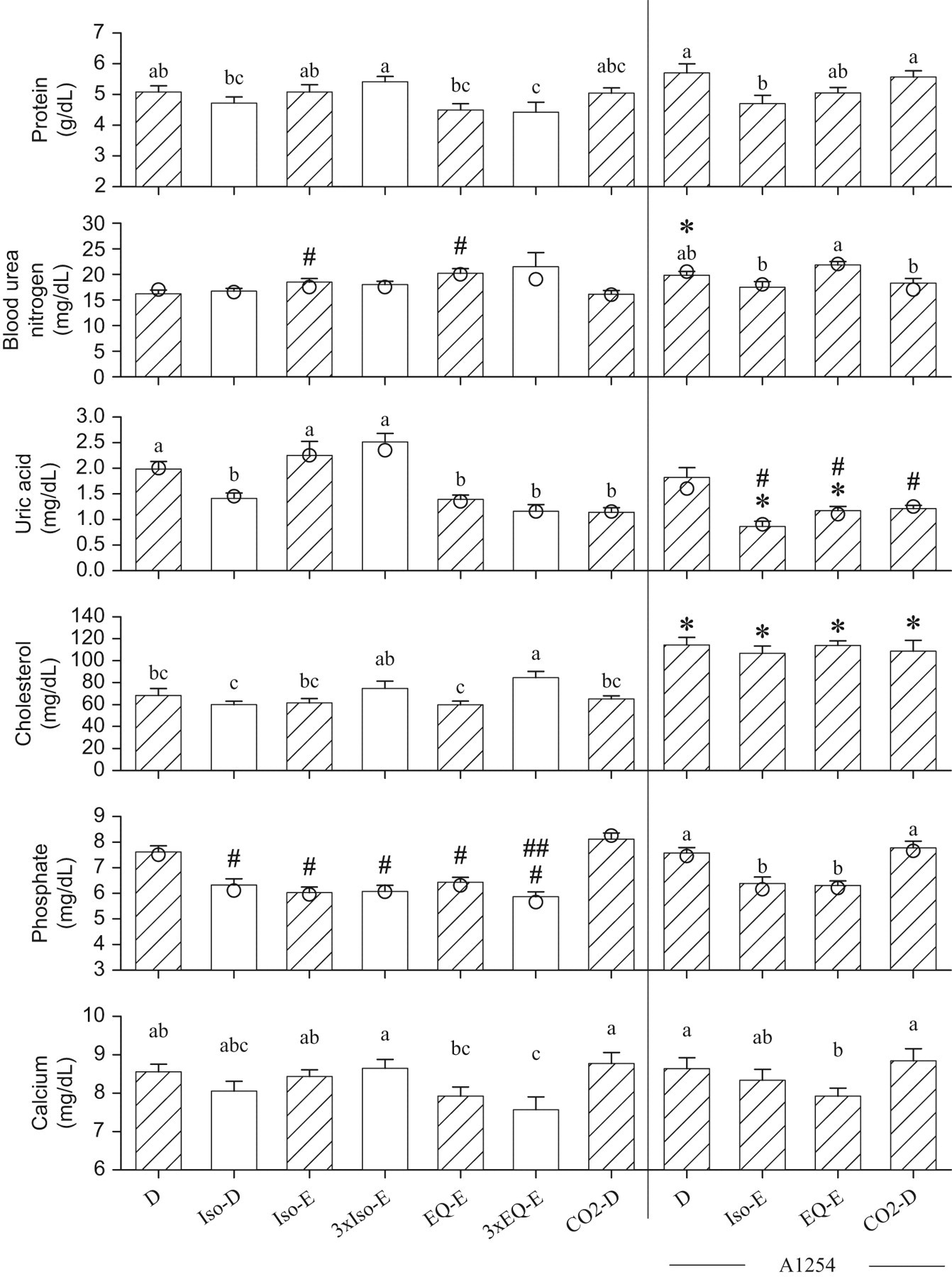
Serum biochemistry endpoints measured from blood samples collected from the trunk after decapitation (D), or following exsanguinations (E) through the abdominal aorta, from rats anesthetized with isoflurane (Iso), or equithesin (EQ), or subjected to CO2 narcosis. The bars represent the means ± SE of 10 rats. Median values shown as circles are presented when data not normally distributed are involved in non-parametric analysis. The hatched bars show groups that are compared to test the effects of A1254. The non-A1254-treated and A1254-treated groups are separated by the vertical bar. #, blood urea nitrogen and phosphate concentrations are significantly different from group D, or uric acid concentrations different from group A1254-D (P < .05); ##, significant difference between rats anesthetized once or thrice (P < .05). *, this A1254-treated group differs from its respective non-A1254-treated group (P < .05). a, b, c: data normally distributed and analyzed by ANOVA followed by the Student-t all comparison procedure; means sharing the same letters are not significantly different. The letter “a” is assigned to the highest mean.
Hepatic cytochrome P-450 activities, as evaluated by EROD, BROD, PROD, and AH activities, were not affected by the anesthetics or CO2 narcosis (Table 2). The A1254 treatment significantly increased the transformation of all investigated substrates. PROD activity in the group A1254-EQ-E was significantly less than in the group A1254-CO2-D (P = .004).
Hepatic Ethoxy- (EROD), Benzyloxy- (BROD), and Pentoxyresorufin-o-dealkylase (PROD), and Aniline Hydroxylase (AH) Activities (nm/min/mg Protein) a
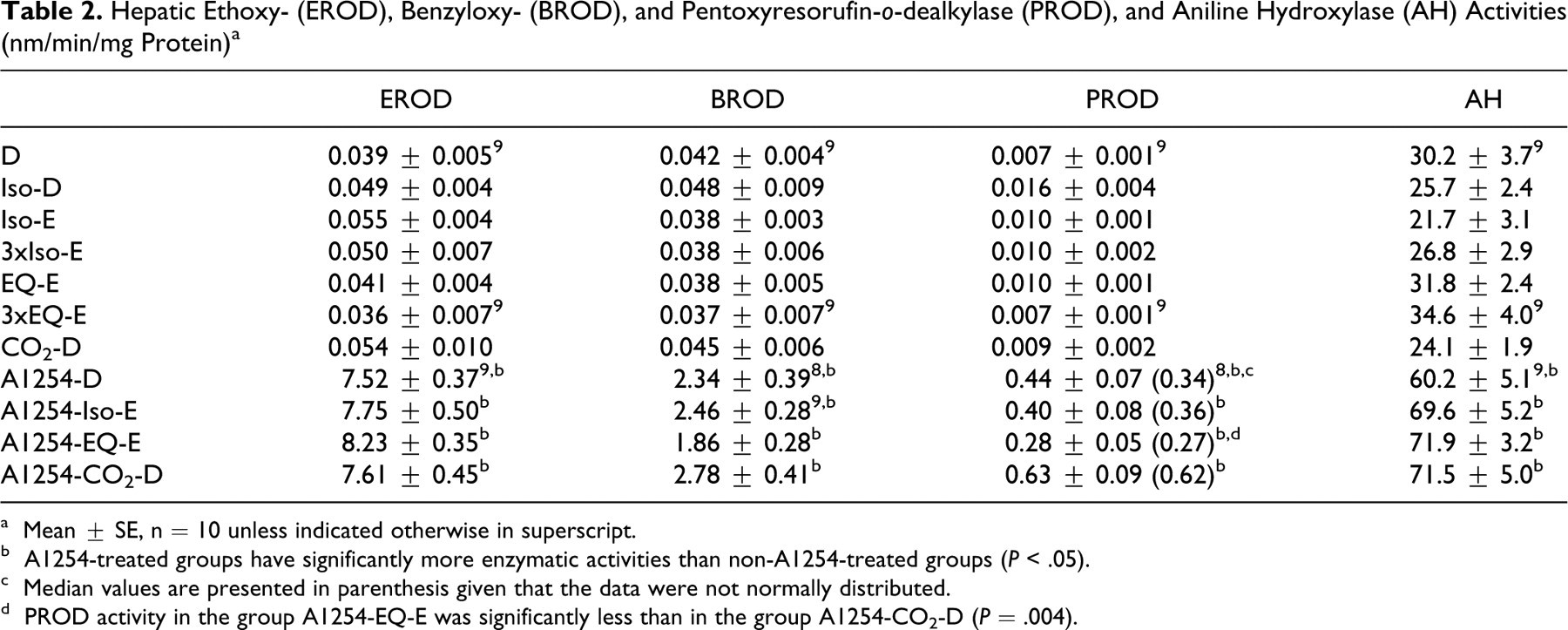
a Mean ± SE, n = 10 unless indicated otherwise in superscript.
b A1254-treated groups have significantly more enzymatic activities than non-A1254-treated groups (P < .05).
c Median values are presented in parenthesis given that the data were not normally distributed.
d PROD activity in the group A1254-EQ-E was significantly less than in the group A1254-CO2-D (P = .004).
Table 3 summarizes the data by comparing the impacts of the selection of terminal procedures (D, Iso-E, EQ-E, CO2-D) on the conclusions about the effects of exposure to A1254. For example, the first line in Table 3 presents that A1254 treatment increased the concentrations of corticosterone significantly (P = .007) when the EQ-E process was used, while it had no statistically significant effects when the terminal procedure involved D, Iso-E or CO2-D. This permits us to conclude (in the last column) that depending on the selection of a terminal procedure, A1254 treatment can be perceived to have no significant effect or to significantly increase the concentrations of corticosterone. Observations of greatest consequences in this table are the selections of terminal procedures generating opposite conclusions, or conclusions changing from nonsignificant to significant effects. Opposite conclusions were observed related to the effects of A1254 on LH concentrations, which changed from a statistically significant increase (decapitated) to a decrease (Iso-E). The effects of A1254 changed from being not statistically significant to significant increases in the case of LD and BUN, but to significant decreases for insulin, AP, glucose, and uric acid. Of less consequence are the effects of A1254 for which only the magnitude of the response was affected (T4, PROD, ALT, and cholesterol). The conclusions were the same regardless of the terminal procedures for EROD, BROD, and AH activities, which were all increased by A1254. Finally, A1254 had no effect on PRL, TSH, total protein, phosphate, and Ca, and these conclusions were also the same regardless of the terminal procedures.
Summary of How the Conclusions About the Effects of A1254 Can Be Modified by the Selection of a Terminal Procedure a
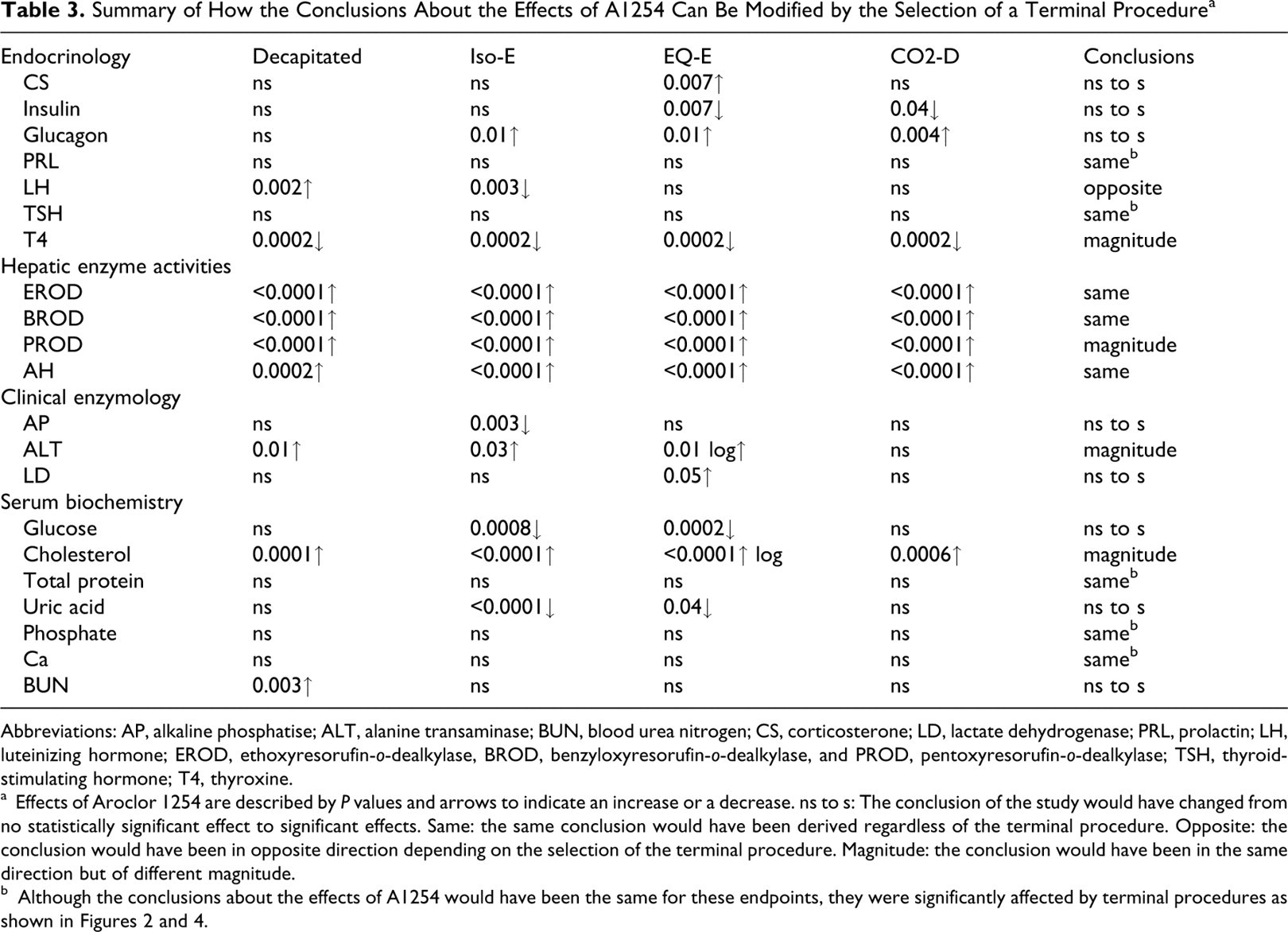
Abbreviations: AP, alkaline phosphatise; ALT, alanine transaminase; BUN, blood urea nitrogen; CS, corticosterone; LD, lactate dehydrogenase; PRL, prolactin; LH, luteinizing hormone; EROD, ethoxyresorufin-o-dealkylase, BROD, benzyloxyresorufin-o-dealkylase, and PROD, pentoxyresorufin-o-dealkylase; TSH, thyroid-stimulating hormone; T4, thyroxine.
a Effects of Aroclor 1254 are described by P values and arrows to indicate an increase or a decrease. ns to s: The conclusion of the study would have changed from no statistically significant effect to significant effects. Same: the same conclusion would have been derived regardless of the terminal procedure. Opposite: the conclusion would have been in opposite direction depending on the selection of the terminal procedure. Magnitude: the conclusion would have been in the same direction but of different magnitude.
Discussion
This study demonstrates that the selection of a terminal procedure can alter endpoints and change the conclusions related to the effects of a test chemical. For example, the observed effect of A1254 on LH was reversed when comparing the decapitated groups relative to the ISO-E groups. The terminal procedures using EQ anesthesia was found to alter more endpoints and induce a greater stress response than when Iso was used. Therefore, the design of an investigation of the effects of a potential toxicant should include consideration of effects of the terminal procedure. The latter can bias conclusions due to a series of factors which may act in combination. These may include (1) different animal handling procedures eliciting activation of the hypothalamo-pituitary-adrenal axis (HPA), (2) delay between exposure to specific anesthetics and loss of responsive reflex (eg, toe pinch), (3) hypoxic changes due to lengthy exsanguination procedures or CO2 exposure, and (4) hypothermia induced by the anesthetic and effects of the anesthetic itself. 47
Stress Response and Glucose Metabolism
The 3 groups treated with EQ (EQ-E, 3xEQ-E, A1254 EQ-E) had high concentrations of corticosterone and glucose, suggesting that EQ treatments induced endocrine stress responses. 48 In line with our observations, pentobarbital, a component of EQ, was previously reported to increase levels of corticosterone in rats. 49 The comparison of the groups EQ-E and A1254-EQ-E showed that the coadministration of A1254 with EQ-E increased the corticosterone concentrations even higher than in the group EQ-E, suggesting that EQ and A1254 might have additive effects. A1254 is toxic to the hippocampus 50 and this might contribute to an increase in corticosterone concentrations given that the hippocampus is a negative regulator of glucocorticoid secretion. 51,52 However, effects of PCBs on corticosterone concentrations are not consistently reported. 53
The results support a deregulation of glucose levels in the 3xEQ-E group. Normally, hyperglyceamia is compensated by the release of insulin to reduce the glucose concentration by inducing its absorption by muscles and adipocytes. This endocrine relationship was evident in the EQ-E data set with insulin being elevated when glucose is elevated in the presence of a normal glucagon level. In contrast, the group 3xEQ-E had a different metabolic response, with a significant increase in glucagon despite concentrations of glucose still significantly higher than group D. Perhaps the elevated levels of glucagon and glucose following repeated exposures to EQ were associated with insensitivity to glucose.
In general, compared with non-A1254-treated rats, the A1254-treated groups (A1254-Iso-E, A1254-EQ-E, A1254-CO2-D) had elevated concentrations of glucagon and low glucose concentrations (a statistically significant relationship in groups treated with A1254-Iso-E and A1254-EQ-E). These results support that the measurement of glucagon is an additional useful biomarker to facilitate the detection of hypoglycaemia in toxicological investigations. Physiologically, the circulating concentrations of glucose are tightly regulated given that glucose is the major source of energy for the brain. 54 Glucagon stimulates hepatic gluconeogenesis, and the high glucagon concentrations in most A1254-treated rats suggest impaired hepatic glucose production. This is consistent with the other indicators of A1254-induced hepatic effects reported here (high P450 inductions and ALT levels). The effect of A1254 on the regulation of glycemia did not reach statistical significance in the decapitated rats, and this conclusion is similar to others using decapitated rats while testing A1254 at lower doses in longer exposure protocol. 55,56
Pituitary Peptide and Thyroid Hormones in the Serum
The serum levels of all the 3 anterior pituitary hormones (PRL, LH, and TSH) were modified by the terminal procedures. The pattern of effects was very similar for both the concentrations of PRL and corticosterone given that both hormones are known to be increased in stressed rats. 57 The elevated PRL concentrations in EQ-treated rats are consistent with previous studies showing pentobarbital-induced increase in PRL. 58,59 An increase in PRL is suggested to counteract stress-induced HPA activity and stress-induced hypocalcaemia, 60 which incidentally were observed here in the EQ-treated rats. In the current study, the A1254 treatment did not affect PRL concentrations. This 5-day treatment was relatively short compared to a 30-day exposure period to A1254 (2 mg/kg/day), which induced a decrease in PRL concentration. 61
The current data suggest that LH concentrations are sensitive to the effects of terminal procedures. An important observation from the A1254-treated groups is the fact that the selection of a specific terminal procedure could have led to opposite conclusions (A1254 increased LH when comparing D vs A1254-D, but decreased LH when comparing Iso-E vs A1254-Iso-E). Other observations from Figure 2 indicate that LH concentrations were increased by most terminal procedures, but the 3xEQ-E rats responded differently with concentrations of LH similar to those in the D group. Long exposures to barbitals inhibit LH secretion by acting on the hypothalamus in both male 62 and female rats 63 ; whereas other anesthethic treatments during terminal procedures were reported to have no effects. 58 Using CO2 asphyxiation as terminal procedure, Muthuvel et al 64 observed a decrease in LH concentrations following a 30-day exposure to A1254 (2 mg/kg/day). Here, the decrease in LH concentration between the CO2-D and the A1254-CO2-D-treated group was not statistically significant, but the dosing regimen was different.
Thyroxine concentrations were decreased in all groups, except 3xIso-E, which had concentrations similar to group D. It is likely that many mechanisms contributed to the reduction in serum thyroxine. Pentobarbital is known to reduce thyroxine concentrations, 65 it is an inducer of UDP-glucuronyl transferase (UDPGT), 66 an enzyme that leads to the glucuronidation and excretion of thyroxine. 67 The A1254 treatment drastically suppressed thyroxine concentrations. Therefore, it is plausible that additional modulating effects of the various terminal procedures were no longer detectable in A1254-treated groups. The mechanisms responsible for a decrease in thyroxine concentration appear to differ depending on the dosing protocol of A1254. While a high dose (600 mg/kg ip) of A1254 increased UDPGT activities, 68 a smaller dose was without effects on UDPGT activity (10 mg/kg/day over 14 days) and the reduction in thyroxine was associated with displacement from the transport protein rather than activation of UDPGTs. 69 A1254-treated rats consistently showed a decrease in thyroid hormones. 27,55,61,70,71 , but effects on TSH are inconsistent and chemical-specific. 55,61,67,71 .
Enzymology and Biochemistry
Although some members of the CYP family have been shown to be sensitive to anesthetic neurodepressants, 36 no effect of anesthetics could be detected on EROD, PROD, BROD, or AH activities (Table 2). As expected, A1254 increased hepatic enzyme activities. 27,56,72,73 PROD activities were slightly lower in A1254-EQ-E treated rats perhaps due to competitive substrate inhibition between PCBs and the pentobarbital component of the EQ mixture (pentobarbital being a CYP2B inducer 74 ). This is a subtle effect given that EQ had no effects on PROD and BROD in the non-A1254-treated groups.
Multiple statistically significant effects were observed among clinical enzymology and biochemistry endpoints (Figures 3 and 4), but these effects can be influenced by various mechanisms. 54 Therefore, only a general and brief discussion of these effects is presented to avoid speculations. Hypercholesterolaemia is known to be induced by PCBs and pentobarbital in the rat 55,56,75,76 ; and in human. 29,77 The elevated cholesterol concentrations, ALT, and CYP activities, observed in all A1254-treated groups regardless of the terminal procedures, are thus robust indicators of A1254-induced hepatic effects. Enzyme leakage due to the extent of cell and tissue damage prior or during blood collection (trunk blood compared to opening the abdominal cavity to access the abdominal aorta), and the severity of hepatotoxicity, are known to alter blood measurements of LD, ALT, and AP, and can have affected these measurements. The immobilization procedures (anesthetics, CO2) induce respiratory acidosis to various extents, which affect blood pH, LD, ALT, and uric acid measurements. 54 Accordingly, our data provide examples of how CO2 (CO2-D vs A1254-CO2-D vs group D) decreased LD and uric acid and increased ALT. Hypophosphatemia was induced by most terminal procedures except CO2, consistent with reported effects of anesthetics. 78 The levels of calcium, phosphate, and magnesium are intimately regulated. 54 Some factors that can affect the concentrations of these cations and that are relevant to our study include stress, 60 sodium pentobarbital increasing parathyroid hormone concentrations, 79 and magnesium sulphate in the EQ mixture (magnesium and calcium are competitive with respect to their absorptive sites 4 ).
In conclusion, all immobilization procedures (CO2 narcosis, Iso and EQ anesthesia) interfered with some endpoints, particularly EQ, which induced glucocorticoid stress reactions and altered other hormone concentrations compared to decapitation without anesthesia. The results demonstrate that terminal procedures can change conclusions related to the measurements of 9 out of 21 endpoints (Table 3). Briefly, the effects of A1254 on the concentrations of corticosterone, insulin, glucagon, glucose, alkaline phosphatase, lactate dehydrogenase, uric acid, and blood urea nitrogen changed from nonstatistically significant to significant, and in the case of LH from a statistically significant increase to a decrease. The possibilities to extrapolate these findings to other PCBs or toxicants are limited given that A1254 is a mixture of PCBs, and effects are dependent upon a series of factors (dose and dosing regimen, species/strain, age, gender, etc) that are different for each study. Nevertheless, the choice of terminal procedure and the use of anesthetics may help to explain differing results between otherwise “identical” studies. For neurochemistry endpoints also, our previous work 14 demonstrated that the selection of a terminal procedure can mask effects, can induce significant changes not attributable solely to the dosing chemical, or alter the apparent effects of the dosing chemical. This is especially true when considering the effects of chemicals being dosed at environmentally relevant levels where effects tend to be subtle and hard to detect at the best of times. Investigation of toxicants should be designed considering animal welfare and interpreted considering confounding effects of terminal procedures, which can contribute to differences across toxicological investigations.
Footnotes
Acknowledgments
We are grateful to Gail Merriken, Kevin Kittle, Judy Edgar, James Elwin, Tanya Hoeksma, Brita Nadeau, Sheila Masson, Karen Leingartner, and Jonathan Cole for technical assistance and to Dr. G Pelletier and Dr. R Poon for reviewing this manuscript.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: Health Canada.
