Abstract
Nonclinical rodent studies with repeat slow intravenous dosing, such as safety assessments of anticancer therapeutics, often require the use of animals with surgically implanted catheters. Catheterization is a relatively short surgical procedure but requires use of anesthesia. Ketamine/xylazine injectable anesthesia is typically used because it has advantages over inhalation anesthesia including ease of administration, safety and predictability of effects, and relatively low cost. However, ketamine/xylazine anesthesia in rodents can also be associated with the development of undesirable corneal lesions of uncertain mechanism such as mineralization of Bowman’s membrane or stroma, erosion/ulceration, inflammation, fibroplasia, and neovascularization. Such findings have the potential to confound study interpretation in programs for which the cornea is a potential target tissue. This case report describes the occurrence of ketamine/xylazine-related corneal lesions observed in surgically catheterized rats in a 16-day toxicity study for an oncology compound.
A non-GLP (Good Laboratory Practice) 16-day 3-dose (dosed on days 1, 8, and 15) intravenous infusion toxicity study of an oncology compound was performed in Sprague-Dawley rats (CD [Crl: CD(SD)], Charles River Laboratories) with indwelling femoral vein catheters previously implanted by the laboratory animal supplier. The cornea was considered a target tissue based on previous study findings of test article–related single-cell necrosis of the basal epithelium. At termination, corneal lesions included epithelial single-cell necrosis in test article–dosed animals and stromal mineralization and inflammation in both control and test article–dosed animals. The incidence of corneal stromal mineralization and inflammation greatly exceeded the facility historical control incidence for 2-week studies of Sprague-Dawley rats without indwelling catheters using oral, subcutaneous, or intravenous routes of administration. These findings were attributed to prestudy anesthesia associated with cannulation procedures.
All procedures were performed in accordance with established federal and state regulations and Test Facility Standard Operating Procedures and reviewed and approved in advance by the Charles River Mattawan Institutional Animal Care and Use Committee. Thirty-two main study rats were implanted with femoral catheters by the supplier 13 days prior to study initiation (day –13). Intraperitoneal injections of a combination of ketamine (Ketathesia, Henry Schein Animal Health: 70-100 mg/kg) and xylazine (AnaSed, Akorn Animal Health: 6-10 mg/kg) were administered along with subcutaneous buprenorphine HCl (Buprenex, Reckitt Benckiser Pharmaceuticals: 0.02 mg/kg) for analgesia. Anesthesia generally lasts 30 to 45 minutes at these dose levels, and the surgical procedure is typically completed within 20 minutes. Petroleum-based eye lubricant (Puralube Ophthalmic Ointment; Dechra) was applied bilaterally before and after catheter placement, and heating pads were placed under the animal cages until recovery.
Animals were shipped to the study site (Charles River Laboratories) on day -9 and retained for an acclimation period until study initiation. The rats were randomly assigned to groups (4 animals/sex/group) and were dosed with vehicle (5% dextrose in water [D5W]) or test article (low, mid, or high mg/kg doses). Animals were single housed. Prestudy ophthalmologic examinations were not required by protocol, but no eye lesions were observed during routine evaluation upon arrival (day -9) which included gross examination of the eyes. Cageside observations (at least twice daily) and detailed clinical observations (3 times per week) occurred throughout the study beginning on day 1. Corneal lesions (eye discolored: cloudy, unilateral or bilateral) were observed clinically in 1 control female and 1 male and 3 females in the high-dose group beginning on day 2 through day 16 of the study. No ocular lesions were observed clinically in animals in the toxicokinetic portion of the study (3 animals/sex/group).
Complete necropsy examinations were performed on one early death male at the mid dose and all surviving animals at the scheduled euthanasia on day 16. The eyes were immersion fixed in modified Davidson’s fixative, processed conventionally, embedded in paraffin, sectioned at 4 to 6 µm to obtain 2 midsagittal sections per eye, and stained with hematoxylin and eosin for histopathology. Microscopic examination using a 5-grade severity scale (minimal, mild, moderate, marked, and severe) was performed by an American College of Veterinary Pathologists (ACVP) board-certified pathologist (L.S.Z.), and a peer review was conducted by a sponsor-designated ACVP board-certified pathologist (M.F.R.).
Macroscopic findings included unilateral or bilateral corneal surface irregularity (1 control female and 2 females in the high-dose group) and bilateral cloudiness (1 female in the high-dose group). Correlative microscopic findings were present in all affected animals except one female in the high-dose group.
Microscopically, test article–related ocular findings were limited to minimal to mild single-cell necrosis of the basal corneal epithelium observed at all dose levels of the test article. Non-dose-related corneal findings that were attributed to prior anesthesia occurred in control and test article–dosed animals as summarized in Table 1.
Number of Rats With Anesthesia-Related Microscopic Corneal Lesions.
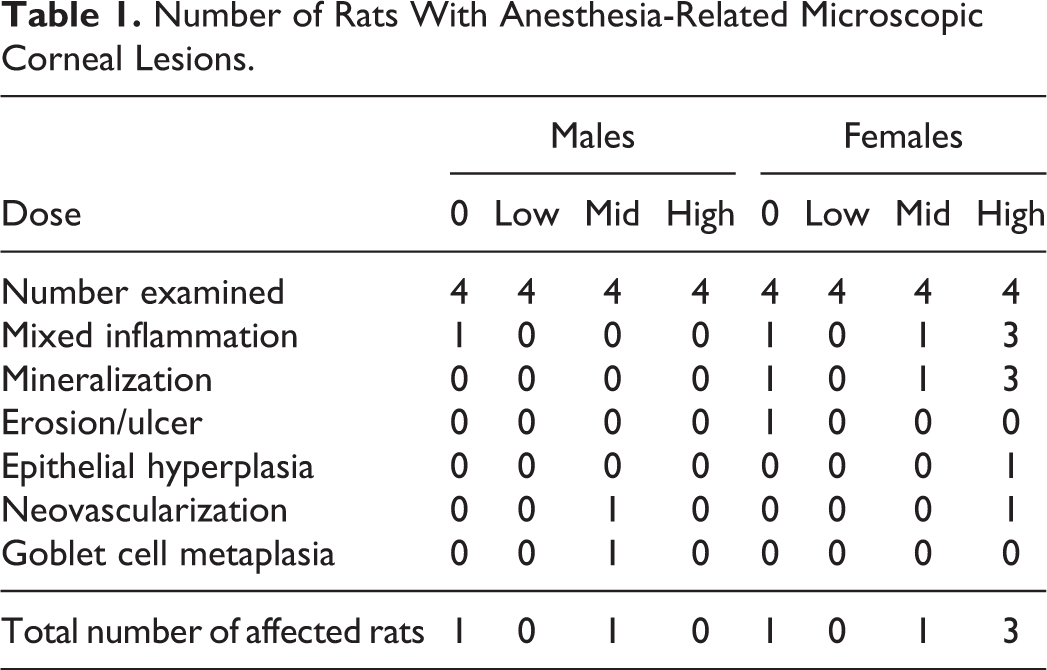
Overall, corneal lesions occurred in 2 (12.50%) of 16 males and 5 (31.25%) of 16 females. For comparison, corneal lesions (epithelial hyperplasia or dysplasia) were only present in 3 (2.50%) of 120 males and 1 (0.83%) of 120 females in recent (5-year period) historical control data for 2-week studies in Sprague-Dawley rats using other methods of administration not requiring prestudy anesthesia (Charles River Mattawan historical control database). Most lesions attributed to anesthesia in the current study occurred in the central aspect of the cornea. Findings were unilateral or bilateral and typically ranged from minimal to mild severity, with moderate findings limited to inflammation in one female in the high-dose group. Given the low magnitude of the microscopic corneal findings in most affected animals, it was unsurprising that these subtle lesions may not have been grossly visible during routine clinical examinations. The most commonly observed findings were mineralization of Bowman’s membrane and/or subjacent stroma with variable-associated inflammation and artifactual cleft formation (Figure 1). The inflammation was composed of variable combinations of neutrophils, mononuclear cells, and/or multinucleated giant cells with or without attendant fibroplasia that usually bordered foci of mineralization or erosion/ulceration. Other corneal findings that were present at a low incidence included neovascularization and reactive changes in the epithelium such as hyperplasia or goblet cell metaplasia that occurred overlying foci of stromal inflammation and/or neovascularization.
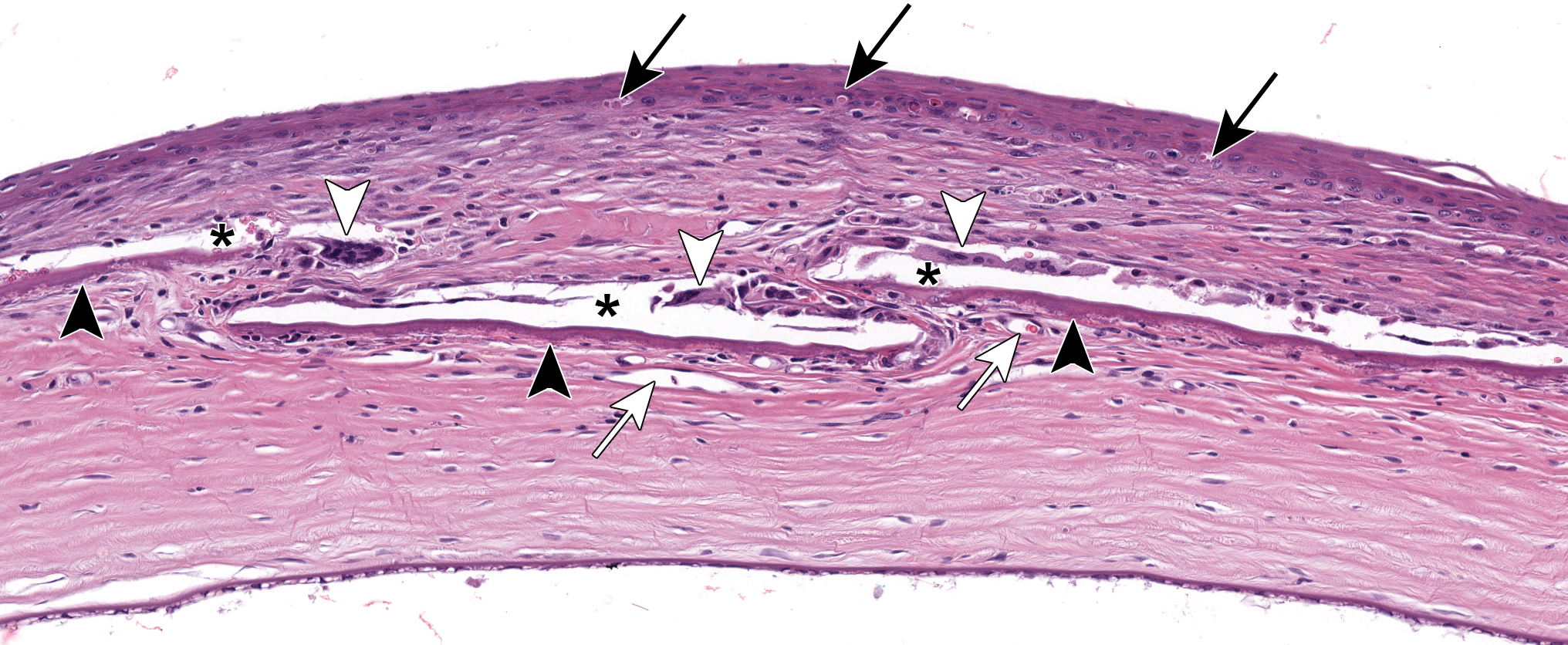
Representative anesthesia-related corneal stromal lesion of moderate severity from a drug-treated rat. The anterior corneal stroma is expanded by fibroplasia with neovascularization (white arrows) encompassing several linear foci of mineralization (black arrowheads). Mineralization is surrounded by multinucleated giant cells (white arrowheads) with adjacent cleft formation (black asterisks: likely artifactual change). Black arrows depict single-cell necrosis of basal corneal epithelial cells, the expected drug-related finding. Original scan, ×20.
Combinations of ketamine hydrochloride (a dissociative agent) and xylazine (an α-2 adrenergic agonist) are commonly used for injectable anesthesia in rodents due to their safety and reliability for procedures of short duration such as intravenous catheter placement. 1 However, ketamine/xylazine anesthesia in rodents can be associated with development of keratoconjunctivitis sicca and/or corneal lesions such as mineralization, erosion/ulceration, inflammation, and neovascularization. 2 –6 Corneal lesions have been primarily attributed to the α-2 adrenergic agonistic effects of xylazine. Similar lesions can be induced by administration of clonidine (a more potent α-2 adrenergic agonist than xylazine), and the incidence and severity of the corneal lesions is prevented or reduced with administration of yohimbine (an α-2 adrenergic antagonist). 2,5 –7 The mechanism of corneal toxicity is unknown but possible causes include hypoxia secondary to persistent vasoconstriction, 5 osmotic stress associated with increased glucose concentration in the aqueous humor due to inhibition of insulin release, mechanical endothelial stress due to increased ocular pressure resulting from prolonged mydriasis, or other unknown direct α-2-adrenergic receptor–mediated effects. 7 Coadministration of ketamine has previously been associated with an increased incidence of corneal lesions compared to xylazine administration alone 2 and may have a contributing role. Corneal surface desiccation and exophthalmos associated with anesthesia can contribute to development of corneal lesions similar to those observed in the current study. 8 However, the usage of eye lubricant and supplemental heat during anesthesia, both of which were used in the current study, have not been shown to be preventative. 2,3 Susceptibility to ketamine/xylazine anesthesia–related corneal lesions in rats has been reported to vary by strain (Wistar, Long-Evans, and Fischer 344 strains are more susceptible than Sprague-Dawley and Lewis rats) and age (juveniles are more susceptible than adults), with no sex predilection. 2,5
Spontaneous corneal mineralization, degeneration, and inflammation have also been reported in multiple rat strains including Sprague-Dawley rats. 9 –14 These spontaneous corneal lesions have been reported in rats ≥7 weeks of age with an age-related increase in incidence. Genetic and environmental factors may contribute to their development.
Prestudy ophthalmologic examinations are recommended to help exclude animals with anesthesia-related or spontaneous corneal lesions that could confound interpretation of ocular findings in toxicologic studies, especially for test articles for which the eye is a potential target tissue (eg, antibody-drug conjugates). Usage of age-dependent dosages of ketamine/xylazine, supplementation with 100% O2 by facemask during anesthesia, and administration of yohimbine as a reversal agent may help reduce the incidence of ketamine/xylazine anesthesia–related ocular lesions in rodents. 2,5,6
Footnotes
Acknowledgments
The authors would like to acknowledge the assistance of Dale Cooper, Senior Director and Attending Veterinarian at Charles River Laboratories, Mattawan, MI.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M. Ramos is an employee of AbbVie Inc, North Chicago, IL.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study and the publication cost were funded by AbbVie Inc, North Chicago, IL.
