Abstract
Rodent lungs are routinely examined after intratracheal instillation (IT) of fixative. This study compares the histopathologic appearance of the lung after IT fixation with air inflation (AI) followed by immersion fixation. Lungs from mice chronically exposed to cigarette smoke were fixed either by IT or by AI. Increased numbers of macrophages with differing distributions were seen in both groups. Lungs fixed by IT had prominent, large macrophages floating in the alveolar lumina, as well as macrophage clusters and loose aggregates, often near terminal airways. Macrophages in lungs fixed by AI were randomly distributed throughout the lung, lying singly along alveolar walls, with large numbers visible in the interstitium. Clusters of macrophages were seen in the airways after AI but were fewer after IT fixation. The effects of intratracheal fixation on lung macrophages need to be considered carefully when assessing the significance of changes in macrophage appearance and distribution.
Keywords
Introduction
Several recent initiatives have refocused interest on the standardization of nomenclature, diagnostic criteria, and significance of changes in lung macrophages in toxicological studies in rodents. The International Harmonization of Nomenclature and Diagnostic Criteria for Lesions in Rats and Mice Project (INHAND) recommended nomenclature for classifying changes in the respiratory tract of rodents (Renne et al. 2009). More recently, the Society of Toxicologic Pathologists (Nikula et al. 2014), together with the recent review by Forbes et al. (2014), have helped to harmonize our understanding of macrophage findings in the rodent lung. Pulmonary macrophages are a normal component of the mammalian lung, providing clearance of particulate material in the lung, increasing in number as a normal physiological response to increases in particulate lung burden. Macrophages can be seen throughout the lung, occasionally near terminal airways where they can form clusters of large rounded macrophages with variable amounts of vacuolated (foamy) cytoplasm. This can be seen in the absence of other histopathologic signs such as inflammatory cells, hemorrhage, alveolar wall thickening, or phospholipidosis. Such clusters can be seen in a variety of studies including non-inhalation studies and are recorded both in control and in test article groups. Macrophages migrate freely over the alveolar surfaces phagocytosing particulates before moving either into the interstitium of the lung or to the terminal airways to travel cranially along the airways to the pharynx on the “muco-ciliary escalator.” Thus, there are 3 macrophage populations in the lung: alveolar macrophages in the airspaces, interstitial macrophages sequestered in the lung interstitium, and airway macrophages migrating cranially. Macrophages recovered by bronchopulmonary lavage are often assumed to be alveolar macrophages, instead of, more accurately, a mixture of alveolar and airway populations.
Little attention has been paid to the airway macrophage populations in the lung since they are difficult to separate. Brain, Gehr, and Kavet (1984) studied airway and alveolar macrophage distribution in the hamster by comparing the effects of intratracheal and intravascular fixation as a means of separating and examining the 2 populations. Using intravascular fixation for comparison, they were able to demonstrate a 70% decrease in airway macrophages after IT fixation. Clumps of macrophages were described in the alveoli after intratracheal fixation “suggesting displacement of cells by the fixative.” Matulionis (1986) exposed mice to cigarette smoke 7 days a week for 5 months and then compared pulmonary macrophages per unit area of lung tissue after immersion fixation, intratracheal fixation, and bronchoalveolar lavage followed by intratracheal fixation. Numbers of lung macrophages were increased after intratracheal fixation compared with immersion fixation. Airway macrophages were common in lungs fixed by immersion fixation but were “seldom observed” after intratracheal fixation. In addition, he described clusters of macrophages “in close proximity to terminal airways” after intratracheal fixation. These cells contained more phagocytosed material than in macrophages fixed by immersion, leading Matulionis to suggest that these cells were older—perhaps representing senescent airway macrophages that had been displaced by fixation. This increase in size in airway macrophages was confirmed by Lehnert et al. (1990) using flow cytometry to compare airway and alveolar macrophage populations. Airway macrophages were bigger, having larger nuclear and cytoplasmic volumes as well as a larger total lysosome profile area (which might thus appear as visible inclusions by microscopy). Rankin et al. (1992) developed a lavage technique for retrieving human airway macrophages and also concluded that they had more vacuoles. Other workers have found a correlation of macrophage size with age, with the largest macrophages being the most mature (Harbison et al. 1984).
Materials and Methods
We were interested in comparing pulmonary macrophages in lungs fixed by conventional intratracheal fixation (IT) with macrophages in lungs fixed after air inflation (AI), which avoids disruption of airway and alveolar contents. All procedures and care of animals were in accordance with the principles for humane treatment outlined by the Institute of Laboratory Animal Resources Guide for the Care and Use of Laboratory Animals of the National Institutes of Health, USDA Guidelines Animal Welfare Regulations, and were reviewed and approved by the GlaxoSmithKline Institutional Animal Care and Use Protocols Committee.
Twenty female C57BL/6 mice (Jackson Laboratory, Bar Harbor, ME) received nose-only exposure to 4% cigarette smoke from 3R4F cigarettes (College of Agriculture, Reference Cigarette Program, University of Kentucky), for 2 hr/day, 5 days/week, for 22 months. Smoke was generated by a Baumgartner-Jaeger CSM 2070i Smoking Machine (CH Technologies Inc., Westwood, NJ). The mice were divided into 2 groups of 10 and the lungs were then fixed, either by IT or by AI. Following euthanasia (isofluorane anesthesia and cardiac exsanguinations followed by cervical dislocation), the rib cage was removed and the trachea and lungs exposed. For intratracheal inflation, the trachea was grasped and pulled caudally, and the lungs were lifted clear after careful dissection. A cannula was then inserted into the trachea and the lungs filled with 10% neutral-buffered formalin until the pleural surfaces were smooth and glistening. The trachea was then tied off and the lungs immersed in fixative prior to processing. For air inflation, a cannula was inserted into the trachea with the lungs in situ and the lungs gently inflated with air from a large syringe until the pleural surfaces were smooth and glistening. A steady gentle inflation pressure was then applied to maintain lungs at approximate total lung capacity while fixative was gently flooded onto the exposed pleural surfaces from a second syringe for approximately 3 to 5 min. The trachea was then tied off and the lungs immersed in 10% neutral-buffered formalin for 24 to 48 hr prior to processing (covered with a paper towel to ensure thorough fixation). The entire lung from each animal was embedded in paraffin and a single sagittal section across all lobes of the right and left lung was cut and stained with hematoxylin–eosin (HE). For immunohistochemical investigations, a goat polyclonal antibody against a peptide mapping at the C-terminus of cathepsin S of rat origin (cat# sc-6505; Santa Cruz Biotechnology, Santa Cruz, CA) was used for detecting cathepsin S in macrophages. Tissue sections were scanned into digital whole-slide images at 20× or 40× magnification using an Aperio ScanScope® AT Turbo digital slide scanner (Leica Biosystems, Vista, CA) and examined using Aperio Spectrum™ and ImageScope.
Results
Histopathologic examination of the 2 groups revealed marked differences in macrophage appearance and distribution. In both groups, macrophages contained large amounts of golden-brown pigment characteristic of chronic exposure to tobacco smoke. In lungs fixed by IT, alveolar macrophages were large, rounded, and prominent, often appearing to float in the alveolar lumina (Figure 1A, B, and C). Pure aggregates of these large spherical macrophages were frequently seen scattered in the deep lung often adjacent to terminal airways (Figure 1A). The interstitium was flattened by the pressure of fixative and so interstitial macrophages could not be easily evaluated (Figure 1B). Airway macrophages were sparsely distributed but still seen occasionally (Figure 1C). Macrophages in lungs fixed by AI appeared as flattened profiles lying along the alveolar walls, conforming to the contours of the alveolus (Figure 1D and E [inset]). The interstitium was not flattened by fixative, and large populations of intra-alveolar and interstitial macrophages could be seen scattered irregularly throughout the parenchyma (Figure 1E). Airway macrophages were seen on airway surfaces at all levels (Figure 1F), but alveolar macrophage clusters in the regions adjacent to terminal airways were not seen.
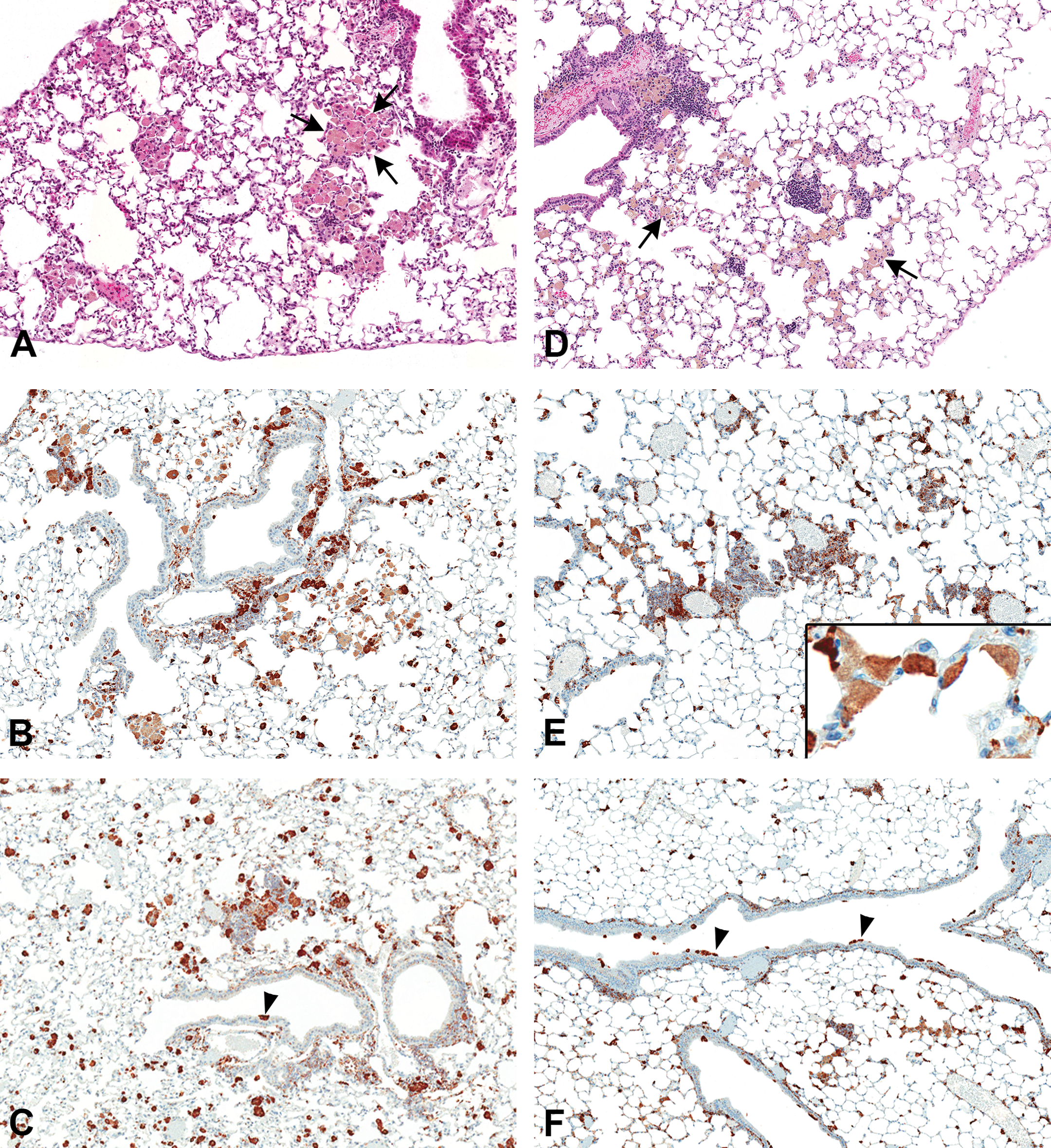
Lung macrophages after intratracheal fixation (A, B, C) and air inflation (AI)/immersion fixation (D, E, F). (A) Aggregates of macrophages in alveoli of the distal lung (arrows), HE, original magnification 10×. (B) Cathepsin S. Immunohistochemistry of lung macrophages, original magnification 10×. (C) Cathepsin S. Immunohistochemistry of airway macrophages—single macrophage on airway wall (arrowhead), original magnification 4×. (D) Lung macrophages scattered on alveolar walls and in the interstitium (arrows), HE, original magnification 10×. (E) Cathepsin S. Immunohistochemistry of lung macrophages, original magnification 10×. Inset showing flattened macrophage profiles on alveolar walls, original magnification 40×. (F) Cathepsin S. Immunohistochemistry of airway macrophages showing small groups on airway walls (arrowheads), original magnification 4×.
Discussion
Inflation of the lung with intratracheal fixative was originally proposed to characterize centrilobular emphysema in human lung specimens (Leopold and Gough 1957; Heard 1958). It is a simple and easy way to maintain the spatial architecture of the lung at approximate total lung capacity, as well as providing a large volume of fixative in intimate contact with airway and alveolar surfaces, ensuring rapid fixation (Dungworth et al. 1976). Several other methods of inflation fixation have been described such as intravascular fixation (Gil 1971), formalin vapor fixation (Weibel and Vidone 1961), and microwave fixation (Turner et al. 1990), but these are labor intensive and impractical for routine toxicologic studies. Because of this, intratracheal instillation of formalin has been the method of choice for many years in rodent toxicology studies, where large numbers of lungs need to be fixed promptly. As a result, most toxicologic pathologists have never seen rodent lungs fixed by any other route.
In this study, simply inflating the lungs with air and then tying off the trachea immediately resulted in rapid collapse of the lung lobes, presumably due to the elastic recoil of the lung. For this reason, we developed a method to maintain a slow steady inflation pressure with air over several minutes while dropping the fixative onto the exposed pleural surfaces. Inflation, though not precise, maintained approximate total lung capacity and allowed us to assess and compare the distribution of macrophages in the lung by the 2 techniques. Our findings were essentially in agreement with those of Brain, Gehr, and Kavet (1984) and Matulionis (1986). After IT fixation, airway macrophages were present but in small numbers, while alveolar macrophages were large and rounded, appearing to float freely in the alveoli. There were numerous macrophage aggregates in the deep lung, usually near terminal airways. The interstitium was compressed and therefore interstitial macrophages were difficult to evaluate. After AI, airway macrophages were frequently visible, often in small groups. Alveolar macrophages were seen flattened along the alveolar walls, usually as flattened profiles. Because the interstitium had not been compressed by the pressure of the fixative, numerous interstitial macrophages were clearly visible.
Microscopic examination of the lung is enhanced by inflation. Air inflation of the lung preserves the normal distribution and appearance of macrophage populations on the airway and alveolar surfaces as well as in the interstitium. However, the routine technique of IT affects the microscopic appearance of the lung and its cellular constituents, particularly the airway and alveolar macrophage populations. It disrupts both populations, resulting in flushing artifacts and altered distribution, making it likely that there is a mixing of the 2 populations in the deep lung. It is not known if the airway macrophages, being bigger and having more prominent vacuoles than alveolar macrophages, influence the overall macrophage appearance in the lung.
A subcommittee of the Society of Toxicology has reviewed the various methods of lung fixation with formalin in rodent toxicology studies and recommended intratracheal instillation of formalin, stating that the “advantages of intratracheal instillation of formalin outweigh the disadvantages” (Renne et al. 2001). Other techniques, including the AI/formalin fixation technique described here are not practical for routine studies. However, this comparative study illustrates the differences in the histopathologic appearance of lung macrophage populations using different fixation techniques. These differences and, in particular, the toxicological significance of macrophage findings in lungs fixed by intratracheal instillation should be carefully considered when assessing microscopic changes in lung macrophages in toxicology studies.
Footnotes
Acknowledgment
The authors would like to thank Drs. Deon Hildebrand and Justin Vidal for many critical discussions. Ms. Bev Maleeff provided invaluable technical support.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author Contribution
Authors contributed to conception or design (EBW); data acquisition, analysis, or interpretation (PLP, RCM); drafting the manuscript (EBW); and critically revising the manuscript (PLP, RCM). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
