Abstract
Previous studies indicated that exposure to fine particulate matter (PM2.5) was related to pulmonary inflammatory diseases through activation of nuclear factor kappa B (NF-κB) signaling pathway to trigger cytokine secretions in human lung carcinoma cells. To investigate the potential mechanisms underlying expression of cytokines via activated NF-κB by PM2.5, human bronchial epithelial cells (BEAS-2B cells) were treated with PM2.5 extracts at different concentrations (6, 13, 25, 50, 100, 200, and 400 µg mL−1) for 6 and 24 h. We found that 100 µg mL−1 PM2.5 increased interleukin 6 (IL-6) and IL-8 expression at 24 h (p < 0.05 or p < 0.01). Moreover, 100 µg mL−1 PM2.5 upregulated phosphorylated IκB kinase (IKK), p65, and IκBα at 6 h, which could be reversed by the IKK inhibitor Bay11-7082 (p < 0.05 or p < 0.01). The p65 subunit of NF-κB was translocated into the nucleus of the cells treated with 100 µg mL−1 PM2.5 at 6 and 24 h. Bay11-7082 partly inhibited PM2.5-induced increases of IL-6 and IL-8 secretion. The results indicated that PM2.5 extract increased IL-6 and IL-8 levels in BEAS-2B cells through activation of IKK/NF-κB pathway. Our study will contribute to better understanding of the mechanism of PM2.5-induced pulmonary inflammatory diseases.
Keywords
Introduction
Particulate matter (PM) can provoke adverse health effects, such as cardiovascular diseases 1 and pulmonary diseases, 2,3 and birth defects. 4 PM2.5 (i.e. fine PM) has an aerodynamic diameter of 2.5 microns or smaller. PM2.5 from biomass burning, coal combustion, motor vehicle emission, dust, and secondary sulfate and nitrate 5,6 is composed primarily of various toxic substances, including metals, organic carbons, and elementary carbons. 7,8 Numerous studies showed that PM2.5 is linked to increased risk of pulmonary diseases 9 –11 ; however, the underlying pathogenic mechanisms for PM2.5-induced respiratory diseases remain largely unclear. Airway inflammation is a prominent feature of respiratory diseases. 12,13 As the first target of inhaled air pollutants, airway epithelial cells release cytokines to induce inflammatory response 14 and trigger respiratory diseases. However, there are variable findings about PM2.5-induced cytokine secretions. In vitro studies showed that the PM2.5 samples increased cytokine secretions in lung epithelial cells, including interleukin 6 (IL-6), IL-8, and tumor necrosis factor α (TNFα). 15 –17 Whereas no increased IL-8 levels were detected in human bronchial epithelial cell line (BEAS-2B) and human lung adenocarcinoma cell line (A549) treated with PM2.5 from Milan (Italy) 18 and the decreased IL-6 and IL-8 levels were detected in BEAS-2B cells treated with PM2.5 samples from a city in Brazil. 19 PM2.5-induced variations of cytokines in humans may be modulated by multiple molecular mechanisms, 20 –22 owing to differences in the origin, morphology, and composition of PM2.5 from different regions or countries. It is thus necessary to further clarify the effects of PM2.5 on cytokine secretion and elucidate the underlying molecular mechanisms of PM2.5-induced inflammatory response.
Nuclear factor kappa B (NF-κB) is implicated to regulate inflammatory response, proliferation, and apoptosis. 23 It is sequestered in the cytoplasm by interacting with the inhibitory proteins IκBs. Upon inflammatory stimulation such as TNFα, lipopolysaccharide, and IκB kinase (IKK), 24 IκBs are degraded and subsequently release NF-κB to facilitate its translocation into the nucleus and transcription of target genes. It has been found that NF-κB was activated in PM2.5-induced inflammatory response. PM2.5 samples from Seoul (South Korea) induced IκBα degradation and NF-κB activation in A549 cells. 25 PM2.5 samples from Dunkerque city in France increased cytoplasmic phosphorylation of IκBα and p65 and nuclear DNA-bindings of p65 and p50 in L132 lung epithelial cells. 26 NF-κB inhibitor (Bay11-70-85) partly blocked PM2.5-induced IL-8 release in A549 cells, indicating that inflammation in response to PM2.5 was regulated by NF-κB pathway. 27 However, the mechanisms by which NF-κB-mediated inflammatory response induced by PM2.5 are still uncertain.
NF-κB can promote the release of cytokines such as IL-6, IL-8, IL-12, and TNFα, whereas IL-1β and TNFα activate NF-κB. The positive regulatory loop contributes to amplified local inflammation. 28 –30 Adverse effects of PM2.5 on respiratory system in humans are more harmful than exposure to coarse PM, because PM2.5 has a potential to deposit in the lower respiratory tract and the lungs with a large surface area per unit mass, leading to more pollutants, were adsorbed. 31 In the present study, we investigated the molecular mechanisms underlying the inflammatory response in relation to the IKK/NF-κB pathway in BEAS-2B cells treated with the PM2.5 extracts from Wuhan, China. This study will further provide molecular basis to uncover the mechanisms by which PM2.5 induces inflammatory response and respiratory diseases.
Materials and methods
Materials
We purchased dimethylsulfoxide (DMSO) and IKK inhibitor (Bay11-7082) from Sigma-Aldrich (St Louis, Missouri, USA) and 3-(4,5-dimethyithiazol-2-yl)-2,5-diphenyl-tetrazolium bromide (MTT) from Amresco (Solon, Ohio, USA). As one of adenovirus-12/SV40 hybrid virus immortalized human bronchial epithelial cell lines, BEAS-2B was kindly supplied by Professor Lun Song (Academy of Military Medical Science). Dulbecco’s modified Eagle’s medium (DMEM) and fetal bovine serum were obtained from Gibco, Gaithersburg, Maryland, USA. Enzyme-linked immunosorbent assay (ELISA) kits for the detection of IL6, IL8, and TNFα levels as well as the nuclear protein and cytoplasmic extraction kits were purchased from Neobioscience Technology (Shenzhen, China) and KeyGen Biotechnology (Nanjing, China), respectively. Antibodies of p-IKKα/β (Ser176/180, Ser177/181), p-IκBα (Ser32), p-p65 (Ser536), IKKα, IKKβ, IκBα, and p65 were obtained from cell Signaling technology (Danvers, Massachusetts, USA). Anti-β-Actin antibody was purchased from MultiSciences Biotechnology (Hangzhou, China). Anti-Lamin A antibody was from Santa Cruz Biotechnology (Delaware Avenue, California, USA). Goat anti-rabbit or mouse secondary antibodies were obtained from Bioworld Technology (St Louis Park, Minnesota, USA).
Collection and extraction of PM2.5 samples
We collected PM2.5 samples in the Shanghai Street in Wuhan in the winter (January 27 to February 14) of 2012. The sampling site was located at the urban district of this city, and there was no other known pollution sources surrounding it. The 2-μm pore size Teflon filters with 90-mm diameter were used to collect PM2.5 samples using middle volume samplers (model TH-150; Wuhan Tianhong Instrument Co., Ltd, Wuhan, China) at a flow rate of 80 L min−1 for 23 h. Then, the filters were transported to the laboratory with ice box and stored at −20°C. The filters with PM2.5 were cut into several fragments, and then immersed in ultrapure water (ELGA PURELAB ultra, 18.2 MΩ) in an ultrasound bath (KQ-500DE; Kunshan Ultrasonic Instrument Co. Ltd, China) for 2 h at 25°C. Particles on the filters were removed by ultrasound treatment in the bath. The extract was lyophilized for 23 h using a freeze-dry system (CHRIST ALPHA1-4; Martin Christ Gefriertrocknungsanlagen GmbH, Germany) and then stored at −80°C. The weather forecast data were simultaneously collected.
Cell culture and treatment
BEAS-2B cells were grown in DMEM with 10% fetal bovine serum and 1% penicillin-streptomycin at 37°C in a humidified incubator with 5% carbon dioxide and then treated with the PM2.5 extract at different concentrations (6, 13, 25, 50, 100, 200, and 400 μg mL−1) for 6 and 24 h, respectively. Bay11-7082 was dissolved in DMSO (final concentration < 0.1%) before the treatment. The concentrations (10 or 2.5 µM) and treatment time points (6 and 24 h) of Bay11-7082 used in this study were chosen based on the results of previous studies. 32 –35 BEAS-2B cells were exposed to Bay11-7082 (10 or 2.5 µM) for 1 h prior to the treatment of the PM2.5 extracts (50 and 100 μg mL−1) for 6 and 24 h, respectively.
MTT assay
MTT assay was used to determine cell viability according to the standard protocol by the manufacturer (Amresco, Solon, Ohio, USA). Briefly, BEAS-2B cells were seeded in 96-well plates at a density of 8 × 103 cells/well with DMEM and then treated with the PM2.5 extracts (6, 13, 25, 50, 100, 200, and 400 μg mL−1, correspondingly, the treated concentrations were equivalent to 2, 4, 8, 16, 31, 63, and 125 μg cm−2) for 6 or 24 h. After removing the medium, cells were incubated with 100-μL DMEM containing 0.5 mg mL−1 MTT for 4 h at 37°C. Intracellular formazan was solubilized with 150-μL DMSO. The plates were shaken for 10 min at room temperature. After the crystal dissolved completely, we detected the absorbances of the samples at 570 nm with a microplate reader (BioTek Instruments Inc., Winooski, Vermont, USA). The results were presented as the percentage of control.
Enzyme-linked immunosorbent assay
BEAS-2B cells were seeded in six-well plates at a density of 2 × 105 cells/well with DMEM and then treated with the PM2.5 extracts (6, 13, 25, 50, and 100 μg mL−1, the treated concentrations were equivalent to the PM2.5 concentrations of 1, 3, 5, 10, and 21 μg cm−2) for 6 or 24 h, respectively. Based on the results above, co-treatment of 100 μg mL−1 PM2.5 with Bay11-7082 (2.5 μM) was performed to test the effect of IKK/NF-κB on cytokine secretions, namely, BEAS-2B cells were pretreated with or without Bay11-7082 (2.5 μM) for 1 h and then treated with 100 μg mL−1 PM2.5 for 24 h. Supernatants were obtained by centrifugation at 14,000 × g for 15 min. The levels of IL-6, TNFα, and IL-8 in supernatants were measured using ELISA kits according to the corresponding manufacturer’s instructions.
Western blot
BEAS-2B cells were seeded in six-well plates at a density of 2 × 105 cells/well with DMEM and then treated with PM2.5 extract of 50 μg mL−1 (10 μg cm−2) or 100 μg mL−1 (21 μg cm−2) for 24 h. BEAS-2B cells were pretreated with or without Bay11-7082 (10 μM) for 1 h prior to the treatment of PM2.5 extract of 50 μg mL−1 (10 μg cm−2) or 100 μg mL−1 (21 μg cm−2) for 6 h. BEAS-2B cells were seeded in 100-mm dishes at a density of 1 × 106 cells/well with DMEM and pretreated with or without Bay11-7082 (10 or 2.5 μM) for 1 h prior to the treatment of PM2.5 extract (50 (9 μg cm−2) or 100 μg mL−1 (18 μg cm−2)) for 6 and 24 h. Whole-cell proteins and nuclear proteins were extracted using radio immunoprecipitation assay lysis buffer and the nucleus and cytoplasm protein extraction kits, respectively, according to the corresponding manufacturer’s instructions. Proteins were electrophoresed on 8% SDS-PAGE gels and transferred to polyvinylidene difluoride membranes. The membranes were incubated with the primary antibodies of p-Ikkα/β, p-IκBα, p-p65, Ikkα, Ikkβ, IκBα, p65, Lamin A, or β-Actin and the secondary antibody of goat anti-rabbit or mouse. The bands with an enhanced chemiluminescence reagent were detected with a chemiluminescent imaging system (GeneGnome; Syngene Inc, Frederick, Maryland, USA).
Statistical analysis
Data were expressed as the means ± standard deviation of at least three independent experiments and analyzed by the one-way analysis of variance with the least significant difference test using SPSS 12.0 software (SPSS Inc., Chicago, Illinois, USA). A statistical significance was considered at p < 0.05.
Results
Cell viability
To identify PM2.5 concentrations used in the following experiments, we firstly tested cell viability using the MTT assay. Cell viability was significantly decreased after treatment with 100, 200, and 400 μg mL−1 PM2.5 for 6 h or 25, 50, 100, 200, and 400 μg mL−1 PM2.5 for 24 h (p < 0.05 or p < 0.01; Figure 1). Therefore, PM2.5 extracts at the concentrations of 6, 13, 25, 50, and 100 μg mL−1 were used to the following experiments.

The cell viability of PM2.5-treated BEAS-2B cells. The cells were treated with 6 μg mL−1 (2 μg cm−2), 13 μg mL−1 (4 μg cm−2), 25 μg mL−1 (8 μg cm−2), 50 μg mL−1 (16 μg cm−2), 100 μg mL−1 (31 μg cm−2), 200 μg mL−1 (63 μg cm−2), and 400 μg mL−1 (125 μg cm−2) PM2.5 extracts for 6 and 24 h, respectively. Results were expressed as the means ± SD of three independent experiments. *p < 0.05 and **p < 0.01 compared with untreated cells. PM: particulate matter; SD: standard deviation.
PM2.5 induced cytokines secretion
Results regarding the levels of IL-6, IL-8, and TNFα were exhibited in Figure 2. We found that at 24 h after treatment, IL-6 and IL-8 levels were raised in the PM2.5-treated groups of 25, 50, and 100 μg mL−1 as well as the 100 μg mL−1 PM2.5-treated group, respectively, compared to the corresponding controls (p < 0.05 or p < 0.01). However, PM2.5 failed to induce TNFα production.

The levels of (a) IL-6, (b) IL-8, and (c) TNFα in PM2.5-treated BEAS-2B cells. The cells were treated with 6 μg mL−1 (1 μg cm−2), 13 μg mL−1 (3 μg cm−2), 25 μg mL−1 (5 μg cm−2), 50 μg mL−1 (10 μg cm−2), and 100 μg mL−1 (21 μg cm−2) PM2.5 extract for 6 and 24 h, respectively. Expression of IL-6, IL-8, and TNFα was measured by ELISA assay. Data were expressed as the means ± SD of three independent experiments. *p < 0.05 and **p < 0.01 compared with the untreated cells. IL: interleukin; TNFα: tumor necrosis factor α; ELISA: enzyme-linked immunosorbent assay; PM: particulate matter; SD: standard deviation.
PM2.5 activated IKK/NF-κB pathway
To determine the mechanism by which PM2.5 triggered cytokine secretions in BEAS-2B cells, we measured the levels of the proteins involved in IKK/NF-κB pathway. As shown in Figure 3(a), 100 μg mL−1 PM2.5 extract upregulated the protein levels of p-IKK, p-IκBα, p-p65, and p65 but downregulated IκBα protein level at 6 h after treatment (all p < 0.05). However, no change was found in the expression of IKKα and IKKβ proteins. As shown in Figure 3(a), at 6 h after treatment, 100 μg mL−1 PM2.5 extract downregulated the expression of p-IKK, p-IκBα, p-p65, and p65 proteins but upregulated the expression of IκBα protein (p < 0.05 or p < 0.01) in BEAS-2B cells pretreated with 10 μM Bay11-7082 (an IKK inhibitor) for 1 h. As shown in Figure 3(b), 100 μg mL−1 PM2.5 extract had no effect on the protein levels of p-IKK, IKKα, IKKβ, p-IκBα, p-p65, and p65 at 24 h time point. However, 100 μg mL−1 PM2.5 extract still led to degradation in expression of IκBα protein at 24 h after treatment (p < 0.01). As shown in Figure 4(a) and (b), only 100 μg mL−1 PM2.5 extract upregulated nuclear p65 at 6 and 24 h in BEAS-2B cells pretreated with 10 or 2.5 μM Bay11-7082 (p < 0.05 or p < 0.01), which may be attenuated by Bay11-7082 at 6 h (10 μM Bay11-7082) but not 24 h (2.5 μM Bay11-7082) after treatment.
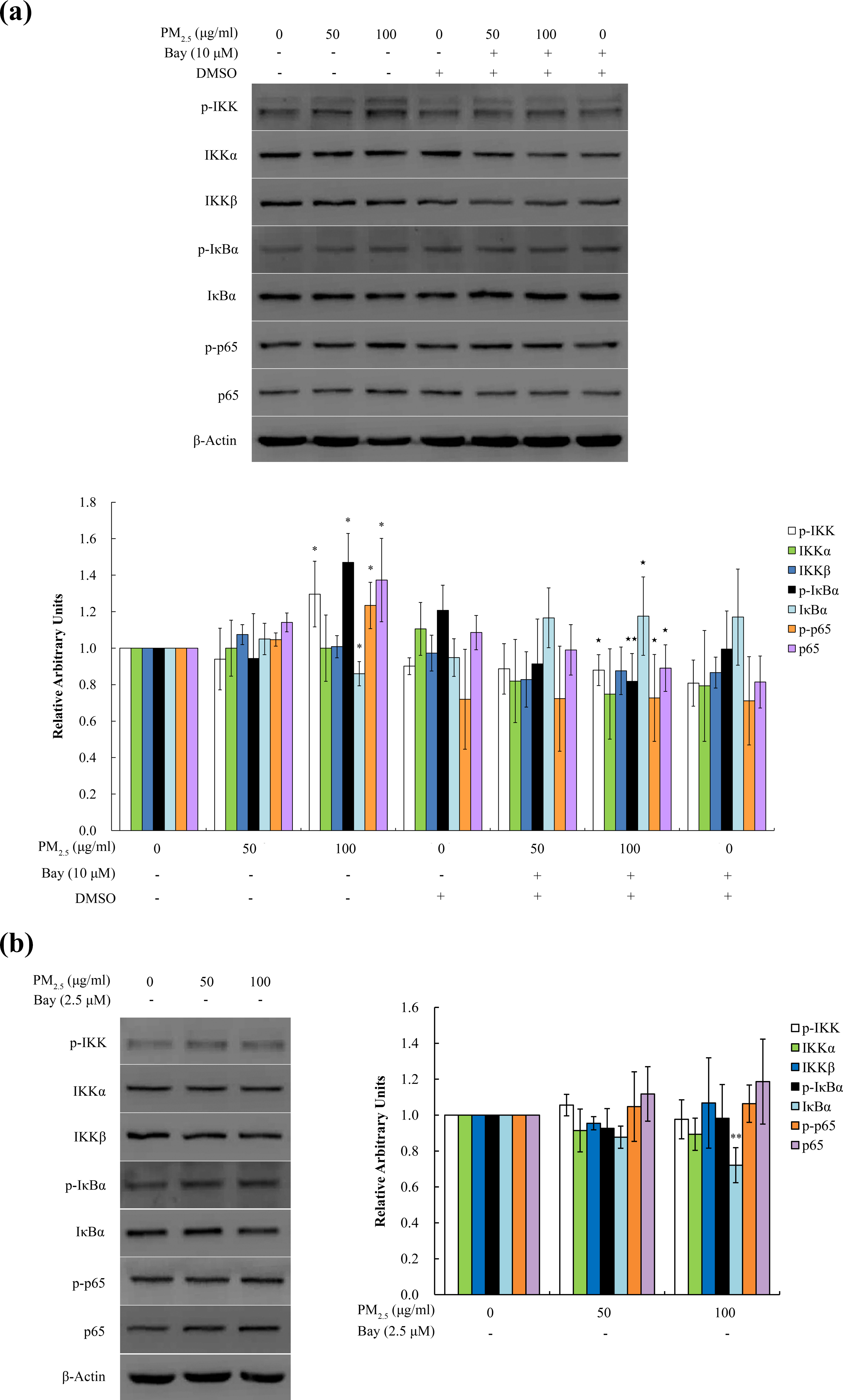
PM2.5 activated IKK/NF-κB signaling pathway in BEAS-2B cells. The cells were treated with 50 μg mL−1 (10 μg cm−2) and 100 µg mL−1 (21 μg cm−2) PM2.5 extract with or without IKK inhibitor (Bay11-7082) for 6 h (a) and 24 h (b). Whole cell lysates were extracted from BEAS-2B cells and the protein levels of p-IKKα/β, IKKα, IKKβ, p-IκBα, IκBα, p-p65, and p65 were measured by Western blot. β-Actin was used as a loading control. One representative blot for β-Actin was shown. The experiments were performed at least three times. The intensity of the bands was quantified using Gene Tool software, and the values were normalized to those of untreated cells. *p < 0.05 and **p < 0.01 compared with untreated cells. ⋆p < 0.05 and ⋆⋆p < 0.01 compared with PM2.5-treated cells. PM: particulate matter; IKK: IκB kinase; NF-κB: nuclear factor kappa B.

Nuclear translocation of NF-κB p65 in PM2.5-treated BEAS-2B cells. The cells were treated with 50 μg mL−1 (9 μg cm−2) and 100 µg mL−1 (18 μg cm−2) PM2.5 extracts with or without IKK inhibitor (Bay11-7082) for 6 (a) and 24 h (b), respectively. Expression of nuclear p65 protein was measured by Western blot. Lamin A was used as a loading control. The intensity of the bands was quantified using Gene Tool software. The experiments were performed in triplicates. *p < 0.05 and **p < 0.01 compared with untreated cells. ⋆p < 0.05 compared with PM2.5-treated cells. PM: particulate matter; IKK: IκB kinase; NF-κB: nuclear factor kappa B.
Cytokines production and the IKK/NF-κB pathway
We further investigated whether PM2.5 led to IL-6 and IL-8 productions by activating IKK/NF-κB signaling pathway. As shown in Figure 5, at 24 h after treatment, 100 μg mL−1 PM2.5 extract decreased IL-6 and IL-8 levels in BEAS-2B cells pretreated with 2.5 μM Bay11-7082 (p < 0.05).
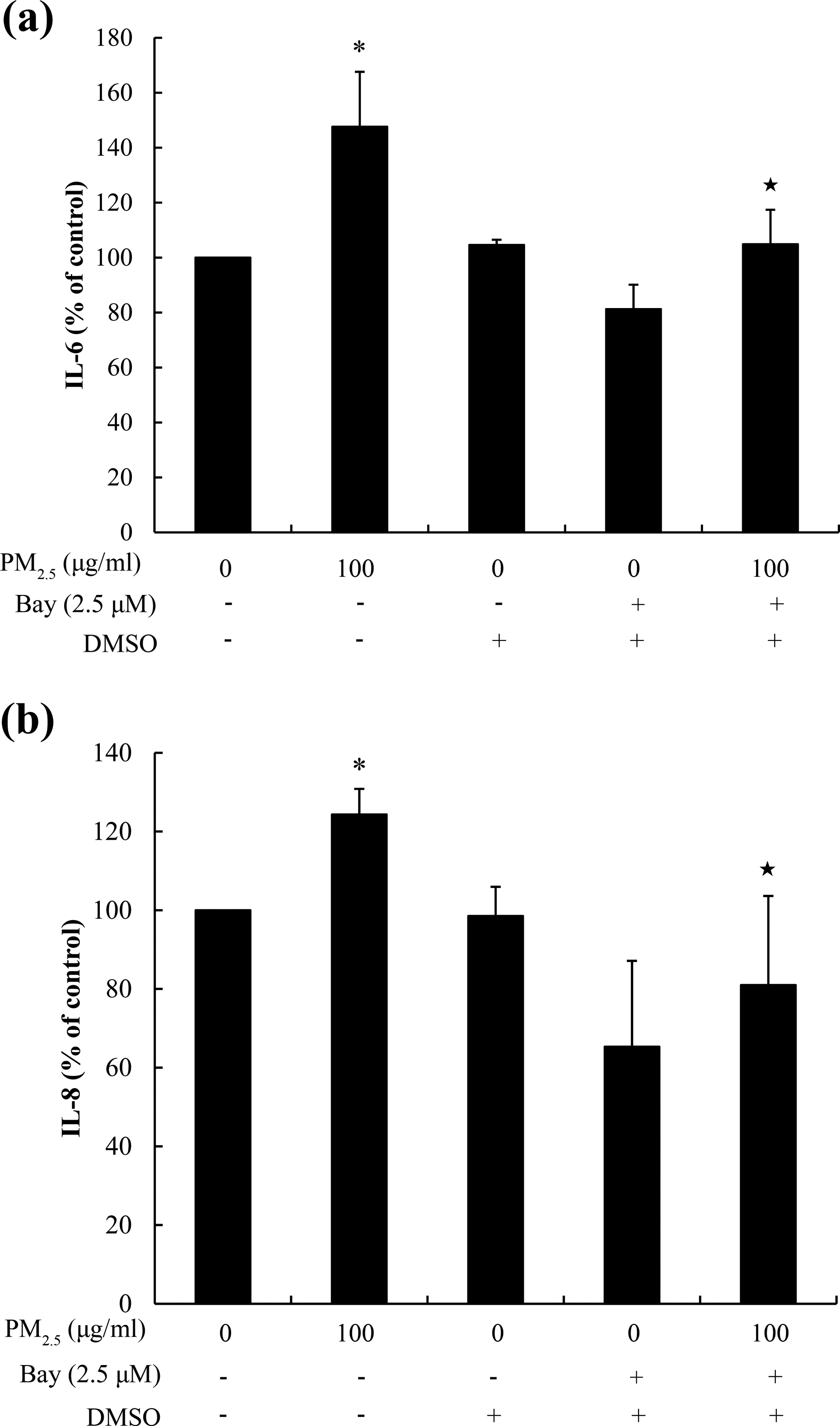
PM2.5 increased the IL-6 and IL-8 levels by the activation of IKK/NF-κB pathway in BEAS-2B cells. The cells were treated with the PM2.5 extract of 100 µg mL−1 (21 μg cm−2) for 24 h with or without Bay11–7082. The levels of IL-6 (a) and IL-8 (b) in the culture supernatants were determined by ELISA. Results were expressed as the means ± SD from three independent experiments. *p < 0.05 compared with untreated cells. ⋆p < 0.05 compared with PM2.5-treated cells. PM: particulate matter; IKK: IκB kinase; NF-κB: nuclear factor kappa B; ELISA: enzyme-linked immunosorbent assay; SD: standard deviation.
Weather forecast for Wuhan during the sampling days
Weather forecast data for Wuhan city during the sampling days are listed in Online Supplemental Table S1. The monitoring data display that during the period of collecting samples, daily mean values of atmospheric pressure, temperature and relative humidity, and daily precipitation are basically stable; however, the daily mean value of wind speed displays greatly fluctuation. Sample collection was stopped due to relative heavy rain on those 2 days (February 5 and 14, 2012).
Discussion
Epidemiological studies have revealed that PM2.5 was related to pulmonary diseases including asthma and chronic bronchitis. 36 These airway diseases were characterized regarding inflammation, remodeling, and hyperresponsiveness of the airway. 37 Several studies have shown that the secretion of inflammatory cytokines (such as IL-6 and IL-8) was induced in certain kinds of cells exposed to PM from various regions. 38 Previous studies indicated that PM2.5 elevated IL-1β, IL-6, IL-8, IL-10, and TNFα levels in airway epithelial cells. 39 –41 The expressional changes of cytokines have been accepted as markers for assessing inflammatory status in relation to exposure to PM based on the experimental in vivo and in vitro evidence. These cytokines participated in the pathogenesis of airway inflammation, for instance, IL-8 and TNFα recruited and activated neutrophils and IL-6 induced the B-lymphocytes differentiation. 42,43 In this study, increased IL-6 and IL-8 but without change in TNFα level implied that PM2.5 may promote the inflammatory reaction in BEAS-2B cells.
IKK/NF-κB pathway could be activated by several signaling pathways via IL-1 receptor, Toll-like receptor, and TNF receptor. 44 Activated IKK induced IκB degradation and NF-κB nuclear translocation. We found elevated level of phosphorylated IKK in 100 µg mL−1 PM2.5-treated cells at 6 h, indicating that IKK was activated by PM2.5 extract. We also found upregulations of p-IκBα, p-p65, and nuclear p65 proteins in the cells treated with 100 µg mL−1 PM2.5 for 6 h, indicating that NF-κB p65 signaling was activated by PM2.5. Moreover, the IKK inhibitor Bay11-7082 reversed PM2.5-induced upregulation of the proteins involved in IKK/NF-κB pathway and inhibited IκBα degradation and nuclear translocation of p65 in the cells treated with 100 µg mL−1 PM2.5 for 6 h. The results suggested that IKK regulated PM2.5-induced NF-κB p65 activation in BEAS-2B cells at 6 h after treatment. However, treatment with PM2.5 for 24 h failed to induce the phosphorylations of IKK, p65, and IκBα, but was still able to decrease IκBα and increase nuclear p65 translocation which could not be inhibited by Bay11-7082. IKK-independent signaling pathways could probably contribute to the activation of NF-κB. A study indicated that ultraviolet irradiation induced IκBα degradation, which triggered NF-κB activation. However, IκBα degradation depended on the phosphorylation at special serine residues in relation to the p38 pathway, but without the phosphorylation at Ser 32/36 by the IKK, 45 whereas the proteasome inhibitors PS-341 and MG-132 induced IκBα degradation in NCL-H157 cells by the lysosomal pathway. 46 Therefore, the observed degradation of IκBα at 24 h may be induced by the IKK-independent signaling pathways in this study.
Cytokine secretions could be regulated via multiple signaling pathways. For instance, MAPK and NF-κB signaling pathways participated in upregulation of IL-6 and IL-8 in response to stimulation with different factors. 47 Production of IL-6 and IL-8 was regulated by protein kinase C, calcium ionophore, 48 activator protein 1, and interferon regulatory factor 1. 49 The results regarding activated IKK and NF-κB at 6 h and elevated IL-6 and IL-8 secretions at 24 h suggested that IKK/NF-κB may mediate the expression of these cytokines in response to PM2.5. Furthermore, we observed that Bay11-7082 inhibited PM2.5-induced expression of IL-6 and IL-8 at 24 h, indicating that IKK/NF-κB was involved in IL-6 and IL-8 induction.
We need note limitations of this study. First, these cytokines as immune transmitters with multiple functions can transmit information in the signaling pathways. However, this study only referred to the mechanisms underlying the expressional changes of few cytokines (including IL-6, IL-8, and TNFα). Additionally, it is recognized that endotoxin (i.e. biological constituent of PM) contributed to inflammatory response in mouse macrophage RAW 264.7 cells exposed to PM from Taiwan, China, which are related to the activation of Toll-like receptor. Moreover, increased TNFα protein levels were inhibited by Polymyxin B, indicating that influence of endotoxin on TNFα expression. 50 Owing to the ambient PM2.5 samples were collected in the winter season of 2012, the detected secretions of IL-6 and IL-8 may be partially due to the contribution of the biological constituents of ambient PM2.5. Third, since we did not focus on the roles of the upstream regulators (such as Toll-like receptor, IL-1 receptor) of the IKK/NF-κB pathway in the secretion of IL-6 and IL-8 in BEAS-2B cells exposed to PM2.5, further studies need to understand the molecular mechanisms for PM2.5-induced inflammatory response, because combination of certain components of PM2.5 or cytokines with the receptors can stimuli the transmission of signals from the extracellular environment to intracellular adaptor and signaling molecules, resulting in the activation of the IKK/NF-κB. Therefore, the findings are limited to fully ascertain the mechanisms underlying local and systemic inflammation induced by PM2.5.
Conclusions
We observed that the PM2.5 extract induced IL-6 and IL-8 secretion in BEAS-2B cells through IKK/NF-κB signaling pathway. The findings contribute to understand the molecular mechanisms underlying PM2.5-induced inflammatory response in human bronchial epithelial cells. It provides significant clues for explaining the roles of inflammatory response in the pathological processes of respiratory diseases related to air pollution.
Supplemental material
Supplemental Material, Supplemented_Table_1_(04292018) - PM2.5 stimulated the release of cytokines from BEAS-2B cells through activation of IKK/NF-κB pathway
Supplemental Material, Supplemented_Table_1_(04292018) for PM2.5 stimulated the release of cytokines from BEAS-2B cells through activation of IKK/NF-κB pathway by J Wang, WJ Zhang, W Xiong, WH Lu, HY Zheng, X Zhou, and J Yuan in Human & Experimental Toxicology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant no 81472947) and the Commonweal Program of Ministry of Environmental Protection, China (no 201409081).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
