Abstract
Aristolochic acid (AA) and tetrandrine (TET) are the major bioactive components in Chinese herbs used for weight loss. The nephropathy caused by the 2 Chinese herbs has not been simultaneously investigated. The aim of this study was to examine the potential nephrotoxicity of AA and TET using Madin-Darby canine kidney (MDCK) cells and mice. The results showed that TET was more potent than AA in inhibiting MDCK cell growth via inducing apoptosis, as determined by annexin-V staining, 4’, 6’-diamino-2-phenylindole (DAPI) staining, DNA fragmentation, and caspase 3 activity. Mice treated with AA (10 mg/kg) by intraperitoneal administration for 3 months showed nephrotoxicity, elevated blood urea nitrogen, and increased renal tubular injuries. In contrast, mice treated with 50 mg/kg of TET in the same time period had moderate hydropic degeneration of the distal tubules in the kidneys. These results suggest that TET is more cytotoxic than AA in MDCK cells but shows less nephrotoxic than AA in mice.
Introduction
In the early 1990s, severe tubulointerstitial nephritis was reported to occur in Belgian women who had followed a particular slimming regimen that included the Chinese herbs,
Chemical structures of tetrandrine (TET) and aristolochic acid I and II.(AA I and AA II)
Aristolochic acid is a mixture of structurally related nitrophenanthrene carboxylic acids, mainly containing aristolochic acid I (AAI) and aristolochic acid II (AAII). 6 Herbal drugs derived from aristolochia plants have been used as medicines in obstetrics and in the treatment of snake bites. 7 The plant extracts have also been used for the treatment of arthritis, gout, rheumatism, and festering wounds. 8 Due to its anti-inflammatory properties, AA had been included in various drug formulations in Germany 9 until it was identified as a potential carcinogen in rodents by Mengs in the late 1980s. Acute AA intoxication results in acute renal failure, whereas chronic administration induces multisystemic tumors in rats. 10,11 In past decades, typical renal interstitial fibrosis and urothelial malignancy have been found in patients exposed to Aristolochia spp. containing AA in European 12 and Asian 13 countries and also in the United States, 14 indicating the etiologic role of AA in the genesis of the diseases. 15 Therefore, it has been proposed to term interstitial nephropathy, in which the unequivocal role of AA has been fully documented, as AA nephropathy (AAN). 16,17 These clinical features seen in patients with AAN include mild tubular proteinuria and metabolic acidosis. 16 Microscopic renal tissue examination of these patients reveals severe degeneration, necrosis and desquamation of renal tubular epithelial cells (RTECs), and edematous interstitium with interstitial fibrosis. 18,19
Tetrandrine belongs to a member of the bisbenzylisoquinoline alkaloids and has antirheumatic, anti-inflammatory, and antihypertensive activities. 20 Several studies have shown that TET inhibits the proliferation of HeLa cells, 21 HL-60 cells, and HepG2 cells (a human hepatoblastoma cell line), 22,23 and induces apoptosis of malignant cells. 24 TET can also suppress growth of ascites tumors in mice. 25 The pharmacological effects of TET have been studied extensively but its toxicity, especially chronic toxicity, is largely unknown. Tetrandrine exhibits hepatotoxicity when used for silicosis treatment in dogs. 26 It induces frameshift mutations and acts as a potent genotoxic enhancer. 27 Micronucleus and sister chromatid exchange (SCE) assays have revealed that TET is a weak genotoxic agent. 28 Tetrandrine ameliorates pulmonary hypoplasia and hypertension. 29 Furthermore, TET has positive effects on acute pancreatitis (AP) in human patients and animal models. 30
Different clinical effects have been observed between TET and AA. However, the nephrotoxicity of these 2 compounds has not been simultaneously studied. Therefore, the aim of this study was to compare the nephrotoxicity of TET and AA in vitro using a renal epithelial cell-derived cell line, Madin-Darby canine kidney cells (MDCK), and in vivo using mice.
Materials and Methods
AA and TET Compounds
The AA compound and TET were purchased from Sigma-Aldrich Company (St Louis, Missouri). The AA compound (AAI: 60%, AAII: 40%) was dissolved in saline (20 mmol/L). TET was dissolved in 0.1 N HCl at a stock concentration of 20 mmol/L. Both AA and TET solutions were passed through a 0.22 μm filter for bacteriologic sterilization. The pH of the final solutions was adjusted at 7.0. They were further diluted with culture medium to desired concentrations.
Cell Culture and MTT Assay
MDCK cells, obtained from American Type Culture Collection (CRL-6253), were cultured in Dulbecco modified Eagle’s medium (DMEM) supplemented with 10% heat-inactivated fetal bovine serum,
Morphological Assessment of Cell Death
Madin-Darby canine kidney cells were treated with AA or TET. After treatment, cells were washed with phosphate buffered saline (PBS), fixed in 2% paraformaldehyde for 30 minutes, and then permeabilized with 0.1% Triton X-100/PBS for 30 minutes at room temperature. Nuclei were stained by incubating the cells with 4’,6’-diamino-2-phenylindole ([DAPI] 1 μg/mL). Stained cells were examined under an Olympus fluorescence microscope.
DNA Fragmentation Analysis
Madin-Darby canine kidney cells seeded at a density of 1 × 106 cells/mL in 10-cm culture dishes were exposed to 10 μmol/L of AA or TET for 48 hours, and then the genomic DNA was prepared with a Wizard Genomic DNA Purification Kit (Promega, Madison, Wisconsin). DNA was precipitated with isopropanol, separated on 2% agarose gel and visualized by UV illumination after ethidium bromide staining.
Annexin-V Staining
Madin-Darby canine kidney cells were treated with 10 μmol/L of AA or TET for 6 and 24 hours. Apoptotic cells were then estimated by flow cytometry using an Annexin-V-fluoresceine isothiocyanate (FITC) Apoptosis Detection kit (Biovision, Palo Alto, California), according to the manufacturer’s protocol. Briefly, after AA or TET treatment, cells were harvested and centrifuged at 200
Caspase 3 Activity
Caspase-3 activity was measured with a Caspase 3 assay kit (Biovision, Mountain View, California). In brief, cultured cells were lysed in a buffer provided with the kit for 30 minutes on ice. Reaction mixtures containing acetyl-Asp-Met-Gln-Asp (Ac-DEVD)-p-nitroanilide (
AA or TET Preparation for Animal Treatment
Stock solution (10 mg/mL) of AA in saline was prepared by filtration through 0.22 μm filter units (Sartorius Biotech GmbH, Goettingen, Germany) prior to aliquoting and storage at −20°C. Tetrandrine was dissolved in 0.1 N HCl at a stock concentration of 10 mg/mL and passed through a 0.22 μm filter for bacteriologic sterilization. The pH of the final solutions was adjusted at 7.0. The AA or TET solution was diluted with normal saline to 1 mg/mL. C3H/HeN mice (8-week-old males; body weight 25-30 g) were used in this study. The research protocol was approved by the Animal Ethics Committee of Taichung Veterans General Hospital. The C3HeN mice were divided into 7 groups (6 mice for each experimental group; n = 6): group 1 was given normal saline by intraperitoneal administration daily (the total volume was 0.1 mL) for 3 months. Groups 2, 3, and 4 were intraperitoneally given AA (1, 10, and 50 mg/kg, respectively) daily for 5 days per week for 3 months. Groups 5, 6, and 7 were given TET (1, 10, and 50 mg/kg, respectively) by intraperitoneal administration daily for 5 d/week for 3 months.
Renal Function Parameters
At the end of the experiment, the mice (n = 6) were sacrificed, and blood was obtained by the cardiac puncture method under anesthesia for the measurement of serum creatinine and blood urea nitrogen (BUN).
Histological Pathology
Both left and right kidneys were evaluated for each animal (n = 6). The kidneys were removed and fixed in 10% buffered formalin and then embedded in paraffin. Each 5 μm section of paraffin-embedded kidney tissue was routinely processed for hematoxylin-eosin and picro-sirius red staining. Glomerulous and interstitial tubular injuries were evaluated using semiquantitative scores in a double-blind manner.
31
Kidney sections were observed under a light microscope at a magnification of ×400 using the Imaging Analysis System (Zeiss, HBO50) connected with a video camera and computer. All glomeruli and tubular lesions in each kidney section were graded in a double-blind manner according to the severity of the lesions as follows: 1 =
Apoptosis Determination by TUNEL Assay
Apoptotic cells were detected by the terminal deoxynucleotidyl transferase (TdT)-mediated dUTP-biotin nick end-labeling (TUNEL) assay, using an In Situ Apoptosis Detection kit (Roche Diagnostics GmbH, Roche Applied Science, Mannheim, Germany), according to the manufacturer’s instructions. Briefly, tissue sections were first deparaffinized in xylene and rehydrated in graded alcohols. They were then pretreated with proteinase K for 15 minutes at room temperature. Endogenous peroxidase was blocked with 3% hydrogen peroxide methanol for 5 minutes at room temperature, followed by an antigen retrieval step, carried out by boiling the slides in 0.01 mol/L citrate buffer for 5 minutes. Tissue sections were incubated with a reaction buffer containing TdT, fluorescein-dUTP for 90 minutes at 37°C. The reaction was stopped by rinsing the sections with 0.01 mol/L PBS, and then the sections were incubated with peroxidase-conjugated anti-fluorescein antibody for 30 minutes at 37°C. The immunocomplex was visualized by exposing the sections to hydrogen peroxidase and 3’, 3’ diaminobenzidine. Finally, sections were counterstained with hematoxylin prior to being mounted for examination by light microscopy. A negative control was prepared by using PBS instead of TdT. As a positive control, normal kidney tissue sections were pretreated with deoxyribonuclease I (DNase I) at 80 units/mL for 10 minutes at room temperature before adding the TdT reactive solution. The numbers of TUNEL-positive cells were counted in at least 20 randomly selected microscopic fields at ×400 magnifications. The average number was calculated as the representative variable for each mouse.
Statistical Analysis
Data were presented as mean ± SD and analyzed by Kruskal-Wallis test and Mann-Whitney
Results
Proliferation Inhibitory Effects of TET and AA in MDCK Cells
To examine the inhibitory effects of TET and AA on the proliferation of MDCK cells, MDCK cells were treated with TET or AA (2.5-80 μmol/L) for 24 and 48 hours, and the cell number was measured by MTT assay. As shown in Figure 2A, both AA and TET inhibited proliferation of MDCK cells in a concentration-dependent manner. Tetrandrine was more potent than AA in inhibiting growth of MDCK cells. The IC50s of TET were estimated to be 10.1 ± 0.2 μmol/L and 7.4 ± 0.9 μmol/L at 24 and 48 hours, respectively. The IC50s of AA were 58.9 ± 0.4 μmol/L and 47.2 ± 0.5 μmol/L at 24 and 48 hours, respectively. However, TET exhibited less inhibitory effects on cell proliferation than AA in the presence of a 0.3% S9 mix in the medium of MDCK cells (Figure 2B and C). Assuming the growth rate of control cells (without treatment) as 100%, cells treated with AA exhibited 45% ± 1% in contrast to 85% ± 6% in cells treated with TET.
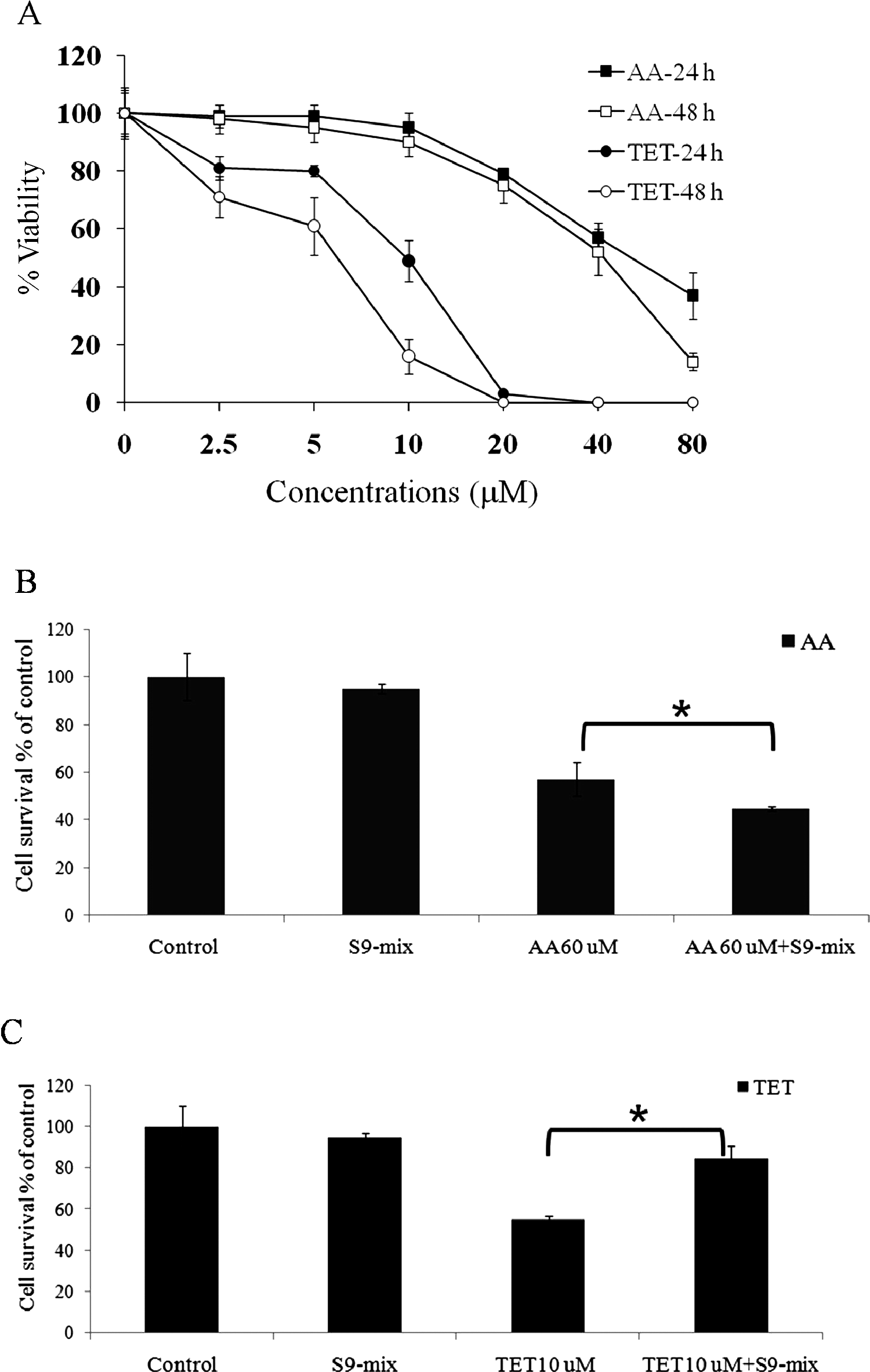
Antiproliferative effects of AA and TET in MDCK cells. A, Cell growth profiles of MDCK cells after incubation with various concentrations of AA and TET for 24 and 48 hours. Data are shown as mean ± SD of 3 independent experiments with triplicate wells. B, Viability of MDCK cells after incubation with 60 μmol/L of AA for 24 hours, as determined by 3-(4,5-dimethythiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) assay. Mean ± SD were obtained from 3 independent experiments. *Indicates a statistically significant difference (
Induction of MDCK Cell Apoptosis by TET
Microscopic analysis following DAPI staining of MDCK cells treated with TET revealed that cells exhibited morphological features of apoptosis. As shown in Figure 3A, MDCK cells treated with 10 μmol/L of TET for 12 hours exhibited moderately fluorescent condensed chromatin and cleaved nuclei. We next investigated the sub-G1 population of MDCK cells using flow cytometry. In the untreated group and group treated with 10 μmol/L of AA for 48 hours, the percentages of apoptotic cells were 4% and 16%, respectively. However, the apoptotic population increased dramatically to 54% and 85% after 24 and 48 hours of TET treatment, respectively (Figure 3B). Moreover, TET treatment resulted in the degradation of chromosomal DNA into small internucleosomal fragments, as evidenced by the formation of 180 to 200 bp DNA ladders on agarose gels (Figure 3C). Annexin V binding assay, as shown in Figure 3D, revealed that apoptosis was clearly detectable in cells with 10 μmol/L of TET for 6 hours (22.09%) and 24 hours (53.69%). In contrast, cells treated with 10 μmol/L AA for 6 and 24 hours exhibited moderate apoptosis rates of 5.13% and 6.82%, respectively. Furthermore, caspase 3 activity analysis showed that treatment with TET, but not AA, caused a significant increase in caspase 3 activity at 24 hours (Figure 3E;
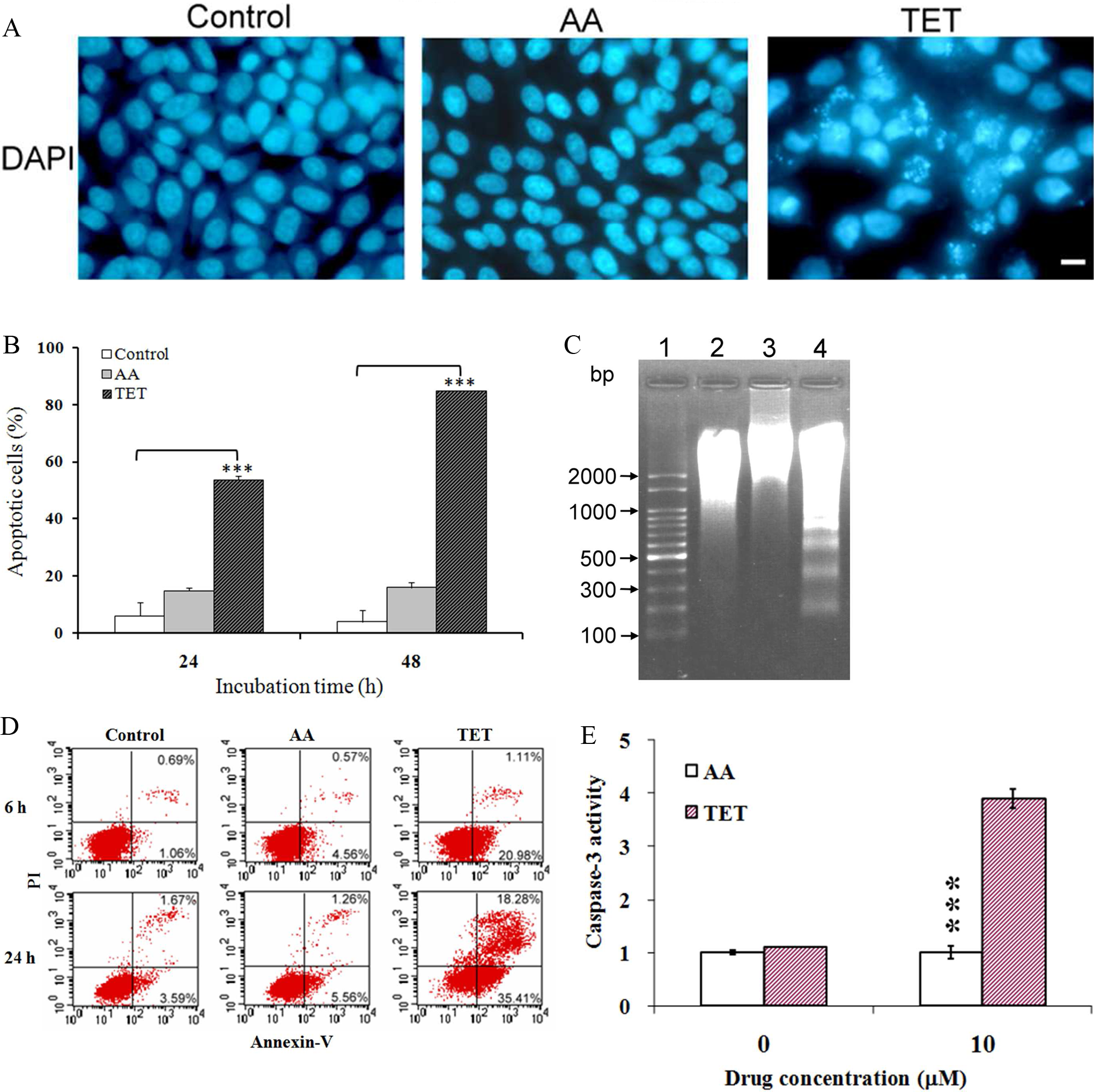
Tetrandrine (TET)-induced apoptosis in MDCK cells. A, Cells were grown in culture medium with or without 10 μmol/L of AA or TET for 12 hours, stained with 1 μg/mL of DAPI, examined with fluorescence microscopy and photographed at ×200 magnification; scale bar, 5 μm. B, Increase of sub-G1 population after TET treatment. The percentage of apoptotic cells was assessed by flowcytometry using an Annexin-V-fluoresceine isothiocyanate Apoptosis Detection kit after incubation of cells with 10 μmol/L of AA or TET for 48 hours. The data represent mean ± SD of 3 different determinations. ***Significantly different from control (
Survival of AA- and TET-Treated Mice
In order to compare the toxicity of AA and TET in vivo, mice were intraperitoneally injected with 3 doses (1, 10, and 50 mg/kg) of AA or TET daily for 5 d/week and the control group was injected with normal saline daily for 5 d/week. The survival of each group was monitored for 3 months. As indicated in Figure 4 , after intraperitoneal injection of 1 and 10 mg/kg of TET, 100% survival was observed. Fifty percent of the mice (n = 3 of 6) treated with 50 mg/kg of TET were dead on days 16, 17, and 31 after treatment. However, all mice in the groups treated with 10 and 50 mg/kg of AA died within a few days (day 1-day 9) due to its acute renal failure.
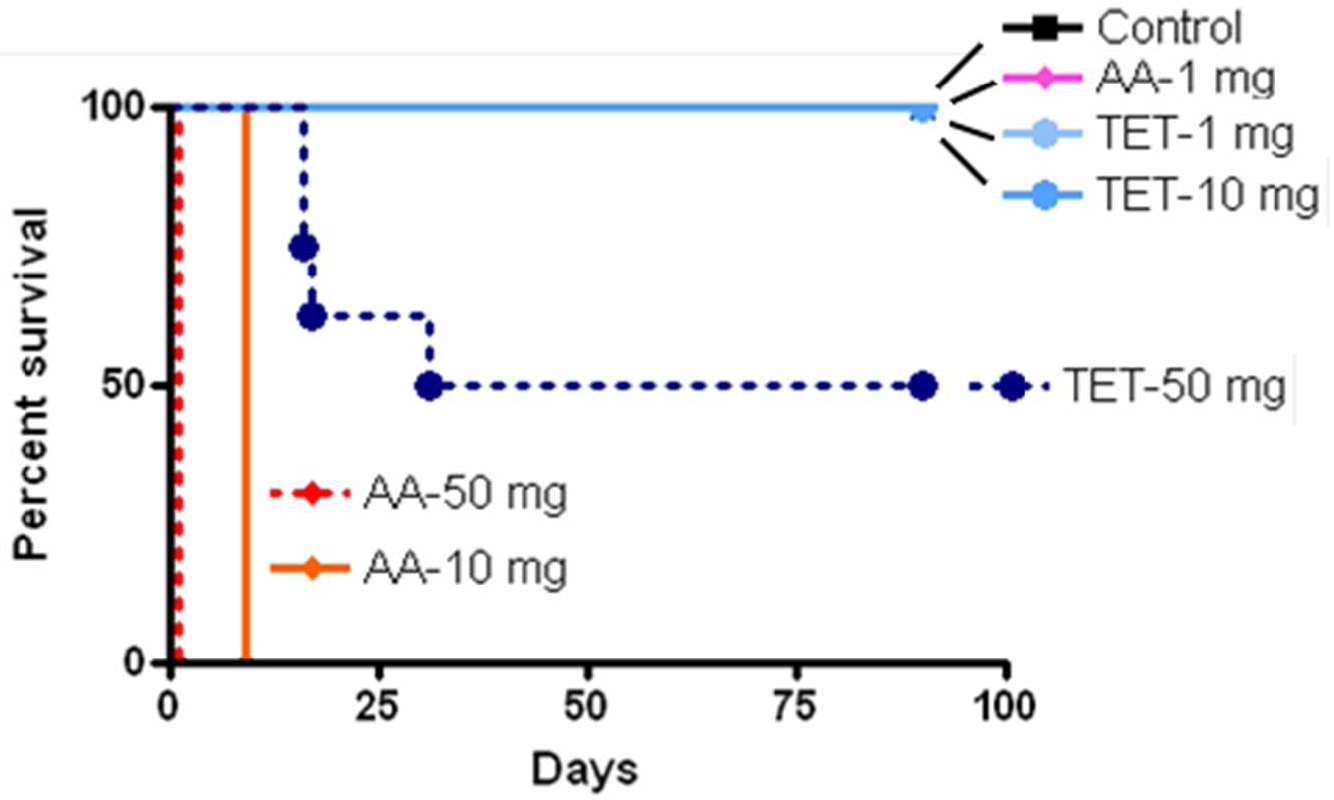
Effects of AA and TET on animal survival. The survival of the control, AA- and TET-treated mice was determined by Kaplan-Meier analysis. C3H/HeN mice were injected with various doses of AA and TET (n = 6) and the survival of treated animals was recorded. TET indicates tetrandrine; AA, aristolochic acid.
Comparison of TET- and AA-Induced Renal Toxicity
Body weight
The effects of TET and AA by intraperitoneal injection on the body weights of mice were analyzed. As shown in Figure 5 , mice treated with 1 mg/kg of AA and 50 mg/kg of TET had a decreased or lower weight gain during the study and weighed less than vehicle-treated mice at the end of the study. There was no significant difference in the body weights between animals treated with 1 and 10 mg/kg TET and control animals.
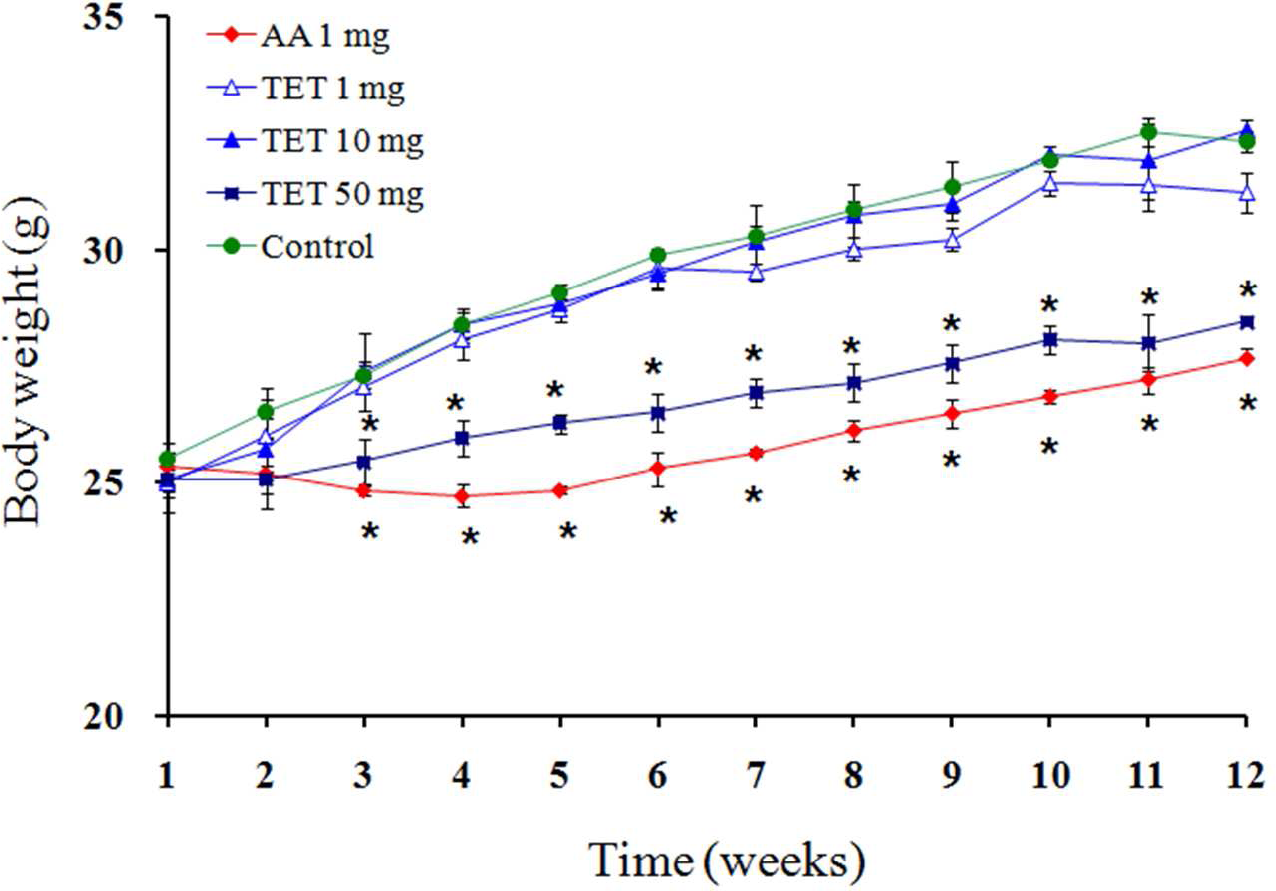
Effects of AA and TET on body weights of mice. Mice received an intraperitoneal injection of 1 mg/kg AA or 1, 10, and 50 mg/kg TET daily for 5 d/week for 3 months. *Significantly different from the control group (
Creatinine and BUN
As indicated in Table 1 , the creatinine and BUN levels of the groups treated with 1 mg/kg and 10 mg/kg of AA were significantly higher than those of the control groups. The creatinine and BUN levels in the groups treated with 10 mg/kg of AA were markedly higher than in the groups treated with 1 mg/kg of AA. There was no significant difference in the creatinine and BUN levels between the TET-treated group and the control.
Biochemical Parameters of Mice After Treatment With TET and AA for 3 months a

Abbreviations: TET, tetrandrine; AA, aristolochic acid; BUN, blood urea nitrogen.
a Each group received TET-1,-10, and -50 (1, 10, and 50 mg/kg daily for 5 d/week), AA-1 and -10 (1 and 10 mg/kg daily for 5 d/week the same dose), or sterile saline (control) intraperitoneally (ip) for 3 months (n = 6).
b and cSignificantly different from the control group (
Kidney histopathology
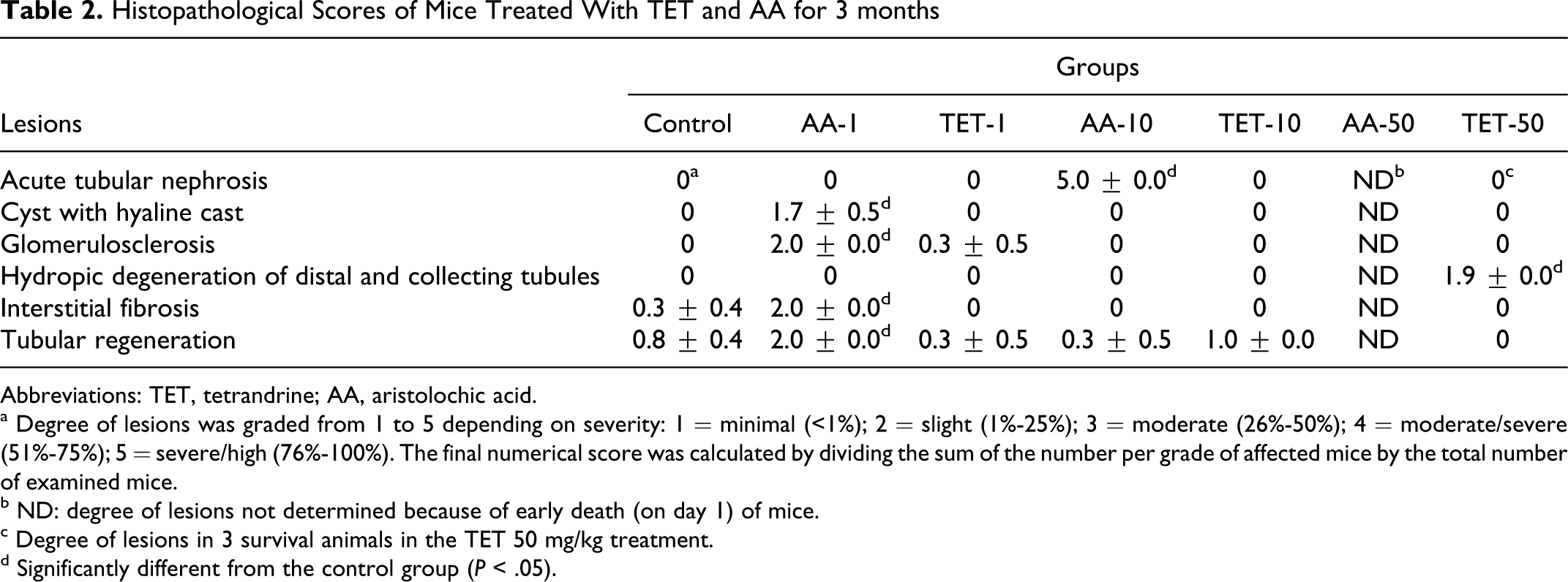
The histology of the kidneys from control mice and from mice treated with AA and TET was examined. As shown in Figure. 6A, B, C, and D, the control groups with normal glomeruli, proximal, distal, and collecting tubules of kidneys. In mice treated with 1 mg/kg AA for 3 months, glomeruli of kidneys showed focal minimal to slight glomerulosclerosis with interstitial fibrosis (Figure 6E, F), and no significant lesion was found in the distal and collecting tubules (Figure 6G, H). Furthermore, no effect was found in glomeruli (Figure 6I), but numerous hyaline casts accumulated in the lumens of the proximal, distal, and collecting tubules in the 10 mg/kg AA-treated mice (Figure 6J-L). In the TET treatment groups, normal architecture of glomeruli and the proximal tubules in the kidneys were observed in the 1 mg/kg and 10 mg/kg TET treatments (Figure 7A-H). Mice treated with 50 mg/kg TET exhibited lesions of acute swelling and hydropic degeneration in the capsular epithelium of glomeruli and renal tubules of the kidney (Figure 7I-L). The histopathological scores on each lesion were graded as shown in Table 2
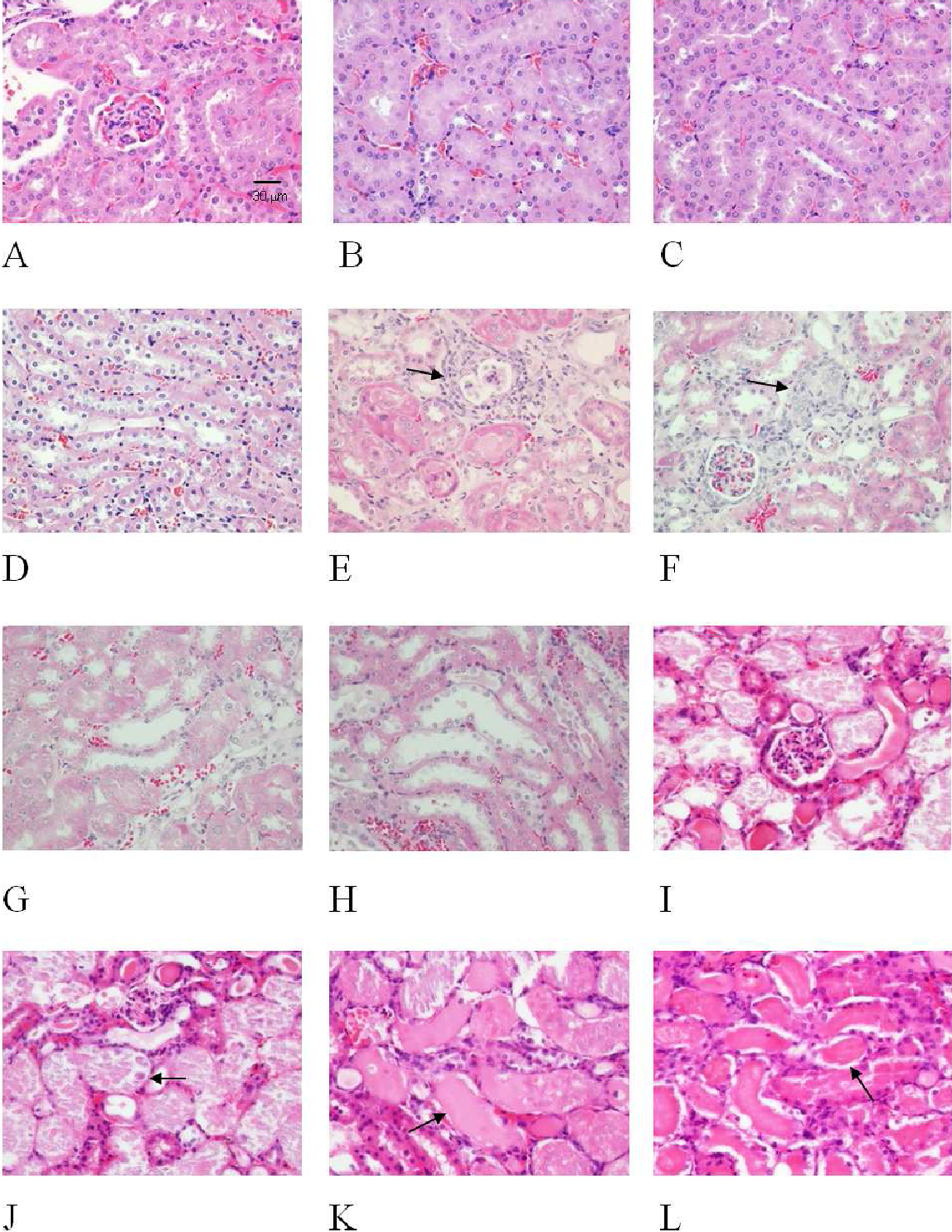
Histological alterations of kidneys in AA-treated mice. Mice were given an intraperitoneal injection daily with 1 and 10 mg/kg of AA for 5 d/week for 3 months. Normal architectures of the glomeruli, proximal, distal, and collecting tubules in the control mice (A-D). The kidneys from AA (1 mg/kg)-treated mice showed focal glomerulosclerosis (E) with interstitial fibrosis (F), no significant lesion was noted in the distal (G) and collecting tubules (H). Mice died early after treatment with 10 mg/kg AA and showed no significant change of the glomeruli in the affected kidneys (I) but had severe tubular necrosis with hyaline casts in the lumens of proximal (J), distal (K), and collecting tubules (L) in the affected kidneys. The arrows indicate the location of the lesion in the tissue as indicated (E, F, J, K, L). (H&E stain, all the photos are printed the same, with the same magnification: ×400, scale bar = 30 μm). AA indicates aristolochic acid; H&E, hematoxylin-eosin.
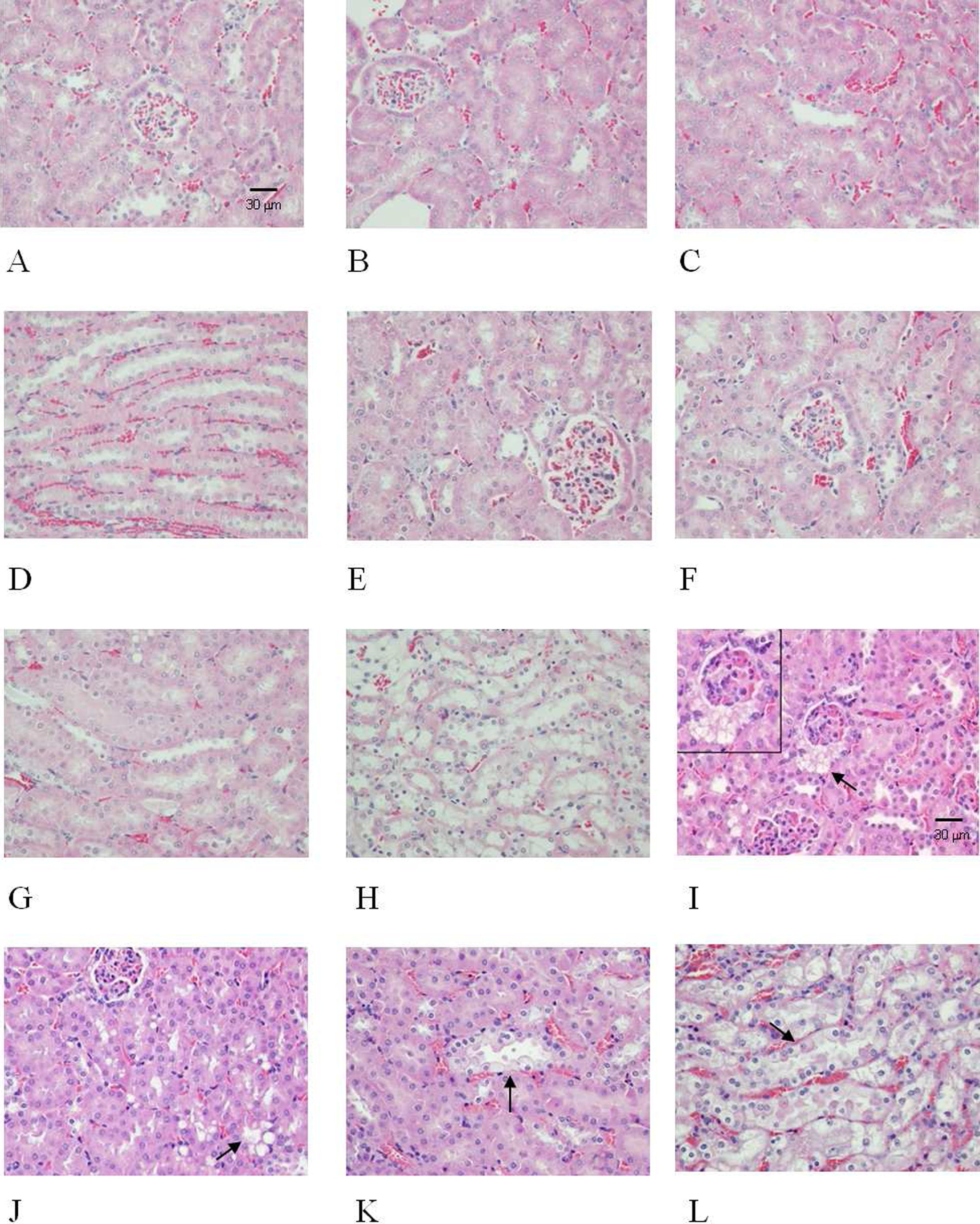
Histological alterations of the kidneys in TET-treated mice. Mice were given an intraperitoneal injection daily with 1, 10, and 50 mg/kg of TET for 5 d/ week for 3 months. No effects on the glomeruli, proximal, distal, and collecting tubules in the kidneys were found after treatment of mice with 1 (A-D) and 10 mg/kg TET (E-H). However, mice treated with 50 mg/kg of TET exhibited hydropic degeneration around the capsular epithelium of the glomeruli (I), and the tubular cells of proximal (J), distal (K), and collecting (L) tubules in the kidneys of the treated animals The arrow indicates the location of the lesion in the tissue (I, J, K, L). The inset in (I) is a picture with higher magnification of the lesion as indicated by an arrow in (I). (H&E stain, all the photos are printed the same, with the same magnification: ×400, scale bar = 30 μm). TET indicates tetrandrine; H&E, hematoxylin-eosin.
Histopathological Scores of Mice Treated With TET and AA for 3 months
Abbreviations: TET, tetrandrine; AA, aristolochic acid.
a Degree of lesions was graded from 1 to 5 depending on severity: 1 = minimal (<1%); 2 = slight (1%-25%); 3 = moderate (26%-50%); 4 = moderate/severe (51%-75%); 5 = severe/high (76%-100%). The final numerical score was calculated by dividing the sum of the number per grade of affected mice by the total number of examined mice.
b ND: degree of lesions not determined because of early death (on day 1) of mice.
c Degree of lesions in 3 survival animals in the TET 50 mg/kg treatment.
d Significantly different from the control group (
TUNEL assay for cell apoptosis and proliferation
TUNEL assay was performed in the paraffin section to investigate whether cell apoptosis was involved in the tubulointerstitial injury caused by AA or TET. As shown in Figure 8A, mice treated with 1 and 10 mg/kg AA exhibited significant cell apoptosis in the tissue examined, whereas mice treated with TET (1 and 10 mg/kg) had very low levels of apoptotic cells in the same tissue. Quantitative analysis (Figure 8B) of TUNEL-positive tubular cells revealed that the groups treated with 1 and 10 mg/kg AA had 12% and 64.5% apoptotic cells, respectively. In contrast, the groups treated with 1 and 10 mg/kg of TET had less than 1% apoptotic cells.
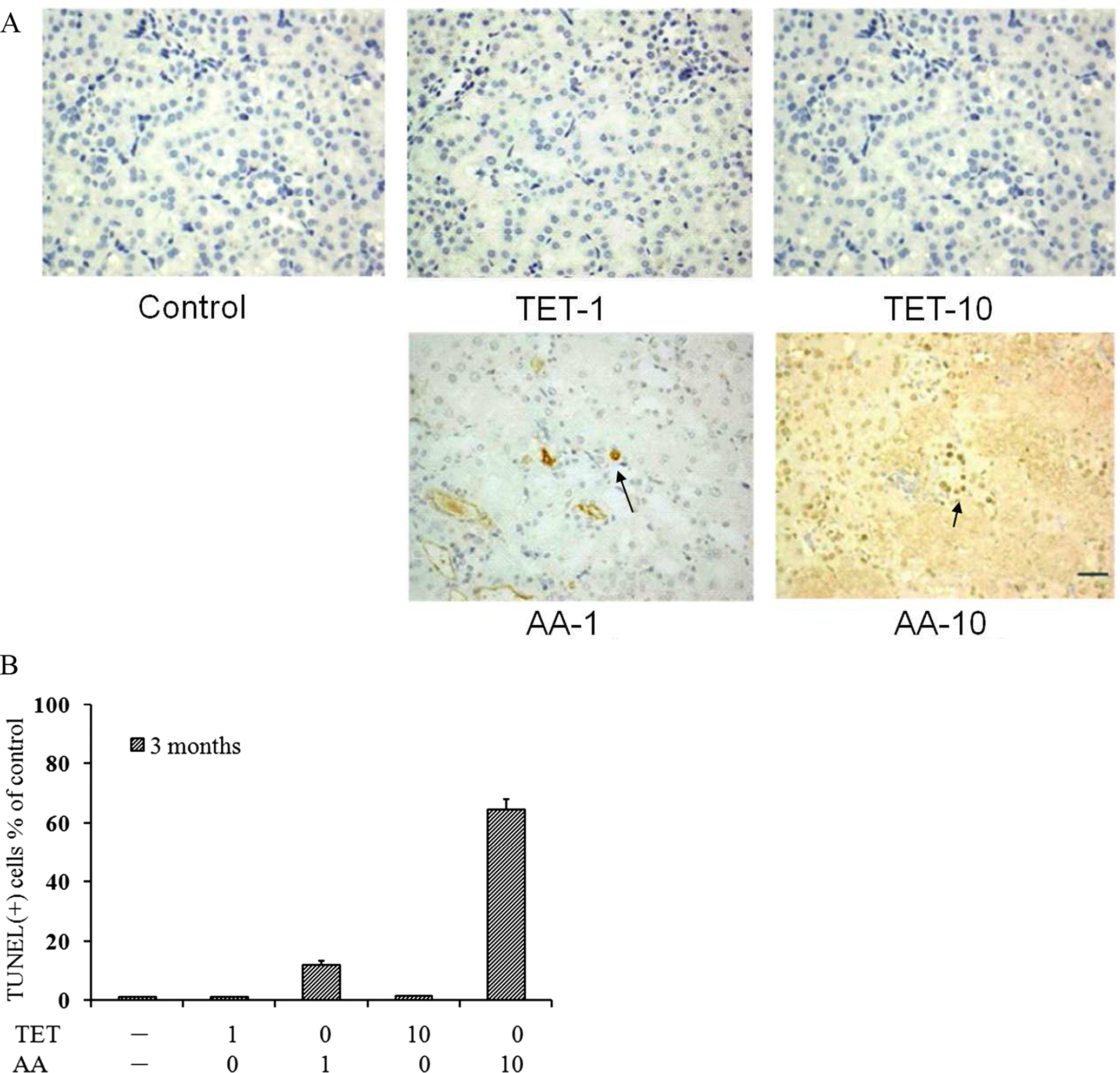
AA-induced apoptotic cell death in the kidneys of mice. A, All mice received an intraperitoneal injection daily of 1 and 10 mg/kg of AA or TET for 5 d/week for 3 months (N = 6), except that mice injected with 10 mg/kg of AA died on day 9 due to acute toxicity. TUNEL assay was performed in the paraffin sections and counterstained with hematoxylin to visualize nuclei. Representative images are shown: original magnifications ×400, scale bar, 30 μm. The arrow indicates the apoptotic cell. B, The number of TUNEL-positive tubular cells was counted (in a double-blind manner) in at least 20 randomly selected microscopic fields at ×400 magnification. The data represent mean ± SD from the measurement of randomly selected lesions. AA indicates aristolochic acid; SD, standard deviation.
Discussion
In the present study, we attempted to compare the renal toxicity of TET and AA. Our study has shown that both compounds exhibit cytotoxicity toward distal renal tubular MDCK cells. A 10 μmol/L IC50 dose of TET dramatically induced cell apoptosis in the distal renal tubules of the MDCK cells. Both compounds at high doses induced toxicity by virtue of different tissue locations and different doses. For example, C3H/HeN mice were injected with 10 mg/kg of AA 5 days a week for 9 days, causing acute necrosis in the proximal tubules with interstitial fibrosis, whereas TET (50 mg/kg) only caused hydropic degeneration in the renal tubular (mainly in the distal and collecting tubules) and around the capsular epithelium of the glomeruli. The dosage of TET required for inducing acute toxicity was 50 times greater than that of AA.
Studies have shown that AA induces apoptosis of renal proximal tubular cells (LLC-PK1 cells), 32,33 HK-2 cells, 34 and distal tubular cells (MDCK cells). 35 –38 However, these studies were carried out using either a mixture of AAs 34 or AAI alone. 33, 34 The IC50 of the AA mixture (AAI and AAII) found in our studies is approximately 60 μmol/L which is comparable to those found in other studies. 32,35 Aristolochic Acid I has been found to be toxic in MDCK cells after incubation for 24 hours (IC50 ≈ 67 μmol/L) and AAII has shown lower levels of toxicity with an IC50 ≈ 127 μmol/L. 6 The IC50 of AAI obtained by these laboratories is similar to our results (IC50 ≈ 60 μmol/L). Our results, therefore, suggest that AAs at a concentration of 60 μmol/L are cytotoxic to cultured renal tubular cells (MDCK cells).
Recent research has also shown that AA appears to mainly damage the renal tubular epithelial cells (RTECs) and causes acute, subacute, and chronic renal tubulointerstitial injuries. 39 In 2005, Lebeau et al 40 confirmed that proximal tubular dysfunction can be detected as early as 2 days after AA administration in vivo, and a more pronounced intoxication may lead to severe tubular atrophy and interstitial fibrosis. Our results (Figure 6E-H, J-L) also showed focal, slight-to-moderate chronic cysts with hyaline casts, tubular regeneration, and glomerulosclerosis in the kidneys of mice treated with AA. There was also interstitial fibrosis when mice were given an intraperitoneal injection of 1 mg/kg of AA for 3 months, but AA at a dose of 10 mg/kg caused death and led to acute proximal tubular necrosis in the kidneys (Figure 6F, J). In contrast, treatments with 1 and 10 mg/kg of TET showed normal kidney histopathology (Figure 7A-H). We noticed that 50 mg/kg of TET only caused slight hydropic degeneration in the renal tubular and around the capsular epithelium of the glomeruli (Figure 7I-L); therefore, renal injury caused by both compounds might be related to their different metabolism and tissue distributions. The safety level of TET was 50 times greater than AA, as seen in these experiments.
In our study, TET showed more apoptotic cytotoxicity in MDCK cells than AA based on morphological alteration data, flow cytometric analysis, DNA fragmentation, and caspase 3 activity. To our knowledge, there are no reports about renal tubular cell treatment with TET. However, a recent report demonstrated that AA evokes a rapid rise in the intracellular Ca2+ concentration of renal tubular cells through the release of intracellular endoplasmic reticulum stress and mitochondrial stress, resulting in activation of caspases and finally apoptosis. 35 These results suggest that the Ca2+ channel and mitochondrial membrane potential balance may be involved in the injury to MDCK cells after exposure to AA and TET, respectively.
Previous reports have identified TET as a nonselective Ca2+ channel blocker, 20 and as an antioxidant and anti-inflammatory agent. 41 Tetrandrine has also been shown to exhibit other various pharmacological effects including antitumor activities. 22 Tetrandrine is known to play a broad therapeutic role. For example, 20 mg/kg of TET was given once a day by gavage to BB rats. These rats exhibited reduced incidence of spontaneous development of diabetes mellitus. The drug (TET) toxicity was not seen in these rats. 42 Furthermore, Lewis rats were treated with TET by intraperitoneal injection on alternate days for 30 days. The incidence of relapsing experimental allergic encephalitis (EAE) was reduced by 41% at a nontoxic dose of 60 mg/kg of TET in these animals. 43 However, it was noted that the administration of TET at a high dose (40 mg/kg) for 2 months may induce focal necrosis of liver cells, abnormal liver function, and acceleration of erythrocyte sedimentation rate. After the continuous administration of TET, an accumulation of the drug in the liver, lungs, kidneys, and adrenal glands was observed. 26 Moreover, a study of beagles and rhesus monkeys given doses of TET ranging from 10 to 150 mg/kg intravenously resulted in severe local tissue reaction, hepatotoxicity, and lymphoid tissue necrosis. Nephrotoxicity occurred in monkeys that received TET at a dose of 100 mg/kg per d for 5 days (BUN = 100 mg/dL). 44 Our results also demonstrate hydropic degeneration in the capsular epithelium of the glomeruli and renal tubular in mice received 50 mg/kg of TET for 3 months. In general, previous reports have suggested that TET regulation of cellular redox states may play a major role in its dual action of cytotoxicity and cytoprotection. 45
Aristolochic acid is a nephrotoxic and carcinogenic compound, 10,11 which has been demonstrated to be genotoxic and mutagenic both in vitro and in vivo. 46 The molecular mechanisms of AA-mediated renal injury in mice were recently identified by Xiao et al. 47 Their research suggests that AAI metabolic enzymes containing hepatic and renal cytosolic NAD(P)H:quinone oxidoreductase (NQO1), hepatic microsomal cytochrome P450(CYP)1A1/2, and renal microsomal NADPH:CYP reductase (CPR) play an important role in detoxification of AA. Xiao et al 47 also reported that the hepatic P450s play a critical role in the detoxification of AAI in vivo. The renal CYP1A activities and levels were much lower in the liver of mice, indicating that the renal P450s may play a less important role in metabolizing AA in mice. Therefore, S9-mix obtained from the liver, pooled from male Sprague-Dawley rats, which contained sulfotransferase and cytochrome P450 enzymes, was used in this study. The results, as shown in Figure 2B and C, indicate that TET shows less cytotoxicity than AA in the presence of S9-mix in vitro. We reason that MDCK cells treated with 10 μmol/L of TET exhibit reduced cytotoxicity, likely via detoxification by S9-mix. MDCK cells treated with 60 μmol/L of AA in the presence of S9-mix exhibit enhanced cytotoxicity. This may cause more toxicity via the S-9 mix metabolic enzyme actions. We speculate that both compounds affect the growth and apoptosis of MDCK cells, depending on the presence and absence of metabolic enzyme actions, but this hypothesis still warrants further investigation.
In summary, our findings support the evidence that AA induces more severe nephrotoxicity than TET in mice, but TET is obviously more toxic to MDCK renal tubule cells than AA in vitro. These 2 compounds exhibit different effects in cultured MDCK cells and mice. These may be due to the different pharmacological properties (eg, different interactions of TET and AA with plasma proteins) and different metabolic mechanisms. Therefore, the molecular mechanisms of AA- and TET-mediated renal injury and cell damage still warrant further investigation.
Footnotes
Acknowledgments
We would like to thank Taichung Veterans General Hospital (TCVGH-955005C) for funding this project, Dr Yung-Tsung Chiu for help with histological staining and the Biostatistics Task Force of Taichung Veterans General Hospital for statistical analysis.
The author(s) declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
