Abstract
The effect of the natural product diindolylmethane (DIM) on cytosolic Ca2+ concentrations ([Ca2+]i) and viability in MDCK renal tubular cells was explored. The Ca2+-sensitive fluorescent dye fura-2 was applied to measure [Ca2+]i. DIM at concentrations 1–50 μM induced a [Ca2+]i rise in a concentration-dependent manner. The response was reduced partly by removing Ca2+. DIM induced Mn2+ influx leading to quenching of fura-2 fluorescence. DIM-evoked Ca2+ entry was suppressed by nifedipine, econazole, SK&F96365 and protein kinase C modulators. In the absence of extracellular Ca2+, incubation with the endoplasmic reticulum Ca2+ pump inhibitor thapsigargin (TG) or 2,5-di-tert-butylhydroquinone (BHQ) greatly inhibited DIM-induced [Ca2+]i rise. Incubation with DIM abolished TG or BHQ-induced [Ca2+]i rise. Inhibition of phospholipase C with U73122 reduced DIM-induced [Ca2+]i rise by 50%. At 1, 10, 40 and 50 μM, DIM slightly enhanced cell proliferation. The effect of 50 μM DIM was reversed by chelating cytosolic Ca2+ with 1,2-bis(2-aminophenoxy)ethane-N,N,N',N'-tetraacetic acid. In sum, in MDCK cells, DIM induced a [Ca2+]i rise by evoking phospholipase C-dependent Ca2+ release from the endoplasmic reticulum and Ca2+ entry via protein kinase C-sensitive store-operated Ca2+ channels. DIM did not induce cell death.
Keywords
Introduction
Emerging evidence provides credible support in favor of the potential role of bioactive products derived from ingesting cruciferous vegetables such as broccoli, brussel sprouts, cauliflower and cabbage. 1,2 Among many compounds, 3,3'-diindolylmethane (DIM) is generated in the acidic environment of the stomach following dimerization of indole-3-carbinol (I3C) monomers present in these classes of vegetables. Extended treatment with physiological concentrations of DIM results in gene expression alteration, growth inhibition and apoptosis of cancer cells in vitro. 3,4 Both I3C and DIM have been investigated for their use in preventing, inhibiting and reversing the progression of cancer as a chemopreventive agent. DIM appears to be a promising chemopreventive agent or chemo-radio-sensitizer for the prevention of tumor recurrence and/or for the treatment of human malignancies. Most of the inhibitory effect of DIM is shown in cancer cells such as prostate cancer cells, 5,6 breast cancer cells, 7 osteosarcoma cells, 8 hepatoma cells 9 and pancreatic cancer cells. 10 The effect of DIM on noncancer cells is less explored. Additionally, Roh et al. 11 showed that DIM induced immunotoxicity via splenocyte apoptosis in neonatal mice. DIM was shown to inhibit breast cancer cell growth via miR-21-mediated Cdc25A degradation 12 and to activate Nrf2 and induce Nrf2 target gene expression in murine fibroblasts. 13 A clinical study in healthy adults showed that a single 300 mg dose of DIM resulted in a mean maximum plasma concentration (C max) of 108 ng/ml (∼0.44 μM) after 1 h. 14 The effect of DIM on renal tubular cells remains unclear.
Ca2+ ions play a crucial role in different biological responses. A rise in intracellular free Ca2+ concentrations ([Ca2+]i) can initiate many pathophysiological cellular processes. 15 However, a unregulated [Ca2+]i rise may cause ion flux, dysfunction of proteins, apoptosis, proliferation and so on. 16 DIM has been shown to induce [Ca2+]i rise and death in cancer cells such as hepatoma cells 9 and osteosarcoma cells. 8 Furthermore, DIM has also been shown to prevent prostate cancer development in humans. 6 Whether DIM could increase [Ca2+]i and evoke death in noncancer cells is unknown. Thus, we investigated the effect of DIM on [Ca2+]i and viability in noncancer cells derived from renal tubular cells (MDCK). The MDCK cell is a useful model for renal research. It has been shown in this cell that [Ca2+]i can increase in response to the stimulation of various ligands such as melittin, 17 anandamide, 18 celecoxib 19 and endogenous ligands such as adenosine triphosphate (ATP) 20 and bradykinin, 21 via causing Ca2+ entry and Ca2+ release.
Fura-2 was used as a fluorescent Ca2+-sensitive dye to measure [Ca2+]i changes in the present study. Both DIM-induced Ca2+ entry and Ca2+ release in MDCK cells were explored. The [Ca2+]i rises were characterized, the concentration–response plots were established and the pathways underlying DIM-evoked Ca2+ entry and Ca2+ release were explored. The effect of DIM on cell viability was also investigated using the tetrazolium assay.
Materials and methods
Chemicals
The reagents for cell culture were obtained from Gibco (Gaithersburg, Maryland, USA). Fura-2/AM and 1,2-bis(2-aminophenoxy)ethane-N,N,N',N'-tetraacetic acid (BAPTA)/AM were purchased from Molecular Probes (Eugene, Oregon, USA). DIM and all other reagents were from Sigma-Aldrich (St. Louis, Missouri, USA) unless otherwise indicated.
Cell culture
MDCK canine renal tubular cells obtained from Bioresource Collection and Research Center (Taiwan) were cultured in Dulbecco’s modified Eagle medium supplemented with 10% heat-inactivated fetal bovine serum, 100 U/ml penicillin and 100 μg/ml streptomycin.
Solutions used in [Ca2+]i measurements
Ca2+-containing medium (pH 7.4) had 140 mM NaCl, 5 mM KCl, 1 mM MgCl2, 2 mM CaCl2, 10 mM HEPES and 5 mM glucose. DIM was dissolved in dimethyl sulfoxide as a 1 M stock solution. Ca2+-free medium contained similar components as Ca2+-containing medium except that CaCl2 was omitted and 2 mM MgCl2 and 0.3 mM EGTA were added. The other chemicals were dissolved in water, ethanol or dimethyl sulfoxide. The concentration of organic solvents in the experimental solutions did not exceed 0.1% and did not affect viability or basal [Ca2+]i.
[Ca2+]i measurements
Confluent cells grown on 6 cm dishes were trypsinized and made into a suspension in culture medium at a density of 106/ml. Cell viability was determined by trypan blue exclusion (adding 0.2% trypan blue to 0.1 ml cell suspension). The viability was greater than 95% after the treatment. Cells were subsequently loaded with 2 μM fura-2/AM for 30 min at 25°C in the same medium. After loading, cells were washed with Ca2+-containing medium twice and was made into a suspension in Ca2+-containing medium at a density of 107/ml. Fura-2 fluorescence measurements were performed in a water-jacketed cuvette (25°C) with continuous stirring; the cuvette contained 1 ml of medium and 0.5 million cells. Fluorescence was monitored with a Shimadzu RF-5301PC spectrofluorophotometer immediately after 0.1 ml cell suspension was added to 0.9 ml Ca2+-containing or Ca2+-free medium, by recording excitation signals at 340 nm and 380 nm and emission signal at 510 nm at 1-s intervals. During the recording, reagents were added to the cuvette by pausing the recording for 2 s to open and close the cuvette-containing chamber. For calibration of [Ca2+]i, after the completion of the experiments, the detergent Triton X-100 (0.1%) and CaCl2 (5 mM) were added to the cuvette to obtain the maximal fura-2 fluorescence. Then the Ca2+ chelator EGTA (10 mM) was added to chelate Ca2+ in the cuvette to obtain the minimal fura-2 fluorescence. Control experiments showed that cells bathed in a cuvette had a viability of 95% after 20 min of fluorescence measurements. [Ca2+]i was calculated as previously described. 22 Mn2+ quenching of fura-2 fluorescence was performed in Ca2+-containing medium containing 50 μM MnCl2. MnCl2 was added to cell suspension in the cuvette 30 s before the fluorescence recoding was started. Data were recorded at excitation signal at 360 nm (Ca2+-insensitive) and emission signal at 510 nm at 1-s intervals as described previously. 23
Cell viability assays
The measurement of cell viability was based on the ability of cells to cleave tetrazolium salts by dehydrogenases. Augmentation in the amount of developed color directly correlated with the number of live cells. Assays were performed according to manufacturer’s instructions specifically designed for this assay (Roche Molecular Biochemical, Indianapolis, Indiana, USA). Cells were seeded in 96-well plates at a density of 10,000 cells/well in culture medium for 24 h in the presence of DIM. The cell viability detecting reagent 4-[3-[4-lodophenyl]-2-4(4-nitrophenyl)-2H-5-tetrazolio-1,3-benzene disulfonate] (WST-1; 10 μl pure solution) was added to samples after DIM treatment, and cells were incubated for 30 min in a humidified atmosphere. In experiments using BAPTA/AM to chelate cytosolic Ca2+, fura-2-loaded cells were treated with 10 μM BAPTA/AM for 1 h prior to incubation with DIM. The cells were washed once with Ca2+-containing medium and incubated with and/or without DIM for 24 h. The absorbance of samples (A 450) was determined using an enzyme-linked immunosorbent assay reader. Absolute optical density was normalized to the absorbance of unstimulated cells in each plate and expressed as a percentage of the control value.
Statistics
Data are reported as mean SEM. of three experiments. Data were analyzed by one or two-way analysis of variances (ANOVA) using the Statistical Analysis System (SAS®, SAS Institute Inc., Cary, North Carolina, USA). There are two independent variables in a two-way ANOVA, which are called factors. The idea is that there are two variables, factors, which affect the dependent variable. Each factor will have two or more levels within it, and the degrees of freedom for each factor is one less than the number of levels. Therefore, there are two factors (time and concentration) in two-way ANOVA. Figure 1 had with or without Ca2+ and Figure 6 had with or without BAPTA-AM as second factors. One-way ANOVA was used in other figures. Multiple comparisons between group mean values were performed by post hoc analysis using the Tukey’s honestly significantly difference (HSD) procedure. A p value less than 0.05 were considered significant.

(a) Effect of DIM on [Ca2+]i in fura-2-loaded MDCK cells. DIM was added at 25 s. The concentration of DIM was indicated. The experiments were performed in Ca2+-containing medium. (b) Effect of DIM on [Ca2+]i in the absence of extracellular Ca2+. DIM (10 or 50 μM) was added at 25 s in Ca2+-free medium. (c) Concentration–response plots of DIM-induced [Ca2+]i rises in the presence or absence of extracellular Ca2+. Y axis is the percentage of the net (baseline subtracted) area under the curve (25–250 s) of the [Ca2+]i rise induced by 50 μM DIM in Ca2+-containing medium. Data are mean ± SEM of three experiments. Three experiments are independent biological replicates. *p < 0.05 compared with open circles. [Ca2+]i: cytosolic Ca2+ concentrations; DIM: diindolylmethane; SEM: standard error of mean.
Results
Effect of DIM on [Ca2+]i
The effect of DIM on basal [Ca2+]i was examined. Figure 1(a) shows that the basal [Ca2+]i level was 50 ± 2 nM. At concentrations between 1 and 50 nM, DIM induced a [Ca2+]i rise in a concentration-dependent manner in Ca2+-containing medium. At a concentration of 50 nM, DIM evoked a [Ca2+]i rise that attained to a net increase of 150 ± 2 nM (n = 3) followed by a slow decay. The Ca2+ response saturated at 50 μM DIM because at a concentration of 70 μM, DIM evoked a similar response as that induced by 50 μM. Figure 1(b) shows that in the absence of extracellular Ca2+, 50 μM DIM induced a [Ca2+]i rise of 65 ± 2 nM and at a concentration of 10 μM, DIM induce a [Ca2+]i rise of 27 ± 2 nM. Figure 1(c) shows the concentration–response plots of DIM-induced responses. The EC50 value was 5 ± 2 mM in Ca2+-containing medium by using a Hill equation. The term EC50 (half maximal effective concentration) refers to the concentration of DIM which induces a response halfway between the baseline and maximum after 250 sec.
DIM-induced Mn2+ influx
Experiments were performed to confirm that DIM-evoked [Ca2+]i rise involved Ca2+ influx. Mn2+ enters cells through similar mechanisms as Ca2+ but quenches fura-2 fluorescence at all excitation wavelengths. 23 Therefore, quenching of fura-2 fluorescence excited at the Ca2+-insensitive excitation wavelength of 360 nm by Mn2+ implicates Ca2+ influx. Figure 2 shows that 50 μM DIM evoked an instant decrease in the 360 nm excitation signal by 90 ± 2 (n = 3) arbitrary units. This suggests that Ca2+ influx participates in DIM-evoked [Ca2+]i rise.

Effect of DIM on Ca2+ influx by measuring Mn2+ quenching of fura-2 fluorescence. Experiments were performed in Ca2+-containing medium. MnCl2 (50 μM) was added to cells 1 min before fluorescence measurements. The Y axis is fluorescence intensity (in arbitrary units) measured at the Ca2+-insensitive excitation wavelength of 360 nm and the emission wavelength of 510 nm. Trace a: control, without DIM. Trace b: DIM (50 μM) was added as indicated. Data are mean ± SEM of three experiments. Three experiments are independent biological replicates. DIM: diindolylmethane; SEM: standard error of mean.
Pathways of DIM-induced Ca2+ entry
Experiments were conducted to explore the Ca2+ entry pathway of the DIM-induced [Ca2+]i rise. Three Ca2+ entry inhibitors: nifedipine (1 μM), econazole (0.5 μM) and SK&F96365 (5 μM); phorbol 12-myristate 13 acetate (PMA; 1 nM; a protein kinase C activator) and GF109203X (2 μM; a protein kinase C inhibitor) were applied 1 min before DIM. These agents all significantly inhibited DIM-induced [Ca2+]i rise to different degrees (Figure 3(a)). Figure 3(b) shows that in Ca2+-free medium, PMA also inhibited DIM-induced [Ca2+]i rise by 46 ± 2 nM (p < 0.05).
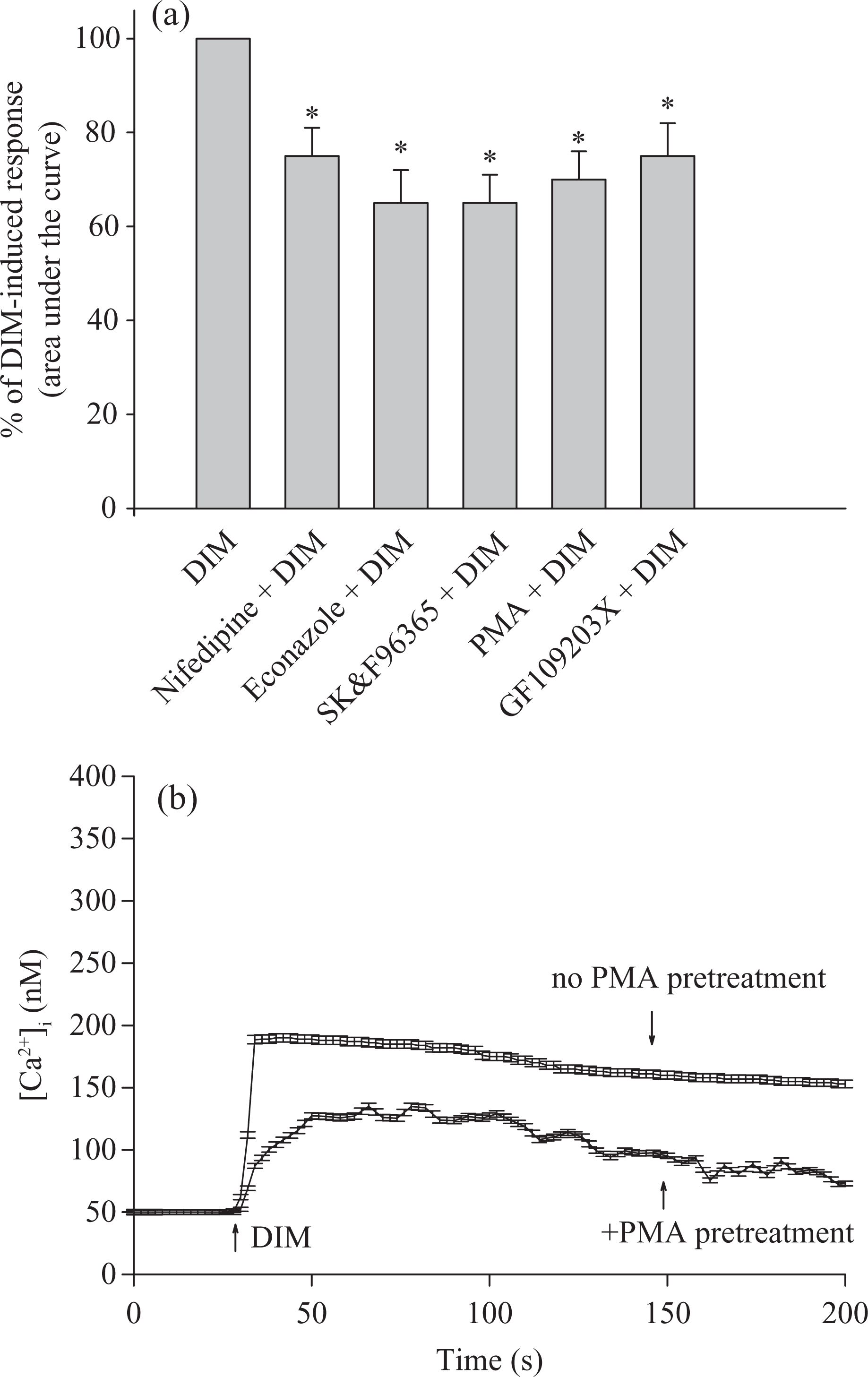
Effect of Ca2+ channel modulators on DIM-induced [Ca2+]i rise. (a) In blocker- or modulator-treated groups, the reagent was added 1 min before DIM (50 μM). The concentration was 10 nM for PMA, 2 μM for GF109203X, 1 μM for nifedipine, 0.5 μM for econazole and 5 μM for SK&F96365. Data are expressed as the percentage of control (first column) that is the area under the curve (25–200 s) of 50 μM DIM-induced [Ca2+]i rise and are mean ±SEM of three experiments. Three experiments are independent biological replicates. *p < 0.05 compared with the first column. (b) Recording of DIM-induced responses in the presence or absence of PMA. Data are mean + SEM of three experiments. [Ca2+]i: cytosolic Ca2+ concentrations; DIM: diindolylmethane; PMA: phorbol 12-myristate 13-acetate; SEM: standard error of mean.
Internal stores of DIM-induced [Ca2+]i rise
Efforts were made to explore the Ca2+ store involved in DIM-induced [Ca2+]i rise. Previous studies have shown that the endoplasmic reticulum is the major Ca2+ store in MDCK cells. 20,21 Figure 4(a) shows that in Ca2+-free medium, addition of 50 μM 2,5-di-tert-butylhydroquinone (BHQ) 24 induced a [Ca2+]i rise of 50 ± 2 nM. DIM added at 500 s induced a small [Ca2+]i rise of 25 ± 2 nM, which was smaller by 68% than the DIM-induced [Ca2+]i rise shown in Figure 4(b). Figure 4(b) shows that BHQ added at 500s after DIM pretreatment failed to increase [Ca2+]i. To confirm the BHQ’s data, another endoplasmic reticulum Ca2+ pump inhibitor thapsigargin (TG, 1 μM) 25 was used in similar experiments. Figure 4(c) shows that TG induced a [Ca2+]i rise of 45 ± 2 nM. DIM added at 500 s induced a small [Ca2+]i rise of 48 ± 2 nM. Figure 4(d) shows that TG added after DIM pretreatment failed to increase [Ca2+]i.

Intracellular Ca2+ stores of DIM-induced Ca2+ release. Experiments were performed in Ca2+-free medium. DIM (50 μM; a, b, c and d), TG (1 μM; c and d) and BHQ (50 μM; a and b) were added at time points indicated. Data are mean ± SEM of three experiments. Three experiments are independent biological replicates. BHQ: 2,5-di-tert-butylhydroquinone; DIM: diindolylmethane; SEM: standard error of mean; TG: thapsigargin.
The role of phospholipase C in DIM-induced [Ca2+]i rise
Phospholipase C-dependent production of inositol 1,4,5-trisphosphate is a key process for releasing Ca2+ from the endoplasmic reticulum. 16 Because DIM released Ca2+ from the endoplasmic reticulum, the role of phospholipase C in this event was examined. U73122, a phospholipase C inhibitor, 26 was used to see whether the activation of this enzyme was required for DIM-induced Ca2+ release. Figure 5(a) shows that ATP (10 μM) induced a [Ca2+]i rise of 51 ± 2 nM. ATP is a phospholipase C-dependent agonist of [Ca2+]i rise in most cell types. 27 In Figure 5(b), first column shows that 50 μM DIM induced [Ca2+]i rise. Second column shows that 2 μM U73122 did not alter basal [Ca2+]i. Third column shows the ATP-induced [Ca2+]i rise compared with DIM control. Fourth column shows that U73122 completely abolished ATP-induced [Ca2+]i rise. Fifth column shows that U73122 and ATP pretreatment partially inhibited 50 μM DIM-induced [Ca2+]i rise. Sixth column shows that U73122 partially inhibited DIM-induced [Ca2+]i rise. Together, Figure 5(b) shows that incubation with 2 μM U73122 did not change basal [Ca2+]i but abolished ATP-induced [Ca2+]i rise. This suggests that U73122 effectively suppressed phospholipase C activity. Figure 5(b) also shows that incubation with 2 μM U73122 did not alter basal [Ca2+]i but reduced DIM-induced [Ca2+]i rise by 50% in the area under the curve. U73343 (2 μM), a U73122 analogue, failed to demonstrate an inhibition (not shown).

Effect of U73122 on DIM-induced Ca2+ release. Experiments were performed in Ca2+-free medium. (a) ATP (10 μM) was added as indicated. (B) U73122 (2 μM), ATP (10 μM) and DIM (50 μM) were added as indicated. Data are mean ± SEM of three experiments that are independent biological replicates. *p < 0.05 compared with first bar (control). Control is the area under the curve of 50 μM DIM-induced [Ca2+]i rise (25–250s). ATP: adenosine triphosphate; DIM: diindolylmethane.

Effect of DIM on viability of MDCK cells. Cells were treated with 0–50 μM DIM for 24 h and the cell viability assay was performed. Data are mean ± SEM of three experiments. Each treatment had six replicates (wells). Data are expressed as percentage of control that is the increase in cell numbers in DIM-free groups. Control had 10,567 ± 712 cells/well before experiments and had 13,466 ± 798 cells/well after incubation for 24 h. *p < 0.05 compared with control. In each group, the Ca2+ chelator BAPTA/AM (10 μM) was added to fura-2-loaded cells followed by treatment with DIM in Ca2+-containing medium. Cell viability assay was subsequently performed. #p < 0.05 compared with the pairing group. Data are mean ± SEM of three experiments. Three experiments are independent biological replicates. BAPTA: 1,2-bis(2-aminophenoxy)ethane-N,N,N',N'-tetraacetic acid; DIM: diindolylmethane; SEM: standard error of mean.
Effect of DIM on cell viability
Given that acute incubation with DIM induced a substantial and lasting [Ca2+]i rise, and that unregulated [Ca2+]i rise often alters cell viability, 15 experiments were performed to examine the effect of DIM on viability of MDCK cells. Cells were treated with 0–50 μM DIM for 24 h, and the tetrazolium assay was performed. In the presence of 1, 10, 40, 50 μM DIM, cell viability slightly increased. At 20 and 30 μM, DIM did not affect viability when compared with the control (Figure 6).
Relationship between DIM-induced [Ca2+]i rise and cell proliferation
The next issue was whether the DIM-induced proliferation was caused by a preceding [Ca2+]i rise. The intracellular Ca2+ chelator BAPTA/AM 28 was used to prevent a [Ca2+]i rise during DIM treatment. Figure 6 also shows that 10 μM BAPTA/AM loading did not alter the control value of cell viability. DIM (50 μM) did not induce a [Ca2+]i rise in BAPTA/AM-treated cells (not shown). In the presence of 50 μM DIM, BAPTA/AM loading partly reversed DIM-induced cell proliferation (n = 3; p < 0.05). At 10–40 μM, DIM did not affect DIM’s effect.
Discussion
Previous studies on DIM were mostly performed on cancer cells. Our study is the first to show that DIM induced a [Ca2+]i rise in noncancer renal tubular cells. The data show that DIM induced a concentration-dependent [Ca2+]i rise in MDCK cells. DIM increased [Ca2+]i by depleting intracellular Ca2+ stores and causing Ca2+ entry from extracellular milieu, because removing extracellular Ca2+ reduced 50% of DIM-induced [Ca2+]i rise. In contrast, DIM has been shown to evoke Ca2+ release from the endoplasmic reticulum of human prostate, cervical cancer cells 3 and pancreatic cancer cells 29 without evidence of inducing Ca2+ entry. In hepatoma cells 9 and osteosarcoma cells, 8 DIM was also shown to cause Ca2+ release and Ca2+ entry. Thus it appears that the mechanisms underlying DIM-induced Ca2+ signal varies with cell type. Mn2+ quenching data also suggest that DIM induced Ca2+ entry. Because Mn2+ and Ca2+ share similar movement pathways in cells, but Mn2+ quenches fura-2 fluorescence at all excitation wavelengths, while Ca2+ does not, 23 quenching of fura-2 fluorescence excited at the Ca2+-insensitive excitation wavelength of 360 nm by Mn2+ would suggest the occurrence of Ca2+ entry into cells. Furthermore, the DIM-induced Mn2+ quenching did not change over time, suggesting that Mn2+ influx continuously occurred throughout the measurement interval.
Removal of extracellular Ca2+ reduced the DIM-induced [Ca2+]i rise throughout the measurement period, suggesting that Ca2+ entry occurred during the whole stimulation interval (200 s). The mechanism of DIM-induced Ca2+ influx was explored. The results suggest that DIM might cause Ca2+ entry via stimulating store-operated Ca2+ entry, which is induced by the depletion of intracellular Ca2+ stores, based on the inhibition of DIM-induced [Ca2+]i rise by nifedipine, econazole and SK&F96365. These three compounds have often been applied as blockers of store-operated Ca2+ entry in different cell types. 30 –33 Because activation of phospholipase C produces IP3 and diacylglycerol, which stimulates protein kinase C, the effect of regulation of protein kinase C activity on DIM-induced [Ca2+]i rise was explored. Both activation and inhibition of protein kinase C inhibited DIM-induced [Ca2+]i rise. Regarding the Ca2+ stores involved in DIM-induced Ca2+ release, the TG/BHQ-sensitive endoplasmic reticulum stores might be the dominant one because TG/BHQ pretreatment both greatly inhibited DIM-induced [Ca2+]i rise; and conversely, DIM pretreatment also inhibited TG/BHQ-induced Ca2+ release. Furthermore, it seems that phospholipase C-dependent pathways played a significant role in DIM-induced Ca2+ release, since the response was inhibited by 50% when phospholipase C activity was inhibited by U73122. Thus it appears that DIM-induced Ca2+ release was caused by an IP3-dependent Ca2+ release from the endoplasmic reticulum.
DIM has been shown to induce apoptosis in several types of cancer cells. This study is the first to show that DIM was not cytotoxic to MDCK cells, and at higher concentrations, DIM even enhanced proliferation by 20%. Thus, these data suggest that the chemopreventive drug DIM may selectively inhibit cancer cells without killing normal cells. In vitro studies, DIM (10–50 μM) caused cell death in breast cancer cells, 7 osteosarcoma cells 8 or hepatoma cells. 9 In vivo studies, DIM (20–50 μM) induced immunotoxicity via splenocyte apoptosis in neonatal mice. 11 However, in our study, DIM (10–50 μM) did not induce cell death in MDCK cells. Thus, DIM may act differently on viability in cancer and noncancer cells.
A previous study 14 explored the plasma concentration of DIM after oral administration. The doses administered were 50, 100, 150, 200 and 300 mg/day in drug-free, nonsmoking, healthy adults. No BioResponse DIM (BR-DIM)-related adverse effects were reported at doses up to 200 mg. A single 200 mg dose of BR-DIM resulted in a mean C max of 104 ng/ml (∼0.42 μM) after 1 h. A single 300 mg dose of BR-DIM resulted in a mean C max of 108 ng/ml (∼0.44 μM) after 1 h. BR-DIM is well tolerated at single doses of up to 200 mg and that increasing the dose to 300 mg did not cause a significant increase in C max. 14 In contrast, our data show that DIM at concentrations between 1 and 50 µM evoked [Ca2+]i rises without altering cell viability in MDCK renal tubular cells. However, in elderly or renal impaired patients, the plasma concentration of DIM after oral administration might be three-fold higher than in healthy adults. 14 Thus, our study may have clinical relevance.
Together, the results show that the natural product DIM induced Ca2+ release from endoplasmic reticulum in a phospholipase C-dependent manner and also caused Ca2+ influx via a protein kinase C-dependent, store-operated Ca2+ entry in MDCK renal cells. DIM also enhanced cell proliferation. A rise in [Ca2+]i can lead to numerous Ca2+-associated cellular responses, thus the effect of DIM on Ca2+ movement should be considered in other types of in vitro research.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This work was supported by Zuoying Armed Forces General Hospital (ZAFGH100-24) to YC Fang and Kaohsiung Veterans General Hospital (VGHKS101-019) to CR Jan.
