Abstract
Space toxicology is a unique and targeted discipline for spaceflight, space habitation, and occupation of celestial bodies including planets, moons, and asteroids. Astronaut explorers face distinctive health challenges and limited resources for rescue and medical care during space operation. A central goal of space toxicology is to protect the health of the astronaut by assessing potential chemical exposures during spaceflight and setting safe limits that will protect the astronaut against chemical exposures while in a physiologically altered state. In order to maintain sustained occupation in space on the International Space Station (ISS), toxicological risks must be assessed and managed within the context of isolation, continuous exposures, reuse of air and water, limited rescue options, and the need to use highly toxic compounds for propulsion and other purposes. As we begin to explore other celestial bodies, in situ toxicological risks, such as inhalation of reactive mineral dusts, must also be managed.
The Space Toxicology: Human Health During Space Operations symposium was chaired by Dr Noreen Khan-Mayberry and cochaired by Dr John T. James as a part of the American College of Toxicology (ACT) annual meeting. The focus on this subdiscipline of toxicology was intended to introduce the greater toxicology community to the unique toxicological risks and human health issues inherent during human spaceflight operations. Space toxicology presents unique challenges due to confined living and reduced gravity. The astronaut crew members experience physiological changes that compound the complexity in managing their exposures to chemical contaminants. This symposium gave: (1) an introduction to the history of Space Toxicology; (2) the processes for setting standards and guidelines for air and water exposure; (3) the process of risk-based monitoring; and (4) National Aeronautics and Space Administration’s (NASA) research on pulmonary toxicity of lunar dusts.
A History of Space Toxicology (John T. James, PhD)
The possibility of toxic exposures during spaceflight was a concern from the beginning of human spaceflight by the United States. As our experience grew, NASA recognized that unique air-quality standards were needed to establish boundaries on air pollution and that the sources of pollution were innumerable. Monitoring strategies were developed to meet the challenges of managing toxic events, and control strategies were implemented to restrict the probability of accidental releases. Despite our best effort, toxic events still occur and from each of these we learn to improve our risk profile to better ensure a healthy and productive crew.
Sources of Toxicological Risk to Space Crews
The earliest toxicological risk that concerned space capsule builders was the possibility of excess off-gassing of materials that would pollute the capsule breathing atmosphere. 1 This was managed by rigorous testing of all materials to ensure that the air revitalization systems could remove pollutants to safe levels. There was also concern that highly toxic propellants could contaminate the extravehicular-activity suits during space walks, and then contaminate the capsule atmosphere when the crew member returned to the capsule. Obviously metabolic products, especially carbon dioxide, had to be adequately managed to prevent adverse effects.
As experience accumulated over decades of spaceflight, new sources made themselves apparent. These included utility compounds such as lubricants, cleaning agents, and hygiene products that can gradually pollute the atmosphere. Sudden and potentially dangerous releases that had to be controlled included leaks of toxic compounds from payload experiments (eg, tissue fixatives), batteries with toxic volatile components such as thionyl chloride, and thermodegradation of polymeric materials (eg, shorting of electronic components). As vehicles aged and systems failed more frequently, we recognized additional sources of air pollution such as volatile products of microbial action, pollution from systems leaks (eg, ethylene glycol), and corrosion of metallic materials (eg, cadmium-plated components) that can produce harmful particulates. Increasing complexity of space vehicles, such as the International Space Station (ISS) and the broader range of experiments conducted aboard spacecraft has made the job of controlling air pollution a substantial undertaking. Some modules of the ISS are more than a decade old and at one point there were 13 astronauts working aboard the vehicle while the space shuttle was docked to it.
Historical Overview of Spacecraft Maximum Allowable Concentrations (SMACs)
NASA in cooperation with the National Academy of Sciences, and later the National Research Council (NRC), has set safe exposure limits for those compounds anticipated to be present in spacecraft atmospheres. The initial effort in 1964 was to set continuous-exposure limits for the Apollo vehicles that were going to the moon and back—a journey that could take up to 2 weeks. This was done for approximately 80 compounds 5 years before the first successful voyage to the lunar surface and back. 2 Even before the first successful lunar landing, missions of up to 1000 days were being anticipated and limits of 90 and 1000 days were set for about a dozen compounds. 3 The grand vision of long missions encountered the reality of high cost, so the mission durations shrunk, and the limits were adjusted to shorter time periods. There was a need for limits for a variety of short-duration flights, and in 1976 limits were established for space shuttle flights. 4,5 After a decade of shuttle flights, NASA began to envision an earth-orbiting space station in which crews would remain for 6-month periods. In addition, real-time on-board air-quality analyzers were being developed. Thus, it made sense to set long-term limits for this space station that reflected stays up to 6 months and also to set short-term limits indicative of our growing ability to detect and quantify products from accidental releases such as combustion events. In the 1990s, SMACs were set for exposures from 1 hour to 180 days. 6 In the past few years, the possibility of long-term missions to distant celestial bodies has reappeared; and in 2008, NASA set limits for many compounds for continuous exposures up to 1000 days. 7
Modern SMACs (those set since 1992) consider the physiological changes induced by spaceflight. 8 For example, the SMAC for benzene is reduced 3-fold because of the excess risk from space radiation which targets the blood-forming cells of the bone marrow just as benzene does. Cardiac arrhythmias have been documented during stressful times in space, so SMACs for compounds that sensitize the myocardium to arrhythmias are reduced by a factor of 5 to compensate for this spaceflight-induced risk. The space station can be noisy and temporary hearing loss is not unusual in returned astronauts, so the limits on ototoxic compounds are reduced accordingly. Astronauts also lose approximately 10% of their red blood cell mass in space, so hematotoxicants have reduced limits for spacecraft atmospheres.
Air-Quality Monitoring—Going High Tech
Historically, NASA has obtained air samples during missions and then analyzed those samples when they are returned to the earth. 9 This approach has the advantage that samples can be thoroughly analyzed by large, complex instruments in the laboratory; however, some compounds are lost to the container walls or sorbents and the results may not be available until months after the samples are acquired. This means that any investigation of the source of unexpected compounds found in the samples is severely hampered. We wanted to alleviate this handicap and eventually allow astronauts to manage air-quality problems with on-board resources. Thus, in the early 1990s, NASA began to develop a suite of instruments that can quantify combustion products and a large group of trace organic compounds. Identifying precisely which combustion products will be the greatest threat to the crew is not simple because it depends on the composition of the material burned, the temperature of pyrolysis, and the availability of oxygen. We are currently targeting carbon monoxide, hydrogen cyanide, and acid gasses as the most likely to be harmful. In terms of trace contaminant quantification, NASA has flown a volatile organic analyzer (VOA) for 8 years 10 and is working with partners to explore the use of a Fourier transform infrared (FTIR) spectrometer, 11 a gas chromatograph/mass spectrometer, 12 and a gas chromatograph/differential mobility spectrometer. 13
Toxicological Events During Spaceflight
Transparent events
Some atmospheric pollution events aboard spacecraft are transparent to the crew. For example, the air conditioner units in the service module (SM) of the ISS, and in the core module of the old Mir space station, periodically leaked Freon 218 (perfluoropropane), which is virtually nontoxic. This volatile compound spreads throughout the station complex and is nearly impossible to scrub from the atmosphere. Formaldehyde is produced by off-gassing and at times has exceeded limits set to protect against mucosal irritation, although no such irritation has been reported. It is always more concentrated in the US Laboratory module than in the SM. 14
Minor events
Toxicological events that are sufficient to elicit minor symptoms in the crew have occurred at least since the days of Apollo. Lunar dust, when floating in the spacecraft atmosphere caused the astronauts to don helmets until the dust was cleared. On rare occasions, the astronauts reported respiratory symptoms from brief exposures to the dust. One ground-based worker seemed to develop increasing sensitivity to the dust when he worked with it on several occasions. 15 During the return voyage of the ill-fated Apollo 13 capsule from its swing around the moon, the ability to scrub CO2 was much diminished. The CO2 levels eventually reached about 15 mm Hg (2%), but no specific symptoms were ascribed to this exposure. 16 On occasion CO2 accumulates in pockets that are poorly ventilated and this has caused minor discomfort and headaches. In space, there is no such thing as “up” so convection does not move warm, CO2-laden breath away from the face. In a stagnant area one can easily rebreathe his or her own exhaled breath repeatedly, causing minor symptoms. Canisters with lithium hydroxide (LiOH) have been used for many years to remove CO2 from the atmosphere; however, if the LiOH dust is not vacuumed from the canisters before they are inserted into the air revitalization system, astronauts can experience minor upper airway irritation.
Fires or pyrolysis events (heating to the point of breakdown of a polymer) are always a concern during spaceflight. In 1997 aboard the Mir space station, the oxygen generator caught fire and was destroyed in an oxygen-rich blaze of rather spectacular proportions. 17 The event was obvious to the crew, so they donned protective masks and did everything they could to stop the fire and then clean up the atmosphere. Although this was a frightening event for many reasons, it was not a major toxicological event because the oxygen-rich fire produced very little CO. It did produce a few ppm of benzene, but this was rapidly scrubbed from the atmosphere by the Russian air revitalization system. Also, during the 1990s, there were several minor events aboard the space shuttle involving pyrolysis of electronic components such as wire insulation, diodes, and resistors. These events produced a strong burned-electronic smell in the cabin and plenty of anxiety, but no serious toxic effects were reported.
Microbial contamination of spacecraft systems that are aqueous based can occur under favorable conditions. Under some conditions, this can result in significant air pollution. During the STS-55 mission in early 1993, the waste management system malfunctioned, so the crew began to place some waste in contingency bags. Periodically the bags' contents had to be emptied (squeezed) through a port for disposal into space. The crew reported that noxious odors had contaminated the areas near the bags and that they were not inclined to continue the emptying process. An air sample was taken and its analysis showed 3 di-methyl sulfide compounds. Using bags identical to the ones on orbit and similar waste material, we demonstrated microbial production of these compounds and penetration of them through the walls of the storage bags. 18
Moderate events
Fortunately moderately serious events have been unusual throughout the course of human spaceflight. The earliest event that caused moderate toxicological effects occurred aboard STS-40, in 1992. 18 On orbit it was noted that the refrigerator was emitting an acrid odor and excess off-gassing was suspected. Crew members periodically went to a different module to get fresh air. Eventually, the unit was unpowered and all openings taped. When the unit was disassembled on the ground, engineers discovered that the fan motor had overheated. This burned its housing, which was made of polyoxyethylene, an excellent source of formaldehyde when heated. The overheating was caused by set screws on the fan shaft couplers coming out against a guide sleeve so that the shaft could not turn. Since there was no thermal protection on the motor, power continued to be supplied to the locked motor, causing overheating. The odor was so strong that if the crew had not been able to get fresh air in another module, the flight may have been stopped early.
During the late 1990s aboard the Mir space station, there were repeated leaks of the ethylene glycol heat-exchange fluid. The vapors caused mucosal irritation in the crew members and if they encountered a sizable bleb of the fluid in the face, then the eyes became extremely irritated. The leaks occurred primarily in the Kvant module where the highest concentrations remained. Once a leak occurred, the fluid lodged on cooler, nearly inaccessible surfaces and remained there almost indefinitely. Ethylene glycol also ended up in the water-recovery system where its removal was problematic. 19
Another moderate pyrolysis event occurred in 1998 aboard Mir about 1 year after the spectacular oxygen-generator fire noted in the preceding paragraph. 20 At face value, this seemed to be a much less significant event; however, toxicologically it was much more important. A hot filter had been prematurely switched into the trace-contaminant removal system and this caused a downstream cellulose filter to burn. A small amount of smoke was observed; however, a few hours later, crew members experienced headaches and nausea. An experimental monitor for CO was aboard and was showing readings above 400 ppm; this was confirmed to be accurate by the amount of CO found in a grab sample that was analyzed on the ground much later. The levels of blood carboxy-hemoglobin were estimated from the airborne concentrations of CO. This was estimated to be as high as 40%, which explained the reported symptoms. The peak blood carboxy-hemoglobin concentration occurred approximately 5 hours after the burn. This explains why the symptoms were delayed. Although much less obvious than the oxygen generator fire, this “small” fire was a toxicologically dangerous event.
In 2002, a moderate event occurred aboard the ISS when portable filters used during extravehicular activity (EVA) were regenerated with heat to discharge pollutants from the filters into the ISS atmosphere. 21 These specific filters had been in a position to absorb ISS air pollutants for 6 months, so they were much dirtier than expected. A few hours into the regeneration process, the crew reported noxious odors. The generation was stopped and the crew took refuge in another module (with hatch closed) until the air revitalization system cleaned the pollutants discharged during the regeneration process. This required about 30 hours. Samples taken during this time showed high concentrations of 1-butanol and ethyl acetate.
Severe event
During the course of human spaceflight, only 1 toxicological event can be classified as severe. In 1975, the United States and Russia did a demonstration project in which spacecraft from the 2 countries docked in space. 22 The Apollo capsule was used by NASA for the rendezvous. After undocking, as it was returning back through the atmosphere, the capsule interior was repressurizing through a pressure-relief valve by allowing outside air to enter. Unfortunately, the thrusters were still active because the capsule’s descent was unstable. This caused nitrogen tetroxide fumes to enter the cabin and decompose into NO2. The capsule concentrations peaked near 700 ppm (rough indirect estimate of NO2). Upon return to earth, post-recovery, the crew members developed respiratory symptoms. They were given chest X rays the next day. These showed patterns consistent with alveolar exudates. Corticosteroid treatment was given and the X rays were normal 5 days after landing. No long-term health effects were reported in this crew.
NASA has developed a set of air-quality standards (SMACs) to define levels to which air pollutants must be controlled to protect crew health. Unfortunately, toxicological events are a normal part of human spaceflight. Reactive compounds useful as propellants can pose a risk to the crew. High-temperature components and many electronic devices invite pyrolysis of polymeric material such as wire insulation or components of circuits. Payloads and systems contain toxic compounds that can escape containment and evaporate into the air to expose crews. On-board air monitors will facilitate our ability to manage these events when they occur; however, the first line of defense is to design out the possibility of a toxic exposure—an almost impossible, task given the weight and cost constraints of our incredibly complex spacecraft.
Spacecraft Maximum Allowable Concentrations (Rochelle Tyl, PhD)
History of SMACs
Spacecraft maximum allowable concentrations are defined as “the maximum concentrations of airborne substances that will not produce adverse health effects, cause significant discomfort, or degrade crew performance,” although short-term SMACs are set for contingencies and allow for temporary, minor effects. NASA requested that the NRC of the National Academies develop guidelines for establishing SMACs for various airborne contaminants and to review previous SMACs for various spacecraft contaminants to determine whether NASA’s recommended exposure limits are consistent with the guidelines recommended by the Committee. The NRC developed criteria and methods for preparing SMACs published in its 1992 report. Since then, NRC’s Committee on Spacecraft Exposure Guidelines has been reviewing NASA’s documentation of chemical-specific SMACs in reports published in 1994, 1996, and 2000. The current report 7 is an update of several of the original SMACs by an Expert Panel convened by the NRC.
The determination of SMACs has become even more important now to maintain sustained occupation in space on the ISS. Toxicological risks must be assessed and managed within the context of isolation, continuous exposures, reuse of air- and water-limited rescue options, and the need to use highly toxic compounds for propulsion. In its present configuration, the ISS can carry a crew of 3 to 6 astronauts for up to 180 days. Several hundred chemical contaminants are found in its closed-loop atmosphere (most are at very low concentrations).
The present NRC Expert Panel objectives were to update the NASA Spacecraft Water Exposure Guidelines (SWEGs) and SMACs from the last evaluation started in 1992, using the best available toxicologic risk assessment methods based on new studies, new data, new technologies, and new concerns.
Unique Population of Astronauts
Astronauts are healthy, bright, dedicated, and educated, but new concerns are: (1) they are of different ethnicities with possible ethnic-specific susceptibilities and concerns; (2) they are not all men; and (3) they experience physiological adaptation to microgravity that can make them especially or uniquely sensitive to toxicants. The durations of spaceflight for which SWEGs and SMACs are to be assigned have been extended to 1000 days for anticipated longer spaceflights (previously just to 180 days). The concerns are for astronaut performance in flight and for any adverse consequences from flight when they return. The SMAC classification scheme by duration and criteria is presented in Table 1 .
Spacecraft Maximum Allowable Concentration Classification

Abbreviation: SMAC, spacecraft maximum allowable concentrations.
Methods and Process for Evaluation and Setting of SMACs
The information evaluated by NASA and the NRC subcommittee to set SMACs includes: (1) physical and chemical characteristics of contaminant (2) relevant in vitro toxicity studies (3) toxicokinetic studies (4) mechanistic studies (5) animal toxicity studies conducted over a range of exposure durations (6) genotoxicity studies (7) carcinogenicity bioassays—2 years rat, 18 months mouse, and (8) human clinical and epidemiology studies. The toxic effects of concern included: (1) mortality, (2) morbidity, (3) functional impairment including mucosal irritation and central nervous system (CNS) depression, (4) specific organ system toxicities such as hepatic, renal, and endocrine, (5) neurotoxicity, (6) immunotoxicity, (7) reproductive toxicity, (8) genotoxicity, (9) carcinogenicity, and (10) cardiotoxicity. The determinants evaluated were: (1) identification of the most sensitive target organ or system affected, (2) the nature of the effect on the target tissue, (3) the exposure duration in relation to the SMAC being developed, (4) the dose−response relationship for the target tissue, benchmark dose analysis may be applied, (5) the rate of recovery, (6) the nature and severity of the injury, (7) cumulative effects, (8) toxicokinetic data, (9) interactions with other chemicals, and (10) the effects of microgravity.
The NRC expert subcommittee noted that toxicity data from human studies are most applicable and are used (when available) in preference to data from animal or in vitro studies. Toxicity data from animal species most representative of humans in terms of toxicodynamic and toxicokinetic properties are used for determining SMACs. Toxicity data from inhalation exposures are most useful for setting SMACs for airborne contaminants, because inhalation is the most likely route of exposure.
NASA toxicologists wrote a draft document for each chemical, identifying the key (driver) studies. The draft document and the key papers on which it is based were provided to the NRC committee. The committee members examined the document with special focus on concerns in their areas of expertise; additional papers were identified by committee members and distributed. The committee had conference calls and met in person to discuss each draft document, and provided questions, comments, additional edits, concerns, and so on, to each author. The author of each document revised it according to the committee’s comments, and the revised document was reviewed again at a subsequent meeting (with additional revisions if necessary). Once the document was accepted by the subcommittee, it was turned over to the NRC editorial staff for final external review and preparation for publication. 6,7
Spacecraft Water Exposure Standards (Summary by John T. James, PhD; Presented by Kenneth Thummel, PhD)
As NASA’s joint effort with the NRC to set air-quality standards was coming to an end in 1998, the agency decided that the team that had developed the air standards should remain intact and tackle the job of setting water-quality standards. After considerable discussion, the joint group determined that water-quality standards for spaceflight must be developed separately from ground-based standards and that the times of exposure for standards would include the following: 1 day, 10 days, 100 days, and 1000 days. The 2 shorter periods were for contingency situations in which drinking water had become polluted and the contaminant levels would be constrained to those that would produce no more than very mild adverse effects. The 100-day and 1000-day guidelines would be set to guard against any adverse effect to a high degree of certainty. It was understood that the composition of the NRC subcommittee would have to evolve to include members more familiar with setting water standards.
The need for water-quality standards was also driven by the reality that NASA would lead an international team in placing and operating a space station in orbit around the earth. One of the key purposes of such a station would be to develop and demonstrate technologies to recover and purify water. The on-orbit sources of water were to be first humidity condensate from the air and second, treated urine. The team had to first address the question, what compounds were most likely to appear in the water sources and which ones could survive purification to contaminate drinking water. A further question was which compounds might be amenable to monitoring in water.
The plan in the late 1990s was to monitor water for total organic carbon (TOC) and a number of individual contaminants on orbit; however, funding issues caused that effort to collapse to the on-board monitoring of TOC only. Nonetheless, the group decided that before the last days of the ISS there would be the capability to monitor many water pollutants on orbit, so the effort to set water guidelines was initiated. In addition, ground-based analysis of samples taken on orbit was on-going, so after-the-fact we would know whether the crew had consumed water that exceeded acceptable guidelines for potable use. While not ideal, this ground-based analytical approach has led to “slow-motion” management of ethylene glycol aboard the Mir space station and cadmium aboard the ISS. 23,24
One of the goals of water purification is to remove organic contaminants to a level that ensures that it is potable. The simplest way to monitor this is to measure the TOC; however, setting a standard for this measurement is problematic if one wants to protect crew health. The previous US limit for this parameter was 0.5 mg/L, whereas the Russian limit was 20 mg/L. Potable water samples from Space Station Mir and later ISS often exceeded the US limit but were typically within the Russian limit. The group set out to develop a standard that was better suited to management of water quality. The primary issue to resolve was what components were most likely to break through the water purification system and affect crew health. Clearly, if the TOC comprised mostly ethanol, then a relatively high level could be tolerated, whereas, if the main constituent of TOC were something much more toxic, say formaldehyde, then the TOC must be lower.
A careful analysis of the potential breakthrough products revealed that the worst compound that could break through was formaldehyde. This was based on data showing that substantial formaldehyde was captured in humidity condensate and if this were to evade the purification processes, then formaldehyde could appear in the product water. At about the same time, NASA and the NRC subcommittee were working on a SWEG for formaldehyde, so once that SWEG was settled, the task of setting a guideline for TOC was simplified. The final 100-day SWEG for TOC was set at 3 mg/L. 25
One point of discussion was whether NASA should develop a standard based on changes in TOC levels as measured by the on-board analyzer. The thinking was that even if the levels were below 3 mg/L, a significant change from the running baseline values would suggest that a new source of contamination had entered the recovered water or the purification system had begun to fail. Since this was not actually a crew-health standard but an operational standard, the NRC subcommittee declined to help with determination of this standard; however, it was recognized that such a standard would likely be useful during water-recovery operations. The key issue is what level of deviation from the running mean constitutes a real change versus a statistical variation. As we gain experience with on-board TOC analysis, we will be better able to address that question.
One of the more challenging SWEGs to set for an individual compound was one for ethylene glycol. This compound had frequently leaked from the thermal loops aboard Mir and had polluted the atmosphere and humidity condensate. At one point, it broke through the purification system in to the potable water. 23 Two troublesome questions emerged as NASA and the subcommittee grappled with guidelines for this compound. The first was whether the guideline should be lower because of the ability of ethylene glycol metabolites to form oxalate crystals in the kidney coupled with the perception that astronauts have a tendency to form oxalate kidney stones, presumably due to the excess release of calcium from bones. The second troublesome question was how to apply benchmark dose analysis to the very limited data on rats that had ingested ethylene glycol in drinking water.
In reference to the question about increasing the risk of forming kidney stones, it was determined that astronauts are in fact not more likely to form stones than persons in the general population. 26 This is primarily due to the fact that persons who are “stone formers” are not selected for the astronaut corps. Data showed that the rate of formation of kidney stones in astronauts during and shortly after a mission is much less (0.7 × 10−5/person per d) than in the general age-matched population (1.14 × 10−5/person per d). In addition, it was estimated that the additional load of oxalate to the kidney would be no more than 5% when ingesting 20 mg/L of ethylene glycol. Therefore, it was determined that no additional safety factor was needed in the ethylene glycol SWEG to protect astronauts from stone formation. 23
Early in the process of setting the SWEG for ethylene glycol, it was apparent that benchmark dose analysis could be applied to male rat data on kidney lesions produced by ingestion of ethylene glycol in drinking water. There were 4 exposure groups and a control group, each consisting of 10 animals. Three kidney lesions were identified: intratubular crystals, renal tubular degeneration, and subacute inflammation (30). Exposures were for 10 days or 90 days. 23 Several difficult questions emerged. What dose metric should we use—water concentration or average consumed by the groups of rats? Which model or models should be used to fit the dose−response data? How well does the selected model/models need to fit? Which level of benchmark risk should be used (1% or 10%) and should the maximum-likelihood value or the 95% lower confidence level be used as a point of departure?
We chose the average amount of ethylene glycol consumed as the best dose metric and renal tubular degeneration as the index lesion. In doing so, we noted that the metabolism of ethylene glycol leads to metabolites that must chemically react with critical cellular components to produce cellular degeneration. Thus a chemical-kinetic model seemed appropriate, and this suggested to us that the log-logistic model was most suitable to fit the data. We found that this model gave a very good fit to the tubular degeneration data. We also selected the lower limit of the 10% risk of tubular degeneration in male rats as the point of departure for 10-day and 90-day exposures. To this, we applied factors for potential species differences, interindividual variability, body weight differences between species, and the volume of water consumed each day. The benchmark doses turned out to be drivers for the 10-day and 100-day SWEGs, when compared to the default No Observable Adverse Effect Level - Low Observable Adverse Effect Level (NOAEL-LOAEL) approach. 23
The joint effort to set guidelines for water purity concluded in 2008, with the publication of volume 3 in the series. 27 In the end, we set SWEGs for 24 individual compounds, for 3 groups of compounds, and for TOC. We expect to update these SWEGs at least every decade and in the meantime will add more SWEGs by developing these at NASA and publishing the results in peer-reviewed toxicology journals to ensure external review by experts.
Risk-Based Monitoring of Spacecraft Pollutants (Noreen Khan-Mayberry, PhD)
As air- and water-quality standards specifically for human spaceflight became available, the need to perform real-time, onboard monitoring of some of the riskiest compounds became evident. 28 NASA has developed and used various instruments to monitor selected combustion products since the early 1990s. We have targeted specific pollutants for monitoring including carbon dioxide, which can build up in “pockets” if crew members are in isolated areas, propellants, and formaldehyde (the latter 2 are specifically due to their high toxicity and use during spaceflight). NASA has also developed broad-spectrum trace volatile organic analyzers for air-quality monitoring aboard the ISS. In addition, a TOC analyzer has been flown to monitor water quality as we begin to recover potable water from urine.
NASA’s knowledge of the compounds present aboard spacecraft gives toxicologists the ability to calculate and predict which of these chemicals might become a toxic hazard. 29 The wide array of materials containing chemicals, including liquids, gases, and particulates, must be assessed in order for a toxicity hazard rating to be determined. In order to assess a known chemical risk and assign a toxicity level, the space toxicologist takes into consideration the route of exposure, exposure duration, the amount of singular chemicals present or chemical mixtures, and/or chemical by-products if necessary, along with physiological changes experienced by astronauts during spaceflight. 28 Collectively, the known chemical risks intrinsic to spaceflight drive the need for environmental monitoring. NASA currently uses a variety of environmental monitors to manage predictable risks of air and water pollutants in the ISS.
Predictable Versus Unpredictable
Toxicological risks in space environments are generally classified as either predictable or unpredictable. Predictable risks are controlled via air scrubbing, containment and materials selection. Regarding predictable risks, space toxicologists have the benefit of knowing the chemicals that are on board spacecraft and can make predictions of which chemicals may be accidentally released. Predictable risks are monitored in some cases, such as ammonia leaks and detection of combustion products. There are a substantial number of particulate and volatile contaminants that have been identified and quantified by NASA’s space toxicology laboratory. Other predictable sources of contaminants are crew member metabolism and materials off-gassing. Unpredictable risks require a broad approach to environmental monitoring that includes compounds that have no history of polluting spacecraft atmospheres.
NASA’s response to predictable, accidental release/releases is handled through flight rules. Vehicle propellants are monitored using a gold-salt method in the vehicle’s air lock, which is an isolated compartment that astronauts use to ingress/egress from the vehicle. By monitoring the airlock, we can identify whether the propellant has contaminated the EVA suits and is brought back into the vehicle. 30 If propellants are detected, NASA’s flight rules define required procedural steps for removing propellants. Ammonia, which is used as an external heat exchange fluid could get into the interior exchange loops and enter the vehicle atmosphere, yet its escape is remote due to several barriers in place. However, if this highly toxic chemical were to migrate from the external thermal loops into the cabin, it can be monitored in ISS. If ammonia were detected, NASA flight rules state the operational procedures, which are based on the detected levels identified by the ammonia monitor.
Toxic Chemical Identification Toxicity Labels in Space
Hazard classification depends on the inherent toxicity and amount of compound that could escape containment, assessing the containability if a chemical compound is released into the air and predicting the volume of dispersion after release. 31 All US hardware/payloads identified for toxicants prior to flight must contain a label so that the crew members and ground personnel can easily identify whether the payload contains any toxic hazards (see Figure 1 ).

Toxicity labels used on ISS and shuttle orbiter. Reprinted with permission fromNASA. ISS indicates International Space Station.
All crews are briefed prior to flight so that they can identify toxic hazard labeling and to educate them of experiments and systems containing known toxic hazards. An example of a typical crew preflight briefing of toxic hazards includes a chart (see Table 2 ) that outlines the apparatus on board the vehicle or space habitat, the chemicals of concern, and the expected toxic effects if a crew member is exposed. The chart can also be used to clarify some myths about chemical exposures, such as Freon 218.
Crew Preflight Briefing of Toxic Hazards Levels (THLs)
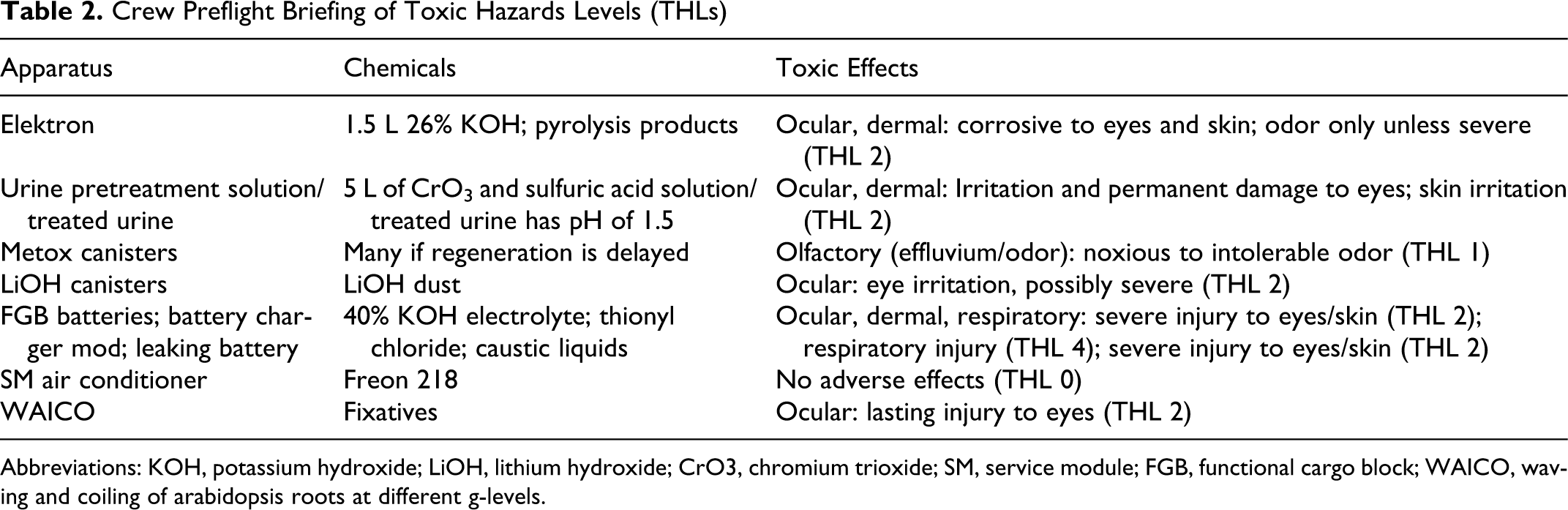
Abbreviations: KOH, potassium hydroxide; LiOH, lithium hydroxide; CrO3, chromium trioxide; SM, service module; FGB, functional cargo block; WAICO, waving and coiling of arabidopsis roots at different g-levels.
Space Toxicology Monitors
Volatile organic compounds may accrue gradually in spacecraft atmospheres or they can be suddenly released as a result of accidents. 28 Such unpredictable sources require a broad-spectrum capability to monitor volatile organic compounds at trace levels. NASA has developed and used various instruments to monitor selected combustion products since the early 1990s as well as instruments for selected high-risk pollutants including carbon dioxide, propellants, and formaldehyde. NASA has also generated broad-spectrum trace organic analyzers (VOA, gas chromatography/differential mobility spectrometry [GC-DMS]) for monitoring air quality aboard the ISS. To ensure the condition of water recovered from urine, a TOC analyzer (TOCA) is utilized on ISS to examine water quality. These instruments provide real-time and archival (returned to earth for analysis postmission) monitoring of chemical contaminants. The ISS has become an expansive development; therefore, analyzers are deployed in selected modules throughout the ISS to deal with any off-nominal events.
Grab sample canisters (GSCs) are used to monitor air quality at the crew’s first entry into a space vehicle/habitat as well as during nominal scheduled times, and contingency samples for use during off-nominal events. The GSC has 3 surrogate standards, the sample is aspirated by vacuum in <5 seconds and is analyzed in the laboratory by GC and GC/MS. Limitations of the GSC are that reactive compounds are lost. The old model (see Figure 2 ) had a valve, which if not sealed properly by crew members, would allow the sample to escape. This issue occurred on rare occasions, causing missed opportunities to determine spacecraft air quality. The new model (see Figure 2) eliminated the valve issue and takes up less space allowing for more samples to be taken. Formaldehyde badges (see Figure 3 ) are used for nominal sampling, these low-cost ($20 USD) peel and stick badges can be used to sample formaldehyde in an area or stuck to a crew member to sample personal exposures. Formaldehyde is trapped in badge matrix by diffusion. The typical sample time is 24 hours (in pairs). During ground-based laboratory analysis, formaldehyde is eluted from badge and analyzed by spectrophotometry. The badges limitation is that it must have sufficient face velocity of air flow.

Grab sample container (GSC) from left to right: original model, new model. Reprinted with permission from NASA.

Formaldehyde badge (FMK). Reprinted with permission from NASA.
Real-time monitors give the crew immediate knowledge of chemical constituents in air; some are broad-spectrum, while others target specific components. Due to the risk that is associated with combustion events such as fires or pyrolysis events, such as smoldering wiring, a device to analyze the chemical products produced during such events was needed. The compound specific analyzer for combustion products (CSA-CP) is a continuous monitor (see Figure 4 ) that uses electrochemical sensors to detect CO, HCN, HCl, and O2. It is strategically placed throughout ISS to deal with combustion events that could occur. 32 The continuous release of carbon dioxide due to human respiration drove the development of a monitor that can be used to test ambient areas as well as the buildup of CO2 in isolated areas. The carbon dioxide monitor (CDM) is a special-purpose infrared spectrometer for the detection of CO2 (see Figure 5 ). It has a 6% upper detection limit and an 18-hour battery life. The sample is pumped into the CDM which contains a water and particle filter and uses infrared absorption to measure CO2. 33 The VOA quantifies compounds by using a dual gas chromatograph/ion mobility spectrometer (see Figure 6 ). The VOA takes an in situ sample or remote sample by bag; it was used by NASA for 8 years aboard the ISS, but it is also very large and has a small dynamic range and no “unknown” chemical library. The GC-DMS is the next-generation replacement of the VOA for detection and quantification of select volatile organic compounds; it is much smaller (see Figure 7 ).

Compound-specific analyzer for combustion products (CSA-CP). Reprinted with permission from NASA.

Carbon dioxide monitor (CDM). Reprinted with permission from NASA.

Volatile organic analyzer (VOA). Reprinted with permission from NASA.

Gas chromatography/differential mobility spectrometry (GC-DMS). Reprinted with permission from NASA.
The TOCA measures conductivity, pH, total carbon, and total inorganic carbon (see Figure 8 ). Key parameter is the TOC. The TOCA uses small amount of hazardous reagents and TOC measurements confounded by addition of formate to some make-up water. 34 The replacement TOCA or TOCA 2 (see Figure 9 ) does not require the crew to add hazardous reagents and allows real-time screening of organic content in consumable potable water. Archival analysis of water is done via water sampling bag. This method has been used to detect highly toxic chemicals such as Cd, the source of which was traced back to a Cd spring.

Total organic carbon analyzer (TOCA). Reprinted with permission from NASA.

Next-generation total organic carbon analyzer (TOCA 2). Reprinted with permission from NASA.
Summary
Spaceflight toxicological risk is managed by controlling predictable risks and managing unpredictable risks with a suite of monitoring devices. Toxic hazard assessments depend on a chemical’s inherent toxicity, the releasable amount, volume of dispersion, and containability after release. On orbit analytical instruments must be small, reliable, and use minimal resources, and trace-contaminant toxicity must be considered a sum at the target organ level. Reclaimed water must be analyzed for toxicants, although NASA does not have that capability yet. As we move out of low Earth orbit and explore other celestial bodies, space toxicology risk assessment will include hazards from reactive, in situ dust and effective on-site monitoring. 35
Pulmonary Toxicity of Lunar Dust (Chiu-wing Lam, PhD)
As NASA plans to visit various planets and celestial bodies in our solar system, the concern of exposing space explorers to various types of ultrafine dusts, such as the highly reactive lunar dust, is considered a real issue. If NASA returns to the moon, one of the proposed plans outlines building an outpost on the lunar surface for long-duration human habitation and research. The Shackleton crater area of the lunar South Pole is the landing site of choice (see Figure 10 ). The crater lies entirely within the rim of the immense Aitken basin (~1500 miles in diameter), which is the largest and oldest impact basin on the moon. This basin is roughly 8 miles deep, and an exploration of its properties could provide useful information about the lunar interior. 36 The crater rim is illuminated by sunlight almost continuously; besides being subjected to smaller extreme temperature fluctuations than those that occur at the near-equatorial Apollo landing sites, the crater rim provides good access to solar energy. The interior of the crater is perpetually dark and very cold; any water that landed on the crater from cometary impacts would lie permanently frozen on or below the surface. An engineers' concept of a full lunar outpost is shown in Figure 11 . 37

Shackleton Crater at the lunar South Pole, the site of choice for building a habitat. Reprinted with permission from NASA.

Engineers' concept of full lunar outpost. Reprinted with permission from NASA.
Besides having occasionally been hit by large comets, the surface of the moon has consistently been bombarded by micrometeoroids for more than 4 billion years. The lunar surface lacks earth’s protective atmosphere, exposing it directly to repeated hypervelocity meteorite impacts. 36 Cooling welds the particles together into glassy and jagged-edged agglutinates, which are pulverized to fine dusts upon subsequent impacts. The texture of lunar soil evolves in a recurrent routine, which is in direct response to continual reworking by the micrometeorite flux. 36 In the course of lunar history, these meteoritic activities have created a relatively even particle-size distribution of the regolith over the whole lunar surface. The regolith contains about 10% to 20% fine dust with particle diameters less than 20 µm. 38 The lunar regolith resides in a near-vacuum environment and is constantly subjected to irradiation from solar ultraviolet light and x-rays in the daytime and solar wind at night; these solar radiations alternately impart positive and negative charges to the regolith. The surfaces of the charged lunar fine dust are expected to be populated with “unsatisfied” chemical bonds, making them very reactive. 39,40 While astronauts are living on the moon, as they go in and out of the habitat (including bringing instruments, hardware, and space suits in for servicing or refurbishing) they will bring dust, which is very adherent, into the living quarters of the lunar outpost. The potential for dust contamination of the lunar habitat can easily be inferred and visualized by examining Apollo 17 astronaut Jack Schmitt’s soiled suit (see Figure 12 ) and reading the Apollo crews' comments about exposure to lunar dust in the command modules during their return journeys to the earth. Respiratory tract irritation resulted from lunar dust exposure was reported by crew members of Apollo 12, 16, and 17 missions. 41

The space suit of Apollo 17 astronaut Dr Jack Schmitt shows it was contaminated with fine lunar dust. Reprinted with permission from NASA.
The lunar regolith is made up of minerals derived from anorthositic, gabbroic, and basaltic rocks that are also common in the earth's crust; aluminosilicate and ferromagnesian silicate minerals including plagioclase feldspar, pyroxenes, and olivine make up the bulk of the lunar regolith. 39 According to Dr Lawrence Taylor, a member of the NASA Lunar Geology Team, the samples of lunar surface soil collected during the Apollo program show that the regolith contains about 1% to 2% very fine dust (≤3 μm), which is respirable by humans; about 80% of the mineral in the fine-dust portion of lunar regolith is silica-rich glass. 42 The solar activities and micrometeoroid bombardments make the very fine surface dust potentially reactive. Fine reactive dust in the habitat can be expected to produce toxicity in the lung if it is inhaled and could pose a health risk to astronauts living on the moon. NASA has established a Lunar Airborne Dust Toxicity Assessment Group (LADTAG), which includes national experts in toxicology and lunar geology, to evaluate the risk of exposure to the airborne dust and to establish safe exposure limits for astronauts working in the lunar habitat; NASA has also directed its toxicology laboratory at the Johnson Space Center (JSC) to investigate the pulmonary toxicity of lunar dust in experimental animals to obtain the needed data. The NASA JSC Toxicology Laboratory has invited National Institute for Occupational Safety and Health and other academic institutes to participate in these important toxicity studies.
General Design of Experiments
Assessment of the pulmonary toxicity of a dust is generally done first in rodents by an intratracheal or intrapharyngeal instillation (ITI/IPI) study, in which the dust of interest can be compared with reference dusts of known toxicity. 43 The ITI/IPI study is then followed by an inhalation study. Lunar dust samples in our (ITI/IPI) study will be tested simultaneously with 2 common reference dusts, titanium dioxide (Rutile T-100, a product of Du Pont Company, was provided by Dr. David Warheit of Du Pont. R-100 is no longer commercially available; replaced with T-101) and crystalline silica (quartz or Min-U-Sil 5, a product of US Silica, was provided by Dr. Patti Erdely, a scientist of the National Institute for Occupational Health and Safety). The pulmonary toxicities of these 2 reference dusts are well characterized: titanium dioxide is low in toxicity while quartz is a fibrogenic dust that can produce a spectrum of lung lesions. The Occupational Safety and Health Administration (OSHA) and the American Conference of Governmental Industrial Hygienists (ACGIHs) have set occupational exposure limits (permissible exposure limit [PEL] and threshold limit value [TLV], respectively) on both dusts. The comparative toxicities of the test dusts in these instillation studies will be useful for LADTAG to establish limits for astronaut exposure to the lunar dust (see Figure 13 ).
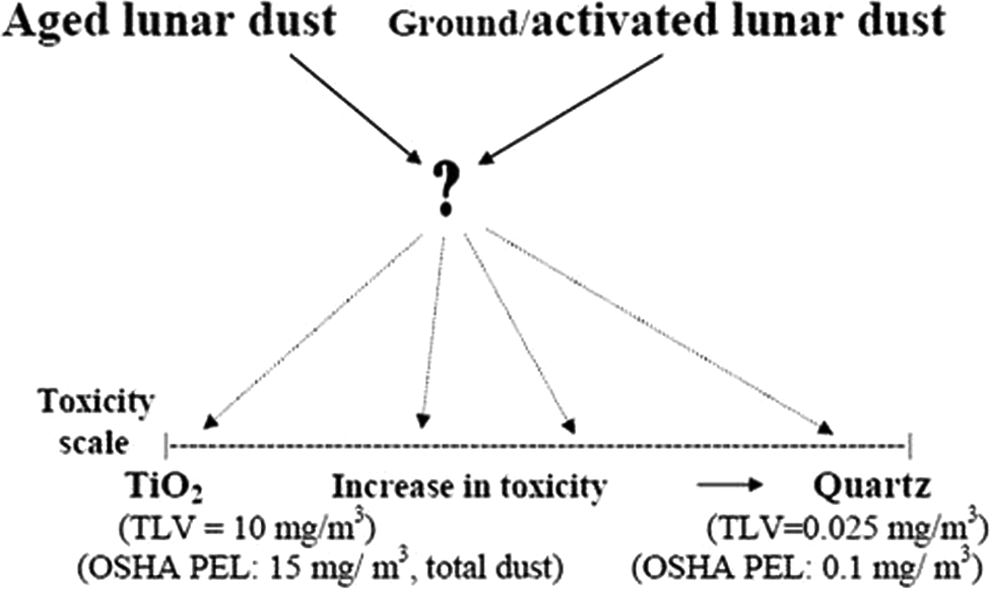
Comparative pulmonary toxicity study of lunar dust with reference dusts in rats by intratracheal instillation.
As discussed above, the dust particles on the lunar surface are subjected to cometary impact and are expected to remain chemically reactive in the high-vacuum lunar environment. 39,40 The lunar soil samples collected during the Apollo missions were exposed to air and moisture on their journeys to earth, and exposed to trace levels of oxygen and water molecules during their prolonged storage on earth. The NASA Geology Team believes that the Apollo lunar dust has been “chemically passivated” by these atmospheric components. 35 When freshly ground earth minerals of the types found on the lunar surface (olivine, augite, and labradorite) were exposed to water, hydrogen peroxide was formed in concentrations ranging from 0.9 to 25 nmol/m2 mineral. 44 The NASA Geology Team believes that grinding will “restore” the chemical reactivity of the passivated lunar dust. They will isolate respirable fractions (diameter <2 μm) of ground and unground lunar dust samples and provide them to the JSC Toxicology Group for evaluation of their pulmonary toxicity.
Intratracheal/Intrapharyngeal Instillation Studies of Lunar Dust
The ITI/IPI on lunar dust will be carried out in mice and rats. The bronchioalveolar lavage fluids (BALFs) will be obtained from rodents 7 and 30 days after the dust instillation to assess pulmonary inflammation and damage; variables to be assessed include lactate dehydrogenase activity, total protein and inflammatory cytokine concentrations, total cell counts, and cell differentials. Lung histopathology will be microscopically evaluated in rodents 1 and 3 months after the dust instillation. The lungs and lymph nodes will be examined for the presence of inflammation, necrosis, fibrosis, and other lesions.
Nose-Only Inhalation Studies of Lunar Dust
The data from these ITI/IPI studies could also be useful for determining the exposure concentrations for the inhalation toxicity study with lunar dust. From the ITI/IPI toxicity data, we could choose 3 exposure concentrations that would be likely to produce moderate, mild, and low/no effects in the lungs of exposed rats. Because of the limited quantity of lunar dust, the inhalation exposure will be carried out in nose-only exposure chambers. We are planning to carry out a 4-week inhalation exposure study. Battelle Northwest Group (Seattle, Washigton), after testing a Vilnius Dry Aerosol Generator (VAG), concluded that aerosolization of small quantities of dry powders with VAG is controllable, consistent, repeatable, and predictable. We set up 2 dust generation-exposure systems, each consisting of a VAG, a cyclone, and an NYU-Jaeger nose-only inhalation exposure chamber (CH Technologies, Westwood, New Jersey). We tested the performance of our 2 exposure systems using a simulated lunar dust (JSC-1Avf, a fine dust sample isolated from a volcanic ash and provided by Dr James Carter of the University of Texas at Dallas, Dallas, Texas). The concentration profile of dust in each chamber was monitored by a Cassella Microdust Pro Real-time Dust Analyzer (Casella USA, Amherst, New Hampshire); the dust in a known volume of chamber atmosphere was collected (1 L/min) continuously for 5 hours on filter paper for quantitative determination of the average dust concentration in the chamber. The aerodynamic diameter of the dust particles was determined by an Aerodynamic Particle Sizer Spectrometer 3321 (TSI Incorporated, Shoreview, Minnesota). Since the TSI 3321 could not give a mass median aerodynamic diameter (MMAD) of our test dust directly, the MMAD was estimated. We have acquired a quartz crystal microbalance (QCM) cascade impactor real-time air particle analyzer (California Measurements, Inc, Sierra Madre, California) that could be used to obtain real-time chamber concentration and aerodynamic particle size information. After further refining our exposure systems and acquiring another nose-only chamber for exposure of control animals, we will carry out a pilot study with rats exposed to the lunar dust simulant. A study with real lunar dust will then be carried out in rats exposed for 4 weeks (5 h/d, 5 d/week). Bronchioalveolar lavage fluids will be obtained from the rats after 7 and 30 days, while lung tissues will be harvested 1 and 3 months after the exposure for pulmonary toxicity assessment.
Surface Activation
Lunar geologists characterize the surface of pristine lunar dust as highly reactive. This surface reactivity is a result of exposure to solar wind bombardment and micrometeoroid impacts. These dusts are considered active on the surface of the moon and contain unsatisfied of “dangling” chemical bonds. Lunar dust also has a very large surface area due to its complex and highly unusual shapes (see Figure 14 ), the dust grains may include embedded glass as well as the presence of nanophase iron. Collectively, these properties of lunar dusts suggest that exposure to fresh or chemically active lunar dust could be more toxic than the aged or passivated dust encountered by the Apollo crews. NASA has investigated the toxicological properties of lunar dust simulants 45 and is in the process of testing authentic lunar dust. The problem is confounded by the fact that there are different types of lunar dust (mare and highland) and each of these types can be in various degrees of “maturity.” 28

Lunar dust grains: agglutinates and complex shapes. Second box from right, bottom row is the comparative weathered “round” dust found on earth.
Summary
We have conducted 2 IPI studies in C57/BL mice and have examined the toxicological parameters in BALFs and histopathology lesions in lungs and lymph nodes. The data analyses have not been completed and will be presented at a later date. After further refinement and a successful pilot rat study with lunar dust simulant, we will conduct an inhalation study with lunar dust in rats. The results of both the ITI and inhalation studies will provide toxicity data needed to assess the health risk of dust exposures on the moon and data for LADTAG to set safe exposure limits of lunar dust.
Conclusion
Space toxicology has a rich and fascinating specialty that has remained largely unknown by the greater toxicology community. Spaceflight presents unique challenges and requires new and innovative methods to support human life as exploration ventures past low Earth orbit into the various planets and other celestial bodies in our solar system. Exploring the unknown is at the core of scientific discovery and spaceflight toxicology is at the heart of sustaining the presence of human explorers in space.
Footnotes
Acknowledgments
We would like to thank the American College of Toxicology for inviting us to present our unique discipline at their annual meeting. A special recognition is given to the National Aeronautics and Space Administration for their continued support of our work. We also want to thank all of those interested in Space Toxicology.
The contributing speakers of this symposium were Drs Chiu-wing Lam, Rochelle Tyl, and Kenneth Thummel. Dr Noreen Khan-Mayberry organized this session. The symposium was chaired by Dr Noreen Khan-Mayberry, cochaired by Dr John T. James.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: the National Aeronautics and Space Administration.
