Abstract
Mexico City (MC) residents exposed to fine particulate matter and endotoxin exhibit inflammation of the olfactory bulb, substantia nigra, and vagus nerve. The goal of this study was to model these endpoints in mice and examine the neuroprotective effects of chocolate. Mice exposed to MC air received no treatment or oral dark chocolate and were compared to clean-air mice either untreated or treated intraperitoneally with endotoxin. Cyclooxygenase-2 (COX-2), interleukin 1 beta (IL-1β), and CD14 messenger RNA (mRNA) were quantified after 4, 8, and 16 months of exposure in target brain regions. After 16 months of exposure, the dorsal vagal complex (DVC) exhibited significant inflammation in endotoxin-treated and MC mice (COX-2 and IL-1β P < .001). Mexico City mice had olfactory bulb upregulation of CD14 (P = .002) and significant DVC imbalance in genes for antioxidant defenses, apoptosis, and neurodegeneration. These findings demonstrate sustained DVC inflammation in mice exposed to MC air, which is mitigated by chocolate administration.
Keywords
Introduction
Air pollution is a complex mixture of particulate matter (PM), gases, and organic compounds present in outdoor and indoor air. Children living in Mexico City (MC) exhibit chronic inflammation of the upper and lower respiratory tracts, accumulation of PM in nasal respiratory epithelium, breakdown of the nasal respiratory epithelial barrier, systemic inflammation, immunodysregulation, brain inflammation, cognitive and olfaction deficits, and brain magnetic resonance imaging (MRI) structural abnormalities. 1 -14 Adult MC residents exhibit upregulation of cyclooxygenase-2 (COX-2) in the olfactory bulb and other brain regions. 6 Children and young adults exhibit upregulation of COX-2, interleukin 1 beta (IL-1β), and the key innate immunity receptor CD14 in their olfactory bulbs, frontal cortex, substantia nigrae, and vagus nerves. 10 Accumulation of immunoreactive β-amyloid and α-synuclein, markers of oxidative stress and trafficking of inflammatory cells, were seen in highly exposed children and young adults, but not in matched residents from less polluted areas. 10 These findings suggest that exposure to severe urban air pollution causes chronic neuroinflammation, a condition that plays an important role in the etiology of neurodegenerative diseases such as Alzheimer disease and Parkinson disease (AD, PD). 14,15
Mexico City residents have been chronically exposed to significant concentrations of particulate matter air pollution for the last 2 decades. 16 –21 Particulate matter with aerodynamic diameters <2.5 μm (PM2.5) concentrations are above the current US National Ambient Air Quality Standards. Lipopolysaccharides (LPS)—a structural component of most gram-negative bacteria—detected in PM10 samples show a range of 15.3 to 20.6 nanograms per milligram of PM10, while Southwest Mexico City (SWMC) show the highest LPS concentrations at 59 EU/mg PM10. 17 –19 Particulate matter-associated LPS could reach the brain by uptake by the cranial nerves, such as the olfactory, trigeminal (oral, nasal) and vagus (respiratory, cardiovascular, and gastrointestinal systems) pathways. Other pathways shown in MC residents include trafficking of macrophage-like cells loaded with PM from the lung capillary bed to the systemic circulation and direct transfer of ultrafine particles (PM with aerodynamic diameters <100 nm) from the systemic circulation and/or red blood cells to brain endothelial cells. 1 –4,6,10,13,14
There is a significant body of literature recognizing that infections associated with intrapartum events, intrauterine infections or maternal infections including urinary tract infections, constitute an important risk factor for adverse outcome, including brain injury, in term and preterm infants. 22 Lipopolysaccharides is a key player in the brain injury described in infected infants and experimentally is associated with upregulation of inflammatory cytokines including IL-1β, tumor necrosis factor alpha (TNF-α), and IL-6 in models of intracisternal LPS administered to neonatal rats. 22 –24 Lipopolysaccharide-activated microglia play a deleterious role in a tissue culture model using rat oligodendrocyte progenitor cells. 25 Given the importance of endotoxin in infants, and the rodent literature showing that systemic LPS exposure causes chronic neuroinflammation and progressive neurodegeneration, 26,27 our objective in this work was to define brain target inflammatory regions in mice exposed to ambient MC air and in mice exposed to small intraperitoneal doses of LPS (IP-LPS).
This study included 4 treatment groups: 2 groups of clean-air-treated mice, with no further exposure, or with intraperitoneal LPS exposure. Two other groups of mice were exposed to MC air, with no further treatment, or receiving daily chocolate administration. The LPS mice were given 10 μL of IP-LPS (750 EU/mL) twice a week. The rationale of the selected dose was based on the MC literature measuring the concentrations of LPS associated with PM in SWMC. 17 –19 The IP route of administration was selected because it is a direct route to the brainstem (via the vagal nerve), a target area in our MC children and young adults. 10 Chocolate supplementation was given as a therapy that could ameliorate the inflammatory effects of air pollutant exposure. Dark chocolate was explored as an antioxidant dietary supplement 28 shown to improve cerebral blood flow and vascular health 29 –31 and cognitive performance. 32,33
We measured mRNA expression of 2 key inflammatory genes: cyclooxygenase-2 and interleukin-1β and the LPS receptor CD14 in target brain regions after 4, 8, and 16 months of the respective exposures. We also measured the expression of nuclear factor-κB (NF-κB), oxidative stress, antioxidant defense genes, and genes related to neurodegeneration in the dorsal vagal complex (DVC) after 16 months of exposure. Our selection of the DVC was based upon our previous observations of the significant vagus nerve inflammation present in SWMC children and young adults, compared to participants residing in clean air environments. 10
Our ultimate concern is the long-term effect of neuroinflammation caused by air pollutants on humans. The results of this work are relevant to people exposed to urban pollution and workers exposed to high endotoxin concentrations in the occupational setting.
Materials and Methods
Animals
Four- to five-week old female Balb-cJ mice were purchased from Taconic Laboratories (Germantown, New York). Animals were maintained under specific pathogen-free conditions. The protocol was approved by the Institutional Animal Care and Use Committee from both the University of Montana and the National Institute of Pediatrics (INP) in MC. At both facilities, mice were housed under Institutional Animal Care and Use Committee-approved conditions in a secured animal facility and were maintained at 20°C to 22°C on a 12-hour light–dark cycle with food (Lab Diet 5008) and water available ad libitum.
Clean-air mice were exposed to 100% fresh air distributed through a heating, ventilating, and air conditioning system (HVAC) and into housing rooms. The nonrecirculating air passed through a 35% coarse mechanical filter (macroenvironment of mice). Individual mouse cages (microenvironment) received room air that diffuses through a High efficiency particulate air (HEPA) filter in the mouse box lid. Mice housed in the animal facility in Southwest Mexico City were exposed 24/7 to the polluted air in an outdoor/indoor housing room. Animals were maintained under constant veterinary care and body weights were recorded each month. The chocolate used in this study was purchased from Chocolates D’Vicar, Coyoacán, Mexico City.
Experimental Design and Treatments
Figure 1 shows the experimental design and description of the treatment and control groups. A total of 106 mice were divided into 4 groups: (1) mice in the control group were exposed to clean air and received no additional treatment, (2) mice exposed to clean air and intraperitoneal injections of lyophilized E coli 0111:B4 (10 μL of 750 EU/mL, IP twice/week), (3) mice exposed 24/7 to SWMC atmosphere by direct exposure to the ambient air in an outdoor/indoor exposure room, (4) mice exposed 24/7 to the SWMC atmosphere and also treated with oral dark chocolate (60% cocoa solids, 9.5 mg polyphenols) 2 g 3 times per week. Our selection of the chocolate dose was made on the basis of a small pilot mice 4-month study where we tested 2 different doses, the one selected in this study and a higher dose (4 g/19 mg polyphenols/3 times per week) and followed weekly body weights and COX-2 protective responses in the DVC. On the selected dose, body weight was not different from the untreated animals and we were able to show a protective DVC response based on COX-2 results (data not shown). Mice in groups 1 and 2 were kept in the animal facility at the University of Montana, while mice in groups 3 and 4 were at the SWMC animal facility. In all, 6 to 12 animals were sacrificed per group by decapitation after 4, 8, or 16 months of the respective treatments. Euthanasia was conducted in accordance with established guidelines and applicable animal care-and-use regulations.
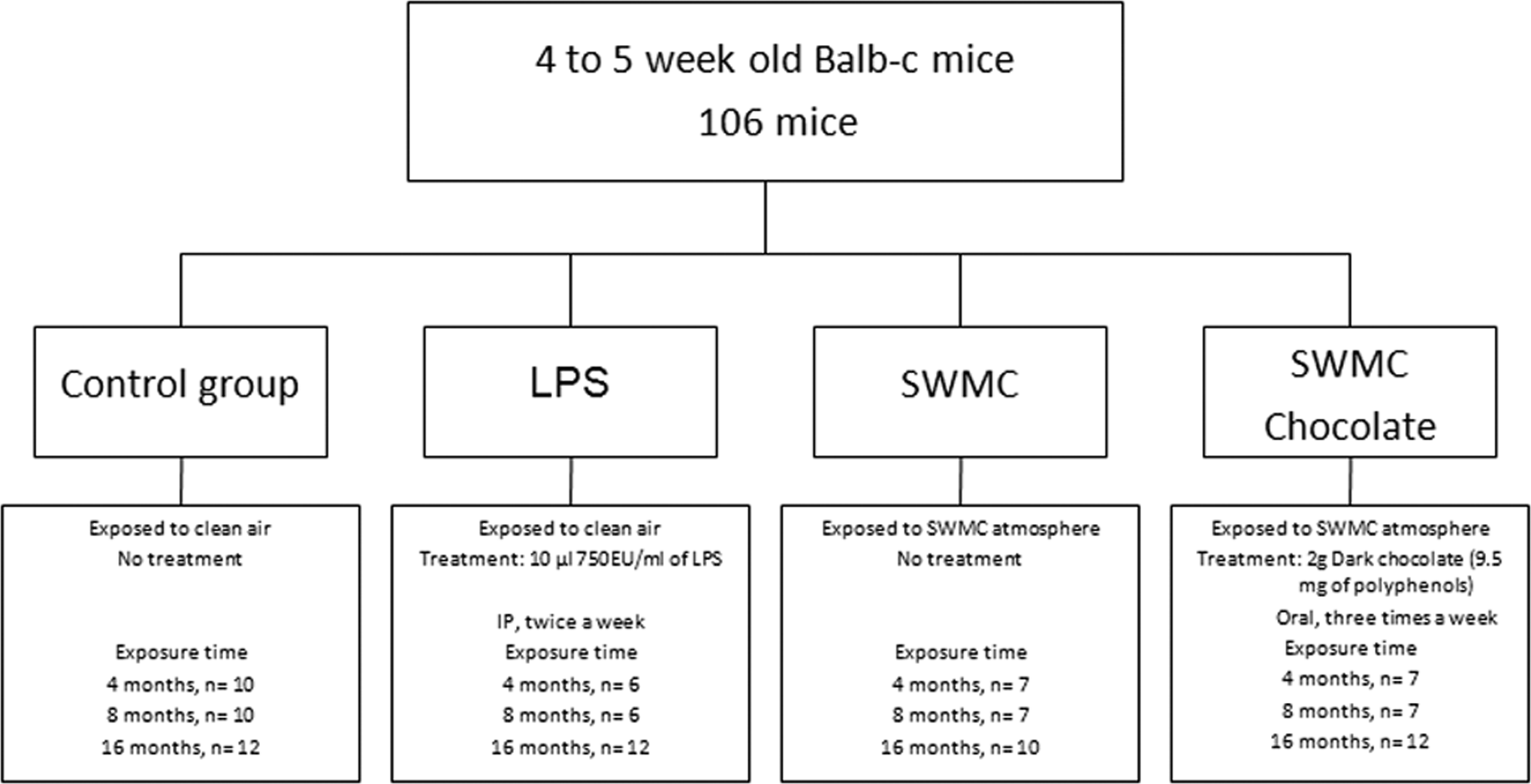
This figure shows the mice experimental design and description of the treatment and control groups. Mice were killed at 4, 8, and 16 months of treatment.
Air Quality Data
Mexico City represents an extreme of urban growth and environmental pollution. 16,20 Lying in an elevated basin at an altitude of around 2240 meters above sea level, the city covers an area of approximately 2000 km2 and is surrounded by a series of volcanic and discontinuous mountain ranges that limit the natural ventilation of the basin. The population grew from fewer than 3 million in 1950 to over 18 million in 2000. As a consequence of the increase in population and the associated industrialization, the basin has more than 30 000 industrial facilities and 4 million vehicles with an estimated annual emission of 2.6 million tons of particulate and gaseous air pollutants. 16 Significant sources of environmental endotoxin 34,35 are present in MC, including daily outdoor deposits of 500 metric tons of animal and human fecal material, 17 –19,36 and its residents have been exposed annually to PM2.5 concentrations above the current annual standards (15 μg/m3 annually) for the last two decades.
Brain Dissection
Brains were frozen and dissected under a microscope immediately after death and the following areas were selected for the Real time polymerase chain reaction (RT-PCR) studies: olfactory bulbs, frontal gray and white matter, full hippocampus, substantia nigrae compacta, lateral and reticular parts, the DVC including the area postrema, the nucleus of the solitary tract, the dorsal motor nucleus of the vagus (DMNV), and trigeminal sensory dorsomedial and ventrolateral nuclei regions bilaterally.
Estimation of mRNA Abundance by Real-Time RT-PCR
Total RNA was extracted from frozen brain samples using Trizol Reagent (InVitrogen Corp, Carlsbad California) according to the manufacturer’s instructions. Random-primed first-strand complementary DNAs (cDNAs) were generated using the SuperScript III First-Strand Synthesis System (InVitrogen, cat# 18080-051). Relative abundances of mRNAs encoding COX-2, IL-1β, and CD14 were estimated by quantitative fluorogenic 5′ nuclease (TaqMan) assay of the first strand cDNAs as described. 6 Primers and fluorophore-labeled TaqMan probes targeting mouse COX-2 (assay Mm00478374-m1), IL-1β (assay Mm00434228-m1), CD14 (assay Mm00438094-g1), and Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (assay Mm99999915-g1) were obtained from Applied Biosystems (Foster City, California) and used according to the manufacturers instructions. The amount of COX-2, IL-1β, and CD14 cDNA in each sample was normalized to the amount of GAPDH cDNA yielding an index: molecules of the gene of interest/attomols of GAPDH.
Polymerase Chain Reaction Arrays
Microarray analysis was conducted with SABiosciences (Frederick, Maryland) polymerase chain reaction (PCR) arrays: oxidative stress and antioxidant defense (PAMM-065), NF-κB signaling pathway (PAMM-025), and the AD array (PAMM-057) following the manufacturer’s instructions using 1 μg of total RNA per PCR array. We pooled the RNA from each sample inside each group and cDNA was synthesized using the C-03 first strand kit (SABiosciences). Relative gene expression was normalized to 5 housekeeping genes (GUSB, HPRT1, HSP90AB1, GAPDH, and ACTB) in each PCR array plate. The fold change for each gene was calculated as 2(ΔΔCt) and shown as upregulated if expression was greater than 2 or downregulated if expression was less than −2.
Statistics
Baseline characteristics of the study groups were compared with the Student t test. A 2-factor fixed effect model was built for each of the genes for comparing different study groups. Two fixed factors were considered: group and age. Each factor had 3 levels: Control, SW, and LPS, while factor age included the age at the time of sacrifice at 5, 9, and 17 months, after 4, 8, and 16 months of treatment, respectively. Since there was only 1 age level when comparing SW and SWCh, a 1-factor fixed effect model was applied. Mixed Model Procedure in SAS 9.2 was used to build the model. Significance was assumed at P < .05. Data are expressed as mean values ± SD.
Results
Air Quality Data
We had access to the atmospheric monitoring data for the mice study period June 2006 through November 2007 from MC monitoring stations <5 miles from the SW animal facility: Pedregal and Coyoacan. Table 1 shows the analysis of the PM2.5 data for the SWMC study area in the period of interest. Figure 2A shows the average concentrations of particulate matter air pollution 10 µm or less in aerodynamic diameter (PM10) recorded in MC from 1995 to 2008. Figure 2B illustrates the concentrations of PM2.5 recorded in MC for the years 2004-2008 including the annual average and the comparison with the air quality standard for PM2.5 (15.0 µg/m3 annual arithmetic mean and 35 µg/m3 24-hour). Mexico City residents and mice in this study have been exposed to concentrations of PM2.5 above the standards ever since PM2.5 monitoring started in 2003.
Summary of the 24-Hours Average PM2.5 Concentration Data for the SWMC Region for the Duration of the Mice Exposures: June 2006 to November 2007

Abbreviation: SWMC, Southwest Mexico City.
a The US EPA 24-hours PM2.5 average air quality standard is 35 μg/m3.
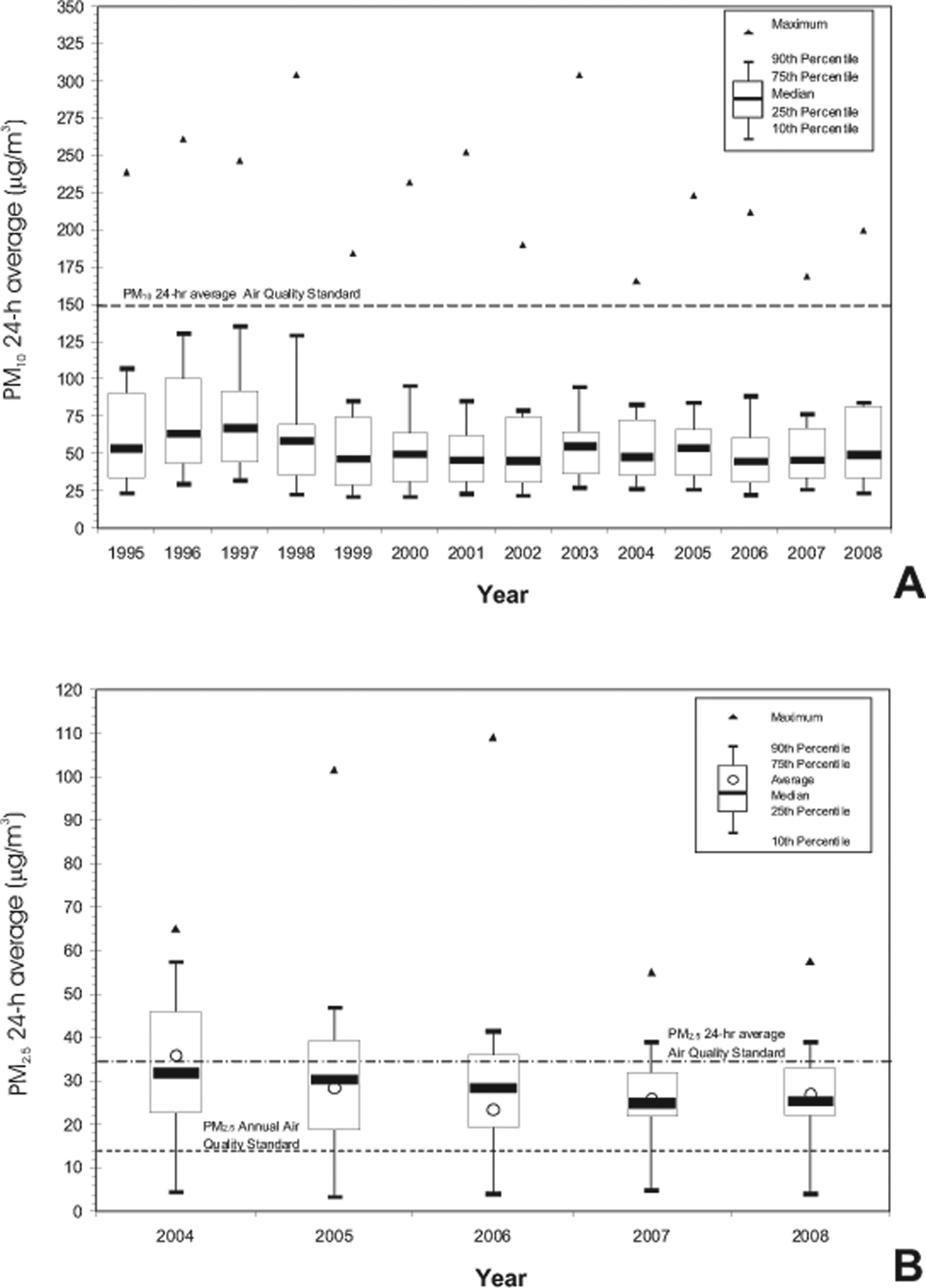
A, The average concentrations of particulate matter air pollution 10 µm or less in aerodynamic diameter (PM10) recorded in Mexico City (MC) from 1995 to 2008. Concentrations of PM10 have been under the air quality standard (150 µg/m3 not to be exceeded more than once per year) for the last 13 years. Historically, Southwest MC PM10 samples have shown the highest endotoxin concentrations at 59 EU/mg PM10. 17 –19 B, The concentrations of PM2.5 recorded in Mexico City (MC) for the years 2004-2008 including the annual average and the comparison with the air quality standard for PM2.5 (15.0 µg/m3 annual arithmetic mean and 35 µg/m3 24-hour average). Residents of MC have been exposed to concentrations of PM2.5 above the standards year after year.
Endotoxin Content in PM10 Samples Collected from South Mexico City
Although concentrations of PM10 have been under the air quality standard (150 μg/m3 not to be exceeded more than once per year) for the last 13 years, South Mexico City PM10 samples have repeatedly shown the highest endotoxin concentrations in the city. 17 –19 Data from Bonner and coworkers 17 showed South Mexico City values in the range from 15.3 to 20.6 nanograms of LPS per milligram of PM10, while more recent data from Rosas-Pérez group 19 showed a range of 0.005 to 0.034 μg mg−1 LPS-PM10. Thus, historically, South Mexico City exhibits significant concentrations of endotoxins associated with PM10. 17 –19
Cyclooxygenase-2, IL-1β, and CD14 mRNA Expression
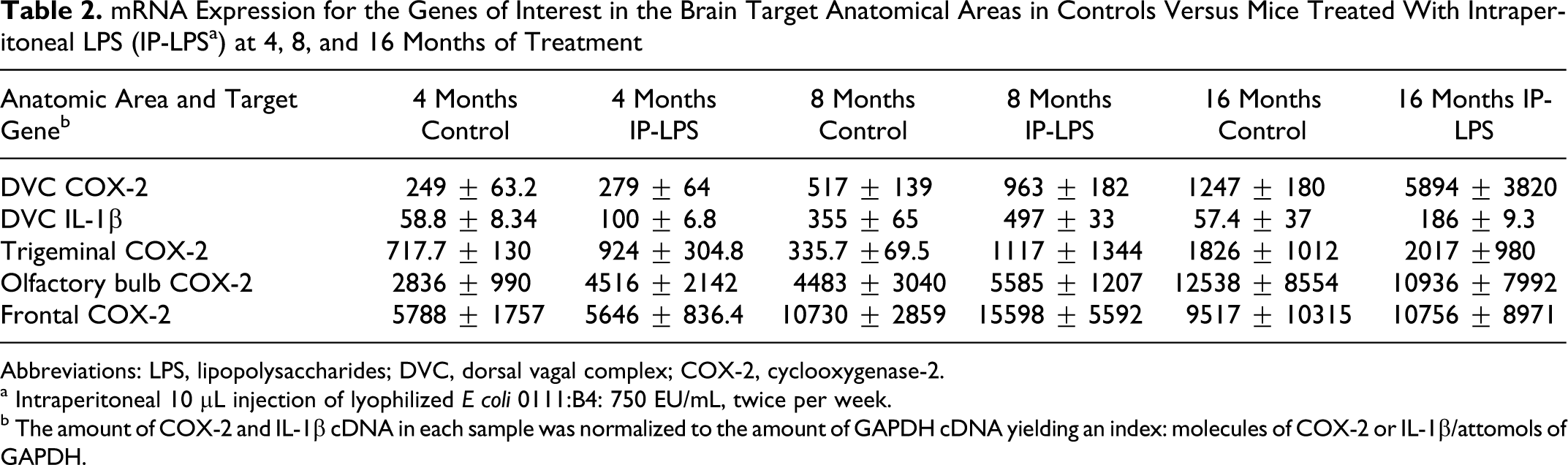
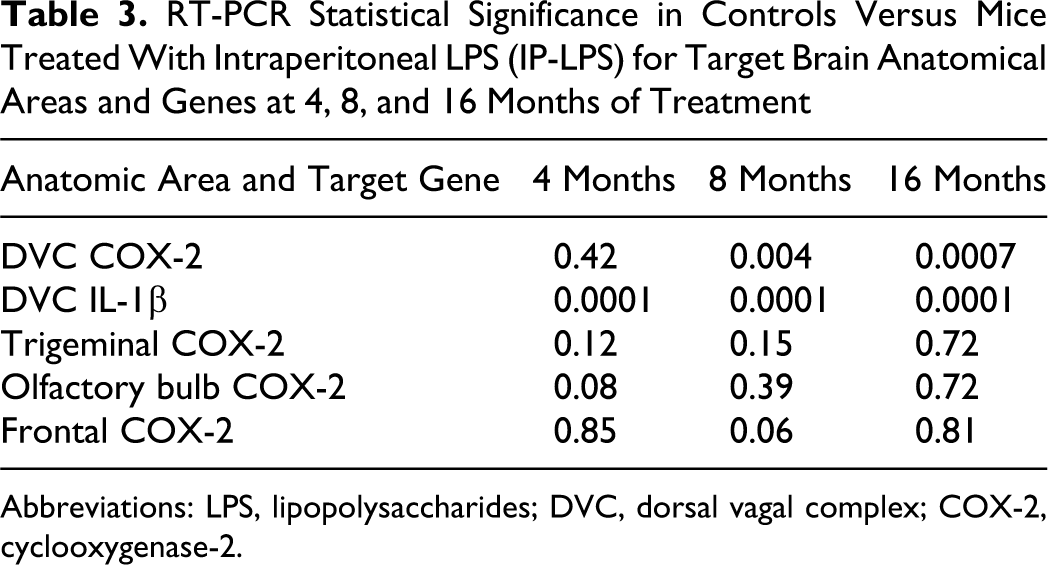
Real-time, rapid-cycle PCR analysis of COX-2, IL-1β, and CD14 mRNAs in the selected brain regions indicated that the corresponding mRNA was present in each of the samples analyzed. Group 2 mice (clean air plus IP-LPS) showed significant upregulation of IL-1β after 4 months of treatment in the DVC (Tables 2 and 3). After 8 months of treatment, the clean air IP-LPS mice exhibited significant upregulation of both COX-2 and IL-1β (P = .0001), and after 16 months of exposure, the DVC exhibited significant upregulation of both COX-2 and IL-1β (P =0.0001; Tables 2 and 3).
mRNA Expression for the Genes of Interest in the Brain Target Anatomical Areas in Controls Versus Mice Treated With Intraperitoneal LPS (IP-LPS a ) at 4, 8, and 16 Months of Treatment
Abbreviations: LPS, lipopolysaccharides; DVC, dorsal vagal complex; COX-2, cyclooxygenase-2.
a Intraperitoneal 10 μL injection of lyophilized E coli 0111:B4: 750 EU/mL, twice per week.
b The amount of COX-2 and IL-1β cDNA in each sample was normalized to the amount of GAPDH cDNA yielding an index: molecules of COX-2 or IL-1β/attomols of GAPDH.
RT-PCR Statistical Significance in Controls Versus Mice Treated With Intraperitoneal LPS (IP-LPS) for Target Brain Anatomical Areas and Genes at 4, 8, and 16 Months of Treatment
Abbreviations: LPS, lipopolysaccharides; DVC, dorsal vagal complex; COX-2, cyclooxygenase-2.
Group 3 mice (MC air) displayed upregulation of COX-2 and IL-1β predominantly in the DVC and the olfactory bulb (Table 4 , Figure 3 ). Upregulation of CD14 was detected in the hippocampus after 5 months of treatment (P = .01), after 8 months of treatment (P = .03), and in the olfactory bulb after 16 months of treatment (P = .002). The hippocampus exhibited upregulation of IL-1β after 4 months of treatment (P = .004) and 8 months of treatment (P = .02), but no significant difference when compared with the controls after 16 months of treatment. The DVC showed upregulation of COX-2 after 4 months of treatment; the upregulation persisted through the 8-month timepoint (P = .02) and was very significant after 16 months of treatment (P = .001). Upregulation of IL-1β in the DVC was significant after 8 and 16 months of treatment (P = .01 and P = .0003, respectively). IL-1β and CD14 were significantly upregulated in the olfactory bulb (P = .001 and P = .002, respectively) after 16 months of treatment, as expected based on the historically high concentrations of PM-LPS in the SWMC atmosphere. The substantia nigra exhibited upregulation of both COX-2 (P = .03) and IL-1β (P = .02) after 16 months of treatment (Table 4).
mRNA Expression for the Genes of Interest in the Brain Target Anatomical Areas in Controls Versus Mexico City-Exposed Mice at 4, 8, and 16 Months of Treatment
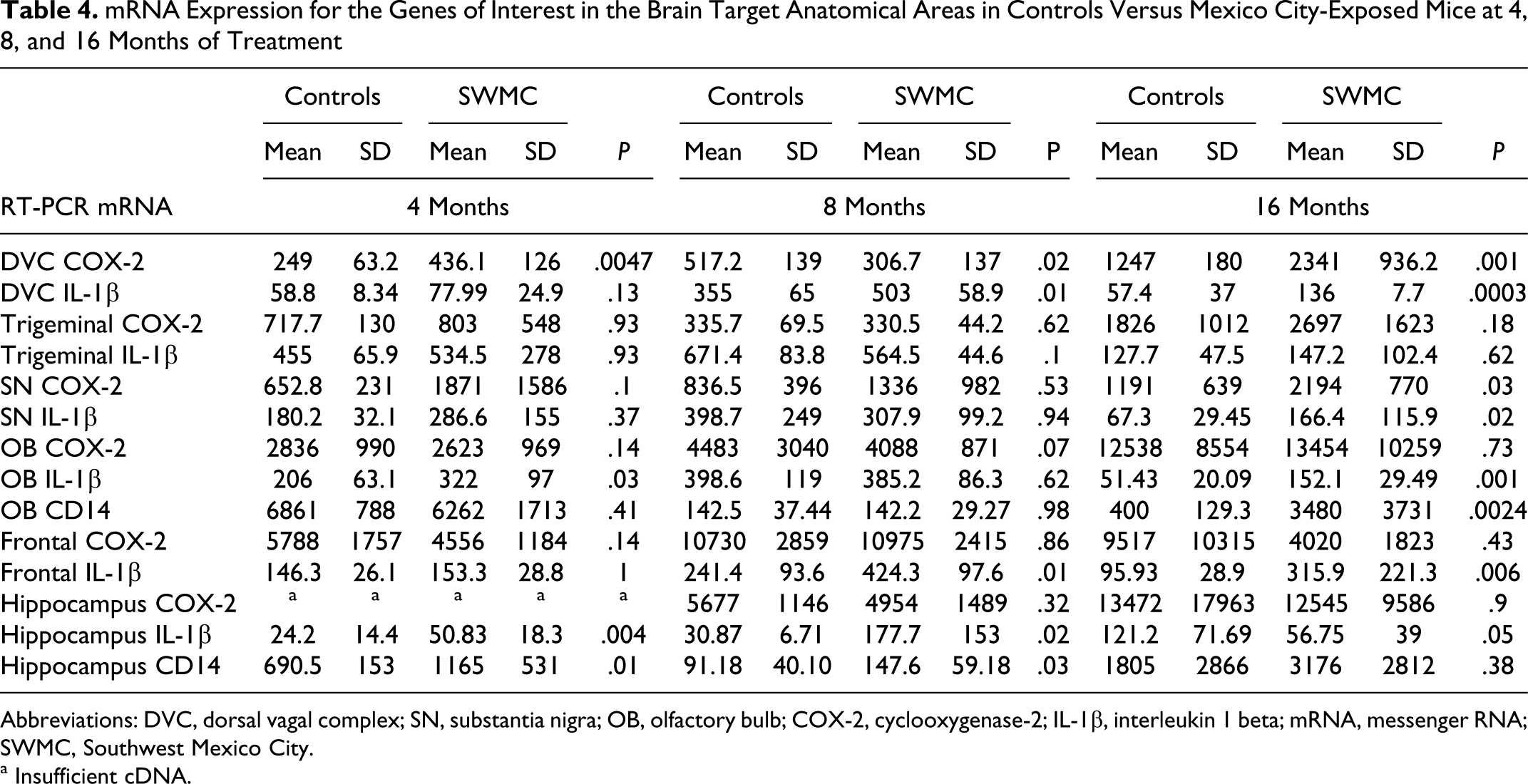
Abbreviations: DVC, dorsal vagal complex; SN, substantia nigra; OB, olfactory bulb; COX-2, cyclooxygenase-2; IL-1β, interleukin 1 beta; mRNA, messenger RNA; SWMC, Southwest Mexico City.
a Insufficient cDNA.
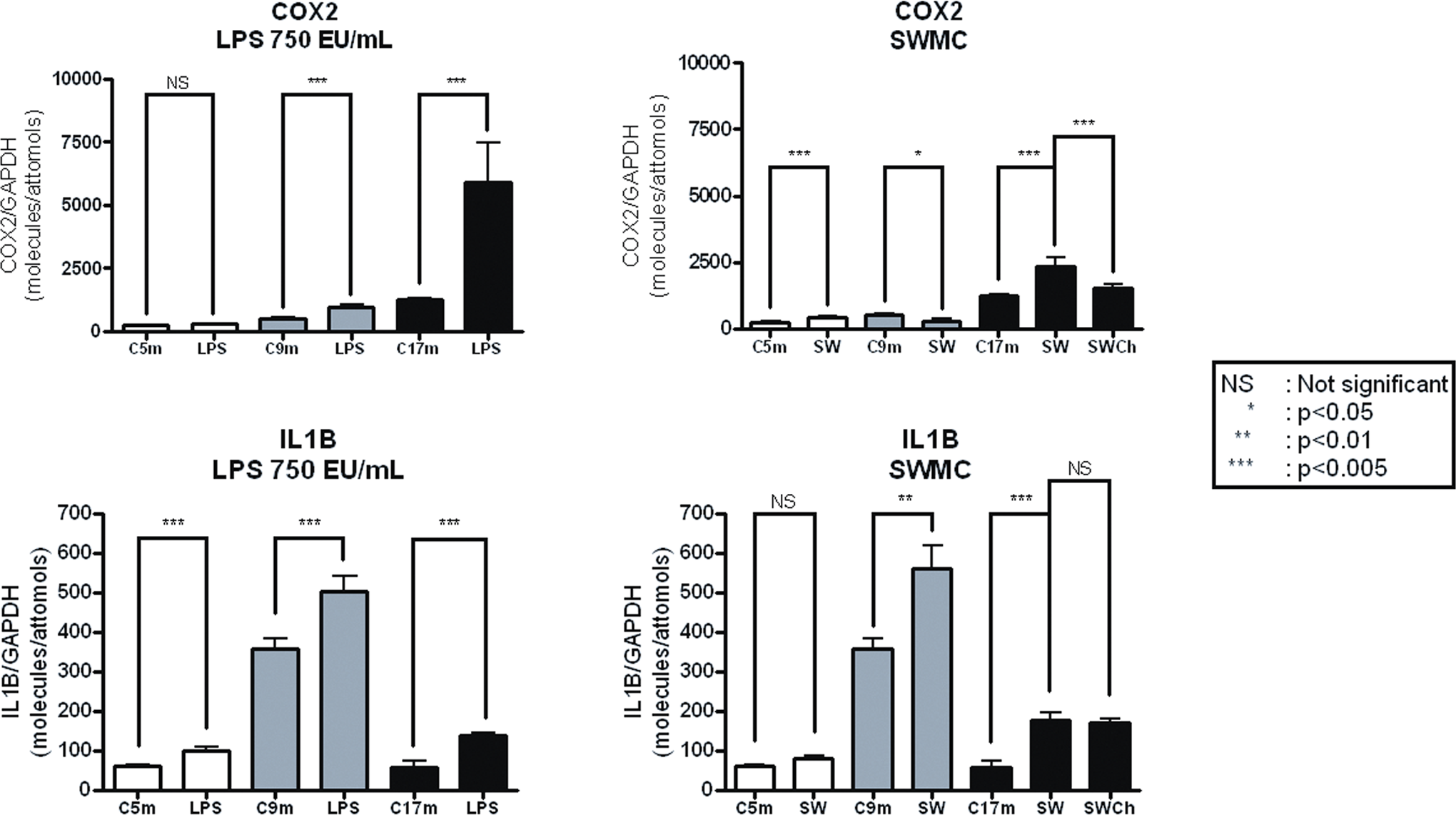
Dorsal vagal complex statistical significance of the cyclooxygenase-2 (COX-2) and interleukin 1 beta (IL-1β) RT-PCR results at 4, 8, and 16 months of treatment. Lyophilized E coli was administered intraperitoneally, 10 µL of 750 EU/mL, 2 times/week (mice group labeled LPS 750 EU/mL). Lipopolysaccharides (LPS)-treated mice and those exposed to the atmosphere in Southwest MC were compared versus Controls exposed to clean air and receiving no treatment. Southwest Mexico City (SWMC) chocolate-treated mice are labeled SWCh. The amount of COX-2 and IL-1β cDNA in each sample was normalized to the amount of GAPDH complementary DNAs (cDNA) yielding an index: molecules of the gene of interest/attomols of GAPDH.
Orally administered chocolate in the SWMC-exposed mice (group 4) resulted in a significant downregulation of IL-1β and CD14 in the hippocampus in the mice exposed to SWMC air P = .007 and 0.04, respectively, compared to mice exposed to SWMC air only. Mice exposed to MC air and treated with chocolate for 16 months showed a significant downregulation of COX-2 in the DVC compared to MC-untreated mice (P < .005; Figure 3). Chocolate treatment did not change expression of IL-1β in the DVC after 16 months of treatment (P = .74; Figure 3).
The results of the 2-factor fixed effect model built for COX-2 and IL-1β in the DVC showed for COX-2 a significant interaction effect between group and age based on the P value (.0001) obtained from the F-test. For comparing control versus LPS, F-test shows that there was a significant difference between the 2 groups since the P value = .0002, while comparing SW versus SWCh, F-test showed also a significant difference between the 2 groups P value = .029. Results for IL-1β showed a significant interaction effect between group and age effects, based on the P value (.0267). For comparing control versus LPS, F test showed a significant difference between the 2 groups since the P value = .0003. Comparing control versus SW, the F test also showed a significant difference between the 2 groups since the P value < .0001, while no significant difference was seen between SW versus SWCh, P value = .74.
Polymerase Chain Reaction Arrays of the DVC After 16 Months of Exposure in Clean-Air Mice Versus SWMC-Exposed Mice Without and With Chocolate Treatment
Signaling pathways related to NF-κB, AD-related genes, and oxidative stress and antioxidant defense genes were explored in the DVC of mice exposed for 16 months. Of utmost interest in the results from SWMC mice versus clean air-exposed mice in the NF-κB array was the upregulation of genes falling within the categories of inflammatory responses (Il-1α), apoptosis-related genes (Casp8, Casp3, Tnsf10, and Ripk2), critical genes regulators of cell survival, proliferation, and activation of the NF-κB transcription factor (Bcl3, Bcl10, and Map3k1), and genes widely recognized as injury-related cytokines, especially those associated with neuron-glia interactions during astrocyte differentiation and activation in response to brain injury (Tgfbr1; Table 5 ). In the AD array, mice exposed to SWMC air had an increase in Chat, the gene encoding an enzyme that catalyzes the biosynthesis of acetylcholine, and Aph1, a multipass transmembrane protein that interacts with presenilin and nicastrin as a functional component of the γ-secretase complex. Downregulated genes in the SWMC mice included acetylcholinesterase and synuclein-β (which inhibit the aggregation of α-synuclein, a protein involved in PD) and lipid homeostasis genes involved in amyloid precursor protein (APP) metabolism and cellular recognition and internalization of low-density lipoprotein receptor lipoproteins (Lrp1 and Lpr8). In the oxidative array, most notable was the increased expression of Sod2, a key antioxidant superoxide dismutase, along with other redox enzymes such as lactoperoxidase, and neuroglobin (Ngb), involved in increasing oxygen availability and providing protection under hypoxic/ischemic conditions.
A Selection of Genes Up and Down Regulated in the Dorsal Vagal Complex in Control Versus SW Mexico City Untreated Mice After 16 Months of Exposure

Results in NF-κB array comparing SWMC air-exposed mice to those exposed to SWMC air and treated with chocolate (Table 6 ) showed a significant upregulation of apoptotic and antiapoptotic-related genes including Tradd, Casp1, and Cflar. Downregulated genes included Raf1, involved in the transduction of mitogenic signals from the cell membrane to the nucleus. In the AD array, chocolate-treated SWMC air-exposed mice had an increase in Ache, Ctsd, Sncb, Psen2, Lrp1, Gja1, and Insr. Bace1, responsible for the proteolytic processing of APP, was downregulated. In the oxidative array, most notable was the increased expression in chocolate-treated mice of glutathione peroxidase 1 (Gpx1), peroxiredoxin (Prdx4), and sulfiredoxin 1 (Srxn1), important antioxidant enzymes contributors to oxidative stress resistance, and sensors for oxidative stress. Nucleotide excision repair (Ercc2) and dynamin 2 (Dnm2), involved in a nucleotide excision repair pathway and vesicular trafficking processes, were also upregulated. Downregulated genes included steroyl-CoA desaturase 1 (Scd1) and cathepsin B (Ctsb; Table 6).
A Selection of Genes Upregulated and Downregulated in the Dorsal Vagal Complex From Southwest Mexico City Untreated Versus Mice Treated With Dark Chocolate
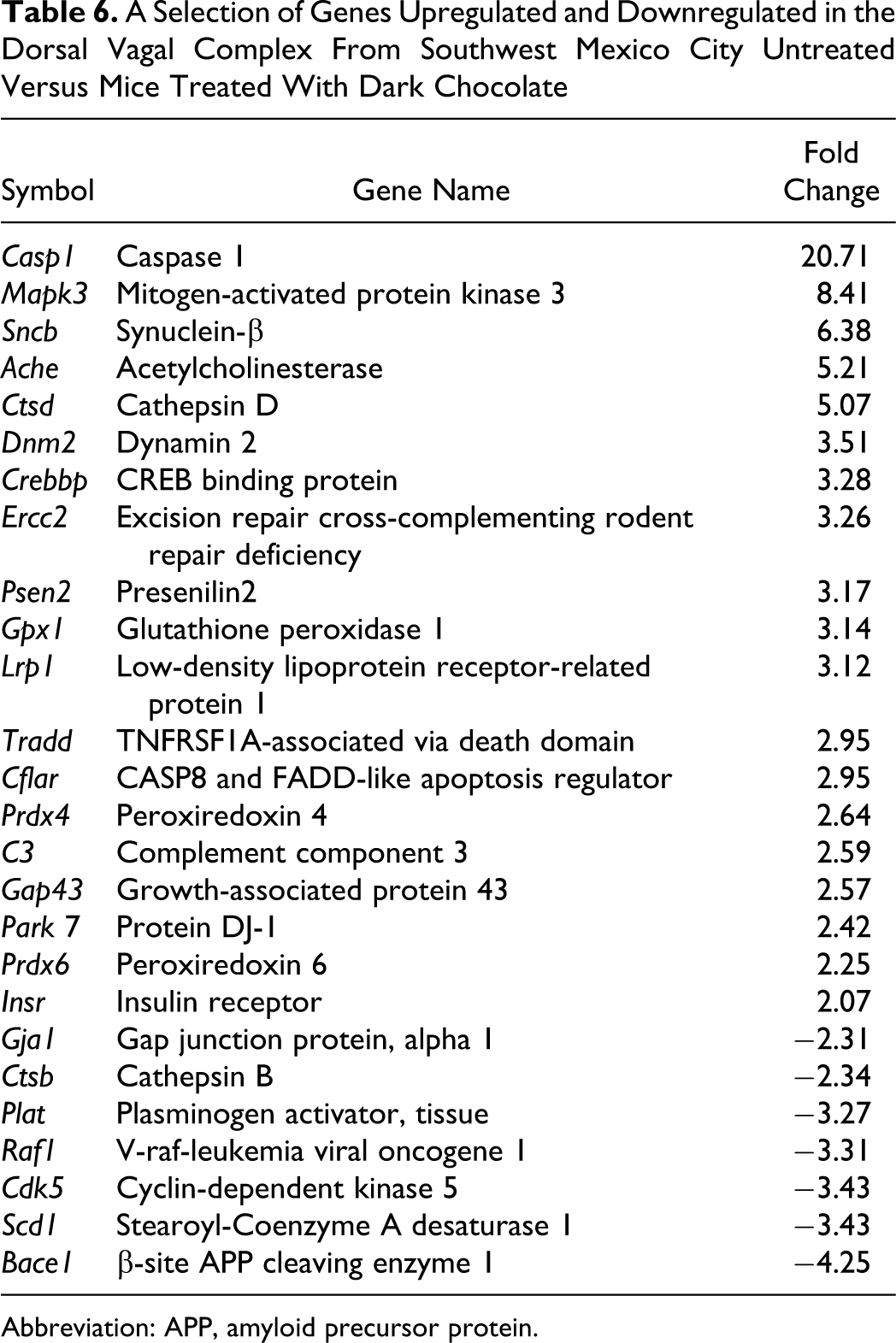
Abbreviation: APP, amyloid precursor protein.
Discussion
The DVC and the olfactory bulb are critical targets of sustained exposures to air pollutants. Our data show that 16 months of exposure to an urban environment with high concentrations of fine particulate matter and endotoxin result in a significant olfactory bulb upregulation of IL-1β and CD14 and of COX-2 and IL-1β in the DVC. Cyclooxygenase-2 and IL-1β in the DVC exhibited a significant interaction effect between group and age. The DVC of mice exposed to the MC atmosphere for 16 months had significant changes in genes critical for inflammatory, apoptotic, oxidant, and antioxidant responses when compared to clean air controls. Low intraperitoneal doses of LPS elicited a similar DVC inflammatory response suggesting that endotoxin is a key player. Chronic oral administration of dark chocolate gave an overall antioxidant and neuroprotective response.
The inflammatory findings in the DVC are important in the context of air pollution for several reasons: First, the DVC has pivotal connections between the central nervous system (CNS) and peripheral viscera and the area postrema, which lacks a blood-brain-barrier, thus playing a role in controlling the entry of blood- or cerebrospinal fluid (CSF)-borne substances to DVC neurons. 37 Components of air pollution, specifically ultrafine and fine PM, could have direct access to the DVC and/or could travel transynaptically in the trigeminal and vagus nerves and reach the medulla. 38,39 The transneural transport has been clearly shown with herpes virus where the virus is identified in vagal preganglionic neurons of the DMNV, nucleus ambiguous, and reticular formation after cardiac viral injections. 38 Secondly, we have reported α-synuclein immunoreactivity, microglia activation, and oxidative stress markers in the DMNV and olfactory bulb autopsy samples of children and young adults exposed to the same atmosphere as the mice in this study. 10,13 Critical for the exposed MC residents, both anatomical targets in mice: the DMNV and the anterior olfactory nucleus and bulb are involved early in the preclinical stages of PD. 40
This study demonstrates a distinctive pattern of COX-2 and IL-1β responses in the DVC of SWMC air-exposed and clean-air plus IP-LPS-treated mice. As expected, given the IP route of exposure, the main target in the IP-LPS mice was the DVC, while the targets in the SWMC air-exposed mice included the olfactory bulbs and the DVC, likely related to the vagus innervation of the exposed respiratory, cardiac, and digestive systems. Cyclooxygenase-2 upregulation in olfactory bulb, substantia nigra, and vagus nerves has been consistently documented in MC residents (including humans and dogs) when compared to clean air-matched controls. 3,6,10,11,13 Cyclooxygenase-2 is increased in a variety of inflammatory states and arachidonic acid metabolites, reactive oxygen and nitrogen species contribute to detrimental effects. 41 –43 Endothelial and perivascular microglia cells express COX-2 after IP-LPS or IL-1β exposure, and strong induction of COX-2 has been shown after peripheral administration of LPS, IL-1β, or TNF-α. 42,44 –48 Cyclooxygenase-2 has been shown to have a critical role in both the pathogenesis and selectivity of the PD neurodegenerative process. 49,50 In the context of air pollution, several potential pathways could induce a DVC COX-2 response, including (1) increases in circulating IL-1β, TNF-α, and IL-6 (ie, systemic inflammation); (2) activation of chemosensitive afferent fibers of the vagus in the respiratory, cardiovascular, and gastrointestinal tracts; 45,51,52 (3) transneuronal transport of particles from the thoracic and abdominal cavity to the medulla via the vagus nerve 10,53 ; and (4) the lack of a BBB in the area postrema, 22,37,53 potentially allowing the direct contact with ultrafine PM.
Interleukin 1β is of the most potent proinflammatory cytokines produced in abundance by activated microglia. We found that IL-1β reached its higher expression after 16 months of exposure to SWMC air and was also highly upregulated from the 4-month time point in IP-LPS mice. Although there are some reports of IL-1β neuroprotection, 54,55 most studies suggest that sustained IL-1β signaling is detrimental to the CNS. 56 –61 The upregulation of IL-1β coincides with the increased expression of initiator and executioner caspases and toll-like receptors suggesting that inflammasome activation is involved in the urban mice inflammatory DVC responses. 62 The innate immune system rapidly detects invading pathogenic microbes and eliminates them. In the case of MC residents (mice, dogs, and humans), the particles with LPS are likely detected as foreign and an inflammatory response is launched with the inflammatory detrimental effects. More importantly, the findings in mice exposed to SWMC air suggests that the DVC is capable of sensing microbial danger signals and metabolic stress, resulting in the proteolytic activation of the proinflammatory cytokine IL-1β. 62,63 Proinflammatory cytokines including IL-1β, IL-6, TNF-α, and interferon γ disrupt intracellular patterns of PD proteins such as α-synuclein, possibly representing a mechanism of cell dysfunction in PD. 64 Thus, the sustained upregulation of IL-1β in the DVC of mice exposed to SWMC air is highly relevant to our findings of immunoreactive α-synuclein and microglia activation in the DVC and the substantia nigrae of MC adolescents and young adults. 10 Heart rhythm alterations, decreased cardiac vagal tone, and autonomic dysfunction are significant in MC children, 7 while olfaction deficits are significantly higher in young adult residents of MC compared to low air pollution-exposed, age-matched controls. 13
In the untreated urban mice, upregulation of antioxidant genes and downregulation of potentially harmful genes support the view that healthy mice under severe exposure conditions evolve defensive mechanisms aimed to protect vulnerable regions and avoid detrimental consequences for the neural tissue. However, in spite of these defense mechanisms, untreated urban mice had significant DVC upregulation of COX-2 and IL-1β along with increased expression of critical genes regulators of proliferation and activation of the NF-κB transcription factor and injury-related cytokines.
Oxidative stress is a common feature of neuroinflammation, 65,66 and dysregulated inflammation contributes to the pathogenesis of CNS diseases. 60 In this work, dark chocolate was explored as an antioxidant dietary supplement 28 shown to improve cerebral blood flow 30 and cognitive performance. 33 The administration of chocolate high in polyphenols to MC mice not only suppressed, reversed, or upregulated DVC gene expression patterns indicative of Aβ accumulation (Bace-1), synuclein aggregation (Sncb), excision repair pathways (Ercc2), biosynthesis of acetylcholine (Chat) but also upregulated the expression of antioxidant genes (Gpx1 and Prdx4), sensors of oxidative stress (Park7), regenerative and neuronal plasticity genes (Gap43 and Sncb), and genes effectively degrading excess α-synuclein in dopaminergic cells (Ctsd). Unexpected findings included the upregulation of caspase-1. 67 The upregulation of caspase-1 goes along with the IL-1β failure to be downregulated in the chocolate-treated mice. The activation of the delicate caspase-cascade system 67 is evident in the highly exposed mice in spite of the chocolate treatment. These results support the notion of the complexity of the cytokine and caspase cascades and the need to expand the chocolate studies to include the evaluation of key proteins such as α-synuclein.
Detailed neuropathological analyses of sporadic PD have suggested that the development of α-synuclein-containing Lewy bodies and/or Lewy neurites in olfactory bulb and the dorsal nucleus of the vagus are 2 of the earliest indications of PD pathogenesis. 68 These events coincide with the appearance of nonmotor dysfunction symptoms, including impaired olfaction and autonomic dysfunction 69,70 and presage decades later, the development of motor dysfunction symptoms, and substantia nigra neurodegenerative changes.
Based on this evidence, Braak et al 68 have proposed that sporadic PD is caused by an environmental neurotrophic factor that is taken up by olfactory and enteric neurons. From there, it spreads to brain structures that are anterograde of the olfactory bulb and retrograde of the DMNV. 40,68,71 Our observations in MC mice and humans suggest that exposures to both fine and ultrafine particulate matter as well as endotoxins could be the missing “environmental neurotrophic factor” Braak refers to in accounting for the PD-like pathology in MC children and young adults. 10 These observations warrant regular monitoring of the raw incidence of PD in MC versus clean-air urban areas.
Although endotoxin concentrations associated with particulate matter in MC have been well documented over the years, 17 –19 a limitation of this study was the lack of direct endotoxin measurements in the study period. The significant upregulation of CD14, a surface differentiation antigen that functions as a receptor for LPS, was used as a proxy for the exposure and indeed MC mice had significant olfactory bulb upregulation of CD14, while clean air and IP-LPS mice did not.
In summary, exposure to air pollution produces significant upregulation of inflammatory genes in the DVC and an important imbalance in antioxidant, apoptotic, and neurodegeneration-related genes. The sustained involvement of the olfactory bulb and the DVC in urban exposed mice suggest that these regions are key air pollutant targets and endotoxin plays a critical role. The DVC persistent inflammation may have deleterious effects in exposed populations as evidenced by the accumulation of immunoreactive α-synuclein in the dorsal motor nucleus of the vagus of MC children and young adults. 10 Although there are no published data on the incidence and prevalence of PD in MC, our children and young adult autopsy studies support the view that sustained inflammation of the DVC could put MC residents at a higher risk for developing PD. Our findings suggest that regular consumption of dark chocolate with high concentrations of polyphenols may reduce the DVC inflammation and have neuroprotective properties in the setting of air pollution exposures.
Footnotes
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: The ASIP SROPP program to Rafael Villarreal-Calderon and P20 RR015583. This work was presented in part at the FASEB 2008 Meeting at San Diego by Rafael Villarreal-Calderon who was the first American Society for Investigative Pathology Summer Research Opportunity in Pathology Program recipient (2007).
