Abstract
Engineered silver nanoparticles (AgNPs), including silver silicate nanoparticles (Ag-SiO2 NPs), are used in a wide variety of medical and consumer applications. Inhaled AgNPs have been found to translocate to the olfactory bulb (OB) after inhalation and intranasal instillation. However, the biological effects of Ag-SiO2 NPs and their potential nose-to-brain transport have not been evaluated. The present study assessed whether inhaled Ag-SiO2 NPs can elicit microglial activation in the OB. Adult Sprague-Dawley rats inhaled aerosolized Ag-SiO2 NPs at a concentration of 1 mg/ml for 6 hours. On day 0, 1, 7, and 21 post-exposure, rats were necropsied and OB were harvested. Immunohistochemistry on OB tissues were performed with anti-ionized calcium-binding adapter molecule 1 and heme oxygenase-1 as markers of microglial activation and oxidative stress, respectively. Aerosol characterization indicated Ag-SiO2 NPs were sufficiently aerosolized with moderate agglomeration and high-efficiency deposition in the nasal cavity and olfactory epithelium. Findings suggested that acute inhalation of Ag-SiO2 NPs elicited transient and differential microglial activation in the OB without significant microglial recruitment or oxidative stress. The delayed and differential pattern of microglial activation in the OB implied that inhaled Ag-SiO2 may have translocated to the central nervous system via intra-neuronal pathways.
Keywords
Introduction
Silver nanoparticles (AgNPs), including silver silicate nanoparticles (Ag-SiO2 NPs), are engineered transition-metal nanomaterials with diverse commercial applications. AgNPs are one of the most widely used metallic nanoparticles due to their broad-antimicrobial properties. 1 AgNPs are used in wound dressings and as protective coatings for medical supplies.2,3 AgNPs are also used in various consumer products, from cosmetics to food storage containers. 4 Exposure to AgNPs can occur through dermal and ocular absorption, oral ingestion, or inhalation.5-9 Inhalation of AgNPs may occur during manufacturing or usage of aerosolized commercial products or drug therapies containing silver, such as over-the-counter nasal spray treatments for respiratory infections. AgNPs are manufactured in various forms, with different compositions, sizes, shapes, and surface coatings, all of which confer unique physical and chemical properties.10-12 Silver silicate (Ag-SiO2) is a type of AgNPs supported on silica, which reduces particle agglomeration. 13 Ag-SiO2 NPs are also used as antimicrobial additives for wound dressings and in catalytic reduction of toxic dyes in industries.14-16
Inhaled AgNPs can become entrapped in the fluid lining layer over the olfactory mucosa before reaching the olfactory epithelium. 17 From there, several possible pathways to the central nervous system (CNS) have been suggested. These include the (1) intra-neuronal pathway via uptake into olfactory nerve and trigeminal nerve, with retrograde axonal transport through neurons to the brain; (2) extra-neuronal pathway via transcellular transport across the sustentacular cells and paracellular transport across the tight junctions between sustentacular cells and olfactory sensory neurons; (3) systemic pathway through translocation across the rich vasculature of the respiratory epithelium, absorption into lymphatics, ingestion following clearance of NPs via the mucociliary escalator with subsequent gastrointestinal absorption into the hepatic portal circulation.18-20 Intra-neuronal nose-to-brain translocation via the olfactory nerve pathway is also utilized by viruses such as herpesvirus, influenza A virus, and SAR-CoV-2 virus, which are similarly sized to AgNPs, to gain access to the CNS.21,22
AgNPs have been found in the olfactory bulb (OB) following inhalation of AgNPs.23-26(p1873) Previous studies suggested that NPs can reach the OB via the olfactory pathway, similar to how odorant information is relayed to the CNS.26-28 The OB receives odorant molecules from the olfactory epithelium in the nasal cavity via the olfactory sensory nerve fascicles.29(p1) The OB has a lamellar structure with several histologically distinct layers (Figure 1). 30 These layers, in the order of most superficial to deep, include the glomerular layer (GL), external plexiform layer (EPL), mitral cell layer (MCL), internal plexiform layer (IPL), and granule cell layer (GCL). Odorant molecules from the nasal cavity arrive at the GL of the OB before information is relayed to cells of the deeper layers of the OB and finally the primary olfactory cortex.29(p2)
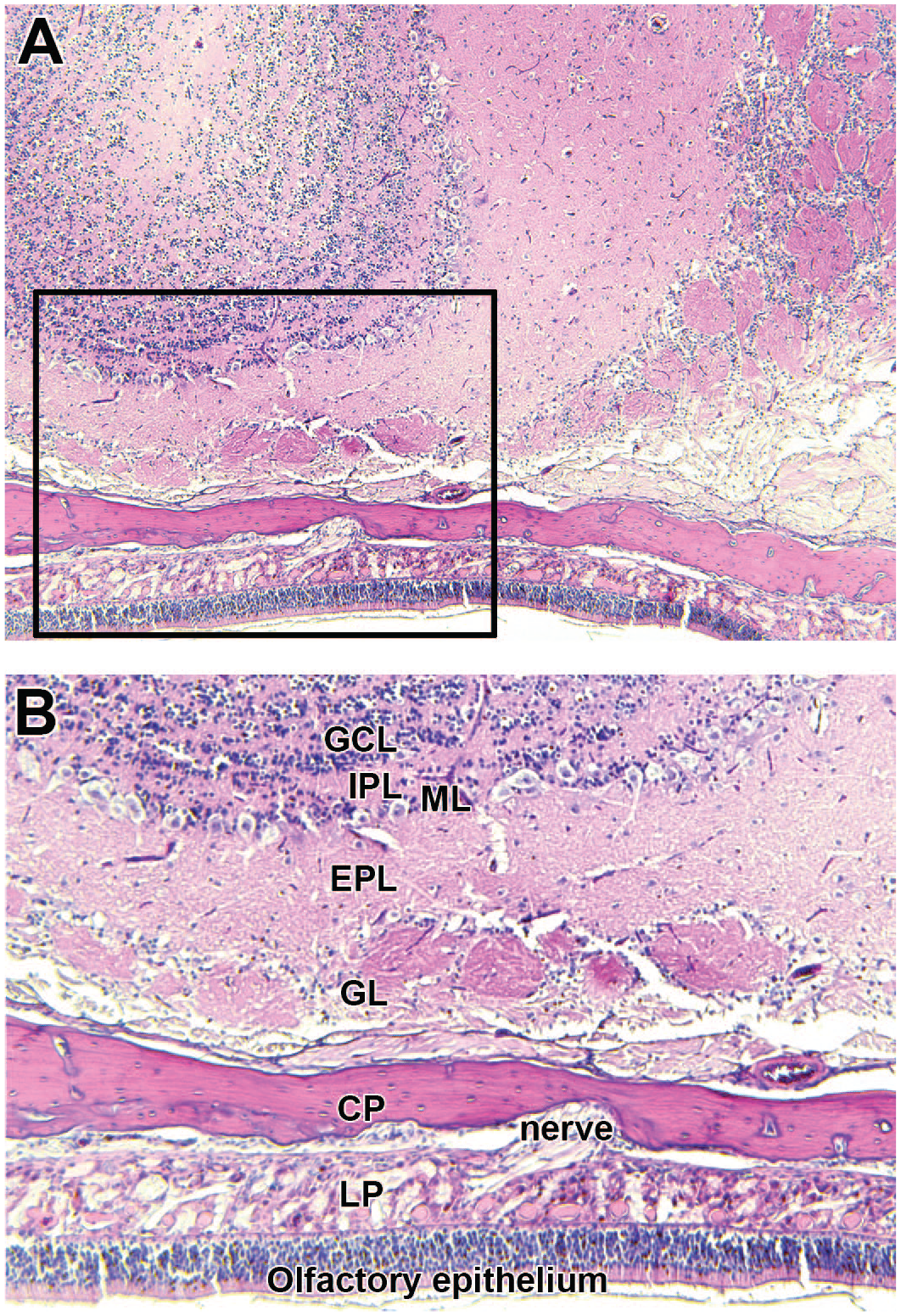
Representative brightfield microscopy images of 5-μm thick, hematoxylin and eosin–stained tissue sections of rat nasal tissue and olfactory bulb (OB). Panel A shows a transverse section of the OB at original objective 5X. Box identifies a section of the OB shown in panel B at original objective 10X. Panel B shows the lamellar structure composed of histologically distinct layers of the OB. CP indicates cribriform plate; EPL, external plexiform layer; GCL, granular cell layer; GL, glomerular layer; IPL, internal plexiform layer; LP, lamina propria; ML, mitral cell layer.
Microglia are resident macrophages of the CNS and are beneficial in immune surveillance, synaptic plasticity, neuronal repair, and neurogenesis.31-34 However, prolonged and/or excessive microglial activation following inhalation of NPs can be detrimental due to associated upregulation of toll-like receptors and release of pro-inflammatory cytokines and chemokines.35-37 Inhaled AgNPs may be sufficiently immunogenic to elicit an in vivo pro-inflammatory response in the OB via microglial activation.26(p1874) The cytotoxicity of AgNPs is thought to be primarily due to the release of ionic silver (Ag+) into the biological environment.38-40
The primary objectives of the present study were to determine whether acutely inhaled Ag-SiO2 NPs activate resting microglia in the OB, and if there is differential activation of microglia in different histological layers of the OB. Rats were exposed to aerosols of Ag-SiO2, and their OB were harvested immediately after exposure on day 0 (D0) and on post-exposure day 1, 7, and 21 (D1, D7, D21, respectively). Ten-micrometer-thick OB sections were immunostained for anti-ionized calcium-binding adapter molecule 1 (anti-Iba1) to identify the site and morphology of microglia in the OB and with heme oxygenase-1 (HO-1) as a marker of oxidative stress in the OB.
Materials and Methods
Powdered Ag-SiO2 NPs
The Ag-SiO2 NPs used in the present study were synthesized, characterized, and supplied in powder form by the National Institute of Environmental Health Sciences Centers for Nanotechnology Health Implications Research Consortium. Ag-SiO2 NPs were synthesized by flame spray pyrolysis as silver (~8 nm) supported on silica (~7 nm) (3.79% w/w).41,42 The powdered Ag-SiO2 NPs were on average 10.64 nm ± 7.10 nm in diameter before suspension in endotoxin-free water to a 1 mg/ml concentration for aerosolization. The primary particle size, primary particle size distribution, and size factor were characterized by transmission electron microscope (TEM) image analysis. Crystallinity, density, and chemical composition were characterized by x-ray diffraction, pycnometer, and inductively coupled plasma mass spectrometry (ICP-MS), respectively.
Animal Care
Adult, 12-week-old male Sprague-Dawley rats were obtained from Envigo laboratory (Fremont, CA). Upon arrival, the rats were randomly assigned to two exposure groups, Ag-SiO2 (n = 4 per time point) or filtered air (n = 8; pooled from D1 and D21 post-exposure). The rats were housed two per cage in filter-top polycarbonate cages in an animal facility with high-efficiency particulate air filters, maintained in a 12-hour dark/light cycle under standard temperature and given ad libitum access to a standard laboratory rodent diet (Purina 5001, Newco Distributors, Rancho Cucamonga, CA) and water. Rats were allowed to acclimate for at least 1 week prior to the experimental exposure. During the acclimation period, the rats were conditioned to remain in the inhalation exposure tubes (Teague Enterprises, Woodland, CA) for progressively longer periods. The conditioning exercises were performed to minimize confinement stress during the experimental exposure. Animals were handled following guidelines from the National Institutes of Health and Institutional Animal Care and Use Committee of the University of California, Davis.
Aerosolization of Ag-SiO2 NPs
Rats were exposed to either filtered room air or Ag-SiO2 aerosol generated by the aerosol nebulization system located at the Center for Health and the Environment, University of California, Davis (Figure 2). The Ag-SiO2 suspension was sonicated (QSonica Sonicators; 500 W, 20kHz, 90% Amp), kept on ice to reduce particle aggregation, and aerosolized with a BGI 6-jet Collision nebulizer (Waltham, MA) into fine droplets. Air for the nebulizer was compressed with an oil-free compressor (California Air Tools, San Diego, CA), dehumidified with compressed air dryers (Wilkerton, Richland, MI), and filtered with a Motor Gard M-610 filter (Motor Gard, Manteca, CA). The fine nebulized droplets passed through a custom-fabricated heater and two diffusion dryers (TSI, Shoreview, MN) to remove any remaining water. The resulting dried particles then passed through a Krypton-85 charge-neutralizer to reduce static and minimize particle agglomeration before entering the 48-port nose-only exposure system connected by steel piping. 43

Aerosolization and exposure system.
Nose-Only Inhalation Exposure
Rats were placed into the nose-only exposure tubes, one rat per tube, for a single, 6-hour exposure to either filtered air or aerosolized Ag-SiO2. Rats were monitored for their presence and activity while in the inhalation chamber for the entire duration of the exposure period. The average temperature and humidity in the exposure room was 56°F and 54%, respectively.
Characterization of the Aerosolized Ag-SiO2 NPs
Air samples were collected throughout the 6-hour exposure period, and aerosol characterization was performed using a protocol previously described. 44 Briefly, the mass concentration and particle size distribution of the aerosolized Ag-SiO2 NPs were determined via gravimetry and an eight-stage Mercer-style cascade impactor, respectively, using samples collected onto 25-mm diameter Pallflex membrane filters (catalog number 28150-925, Pall Life Sciences, Port Washington, NY). The silver mass was determined by x-ray fluorescence (XRF; Chester Labnet, Tigard, OR) with samples collected on 25-mm diameter Pall Teflo filters (catalog number 28139-131, Pall Life Sciences); and the average particle diameter was measured using aerosolized particles collected onto formvar carbon film (carbon grids 200 mesh, catalog number FCF200-Cu, Electron Microscopy Sciences, Hatfields, PA) and examined via a Phillips CM-12 transmission electron microscope operating at 120 kV.
Tissue Collection and Preparation
Ag-SiO2 NPs-exposed rats were euthanized on D0 (immediately after the exposure), D1, D7, and D21 post-exposure with an intraperitoneal injection of Beuthanasia-D at 7.5 ml/kg (MWI Veterinary Supply Company, Los Angeles, CA 90074). Control animals were necropsied on D1 and D21 after the exposure. Cardiac puncture was performed, and blood was collected in a 12-ml round bottom tube for centrifugation at 2000 revolutions per minute (rpm) for 15 minutes. The resulting blood plasma was collected and stored at −80°C for later use. Following blood collection, the trachea was cannulated, and the left main bronchus was clamped. The right middle, caudal, and accessory lobes were lavaged, flash-frozen, and stored at −80°C for later use. Briefly, the lobes were lavaged three times with a single 7-ml aliquot of sterile phosphate-buffered saline (PBS, Sigma Aldrich, St Louis, MO) in a 12-ml syringe for collection of bronchoalveolar lavage fluid (BALF). The BALF was collected in a 15-ml round bottom tube and centrifuged for 15 minutes at 2000 rpm and 4°C. The resulting BALF supernatant was decanted and stored for further analysis, while the cell pellet was resuspended in 2 ml of sterile PBS. Trypan blue was used to count the total viable and non-viable cells. A 100-µl aliquot of resuspended cells was used to prepare cytospin slides for cell differentials. The left lung was unclamped and inflation-fixed at a hydrostatic pressure of 30 cm with 4% paraformaldehyde for 1 hour. The fixed lung was subsequently stored in 4% paraformaldehyde for 24 hours before transfer to 70% ethanol. A separate manuscript is being prepared to evaluate the potential pulmonary toxicity following inhalation of Ag-SiO2 NPs.
The heads were decapitated, brains were removed, and the remaining head was fixed in 4% paraformaldehyde for 24 hours and stored in 70% ethanol. The heads and nasal cavities were decalcified along with the OB using 0.5-M ethylenediaminetetraacetic acid (EDTA, pH = 8; Thermo Fisher Scientific, Waltham, MA) at room temperature for 2 weeks. A total of four transverse sections of nasal cavity per rat were obtained following a standard protocol. 45 Then, one sagittal section of the decalcified heads was obtained per rat. Sagittal tissue orientation was chosen so that the nasal cavity and OB tissues are on the same slide for silver detection in the nose and the OB with autometallography. All tissues were embedded in Paraplast (Fisher Scientific, Swedesboro, NJ), cut with a microtome to 5-µm and 10-µm thick sections and put onto slides (catalog number 12-550015, Fisherbrand, Thermo Fisher Scientific, Waltham, MA).
Immunohistochemistry
For each rat, three slides of OB were immunostained for anti-Iba1, and one slide of OB was immunostained for HO-1 to visualize microglia and assess for oxidative stress, respectively. The OB sections were deparaffinized in three changes of toluene for 5 minutes each and rehydrated in 100%, 95%, and 70% ethanol for 2 minutes each before immersion in EDTA (Thermo Fisher Scientific, pH = 8) at 123°C for 2 minutes, and 85°C for 10 seconds. The sections were then cooled for 45 minutes and washed for 5 minutes under gentle running tap water. Endogenous peroxidase was blocked with 3% hydrogen peroxide for 10 minutes, followed by a quick dip in distilled water and three washes in phosphate-buffered saline with 1000 µl Tween 20 (PBST; Sigma Aldrich) at 2 minutes each. Nonspecific proteins were blocked with Protein Block (catalog number X0909; Dako, Carpinteria, CA) for 20 minutes at room temperature. The sections incubated with anti-Iba1 (ab 153696, Abcam Inc, Cambridge, MA), at a 1:1000 dilution with PBST, were incubated overnight at room temperature. The sections incubated with HO-1 (ab 13243, Abcam Inc), at a 1:700 dilution with PBST, were incubated for 1 hour at room temperature. Negative-control tissue sections were not incubated with the primary antibody. Sections were then washed in PBST following a protocol of two washes of brief 20 dips followed by two washes at 5 minutes each. The sections were then incubated with the secondary antibody, horseradish peroxidase-labeled polymer for rabbit (catalog number K4403; Dako), for 30 minutes at room temperature and washed twice in PBST with occasional agitation for 5 minutes each. Each section was incubated with 100 µl of 3,3’-Diaminobenzidine (DAB) and substrate (catalog number K3468; Dako) for 5 minutes at room temperature. DAB solution was tipped off each section onto lab paper. Sections were washed briefly in distilled water twice and counterstained with Harris Hematoxylin (MasterTech, Inc, Lodi, CA) and coverslipped with ClearMount permanent mounting medium (Thermo Fisher Scientific). Immunohistochemistry staining was evaluated qualitatively.
Microglial Visualization and Characterization
The OB sections immunostained for Iba1 were divided into four histologically distinct layers for photomicrography. These layers consisted of the olfactory nerve layer (ONL), glomerular layer (GL), external plexiform layer (EPL), and inner layers (IL). Each IL consisted of the mitral cell layer, internal plexiform layer, and granule cell layer, grouped as one layer because these layers were difficult to distinguish from each other at original objective 20X. Microglial abundance and morphology were evaluated in three randomly sampled non-overlapping fields per histologically distinct layer for a total of twelve fields per histological section, one section per animal, four animals in each Ag-SiO2 post-exposure group and eight animals in sham control at original objective 20X on a Zeiss AxioLab.A1 microscope. All microglia were manually counted in each of the twelve randomly captured, non-overlapping fields to determine microglial abundance, and microglial morphology was evaluated to determine resting and activation states. The ratio of activated to resting microglia was calculated for sham control and each of the five exposure groups. The protocol was repeated for each of the four histologically distinct layers (ONL, GL, EPL, and IL) in the OB sections. Four investigators evaluated microglial abundance and morphology in a blinded fashion, and their scores were averaged.
Microglial morphology was categorized as resting or activated as a mean of evaluating the OB response to inhalation of Ag-SiO2 and as an indirect assessment of NP translocation to the CNS. Microglial morphology is associated with different microglial phenotypes.46,47 Resting microglia surveils for changes in the microenvironment and has a small cell body with extensively ramified processes. The processes are highly motile, undergoing continuous cycles of de novo formation and disappearance without a net change in the total number of processes to dynamically scan the environment without disturbing pre-existing neuronal structures.48,49 Once microglia sense changes in the microenvironment such as the presence of pro-inflammatory stimuli such as lipopolysaccharide and interferon gamma or cytokines such as interleukin 4 and interleukin 10, they change their morphology into an amoeboid form with enlarged cell bodies and retracted, thickened processes to mediate phagocytic activity, suppress inflammation, or mediate tissue remodeling. 50 This study classified microglia as resting or activated based on previously accepted criteria.26,27,51 Resting microglia were defined as cells with at least two highly ramified processes extending at least twice the length of a small nucleus. Activated microglia were defined as cells with significantly shortened ramified processes extending from a slightly enlarged cell nucleus.
Oxidative Stress
The OB sections immunostained for HO-1 were treated in similar fashion as sections immunostained for Iba1. HO-1 expression was evaluated qualitatively, as present or not present, in twelve randomly sampled non-overlapping fields per histologically distinct layers per histological section, one section per animal, four animals in each Ag-SiO2 post-exposure group and eight animals in sham control at original objective 20X on a Zeiss AxioLab.A1 microscope.
Statistical Analysis
Data were analyzed with Jamovi 1.6 statistical software (Sydney, Australia). No animals were excluded from statistical analysis. Shapiro-Wilk and Levene’s tests were performed to confirm normality and homoscedasticity, respectively. 52 Statistically significant differences in microglial abundance and microglial activation were determined by Welch’s one-way analyses of variance (ANOVAs). 53 Games-Howell post hoc tests were used to identify significant differences between specific groups. These tests were performed at a significance level of P < .05. Data are presented as group means ± standard error.
Results
Ag-SiO2 Aerosol Characterization
Aerosol characterization suggested that the Ag-SiO2 NPs in nanopure water were sufficiently aerosolized with moderate agglomeration. The mean Ag-SiO2 aerosol mass concentration measured by gravimetry was 4.9 ± 2.3 mg/m3, and the Ag concentration measured by XRF was 0.17 ± 0.01 mg/m3. These results suggested that suspension of Ag-SiO2 NPs in nanopure water did not alter the mass concentration of the aerosol. The median aerosol diameter measured by cascade impactor was 1.9 ± 0.3 μm, and the size (diameter) distribution of the Ag-SiO2 NPs based on TEM ranged from 21 to 370 nm (Figures 3 and 4). The aerosol diameter measured by the cascade impactor suggested that there was particle agglomeration, but size distribution based on TEM suggested that approximately 50% of Ag-SiO2 NPs were less than 100 nm in diameter and small enough to be deposited in the nasal epithelium. 54

Transmission electron microscopy image at original objective 120 kX of a silver silicate (Ag-SiO2) nanoparticle obtained during a single, 6-hour aerosolization period.
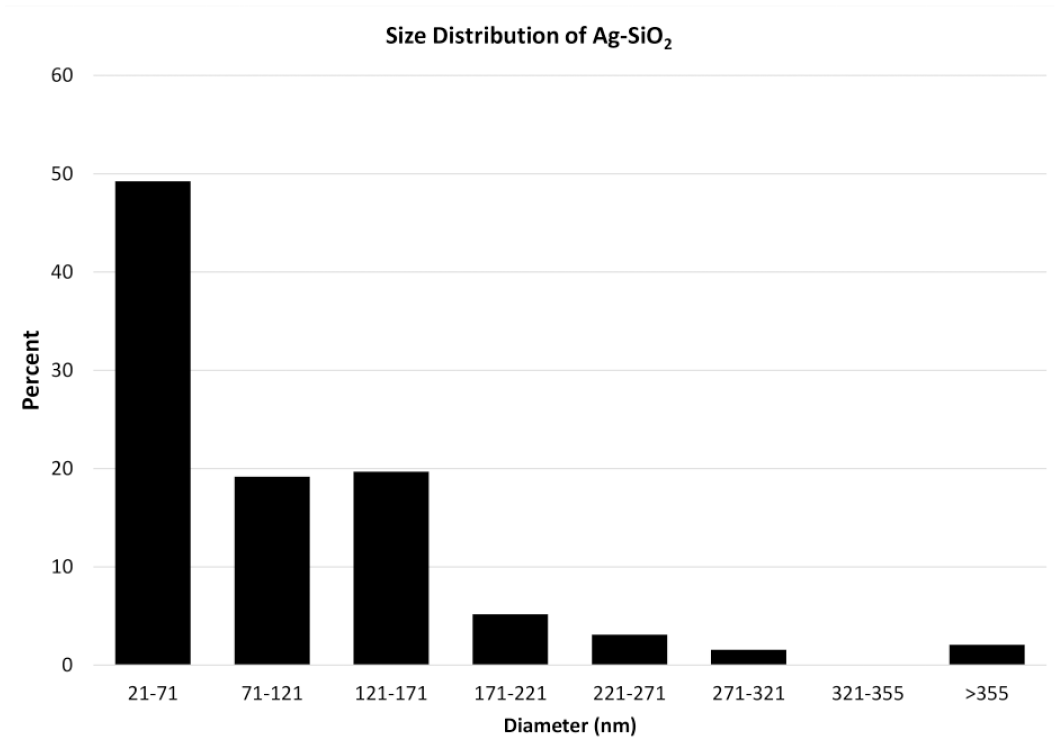
Graph of the size (diameter) distribution of aerosolized silver silicate (Ag-SiO2) nanoparticles measured by transmission electron microscopy.
Transient and Differential Microglial Activation in the OB Without Microglial Recruitment
Microglial morphology in the OB was categorized as resting or activated (Figure 5). Microglial numbers were counted to determine microglial abundance in the OB (Figure 6, panel A) and in each histologically distinct layer of the OB in sham control and Ag-SiO2 post-exposure groups (Figure 6, panels B, C, D, and E). The ratio for activated to resting microglia were calculated for the entire OB (Figure 7, panel A) and for each histologically distinct layer of the OB (Figure 7, panels B, C, D, and E) in sham control and Ag-SiO2. After Ag-SiO2 versus filtered air exposure, there was significant (P = .02) microglial activation in the OB, compared with sham control, with elevated ratios of activated to resting microglia noted on D1 and D7 post-exposure and a return to control concentration by D21 (Figure 7, panel A). In the ONL, there was a notable but non-significant (P = .08) increase in the ratio of activated to resting microglia on D1 and D7 compared with sham control (Figure 7, panel B). In contrast, the GL of the OB in exposed rats demonstrated a significant increase (P = .01) compared with sham control in the proportion of activated to resting microglia on post-exposure D1 [Figure 7, panel C, and Figure 8). In the EPL and IL, there were no notable inter-group differences in the ratio of activated to resting microglia (Figure 7, panels D and E). There was no significant difference in microglial abundance between sham control versus exposed group at any time point post-exposure, which suggested a lack of microglial recruitment (Figure 6).
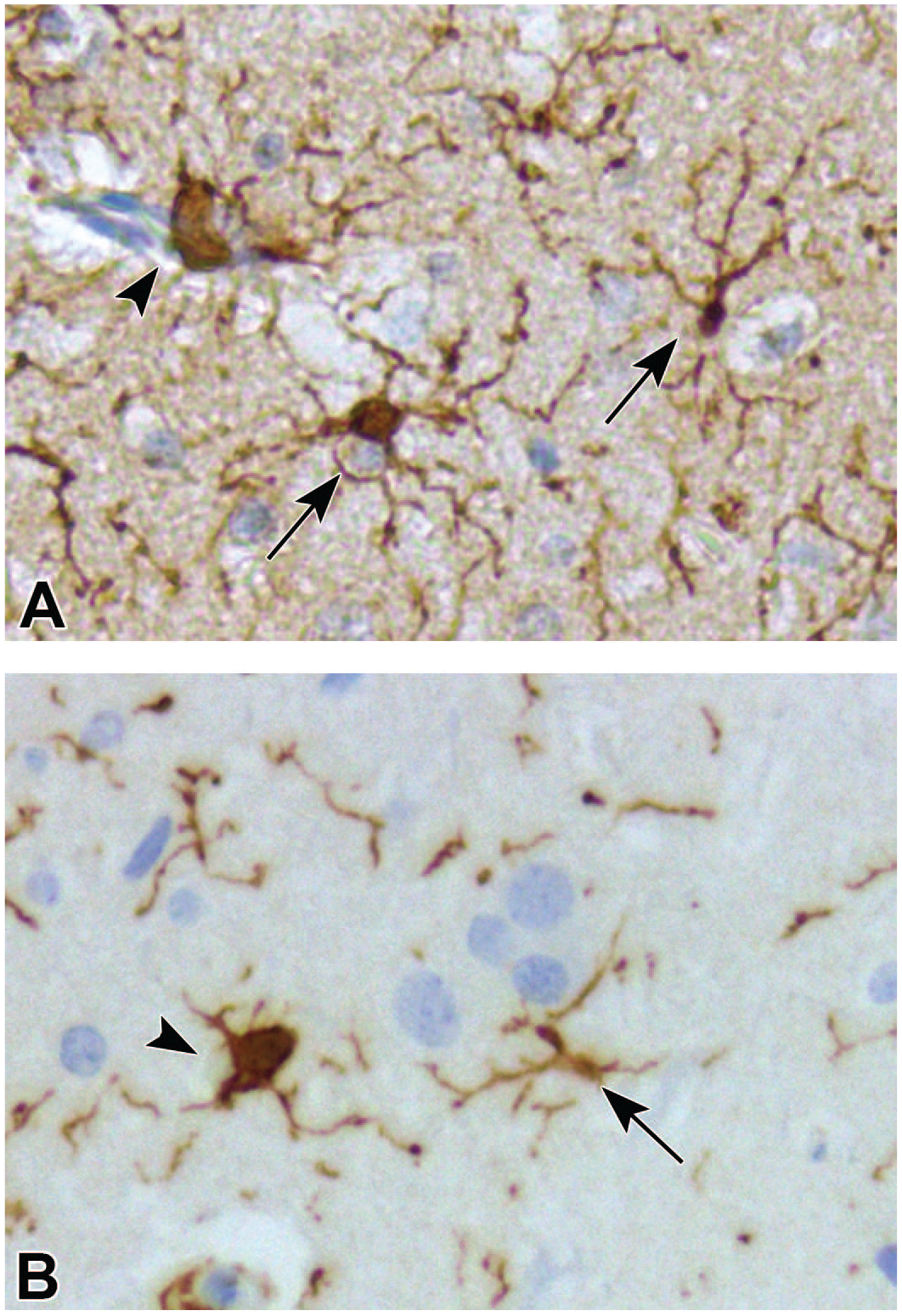
Representative brightfield microscopy images of olfactory bulb tissue sections, at original objective 20X, with anti-ionized calcium-binding adapter molecule 1 (anti-Iba1; panel A) and anti-heme oxygenase-1 (anti-HO-1; panel B) immunohistochemical stains. Resting microglia are indicated by arrows and characterized by a small cell body with ramified processes. Activated microglia are indicated by arrowheads and characterized by a large cell body with retracted processes.
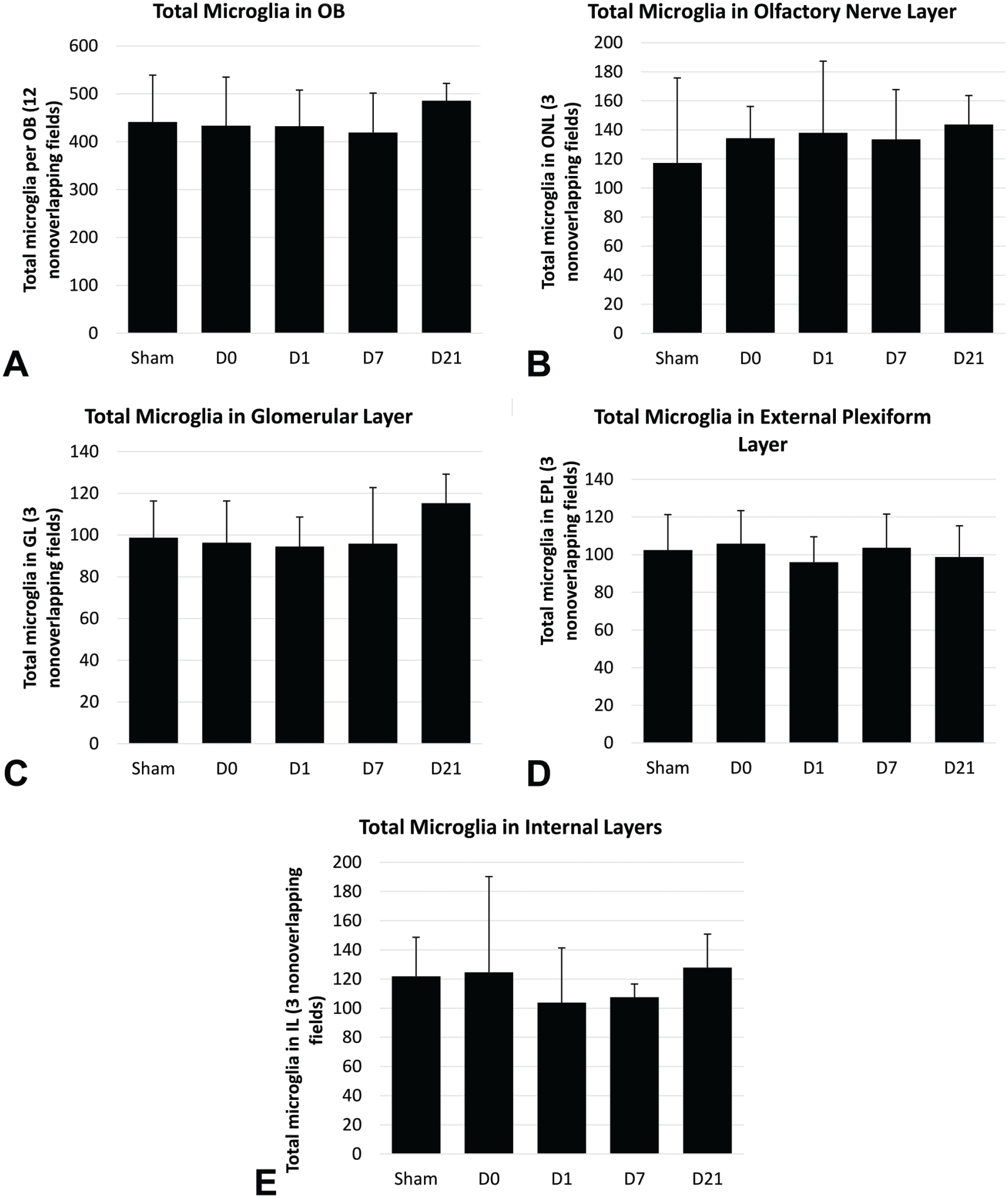
Total (panel A) and differential (panels B-E) microglial cell counts in the olfactory bulb (OB). Microglial cells were counted in twelve non-overlapping fields per animal. Differential microglial cell counts were determined using three non-overlapping fields per histologically distinct layer per olfactory bulb per animal. The analysis was done at original objective 20X, and four animals in each Ag-SiO2 post-exposure group and eight animals in sham control were examined. Welch’s one-way ANOVAs and post hoc Games-Howell tests were used to compare groups exposed to filtered air (control) or aerosolized silver silicate nanoparticles and euthanized 0, 7, 14, or 21 days post-exposure (D0, D7, D14, and D21, respectively). Each value shown in the graphs is a group mean ± standard error.
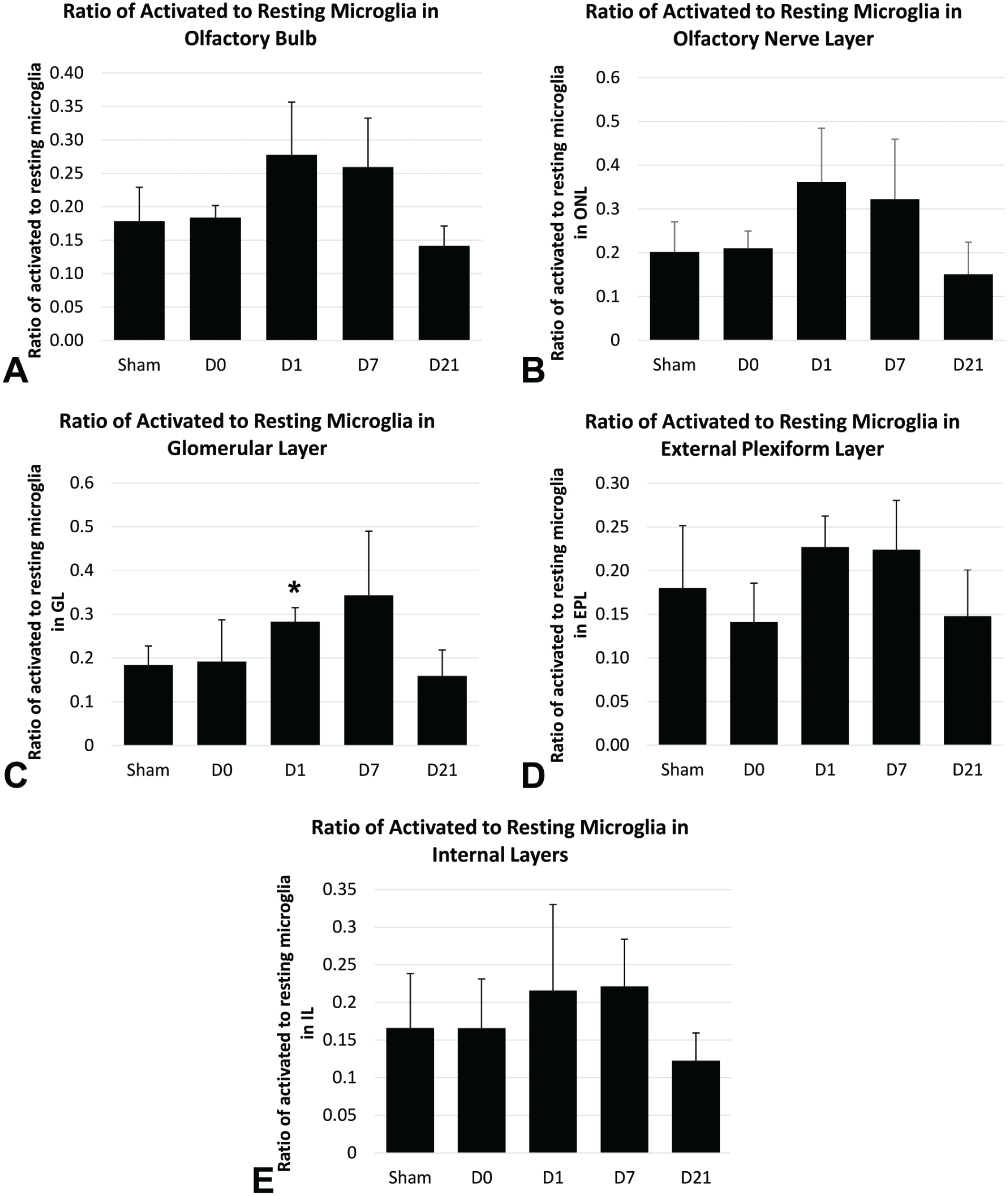
Inter-group comparisons of the ratio of activated to resting microglia in the olfactory bulb (panel A) and histologically distinct layers of the olfactory bulb (panels B-E). Microglia were morphometrically evaluated in twelve non-overlapping fields per olfactory bulb (OB) per animal, with three fields per histologically distinct layer. The analysis was done at original objective 20X, and four animals in each Ag-SiO2 post-exposure group and eight animals in sham control were examined. Welch’s one-way ANOVAs and post hoc Games-Howell tests were used to compare groups exposed to filtered air (sham control) or aerosolized silver silicate nanoparticles and euthanized 0, 7, 14, or 21 days post-exposure (D0, D7, D14, and D21, respectively). Each value shown in the graphs is a group mean ± standard error. The asterisk (
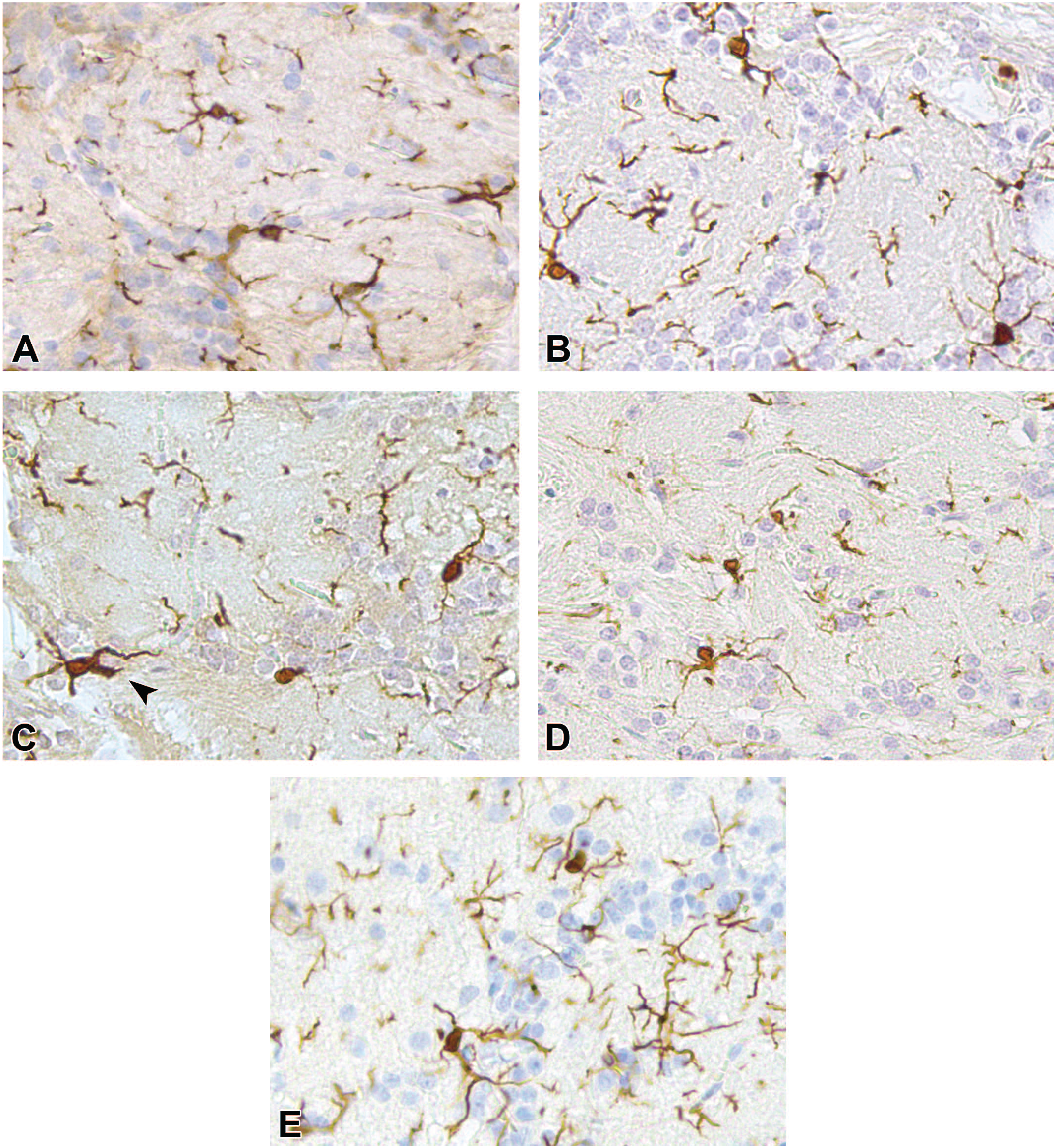
Representative brightfield microscopy images of the glomerular layer of the olfactory bulb tissue sections, at original objective 20X, with anti-ionized calcium-binding adapter molecule 1 immunohistochemical stain to visualize microglial cells (sham control [A], day 0 post-exposure [B], day 1 post-exposure [C], day 7 post-exposure [D], day 21 post-exposure [E]). Arrowhead points to activated microglia.
Mild Oxidative Stress in the OB
Sparse HO-1 immunoreactivity was noted in the ONL on D0 and the EPL on D1 and D7 (Figure 9). Negative and sham controls showed no immunoreactivity.
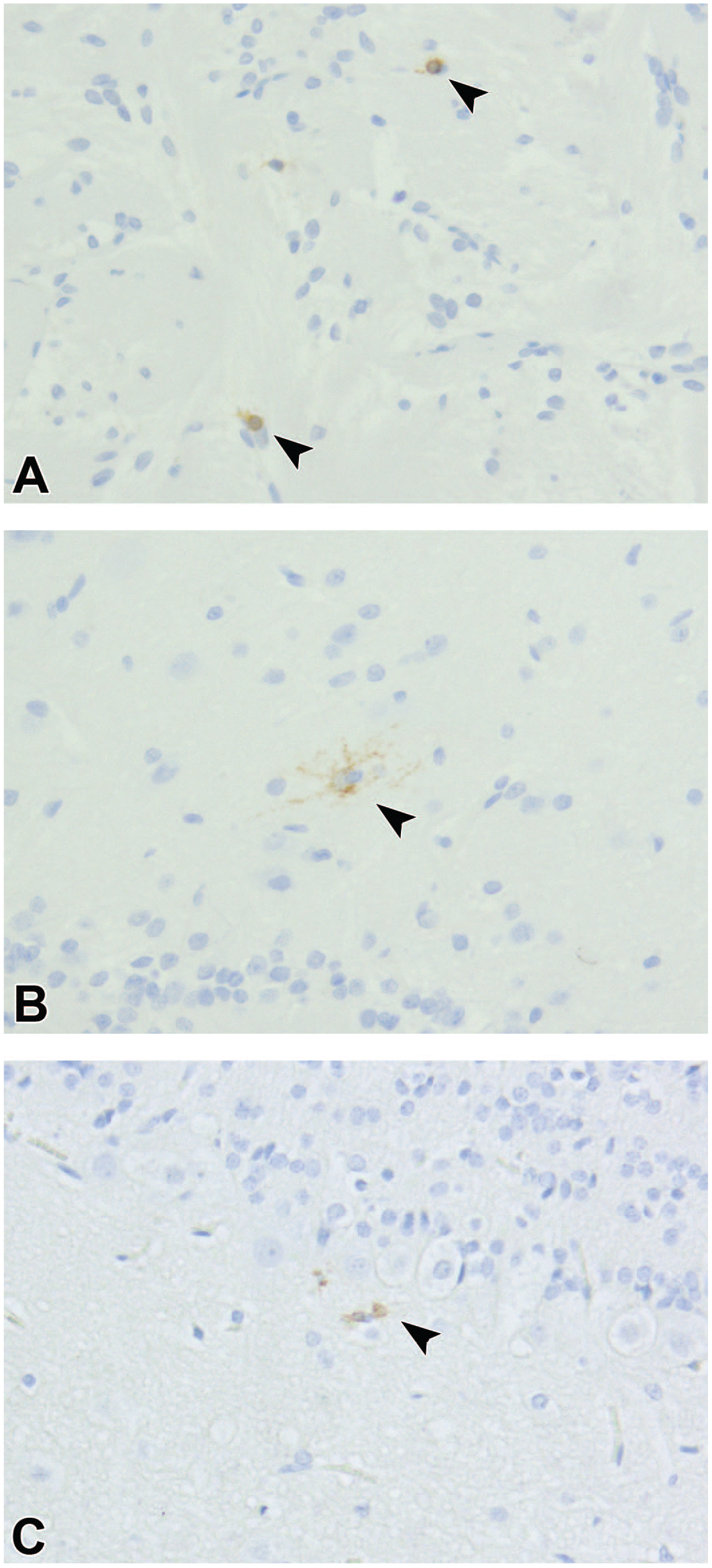
Representative brightfield microscopy images of olfactory bulb tissue sections, at original objective 20X, with anti-heme oxygenase-1 immunohistochemical stain showing immunostaining on post-exposure day 0 (A), day 1 (B), and day 7 (C). Arrowheads point to cells with heme oxygenase-1 staining, a marker for oxidative stress.
Discussion
The primary objectives of the present study were to determine whether acute inhalation of Ag-SiO2 NPs induced microglial activation and, and if so, whether microglial activation differed between the distinct histologic layers of the OB. Based on our results, we concluded that an acute (single, 6-hour) inhalation exposure to Ag-SiO2 NPs elicited slightly delayed and transient microglial activation on D1 and D7 post-exposure, particularly in the GL, without significant microglial recruitment to the OB. There was also evidence of mild oxidative stress in the OB at D0, D1, and D7 post-exposure. The temporal and differential pattern of microglial activation in the OB implied that inhaled Ag-SiO2 NPs are immunogenic in the biologic environment and may have translocated to the CNS via intra-neuronal and systemic pathways to induce a mild pro-inflammatory response.
In this study, the aerosolized silver concentration averaged 0.17 ± 0.01 mg/m3. The Occupational Safety and Health Administration (OSHA) 55 set the exposure limit for silver at 0.01 mg/m3 per 8-hour time weighted average. Maximal time weighted average of aerosolized silver concentration of up to 0.289 mg/m3 has been detected in silver manufacturing plants. 56 Our study’s aerosolized silver concentration can be correlated to an environment with high occupational exposure to aerosolized silver without personal safety measures.
A trend was observed for an increased ratio of activated-to-resting microglia in groups exposed to Ag-SiO2 NPs compared with filtered air controls, with the highest elevation noted on D1 and D7 post-exposure, which returned to control concentration by D21 (Figure 7, panel A). These observations suggested a transient pro-inflammatory response in the OB to inhaled Ag-SiO2 NPs. The inflammatory response mediated by microglia can help directly sequester NPs for digestion. Microglial cells may recognize NPs through surface toll-like receptors (TLR2 and TLR4) and can internalize NPs via phagocytosis, micropinocytosis, and clathrin-mediated endocytosis.36,37,57,58 Microglia may also be activated to remodel damaged neuronal tissues, as AgNPs and Ag+ have been shown to induce apoptosis mediated by reactive oxygen species and phagocytosis mediated by tissue necrosis factor-α and interleukin-1β.38,59-61
Activation of microglia in the OB implied that inhaled Ag-SiO2 NPs may have translocated to the CNS to induce a pro-inflammatory response. However, it is unclear from the present study which route NPs take to enter the CNS. Delayed microglial activation on D1 and D7 post-exposure without significant activation on D0 is suggestive of intra-neuronal pathway as a possible route for inhaled Ag-SiO2 NPs to arrive at the OB. This presumption is supported by previous studies, which demonstrated that particles traveling from the olfactory epithelium to the OB take minutes to make this journey via extra-neuronal pathways, 18 to 36 minutes via fast axonal transport (a type of intra-neuronal transport), and approximately 5 days with slow axonal transport (another type of intra-neuronal transport).20,27
Systemic pathway is another possible route for Ag-SiO2 NPs to enter the CNS. A kinetic time course study showed that inhaled AgNPs of similar diameter to ours were detected in the OB with peak concentration immediately after a 6-hour inhalation exposure period and again at D7 post-exposure. 62 Patchin et al. 26 found significant microglial activation in the OB immediately after exposure to similarly sized inhaled AgNPs and at D1 and D7 post-exposure. These temporal profiles are similar to our finding of microglial activation in the OB on D1 and D7 post-exposure. The first peak in Ag deposition and early microglial activation may represent translocation of NPs via the intra-neuronal olfactory pathway as well as systemic absorption of NPs via the gastrointestinal tract and the hepatic portal circulation. Recordati et al. 63 showed that ingestion of AgNPs was correlated with structural alteration of the blood-brain barrier, which may facilitate AgNPs entry to the CNS. The second peak in Ag deposition and delayed microglial activation may represent NPs entering the systemic circulation and lymphatics following delayed clearance from the nasal cavity and lungs.26,62,64
Recruitment of additional microglia to the OB is another marker of pro-inflammatory response that has been reported following exposure to NP. 65 We did not find a significant increase in the number of microglia in the OB in animals exposed to Ag-SiO2 NPs compared with controls at any post-exposure time point (Figure 6). This suggested a non-inflammatory response to inhaled Ag-SiO2 NPs. This is further supported by a mild oxidative stress response indicated by sparse HO-1 immunostaining in the OB. These findings, taken altogether, suggested an insignificant inflammatory response in the OB to inhaled Ag-SiO2 NPs. The exposure time to NPs in this study (single, 6-hour inhalation) may not be sufficiently long to elicit microglial recruitment to the OB. Wang et al. 65 found microglial recruitment to the OB following every other day instillation of NPs over a period of 40 days. Second, the silicate core of the Ag-SiO2 NPs conferred stability to the structure and may have caused the AgNPs to oxidize more slowly to Ag+, thus minimizing injury to cells in the OB. 13 Aggregation of Ag-SiO2 NPs during aerosolization, as shown by the aerosol diameter measured by the cascade impactor, may have also reduced NPs deposition onto the nasal epithelium, which in turn diminished cellular uptake efficiency and cytotoxic effects on microglial cells in the OB. This presumption is supported by previous studies that demonstrated decreased cellular uptake rates of large-sized NPs compared with smaller-sized NPs of the same composition.66,67 Further studies with sub-acute and chronic exposure durations are needed to fully evaluate the extent of Ag-SiO2 NP-induced inflammation in the OB.
Differential activation of microglia was noted among the histologically distinct layers of the OB. This differential activation of microglia may potentially be due to NPs failing to penetrate deeper into the OB. However, previous studies found metal NPs along with associated microglial activation and histologic brain remodeling in the OB and more distant brain regions such as the hippocampus, striatum, and lateral ventricle following inhalation and intranasal instillation of metal NPs.37,60,65,68,69 These findings suggested that the acute inhalation exposure duration may have precluded delivery of a dose of metal NPs equivalent to chronic inhalation or intranasal instillation leading to less NPs deposition onto the olfactory epithelium, less NPs translocation to the deeper layers of the OB, and less microglial activation than expected.
The major limitation of this study is that the activation of microglia in the OB showed implied rather than actual translocation of AgNPs from the nasal cavity to the OB. We were unable to visualize or quantify silver in the nasal cavity, the olfactory nerves, or in the OB using autometallography or ICP-MS. Other studies used these methods to demonstrate presence of silver in these tissues, but we were unable to replicate this result.25,26 One reason for this may be due to the interaction between EDTA used for decalcification of the nasal cavity and the silver enhancement solution used in autometallography. Autometallography utilizes gold to initiate the reduction of ionic silver to metallic silver. 70 EDTA is commonly used as a metal chelator and is capable of chelating gold.71-73 These reagents may have cross-reacted to cause gold to precipitate out of the solution rather than aid in ionic silver reduction for visualization. Another explanation may be that the amount of AgNPs deposited was insufficient for direct silver visualization due to the low dose of exposure or due to NP agglomeration precluding efficient NP deposition onto the nasal epithelium as discussed above. Further studies are needed to investigate the in vivo translocation of inhaled Ag-SiO2 NPs to the CNS.
Another limitation of the study was that we did not differentiate between activated microglia from CNS-associated macrophages, which have a similar amoeboid morphology. Microglia and CNS-associated macrophages are resident immune cells of the CNS but reside in different niches. Microglia reside in the brain parenchyma, whereas CNS-associated macrophages are non-parenchymal and reside in peripheral regions such as the perivascular space, choroid plexus, and meninges. 74 Iba1, the immunohistochemical marker chosen for this study, is widely used to highlight microglia, but it can also bind to macrophages.75,76 Our OB sections likely had perivascular macrophages but not choroidal or meningeal macrophages, as these structures were not attached to the OB sections used for analysis. Without differentially staining for these cells, we may have classified some perivascular macrophages as activated microglia. However, perivascular macrophages are normal immune cells of the CNS and are present in both sham control as well as exposure groups. Moreover, perivascular macrophages may also proliferate in response to CNS injury. 77 Therefore, by comparing the ratio of activated to resting microglia rather than total count of activated microglia, we were still able to utilize morphometric analysis of microglia to indirectly assess the inflammatory response in the OB in response to inhaled Ag-SiO2 NPs. Further studies are needed to assess the activation pattern of microglia and CNS-associated macrophages following inhalation of Ag-SiO2 NPs, as their activation pattern may have implications on the route NPs may take to enter the CNS. Activation of microglia but not CNS-associated macrophages may suggest a primarily intra-neuronal transport of NPs to the CNS to induce an inflammatory response in parenchymal immune cells. On the contrary, activation of CNS-associated macrophages but not microglia may suggest a primarily extra-neuronal or systemic transport of NPs.
Conclusion
Acute inhalation of Ag-SiO2 NPs elicited transient and differential microglial activation without significant microglial recruitment to the OB. The delayed and differential pattern of microglial activation in the OB implied that inhaled Ag-SiO2 NPs may have translocated to the CNS via intra-neuronal pathway. The absence of significant microglial recruitment and mild oxidative stress in the OB was indicative of an insignificant inflammatory response in the OB that might reflect the short duration of the acute inhalation exposure, the chemical stability of the Ag-SiO2 NPs conferred by the silicate core, or moderate Ag-SiO2 NPs agglomeration during aerosolization that interfered with NPs deposition onto the nasal epithelium, which in turn diminish the number of NPs available to reach distant organs to induce cytotoxic effects.
Footnotes
Acknowledgements
The authors thank Dale Uyeminami, Neha Singh, and Ching-Wen Wu for technical assistance and Rona Silva for proofreading.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Grant support for this study were from the National Institute of Environmental Health Sciences U01 ES027288, P30 ES023513, P51 OD011107. The silver nanomaterials used in this study were procured, characterized, and provided to investigators by the National Institute of Environmental Health Sciences (NIEHS) Centers for Nanotechnology Health Implications Research (NCNHIR) Consortium. Financial support was provided by the Students Training in Advanced Research (STAR) Program through a University of California-Davis School of Veterinary Medicine Endowment Fund.
