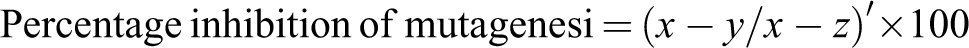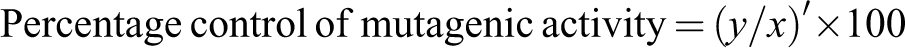Abstract
In the current study, isolation of glucosinolate degradation products was done in 4 different incubation solutions with different pHs based on the fact that distinct hydrolytic products are formed at different pHs. All the extracts were tested against direct-acting mutagens (4 nitro-o-phenylenediamine [NPD]), sodium azide, and indirect-acting mutagen (2-aminofluorene [2AF]). It was observed that extracts inhibited mutagenesis induced by the S9-dependent mutagen (2AF) more significantly than direct-acting mutagens. Two different modes of experimentation (pre-incubation and co-incubation) were used, and it was observed that the extracts showed better results in the pre-incubation mode of experimentation. Out of the 4 extracts tested, 0.1 mol/L of HCl extract was found to be the most effective in inhibiting mutagenesis with both TA 98 and TA 100 strains of Salmonella typhimurium. All other extracts also showed pronounced antimutagenic potential. The results of this study indicate the presence of potent antigenotoxic factors in broccoli, which are being explored further for their mechanism of action.
Introduction
Glucosinolates are organic anions consisting of a β-
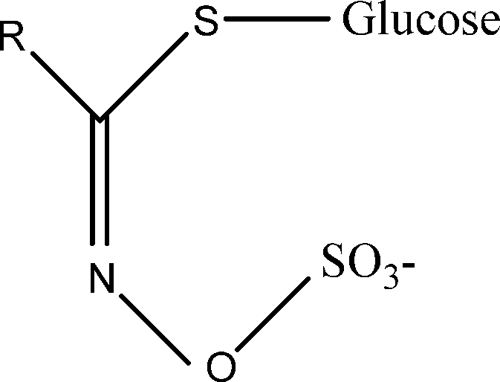
Basic structure of glucosinolate.
Glucosinolate degradation products possess a wide array of biological activities. These compounds are potent biocides and there are many reports regarding their fungicidal, bactericidal, nematicidal, insecticidal, and herbicidal activities. 4 –9 Glucosinolate hydrolysis products, especially ITCs, are also known as potent chemoprotective agents. They are believed to exert their protective effect either by impairing the cytochrome P450-mediated bioactivation of chemical carcinogens to their genotoxic intermediates (phase I metabolism) or by inducting phase II detoxification enzymes such as quinone reductase and glutathione S-transferases, aldehyde reductase, and so on. 10
Keeping in mind the promising cancer-preventing potential of broccoli, coupled with the fact that broccoli has a high content of glucosinolates, the current study was designed to extract the glucosinolate hydrolytic products at varying pH and to evaluate the antimutagenic activity of seed extracts of broccoli using Ames assay with Salmonella typhimurium tester strains TA 98 and TA100. As seeds contain much higher content of glucosinolates than any other plant part, they, therefore, were chosen for this study. Carlson et al, 11 reported that the total glucosinolate content of broccoli seeds is approximately 151.6 mmol/g of seeds whereas that of broccoli florets is only 1.9 mmol/g of fresh weight.
Broccoli (Brassica oleracea L var italica Plenck) belonging to the family Brassicaceae is known for its vitamin-rich, high-fiber content, low in calorie and anticarcinogenic properties. Glucoraphanin, the major glucosinolate, present in broccoli gets hydrolyzed to give sulforaphane ([SFN]; 1-isothiocyannato-4-(methylsulfinyl) butane), which is a potent inducer of phase II enzymes for example, glutathione transferases, epoxide hydrolase, nicotinamide adenine dinucleotide phosphate (NAD(P)H), quinone reductase, and glucuronosyltransferases. 12 Sulforaphane is also known to modulate the phase 1 metabolism either through direct interactions with cytochrome P450 enzymes or by regulating their transcript levels within the cell. 13
Materials and Methods
Chemicals
All chemical reagents (tris base, hydrochloric acid, monobasic dihydrogen phosphate, dibasic monohydrogen phosphate, sodium chloride, anhydrous sodium sulfate, histidine, biotin, glucose, calcium chloride, magnesium sulphate, ammonium chloride, etc) and all solvents (hexane and dichloromethane) were of analytical grade. 4 Nitro-o-phenylenediamine (NPD) was purchased from Sisco-Chem Industries, Mumbai (India); 2-aminofluorene (2AF) from TCI, Tokyo, Japan; Nicotinamide adenine dinucleotide phosphate (NADP); Glucose-6-phosphate (G-6-P); and sodium azide from Hi Media, Mumbai, India. All other chemical reagents were purchased from Qualigens Fine Chemicals, Mumbai, India. Tester strains TA 98 and TA 100 was procured from IMTech (CSIR), Chandigarh, India. All stock solutions were prepared in dimethyl sulfoxide (DMSO).
Preparation of Extracts
Seeds of broccoli were procured from Punjab Agriculture University, Ludhiana, Punjab, India. Glucosinolate hydrolytic products were extracted using the method given by Vaughn and Berhow, 3 with slight modifications. Briefly, seeds were ground to fine powder in a grinder. Seed powder was defatted with hexane and the defatted seed meal was then allowed to dry completely in a fume hood. Seed meal (20 g) was taken in 4 conical flasks and hydrolysis was carried out separately in 50 mL of 0.05 mol/L potassium phosphate buffer (pH 7.0), 0.05 mol/L Tris buffer (pH 10.0), 0.1 mol/L HCl (pH = 2), or 2 mol/L HCl (pH ≈ 0, highly acidic). Methylene dichloride (150 mL) was then added to each flask and the flasks were placed in an incubator shaker set at 25°C and 200 rpm for 8 hours for hydrolysis to take place. After hydrolysis, 15 g of each of sodium chloride and anhydrous sodium sulfate were added to each flask and mixed thoroughly on shaker. The supernatant was filtered through Whatman no 1 filter paper and the residue was washed 3 times with excess methylene dichloride (CH2Cl2) followed by filtration. The solvent was evaporated in a rotary evaporator to get crude extracts.
In vitro Antimutagenic Assay
The antimutagenicity of the extracts was determined with Salmonella typhimurium TA 98 and TA 100 strains using Salmonella histidine point mutation assay proposed by Maron and Ames, 14 with slight modifications suggested by Bala and Grover. 15 The Salmonella typhimurium TA98 strain has frameshift mutation, whereas TA100 carries base-substituted mutation. 4 Nitro-o-phenylenediamine and sodium azide were used as mutagens for TA98 and TA100 strains, respectively. Bacterial culture (10 mL) was grown overnight in Luria broth liquid medium. All the extracts and mutagens were dissolved in DMSO, except sodium azide that was dissolved in distilled water. Two methodologies were followed, that is co-incubation and pre-incubation. In the co-incubation method, 100 μL each of bacterial culture, mutagen, and different concentrations of extract was added to 2 mL of top agar. In the pre-incubation method, equal volumes of mutagen and extract were mixed and incubated at 37°C for 30 minutes, and 200 μL of this was added to 2 mL of top agar with 100 μL of bacterial culture. The top agar was then poured and evenly spread on minimal agar plates followed by incubation at 37°C for 48 hours. Each concentration was tested in triplicate. Spontaneous (100 μL bacterial culture), positive control (100 μL bacterial culture + 100 μL mutagen) and negative control (100 μL bacterial culture + 100 μL extract) were also run.
Many classes of chemical carcinogens are activated by an oxidative reaction catalyzed by microsomal mixed function oxidases. The highest activity of these enzymes is found in liver. As bacteria have limited metabolic capability, so an exogenous activation system needs to be added. For this purpose, a rodent metabolic activation system was introduced into the test system.
16
Post-mitochondrial supernatant (S9 fraction) from rat liver homogenate was used to study the effect of extracts on the mutagenicity of indirect-acting mutagen that is 2AF. Male albino rats (150-200 g weight) were given 0.1% phenobarbitol (mixed-function oxidase inducer) in drinking water for 6 days. On the seventh day, the rats were sacrificed by cervical dislocation and livers were excised. The excised livers were weighed and washed several times with chilled sterilized 0.15 mol/L KCl solutions to ensure blood-free sterile preparation. The washed livers were transferred to beakers containing chilled 0.15 mol/L KCl solution (3 mL/g wet liver). Livers were minced and homogenized. The homogenate was centrifuged at 9000 rpm for 10 minutes, and the supernatant was decanted off in the flask and distributed in 2 mL cryovials and stored immediately in liquid nitrogen till needed. S9 mix (5%) was made on the day of experiment by adding sterile distilled water (16.75 mL), 0.2 mol/L sodium phosphate buffer, pH 7.4 (25 mL), 0.1 mol/L NADP (2 mL), 1 mol/L G-6-P (0.25 mL), MgCl2-KCl salt solution (1 mL), and rat liver S9 (5 mL). S9 mix was maintained at 0°C to −4°C while performing the experiment. S9 mix (0.5 mL), bacterial culture (0.1 mL), and mutagen (0.1 mol/L) were mixed with soft agar and poured onto the minimal plates. After 48 hours incubation, the number of revertent his+ bacteria colonies were scored. The inhibitory activity is expressed as percentage decrease in reverse mutation.
Statistical Analysis
The experimental data are presented as mean ± SE of 2 repeated experiments with triplicate plates/dose/experiment.
Results
In Ames assay, it was observed that all the extracts inhibited mutagenesis in a dose-dependent manner. A linear relationship was found between dose and percentage control of mutagenic activity. Better inhibition was found in pre-incubation mode of experimentation as compared to co-incubation. Negative control was used to check the toxicity of different concentrations of extracts against the bacteria. It was quite evident from the negative control that all the concentrations tested were nontoxic because a well-developed lawn with almost similar number of colonies as that of spontaneous was observed for all the extracts. For TA98 strain, the extracts showed 100% inhibition of mutagenicity at the highest concentration that is 1000 µg/0.1 mL per plate in both the modes of experimentation, except for 2 mol/L HCl extract, which exhibited maximum antimutagenicity of 34.81% (without metabolic activation) in co-incubation mode. This extract showed 54.02% antimutagenicity in pre-incubation mode at the same concentration that is 1000 µg/0.1 mL per plate (Table 1). Of the 4 extracts tested, 0.1 mol/L HCl extract was found to be the most effective followed by tris buffer extract. The extracts were also found to be effective in inhibiting the mutagenicity caused by sodium azide. However, the extent of inhibition with TA100 strain was slightly less as compared to TA98 strain. Tris buffer extract exhibited a percentage inhibition of 84.30% against indirect-acting mutagen, 2AF at lowest concentration tested that is 50 µg/0.1 mL per plate with TA 98 strain. Moreover, at concentration of 250 µg/0.1 mL per plate, this extract showed 100% inhibition of mutagenesis, which remained constant thereafter (Table 2). However, this extract exhibited the maximum inhibition of 54.79% with TA 100 strain with metabolic activation.
Effects of 2 M HCl Extract From Broccoli on Mutagenicity of NPD, Sodium Azide, and Aminoflourene in TA98 and TA100 Tester Strains of Salmonella typhimurium
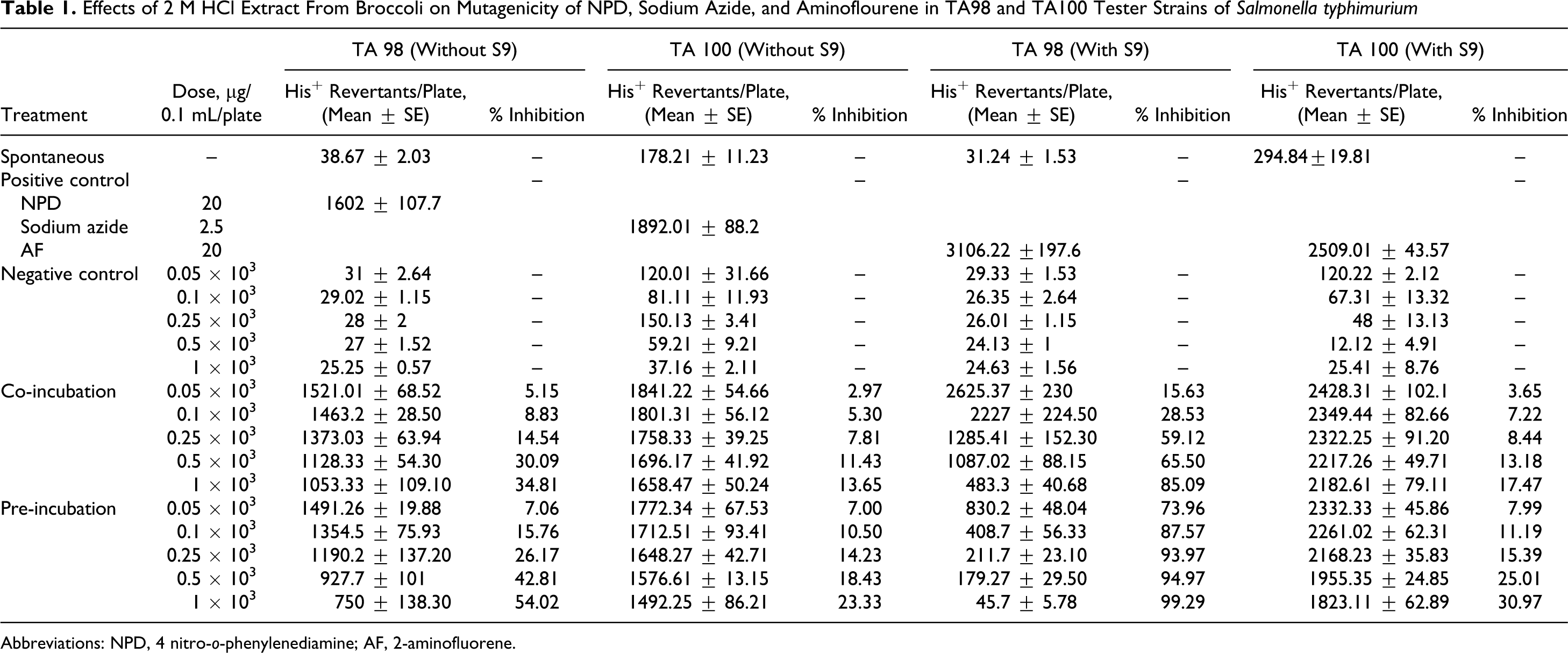
Abbreviations: NPD, 4 nitro-o-phenylenediamine; AF, 2-aminofluorene.
Effects of Tris Buffer Extract From Broccoli on Mutagenicity of NPD, Sodium azide, and Aminoflourene in TA98 and TA100 Tester Strains of Salmonella typhimurium
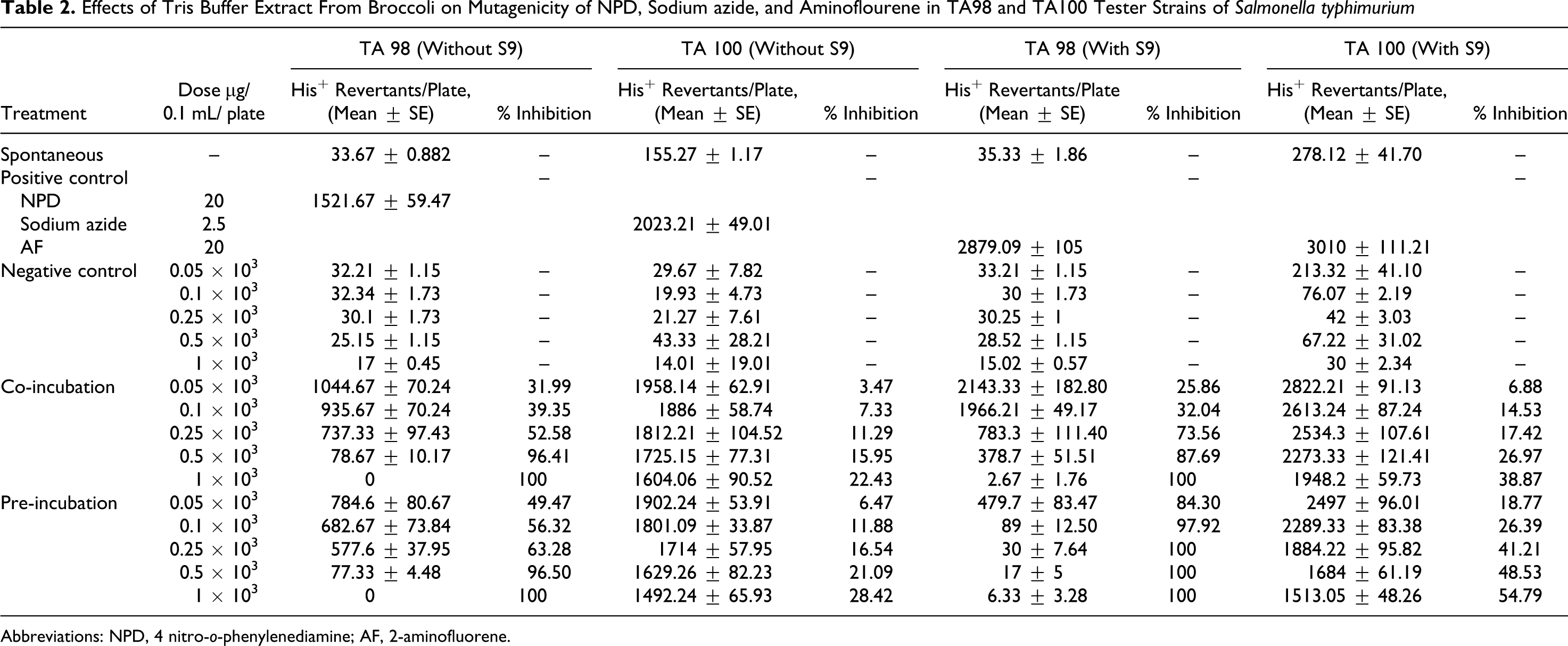
Abbreviations: NPD, 4 nitro-o-phenylenediamine; AF, 2-aminofluorene.
It was further observed that the extracts were more effective in the presence of S9 fraction from rat liver homogenate. The 2-mol/L HCl extract which exhibited maximum inhibition of just 54.02% showed 99.29% antimutagenicity with metabolic activation at the same concentration that is 1000 µg/0.1 mL per plate. With TA100 strain, the phosphate buffer extract showed maximum inhibition of 55.83% in pre-incubation mode with metabolic activation, whereas 0.1 mol/L HCl extract, which was found to be the most active with TA 100 strain, exhibited inhibition of 63.81% (Tables 3 and 4 ). The percentage inhibition by different extracts viz 2 mol/L HCl extract, tris buffer extract, phosphate buffer extract, and 0.1 mol/L HCl extract are represented in Tables 1, 2, 3, and 4, respectively.
Effects of Phosphate Buffer Extract From Broccoli on Mutagenicity of NPD, Sodium Azide, and Aminoflourene in TA98 and TA100 Tester Strains of Salmonella typhimurium
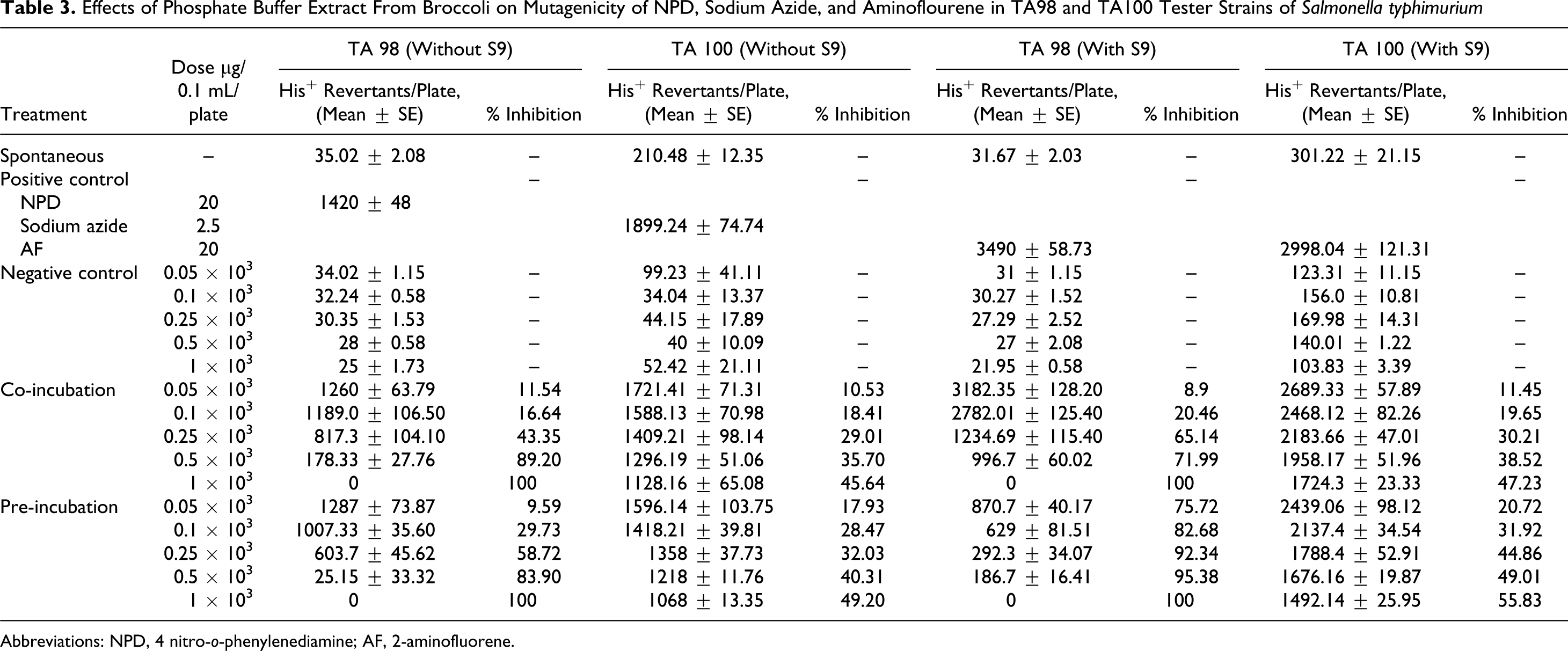
Abbreviations: NPD, 4 nitro-o-phenylenediamine; AF, 2-aminofluorene.
Effects of 0.1 M HCl Extract From Broccoli on Mutagenicity of NPD, Sodium Azide, and Aminoflourene in TA98 and TA100 Tester Strains of Salmonella typhimurium
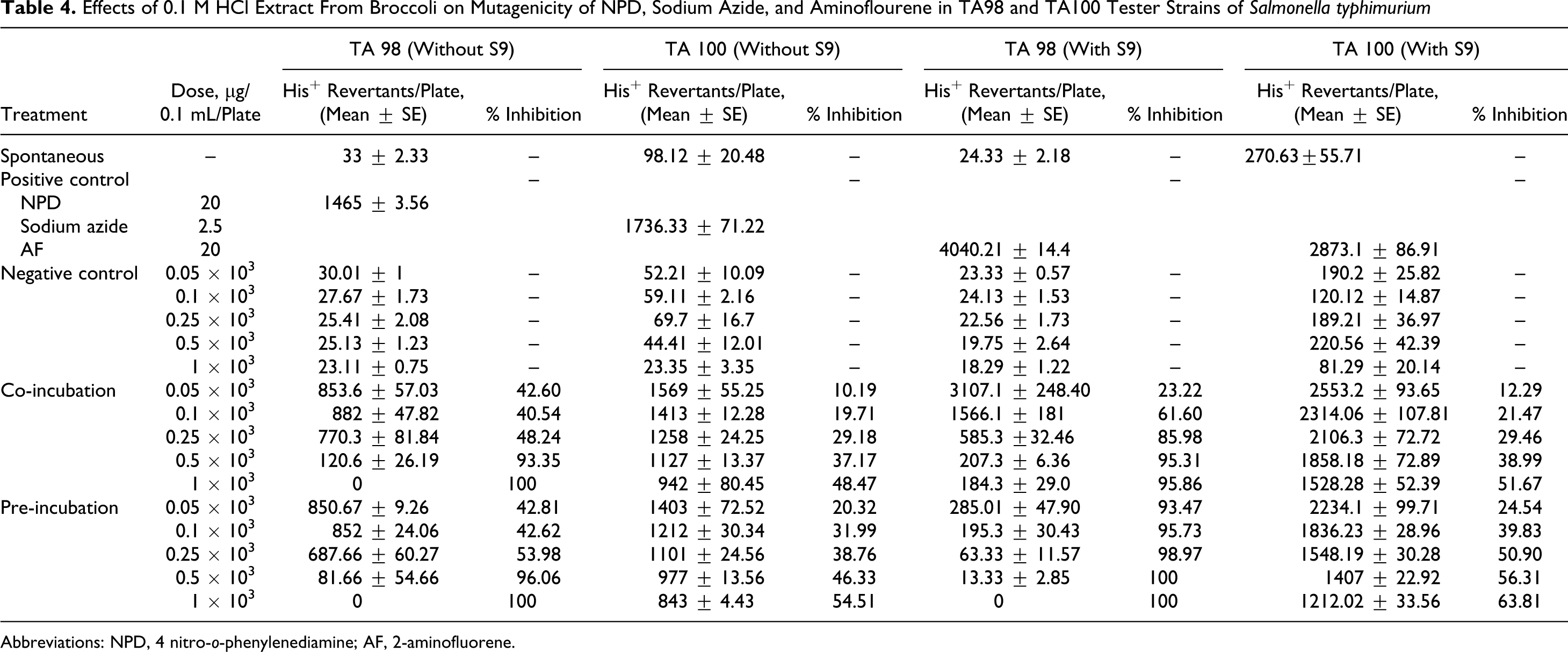
Abbreviations: NPD, 4 nitro-o-phenylenediamine; AF, 2-aminofluorene.
Discussion
The aims of the current study were to isolate glucosinolate hydrolytic products from the seeds of broccoli and to evaluate the antimutagenic activity of the extracts rich in these hydrolytic products. Broccoli was chosen as the study material because it is rich in phytochemicals that inhibit the activity of phase I enzymes and/or induce phase II detoxification system. 17 Broccoli contains higher quantities of glucoraphanin, precursor of SFN, which is an important inducer of anticarcinogenic protective enzymes. About 80% of the total glucosinolates in broccoli are present as glucoraphanin (4-methyl-sulphinylbutyl glucosinolate), an aliphatic glucosinolate whose hydrolytic product is SFN. Broccoli seeds contain glucoraphanin levels ranging from 5 to 100 μmol/g. 18 The isolation of glucosinolate degradation products was based on the fact that distinct hydrolytic products are formed at different pHs. Hydrolysis of glucosinolates was done in 4 different incubation solutions with distinct pHs. These incubation solutions produce extremely acidic (2 mol/L HCl), acidic (0.1 mol/L HCl), neutral (0.05 mol/L potassium phosphate buffer), and basic (0.05 mol/L Tris buffer) conditions, with neutral or basic pH favoring the formation of ITCs and acidic pH favoring the formation of nitrile. Sodium chloride and anhydrous sodium sulfate were used to make the extracts aqueous free.
Among all the extracts tested, 0.1 mol/L HCl extract was found to have the most significant antimutagenicity in both the strains. However, more pronounced effect was observed with TA98 tester strain having frameshift mutation, indicating that nitriles, the most predominant hydrolytic products formed at acidic pH, are the most active antimutagens against NPD. All other extracts were also found to possess strong antimutagenic potential against this mutagen. The extracts also effectively inhibited the mutagenicity of sodium azide. Furthermore, it was observed that all the extracts showed better inhibitory effect in the pre-incubation mode of experimentation, where the test sample was mixed with equal quantity of mutagen and incubated at 37°C for 30 minutes prior to plating on minimal agar plates. These better results in pre-incubation mode could be due to the increased time of contact between the mutagen and the test sample, allowing the plant extract to interact completely with the mutagen.
There are various reports available in literature, which suggest that chemopreventive activity of different hydrolytic products of glucosinolates is due to inhibition of the cytochrome P450-mediated bioactivation of chemical carcinogens. Hecht et al 19 conducted a clinical trial in smokers, which led to the conclusion that consumption of 170 g/d of watercress, which is rich in phenylethyl ITC, decreased the activation of a procarcinogen found in tobacco. Sulforaphane is one of the most explored ITCs and is currently under active investigation for its chemopreventive properties against various cancers. Mechanistic studies have shown that cancer chemopreventive activity of SFN is as a result of modification of the activity of xenobiotic metabolizing enzymes (phases I and II enzymes). Maheo et al 20 reported that SFN causes dose-dependent inhibition of CYP1A1 and CYP2B1/2 in rat hepatocytes. Similarly, in human hepatocytes, it decreased CYP3A4 activity by decreasing its messenger RNA (mRNA levels). 20 In another study conducted by Barcelo et al, 21 SFN was found to be a competitive inhibitor of CYP2E1, an enzyme involved in the metabolic activation of heterocyclic amines. In the current study, it has been observed that the extracts inhibited mutagenesis induced by the S9-dependent mutagen (2AF) more significantly than direct-acting mutagens (NPD and sodium azide). 2-Aminofluorene is a synthetic arylamine, and in biological systems, it can be converted into a well-known carcinogen, 2-acetylaminofluorene that directly interacts with DNA. This metabolism takes place in liver and cytochrome P450 enzymes catalyze this conversion of promutagen into active mutagen. 22 Hence, the results of this study can be correlated with the previous reports, suggesting that the inhibition of cytochrome P450 enzyme activity might be considered competent enough for restraining the metabolic activation of 2AF and thus protecting DNA against the damaging effects of active mutagen/carcinogen.
The results of this study revealed that the extracts of broccoli have potent antigenotoxic compounds. These results are in concurrence with the previous studies conducted on other plant parts of broccoli. Anupama et al 23 conducted a study to evaluate the antimutagenic effect of broccoli flower head against mitomycin-C-induced sister chromatid exchange in cultured human peripheral blood lymphocytes and reported the presence of potent antimutagenic principles in broccoli. Another study by Murugan et al 24 corroborates with the above-mentioned findings, wherein the antimutagenic potential of broccoli flower head was confirmed using Ames Salmonella reverse mutation assay. In the current study, it was observed that the extracts exhibited better activity against frameshift mutagen NPD. However, the results against the base pair mutagenesis induced by sodium azide also revealed good antimutagenic potential of extracts. Furthermore, because proteins are one of the prime targets for oxidative damage and cysteine residues are particularly sensitive to oxidation, the thiol group in cysteine can be oxidized to both reversible (sulfenic acid [SOH], disulfide bond formation [SOS]) and irreversible oxidative states (sulfinic [SO2H] and sulfonic acids [SO3H]). Cysteinyl residues, which are often critical to the maintenance of protein structure and enzymic activity, can be disrupted via the oxidation of the free thiol groups. 25 Although the thiol groups in cysteines are strongly buffered against oxidation by keeping the internal cellular environment in a relatively reduced state, the reactive metabolites can bind to the thiol groups. The inhibition of mutagenicity may be, therefore, due to direct inhibition of microsomal enzymatic activities, preventing bioactivation or binding of reactive metabolites to the protein SH group or other nucleophilic sites present in the active constituents of the extracts, thus eliminating any biological activity. Moreover, extracts were found to be genotoxically safe at the tested concentrations.
Conclusion
All the extracts exhibited effective antimutagenicity against mutagenesis induced by both the mutagens, suggesting that broccoli seeds contain potent antigenotoxic compounds. These preliminary investigations sturdily recommend more intensive evaluation of the chemoprotective effects of the different glucosinolates degradation products.
Footnotes
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: from Council of Scientific & Industrial Research (CSIR) in the form of SRF to Geetanjali Rampal and Tarunpreet Singh Thind.
Acknowledgments
Financial assistance from Council of Scientific & Industrial Research (CSIR) in the form of SRF to Geetanjali Rampal and Tarunpreet Singh Thind is duly acknowledged. The authors are also thankful to Guru Nanak Dev University, Amritsar, for providing the necessary laboratory facilities for the work.