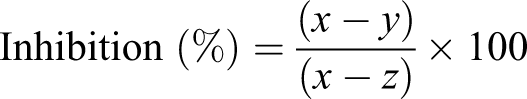Abstract
The current study aims to evaluate the antioxidative and antimutagenic activities of methanol extract and different fractions of Chukrasia tabularis leaves. The antioxidative potential was evaluated using 2,2′-azinobis (3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) cation and superoxide anion radical-scavenging assay. The antimutagenic potential was evaluated against direct-acting mutagens, 4-nitro-o-phenylenediamine and sodium azide; and S9-dependent mutagen, 2-aminofluorene in TA98 and TA100 strains of Salmonella typhimurium using Ames assay. It has been found that methanol extract and its fractions were more efficient against S9-dependent mutagen in pre-incubation mode of treatment as compared to direct-acting mutagens in both the strains. Methanol extract and its fractions also exhibited strong radical-scavenging potential. High-performance liquid chromatography (HPLC) analysis of methanol extract showed the presence of gallic acid, epicatechin, 7-hydroxycoumarin, and rutin. From the study, it could be concluded that antioxidative and antimutagenic activity of methanol extract and its fractions was related to the synergistic interactions among different chemical compounds.
Introduction
The mutation in DNA is a critical step in carcinogenesis as it leads to either arrest or induction of transcription, replication errors, induction of signal transduction pathways, and genomic instability, all of which are associated with carcinogenesis. 1 –3 It is widely hypothesized that oxidative DNA damage, occurring under the influence of reactive oxygen species (ROS), acts as a major contributor in the process of mutagenesis and carcinogenesis. 8-Hydroxy-2-deoxyguanosine (8-OHdG) is the most important DNA lesion that is formed due to the attack of ROS at C-8 position of 2-deoxyguanosine and is also considered the product of promutagenic base modification. 4 –6 The frequency of such modifications is high in oxidative stress and is beyond their repair by base excision repair mechanisms. The replication of DNA prior to the repair of this modified base (8-OHdG) result in GC to TA transversion mutation that are also reported in the genes whose dysfunction is involved in the carcinogenesis. 7,8
The p53 tumor suppressor gene and the ras family of proto-oncogenes are known to be important cancer-related genes. The GC to TA transversion mutations in these genes diminish their tumor-suppressing activity and the fact is justified due to their occurrence in more than 50% of human cancers. 9 Reactive oxygen species are also involved in the modification of other bases and the resultant bases such as 2-hydroxyadenine (2-OH-Ade), 8-hydroxyadenine (8-OH-Ade), 5-hydroxycytosine (5-OH-Cyt), and 5-hydroxyuracil (5-OH-Ura) are also found to be promutagenic due to miscoding potential. 10 In view of the above-mentioned facts and the induction of somatic mutations as a result of DNA adduct formation, oxygen free radicals might be considered an important class of carcinogens.
These ROS-induced mutations are the result of the exposure of living organisms to severe environmental conditions including high-altitude (excessive amount of UV radiations), ionizing radiations, and hazardous chemicals. The excessive cooking of food rich in proteins produces heterocyclic amines like Trp-P-1 (3-amino-1,4-dimethyl-5h-pyrido[4,3-b] indole), IQ (2 amino-3-methylimidazo [4,5-f] quinoline), and MeIQx (2 amino-3,8-dimethylimidazo [4,5-f] quinoxaline) that furthermore stimulate the generation of ROS. 11 –13 Besides cancer, the accumulation of these mutations results in development of aging, genetic defects in offspring (if mutations occur in germ line), and various degenerative diseases.
The effect of these mutagens and free radicals could be minimized or neutralized by increasing the exposure of living organism to antimutagenic/antioxidative agents especially with the help of secondary metabolites present in plants. The phytonutrients are important in the physiology of living organisms as they facilitate cell-to-cell communication, repair DNA damage arising from toxic exposure, enhance immune response, cause apoptosis in cancer cells, serve as antioxidants, and protect membrane polyunsaturated fatty acids from oxidation that causes biomembrane disruptions of cells and organelles. 14 –17 The dietary components also act as inhibitors of cytochrome P450-dependent mixed function oxidases and thus suppress the bioactivation of chemical carcinogens to genotoxic agents. 18 –20
The antimutagenic potential has been assessed by Ames assay given by Maron and Ames (1983) that has been proved to be valuable tool in the identification of DNA damages and its manifestation. 21 On the contrary, evaluation of antioxidative potential of these secondary metabolites against different in vitro generated radicals would help in understanding the possible mode of action against mutagenic agents. Considering these points, the current study was planned to investigate the antimutagenic properties of methanol extract of Chukrasia tabularis A Juss leaves and its different fractions against the direct-acting mutagens that is 4-Nitro-o-phenylenediamine (NPD) and sodium azide; S9-dependent mutagen that is 2-aminofluorne (2-AF) in TA98 and TA100 strains of Salmonella typhimurium. The antioxidative potential of different extract and fractions was evaluated using 2,2′-azinobis (3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) radical cation and superoxide anion radical-scavenging assays, and the identification of bioactive constituents in these extracts was done using high-performance liquid chromatography (HPLC) analysis.
Materials and Methods
Plant Material
Chukrasia tabularis A Juss (belonging to family Meliaceae) is commonly known as chickrassy, lal devdari, chittgong wood. The bark and leaves of the plant were reported to have biological activities including antipyretic, antimalarial, and antioxidative due to the abundance of secondary metabolites. 22,23 The current study aims to evaluate the antioxidant and antimutagenic activities of C tabularis leaves that were collected from the tree growing in the Guru Nanak Dev University campus, Amritsar. Botanical identification was made from herbarium of Department of Botanical and Environmental Sciences, Guru Nanak Dev University (GNDU), Amritsar, where a voucher specimen (accession No 6422/2236 dated April 7, 2006) was deposited.
Preparation of Extract and its Fractions
The leaves were thoroughly washed with tap water, dried at room temperature, and ground to fine powder. The powdered leaves were extracted with 80% methanol (MEL) using maceration method. The methanol extract so obtained was made aqueous and further fractionated using different solvents namely hexane, chloroform, ethyl acetate, and n-butanol to obtain hexane fraction (HFL), chloroform fraction (CFL), ethyl acetate fraction (EAFL), n-butanol fraction (BFL), and water fraction (WFL), respectively. The solvents of different polarities used while fractionation partitioned and separated the various metabolites of methanol extract per their solubility. The supernatant obtained was pooled and filtered after extracting the respective solvents, 3 times, using Whatman no 1 sheet and concentrated by vacuum rotary evaporator (Strike 202, Stereo glass, Italy) followed by lyophilization to obtain the dry residue from the respective fractions (Flowchart 1). The extracts and fractions were then evaluated for their antioxidative and antimutagenic activities in different in vitro assays.
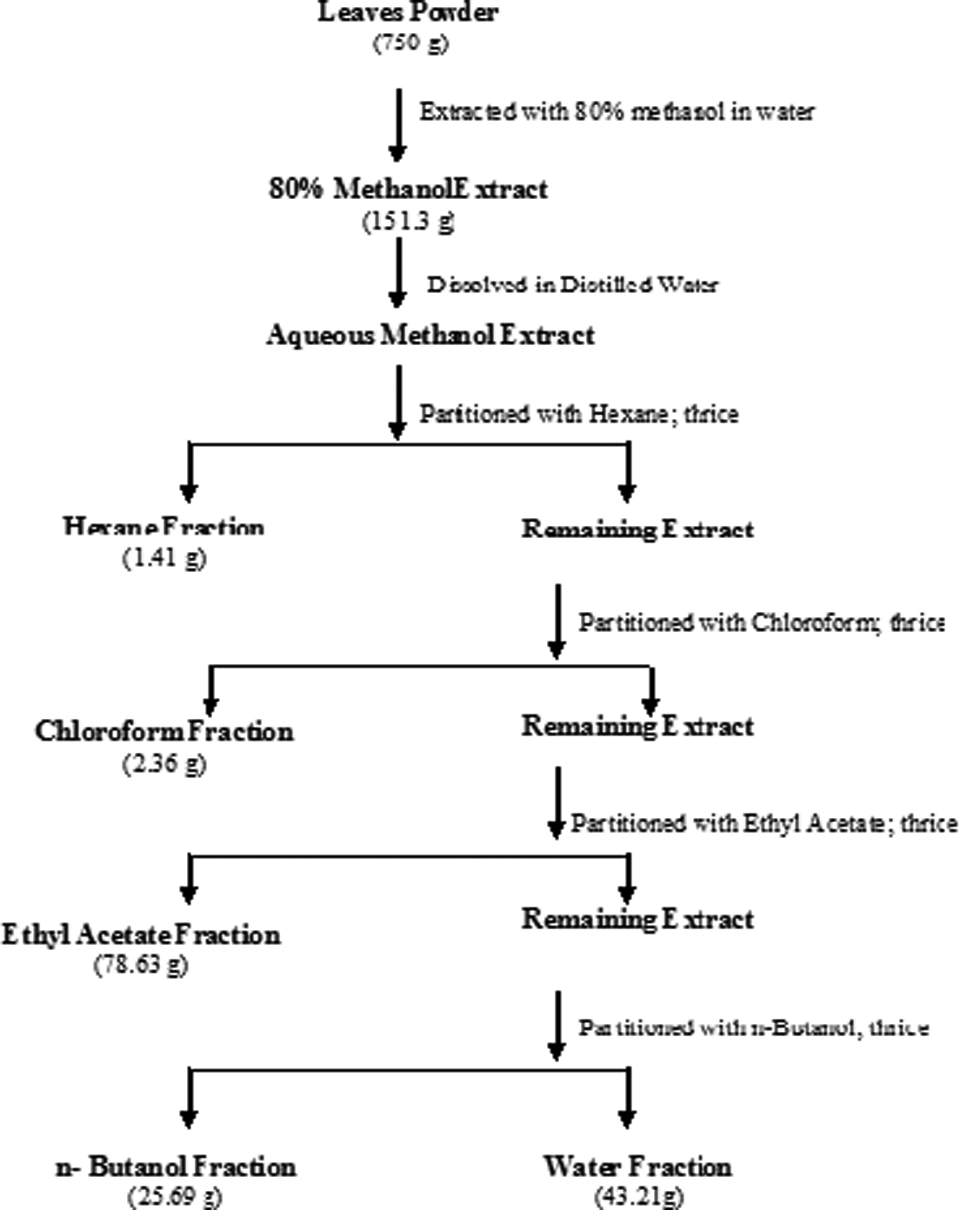
Extraction procedure of leaves of Chukrasia tabularis
Chemicals
2,2′-azinobis (3-ethylbenzothiazoline-6-sulfonic acid), NPD, sodium azide, and 2-aminofluorene (2-AF) were obtained from Sigma Chemical Co (St Louis, Missouri). All chemical reagents (potassium persulfate, ethanol, phenazine methosulfate [PMS], reduced nicotinamide adenine dinucleotide [NADH], nitroblue tetrazolium chloride [NBT], monobasic dihydrogen phosphate, dibasic monohydrogen phosphate, sodium chloride, histidine, biotin, glucose, calcium chloride, potassium chloride, magnesium chloride, magnesium sulphate, ammonium chloride, glucose-6-phosphate, reduced nicotinamide adenine dinucleotide phosphate [NADPH], agar, luria broth, etc) and solvents (hexane, methanol, ethyl acetate, n-butanol, chloroform, dimethyl sulfoxide [DMSO]) were of analytical grade. Tester strains TA98 and TA100 of Salmonella typhimurium was procured from Institute of Microbial Technology (CSIR), Chandigarh, India.
For HPLC analysis, gallic acid, catechin, epicatechin, rutin, 7-hydroxycoumarin, quercetin, were purchased from Sigma-Aldrich Bangalore, Chromadex, Bangalore, Life Technology, New Delhi, India. All HPLC grade solvents (methanol, water) were purchased from JT Baker, Mumbai. Trifluoroacetic acid was purchased from E Merck, Mumbai, India.
Antioxidative Studies
2,2′-Azinobis (3-ethylbenzothiazoline-6-sulfonic acid) radical cation-scavenging assay
2,2′-Azinobis (3-ethylbenzothiazoline-6-sulfonic acid) radical cation-scavenging assay given by Re et al was followed to determine the hydrogen-donating ability of extracts and fractions.
24
ABTS.+ were generated by mixing 2 reagents (7 mmol/L of ABTS and 140 mmol/L of potassium persulfate) in a proportion to make the final concentration of potassium persulfate to 2.45 mmol/L in mixture. The reaction mixture was kept at 30°C for 12 to 16 hours in dark. After 16 hours, the reaction mixture was diluted with ethanol or phosphate buffered saline ([PBS] pH = 7.4) to obtain the absorbance of 0.700 ± 0.020 at 734 nm. ABTS+ so formed was stable for more than 2 days when stored in dark at room temperature. To determine ABTS+- scavenging ability, 1.9 mL of ABTS+ solution was mixed with 0.1 mL of extract or fraction solution, and the absorbance was measured spectrophotometrically for 0 to 6 minutes at 734 nm. The absorbance of ABTS+ blank solution was measured in each assay to correct for radical decay, and rutin being a phenolic compound was used as positive control. All tests were performed in triplicate. The percentage inhibition of absorbance at 734 nm was calculated using the formula:
Superoxide anion-scavenging assay
For assessing the superoxide anion-scavenging ability of different extracts/fractions of C tabularis, the method described by Nishikimi et al (1972) was followed with slight modifications.
25
The superoxide anions were generated nonenzymatically in a phenazine methosulphate-NADH system and assessed by development of blue formazan dye on reduction of nitro blue tetrazolium. Briefly, 1 mL of plant extract or fractions of different concentrations (20-200 µg/mL) was mixed with 156 µmol/L NADH (1 mL), 60 µmol/L NBT (1 mL), and 468 µmol/L phenazine methosulphate (1 mL) in phosphate buffer (pH = 8.3). The reaction was initiated with the addition of PMS. The reaction mixture was incubated at 25°C for 10 minutes. The absorbance of colored complex was measured at 560 nm and the inhibition percentage was calculated using the formula.
Antimutagenic Studies
The antimutagenicity of the methanol extract of C tabularis and its different fractions was determined using Salmonella histidine point mutation assay proposed by Maron and Ames (1983) with slight modifications suggested by Bala and Grover. 21,26 Two methodologies were followed, that is co-incubation and pre-incubation to characterize antimutagenicity to desmutagenicity or bioantimutagenicity. In co-incubation method, 100 μL each of bacterial culture, mutagen, and different concentrations of extract was added to 2 mL of top agar (45°C). In pre-incubation method, equal volume of mutagen and extract was mixed and incubated at 37°C for 30 minutes. This mixture, 200 μL, was added to 2 mL of top agar with 100 μL of bacterial culture. 4-Nitro-o-phenylenediamine (20 µg/0.1 mL per plate) for TA98 strain and sodium azide (2.5 µg/0.1 mL per plate) was used as direct-acting mutagen for TA98 and TA100 strain, respectively. The top agar was then poured and evenly spread on minimal agar plates followed by incubation at 37°C for 48 hours. Spontaneous (0.1 mL bacterial culture), positive control (0.1 mL bacterial culture + 0.1 mL mutagen either direct-acting or indirect-acting) and negative control (0.1 mL bacterial culture + 0.1 mL extract) were also run. The concentrations of extract and fractions used for investigating the antimutagenicity were 100, 250, 500, 1000, 2500 µg/0.1 mL per plate, and each concentration was tested in triplicate.
The effect of extracts and fractions on the mutagenicity of indirect-acting mutagen that is 2-AF (20 µg/0.1 mL per plate) was studied using post-mitochondrial supernatant (S9 fraction) of rat liver homogenate. The metabolic activation was provided to indirect-acting mutagen (2-AF) by the addition of 0.5 mL of S9 mix (5%) per plate along with 100 μL mutagen.
Preparation of S9 fraction
The S9 fraction was prepared from rat liver homogenate per the method given by Garner et al (1972). 27 For this, male albino rats (150-200 g weight), was given 0.1% phenobarbitol (mixed-function oxidase inducer) in drinking water for 6 days. On the seventh day, the rats were sacrificed by cervical dislocation and livers were excised out. Livers were weighed and washed several times with chilled sterilized 0.15 mol/L KCl solutions to ensure blood free sterile preparation. The washed livers were transferred to beakers containing chilled 0.15 mol/L KCl solution (3 mL/g wet liver). Livers were minced and homogenized. The homogenate was centrifuged at 9000 rpm for 10 minutes, and supernatant was decanted off in the flask and distributed in 2 mL cryovials and stored immediately in the liquid nitrogen till needed. The sterility of the preparation is determined by plating (0.1 mL) on minimal agar plates containing histidine. S9 mix (5%) was made on the day of experiment and maintained at 0°C to 4°C. S9 mix (0.5 mL), bacterial culture (0.1 mL), and mutagen (0.1 mL) were mixed with top agar and poured onto the minimal plates. After incubation for 48 hours, the number of revertent his + bacteria colonies was scored. Extract solution, 0.1 mL, of of different concentration was added to above said mixture while assessing the antimutagenic potential.
The antimutagenic activity of each extract and fraction was expressed as percentage decrease in reverse mutations as follows:
Toxicity of extracts
The toxicity of extract and fractions of C tabularis leaves was valuated by running negative control for each concentration of extracts and fractions used (100, 250, 500, 1000, 2500 μg/0.1 mL per plate). For toxicity testing, 0.1 mL of bacterial culture and 0.1 mL of extract solution was added to 2 mL of top agar and poured onto the minimal plates. After incubation for 48 hours, the number of revertant his + bacteria colonies was scored.
Chemical Characterization Using HPLC
Sample preparation
For HPLC analysis, 100 mg of leaves powder was taken and extracted with methanol; thrice. The supernatant was collected and dried on rotary vacuum evaporator at 40°C. The resultant dried extract was dissolved in solvent (methanol: water) and analyzed for the presence of known standard compounds using the linear gradient elution method.
Preparation of standard solution
Standard stock solution of phenol was prepared by dissolving gallic acid (1 mg), catechin (2 mg), epicatechin (3 mg), 7-hydroxycoumarin (1 mg), quercetin (5 mg), and rutin (5 mg) in 2 mL of methanol:water (90:10).
Apparatus and chromatographic conditions
High-performance liquid chromatogrphy analysis was performed on a Shimadzu Prominence HPLC system, equipped with LC-20AT quaternary gradient pump, SPD-M20A diode array detector (DAD), CBM-20A communication bus module, CTO-10AS VP column oven, Rheodyne injector, and Shimadzu LC solution (ver 1.21 SP1) software. Chromatography was carried out on a Lichrocart 250-4 column from Lichrospher (250 mm × 4.0 mm × 5µmol/L particle size). At a column temperature of 27°C and a flow rate of 1 mL/min using solvent A (0.03% trifluroacetic acid [TFA] in water) and solvent B (methanol) with a linear gradient elution: 25% B (4 minutes), 25% to 40% (6 minutes), 40% to 50% B (8 minutes), 50% to 45% (12 minutes), 45% to 35% B (12 minutes), 35% to 20% B (15 minutes), 20% to 5% B (18 minutes), and 5% to 25% B (20 minutes) at λ 280 nm.
Statistical Analysis
The experimental data were expressed as mean ± standard error (SE). One-way analysis of variance (ANOVA) and Tukey multiple comparison and Tukey Honestly Significant Difference (HSD) post hoc test were carried out to determine significant differences (P ≤ .05) between the means.
Results
Antioxidative Studies
Figures 1 and 2 show the hydrogen donating and superoxide anion radical-scavenging potential of methanol extract of C tabularis leaves and its different fractions.
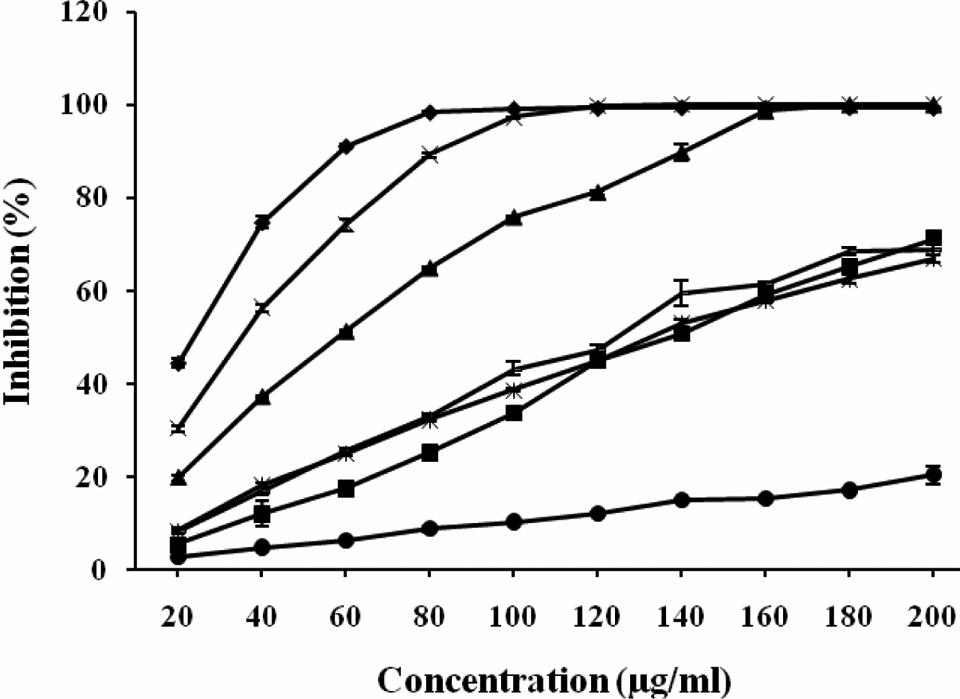
2,2′-Azinobis (3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) radical cation-scavenging potential of methanol extract of Chukrasia tabularis leaves and its different fractions. The results are expressed as inhibition (%) ± SE. Here, ▪ = rutin, × = methanol extract of leaves, • = hexane fraction of leaves, — = chloroform fraction of leaves, ♦ = ethyl acetate fraction of leaves, ▴ = Butanol fraction of leaves, and = water fraction of leaves.
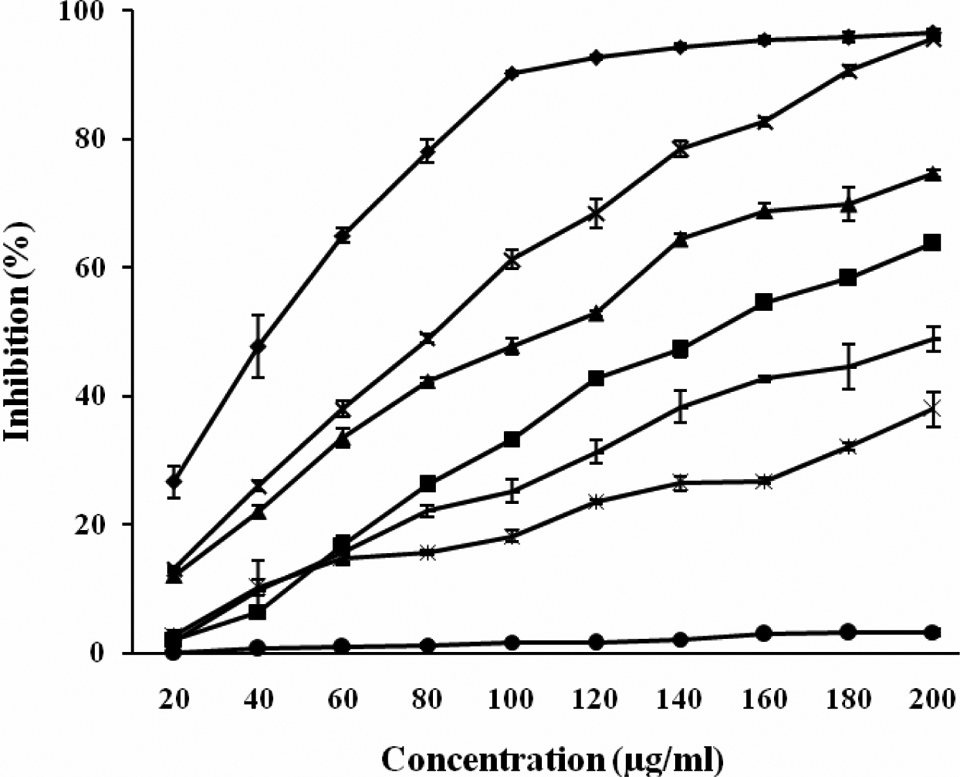
Superoxide anion radical-scavenging potential of methanol extract of Chukrasia tabularis leaves and its different fractions. The results are expressed as inhibition (%) ± SE. Here, ▪ = rutin, × = methanol extract of leaves, • = hexane fraction of leaves, — = chloroform fraction of leaves, ♦ = ethyl acetate fraction of leaves, ▴ = butanol fraction of leaves, and = water fraction of leaves.
ABTS radical cation-scavenging assay
Figure 1 presents the ABTS+-scavenging ability of different extracts and fractions of C tabularis leaves in comparison to standard antioxidant compounds that is rutin. It was found that ethyl acetate fraction of leaves (EAFL) exhibited an inhibition value of 98.54% at 80 μg/mL. The MEL and BFL has comparatively lower activity with inhibition value that is 89.27% and 64.87%, respectively, at the same concentration. However, CFL, WFL, and HFL were found to be least effective as they exhibited inhibitory effect of 33.22%, 32.44%, and 8.79%, respectively, at the same concentration that is 80 μg/mL. All the extracts and fractions have exhibited the hydrogen donation ability in a dose-dependent manner. Among different extract/fractions of leaves, EAFL was found to be most effective one with an IC50 value of 14.394 μg/mL. The IC50 value of EAFL was found to be even lower than standard antioxidant compound used that is rutin (IC50 value = 139.851 μg/mL that corresponds to its highest hydrogen donating activity than rutin. The significance of the results obtained and concentrations used was also checked using one-way ANOVA and Tukey HSD post hoc test and the results were found to be statistically significant at P ≤ .05.
Superoxide anion radical-scavenging assay
Figure 2 depict the superoxide anion-scavenging ability of different extract and fractions of C tabularis leaves. It was found that among different extract and fractions, EAFL was the most effective one with an inhibitory effect of 90.24% at 100 µg/mL, whereas MEL exhibited 61.25% inhibition at the same concentration. The effect of BFL and CFL was moderate as they possessed the inhibitory activity of 47.82% and 25.27%, respectively, at the same concentration that is 100 µg/mL. Water fraction of leaves showed 18.26% scavenging of superoxide anion radicals whereas HFL was least effective with an inhibitory effect of 1.771% at 100 µg/mL concentration. The inhibitory effect that EAFL possessed was found to be higher than rutin that exhibited O2-scavenging potential of 33.37% at 100 µg/mL (Figure 2). The different extracts and fractions showed superoxide anion radical-scavenging potential in a dose-dependent manner. The results obtained were found to be statistically significant at P ≤ .05, using one-way ANOVA and Tukey HSD post hoc test.
Antimutagenic Studies
It was found that methanol extract of C tabularis leaves and its different fractions were nontoxic at all the concentrations used in the experiments, as the number of revertant colonies in negative control plates were found to be similar to that of spontaneous revertant colonies (For TA98: 25-60 and for TA100: 100-240). The extract and its different fractions showed moderate-to-strong inhibitory activity in a dose-dependent manner and the highest activity was recorded against indirect-acting mutagens that is 2-AF, in the presence of S9 mixture and the ethyl acetate fraction was found to be the most effective one among different extracts and fractions.
Figure 3 shows that the methanol extract of C tabularis leaves exhibited significant antimutagenic response against both the mutagens that is NPD and 2-AF in TA98 strain. It was observed that the MEL was more effective when pre-incubated with mutagens. The MEL showed 51.73% and 47.48% reduction in the number of his+ revertants induced by NPD in pre-incubation and co-incubation treatments, respectively, at the highest dose tested (2500 μg/0.1 mL per plate). It completely reduced the S9-mediated mutagenicity of 2-AF at the dose of 1000 μg/0.1mL per plate in pre-incubation treatment, whereas in co-incubation mode of experimentation about 99.61% inhibition was recorded at the highest tested dose of 2500 μg/0.1 mL per plate.
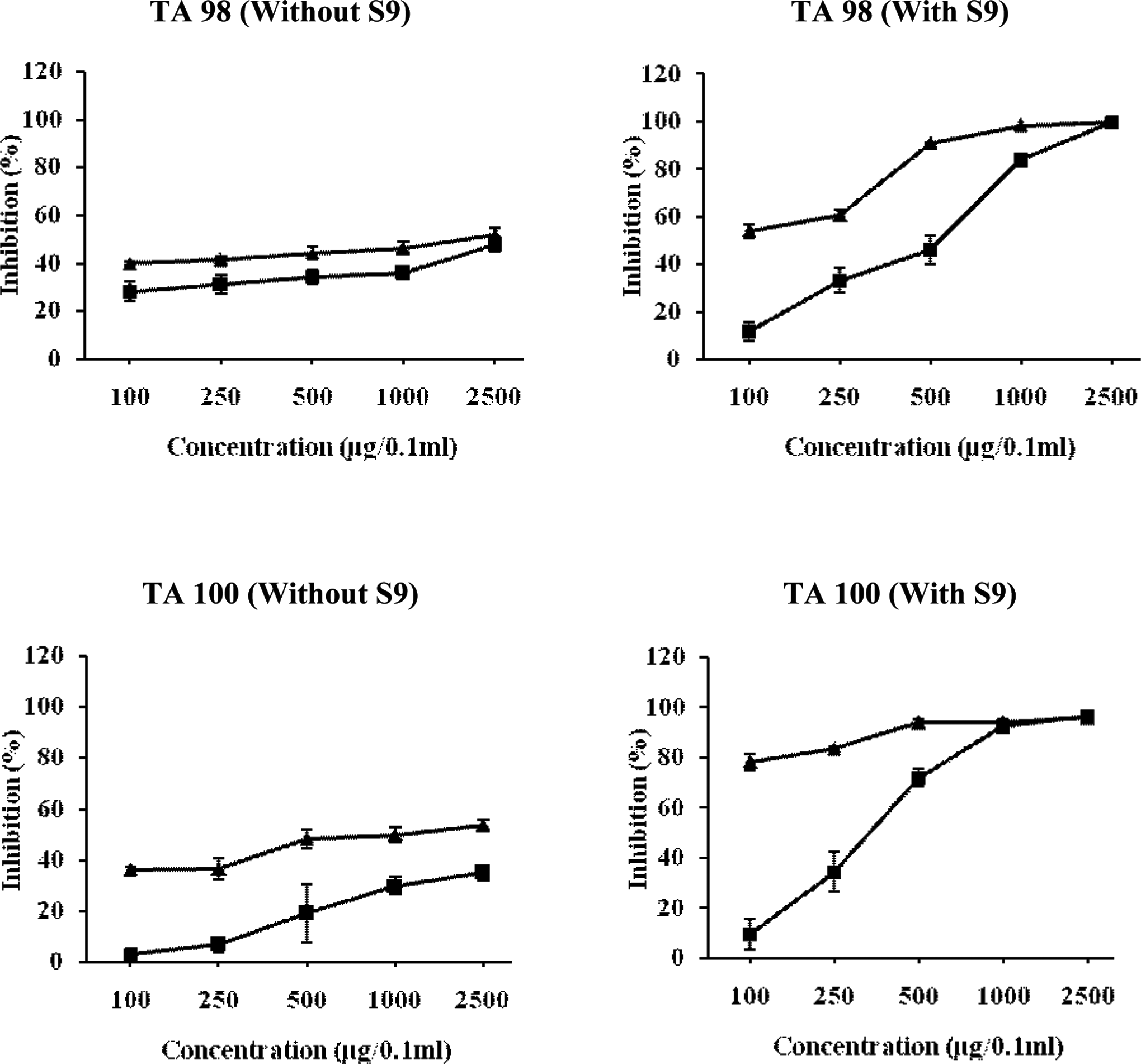
Relationship between different concentrations of methanol extract of Chukrasia tabularis leaves and antimutagenic mutagenic activity of NPD, sodium azide, and 2-AF in TA98 and TA100 tester strains of Salmonella typhimurium. The results are expressed as inhibition (%) ± SE. Here, ▴ = pre-incubation mode of experimentation, and ▪ = co-incubation mode of experimentation. 2-AF indicates 2-aminofluorne; NPD, 4-Nitro-o-phenylenediamine.
In TA100 strain, a maximum of 53.76% reduction in sodium azide-induced mutagenesis was observed when the extract (MEL) was pre-incubated with sodium azide as compared to moderate reduction of 34.95% at the highest concentration in co-incubation mode of treatment. However, against indirect-acting mutagens, pre-incubation yielded greater antimutagenic response of 93.95% inhibition of his+ revertants at 500 μg/0.1 mL per plate with an ID50 value of 0.6598 μg/0.1 mL per plate (as calculated from regression equation), whereas in co-incubation treatment, the MEL exhibited an ID50 value of 344.127 μg/0.1 mL per plate concentration (Figure 3).
Figure 4 shows the effect of hexane fraction of C tabularis leaves on NPD and sodium azide-induced mutations in TA98 and TA100 strains. A moderate inhibitory effect of 32.74% was observed in pre-incubation mode of experimentation against NPD, whereas in co-incubation treatment, HFL showed weak inhibitory effect of 20.57% at a dose of 2500 μg/0.1 mL per plate. The HFL showed weak-to-moderate antimutagenic activity against sodium azide-induced mutagenicity in TA100 strain. However, the HFL strongly arrested the mutagenicity of promutagen 2-AF in the presence of microsomal mammalian activation system. In TA98 strain, a maximum of 71.76% and 94.32% inhibitory effect was observed in co- and pre-incubation assays, respectively, at the highest dose (2500 μg/0.1 mL per plate), whereas in TA100 strain, the antimutagenic effect of HFL in terms of ID50 value was found to be 177.36 μg/0.1 mL and 26.257 μg/0.1 mL in co- and pre-incubation experiments, respectively (Figure 4).
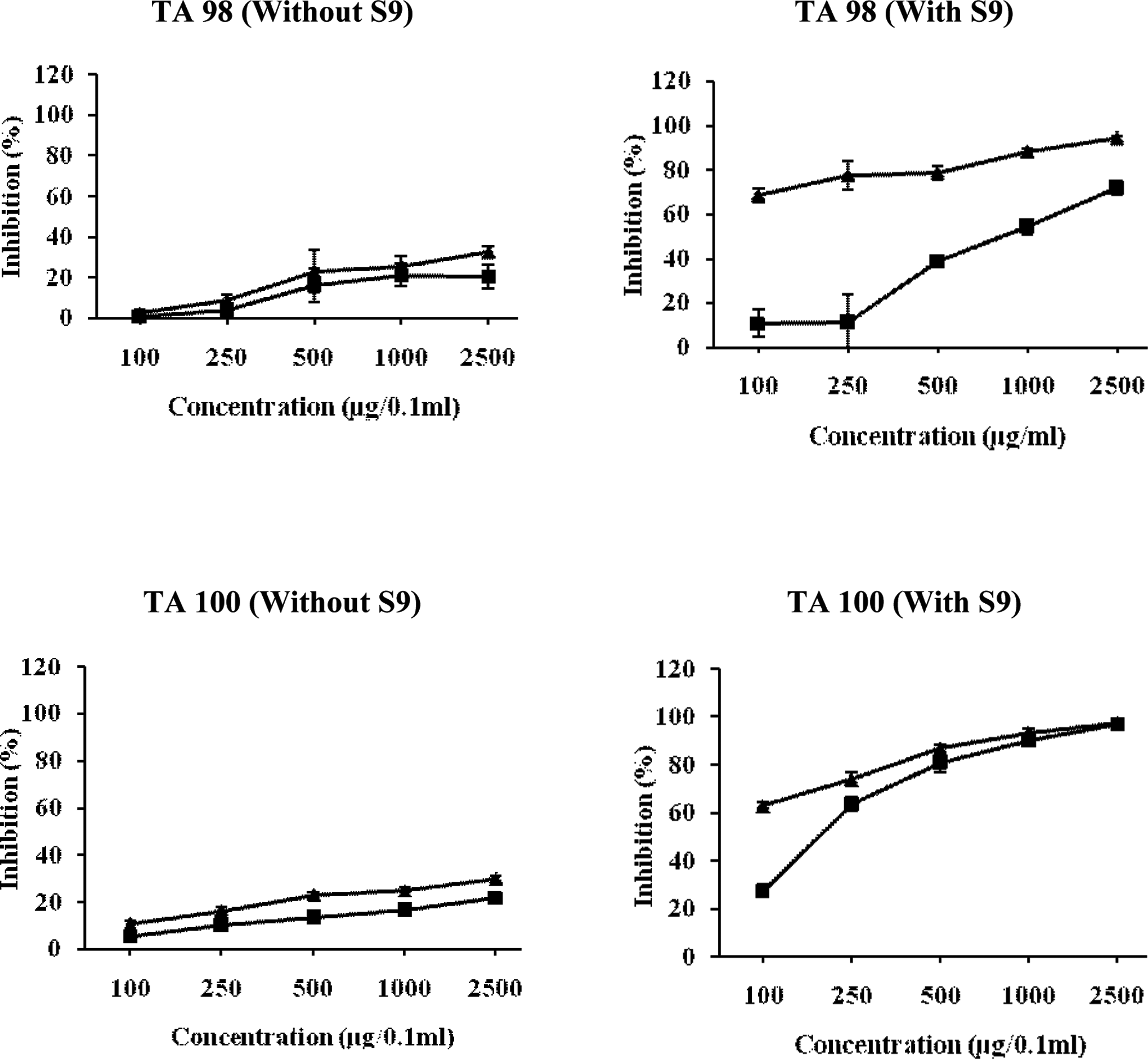
Relationship between different concentrations of hexane fraction of Chukrasia tabularis leaves and antimutagenic mutagenic activity of NPD, sodium azide, and 2-AF in TA98 and TA100 tester strains of Salmonella typhimurium. The results are expressed as inhibition (%) ± SE. Here, ▴ = pre-incubation mode of experimentation and ▪ = co-incubation mode of experimentation. 2-AF indicates 2-aminofluorne; NPD, 4-Nitro-o-phenylenediamine.
Figure 5 reveals that chloroform fraction of C tabularis lowered the number of NPD-induced his+ revertants maximally by 57.11% in the TA98 strain in pre-incubation treatment, whereas in co-incubation treatment, the number of his+ revertants were reduced by 46.57%. The results obtained in TA100 strain were quite similar to that of TA98 strain with reduction of 52.55% of sodium azide-induced mutagenesis in the pre-incubation mode of experimentation, whereas in co-incubation mode, it exhibited a maximum reduction of 38.66% at the highest dose (Figure 5). The CFL also exhibited strong antimutagenicity with maximum reduction of 96.64% against the promutagen-induced his+ revertants in pre-incubation studies at a dose of 2500 μg/0.1 mL per plate in TA98 strain, whereas in co-incubation mode, the maximum reduction was 88.94% at the highest tested dose. In TA100 strain, the CFL suppressed 2-AF-induced mutagenicity by 86.67% and 91.00% in co-incubation and pre-incubation assays, respectively, with an ID50 value of 12.426 μg/0.1 mL and 0.045 μg/0.1 mL (Figure 5).
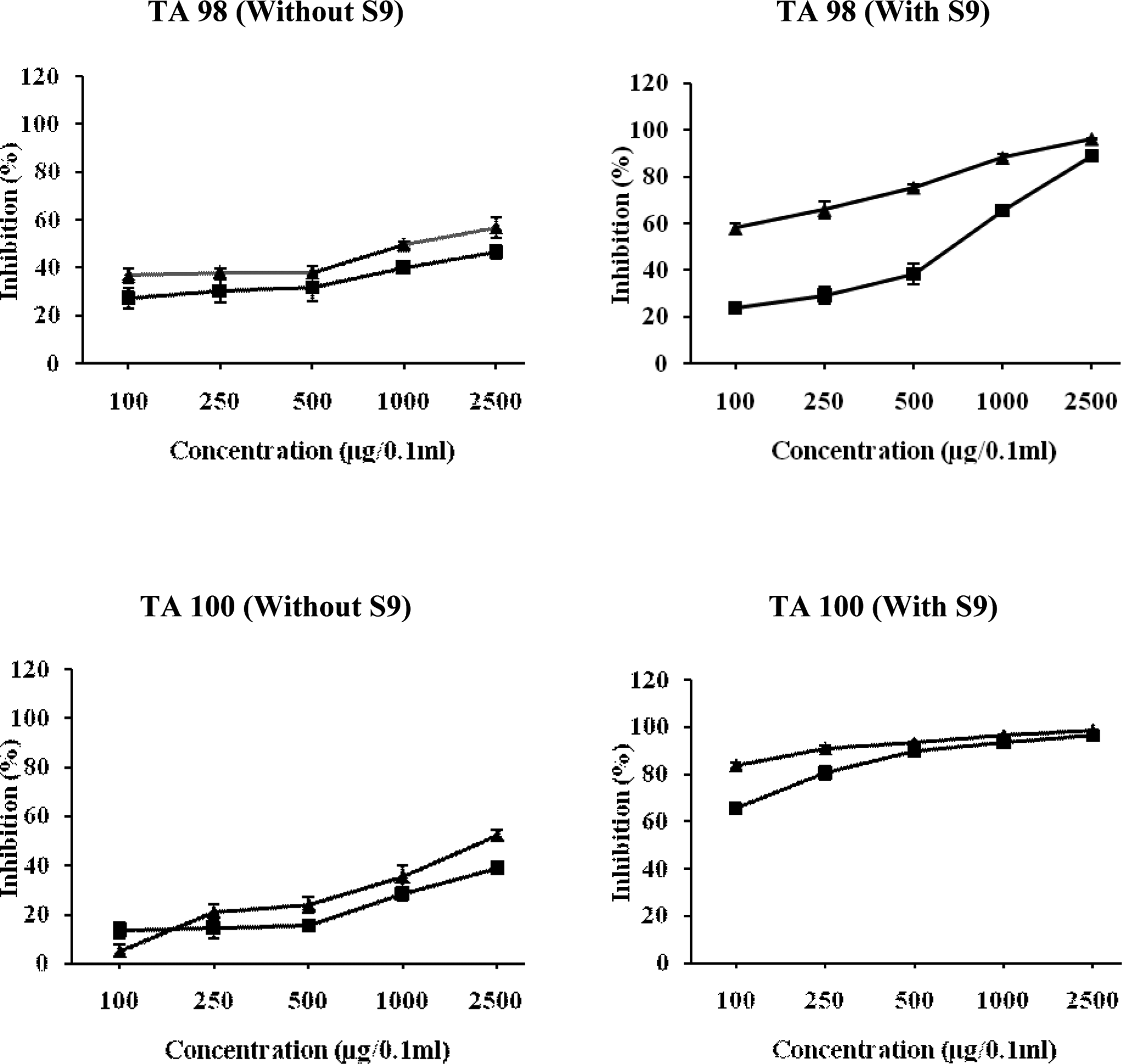
Relationship between different concentrations of chloroform fraction of Chukrasia tabularis leaves and antimutagenic mutagenic activity of NPD, sodium azide, and 2-AF in TA98 and TA100 tester strains of Salmonella typhimurium. The results are expressed as inhibition (%) ± SE. Here, ▴ = pre-incubation mode of experimentation and ▪ = co-incubation mode of experimentation. 2-AF indicates 2-aminofluorne; NPD, 4-Nitro-o-phenylenediamine.
Figure 6 shows that EAFL exhibited moderate inhibitory effect on the mutagenicity of direct-acting mutagen, NPD at the highest tested dose (2500 μg/0.1 mL per plate) with an inhibition of 42.68% in the pre-incubation approach, whereas in co-incubation experiment, it showed an inhibitory effect of 30.9%. Furthermore, it was observed that at the maximum dose tested, antimutagenic effect against sodium azide was more prominent than in NPD with an inhibitory effect of 80.95% and 77.80%, respectively, in co- and pre-incubation treatments (Figure 6). However, against indirect-acting mutagens (2-AF), the EAFL almost completely inhibited the mutagenicity in TA98 strain at 500 μg/0.1 mL per plate concentration in pre-incubation treatment, whereas in co-incubation treatment, it showed almost similar inhibitory effect but at the highest tested dose (2500 μg/0.1 mL per plate). In TA100 strain, the diminution of number of revertant colonies was found to be 98.50% in the pre-incubation and 85.37% in co-incubation mode of treatment at the concentration of 250μg/0.1 mL per plate with an ID50 value of 2.436 × 10−21 μg and 18.065 μg, respectively (Figure 6).
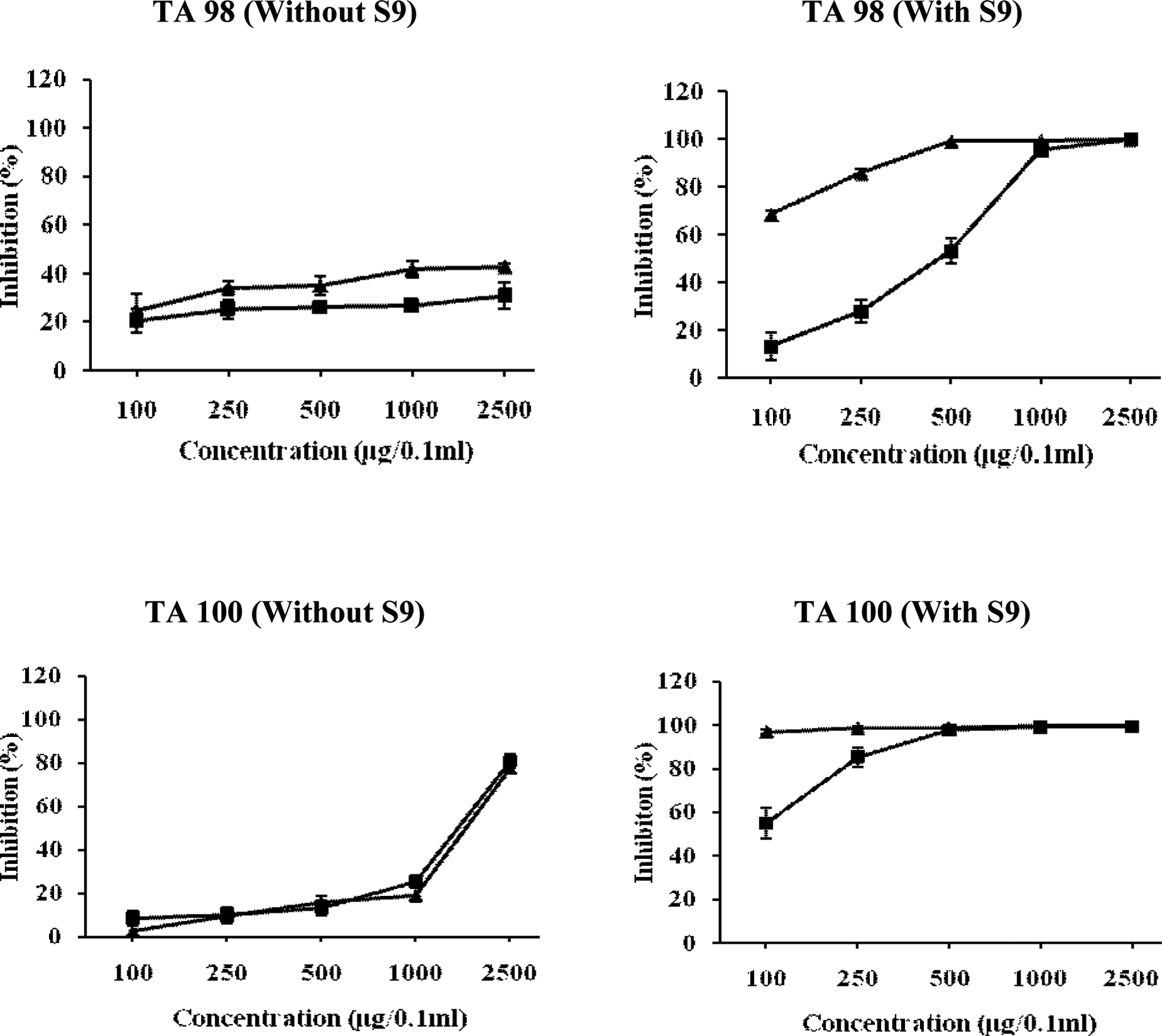
Relationship between different concentrations of ethyl acetate fraction of Chukrasia tabularis leaves and antimutagenic mutagenic activity of NPD, sodium azide, and 2-AF in TA98 and TA100 tester strains of Salmonella typhimurium. The results are expressed as inhibition (%) ± SE. Here, ▴ = pre-incubation mode of experimentation and ▪ = co-incubation mode of experimentation. 2-AF indicates 2-aminofluorne; NPD, 4-Nitro-o-phenylenediamine.
Figure 7 shows that the highest dose (2500 μg/0.1 mL per plate) of butanol fraction reduced the number of his+ revertants induced by NPD in TA 98 strain by 37.83% and 40.64% in the co- and pre-incubation experiments, respectively. In TA100 strain, the modulatory effect was found to be very weak that is about 18.59% and 34.94% in co-incubation and pre-incubation treatment, respectively, at 2500 μg/0.1 mL per plate. However, butanol fraction significantly inhibited the 2-AF-induced mutations in TA98 and TA100 strains in a dose-dependent manner. The pre-incubation of 2-AF with S9 mix and BFL prior to plating increased the antimutagenic effect to 97.97% in TA100 strain, whereas in TA98 strain, the butanol fraction of leaves reduced the promutagen-induced his+ revertants by 98.56% and 95.82% in pre- and co-incubation studies, respectively, at a dose of 500 μg/0.1 mL per plate (Figure 7).
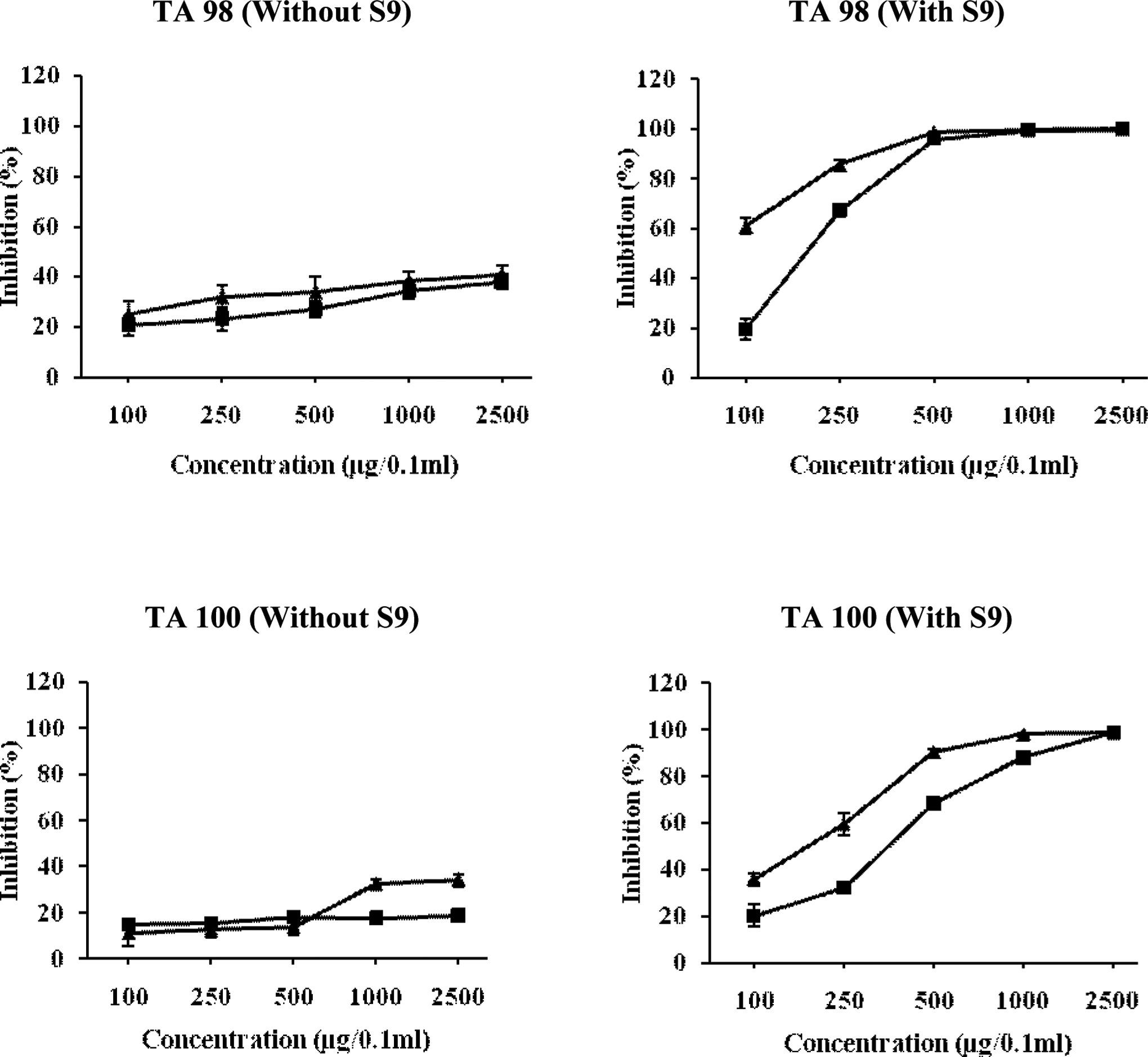
Relationship between different concentrations of butanol fraction of Chukrasia tabularis leaves and antimutagenic mutagenic activity of NPD, sodium azide, and 2-AF in TA98 and TA100 tester strains of Salmonella typhimurium. The results are expressed as inhibition (%) ± SE. Here, ▴= pre-incubation mode of experimentation and ▪ = co-incubation mode of experimentation. 2-AF indicates 2-aminofluorne; NPD, 4-Nitro-o-phenylenediamine.
Figure 8 shows that water fraction of leaves was more effective in TA98 strain as compared to TA100 strain. The pre-incubation of NPD with water fraction reduced the number of his+ revertants by 33.83% at highest tested dose (2500 μg/0.1 mL per plate), whereas in co-incubation treatment, the inhibitory effect was found to be 43.54% at same dose. It is also clear from Figure 8 that in TA 100 strain, water fraction of leaves showed dose dependent, albeit a weak response with reduction in number of his+ revertants only by 16.95% and 18.78% in the co- and pre-incubation modes of experimentation, respectively, at the highest tested dose (2500 μg/0.1 mL per plate). The water fraction significantly inhibited the activity of 2-AF with an inhibitory effect of 62.90% and 94.78%, respectively, in co-incubation and pre-incubation mode of treatments at a dose of 2500 μg/0.1 mL per plate in TA98 strain. In TA100 strain, the WFL exhibited strong antimutagenic activity with an inhibitory effect of 98.28% and 99.11% in co- and pre-incubation modes of treatments, respectively, at a dose of 2500 μg/0.1 mL per plate (Figure 8).
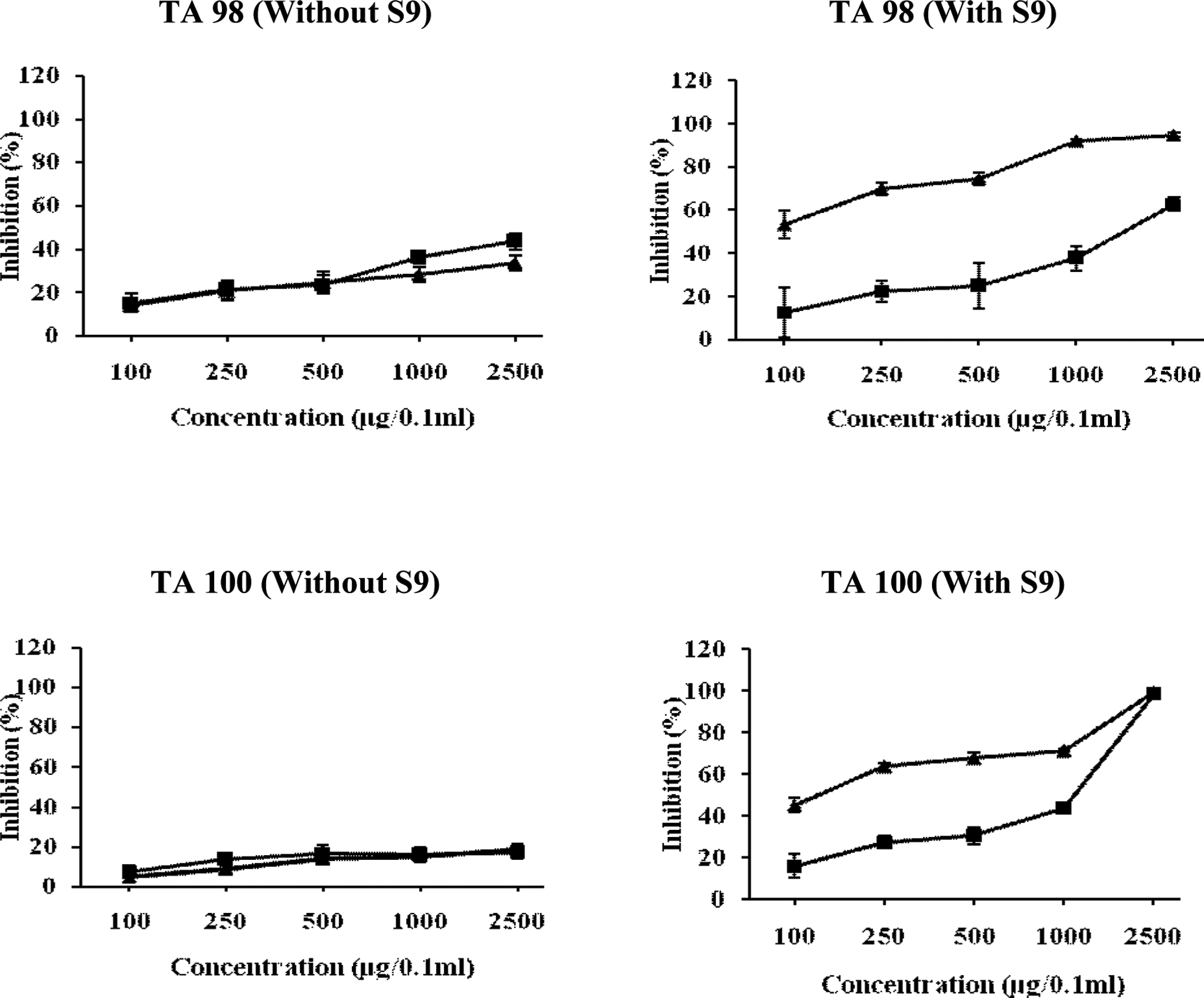
Relationship between different concentrations of water fraction of Chukrasia tabularis leaves and antimutagenic mutagenic activity of NPD, sodium azide, and 2-AF in TA98 and TA100 tester strains of Salmonella typhimurium. The results are expressed as inhibition (%) ± SE. Here, ▴= Pre-incubation mode of experimentation and ▪ = co-incubation mode of experimentation. 2-AF indicates 2-aminofluorne; NPD, 4-Nitro-o-phenylenediamine.
High-Performance Liquid Chromatography Analysis
High-performance liquid chromatography analysis for the identification of known phenols (gallic acid, catechin, epicatechin, 7-hydroxycoumarin, rutin, and quercetin) in methanol extract of C tabularis leaves was based on their retention time (RT) in solvents A (0.03% TFA in water) and B (methanol) which were run in linear gradient (25% B, 40% B, 50% B, 45% B, 35% B, 20% B, 5% B, and 25% B). First, the mixture of known phenols was subjected to HPLC in the mobile phase and the retention time for the phenols was noted (Figure 9 ), then the methanol extract of leaves was analyzed in the same mobile phase. On the basis of retention time, peaks were analyzed.
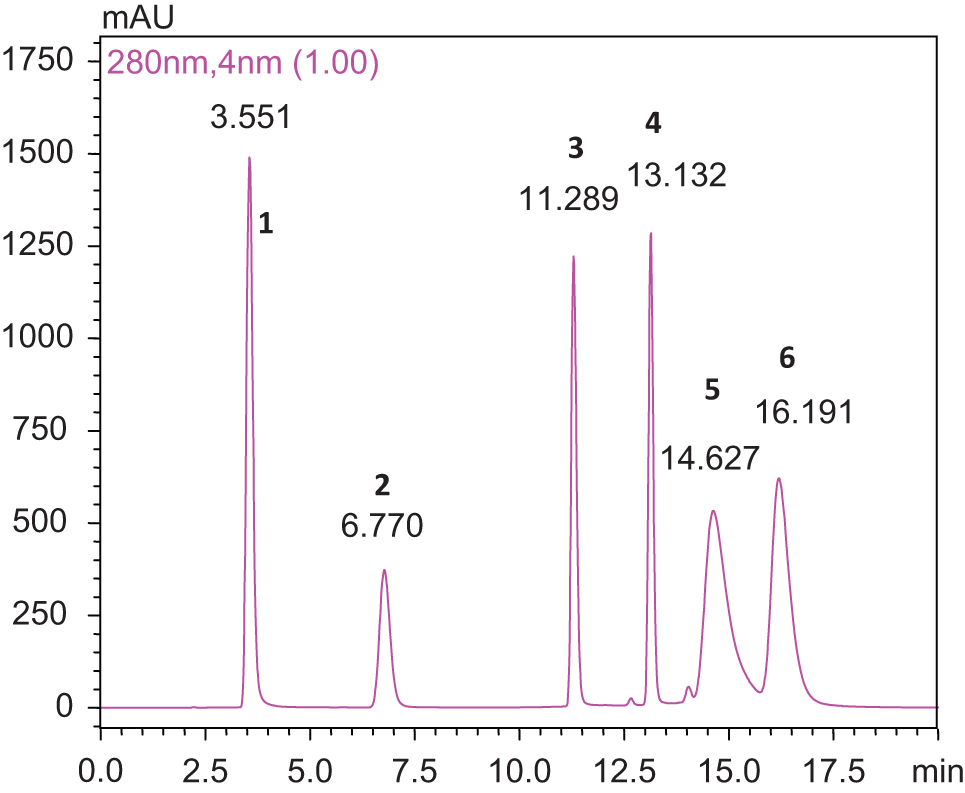
Chromatogram showing retention time of standard phenols that is gallic acid (peak 1), catechin (peak 2), epicatechin (peak 3), 7-hydroxycoumarin (peak 4), quercetin (peak 5), and rutin (peak 6).
Figure 10 represents the HPLC chromatogram of methanol extract of C tabularis leaves (dissolved in HPLC grade methanol) obtained after elution with solvents A (0.03% TFA in water) and B (Methanol) in linear gradient. It was found that the compounds per their elution in mobile phase had retention time (RT) of 3.366 minutes, 7.989 minutes, 10.887 minutes, 12.177 minutes, 12.864 minutes, 13.226 minutes, 13.845 minutes, and 15.898 minutes (Figure 10). After comparison with standard phenol mixture, it was found that leaves of C tabularis had gallic acid (RT = 3.366 minutes), epicatechin (RT = 10.887 minutes), 7-hydroxycoumarin (RT = 13.226 minutes) and rutin (RT = 15.898 minutes). The chromatogram of methanol extract of leaves showed major peak at 13.226 minutes and thus represented the predominance of 7-hydroxycoumarin in extracts, followed by rutin.
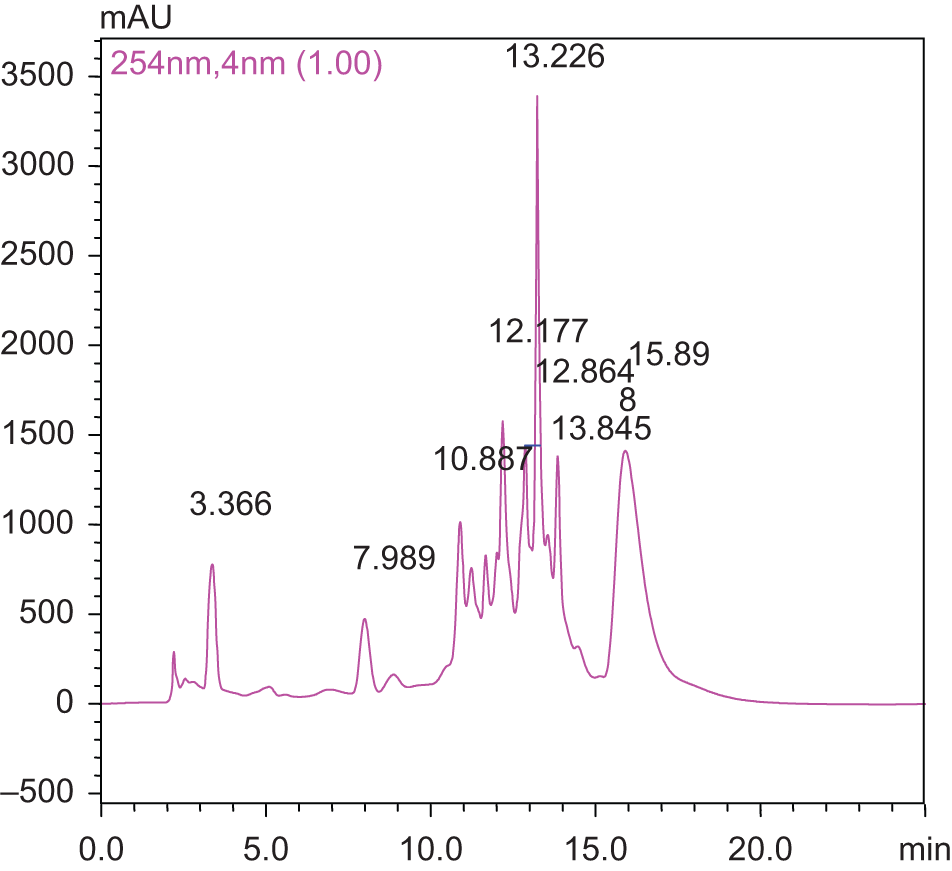
Chromatogram of Chukrasia tabularis leaves extract.
Discussion
The initiation and promotion of mutagenesis and carcinogenesis is reported to be effected by ROS either directly or indirectly. These species also alter the integrity of membrane bound enzymes by enhancing lipid peroxidation. 28,29 It is assumed, therefore, that the antioxidant potential of chemopreventive agents that is phytochemicals might have contribution in imparting antimutagenic and anticancer activities as these agents act by either suppressing or blocking the initial phase of carcinogenesis or preventing the transformation of neoplastic cells to cancer. The antioxidative properties of phytochemicals are linked to their ability to scavenge free radicals (.OH, O2 -., NO.) that due to their short life span and potential to initiate chain reactions put forth damaging effects on biomolecules. 20,30,31 The scavenging of ROS by dietary antioxidants is supposed to cause the downregulation of transcription factors (activator protein 1 [AP-1], nuclear factor kappa-light-chain-enhancer of activated B cells [NFκB], β-catenin-T cell Factor [TcF], tumor protein 53 [p53], and tumor necrosis factor-α [TNF-α]) that are activated by ROS on interaction with mitogen-activated protein kinases (MAPKs). 32,33 These dietary supplements affect the metabolism and disposition of carcinogen due to their influence on metabolizing enzyme system (cytochrome P450 and glutathione-S-transferase) of body. The dietary compounds also act as bioantimutagenic agents due to their tendency to scavenge mutagens through either binding or adsorption that causes the hindrance of the replication process of damaged DNA. In addition to this, dietary supplements are involved in the indirect inactivation of mutagens by acting as desmutagens. 34,35 The multifunctional protective properties of phytochemicals are related to their chemical structure in terms of the presence of OH groups in varying number and arrangements around the nuclear structure. The arrangement of different functional groups contributes to their tendency to act as multidentate ligand. 14,36,37
In the current study, ABTS+ has been used to determine the hydrogen-donating ability of methanol extract of C tabularis leaves and its different fractions. ABTS+ is more versatile and has been used to assess the scavenging activity for both the polar and nonpolar compounds due to its solubility in aqueous and organic solvents. In addition to this, lesser spectral interference during the analysis and stability in a wide range of pH values are another advantage linked with its use to determine antioxidant activity. The absorption maximum (734 nm) that is used in this assay is not normally encountered by natural products. 24,38 It was found that methanol extract and its different fractions exhibited ABTS+-scavenging activity in a dose-dependent manner and the ethyl acetate fraction of the leaves exhibited the highest affinity to scavenge ABTS+ with an IC50 value of 14.394 µg/mL. This value was lowest among different extracts and fractions of leaves. The hydrogen-donating activity was found to be related to the presence of phenols, as Ozgen and coworkers reported in 2006. Quercetin and gallic acid exhibited the highest activity in ABTS+ and FRAP assays, whereas chlorogenic acid, quercetin, and caffeic acid had the highest antioxidant capacity in the 2,2’-diphenyl-1-picrylhydrazyl (DPPH) assay which also determines hydrogen-donating activity. 39
Similarly, in a superoxide anion-scavenging assay, it was found that methanol extract and its fractions showed a dose-dependent response with the EAFL acting as an effective scavenger of superoxide anion radicals with the lowest IC50 value of 37.903 µg/mL among different extracts and fractions. These observations are in agreement with the earlier reports of Chun and coworkers (2003), which related the O2 scavenging of fresh plums to the presence of anthocyanins and chlorogenic acids. They found that different polyphenols exhibited O2 -. scavenging activity in alkaline medium by electron spin resonance [ESR] spectrometry in the order quercetin > quercritin > cyanidin > quercetin-3-rutinoside > peonidin. Presence of hydroxyl groups and absence of sugar moieties are the major contributory factors for superoxide anion radical-scavenging action. 40
The Ames assay has been useful in correlating in vitro bacterial mutagenesis with in vivo carcinogenicity in animals. The S9 mixture is used as an approximation of mammalian metabolic system and it helps to confirm the mutagenicity of indirect-acting mutagens including aromatic amines or polycyclic amines. These mutagens are biologically inactive but their metabolization by cytochrome P-450 enzymes of S9 mixture (prepared from S9 fraction of rat liver homogenate) convert them to DNA-reactive, electrophilic forms. A critical analysis of the results obtained in the current study indicated that almost all the extracts and fractions of C tabularis leaves reduced the mutagenicity caused by the mutagens more or less in a dose-dependent manner. The inhibitory effect was found to be more pronounced in pre-incubation mode of treatment than co-incubation one. The methanol extract and its different fractions showed strong inhibitory effect against indirect-acting mutagen (2-AF) as compared to NPD and sodium azide, direct-acting mutagen of TA98 and TA100 strains of S typhimurium. Among different extracts and fractions, it was found that the methanol, chloroform, and EAFL showed moderate-to-very-strong antimutagenic activity against direct- and indirect-acting mutagens (Figures 3-8).
The higher antimutagenic potency of methanol extract and its different fractions against 2-AF might be through the interference of polyphenols in the metabolic activation of promutagen or might involve the interaction with ultimate metabolite. The phase I enzymes particularly cytochrome P450s enzyme system catalyses the formation of N-hydroxy derivative that is N-hydroxy-2-aminofluorene and this metabolic intermediate interacts directly with DNA. The plant metabolites exert protective effect by altering the structure and function of cytochrome P450s enzymes that further altered the reaction rates and differential pathways related to the metabolism of mutagens and carcinogens. However, another finding has suggested that the interaction of polyphenols with proteins might act as key factor in the mechanism of inhibition of mutagenicity induced by S9-dependent mutagens. 41,42 The postulation is further strengthened by the fact that in most cases wherein fractions were pre-incubated for 30 minutes with 2-AF and S9 mix, the inhibitory effect was more prominent as compared to co-incubation treatment.
The higher potency against indirect-acting mutagens was earlier reported by Gupta and coworkers in 2002. They found that tea extract and its active principles (theaflavins and thearubigins) resulted in concomitant fall in the number of histidine revertant colonies that were increased by metabolically activated mutagens 2-AF (TA97a, TA98, and TA100) and Danthron (TA102). The inhibitory effect of tea extract and its active principles might be related to their ability to detoxify the activity of carcinogen or inhibiting the conversion of mutagenic precursors to active forms. 43
Furthermore, it was found that the different extract and fractions of C tabularis leaves showed comparatively low effect against direct-acting mutagens that is NPD and sodium azide in TA98 and TA100 strains, respectively, as compared to S9-dependent mutagens which might be related to their ability to enhance DNA-excision repair system in mutant strains. However, among direct-acting mutagens, the effect was much lower against sodium azide in TA100 strains as compared to NPD in TA98 strain. Sodium azide and aromatic amines including NPD showed their mutagenic effect by the formation of azidyl radicals, reactive intermediates, and electrophiles. Therefore, chemopreventive agents that act as scavenger of reactive intermediates and radicals are expected to show strong antimutagenic effect in Ames assay. However, the possible explanation for this comparatively weak protection could be the involvement of in vivo antioxidative enzymes in protection against oxidative like mutations. 44,45
The HPLC analysis of methanol extract of C tabularis leaves showed the presence of gallic acid, epicatechin, 7-hydroxycoumarin, rutin, and other unidentified compounds of phenolic nature. From the chemical investigation, it could be postulated that the antimutagenic and antioxidative activity of these extracts and fractions is related to the synergistic interaction among different chemical compounds. The chloroform fraction of leaves was found to be rich in chlorophyll that might have contributed to its antimutagenic potency.
Conclusion
From the current study, a conclusion can be made that the methanol extract of C tabularis leaves and its different fractions exhibited strong antioxidant potential against superoxide anion radicals and ABTS radical cations generated in vitro either due to their potential to donate hydrogen ions or free radical-scavenging ability. The extract and fractions of C tabularis leaves were more efficient antimutagenic agents against indirect-acting mutagens as compared to direct-acting mutagen. It further indicated that extracts might be involved in the inactivation of phase I enzymes and induction of phase II enzymes so as to prevent the conversion of procarcinogen to carcinogen. Besides affecting the metabolic enzymes, the inhibitory activity of the extracts may be due to the scavenging of ROS formed due to metabolic activation. High-performance liquid chromatography analysis helps in the identification of active constituents (gallic acid, epicatechin, rutin, and 7-hydroxycoumarin) that contribute to the antioxidative and antimutagenic effect of different extracts and fractions.
Footnotes
Acknowledgments
Financial assistance from University Grants Commission, New Delhi, India, is duly acknowledged. Dr PS Ahuja, Director, Institute of Himalyan Bioresource Technology (IHBT) Palampur (Himachal Pradesh) is duly acknowledged for providing necessary laboratory facilities to pursue the work on fractionation of the active extracts.
The authors declared no conflicts of interest with respect to the authorship and/or publication of this article.
The authors disclosed receipt of the following financial support for the research and/or authorship of this article: The form of Senior Research Fellowship from University Grants Commission, New Delhi, India.