Abstract
The exposure–response relationship and threshold for nasal epithelial effects of naphthalene (NP) vapor in F344 and SD rats were investigated in 1-day (6 hours) and 5-day (6 h/d) studies at concentration ranges of 0 to 30 ppm. Lesions related to 1-day exposure were predominately necrosis of the olfactory epithelium (OE). The severity of OE lesions was concentration dependent and ranged from minimal (≤1 ppm) to marked (10-30 ppm). In the 5-day study, degeneration of OE was observed in both strains, both sexes, with increasing incidence and severity that correlated with concentration. The epithelial degeneration lesion was minimal to moderate in severity. At 0.1 ppm, minimal OE lesions were observed in female SD rats only (20% incidence). Animals exposed to 10 ppm NP followed by 14 days without exposure also had OE lesions, but of lower severity, showing evidence of good recovery. In both studies, differences between sex or strain were not remarkable.
Naphthalene (NP) is a naturally occurring component of fossil fuels that is isolated for use as an intermediate in the chemical, plastics, and dye industries as a moth repellant, an air freshener, and a surface-active agent. 1 Naphthalene also is formed via incomplete combustion of organic matter, resulting in its presence in wood smoke and tobacco smoke. The toxicologic and carcinogenic potential of NP in Fischer 344 (F344) rats and B6C3F1 mice following 2 years of inhalation exposure of NP vapor was studied by investigators of the National Toxicology Program (NTP). 2-5 At the lowest concentration tested (10 ppm), nasal respiratory epithelial adenomas were observed in male rats, and nasal olfactory epithelial neuroblastomas were observed in female rats. However, nasal tumors were not observed in B6C3F1 mice. An increased incidence in pulmonary adenoma was observed in female mice exposed to 30 ppm NP. In both species and both sexes, exposure to NP caused increases in the incidences of various nonneoplastic lesions, indicating that cytotoxicity of the nasal cavity was neither species nor sex dependent. Metabolites of NP are suspected to play a major role in the toxicity and tumorigenicity of NP. 6-8 Species and strain differences in metabolic capacity may influence the severity and location of NP-induced effects. For example, nasal cavity metabolism of NP (via P450-dependent monooxygenases) appears to be an important factor in cytotoxicity and location of NP-induced injury. 9-11
Given the differences observed between species and sexes in laboratory animal studies, the toxicity of NP and its relevance to human health risk assessment are complex, and scientific uncertainties that need further elucidation were discussed in an NP science symposium conducted in 2006. Reviews and reports from the symposium were published in 2008 in Regulatory Toxicology & Pharmacology. 12 Symposium participants identified an uncertainty of effect concentrations of nasal cavity cytotoxicity following acute or subchronic exposure of NP vapor, due in part to the limited number of in vivo inhalation studies that have been conducted. The objective of the studies described here was to establish an exposure–response relationship and threshold for nasal epithelial effects in F344 and Sprague-Dawley (SD) rats following both a single 6-hour inhalation exposure (1-day study) and a 5-day, 6-h/d inhalation exposure pattern (5-day study) to NP vapor. Exposure concentrations of NP were selected based on those used previously in the NTP 2-year study and lower. To evaluate recovery from cytotoxic effects, a subgroup of animals from the highest exposure group in the 5-day study (10 ppm) was necropsied 14 days following the last NP exposure.
Methods
Test Substance
Naphthalene (CAS #91-20-3) was obtained from Sigma-Aldrich (St Louis, MO). The certificate of analysis for lot 09621TD indicated that the material was 99.9% pure.
Test Animals, Group Assignments, and Husbandry
For the 1-day study, 33 male and 33 female Fischer-344 rats were obtained from Charles River Laboratories (Kingston, NY), and 33 male and 33 female Sprague-Dawley rats were purchased from Charles River Laboratories (Raleigh, NC). Five rats per sex per strain were assigned to NP concentration groups of 0, 0.1, 0.3, 1, 10, and 30 ppm. For the 5-day study, 53 rats per sex per strain from the same suppliers were obtained. The number of rats per sex per strain assigned to NP concentration groups was 5 (0 ppm) or 10 (0.1, 1, and 10 ppm). In addition, 5 control (0 ppm) or 10 high-concentration (10 ppm) rats per sex per strain were assigned to the 14-day postexposure recovery groups. For both studies, animals were approximately 4 weeks of age upon receipt at the testing laboratory. Animals were acclimated in the animal facility for approximately 2 weeks prior to beginning the exposure. Animals were fed pelleted food (NIH-07, Zeigler Bros, Gardners, PA) and water (reverse-osmosis purified) ad libitum, except during the exposure, when food was removed. Animals were identified by ear tags. Prior to NP exposure, animals were randomly assigned to study groups based on body weight. Animals were also acclimated to the environment of the whole-body exposure chambers 1 day prior to exposure to NP.
Animals were exposed in a 1-m3 whole-body exposure chamber (H1000, Lab Products, Seaford, DE). One chamber was used for each concentration including a control chamber. A stainless steel cage unit, housing up to 24 rats (R-24, Lab Products), was used to contain the animals during inhalation exposures. Stainless steel pans were placed under each R-24 cage unit to collect urine and feces. The incoming air for the exposure chamber was HEPA filtered. Environmental conditions in the 1-m3 chambers were recorded during exposure. A temperature and humidity probe (200R, Rotronic Instrument, Huntington, NY) measured the temperature and humidity in each exposure chamber. Airflow in the chamber was determined by measuring the pressure drop across a calibrated orifice plate. The temperature, humidity, and airflow measurements were periodically recorded by a building automation system (Andover Continuum System, Andover Controls, Andover, Mass), and a report was printed at the end of each exposure day.
Generation and Characterization of Naphthalene Vapor Exposures
Naphthalene vapors were generated by heating solid NP and using nitrogen gas to carry the vapor into the 1-m3 chamber inlet (Figure 1 ) via a small or large generator. A generator consisted of a 2-piece glass container with a gas inlet and outlet at either end. The small generator was approximately 3 cm outer diameter by 20 cm long, with approximately 1 to 2 g of NP placed on the bottom of the generator. The small generators were placed in an oven to control temperature, with the inlet and outlet lines passing through holes in the oven. Two temperature probes, 1 for each generator, were placed through a hole into the oven to monitor temperature. These small generators were used for the 0.1-, 0.3-, and 1-ppm exposure chambers. The large glass system was approximately 10 cm outer diameter by 45 cm long, with approximately 50 g of NP contained in a small glass dish at the bottom of the chamber. A heat tape wrapped around the large glass generator provided the heat to sublimate the NP. A temperature probe was placed between the outer surface of the glass vessel and the heat tape. The large generation system was used for the 10- and 30-ppm exposures. Concentrations in the chambers were controlled by adjusting a combination of the heating temperature and nitrogen flow rate through the generators.
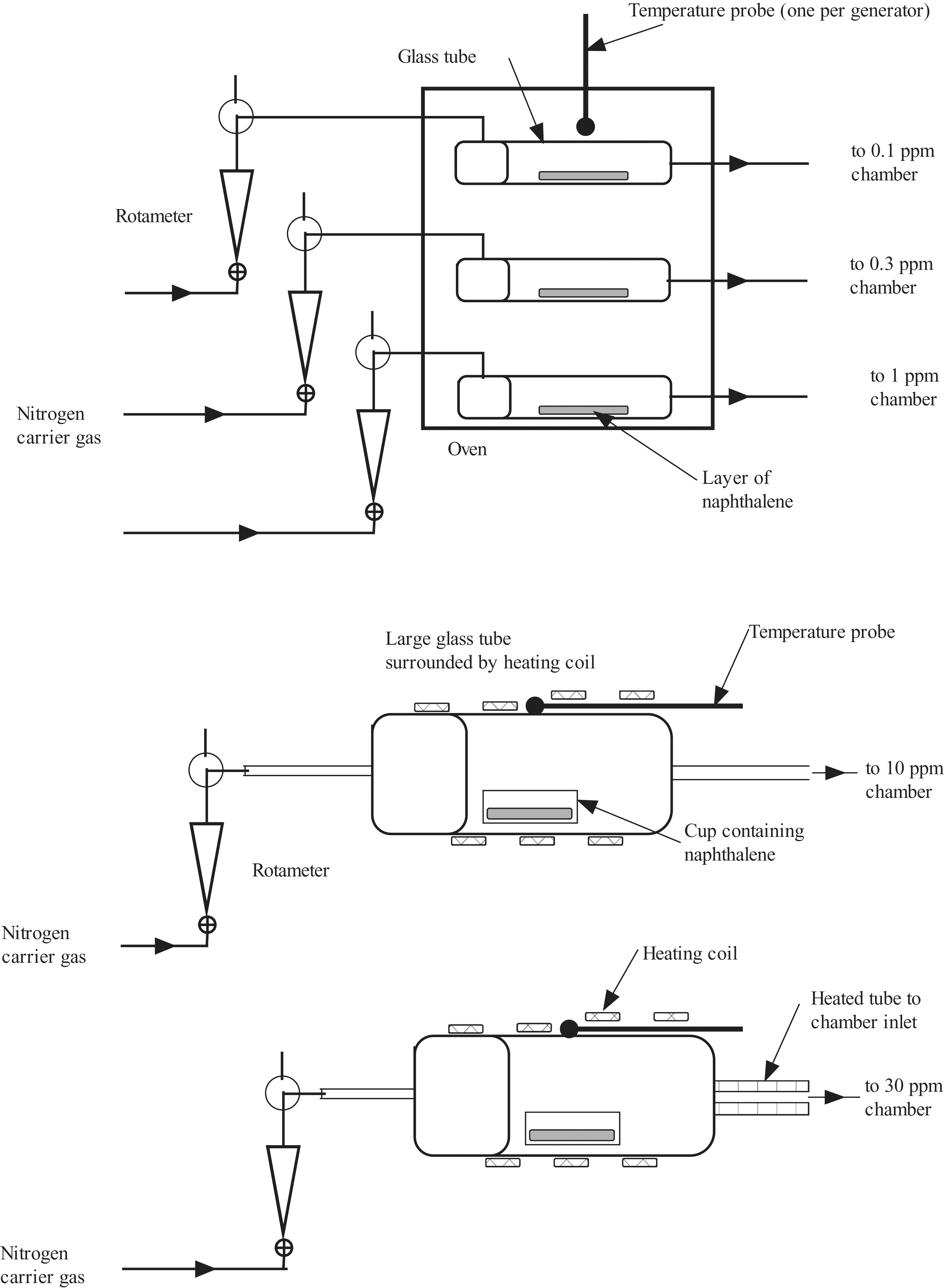
Schematic of naphthalene vapor generation system used in the 1-day study.
During the exposures, NP concentration was measured using a gas chromatograph (GC, 5890 Series II, Hewlett Packard, Palo Alto, CA) equipped with either a fused silica film (1-day study) or a DB-FFAP film (5-day study), 15 m × 0.53 mm inner diameter capillary column and flame ionization detector. The GC was calibrated using Tedlar bags containing a range of concentrations of NP. These standards were prepared by injecting known volumes of solutions of NP dissolved in chloroform into a Tedlar bag and diluting with nitrogen gas. The bag standards were sampled by the GC to produce a calibration curve. A multiport gas sampling valve system was used to convey a sample from each chamber to the GC. A measurement of NP concentration was taken from each chamber periodically. The GC was programmed to integrate automatically the chromatographic peak and calculate the concentration of the NP.
A check for aerosol formation was conducted in each exposure chamber. A scanning mobility particle sizer (SMPS, 3934, TSI, Shoreview, MN) was used to measure particle concentration in the size range from 4 to 1000 nm. The range of particle sizes measured by this instrument covered the expected particle sizes that could be produced by the condensation of NP vapors to aerosol particles. The SMPS was used to take 3 consecutive readings from each chamber, including the control chamber, while an NP atmosphere was being generated in the chamber. Prior to the conduct of exposures, aerosol measurements were taken from each chamber. During exposures, aerosol measurements were taken from the high-concentration and control chambers. In addition, in the 1-day study, filter samples were taken during exposure from the high-concentration (30 ppm) chamber and the control chamber. A flow of chamber atmosphere was pulled through a weighed glass fiber filter at a constant rate. After sampling, the filter was weighed again, and the mass concentration was determined from the net weight, flow rate, and sampling time.
In addition to the NP vapor samples collected from the exposure chambers, NP vapor samples were taken from 2 separate sources for quality control. A diffusion generator (Dynacalibrator 340, VICI Metronics, Santa Clara, CA) was set up to generate a steady concentration of NP. A small amount of solid NP in a diffusion bottle was placed in a temperature-controlled oven of the diffusion generator. At a set temperature, NP diffused out of the bottle at a constant rate into a constant dilution airflow. The diluted NP flow was sampled by the multiport sampling valve system. A Tedlar bag standard containing 3.1 ppm of NP vapor was prepared from a certified standard cylinder (3.1 ppm NP in nitrogen, Scott Specialty Gases, Plumsteadville, PA). The bag standard was also sampled via the multiport gas sampling valve. The diffusion generator output and 3.1-ppm bag standard were part of the regular rotation of the multiport sampling valve during the exposure.
Each exposure chamber was checked for the uniformity of distribution of vapor by measuring NP concentration at 5 different locations during a trial run prior to exposures. Measurements were made after the generator was operating stably and with appropriate caging and catch pans in place. The coefficient of variation of the measurements was less than 10% for all chambers, and the NP was considered to be uniformly distributed within the chamber.
Clinical Observations and Body Weights
Daily cage-side observations were performed for mortality and overt clinical signs of toxicity. Animals were weighed prior to NP exposure and prior to scheduled necropsy.
Animal Necropsy and Histopathology
On the day following the last NP exposure (or 14 days postexposure for the recovery groups), animals were killed by intraperitoneal administration of pentobarbital followed by exsanguination (transection of the abdominal aorta). The lungs were inflated to approximately 30 cm pressure, and the nasal cavities were flushed with 10% neutral buffered formalin (NBF). The respiratory tract pluck, consisting of the lower jaw, tongue, larynx, trachea, bronchi, and lungs, was collected and fixed in NBF. The head was removed, skinned, trimmed of excess tissue, and placed in NBF. Rat heads were fixed in NBF for approximately 72 hours, rinsed in running tap water for 30 minutes, and decalcified in 5% formic acid for approximately 90 hours. Cross-sectional blocks of the nasal tissues were prepared at 6 standard levels that include the 4 central areas described by Young 13 plus an area at the nasal tip and an area near the posterior end of the ethmoid turbinates. 14 More specifically, the sections are labeled tip, level I, level II, level III, level IV, and level V (Figure 2 ), corresponding to the levels used by Morgan et al. 14 The tip and level I are mostly squamous and respiratory epithelium. Olfactory epithelium is first observed along the dorsal meatus of level II. Level III has olfactory epithelium lining the dorsal meatus, dorsal tips of the ethmoid turbinate, and low in the nasal septum (septal olfactory organ 15 ). Levels IV and V have mostly olfactory epithelium dorsally lining the meatus, ethmoid turbinates, and much of the nasal septum. The blocks were embedded in paraffin wax, sectioned (5 microns), deparaffinized, stained with hematoxylin and eosin, and examined microscopically.
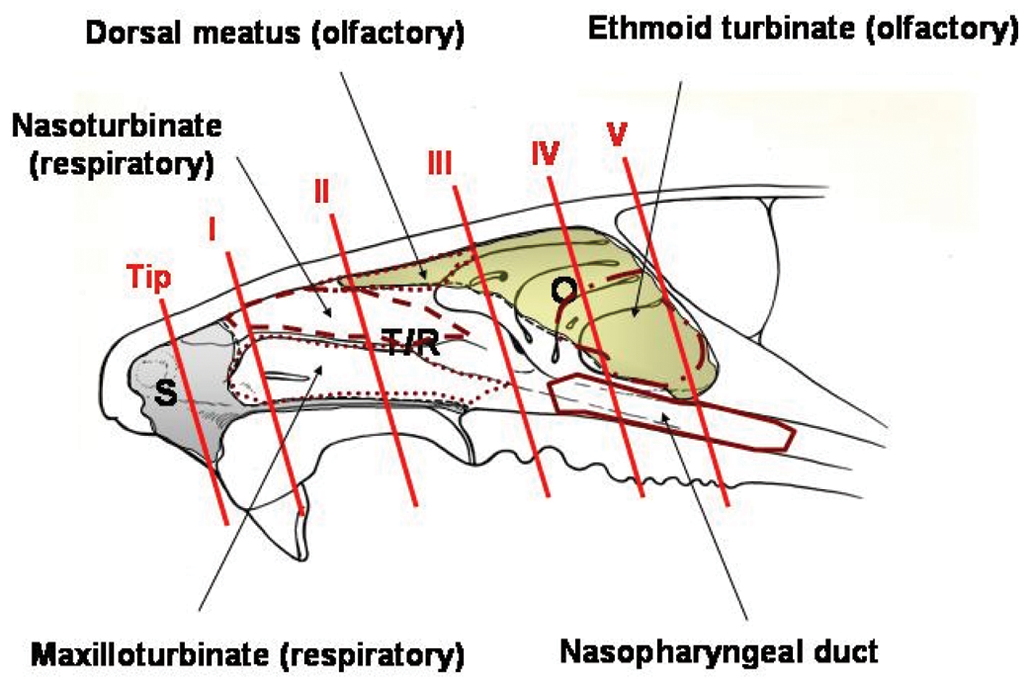
Diagram to describe the locations of the sections collected for histopathological examination. See text for description of tip and levels I, II, III, IV, and V. S, area of predominantly squamous epithelium; T/R, area of predominantly transitional and respiratory epithelium; O, area of predominantly olfactory epithelium. The nasopharyngeal duct is outlined in solid red. The nasoturbinate is outlined by a dashed red line. The dotted red line (lower left) outlines the maxilloturbinate, whereas the dotted red line (upper middle) outlines the area described as dorsal meatus.
Results
Exposure Concentrations
Table 1 shows mean and standard deviation exposure concentrations of NP for the 1-day and 5-day animal studies. Because of a 1-day stagger start for exposure of male and female rats, results for each sex are shown separately. Quality control measurements of NP concentrations are also shown. Mean exposure concentrations were close (±10%) to target concentrations, except for the 1-day 10-ppm target exposure, for which the means were 11.6 and 12.3 ppm NP for male and female rats, respectively. Results of aerosol measurements in the exposure chambers (data not shown) indicated there was no condensation of NP vapor to an aerosol at any exposure concentration. Daily mean environmental conditions for all animal exposures ranged from 22°C to 26°C, 45% to 56% relative humidity, and 224 to 231 L/min airflow.
Mean and Standard Deviation Exposure Concentrations (given as ppm) of Naphthalene (NP) for the 1-Day and 5-Day Animal Studies
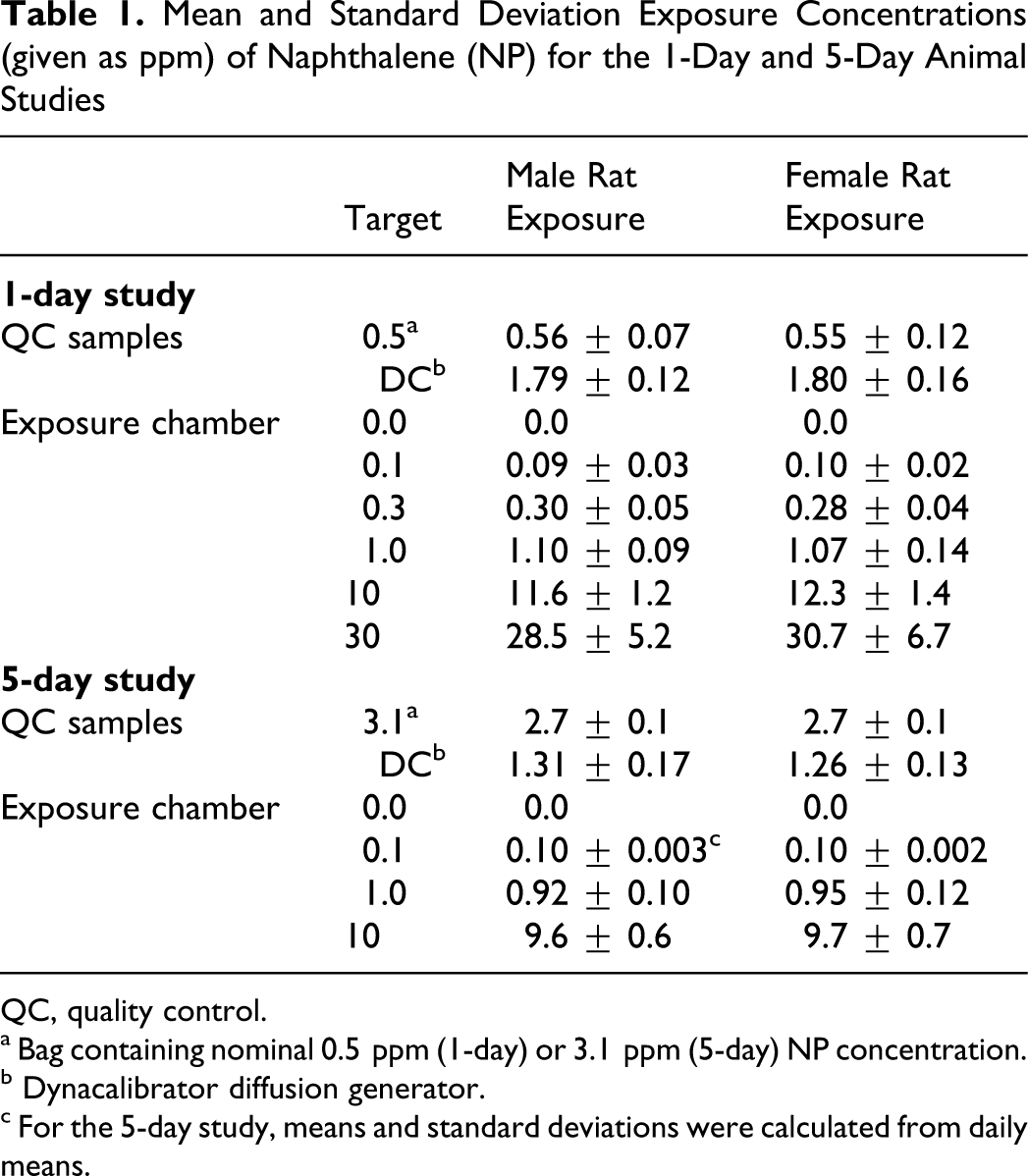
QC, quality control.
a Bag containing nominal 0.5 ppm (1-day) or 3.1 ppm (5-day) NP concentration.
b Dynacalibrator diffusion generator.
c For the 5-day study, means and standard deviations were calculated from daily means.
Clinical Observations, Body Weights, and Gross Necropsy Findings
There were no clinical observations attributable to NP exposure during or following exposures for the 1-day and 5-day studies. There were no statistically significant differences in mean body weights between control and NP exposure groups for the 1-day and 5-day studies, including the 14-day recovery period that was part of the 5-day study design (Figure 3 ). However, the group means of animals exposed to 10-ppm NP tended to be lower than those of control animals throughout the 5-day study. At necropsy, there were no gross pathological lesions attributable to NP exposure in rats of both the 1-day and 5-day studies.
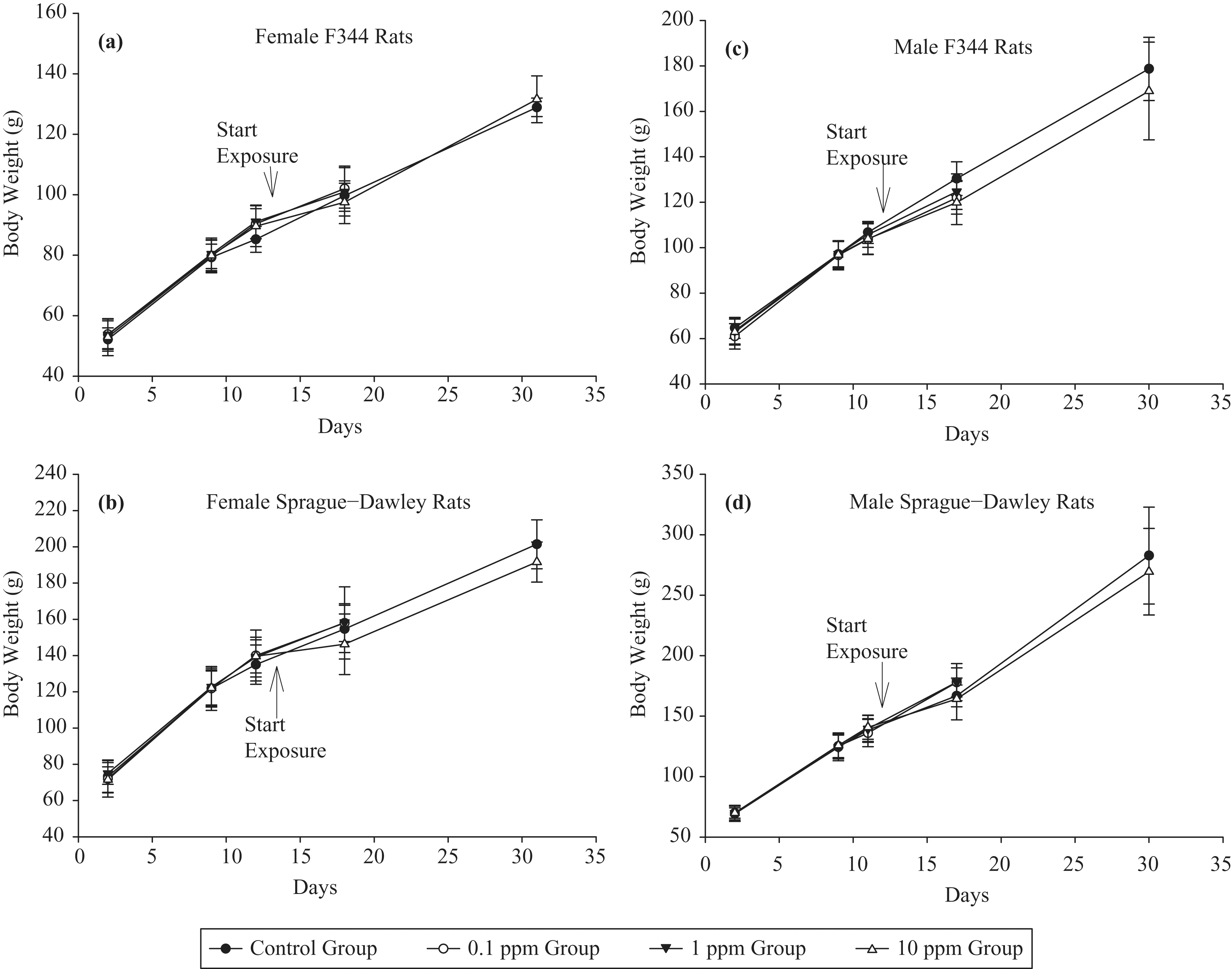
Mean body weights of (a) female F344, (b) female SD, (c) male F344, and (d) male SD rats exposed to NP vapor in 5-day study.
Nasal Cavity Histopathology
One-day study
Some lesions related to NP exposure were observed in some of the respiratory epithelial locations, but most were seen predominantly as necrosis in the olfactory epithelium. Incidences of nasal olfactory and nasal respiratory necrosis are shown in Tables 2 and 3 , respectively. The severity of necrosis was NP concentration-dependent and graded minimal to moderately severe. The necrosis was characterized by cytoplasmic vacuolation, loss of proper epithelial orientation, condensation of the cytoplasm, pyknotic and karyorrhectic nuclei, and sloughing of the necrotic epithelium. Little inflammatory cell infiltration was present. Figure 4 shows necrosis of the olfactory epithelium of the dorsal meatus and ethmoid turbinates in male SD rats exposed to 0 (control) or 30 ppm NP. Olfactory epithelial necrosis was widespread in the dorsal meatus, in the upper septum, and on the medial aspects of the ethmoid turbinates as they approached the septum. At lower concentrations of NP (10 and 1 ppm), the extent and severity of olfactory epithelial necrosis lessened, and necrosis was not observed in F344 rats below 1 ppm (Table 2). The minimal necrosis observed in several but not all SD rats at 0.3 or 0.1 ppm NP was primarily at level III (dorsal meatus or the tip of the ethmoid turbinate in that area), which is lined predominantly with olfactory epithelium. One control SD female had minimal necrosis in this region. Respiratory epithelial necrosis was observed in areas anterior to level III in all rats exposed to 10 or 30 ppm NP (Table 3). One of 20 rats exposed to 1 ppm NP had minimal respiratory epithelial necrosis.
Incidence of Nasal Olfactory Epithelium Necrosis in Fischer 344 (F344) and Sprague-Dawley (SD) Rats Exposed to Naphthalene (NP) Vapor for 1 Day (6 Hours)
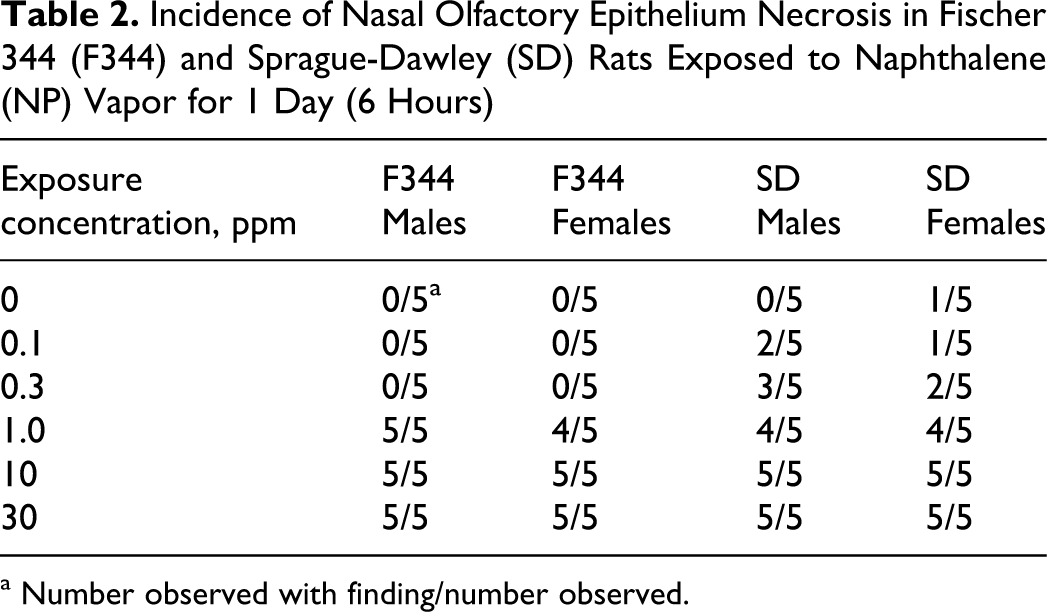
a Number observed with finding/number observed.
Incidence of Nasal Respiratory Epithelium Necrosis in Fischer 344 (F344) and Sprague-Dawley (SD) Rats Exposed to Naphthalene (NP) Vapor for 1 Day (6 Hours)
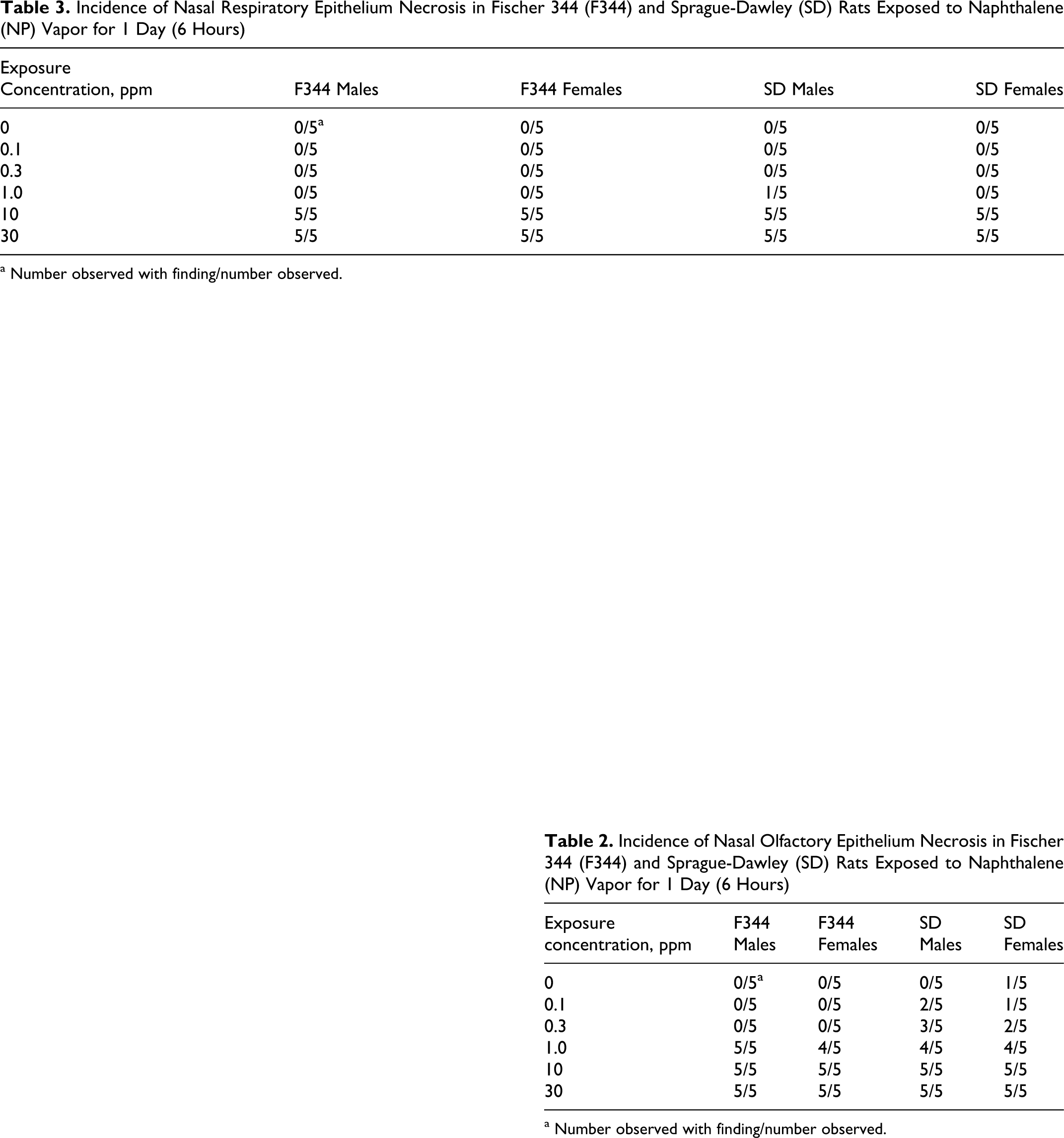
a Number observed with finding/number observed.
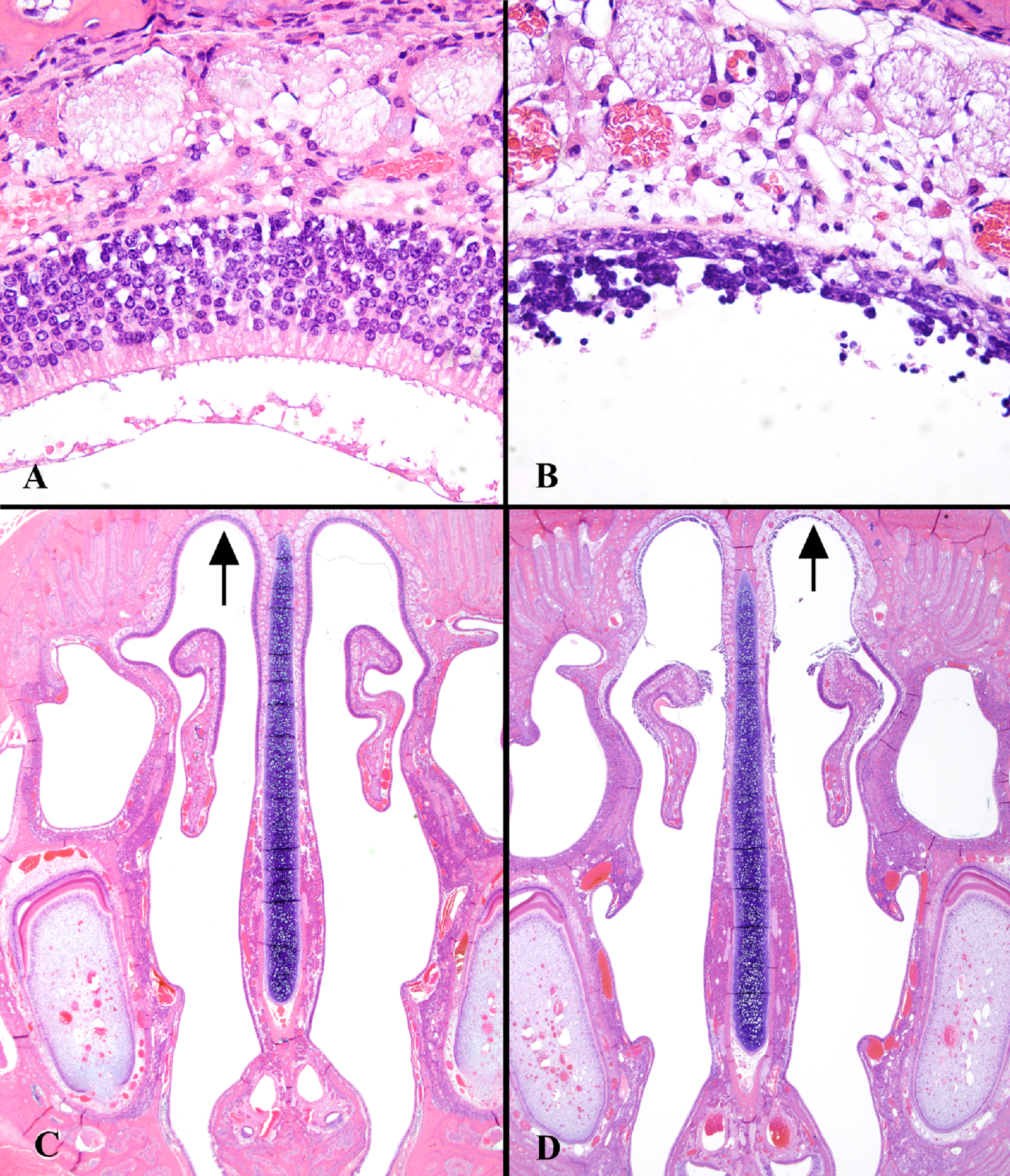
Olfactory epithelium of the dorsal meatus and ethmoid turbinates (level III) in (A and C) control and (B and D) 30-ppm NP male SD rats (1-day study). Areas indicated by arrows in lower photomicrographs (C and D, ×10) are shown in greater detail in upper photomicrographs (A and B, ×40).
Five-day study
NP-induced lesions were again observed in the olfactory epithelium and were diagnosed as degenerative changes with evidence of prior and ongoing necrosis. Degeneration of olfactory epithelium was observed in both F344 and SD strains and in both sexes with increasing incidence and severity that correlated with NP exposure concentration (Table 4). Lesions ranged from minimal to moderate in severity. They were generally multifocal and characterized by cytoplasmic vacuolation, loss of proper epithelial orientation, condensation of the cell cytoplasm, thinning of the epithelial layer due to decreased olfactory epithelial cells, pyknotic and karyorrhectic nuclei, and sloughing of the epithelium. Areas of re-epithelialization were noted beneath the sloughed epithelium that sometimes covered the surface with flattened cells similar to squamous cells but were not layered enough to classify as squamous metaplasia. Minimal to mild mixed inflammatory cell infiltration was present in the lamina propria, and occasionally neutrophils were observed in the nasal lumen. At 10 ppm, olfactory lesions were widespread in all anatomic areas of nasal olfactory epithelium. At 0.1 ppm, minimal olfactory epithelial lesions were observed in female SD rats only (20% incidence). Animals exposed to 10 ppm followed by 14 days of no NP exposure also had olfactory epithelial degeneration but at much lower severity and incidence (Table 4). Figure 5 shows similar nasal cavity cross sections (dorsal meatus—level III) in 3 male SD rats exposed to control, 10 ppm, or 10 ppm plus 14-day postexposure recovery. Epithelial degeneration can be clearly observed following 10-ppm exposure for 5 days (Figure 5B) along with a remarkable degree of recovery (Figure 5C) following 14 days of nonexposure. Olfactory epithelial degeneration in 1-ppm–exposed rats was confined to nasal levels II and III (dorsal meatus, tips of the ethmoid turbinates, and septal olfactory organ 15 ).
Incidence and Severity of Nasal Olfactory Epithelium Degeneration in Fischer 344 (F344) and Sprague-Dawley (SD) Rats Exposed to Naphthalene (NP) Vapor for 5 Days and in Rats Allowed 14 Days Recovery Following 5 Days of Exposure
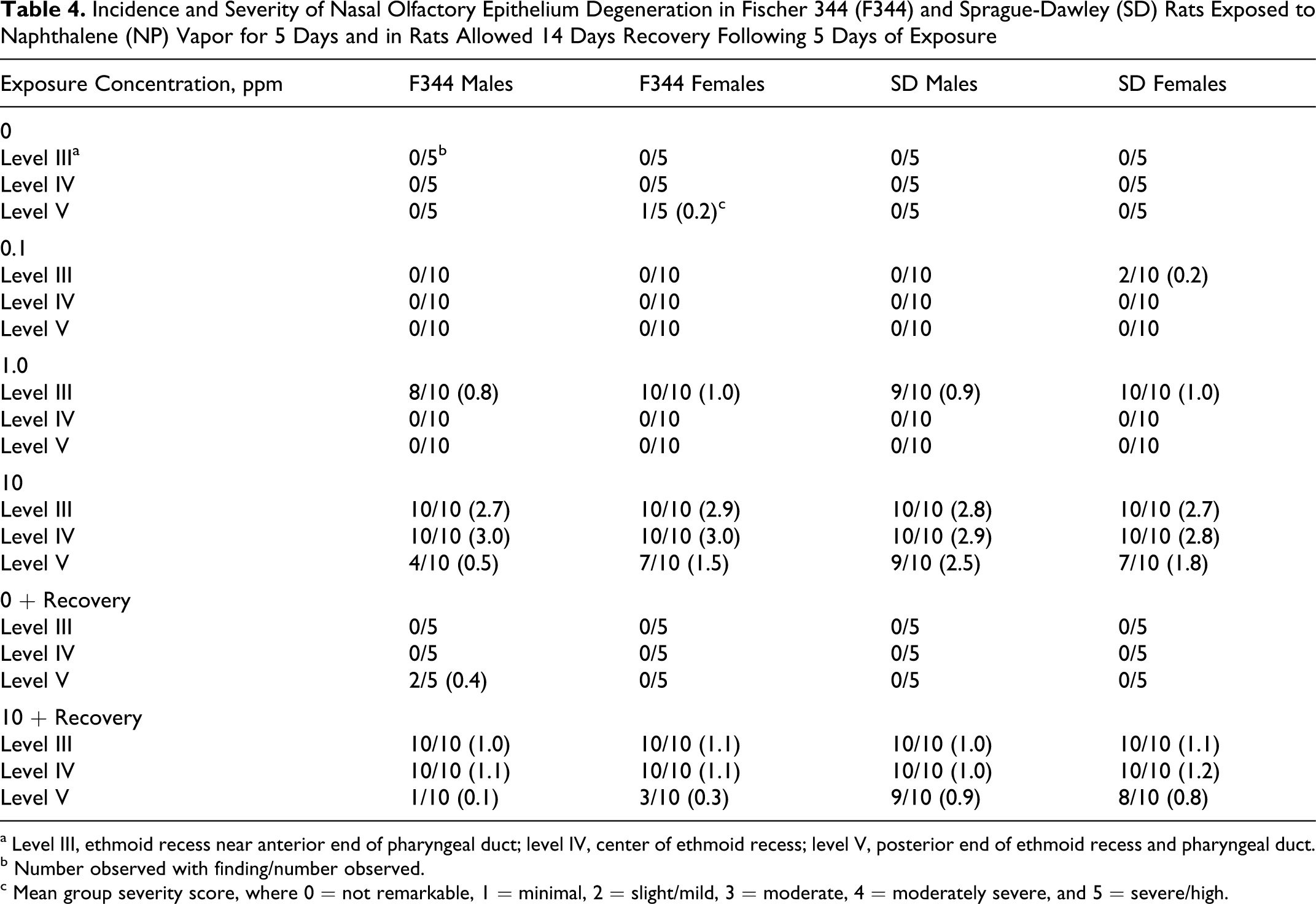
a Level III, ethmoid recess near anterior end of pharyngeal duct; level IV, center of ethmoid recess; level V, posterior end of ethmoid recess and pharyngeal duct.
b Number observed with finding/number observed.
c Mean group severity score, where 0 = not remarkable, 1 = minimal, 2 = slight/mild, 3 = moderate, 4 = moderately severe, and 5 = severe/high.
Olfactory epithelium of the dorsal meatus (level III) of male SD rats in (A) control, (B) 10-ppm NP, and (C) 10-ppm NP followed by 14 days postexposure recovery exposure groups (5-day study). ×20.
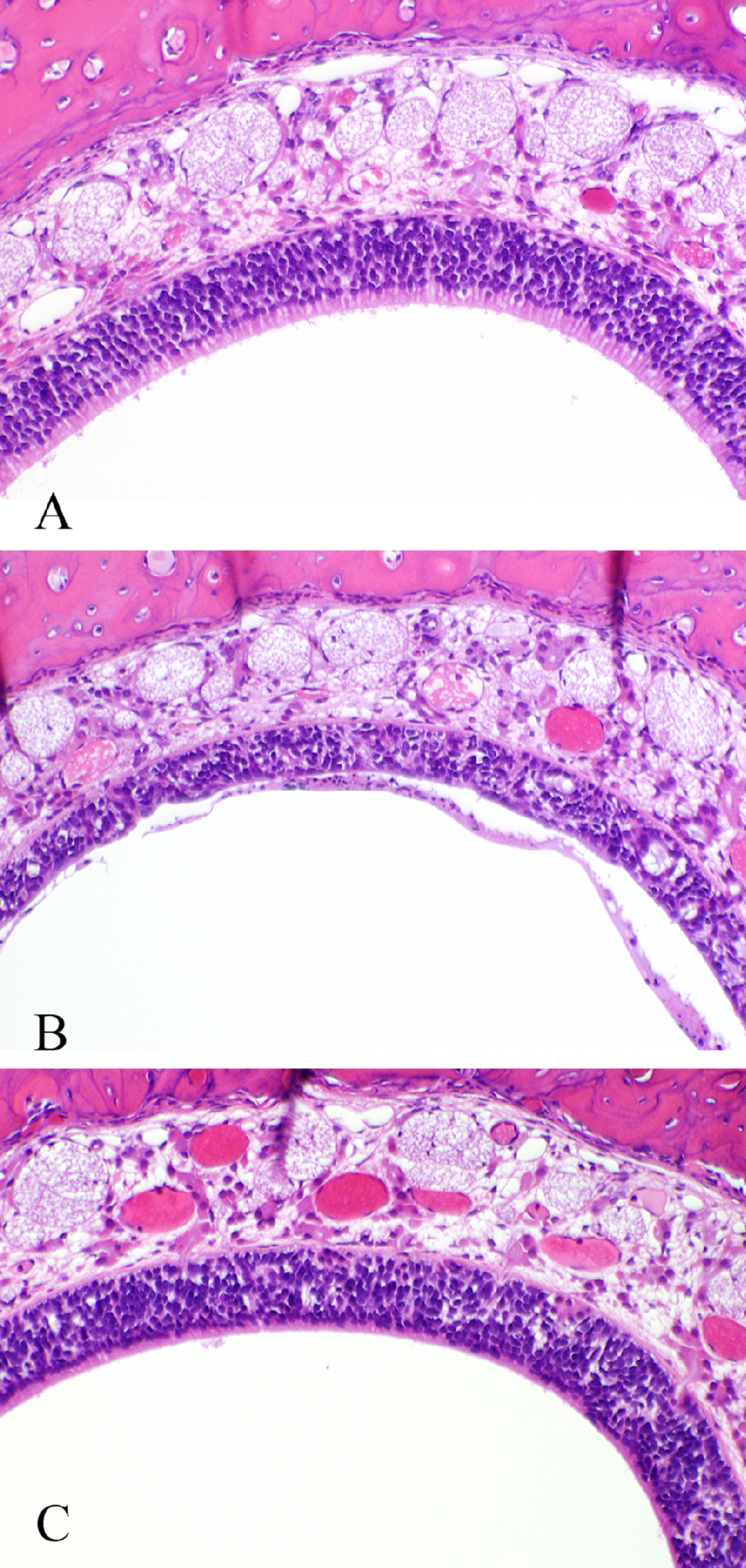
Rats exposed to 10-ppm NP also had minimal to mild nasopharyngeal goblet cell hyperplasia/hypertrophy (Table 5 and Figure 6 ). There was a small incidence (10%-20%) of minimal goblet cell hyperplasia/hypertrophy in animals of the control and the 0.1- and 1-ppm NP exposure groups (Table 5). There was considerable recovery of this lesion observed in animals exposed to 10 ppm and allowed 14 days postexposure recovery. No SD rats of the recovery group had goblet cell hyperplasia/hypertrophy in the nasopharyngeal duct (Table 5).
Incidence and Severity of Nasopharyngeal Goblet Cell Hyperplasia/Hypertrophy in Fischer 344 (F344) and Sprague-Dawley (SD) Rats Exposed to Naphthalene (NP) Vapor for 5 Days and in Rats Allowed 14 Days Recovery Following 5 Days of Exposure
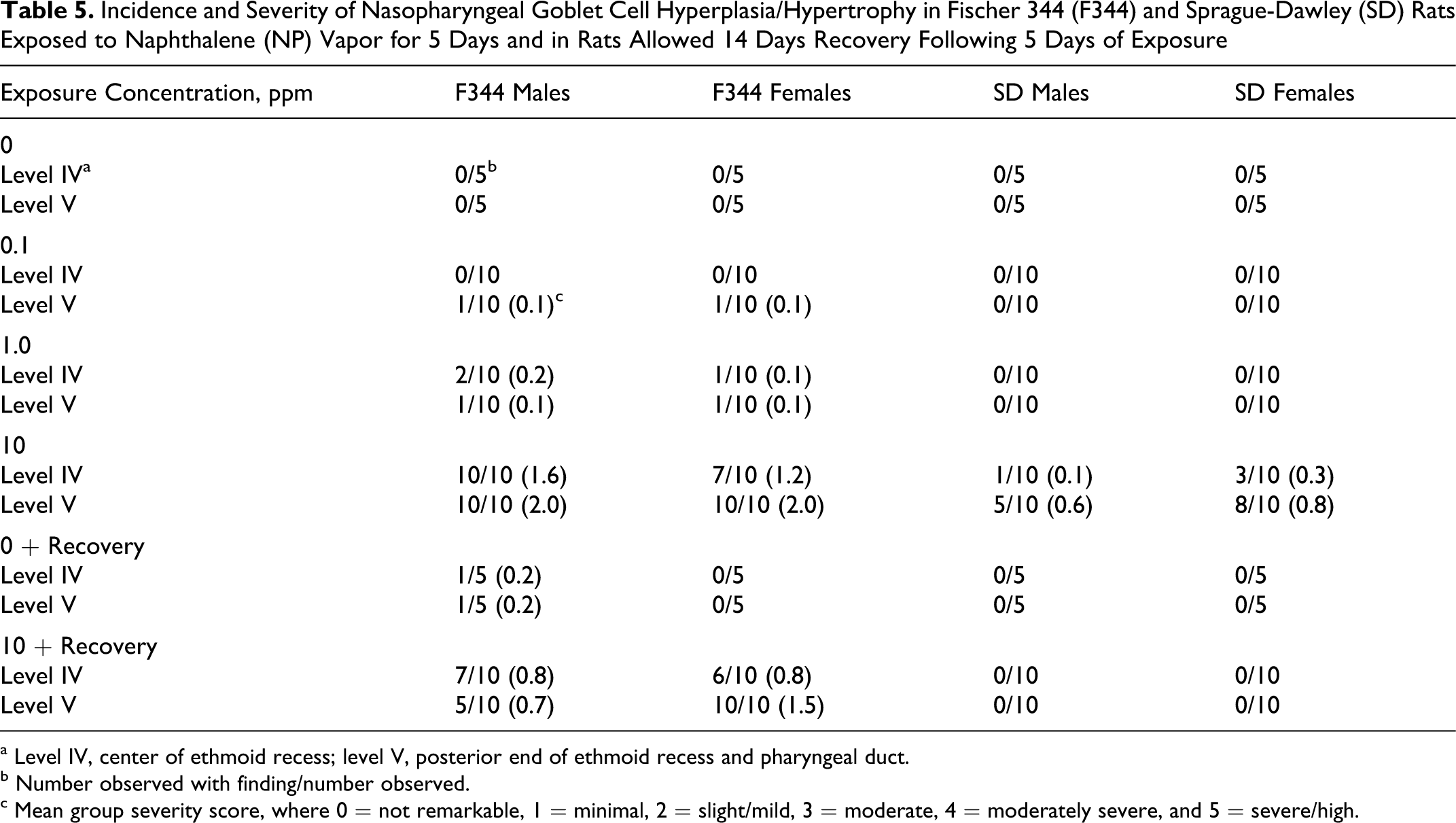
a Level IV, center of ethmoid recess; level V, posterior end of ethmoid recess and pharyngeal duct.
b Number observed with finding/number observed.
c Mean group severity score, where 0 = not remarkable, 1 = minimal, 2 = slight/mild, 3 = moderate, 4 = moderately severe, and 5 = severe/high.
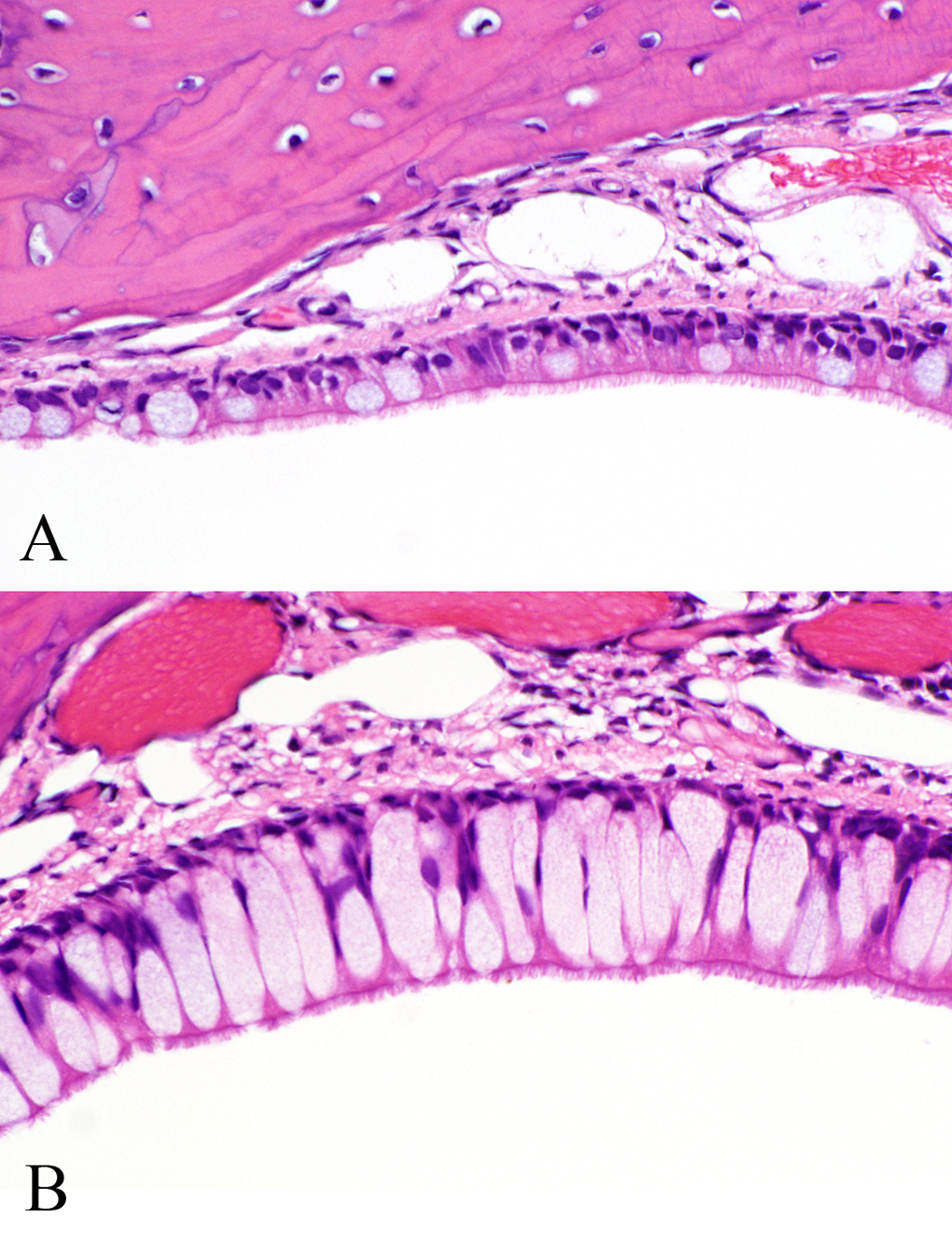
Respiratory epithelium of the nasopharyngeal duct (level IV) of female F344 rats in (A) control and (B) 10-ppm NP exposure groups (5-day study). ×40.
Discussion
Several conclusions can be drawn from the results of the 1-day and 5-day NP inhalation toxicity studies. Nasal cavity necrosis and epithelial degeneration were more prevalent in the olfactory epithelium compared with the respiratory epithelium. This finding is in agreement with that of Lee et al, 11 who observed cellular injury (necrosis) exclusively in the olfactory mucosa of SD rats exposed to 3.4-ppm or 23.8-ppm NP for 4 hours. Furthermore, Lee et al 11 observed that activity of CYP450 in the olfactory region of the rat nasal cavity was significantly greater than in the nonolfactory regions, supporting other research findings that NP metabolism was a contributing factor of NP toxicity. The apparent sensitivity of the olfactory epithelium to NP vapor explains in part that the effects were predominantly observed in the olfactory ethmoid turbinates and the adjacent olfactory dorsal meatus regions. Nasal airflow patterns in the rat also contribute to the location of nasal cavity injury. 16 At low concentrations, the respiratory epithelium appears less sensitive to NP-induced effects; however, concentrations of 10 ppm and higher produced necrosis of the respiratory epithelium following a single, 6-hour exposure (Table 3). The nasal respiratory epithelium has CYP450 activity, 11,17 so again, a combination of concentration, airflow distribution, and metabolism is a likely factor contributing to regional dosimetry of NP in the nasal cavity. Nasal tissue uptake and dosimetry of NP are being investigated, and preliminary findings have been presented. 18,19
In the current studies, there was no apparent indication of differences in exposure response between sexes; however, there was some suggestion of differences between rat strains. In NTP’s 2-year inhalation study using F344 rats, 3 a tumorigenic response was observed in both sexes; however, the location of the tumors varied. In male rats, the incidence of adenomas in nasal respiratory epithelium was increased, whereas in female rats, the increased incidence was observed for olfactory epithelial neuroblastoma. Results from our 1-day and 5-day inhalation studies indicate no nasal cavity histopathological differences between sexes for both strains of rats for NP exposure concentrations ranging from 0.1 to 30 ppm. Because a majority of rat studies using NP appeared to be conducted in either the F344 or the SD strain, it was of interest to explore the possibility of strain differences in nasal cavity response following short-term, low-concentration exposures of NP. A suggestion of histopathological differences between strains was observed at the lowest exposure concentrations used (≤1 ppm NP). Following a single 6-hour exposure, some SD rats had minimal olfactory epithelium necrosis at 0.3 ppm (≤50% incidence, combined sexes), but this finding was not observed in the F344 rats exposed to 0.3 ppm (Table 2). There was a single incidence of respiratory epithelial necrosis in an SD rat exposed to 1 ppm compared with no observed necrosis in F344 rats exposed to 1 ppm (Table 3). The incidence of slight nasal olfactory epithelial degeneration in the 5-day study was 10% (combined sexes) in SD rats exposed to 0.1 ppm but 0% in F344 rats. However, the F344 rat appeared slightly more sensitive to goblet cell hyperplasia/hypertrophy at 10-ppm or less NP and showed less recovery following exposure compared with the SD rat (Table 5). Overall, strain differences in rats were considered minimal following acute (1-day) or subacute (5-day) exposure to NP.
Recovery from nasal epithelial lesions after subacute NP exposure was observed in all 10-ppm exposure groups and was independent of rat strain or sex. The extent of recovery was best indicated by a lower average score of lesion severity and in some cases a lower group incidence of the finding (Tables 4 and 5). In general, the severity of the olfactory epithelial damage and the damaged cell-types determine the extent of recovery after injury. 20 A series of phases beginning with basal cell proliferation, followed by formation of regenerative epithelium and gradual differentiation, leads to recovery of near-normal morphology. 20 Recovery from goblet cell hyperplasia/hypertrophy in the nasal cavity has been observed in SD rats following chemical insult, 21-23 and the response itself during chemical exposure may be part of an adaptive process. 23 Results from the current 5-day study suggest that a remarkable amount of recovery of all observed lesions occurs within 2 weeks.
The nasal cavity lesions observed in this study were clearly concentration dependent. In general, a concentration of 10 ppm caused olfactory epithelial necrosis and degeneration of moderate severity in nearly all animals exposed to NP. However, a concentration of 1 ppm showed only minimal severity of epithelial cell necrosis and degeneration, and though present in most animals exposed, the lesion was not observed in all transverse sections examined (Table 4). Olfactory epithelial necrosis and degeneration were always of minimal severity in animals exposed to NP concentrations of 0.3 ppm or less, were not observed in all transverse sections examined, and were only present in a small number of animals examined. In some cases, control animals showed minimal olfactory epithelial degeneration (Table 4). In conclusion, NP concentrations 1 ppm or less show dramatically lower incidence and severity of rat nasal cavity lesions compared with NP concentrations 10 ppm or more following acute (6 hours) or subacute (6 h/d × 5 days) exposure.
Footnotes
Acknowledgments
We appreciate the advice and support provided by the following individuals: Dr. Gabrielle Willson for pathology support; Drs. Harvey Clewell III and Vincent Piccirillo for study design and discussion; Dr. Mark Sochaski for analytical chemistry support; R. Arden James and Marianne Marshall for inhalation exposure support; Paul Ross for animal care support; and the necropsy/histopathology support staff for the careful consistent processing of nasal cavity sections.
Notes
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: The Naphthalene Research Committee.
