Abstract
The aim of this work is to determine whether consuming tap water containing arsenic (20 μg/L) alters oxidative stress levels in female rats and changes vascular response. Whereas nitric oxide produces complete relaxation, arsenic (7 months of exposure) impairs the acetylcholine-induced endothelial relaxation in the rat aorta compared with control rats. Arsenic exposure results in a marked elevation in reactive oxygen species in blood, and δ-aminolevulinic acid dehydratase activity, which is a sensitive biomarker for arsenic toxicity and oxidative stress, is significantly decreased in erythrocytes from 7-month-old rats. Diastolic blood pressure increases significantly in 7-month-old arsenic-treated versus control rats. The percentage of change in peripheral resistance increases. The results indicate that chronic environmental exposure to low levels of arsenic alters the release of vasoactive substances, causes changes in oxidative stress, and increases blood pressure in female rats.
The north of Chile could be considered a natural laboratory for studying the effects of air and water contamination on organisms. The population of Antofagasta (latitude 23° 38′ S, longitude 70° 24′ W) is chronically exposed to arsenic in drinking water. Indeed, northern Chilean regions suffer the effects of tap water arsenic contamination. Between 1958 and 1970, Antofagasta City inhabitants consumed tap water with high arsenic content, approximately 860 μg/L. 1 We have determined that the arsenic concentration in tap water from Antofagasta City, since September 2004, has been approximately 20 μg/L, 2 whereas the amount recommended by the World Health Organization (WHO) is 10 μg/L in tap water. 3 Other studies indicate that vegetables cultivated in this area are also contaminated: a high concentration of arsenic has been found in corn and potatoes. 4
Arsenic, lead, and cadmium contribute to endothelial dysfunction. 5,6 Several studies have indicated that vascular oxidative stress impairs nitric oxide (NO) production, resulting in decreased NO bioactivity in cardiovascular diseases. 7,8 Kumagai and Pi 9 reviewed the evidence for arsenic-mediated alteration in NO production and oxidative stress. Low chronic exposure to arsenic in tap water was proposed to play an important role in oxidative stress and the vascular response to vasoactive substances. Blood concentrations of oxidative stress biomarkers are highly correlated in other tissues. 10
Endothelial NO is the main vasoactive substance that contributes to the maintenance of basal vascular tone and blood pressure. 11 Hence, a reduction in the availability of NO can result in vasomotor dysfunction. 12
The toxicological importance of very low exposure to arsenic in tap water on animal organisms is not fully understood. The purpose of the present study was to determine whether chronic exposure to a low concentration of heavy metal produces oxidative stress that is associated with changes in vascular response (as the peripheral resistance) during the normal process of maturation. By focusing on a large artery, such as the aorta, this study may provide useful information on action mechanisms of vasoactive substances. Female rats were chosen for the present study because their arteries release a greater quantity of endothelial relaxing factors, such as NO, than do those of male rats 13,14 ; therefore, female rats would be more likely to present alterations of NO in the maturation process. Given that the total release of NO and endothelial NO is greater in premenopausal women than in men 15 and estrogen receptors mediate NO release in human endothelia, 16 this could have wider significance.
Materials and Methods
Drugs
The following drugs were used in this study: Tetramethoxypropane (Sigma-Aldrich, St Louis, Mo), thiobarbituric acid (Merck, Darmstadt, Germany), butylated hydroxytoluene (Merck), δ-aminolevulinic acid (Merck), Cl2Hg (Merck), 4- (dimethylamino) benzaldehyde (Merck), L-phenylephrine hydrochloride (Sigma-Aldrich, St Louis, Mo), acetylcholine (ACh) chloride (Sigma-Aldrich, Munich, Germany), Nω-Nitro-L-arginine (Aldrich Chemical, Sheboygan Falls, Wis), sodium nitroprusside (Merck). Drugs were dissolved in distilled de-ionized water. Acetylcholine (10-3 M) solution in Krebs-Ringer bicarbonate (KRB) was freshly prepared before each experiment.
Animals
Female Sprague-Dawley rats from the breeding colony at the Antofagasta University that were aged 2 months (218 ± 10 g; n = 10) and 7 months (346 ± 8 g; n = 10) were used. All rats were housed in groups of 3 in a temperature-controlled, light-cycled (8
Determination of Arsenic in the Hair of Rats
The arsenic in tap water is associated with arsenic in the hair of animals. 16 A sample of about 0.2000 ± 0.0001 g of hair from arsenic-treated rats was treated with 2 mL of HNO3 (65% suprepure) in a PMD Kürner (Rosenheim, Germany) microwave oven (3-5 minutes, power setup 6-8). For samples of high silicate content, 0.1 mL of hydrofluoric acid (30% suprepure) was used. After cooling, the solutions were diluted to 25 mL with de-ionized water (18 MΩ).
Total arsenic was determined by hydride generation atomic absorption spectrometry. A Perkin Elmer (Waltham, Ma) Analyst 700 atomic absorption spectrometer was used in combination with a Perkin Elmer FIAS-400. Different calibration curves were constructed for the analysis. Arsenic species used to construct a calibration curve were As (III) in this study for total arsenic. As arsenic is generally present as a trace constituent in the media of interest, the method applied must be capable of accurately determining arsenic at low concentrations. The accuracy of the method was estimated by spiking known amounts of arsenic and evaluating bias (losses, contamination, or interferences), such as the recovery of the arsenic in the matrix. Both the original natural sample and the spiked sample are analyzed using the whole procedure several times. The bias (recovery test) is the systematic error of that measuring system.
The analytical precision (relative standard deviation), determined by quality assurance and quality control procedures, using duplicates, blanks, internal standards and reference materials, was better than ±10%.
Blood δ-Aminolevulinic Acid Dehydratase
The activity of erythrocyte δ-aminolevulinic acid dehydratase (ALAD) is a sensitive biomarker for arsenic toxicity 17-19 and oxidative stress. 20 The method is based on the conversion of δ-aminolevulinic acid by enzyme to porphobilinogen. The activity of blood ALAD was assayed according to the Berlin and Schaller 21 procedure published in 1974. Total volume of 0.2 mL of heparinized blood was mixed with 1.3 mL of distilled water and incubated for 10 minutes at 37°C for complete hemolysis. After we added 1 mL of standard δ-aminolevulinic acid (0.01 M), the tubes were incubated for 60 minutes at 37°C. Porphobilinogen formation is linear for at least 2 hours at 37°C. Enzyme activity was stopped after 1 hour by adding 1 mL of HgCl2 (1.35 g per 100 mL of 10% trichloroacetic acid [TCA]). After centrifugation of the reaction mixture (3000 rpm × 10 minutes), an equal volume of Ehrlich reagent was added to the supernatant and the absorbance was recorded at 555 nm after 5 minutes. The activity of blood ALAD was calculated by absorbance (Abs) and hematocrit (Ht) according to this relationship: ALAD activity = Abs × 100 × 2 × 35 / Ht × 60 × 0.062, where 2 = conversion factor from δ-aminolevulinic acid to porphobilinogen, 35 = dilution factor, 60 minutes = incubation time, and 0.062 L·μmol-1·cm-1 is the extinction coefficient of porphobilinogen.
Determination of the Lipid Peroxidation Products in Plasma
The thiobarbituric acid reactive substances (TBARS) in plasma were measured. 22 Plasma samples (400 μL) with an added 200 μL of mixture solution (8% sodium dodecyl sulfate, 1.5 mL of acetic acid 20% pH 3.5, 1.5 mL of thiobarbituric acid 0.8%, 300 mL of butylated hydroxytoluene 4%, and 300 mL of water) were heated at 95°C for 1 hour. To this was added 1.0 mL of water and 5.0 mL of butanol-pyridine (15:1 vol/vol). After centrifugation (3000 rpm × 10 minutes) of the reaction mixture, the absorbance was measured at 532 nm. Tetramethoxypropane was used as a stabilizer because malondialdehyde is not stable.
Determination of Reduced Glutathione in Blood
The level of reduced glutathione (GSH) in erythrocytes was determined by the Ellman 23 method. Erythrocyte suspensions were deproteinized by addition of TCA to a final concentration of 2%. After centrifugation, 5,5-dithio-bis(2-nitrobenzoic acid) was added, to a final concentration of 0.47 mM. The formation of 5-thio-2-nitrobenzoic acid, which is proportional to the concentration of acid soluble thiols, mainly GSH, was monitored at 25°C. The absorbance was measured at 412 nm in a Pharma Spec UV-1700 spectrophotometer (Shimadzu, Duisburg, Germany) against a reagent blank. The concentrations of reduced glutathione in samples were calculated from a standard curve of absorbance versus concentration of glutathione and expressed as nanomoles per milligram of total protein.
Isolation of Aortic Rings
Female Sprague-Dawley rats, aged 2 months (218 ± 10 g; n = 10) and 7 months (346 ± 8 g; n = 10), were killed by cervical dislocation. The thoracic aorta was quickly excised and placed in a cold (4°C) physiological KRB buffer containing (in mM) 4.2 KCl, 1.19 KH2PO4, 120 NaCl, 25 Na2HCO3, 1.2 MgSO4, 1.3 CaCl2, and 5 D-glucose. The aorta was cleaned of all adipose and connective tissue, and the midthoracic region was cut into rings (each 2-3 mm long, 2-3.5 mg). Extreme care was taken during preparation of the rings to avoid stretching the tissue. During the equilibration period, Krebs solution was maintained at 37°C and at pH 7.4 by constant bubbling with 95% O2 and 5% CO2.
Vascular Reactivity Experiments
In each experiment, we studied 4 to 8 adjacent aortic rings from the same animal, using the method for isometric tension measurements. 24 The rings were mounted on two 25-gauge stainless steel wires; the lower one was attached to a stationary glass rod and the upper one to an isometric transducer (Radnoti, Monrovia, Calif). The transducer was connected to a Biopac Systems, Inc. (Goleta, Calif) converter (MP100) and amplifier (DA100) for continuous recording of vascular tension using the AcqKnowledge 3.9.1 computer program (Biopac Systems). After the equilibration period, the aortic rings were stabilized by 2 successive near-maximal contractions with KCl (60 mM) for 10 minutes. Ten minutes after the contraction with phenylephrine (10-6 M), the accumulative concentrations of ACh were added to the medium (ACh 10-9 to 10-5 M). We did this to determine whether ACh-induced relaxation is necessary to contract the aortic rings with phenylephrine.
This protocol was repeated in different conditions: aortic rings untreated or incubated for 30 minutes with Nω-nitro-L-arginine (L-NNA, a NO synthase inhibitor [10-4 M]). To ensure that the resting tone of isolated aortas from female rats was of similar magnitude, resting tone of the blood vessels was assessed by relaxation with ACh (10-5 M) after the final contraction by phenylephrine (10-6 M).
Measurement of Blood Pressure
The rats were anesthetized by intraperitoneal administration of thiopental (Sigma, St Louis, Mo) at a dose of 60 mg/kg body weight. A catheter of polyethylene PE-50 tubing (Clay Adams, Sparks, Md) filled with heparinized saline (100 UI/mL) was placed in the carotid artery for the measurement of blood pressure. The arterial was connected to pressure transducer (DA100B, TSD 120 Biopac Systems), and blood pressure was measured using the AcqKnowledge III computer program. An interval of 15 to 30 minutes was allowed for the blood pressure to stabilize before the baseline blood pressure parameters were recorded.
Statistical Analysis
Values are expressed as mean ± standard error of the mean; n denotes the number of animals studied. One-way analysis of variance for repeated measures was carried out to detect significant differences, followed by Student-Newman-Keuls test to distinguish significant differences between the mean data from groups (eg, vascular reactivity experiments). The half-maximal concentration (EC50) of phenylephrine was determined from log-probit plots of the individual response versus concentrations. Finally, in some experiments the t test was used to distinguish significant differences among the means of rat data groups. We considered P < .05 to be statistically significant.
Results
Arsenic in the Hair of Rats
To test the presence of arsenic in the rat, we measured the arsenic concentration in the hair. Arsenic in tap water is correlated directly to arsenic levels in hair: for every 1 μg/L increment of arsenic in water, there is an increase of 0.9 ng/g arsenic in hair. 16 Only in the 7-month-old arsenic-treated rats was a high level of arsenic detected in the hair (0.63 ± 0.11 μg/g dry hair; n = 5).
Relaxation Induced by ACh in Aortic Rings From Female Rats
To compare the effect of age on the ACh-induced relaxation, we studied rats with access to tap water and pure water. The result indicated that in both arsenic-treated and control rats, the vascular relaxation was incomplete at 7 months of age. However, the effect was statistically more drastic in rats that were drinking tap water from Antofagasta, which contained approximately 20 μg of arsenic per liter (Figure 1 and Table 1, Figure 2). The mean response with the maximal concentration ACh (10-5 M) was 58% ± 6% of 60 mM KCl-induced contraction in 7-month-old arsenic-treated rats versus 77% ± 5% in control rats of the same age (Figure 1 and Table 1; P < .05); the sensitivity (EC50) to ACh was significantly reduced in 7-month-old arsenic-treated rats (245 ± 30 nM) compared with control animals of the same age (60 ± 22 nM; P < .001). In 2-month-old rats, the maximal concentration of ACh (10-5 M) and EC50 were not different between arsenic-treated and control rats (Figure 1 and Table 1). Therefore, these findings showed a statistically significant difference between control and arsenic-treated animals at 7 months of age.
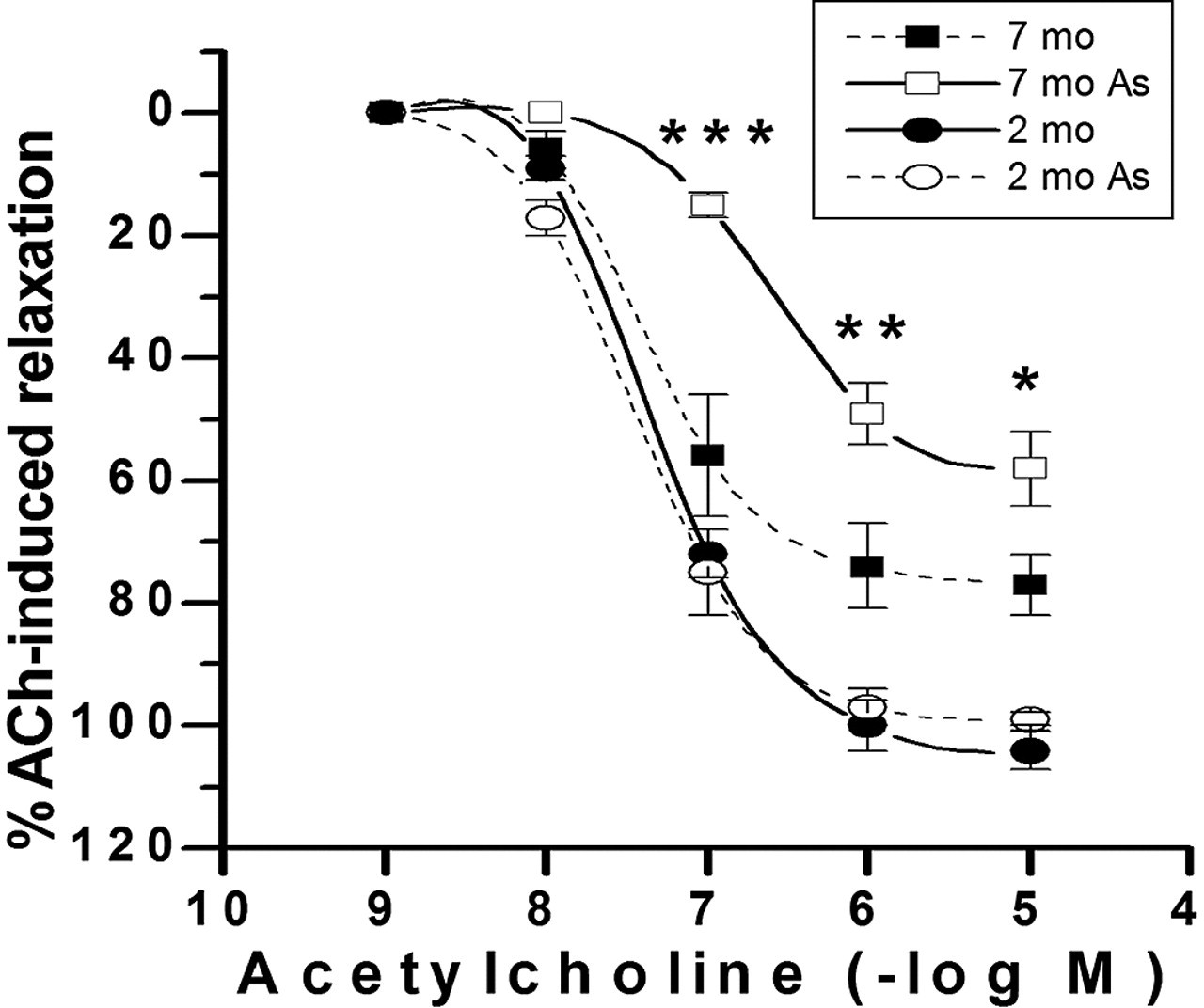
Concentration–response curves for acetylcholine (ACh) in endothelium-intact aortic rings from 2-month-old female and 7-month-old female Sprague-Dawley rats that were exposed to arsenic and animals that were not exposed. Arteries were preconstricted with 10-6 M phenylephrine. The results are mean ± standard error of the mean of 5 rats, with each point assayed in triplicate. Asterisks indicate statistically significant differences (*P < .05, ***P < .001). Points without error bars have a standard error of the mean smaller than symbol size.
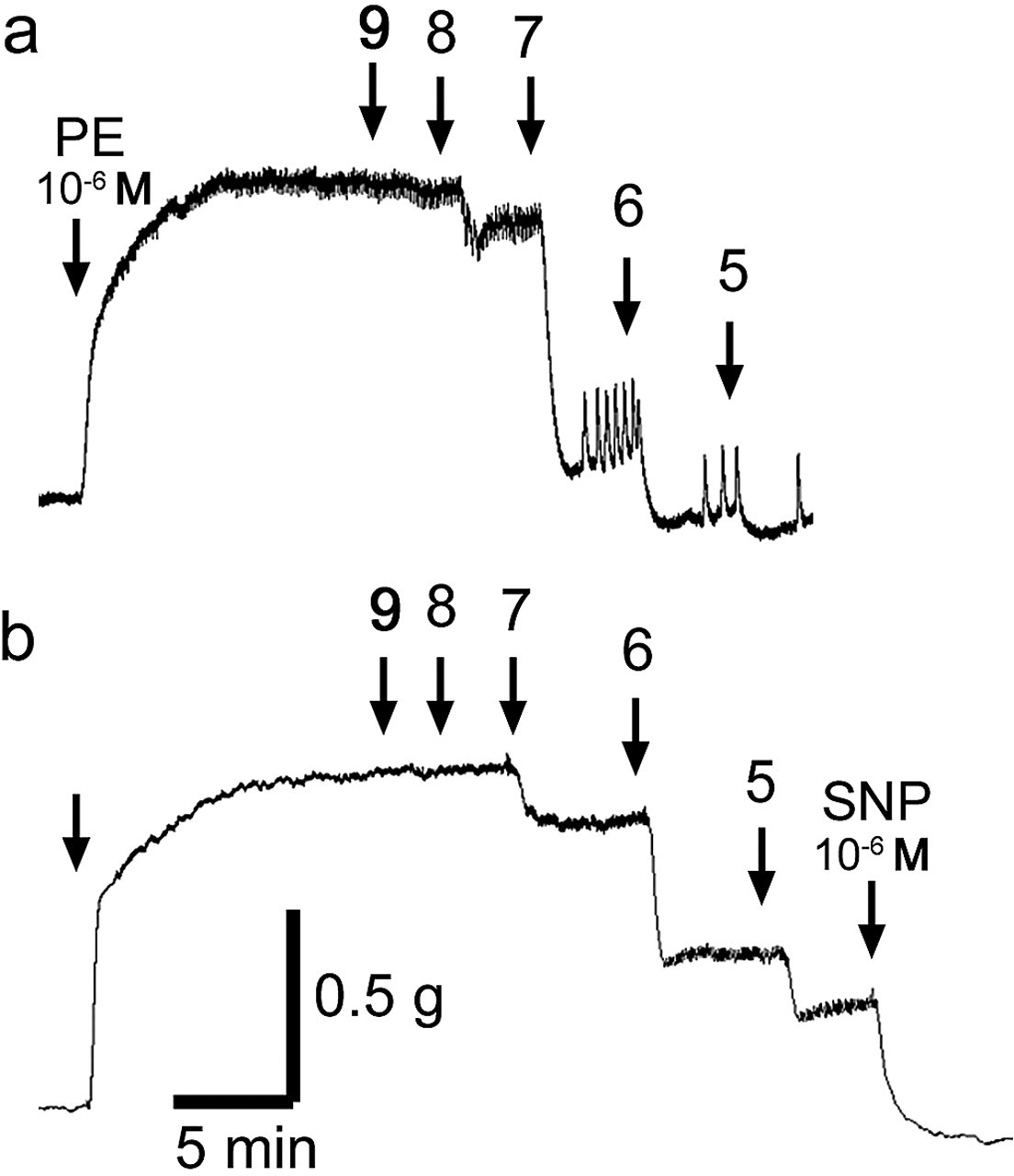
Original tracings showing the time course of the acetylcholine (ACh)-induced relaxation in intact aortic rings from (a) 2-month-old and (b) 7-month-old female rats treated with arsenic. The aortic ring was contracted with phenylephrine (PE, 10-6 M) previously. The figure shows cumulative concentration response for ACh-induced relaxation (10-9 to 10-5 M ACh).
Vascular Response to Acetylcholine in Rat Aortic Rings, Chronically Exposed to Arsenic (Arsenic-Treated)

NS, not significant; EC50, half-maximal concentration; Emax, maximal effort.
Values are mean ± standard error of the mean.
a P < .05;
***P < .001 compared with 2-month-old rats.
Relaxation Induced by Sodium Nitroprusside (NO Donor)
To study whether vascular smooth muscle relaxation mechanism is altered by arsenic effect, the aorta was stimulated with a NO donor. The relaxation to sodium nitroprusside (10-6 M) was complete in aortas of 2- and 7-month-old arsenic-treated rats (Figure 3). Similar results were observed in control rats (Figure 3).
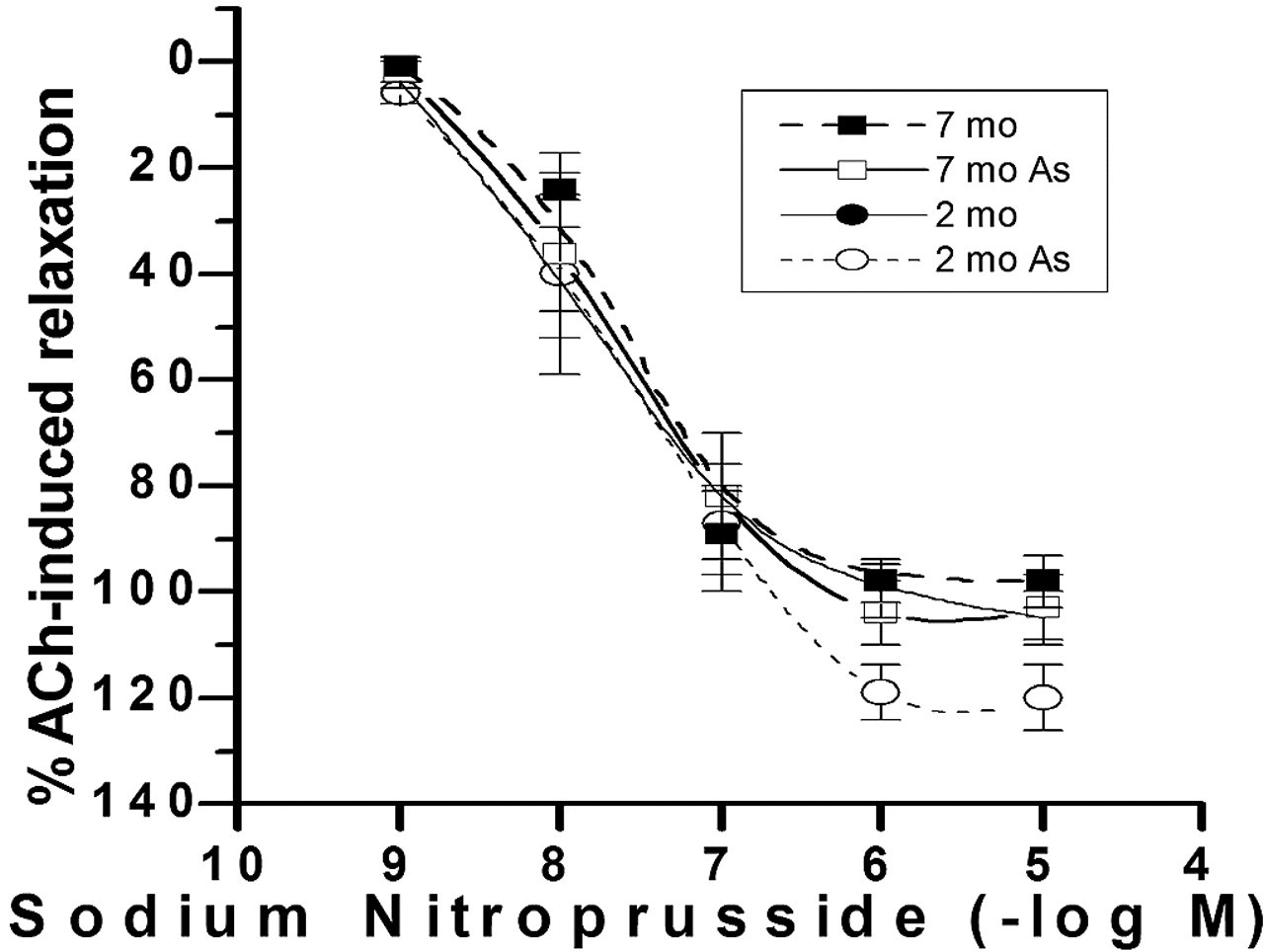
Sodium nitroprusside concentration–response curves in endothelium-denuded aortas from 2-month-old female and 7-month-old female rats that were exposed to arsenic. Arteries were preconstricted with 10-6 M phenylephrine. Each data point represents the mean ± standard error of the mean of 5 experiments.
Role of NO in Vascular ACh-Induced Relaxation
The endothelium-dependent relaxation due to ACh is mainly related to NO synthesis. Therefore, we examined the effect of a NO synthase inhibitor (L-NNA) on ACh-mediated vasorelaxation. As shown in Figure 4, the NO was involved: L-NNA (10-4 M) significantly blunted the ACh-induced relaxation (10-7 to 10-5 M) in both age groups of arsenic-treated rats: 12% ± 10% in 2-month-old rats (ACh 10-5; P < .01 vs control) and 17% ± 5% in 7-month-old rats (ACh 10-5; P < .01 vs control).
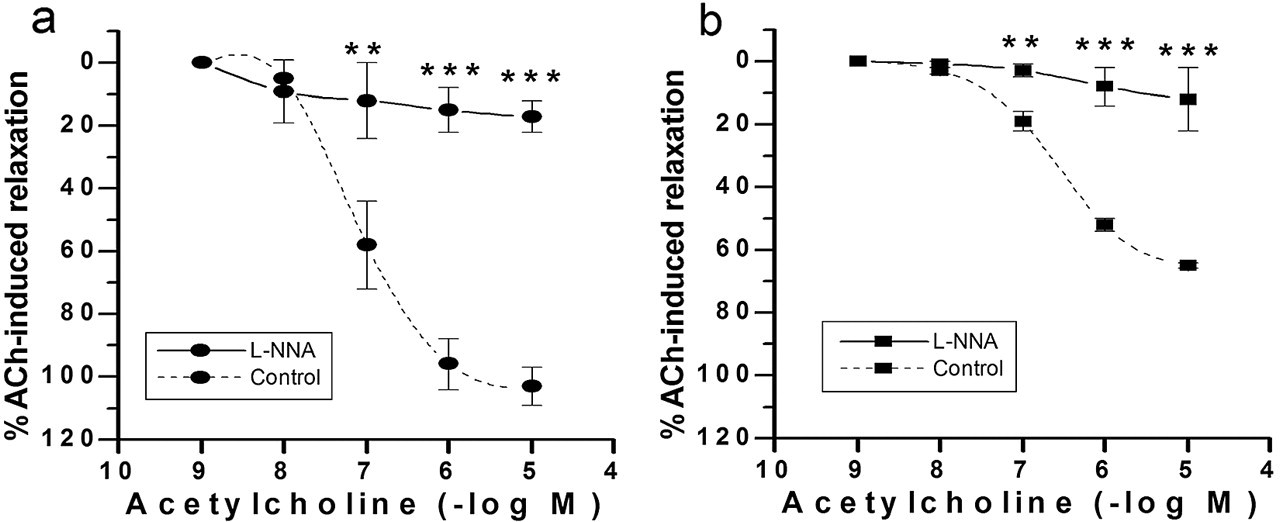
Acetylcholine (ACh) concentration–response curves in endothelium-intact aortas from 2-month-old (a) and 7-month-old (b) female rats in the presence or absence (control) of 10-4 M L-NNA. Arteries were preconstricted with 10-6 M phenylephrine. The results are mean ± standard error of the mean of 5 rats with each point assayed in triplicate. Asterisks indicate statistically significant differences (**P < .01, ***P < .001). Points without error bars have a standard error of the mean smaller than symbol size.
Oxidative Stress in Rats Drinking Tap Water
Chronic exposure to arsenic in tap water produces oxidative stress. 9 Hence, we analyzed the oxidative stress in the blood of arsenic-treated rats. As shown in Table 2, TBARS were increased in 7-month-old arsenic-treated rats (450 ± 64 nmol/L; P < .01) versus 2-month-old arsenic-treated rats (226 ± 37 nmol/L). The level of GSH decreased in 7-month-old arsenic-treated rats (17 ± 2 nmol/mg total protein; P < .001) versus 2-month-old arsenic-treated rats (74 ± 6 nmol/mg total protein). In addition, ALAD significantly decreased in 7-month-old arsenic-treated rats (6.2 ± 0.8 U/I) compared with 2-month-old arsenic-treated rats (14.5 ± 1.5 U/I; P < .001). The reduction of ALAD activity is a marker of oxidative stress. 19
Oxidative Stress in Female Rats Chronically Exposed to Arsenic
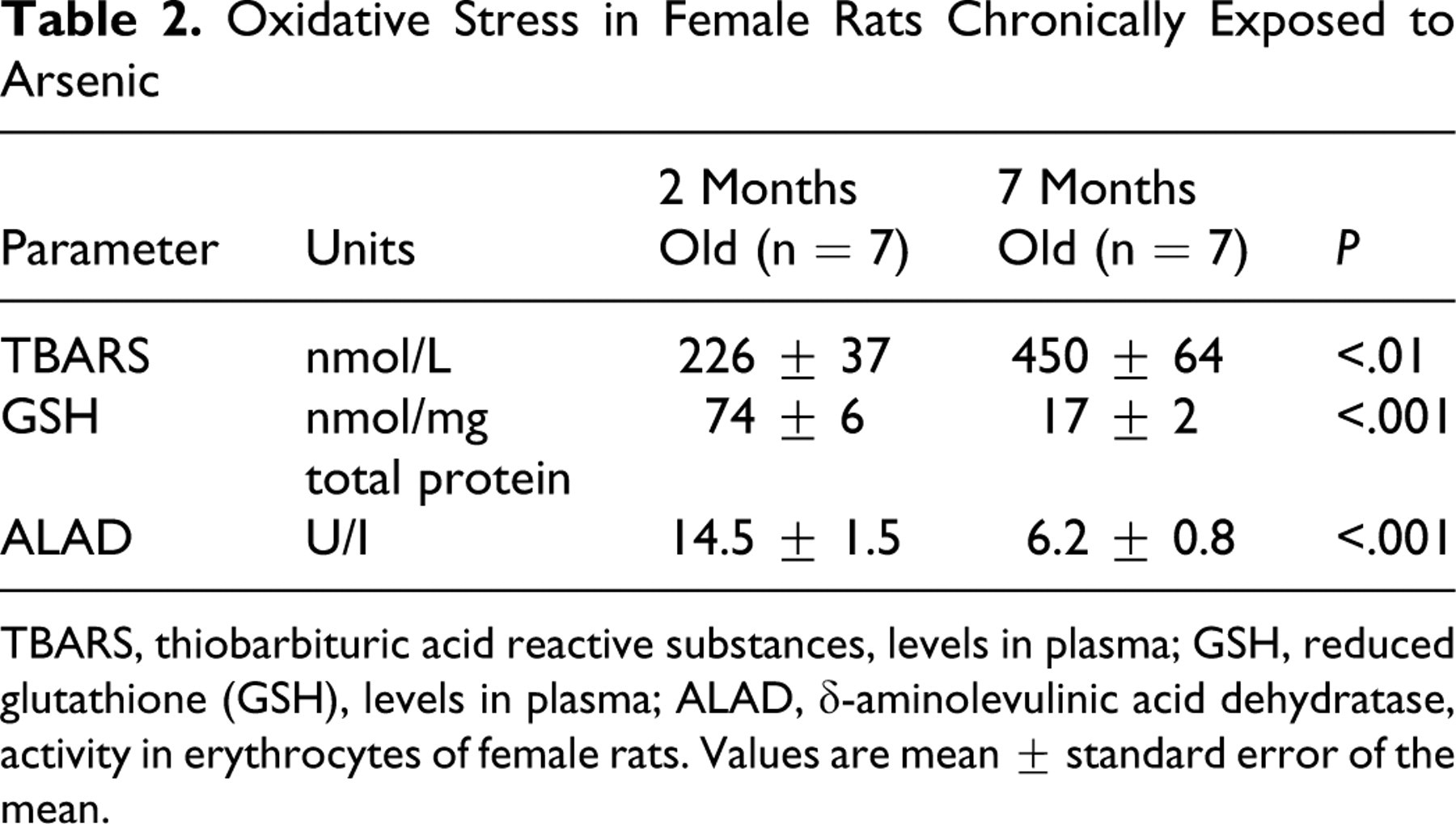
TBARS, thiobarbituric acid reactive substances, levels in plasma; GSH, reduced glutathione (GSH), levels in plasma; ALAD, δ-aminolevulinic acid dehydratase, activity in erythrocytes of female rats. Values are mean ± standard error of the mean.
Factors That Influence Arterial Blood Pressure in Rats Drinking Tap Water
To determine whether the findings of this study may have clinical implications, we measured different cardiovascular parameters in female rats. We did not find differences in systolic or diastolic blood pressures and heart rate between control and arsenic-treated 2-month-old rats. Figure 5 summarizes systolic and diastolic blood pressures between 2- and 7-month-old controls and 2- and 7-month-old arsenic-treated rats. Diastolic blood pressure increased in 7-month-old arsenic-treated rats (140 ± 6 mm Hg) compared with 7-month-old control rats (122 ± 5 mm Hg; P < .05) and 2-month-old control rats (110 ± 2 mm Hg; P < .01).
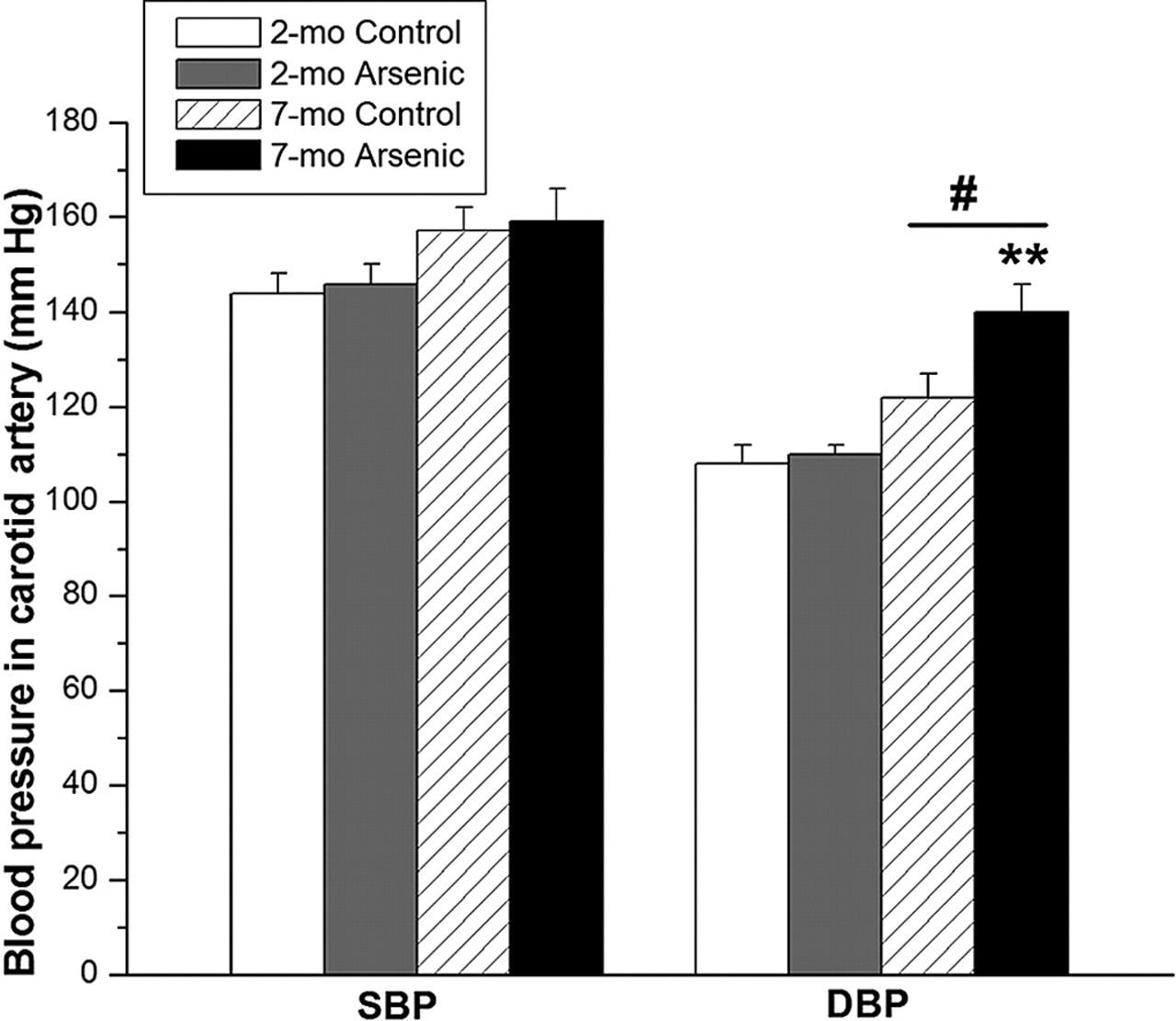
Systolic blood pressure (SBP) and diastolic blood pressure (DBP), of 2-month-old controls (open bars), 2-month-old arsenic-treated rats (gray bars), 7-month-old controls (diagonally lined bars), and 7-month-old arsenic-treated rats (black bars). Values are mean ± standard error of the mean of 5 experiments in mm Hg. Statistically significant differences: **P < .001 vs 2-month-old control and/or arsenic-treated rats; # P < .05 vs 7-month-old controls.
The mean arterial pressure (MAP) increased in arsenic-treated rats (Table 3). The MAP was 124 ± 2 mm Hg in 2-month-old controls versus 148 ± 7 mm Hg in 7-month-old arsenic-treated rats (P < .05). We calculated the cardiac output (CO) and peripheral resistance (PR) as the percentage of change between relative CO and PR in 7-month-old arsenic-treated versus control rats. As shown Table 3, the percentage of change in CO decreased significantly in 7-month-old arsenic-treated rats (–95%; P < .01) compared with control rats of the same age, and the percentage of PR change increased in 7-month-old arsenic-treated rats (51%; P < .001) compared with control rats of the same age. No differences were found in MAP, CO, or PR in 7-month-old control rats versus 2-month-old control rats.
Mean Blood Pressure (MAP), Pulse Pressure (PP), Heart Rate (HR), Cardiac Output (CO), and Peripheral Resistance (PR) of Controls and Arsenic-Treated Rats
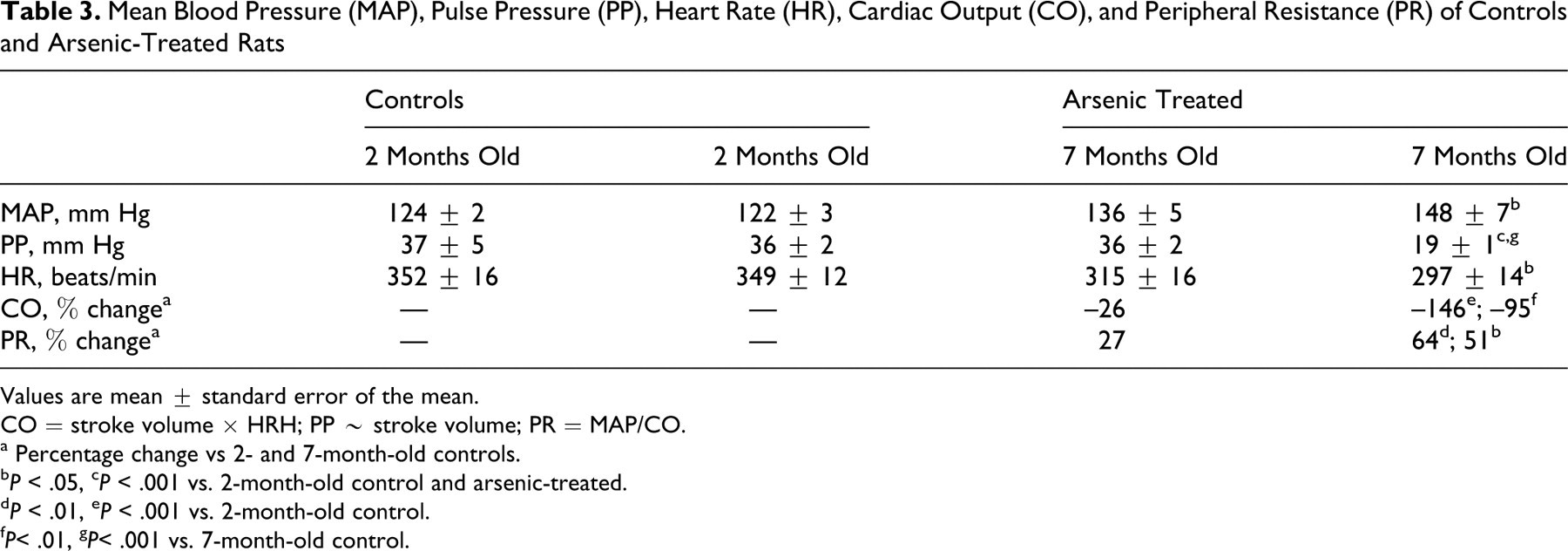
Values are mean ± standard error of the mean.
CO = stroke volume × HRH; PP ∼ stroke volume; PR = MAP/CO.
a Percentage change vs 2- and 7-month-old controls.
b P < .05,
c P < .001 vs. 2-month-old control and arsenic-treated.
d P < .01,
e P < .001 vs. 2-month-old control.
f P< .01,
g P< .001 vs. 7-month-old control.
Discussion
Since 2004, the arsenic concentration in tap water from Antofagasta City has been close to 20 μg/L. 2,25,26 This means that the arsenic concentration in tap water from Antofagasta City is almost double the amount recommended by the WHO—10 μg/L. 3 We found that arsenic accumulated in the hair of the rats (0.63 ± 0.11 μg/g of dry hair) that were exposed to this ecotoxicological metal for 7 months.
The wall of a blood vessel consists of an endothelial cell monolayer that rests on the smooth muscle cells, which are rich in elastic and collagenous fibers. Endothelium regulates blood flow by secreting vasoactive substances, which stimulate the smooth muscle cells resulting in vasoconstriction or vasodilation. Acetylcholine induces the release of endothelial NO by acting on muscarinic receptors, and NO stimulates the soluble guanylate cyclase in smooth muscle cells, producing vasodilation; removal of the vascular endothelium blunts the relaxation by ACh. 11,27 To test whether arsenic altered the endothelial relaxation mechanism, experiments were conducted in intact aortic rings in the presence of ACh and a NO synthase inhibitor (L-NNA) or NO donor (sodium nitroprusside).
We observed that ACh-induced relaxation in aortas of 7-month-old rats was significantly impaired in arsenic-treated rats compared with control rats of the same age, and no changes were observed in 2-month-old control or arsenic-treated animals. Although we did not demonstrate that arsenic directly affects the ACh-induced relaxation of the rat aorta, these results are in agreement with others studies, which demonstrated that the in vitro incubation with arsenic inhibits ACh-induced relaxation of the aortic rings from rats in a concentration-dependent manner. 28
The pre-incubation of aortic tissue with L-NNA (a NO synthase inhibitor) significantly blunted the ACh relaxation in both groups of arsenic-treated rats (2 and 7 months old), indicating that vascular relaxation is caused by NO. The relaxation in response to sodium nitroprusside (an NO donor) was complete in all groups of rats (2 and 7 months old, control and arsenic treated), suggesting that the vascular smooth muscle relaxation mechanism was not altered in 7-month-old arsenic-treated rats.
Pi et al 29 demonstrated that arsenic suppresses the activity of endothelial NO synthase in human umbilical vein endothelial cells. 29 Other studies have noted that prolonged exposure to arsenic of inhabitants of Inner Mongolia, who continued to drink well water containing inorganic arsenic, reduced the level of NO in their blood. 29
It is possible that age also reduces the endothelial NO release in rat aorta, 30 but it is difficult to explain such marked reduction of sensitivity (EC50) and maximal effect (Emax) to ACh, which was not observed in control rats. In addition, the significant increase in diastolic blood pressure in 7-month-old arsenic-treated rats versus control animals of the same age appears to be an effect of treatment.
The significant increase of TBARS in plasma and decrease of GSH in erythrocytes in 7-month-old arsenic-treated rats suggest that oxidative stress could be altered by arsenic and/or age. Although we did not determine GSH and TBARS concentrations in control rats, several studies have demonstrated that there is no difference in GSH or TBARS concentrations in blood between immature rats (2 months old) and adult rats (6 months old). 31-33 Moreover, the significant reduction of ALAD activity in erythrocytes from 7-month-old rats that we observed is a very sensitive biomarker for arsenic toxicity 17,18,34 and is another effect of oxidative stress 19 that is independent of variation of age. 20 Therefore, these data indicate that the oxidative stress in female rats was mainly due to the arsenic exposure.
To determine the physiological significance of these results, we determined systolic and diastolic blood pressure and heart rate. The diastolic blood pressure was increased significantly in 7-month-old arsenic-treated rats over control animals of the same age; also, the MAP, CO, and PR were different in 7-month-old arsenic-treated rats versus 2-month-old rats. The PR was determined by MAP and CO according to this relationship: PR = MAP / CO. The CO was calculated as stroke volume × heart rate. The stroke volume (as proportional pulse pressure) and heart rate decreased in 7-month-old arsenic-treated rats; thus, CO decreased. The reduction of stroke volume suggests that volumes of blood that returned from the venous system decreased. Therefore, the increased blood pressure observed in 7-month-old arsenic-treated female rats may be explained, in part, by an increase of PR.
Conclusion
Arsenic-treated rats presented impaired vascular response that may contribute to differences in vascular tone and factors that influence arterial blood pressure. Few studies are available concerning the effect of very low arsenic concentrations in tap water on cardiovascular response during the normal process of animal maturation. These findings are relevant because of the high mortality index of cardiovascular and stroke-related diseases among the population of northern Chile.
Footnotes
Acknowledgements
We thank Katherine Johnson (Escuela de Inglés, Facultad de Humanidades, Universidad Católica del Norte) for her helpful collaboration in the preparation of this manuscript.
The authors declared no conflicts of interest with respect to the authorship and/or publication of this article.
This work was supported, in part, by grants from Fondo Interno de Investigación Científica de la Universidad Católica del Norte (DGIP 220203-10301206 y DGIP 220203-10301239) and Dirección General de Investigación (DIRINV 1339-2007) de la Universidad de Antofagasta.
