Abstract
A cumulative risk assessment is generally intended to address concurrent exposure by all exposure routes to a group of chemicals that share a common mechanism of toxicity. However, the contribution of different exposure routes will change over time. This is most critical when estimating risks to infants and children because their exposure sources change rapidly during the first few years of life because of dietary and behavioral changes. In addition, there may be changes in sensitivity to toxicants during this time period, associated with various developmental stages. Traditional risk assessments do not address this progression. Examples of how these factors might be incorporated into an early life risk assessment are provided for lead, dioxins and furans, and organophosphate pesticides. The same concepts may apply to other potentially susceptible subpopulations, such as the elderly.
Cumulative risk has most frequently been assessed for acute exposures, in which chemicals that share a common mechanism of toxicity are considered together with various combinatorial strategies. This is of limited usefulness for risk assessment of chemicals in drinking water, which is the focus of our group, because most risk assessments are based on long-term exposures. The situation in these cases is much more complex because exposures change over time and because exposure history can affect subsequent sensitivity to toxicants. Cumulative risk, in this sense, is much more than assessing the significance of concurrent exposures by all exposure routes to a group of similar chemicals; it must integrate exposures over time and consider the changing physiological status and chemical exposures over time. This is most critical for risk assessment for infants and children because of their rapid growth and development, their changing diet, and their behavioral changes during the first few years of life. Assessing the cumulative impact for exposures during this period is a formidable challenge and brings up many questions. This brief article cannot attempt to provide answers for all the questions and resolve the issues of optimum strategy for assessing this time-integrated cumulative risk but will attempt to describe the issues and pose the question of how they should be addressed.
Critical Changes During Development
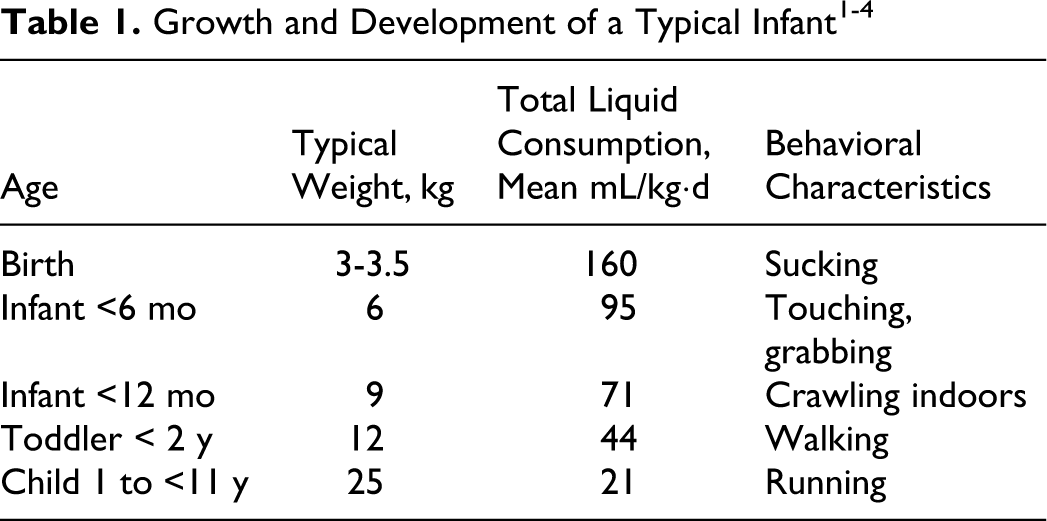
To address the critical issues for risk assessment in the developing infant, the growing infant and the stages through which they pass need to be described. The changing exposure patterns should be explicitly considered and acknowledged in the risk assessment. A basic description is provided in Table 1.
As infants go through a 10-fold increase in weight during their early years, toxicants accumulated in the body during the early years will be diluted 10-fold. Note the very high liquid consumption rates of infants, on a body weight basis. 1 Breathing rate and total food consumption change in a comparable pattern, related to a higher metabolic rate of the growing infant. 2-4 The changing behavior patterns with growth and development must also be considered. All these factors will affect the relative exposure to toxic chemicals during the early years and should be considered in a cumulative risk assessment.
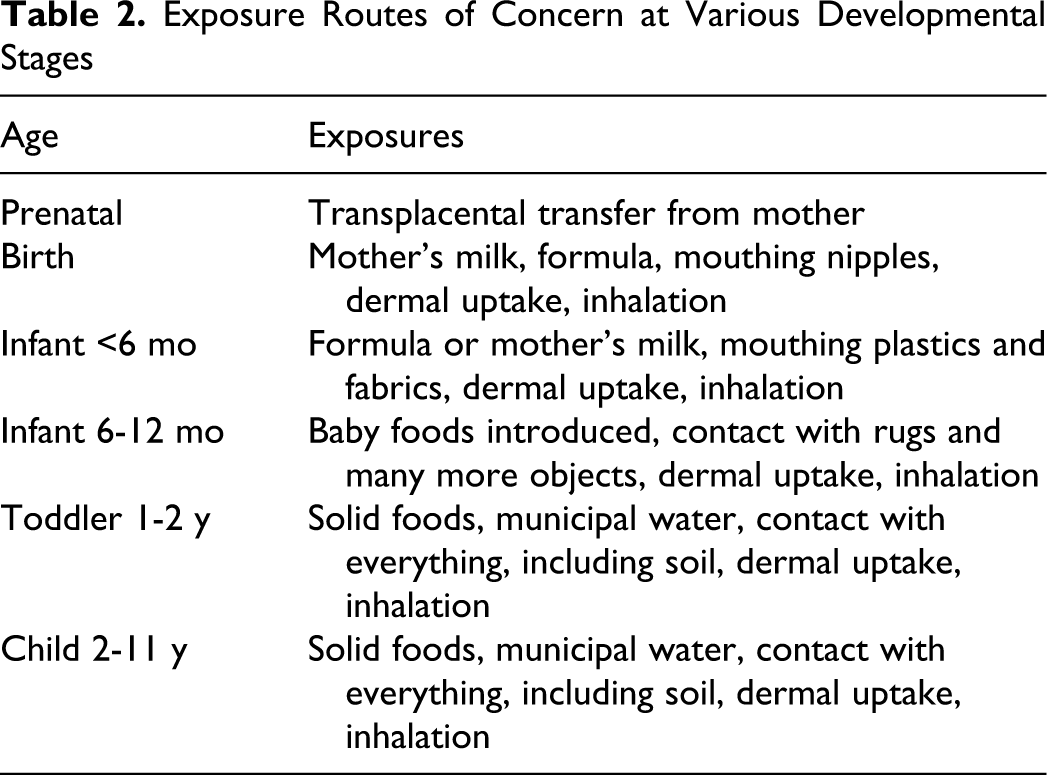
During the progression from fetus to infant to child, the main exposure routes will also change, as discussed at length in the US Environmental Protection Agency (EPA) children’s exposure factors handbook 3 and summarized in Table 2.
Exposure Routes of Concern at Various Developmental Stages
Chemicals with different properties will be delivered to the growing fetus and infant in different doses or proportions through the various exposure routes, which means that doses of environmental chemicals of concern will vary by life stage. Table 3 provides a simplified example of this for 3 representative chemical types: dioxins and polychlorinated biphenyls (PCBs), representing highly lipophilic chemicals; lead, a toxic heavy metal that deposits in bones; and organophosphate pesticides, which have a short residence time in the body.
Relative Exposure to Environmental Chemicals, Considered on a Body Weight Basis
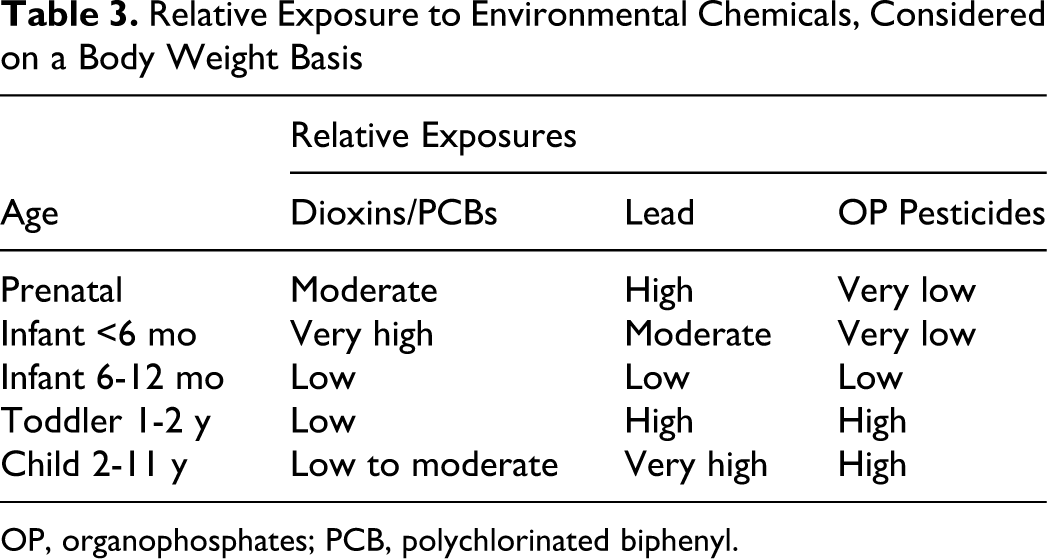
OP, organophosphates; PCB, polychlorinated biphenyl.
These differences in exposure pattern of the growing infant will make a tremendous difference to the body concentration of chemicals, as the infant passes through various stages of growth and development. Table 4 is a more specific example of the potential exposure patterns for dioxins and furans, adapted from a recent presentation by Leung et al. 5
Dietary and Dermal Intakes of Polychlorinated Dioxins/Furans (pg TEQ/kg/d)
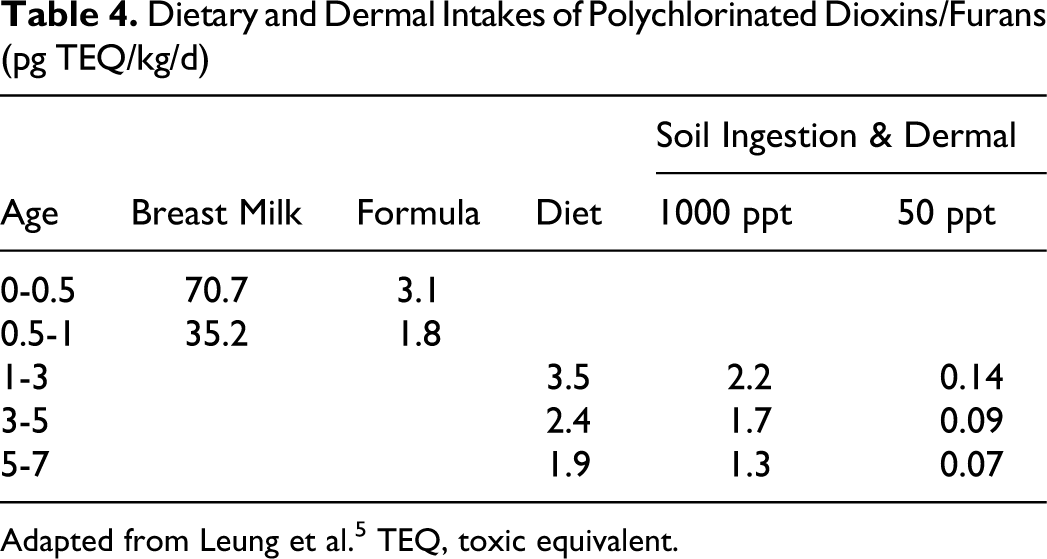
Adapted from Leung et al. 5 TEQ, toxic equivalent.
The following figures illustrate what this means in terms of tissue concentrations. Figure 1 shows the theoretical rate of accumulation of a chemical in the growing infant with a constant exposure, on a dose/kg basis, over the first 2 years of life, plus a proportionate small amount delivered to the fetus. The pattern of accumulation depicted accounts for dilution of the body burden with growth; excretion and metabolism are ignored. As shown in the figure, the tissue concentrations would increase steadily with this theoretical exposure pattern, despite the 4-fold increase in body weight over the 2-year period.
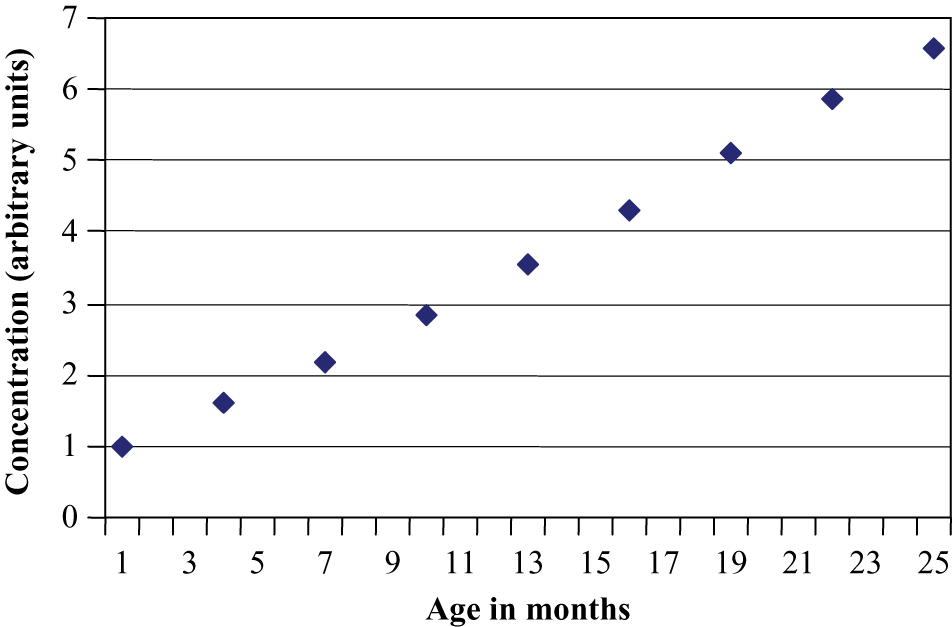
Cumulative body burden in infants with a constant exposure per kg body weight during the first 2 years of life.
However, the actual exposures to an infant are not constant over time because of the changes in exposure sources. Figure 2 shows approximately what will happen to the net concentration of dioxin in an infant’s body, with the relative exposures discussed in Tables 3 and 4, for a breastfed infant.
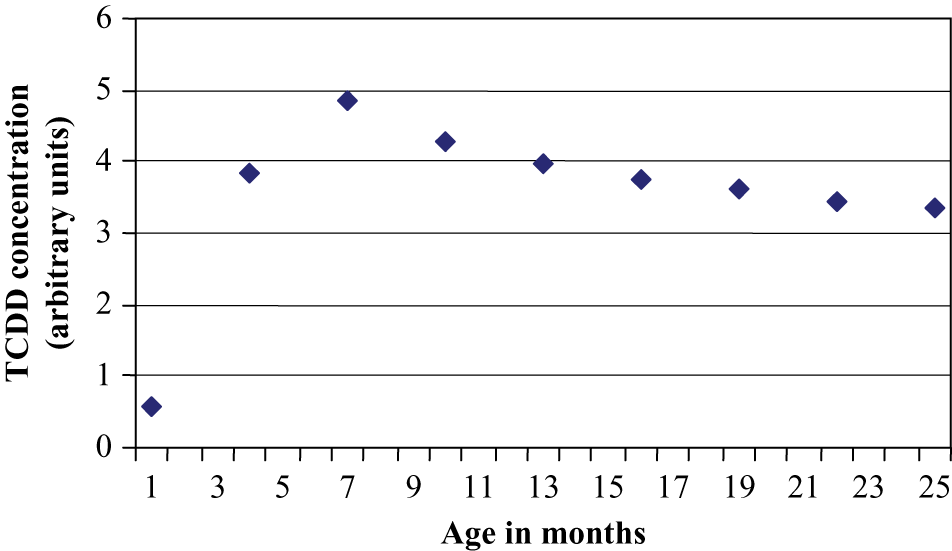
Relative dioxin concentration in infants.
Figure 2 also does not consider the effects of metabolism and excretion, but because dioxin’s half-life is quite long (~6 to 10 years), 6-8 accounting for these factors does not change the resulting pattern. In addition, the changing proportion of body fat in the fetus and infant will affect the whole-body distribution, the elimination rate, and the actual blood concentration for lipophilic chemicals such as dioxins and furans. The striking difference in the whole-body concentrations in this case, compared with the steady dose rate example in Figure 1, is worthy to note. The high early exposure should be a matter of special concern about potential adverse effects during this critical developmental period.
The lead exposure pattern shown in Table 3 represents a theoretical urban exposure pattern where there may be lead paint residues plus a typical soil contamination with lead. In this example, the infant’s mother has had significant environmental exposures to lead over her lifetime, which has deposited in her bones. The large, early delivery of lead to the fetus is associated with mobilization of calcium from the mother’s bones, to support fetal bone development. 9-11 This process continues during breastfeeding, as the mother continues to deliver calcium for bone building to her baby. 12 Lead exposure decreases as the infant is switched to formula, then to solid foods, but then increases as the infant begins to explore his or her surroundings, begins to chew on things, and has access to house dust and soils. The highest exposures of children are associated with pica, in which children are actively consuming soil. 3
Exposure to organophosphates (OP) pesticides, also summarized in Table 3, is relatively low during prenatal and early postnatal life because of the short biological half-life of these chemicals; the “area under the curve” of blood concentration is relatively low in both mother and fetus. OP pesticides will pass through the placenta but do not accumulate in the fetus. 13 As the infant is introduced to soft foods, such as strained fruits and vegetables, potential OP doses reach much higher levels than at any other life stage. This is because of the simpler diets of infants, compared with adults, and because infants have much higher food consumption, on a body weight basis, than do adults and even adolescents. 3
These 3 examples illustrate fundamental differences in exposure patterns during development for various chemical classes, but much more needs to be added to this to develop comprehensive guidance for cumulative risk assessment during early development.
Challenges for Cumulative Risk Assessments in the Fetus and Infant
The overall guiding principles for consideration of early life exposures are as follows: Early life consumption rates are very high. Dose rate varies differently by life stage and exposure pattern for different types of chemicals. Early development is a series of critical periods where processes must take place on an orderly schedule. Sensitivity to disruption may vary during these critical periods. Interaction of all these factors results in potential vulnerable periods for chemical exposures.
Disruption of developmental processes can result in delayed or premature development, which can result in lifelong changes, depending on the severity of the effect.
14
One example of this is differentiation of the sexual organs, which occurs primarily before birth in humans.
15
Another example is the migration of neurons through the developing brain, followed by establishment of the pattern of receptive fields in the cerebral cortex.
16
The latter process continues long after birth.
17,18
If we are to assess cumulative impacts from exposures to chemicals during gestation and infancy, can we take all of these factors into account? How do we group chemicals for this purpose, and where should we start? The 3 classes of chemicals already introduced will be used as examples.
Organophosphates
Most progress has been made so far in developing cumulative risk assessment guidance for OPs, and the particular problems of evaluating the possible effects of combined OP exposures were described several years ago in the groundbreaking National Research Council (NRC) report, Pesticides in the Diets of Infants and Children. 19 This work continued with the US EPA OP cumulative risk assessment, published in 2002, 20 and the recently released 2006 update. 13 These cumulative risk reports do an excellent job of exposure modeling for infant exposure to multiple OP pesticides and then use the relative potency factor to add up the effective contribution from a number of similar chemicals. Food, water, and residential exposures are all included. The relative potency factors are based on brain acetylcholinesterase (AChE) inhibition in female rats after 23 days or more of daily administration.
These US EPA efforts are a tremendous advance for cumulative risk assessment but do not yet explicitly consider one of the principles just described, which is whether or how much the sensitivity of the developing systems vary with time. As both the peripheral and central cholinergic systems develop, it seems plausible that they could vary in sensitivity to cholinesterase inhibitors. Reproductive and developmental studies on OPs indicate some examples of extra sensitivity during development, 19,21,22 although decreased sensitivity may also occur. 23 Because OPs inhibit many serine esterases in addition to AChE, 24 and the critical periods and inhibition recovery times for these esterases will not be identical to that for AChE, the current procedures focusing exclusively on AChE are not likely to adequately address the potential cumulative impacts during this early period (eg, see Furlong et al 25 ). Therefore, basing the relative potency factors for OPs exclusively on their effects in adult women is potentially missing the point for a cumulative risk assessment for infants, who are one of the populations of special concern. Further work exploring this issue is needed.
Dioxin-Like Chemicals
Dioxins and dibenzofurans are the second best-studied group for cumulative risk assessment. Development of toxicity equivalent factors for these chemicals based on binding to the Ah receptor was a tremendous advance in categorizing their cumulative hazard. 8,26,27
Although not all the effects of dioxin-like chemicals are necessarily mediated through binding to the Ah receptor, 28 the major effects of concern for the dioxins and furans are apparently mediated through Ah. The reproductive, neurodevelopmental, immunological, and endocrine effects are most relevant, after cancer. 8 A wide variety of reports suggests that each of these types of effects can occur both through fetal exposures and postnatally, through the mother’s milk. 29-33 Neuronal effects can occur through direct actions on Ah receptors in neurons, not just through endocrinological disruption of the growing fetus. 34 In addition, it is necessary to consider the related compounds PCBs and polybrominated diphenyl ethers (PBDEs) in an assessment of dioxin-like activity. There is coexposure to all these compounds, based on their accumulation in the food chain. However, the ratio of the various contaminants can vary, partly because of the history of industrial pollution in various areas and partly due to environmental weathering of the mixtures. 7 The relative individual exposures will then vary, based on dietary choices. 35
When the exposure mixtures of concern are expanded to this broad grouping of lipophilic, dioxin-like chemicals, assessing the potential cumulative exposures and effects gets very complicated. Not all of the exposure can be characterized by that simplification of perinatal exposures to dioxin toxic equivalents (TEQs) described in Table 4 and Figure 2. The human distribution, metabolism, and resulting half-lives of the various congeners also vary considerably.
Toxicological endpoints of concern for this group of chemicals can be loosely divided into effects occurring during the pre- and postnatal periods, as shown in Table 5.
Potential Interactive/Cumulative Effects of Dioxins, Dibenzofurans, PCBs, and PBDEs
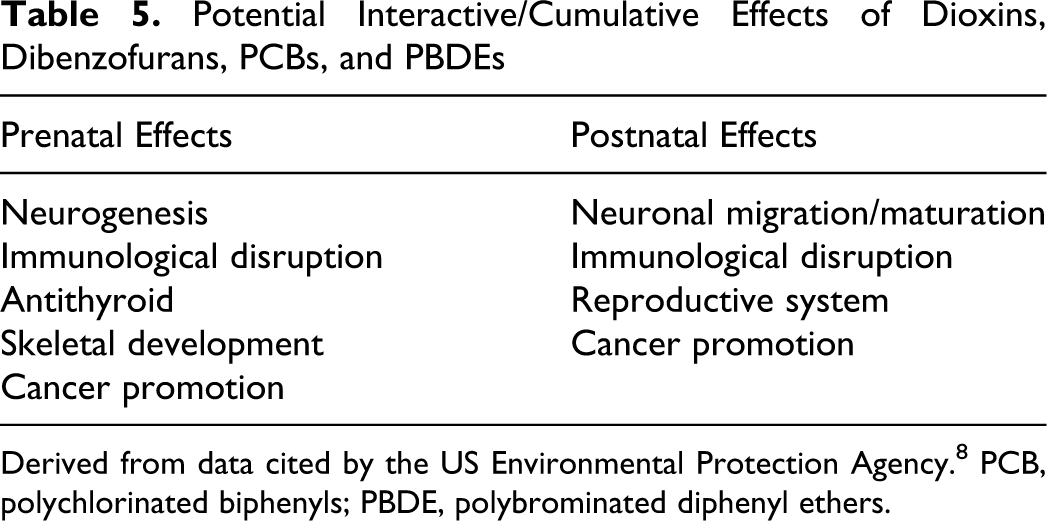
Derived from data cited by the US Environmental Protection Agency. 8 PCB, polychlorinated biphenyls; PBDE, polybrominated diphenyl ethers.
Distinguishing critical periods and effects is necessary for a time-dependent evaluation of the exposures. However, the division shown in Table 5 is undoubtedly an oversimplification. We should not assume that each of the effects on this list occurs through identical receptors throughout development. Similarly, we should not assume that all the chemicals and chemical groups interact with each of the receptors with the same relative affinity. In other words, the TEQs for the mixture of chemicals may be different for each effect. To quantitate the changing TEQs for these chemicals for the various effects for a cumulative risk assessment, it might be necessary to acknowledge the developing organism more explicitly, as suggested, for example, in Figure 3.
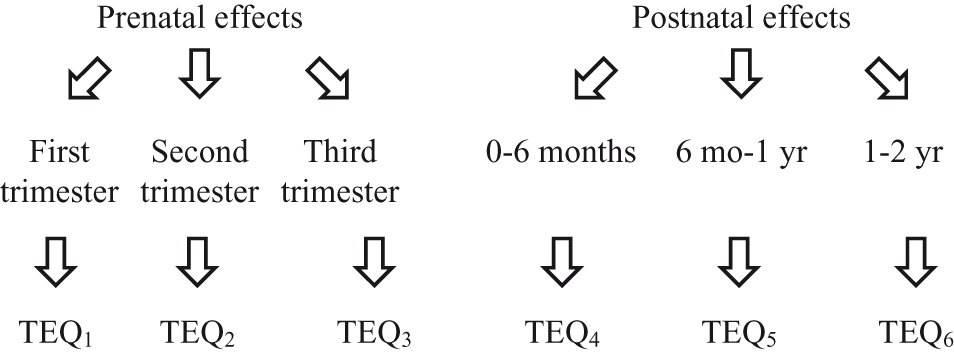
Potential interactive/cumulative effects of dioxins, dibenzofurans, polychlorinated biphenyls (PCBs), and polybrominated diphenyl ethers (PBDEs).
For a cumulative risk assessment for the various endpoints, we should also acknowledge that the TEQs are not necessarily fixed with respect to the different toxic endpoints. The cumulative response, then, might be estimated as shown in Table 6.
Estimation of Effects of Dioxin-like Chemicals on Different Toxic Endpoints
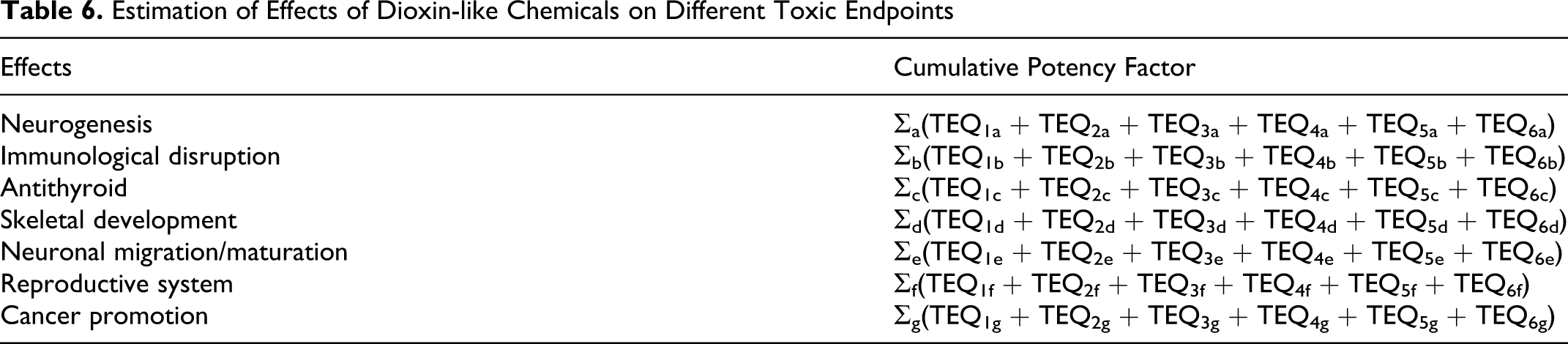
Each toxic endpoint may have a different critical period and a different summed toxicity quotient for the chemical exposures during that period, acting on the sensitive receptors. This is probably what is required to perform a truly data-driven cumulative risk assessment for this important class of chemicals.
A further question that should be asked is whether an effect of dioxin-like chemicals on an early stage of development sensitizes (or protects) the developing organism to other effects at a later stage in development. This is likely to be true for several types of effects. For example, it is known that mild early hormone effects may delay neuronal migration, which may affect sexual maturation years later. 14 Each of these physiological systems in the developing organism may be independently affected by the dioxins, furans, and PCBs, and a combined, interactive mechanism of action seems likely. 36-41
Lead
Cumulative risk from lead exposure, the last of the examples, appears to be conceptually much simpler than the other two. To reiterate, lead exposure is relatively high in the fetus if the mother has had significant lifetime exposure to lead, which deposits in her bones and then is mobilized with calcium to support the growth of fetal bone.
9-11
Lead is also delivered in the mother’s milk to the newborn.
12
As the infant begins crawling and mouthing behavior, she or he will start to accumulate lead from the general environment.
3,42
Oral and inhalation exposure to soil and residues of leaded paint will be the largest sources, with exposure from drinking water an added risk. Most foods are now low in lead. The major sources of lead exposure are as follows: Ingestion of contaminated urban soil or dust Ingestion of lead-based paint residues, which may be a problem in older houses with peeling paint Drinking water that has passed through lead pipes or lead soldered fittings Breathing air contaminated by combustion of leaded fossil fuels; by smelting, refining, and manufacturing operations; and by tobacco smoke Eating food grown on soil that contains lead or has been stored in leaded glass or in tins containing lead solder Occupational exposures (manufacture of lead-acid batteries, lead shot and fishing weights, leaded glass, radiators, ships; iron processing; construction and demolition of steel structures; firing ranges; lead ore production and smelting; recycling)
It is well known that lead exposure during early periods of life very sensitively disrupts brain development in multiple ways with multiple effects. It affects human intelligence,
43,44
it leads to hyperactive disorders with decreased attention span,
45
and it interferes with postural balance.
46
The actual mechanisms of this effect are not well understood, although many biochemical effects of lead are noted at various concentrations. Recent epidemiological studies
47,48
attempt to document a critical period for lead exposure during gestation but provide apparently conflicting conclusions (either the first or the third trimester seemed to be most sensitive to lead).
Because lead is sequestered in bones, might the early fetal dose be higher, before the lead is removed from the fetal circulation by storage in bone? Further along in development, at what age might a child become more resistant to the neuronal effects of lead? Also, it has been suggested that middle-aged men are most susceptible to hypertensive effects of lead but that high exposures during childhood can be a precipitating factor. 49,50 Each of these hypotheses suggesting time-dependent sensitivity changes would require more research to substantiate the potential sensitive period.
Conducting a cumulative risk assessment for lead is not, therefore, a straightforward task. But concentrating on the prenatal period through the first 2 years of life, it might be possible to develop a relative toxicity factor for several well-defined periods. An example of this approach is suggested in Table 7.
Cumulative Risk Assessment for Infant Exposure to Lead
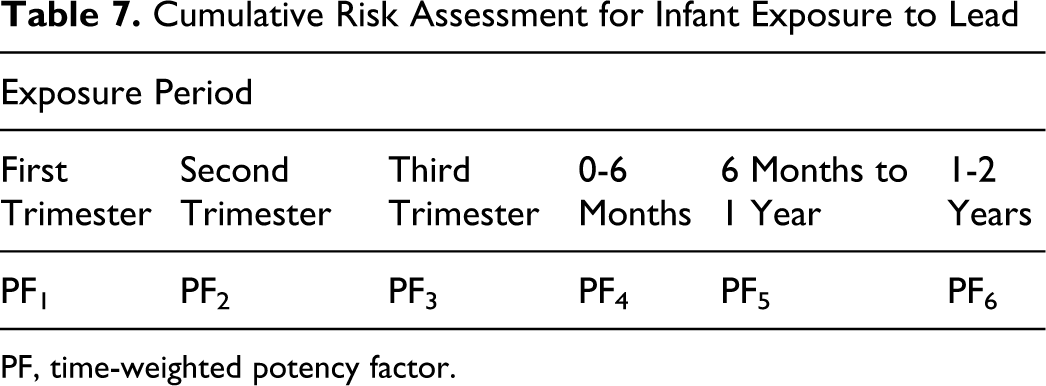
PF, time-weighted potency factor.
If exposure to lead is more dangerous in a particular trimester, for a later effect on IQ, then the cumulative risk assessment might weight exposure during this period more. If the putative effect of lead on blood pressure is mediated through effects on the brain, it might track with the IQ effects. If the effect is mediated through effects on the kidney or directly on the cardiovascular system, it might result in different sensitive periods and relative potency factors. Performing a cumulative risk assessment for lead with these principles in mind could help us direct attention to controlling the most critical exposures. As an additional caution, a better understanding of the mechanisms of lead interactions with many biological systems would be required to conduct this analysis, despite the huge amount of research that has been conducted on lead to date.
Conclusions
Conducting a formal risk assessment that quantitatively considers these questions of differential exposures and critical periods would be vastly more complex than existing protocols. It would also require much more mechanistic information. When and if the data to fill in the missing pieces become available, the quality of cumulative risk assessments for the prenatal/neonatal period will be able to be greatly improved. But in another sense, just recognizing the complexity of developing systems and the multiple simplifications that have been made with regard to children’s risk assessment should help ensure a more cautious approach.
Another aspect of the situation is that for cumulative risk assessments, parallel dose-response curves have commonly been assumed for combined exposures. However, as total chemical exposures increase, nonparallel dose-response interactions can occur. There may be additivity at low doses and synergism at high doses. It is known, for example, that dioxins affect the cytochrome P450-mediated metabolism of a variety of chemicals. But the cytochromes progress through a complicated developmental cycle in fetus and neonate, and the sensitivity to induction of each form is not well studied. So it might be appropriate to consider whether this effect might lead to toxic interactions during a particular critical period, such as the period about 6 months of age where concentrations of dioxin-like chemicals are expected to be highest.
Some of these same concepts are likely to apply to other potentially susceptible subpopulations in other circumstances. The elderly as a susceptible population is one concern. Another is adolescents going through the rapid changes of puberty. This concept of cumulative risk assessment could be extended to the specific life stages, exposures, and other effects of most concern.
The final consideration should be, is a large enough safety margin being provided with the uncertainty factors presently used in risk assessment, so the poorly documented cumulative exposures and potential effects are not a threat? We can hope so, but until more is known about sensitive periods and cumulative effects, this remains an open question. More consideration of these issues, such as that provided in a recent investigation of children’s coexposures to cadmium, lead, mercury, and arsenic, 51 would be extremely useful.
Footnotes
The author declared no conflicts of interest with respect to the authorship and/or publication of the article.
The author(s) received no financial support for the research and/or authorship of this article.