Abstract
The increased concern on the consequence of exposure to multiple chemical combinations has led national regulatory authorities to develop different concepts to conduct risk assessments on chemical mixtures. Pesticide residues were identified as “problem formulation” in the respective European regulations and in this context, the European Food and Safety Authority has suggested to group pesticidal active ingredients (AIs) into cumulative assessment groups (CAGs) based on the toxicological properties of each AI. One proposed CAG, on the liver, currently consists of 15 subgroups, each representing a specific hepatotoxic effect observed in toxicity studies. Dietary cumulative risk assessments would then have to be conducted assuming dose additivity of all members of each CAG subgroup.
The purpose of this publication is to group AIs based upon the knowledge of the pathogenesis of liver effects to discriminate between primary end points (direct consequence of chemical interaction with a biological target) and secondary end points (which are a consequence of, or that arise out of, a previous pathological change). Focusing on the relevant primary end points strengthens and simplifies the selection of compounds for cumulative risk assessment regarding the liver and better rationalizes the basis for chemical grouping. Relevant dose additivity is to be expected at the level of the primary/leading pathological end points and not at the level of the secondary end points. We recognize, however, that special consideration is needed for substances provoking neoplasia, and this category is included in the group of primary end points for which chemicals inducing them are grouped for risk assessment.
Using the pathological basis for defining the respective CAGs, 6 liver subgroups and 2 gallbladder/bile duct groups are proposed. This approach simplifies the cumulative assessment calculation without obviously affecting consumer safety.
Keywords
Introduction
Traditionally, toxicology and risk assessment has focused upon single chemicals, and this is also reflected in most chemical-related legislation.
However, the general public as well as professionals in industrialized and increasingly in third world countries are exposed to multiple and most frequently unknown chemical combinations in their everyday life from various sources including their food, drink, occupation, and general lifestyle. There are therefore a potentially infinite number of combined or cumulative dietary and/or nondietary, exposure scenarios, which can only be addressed by the identification of so-called “problem formulations,” as proposed in the Organization for Economic Cooperation and Development Series of Testing and Assessment No. 296—Consideration for Assessing the risks of combined exposure to multiple chemicals. 1 Dietary exposure to pesticide residues has been identified to be such a problem formulation in respective European regulations. According to the maximum residue level (MRL) regulation and the European Union (EU) pesticides 396/2005 and 1107/2009, 2 known cumulative and synergistic effects of pesticide residues shall be taken into account when deciding on the setting of MRLs and on the approval of active substances and plant protection products, provided the methods to assess such effects are available. A respective US Environmental Protection Agency (US EPA) Framework for cumulative risk assessment to specific pesticides in the diet offers a simple, flexible, structure for conducting and evaluating cumulative risk assessment within the US EPA. 3
The hazard basis of both (European and United States) cumulative risk assessment approaches are groups of similarly acting substances. In the United States, up to now, 5 relatively narrow cumulative assessment groups (CAGs) have been proposed, based on mechanistic data: organophosphates, N-methyl carbamates, triazines, chloroacetanilides, and pyrethrins/pyrethroid. 4,5 Under the US EPA approach, the basic principle used was to include active substances into a CAG only if data are available to proof, that substances have the same mode/mechanism of action, which leads to relatively small groups of substances, following the same mode of toxicological action.
In Europe, the European Food Safety Authority (EFSA) approached the problem by grouping substances in CAGs, based on effects observed in any of the toxicological studies (mice, rats, dogs, or rabbits); for example, histopathological or weight effects on the same target organs, independent of eventually available mode of action (MOA) data. 6 Following this approach, CAGs are larger and might contain substances, which do not contribute to the common toxicity. The first publication of the respective CAGs was focused on the thyroid gland and the nervous system. 6 In these approaches, dose additivity is the default assumption for the combination toxicity model, when similar toxicity is assumed on an organ toxicity basis, even if dissimilar modes of actions can be assumed. 7 Updated CAGs on the nervous system and the thyroid were subsequently published as draft scientific opinions in 2018 and 2019. 8,9 Further documents were published, on an organ by organ basis, for public consultation on the nervous system, the thyroid gland, the reproductive and developmental systems, the liver, the adrenal gland, and the eye. 10
This document critically assesses the current basis for deriving the EFSA liver CAGs and proposes a subcategorization, using a systematic approach to the pathological basis currently applied to derive the CAGs, to help guide whether the induced pathological effects are a primary effect of each chemical, in which case they can justifiably be used to derive CAGs, or are secondary consequences of primary chemical events, in which case they should not be used to form CAGs.
If applicable to the liver, we would propose that a similar approach should equally be applied to the other organs for the purposes of cumulative risk assessment.
The EFSA Approach to CAGs for the Liver
For the purposes of the exercise, the contractor the International Centre for Pesticides and Health Risk Prevention (AOSACCO/ICPS Italy) responsible for collating the liver part of the EFSA discussion document collected toxicity data for all pesticidal active substances approved between January 1, 2012, and May 31, 2013, as well as those pending approval, in addition to those that were not approved for use within the EU, but were detected as residues, and retrieved by EFSA from the Annual Report of the Rapid Alert System for Food and Feed 11 2011 and the 2010 Annual Report on Pesticide Residues in Food. 6 This constituted a total of 134 pesticides. However, for 5 substances of the 134 substances to be screened, neither draft assessment reports nor other international body reports were available, therefore only 129 could be evaluated. For analyses of the liver, biliary system, and gall bladder, a total of 22 toxicologically significant effects were used to derive 15 CAGs (Figure 1 adapted from ref 10). Hence chemicals would be grouped together into a CAG if they had one or more of these end points in common, indicating that they had the potential to exacerbate the effects of any single chemical on its own. Based on these criteria, a large number of the chemicals were included into the CAGs and classified as showing hepatic hypertrophy (99 substances), cholestasis (58 substances), fatty changes (40 substances), hepatocellular neoplasms (35 substances), and hepatocyte cell degeneration (33 substances).
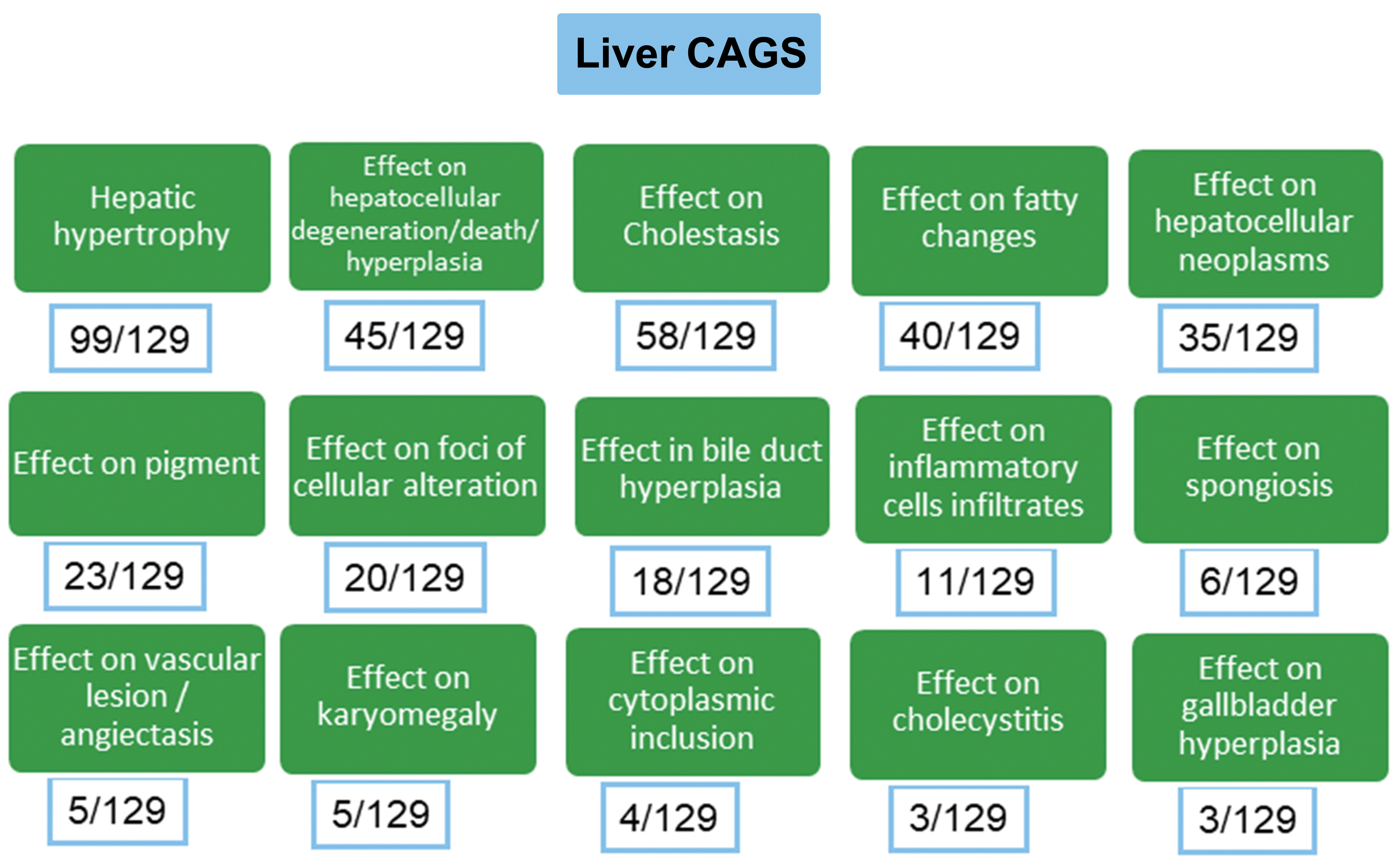
The EFSA proposal for cumulative assessment groups (CAG) from the external scientific opinion of 2016; 129 chemicals were evaluated and the figures in boxes underneath each CAG represent the number of chemicals grouped under each CAG. EFSA indicates European Food Safety Authority.
In the conduct of the EFSA evaluation, it was not possible to arrange CAGs on the basis of common MOA because “For most substances, that exert effects on the liver, no data on the MOA were available since it was rarely investigated and the literature was lacking of consistent information. 10(p45)”
Alternative Approaches to CAGs, Based on Knowledge of Pathophysiology in the Liver
Although cumulative grouping based upon common pathological end points, without a knowledge of the MOA involved, has higher uncertainties compared to a knowledge of a common MOA, 12 -14 it is still considered a sufficiently pragmatic, and practical, approach for combined toxicity assessments. Clearly, 2 different chemicals that activate the same receptor will, in combination, act together to enhance the activation of the given receptor, if toxicokinetic properties don’t lead to changes in internal exposures and/or toxicodynamic effects act to alter interaction at the active site. 12 –14 As toxicological MOA data for pesticides are generally insufficient for cumulative risk assessment purposes for the vast majority of currently registered pesticides, groupings based upon common modes of action, at the current time, are not yet a feasible option.
However, if common phenomenological toxicological effects in in vivo studies are used for grouping, then the knowledge of the pathophysiological events and subsequent regenerative repair processes in the target organ (in this article, the liver) should be taken into consideration to derive appropriate scientifically based, and robust, criteria for grouping insofar as this is possible with the current understanding of pathological processes.
The approach proposed in this article discriminates between “primary” end points (direct consequence of chemical interaction with a biological target) and “secondary” end points (which are a consequence of, and are arising from, a previous pathological change). Primary end points are essentially those that can be produced via molecular initiating events (MIEs) in the jargon of the adverse outcome pathways. 15 In contrast, secondary end points, while they may be “key events” (KE) in the production of the certain hepatic pathologies, require to be preceded by specific MIEs in order for the secondary events to occur. The rationale for arranging the various hepatic pathologies into primary or secondary events is summarized in Table 1.
Summary of the Reasoning Behind Allocating Any Particular Pathology Into a “Primary” Versus a “Secondary” Effect.

Abbreviations: CAG, cumulative assessment group; MIE, molecular initiating event.
The primary pathological effects are assumed to be induced initially after systemic exposure of laboratory animals to the substances under investigation and that they drive the organ toxicity. In contrast, effects that only arise as secondary changes to various precursor pathologies do not reflect key organ toxicities and will not interact following exposure to 2 chemicals, showing the same secondary pathology, to potentiate the combined liver toxicity.
In Figure 2, the phenomenological pathological effects used to identify common specific end points in the EFSA external report 2016 10 have been reorganized based upon whether they are primary or secondary effects taking into account the sequence of the key pathological events.
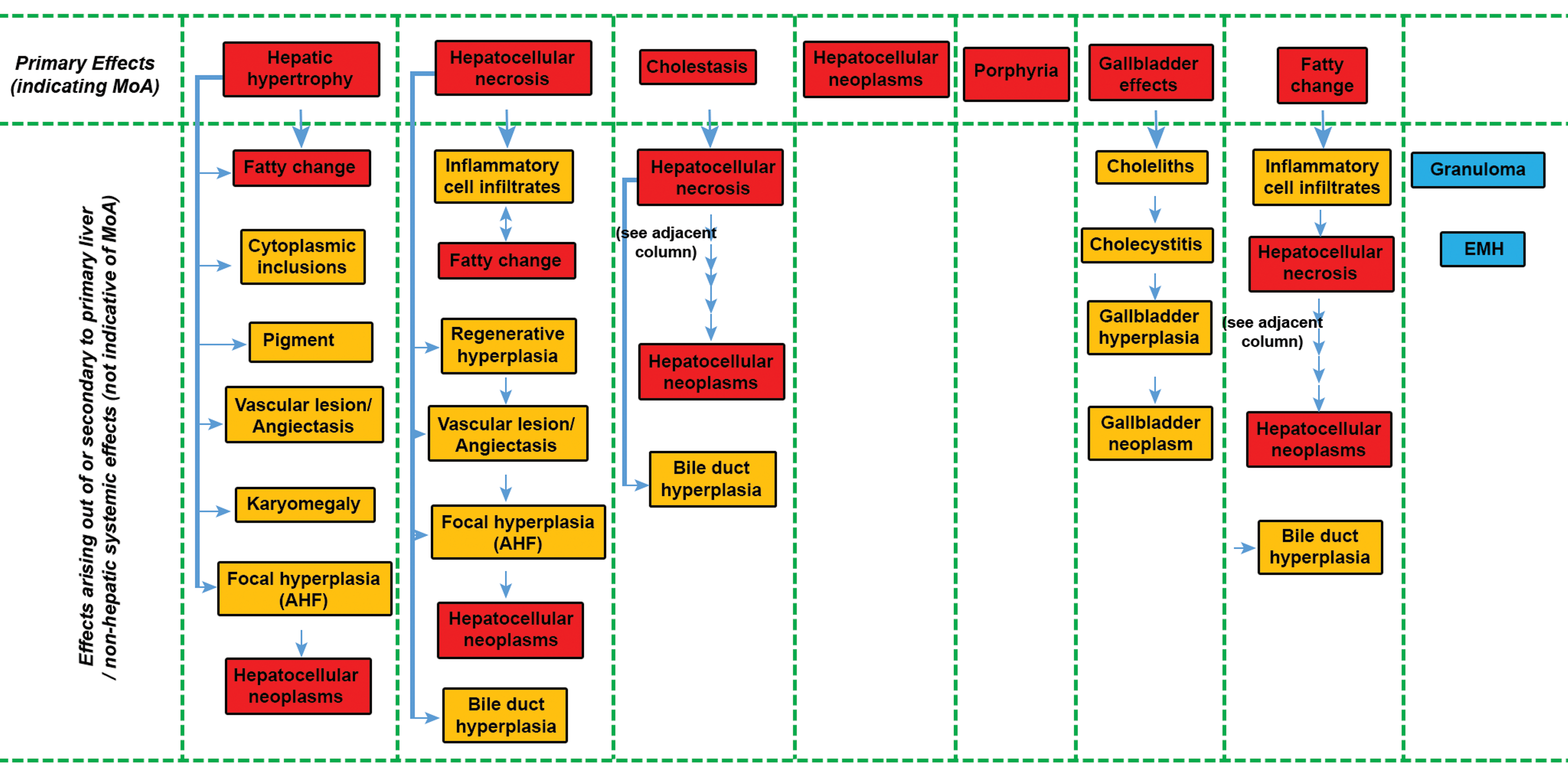
This figure has grouped the various histopathological effects used by the European Food Safety Authority (EFSA) exercise under whether they fall into primary or secondary effects based upon their known etiology. Primary effects are in red boxes while secondary effects are in yellow boxes. Blue boxes list secondary lesions that are unrelated to a principle effect on the liver and generally reflect a systemic toxicity or one affecting a different organ from the liver. The columns represent “secondary” events that occur following a “primary” toxicity or MIE (top row). EFSA indicates European Food Safety Authority; MIE, molecular initiating event; MoA, mode of action.
Primary effects are those end points to be considered for CAGs as they initiate the toxic effects, will occur as direct effects of exposure to hepatotoxic chemicals, and will be expected to be increased in degree (severity) upon exposure to 2 or more chemicals inducing the same primary effect.
An exception to this rule is the inclusion of “hepatocellular neoplasm” as a primary end point for the purposes of deriving the CAGs. These can arise biologically from diverse chemical groups as a consequence of a number of, often disparate, preceding KE but it is considered that chemicals inducing neoplasia in the same organ cannot be separated irrespective of the MOA. In this respect, we consider it reasonable to use “hepatocellular neoplasms” as one group in building CAGs for the liver in agreement with the original EFSA proposal. 10
Furthermore, in many cases, MOA data will be available, or subsequently become available, for substances inducing liver tumors in order to assess the human relevance. These data will permit the inclusion or exclusion of substances from a liver CAG dependent upon the relevant MOA.
Appropriate Definition of the Pathological Terms Used in Toxicity Reports in Deriving CAGs
In order to assign a pathological term to a particular hepatic change, it is essential that each term has a precise, unambiguous, pathological definition as a critical starting point for any objective review of study reports for the purposes of the current cumulative assessment exercise. Clearly, these terms also need to be correctly used by the study pathologist in describing what is present in the tissues. Therefore, the use of standardized pathological nomenclature has been addressed by the societies of toxicologic pathology in their International Harmonization of Nomenclature and Diagnostic Criteria (INHAND) initiative, 16 which should help to ensure that the correct term is applied to the lesions at least for the present and future studies. Assessment of pre-INHAND historical studies could potentially provide some difficulties where current, nonstandard, pathological terms may have been used, but review by a panel of experienced pathologist working alongside the respective regulatory agencies should be able to resolve any nonstandard terminology applied at the time of the study.
In addition, other, nonhistopathological, end points have been used in the original exercise by EFSA to conclude particular CAG terms. An example of this is the use of increases in clinical enzymes, such as alanine aminotransferase (ALT) and alkaline phosphatase (ALP), to interpret a finding of hepatic necrosis. Although it is true that increases in these enzymes can, under certain circumstances, indicate hepatic necrosis or cholestasis, the degree of increase, and the combination of enzyme increases, is important in distinguishing physiological adaptation from hepatic degeneration/necrosis. 17 Increases in single enzymes alone are never pathognomonic of hepatic pathology but are more indicative of the normal physiological changes that occur in the liver in response to increased metabolic demand. 18
Defining the Pathological Terms Used to Derive the Primary CAGs
To achieve consistency in the use of the pathological terminology for defining primary events, on which CAGs should be classified, we have provided the following definitions. We have deliberately decided not to offer definitions for the secondary end points since these are not considered relevant to this document.
Hepatocellular Necrosis
Necrosis affecting the hepatocytes within the liver can be observed in a number of forms including that showing a zonal distribution, affecting large numbers of hepatocytes (Figure 3A), in a focal form, where a small group of hepatocytes will be affected in single or multiple areas of a liver lobe without obvious zonal distribution (Figure 3B), and necrosis affecting single cells randomly distributed in the liver (Figure 3C). The type and extent of necrosis seen are dependent upon the age of the animal at the time of exposure, the dose of chemical administered, the time when the observation is made following dosing, the animal species being studied, the gender of the species being dosed, and also on the MOA of the chemical in inducing the necrotic effect. 19 –22
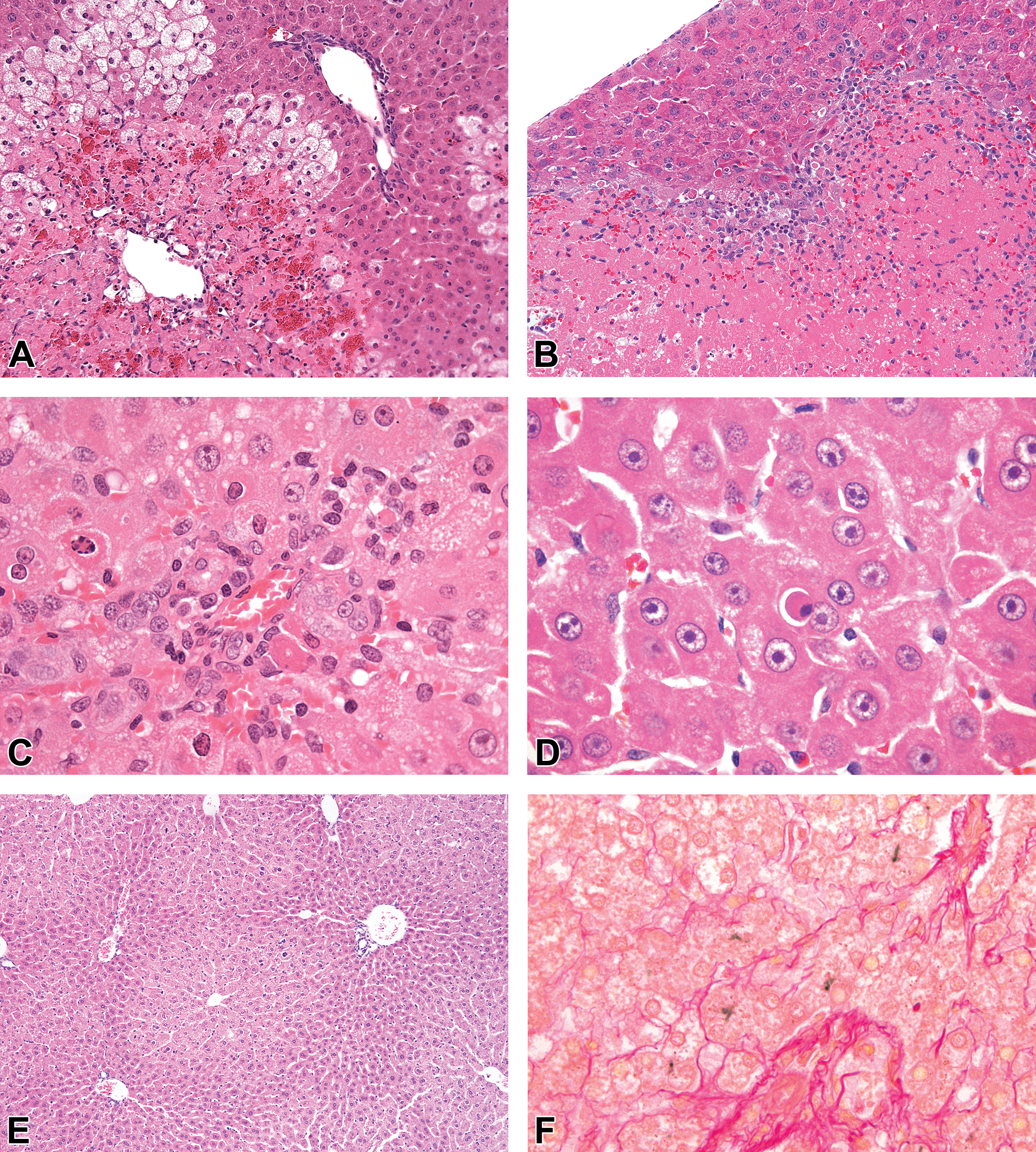
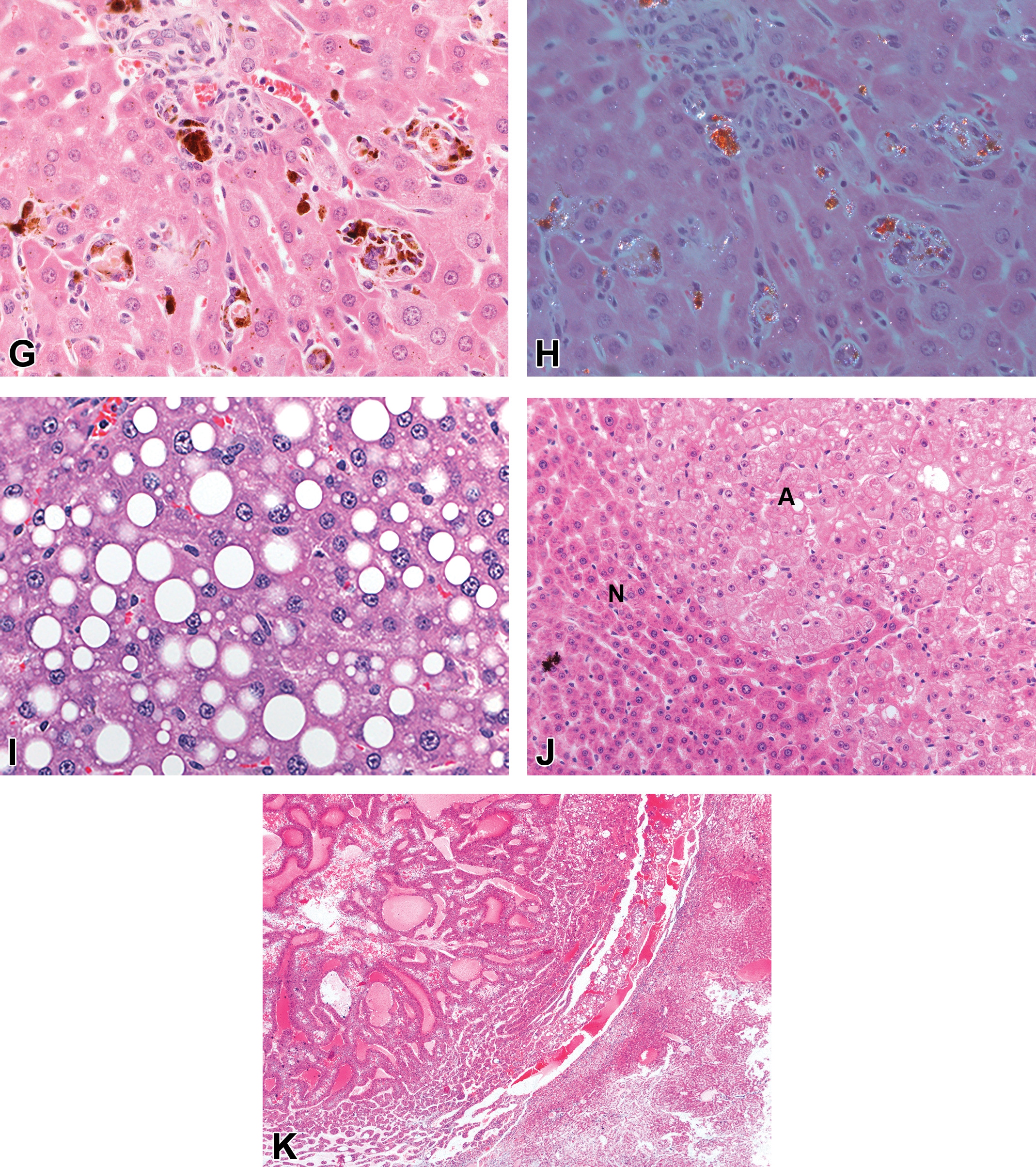
A, An example of zonal necrosis in the liver. The present example shows centrilobular necrosis (left side of photograph) induced in the rat by carbon tetrachloride and typically will affect the majority of liver lobules. Hematoxylin and eosin (H&E) original objective ×10. B, An example of focal necrosis in rat liver where groups of hepatocytes have undergone necrosis. This is commonly a spontaneous lesion which can be multifocal but which fails to show any lobular predilection. H&E original objective ×20. C, Single cell necrosis in rat liver where the affected cells are individually targeted, are randomly distributed throughout the lobule, and again generally fail to show any specific lobular distribution. Unlike apoptotic cells, these cells maintain an irregular outline with palely eosinophilic cytoplasm and the nuclei undergo karyorrhexis or karyolysis rather than showing condensation. H&E; original objective ×63. D, An apoptotic cell in the liver of a rat showing increased eosinophilic staining of the cytoplasm, condensation of the nuclear chromatin, rounding off of the cell margin, and the typical presence of a clear halo around the apoptotic cell. H&E original objective ×63. E, Centrilobular hypertrophy in rat liver showing a zone of enlarged hepatocytes, in this case around the central vein, with the cells showing homogeneous eosinophilic cytoplasm. H&E original objective ×5. F, Bile plugs (green) present in the canaliculi in a case of cholestasis in the dog liver. Fouchet stain for bile; original objective ×40. G, Porphyrin deposits (brown) in hepatocytes and Kupffer cells in the periportal region of the lobule from a mouse given an agrichemical. H&E; original objective ×40. H, The same field as in (G) with the exception of the section being viewed under polarized light when the porphyrin crystals become birefringent. Birefringence per se is not a specific feature of porphyrin deposits but combined with red fluorescence under UV light, is pathognomonic of porphyrin. H&E under polarised light; original objective ×40. I, Fat vacuolation in the liver of a rat. The vacuoles are described as macrovesicular and show a clear, regular, round profile in the hepatocytes. H&E original objective ×40. J, Microscopic appearance of an hepatocellular adenoma in a mouse liver where the neoplastic hepatocytes are slightly compressing the normal liver cells which show a more eosinophilic staining (on the left hand side of the photograph) compared to the pale staining neoplastic cells on the right hand side of the photograph. H&E original objective ×10. K, A photomicrograph of an hepatocellular carcinoma from a rat given a genotoxic carcinogen. The carcinoma shows the development of glandular structures alternating with more solid areas of neoplastic cells. H&E original objective ×5.
Zonal necrosis (periportal and centrilobular) most commonly occurs with chemical exposure following metabolic activation to toxic metabolites. As necrosis develops, the cytoplasm of the affected hepatocytes becomes increasingly homogeneous and pale in the extent of their eosin staining, and the nuclei gradually disappear (karyolysis). The endothelial cells lining the hepatic sinusoids can also become necrotic as a secondary, and more severe, effect of hepatic necrosis, and when this occurs the sinusoidal space can become dilated (angiectasis) and is frequently associated with an accumulation of red blood cells in the affected sinusoids (congestion). Zonal necrosis may have a minimal associated inflammatory reaction and can be accompanied by hydropic degeneration, fatty change, and “ballooning” in adjacent, less severely affected, hepatocytes.
Zonal necrosis in rodent liver is always associated acutely with increased serum hepatic enzymes, such as ALT and aspartate aminotransferase (AST), and glutamate dehydrogenase. There may also be elevations in serum ALP and γ-glutamyltranspeptidase (GGT), although these are more normally associated with periportal hepatocellular damage and bile duct effects, and frequently occur later in time, accompanying regenerative processes that follow the initial hepatocellular necrosis. It is unknown for any single enzyme to be increased following necrosis, and the norm is for a spectrum of enzymes to be elevated, with ALT and AST being the most diagnostic of the commonly used biomarkers. 17 It is recommended that increases in the plasma concentration of single enzymes never be used to assign a chemical into a necrosis CAG unless accompanied by histopathological evidence. In contrast, where increases in the expected spectrum of liver-derived enzymes occur, even in the absence of histopathological evidence of necrosis, this provides reasonable evidence that hepatic necrosis will develop as a consequence and should result in the chemical in question being assigned to a necrosis CAG.
Although there has been considerable controversy regarding the differentiation of apoptosis from so-called “single cell necrosis,” a recent publication by Elmore et al, sponsored through the INHAND initiative, has added clarity to a topic that has in the past led to some confusion. 23 The features of apoptotic hepatocytes (Figure 3D) are a “small pyknotic nucleus (due to nuclear condensation) and a hyper-eosinophilic cytoplasm within a rounded cellular profile (due to cytoplasmic condensation).”(p5)23 Currently, the recommended diagnostic term for individual cells showing features of necrosis, where some uncertainty arises as to the exact origin of the cells, is “apoptosis/single cell necrosis.” This is based upon the fact that apoptotic cells rapidly lose their characteristic nuclear and cytoplasmic condensation to become what is more recognizable as necrotic cells. 23 Because the hematoxylin and eosin section is a snapshot of the physiology of the organ, it is no longer felt appropriate to use the term single cell necrosis simply because the underlying molecular basis, defining the morphology, remains unknown. A full and complete description of the differentiation of apoptotic cells from apoptotic/single cell necrosis is to be found in the publication by Elmore et al 23 and will not be further elaborated on in this publication but the reader is recommended to read the publication for a fuller description. Although apoptotic hepatocytes are seen without inflammatory cell accompaniment, the apoptotic/single cell necrosis is frequently accompanied by inflammatory cells of various forms. Apoptosis/single cell necrosis can be induced in the liver with low doses of chemicals which at higher doses cause zonal necrosis, with cytotoxic anticancer drugs, and can occur as a consequence of viral or bacterial infections. 23
The appearance of apoptosis/single cell necrosis is sometimes accompanied by elevations in plasma enzymes, such as ALT, AST, ALP, or GGT but where the degree of effect is small, or where a pure apoptotic cell response is observed, there will be no elevation in plasma enzymes indicative of abnormal liver physiology.
Hepatic Hypertrophy
This term is commonly defined using evidence from a number of different sources. An increase in liver weight is often referred to as “liver hypertrophy” but “hepatocellular hypertrophy,” commonly present in the centrilobular regions of the liver, is the pathological term which most often accompanies mild to moderate increases in liver weight and hepatocellular size (Figure 3E). Hepatocellular hypertrophy may occur in the absence of any other changes in the liver, but when the degree of liver weight exceeds a certain percentage of control liver weight inflammatory changes, and elevations in circulating, liver derived, enzymes, such as ALT and ALP, will accompany the increased liver weight, hepatic hypertrophy is considered adverse. 17 The presence of hepatocellular hypertrophy is frequently associated with the induction of metabolic enzymes such as those associated with the cytochrome P-450 superfamily, an increase in endoplasmic reticulum, an increase in those enzymes associated with peroxisomes, or an increase in the number of mitochondria. 18
The enlarged hepatocytes frequently show staining differences from the control livers and the cytoplasm may be more homogeneous and eosinophilic (endoplasmic reticulum proliferation) or granular and eosinophilic (peroxisome and mitochondrial proliferation). The changes usually show a zonal pattern of distribution (centrilobular, periportal, midzonal) but for some chemically induced changes the whole of all lobules may show the changes. This pattern of change will be repeated throughout the liver lobules and liver lobes. Depending upon the degree of cellular hypertrophy, the enlarged hepatocytes may compress the associated sinusoids, and concurrent focal or single cell necrosis can occur at high doses of chemicals, where only hypertrophy is observed at lower doses.
Subsequent to the induction of hypertrophy, continued exposures can result in secondary changes including increased pigmentation, bile duct hyperplasia, and fatty change that can develop with increasing time and/or increasing dose. These changes are typically secondary to other events and can be produced following many different primary hepatic changes in addition to hypertrophy.
The induction of pure adaptive hepatocellular hypertrophy, in the absence of accompanying hepatic degenerative changes, is not normally associated with increased release of hepatic enzymes whereas hypertrophy exceeding adaptation can be accompanied by increased serum enzymes and other secondary effects such as hepatocellular degeneration and inflammatory cell infiltration.
Of all of the chemical-induced primary effects on the liver, often the most information in terms of mechanism of action are for those chemicals that induce hepatic hypertrophy. These chemicals appear to work through activation of one or other of the nuclear hormone receptors that include the CAR/PXR (constitutive androstane receptor/pregnane X receptor, PPAR (peroxisome proliferator activated receptor), and AhR (aryl hydrocarbon receptor) nuclear receptors. 18,24 –26
Chemicals that induce adverse hepatocellular hypertrophy should all be placed into a common CAG, irrespective of the MOA involved in inducing that hypertrophy.
Cholestasis (Biliary)
Cholestasis is an important primary disease endpoint that is one of the major causes of idiosyncratic adverse drug reactions in man, and one which is poorly predicted from preclinical laboratory animal studies 27 (Figure 3F). Cholestasis is a rare hepatic response to xenobiotics in rodents but can be seen more commonly in the liver of dogs and monkeys. 28 –30 Cholestasis itself is not solely a histopathological diagnosis but a clinical one interpreted from the integration of multiple pathological indicators, but where confirmed, is clearly a primary pathological event. The interpretation of cholestasis is based upon a weight of evidence evaluation involving elevations in serum bilirubin, elevations in serum enzymes, and histopathological evidence of bile pigment accumulation. Although bile duct hyperplasia can be seen in cases of chronic cholestasis, of itself, it is not indicative of cholestasis and should not be used to conclude that cholestasis is occurring. Bile duct hyperplasia is a common secondary pathological response to a variety of different liver toxicities, and grouping chemicals together on the basis of bile duct hyperplasia would be incorrect as there would be little chance of 2 chemicals, having different MOAs for producing liver toxicity, that eventually result in bile duct hyperplasia, interacting in a cumulative way.
The diagnostic features of bile accumulation (cholestasis) are the appearance of elongated, pale, green-brown pigmented plugs generally within the bile canaliculi, and/or as finely granular pigment within the cytoplasm of hepatocytes, an appearance that is much more commonly seen in human and dog liver than in rodents. In severe cases of cholestasis, bile pigment can also be seen within the cytoplasm of Kupffer cells. The histological presence of bile plugs within the liver is a clear indicator of cholestasis but the identification of a pigment as bile in liver sections needs to be confirmed using a stain such as Fouchet (Hall’s bilirubin stain) since many other pigments can be observed more commonly in the liver. 31 The presence of bile plugs within the liver indicates an inhibition of bile flow through the canaliculi and biliary conducting system. In human medicine, the production of cholestasis has been divided into “extrahepatic cholestasis” where physical blockage of the bile flow in the bile ducts occurs, and “intrahepatic cholestasis” where inhibition of bile secretion occurs from within the hepatocytes. Extrahepatic cholestasis is generally seen as a secondary response to damage to the bile ducts, while intrahepatic cholestasis is a primary effect of drugs/chemicals on the bile transporter proteins that are present on the basolateral and apical membranes of the hepatocytes.
Hepatic damage through a cholestatic MOA is an important cause of drug-induced liver injury in humans and it is broadly acknowledged that laboratory animal studies are generally poor predictors of cholestasis in man. 32 There are several chemicals that have been shown to inhibit the bile transporters on the hepatocyte canalicular membrane in vitro and induce idiosyncratic liver disease in man but the same chemicals have rarely, if ever, been shown to induce hepatic bile accumulation in rodent studies indicating fundamental species differences in the way in which these biological systems work between humans and rodents. The frequency of detecting this effect in the dog is marginally better than it is in the rodent but the overall conclusions from an analysis of the data are that laboratory animal studies, for several reasons, are insensitive at detecting hepatotoxic chemicals acting through a cholestatic MOA in man.
Pathophysiologically, cholestasis involves either an inhibition of bile flow out of the liver, through a defect in the biliary conducting system, or by inhibition of bile secretion into the biliary system through inhibition of ATP-dependent bile transporters localized in the biliary canalicular membrane of hepatocytes. The definitive indicator of cholestasis is an elevation of serum bilirubin and/or bile acids although elevations in several other serum enzymes, such as ALP, GGT, and 5′ nucleotidase, occur consequential to the liver damage that results from the accumulation of bile acids in the liver. Hence, while elevated serum activities of ALP and GGT can be seen in cholestasis, and other biliary diseases, they are also increased in chronic hepatocellular injury in general, such as that induced by alcoholic cirrhosis, and are also elevated in a variety of nonhepatic disorders 33 that are unrelated to a cholestatic MOA. Hence, a conclusion of a chemical-induced cholestasis, and a consequential inclusion in a cholestatic CAG, needs to show increases in serum bilirubin supported by elevations in other biomarkers, the most important of which is histopathological evidence of bile accumulation.
Pigmentation due to Porphyrin
The diagnostic features of porphyrin are accumulation of a dense dark brown to red-brown pigment present within the hepatocytes, bile ductules, and bile canaliculi (Figure 3G), which appears bright red and birefringent when viewed in polarized light (Figure 3H). When fresh frozen sections are viewed in ultraviolet light, the porphyrin pigment shows a brilliant red fluorescence which fades on continued exposure. Because porphyrin is mostly secreted in the bile, it tends to concentrate in the periportal regions of the liver lobule and because it is a toxic agent 34 it is frequently accompanied by hepatocyte necrosis and an acute inflammatory cell reaction. Unlike other forms of pigmentation, which generally occur secondary to a predisposing hepatic damage, porphyrin pigmentation is most often caused by inhibition of one or other of the enzymes involved in the metabolism of heme, normally by a metabolite of the chemical inducing the effect, and as such is a primary toxicological event and not a sequel to other pathological changes in the liver. While liver enzymes such as ALT, ALP, and AST may well be increased as a secondary consequence of the hepatic damage resulting from the porphyrin accumulation, inclusion of a chemical into a porphyrin CAG needs to show the presence of porphyrin deposits in the liver by histopathology.
Fatty Change
Fatty change, or fat accumulation (steatosis, lipidosis), is a primary event that develops following perturbation of hepatic lipid metabolism/catabolism and/or disposition. It can be induced directly by chemicals that inhibit protein synthesis, such as carbon tetrachloride, since lipoproteins are essential for the transport of lipids but can also result physiologically as a consequence of feeding high-fat diets. The critical role of mitochondrial dysfunction in the development of both human liver diseases and chemically induced steatosis in laboratory animals has also been increasingly recognized as a driver in the initial development of hepatic fatty change. 32,35,36 Hepatic phospholipid accumulation is an important variant that can affect hepatocytes, cholangiocytes, and Kupffer cells within the liver 37 following the inhibition of breakdown of phospholipids by chemicals that either bind to the phospholipids, making it more difficult to digest, or by direct inhibition of the enzymes responsible for breaking down phospholipids, such as phospholipase A. 38 –41
Fatty change has been described using various synonyms including lipidosis, vacuolation, macrovesicular and/or microvesicular steatosis, and phospholipidosis. The lipid droplets within the hepatocytes may be in the form of single, large, round, clear vacuoles within each cell, frequently with the nucleus and remaining cytoplasm displaced to the periphery. This appearance has been termed “macrovesicular” fatty change or steatosis (Figure 3I). A second form of fatty change is seen in hepatocytes that may be partially or completely filled with large numbers of small lipid vacuoles, and the cytoplasm of affected hepatocytes may have a ‘‘foamy’’ appearance. This effect has been termed “microvesicular” fatty change and the small vacuoles do not normally displace the nucleus to the periphery in contrast to macrovesicular fatty change. Although macrovesicular fatty change can be seen as an adaptive physiological change in response to high-fat diets, microvesicular fatty change is almost always considered pathological and adverse 35 and is frequently accompanied by necrosis and other degenerative hepatic changes such as cell ballooning and hydropic degeneration. 41
To distinguish vacuolation due to fat, from vacuolation due to glycogen or water, the use of special stains, such as oil red O or Sudan Black, is recommended on formalin-fixed, or fresh frozen, sections as wax-embedded material is not suitable due to solvent extraction of any contained lipid. Vacuolation due to the accumulation of phospholipids can be differentiated from neutral lipid by staining with Pearse’s method using Luxol fast blue, or with antibodies against LAMP-2, a lysosome-associated protein, or adipophilin, a protein that forms the membrane around nonlysosomal lipid droplets. 40 The use of electron microscopy has also been advocated as a specific and sensitive way of differentiating and detecting the accumulation of phospholipid within cells as it has a distinctive ultrastructural appearance. 42 –46
Physiological fat accumulation, or low level accumulation of fat, is not normally associated with increased plasma enzymes or increased liver weight, but in so-called “toxic lipidosis,” where hepatic cell degeneration and necrosis occur alongside the lipid accumulation, increased liver enzymes, such as ALT, AST, and ALP, will be seen but for inclusion into a fatty liver CAG, a histopathological description of fatty change, fat vacuolation, or other synonyms, such as steatosis, will need to be present.
Neoplasia
There are a number of both benign and malignant neoplasms that arise within the liver, including those derived from the hepatocytes and bile duct epithelium, and henceforth described as hepatocellular or biliary neoplasms (Figures 3J-K). Hepatic neoplasms not arising from these 2 tissues, such as vascular or other mesenchymal neoplasms, are not considered in this analysis. Hepatocellular neoplasms are common spontaneously and can be induced by certain chemicals via both genotoxic and nongenotoxic modes of action. Nongenotoxic rodent liver carcinogens act through inducing changes in hepatic function that generally result in a sustained elevation in hepatocyte proliferation and are a consequence of chemicals that cause primary events through either a hepatotoxic (necrotic) MOA or through the activation of nuclear receptors, induction of liver growth, and an adaptive MOA. No serum clinical chemistry markers are used consistently in the diagnosis of hepatic neoplasms and they are almost always diagnosed at necropsy. Some modes of action, leading to rodent liver tumors, are known to be nonhuman relevant (e.g. CAR-mediated, PPAR 35,36 ) and those data should be used to only group those AIs together, where human relevance of the liver neoplasms seen in rodent studies cannot be excluded.
The use of human relevant hepatic neoplasia, as a relevant primary end point under which chemicals can be grouped, irrespective of their initial MoA, implies that 2 chemicals, which independently induce rodent hepatic neoplasia, should clearly be included in the same CAG.
Primary Gall Bladder and Biliary Pathologies
There is no direct relationship between toxicity seen within the liver and that occurring in the gallbladder, and modes of action affecting gall bladder toxicity are not generally applicable to hepatic toxicity and vice versa. 47 It is recommended that the 2 organs be handled as separate in similarity between the liver and the pancreas, with regard to their aggregate exposure to chemically induced toxicity. Primary effects on the biliary system that result in blockage of bile outflow, can secondarily affect the liver most commonly as inflammation which can be seen in the periportal regions of the lobules. The chemical induction of these lesions is however fairly rare and most of the biliary changes seen in the liver are unrelated to primary changes in the biliary system. 47,48
The rationale for arranging the various hepatic pathologies into primary or secondary events is summarized in Table 2.
Summary of the Reasoning Behind Allocating Any Particular Gall Bladder and Bile Duct Pathology Into a “Primary” Versus a “Secondary” Effect.
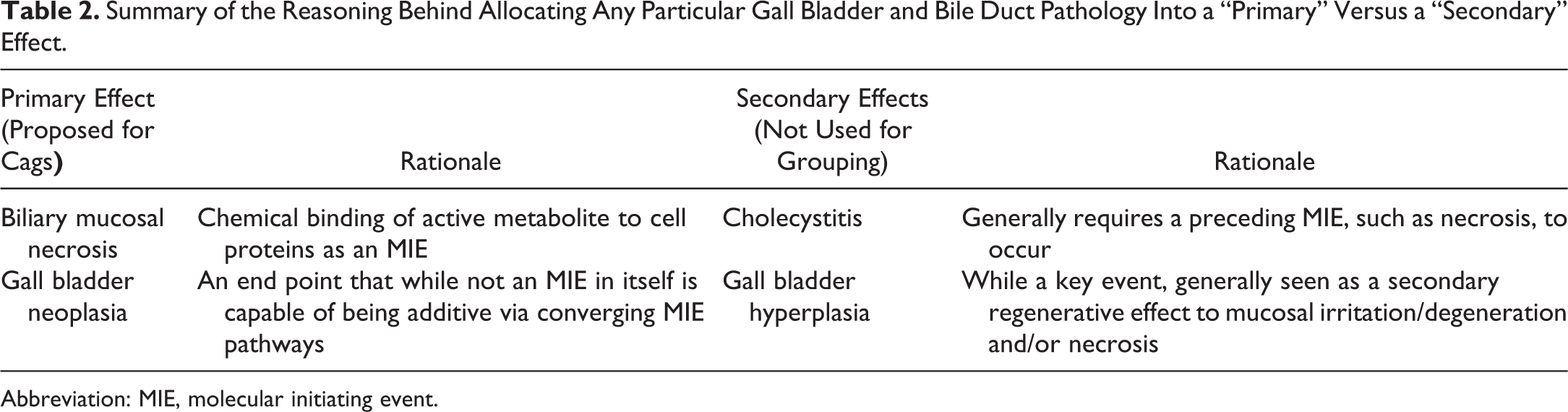
Abbreviation: MIE, molecular initiating event.
In contrast to the liver, the gall bladder and bile ducts have a much more limited number of primary pathologies rather than a secondary effect to a precursor lesion. Many drug-induced human bile duct pathologies have an immune-mediated etiology and are subsequently secondary to the haptenization of the drug with endogenous protein. 49 This type of reaction is not considered within the category of primary CAGs for either of these tissues.
Biliary Mucosal Necrosis
There are a limited number of chemicals that can induce necrosis of the mucosa of the biliary system as a primary effect, which is often accompanied by hepatocellular degeneration and inflammation, but which occurs prior to the hepatic damage. 47,50,51 The industrial chemical intermediate, dibutyltin dichloride, has been used as a model for the induction of acute pancreatitis in laboratory rodents, but which also induces a primary biliary mucosal necrosis of the common bile duct and larger interlobular ducts of the liver. 52 –55 The necrosis of the epithelial cells is followed by a series of secondary pathological changes such as submucosal edema, extrahepatic cholestasis, and inflammation both within the affected tissues and throughout the periportal regions of the liver lobules. Serum bilirubin, ALP, GGT, and ALT are frequently elevated due to the secondary effects on the liver 56 but are not pathognomonic of primary biliary toxicity.
Gall Bladder Neoplasia
Gall bladder neoplasias are clearly different from those arising within the liver itself. The causative feature, as with hyperplasia and inflammation, is down to the organ’s function in storing bile for prolonged periods of time with any toxins present being in prolonged contact with the biliary mucosa in inducing its toxicity through chemical concentration and irritancy. This in contrast to the liver where metabolic activation is the most common route for toxicity.
Gall bladder neoplasms have limited variations in their histology 54,57,58 and can present as both benign and malignant forms, most commonly projecting out into the bladder lumen but occasionally invading through the basement membrane of the bladder mucosa. Clearly chemicals that include gall bladder neoplasia should be combined into a common CAG.
Discussion
Historically, the evaluation of the safety of pesticides has depended upon single chemical, and single exposure scenarios, such as via the inhalation route, oral, or via the dermal exposure route. Much of this effort was driven by the fact that the data, upon which the risk assessment was based, were derived from single chemical exposures in an experimental laboratory setting using classical toxicological principles. Although this approach has the merit of being accurate and robust on a single compound basis, it does not reflect real-life scenario where individuals are often exposed to multiple chemicals via multiple routes of exposure. In addition, it has been shown that exposures to multiple chemicals have the potential to result in additive effects, depending on their toxicological profile.
Approaches that utilize mechanism/MOA data are currently the gold standard for deciding if chemicals should be grouped together for the purposes of risk assessment. 58,59 However, mechanistic data on molecular targets and an in depth understanding of the MOA in inducing toxicity are lacking for the vast majority of registered commodity chemicals or natural substances.
In the EFSA Scientific reports, 6-7,10,13 it is proposed to group chemicals on the basis of the same phenomenological and pathological term to define effects observed in the toxicity studies. However, the proposed liver grouping approach 10 is not reflective of common biological end points but merely reflect common pathophysiological end points resulting from disparate initial lesions, indicating the limited number of ways in which a tissue or organ, can repair itself following damage. Furthermore, some of the proposed groups are unspecific and of very limited relevance. As illustrated in Figure. 1, displaying the various substances in the CAGs proposed according to EFSA, it is evident that substances are redundantly grouped into several CAGs according to criteria and end points that have little possibility for interaction by chemicals inducing the respective end point.
By using the concept of primary versus secondary pathological end points, the respective CAGs become more scientifically justified and results in a reduction in the number of groups from the original 15 CAGs, with 22 phenomenological effects, to 6 CAGs for the liver and 3 for the gallbladder (Figure 4). This new grouping does not negatively impact the safety assessment, because all of the substances presenting at least one secondary effect are included in one of newly proposed primary effect group. Therefore, the primary/lead effect drives the allocation to a specific CAG and the performance of a risk assessment based upon common primary effects.
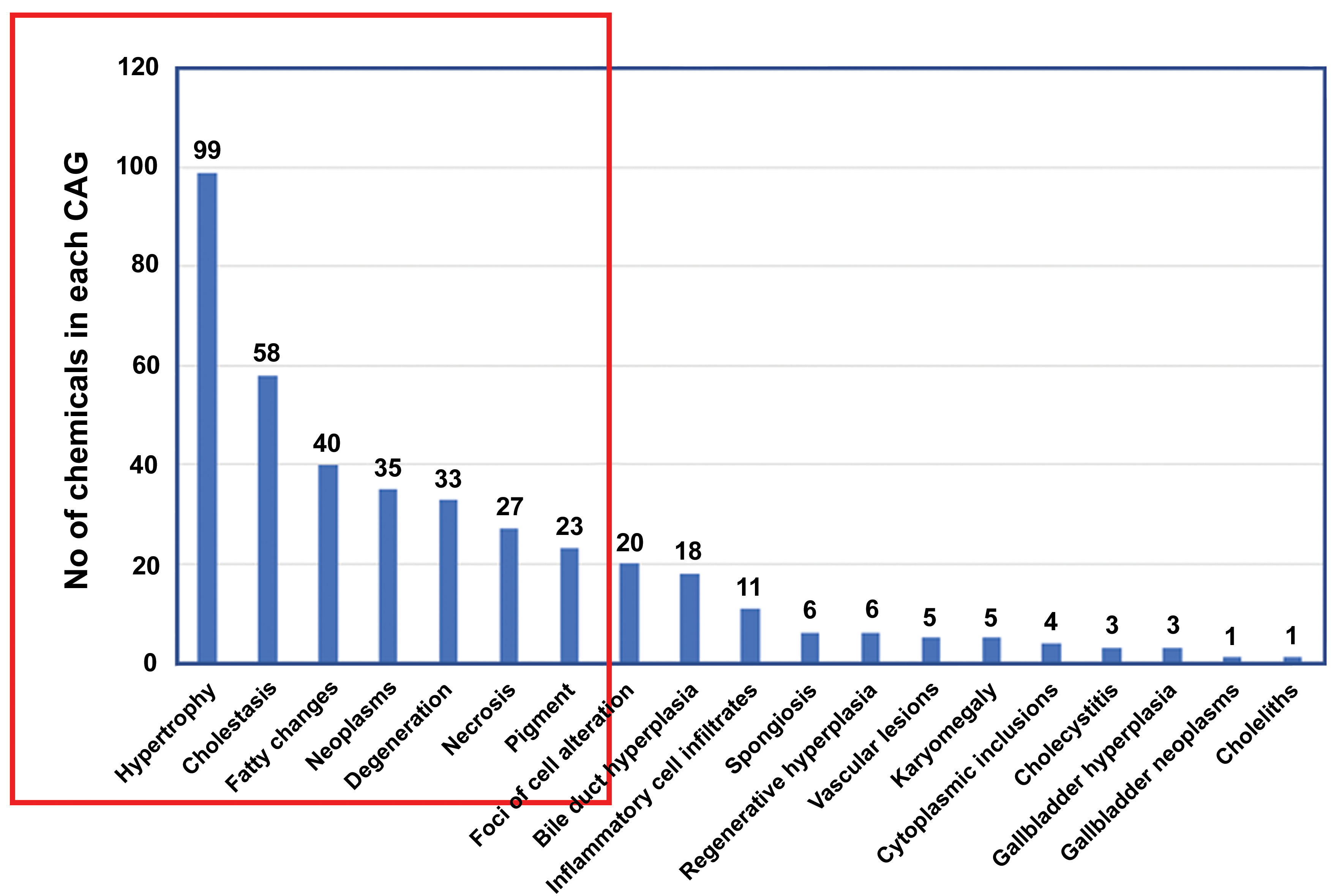
The number of active substances for each specific effect from CAG numbers 2A to 2Q (blue vertical bars) from EFSA’s original proposal (10, page 39). The red box illustrates the modified CAGs resulting from the analysis carried out in this manuscript. The 6 CAGs recommended by the current approach (inside the red box) represent the majority of the chemicals included in EFSA’s original proposal. The inclusion of all “pigment” in the EFSA proposal has been replaced by porphyrin in the current recommendation as other pigments are secondary events. Additionally, the 2 EFSA categories, “hepatocyte cell degeneration” and “hepatocellular necrosis” have been combined in the current recommendation as simply “necrosis.” Further analysis of each substance allocation to CAG level 2 showed that those presenting secondary effects are also included in at least one of the 6 currently recommended CAGs. Redrawn and adapted from ref 10. CAG indicates cumulative assessment group; EFSA, European Food Safety Authority. The figure was an adaptation from the original taken from the ECHA document which includes both categories of “necrosis” and “degeneration”. For pragmatic purposes, mainly because of the use of the non-specific term “degeneration” by pathologists, we have recommended, in this manuscript, that the formerly two categories of “necrosis” and “degeneration” be combined into a single one, called “necrosis”. Thus although there are seven categories within the red box on the figure, there are actually only 6 categories that we recommend using.
While the effect type “hepatocellular neoplasms” is considered to be one CAG, although it is not a primary liver pathological end point as used in the definition, given its relevance and the fact that substances that independently induce hepatocellular neoplasms as the critical effect might interact to increase their risk. It is furthermore important to note that only “human relevant” liver tumors should be considered for grouping, since in general only adverse and human-relevant effects are to be considered for human cumulative risk assessments. 6–7 Considering the fact, that deterministic, and even more so, probabilistic cumulative risk assessment is time-consuming, requires large databases (monitoring and consumption data bases), and extensive use of robust IT tools and algorithms (eg, MCRA model; https://mcra.rivm.nl), a simplification on the side of hazard groupings is considered especially necessary. In addition, if redundant and irrelevant CAGs can be avoided, and only the critical target organ effects of each specific compound are used for hazard grouping and cumulative risk assessment, this has the potential to offer great benefit both to society and to the chemical industry.
Conclusions
This exercise has critically analyzed the existing methodology used in the External EFSA Report, 2016, to formulate the respective liver CAGs. A proposal is made that the current groups should be significantly simplified to more accurately reflect the known pathophysiology of toxic lesions to the liver by excluding those lesions that occur secondarily to the pivotal hepatic toxicity arising from interaction of biological tissues with chemically reactive products. This has resulted in a reduction in the number of CAGs from the current 15 to 6, based upon primary hepatic pathology and relevance (necrosis, hepatic hypertrophy, cholestasis, porphyrin pigmentation, fatty changes, and hepatocellular neoplasms), and 3 gallbladder CAGs. Hepatocellular neoplasms are included as a CAG even though neoplasia is not, as such, a primary hepatic lesion, but this group is included to reflect its end point importance and the fact that chemicals that result in hepatic neoplasia have the potential to interact to increase the incidence and probability of this serious disease developing.
We believe that this refinement is scientifically justifiable, better reflects the available data, and will adequately protect the public from multiple exposures to chemicals producing similar toxic end points via a justifiable, and scientifically supportable, grouping scheme.
We would propose, therefore, to remove those CAG levels that are based upon secondary end points and as a consequence to reduce the number of cumulative assessment categories for the liver to more accurately describe the process for risk assessment. We would also recommend separating the gall bladder and bile duct CAGs from the liver ones on the grounds that the 2 organ systems are fundamentally different in the way in which they react toxicologically, biologically, and physiologically.
Footnotes
Authors’ Note
All the authors contributed to the conception, design, writing review, and revision of this manuscript. All authors gave final approval and agreed to be accountable for all aspects of the manuscript in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately resolved.
Declaration of Conflicting Interests
Dr Foster received financial support for the preparation of this manuscript from the European Crop Protection Agency (ECPA) and Drs Semino-Beninel and Melching-Kollmuss are employed by companies whose business could potentially be impacted by the introduction of the new guidelines on cumulative risk assessment. The views expressed in this manuscript are based on the professional judgment of the authors and do not necessarily represent those of ECPA or of the businesses with whom the authors are employed.
Funding
JRF received funding for the preparation and publication of this manuscript from the European Crop Protection Association. GS-B and SM-K were employed respectively by Bayer CropScience and BASF SE during the preparation and review of the manuscript.
