Abstract
Cyclotrimethylenetrinitramine (RDX) has been used extensively as an explosive in military munitions. Mechanisms for seizure production, seen in past animal studies, have not been described. Increased calcium levels contribute to excitotoxicity, so in this study neuroblastoma cells are loaded with calcium-indicating dye before application of 1.5 µM to 7.5 mM RDX, with fluorescence recorded for 30 cycles of 11 seconds each. The lowest concentration of RDX increases calcium fluorescence significantly above baseline for cycles 2 to 8; millimolar concentrations increase calcium fluorescence significantly above baseline for cycles 2 to 30. Increases in calcium, like those of 200 nM carbachol, are prevented with 10 mM of calcium chelator ethylene glycol-bis(β-aminoethyl ether)-N,N,N,N tetra-acetic acid (EGTA, tetrasodium salt). Calcium channel blocker verapamil (20 μM), Ca2+-ATPase inhibitor thapsigargin (5 μM), and general membrane stabilizer lidocaine (10 mM) partially attenuate carbachol- and RDX-induced increases in calcium, suggesting that RDX transiently increases intracellular calcium by multiple mechanisms.
Cyclotrimethylenetrinitramine (RDX, an abbreviation for rapid detonation explosive or royal demolition explosive 1 ), an uncharged cyclic molecule with 3 carbons and 3 nitramine groups, is a relatively insoluble munitions compound. 2 There is concern about exposure of people and animals to RDX attributable to environmental contamination because it has been detected in soils and water at production and disposal sites or load assembly and packing facilities. 3-8 RDX is rapidly absorbed by the oral route; is distributed to kidney, liver, heart, and brain; and undergoes metabolism. Parent compound and metabolites are excreted in the urine. 8,9
Studies that have examined the effects of RDX in rodents have noted that seizures may appear in some animals. 2 For example, seizures were observed in rats after a single oral dose of 25 mg/kg. 10 The seizures did not always result in death, but animals did not die unless the seizures occurred. Previous studies in rats and swine indicated that RDX (300 μM or 7 μg/g) can be detected in brain within 0.5 to 1 hour after oral exposure. 9 Concentrations of RDX in plasma of in rats demonstrating audiogenic-induced seizures after a single oral dose of 10 to 12.5 mg/kg RDX have been as high as 240 to 370 μM (5.34-8.28 μg per milliliter of plasma). 10 Investigations into the causes of seizures and death have been unsuccessful, because no pathological changes were noted on necropsy.
Attempts have been made to delineate possible RDX-related mechanisms that could contribute to seizures preceding death in laboratory rodents, but none have been precisely identified. Seizure-related sudden death suggests that rapid changes in excitatory neurotransmitters or ion-mediated electrical properties of the central nervous system are involved. 11 Effects on glutamate concentration in brain of rats attributable to RDX have been hypothesized 1 ; this effect is associated with elevation of intracellular free calcium levels. 12,13 Although in vitro experiments cannot be directly extrapolated to the in vivo situation, experiments can be done to determine contributions of excitatory neurotransmitters, changes in intracellular calcium, and mitochondrial effects of RDX using neuronal cells in culture. Experiments in other laboratories have established association of the excitatory neurotransmitter glutamate with increases of intracellular calcium in human neuroblastoma SH-SY5Y cells, 14,15 which prompted the use of this cell line for the studies described.
The current experiments demonstrated that increases in intracellular free calcium can be altered in neuronal cells by high concentrations of RDX. This raises the possibility that ion-induced changes (excitability) provide a potential mechanism associated with seizures as seen in vivo. 13 Because RDX-induced spikes of increased intracellular calcium concentrations in human neuroblastoma SH-SY5Y cells seen during our experiments may be attributable to mechanisms associated with calcium channels, general disruption of cellular membranes, or nonspecific factors, the experiments included agents that modulate calcium levels in cells, such as a calcium chelator, calcium channel blockers, and a membrane stabilizer.
Method
Materials
Medium and other materials for cell culture were obtained from Mediatech (Manassas, Va). Test compounds were obtained from Sigma Chemical (St. Louis, Mo). The calcium-measuring kit was obtained from Invitrogen Corporation (Carlsbad, Calif).
Chemical
Hexahydro-1,3,5-trinitro-1,3,5-triazine (RDX, CAS no. 121-82-4) was obtained from Holston Army Ammunition Plant (Kingsport, Tenn) and was further purified in the laboratory (99.99% purity) by Dr William M. Koppes, Naval Surface Warfare Center, Indian Head, Maryland.
Cell Culture
SH-SY5Y human neuroblastoma cells (ATCC CRL-2226, passages 27-39, Manassas, Va) were removed from storage in 1% DMSO in liquid nitrogen and transferred to T75 cm2 flasks containing Dulbecco’s modified eagle medium plus 15% fetal bovine serum (FBS, Atlanta Biologicals, Lawrenceville, Ga). Initial experiments were done using cells grown in 15% serum 16 ; other experiments, specifically identified, were done using cells grown without FBS because certain xenobiotics can bind to serum proteins. For serum-free cells, the cultures were removed from frozen storage and transferred to 10% FBS in Ham’s F-12 medium for 2 passages; then the FBS was reduced to 5% serum (in 50% Ham’s F-12 + 50% Cellgro complete medium) for 2 passages before the cells were placed in a 96-well plate with N2-supplemented Cellgro complete medium (Invitrogen) without FBS or phenol red.
Viability in the presence of RDX was determined 24 hours after exposure using Trypan blue. For additional experiments determining viability, SH-SY5Y cells were incubated with RDX for 1 and 24 hours before the Alamar blue dye or 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT, Sigma) were added 17 for an additional 4 hours before reading on a Spectra Max 250 UV-VIS microplate reader (Molecular Devices, Sunnydale, Calif). General membrane integrity was determined using a fluorescent probe, the acetoxymethyl ester derivative of 5-carboxyfluorescein diacetate (CFDA-AM, Invitrogen), that cleaves to a green fluorescent membrane impermeable product (excitation 485 nm, emission 530 nm) described previously. 17,18 SH-SY5Y cells were incubated with RDX for 1, 3, and 24 hours before CFDA-AM was added, and the plates were read on a SpectraMax M5 multidetection reader. The mitochondrial-specific cationic fluorescent dye R123 dye (Sigma) was used to determine transmembrane potential. 19 For all cytotoxicity experiments, cell suspensions (180 μL) were plated at a density of 3.6 ×104 cells per well into 96-well plates; the experiments were performed after overnight incubation. Stock solutions of 0.75 M RDX were prepared in DMSO. This stock solution was diluted in culture media so final concentrations of RDX were between 7.5 mM and 1.5 μM. DMSO 1% and ethanol 98% were used as the vehicle control and positive control, respectively.
Cellular Ca2+ Measurement
Initial experiments were conducted with 1.5 to 7.5 mM RDX to determine concentrations capable of elevating intracellular calcium levels. The procedure was similar to that used previously in our laboratory. 20 For experiments in which mechanisms associated with increases in calcium were examined, RDX in concentrations sufficient to produce relatively high consistent and immediate peaks of calcium influx (7.5 and 1.5 mM) were used. For these experiments, human SH-SY5Y cells (4 × 104 cells/well, 200 μL) were grown overnight in 96-well plates prior to cellular Ca2+ measurements. 21 Carbachol (200 nM) was used to provide a positive control for release of calcium ions from intracellular stores, following the instructions in the calcium assay kit. 22 Cells in medium were pretreated with 20 μL of one of the following prior to addition of RDX: 10 mM of the calcium chelator ethylene glycol-bis(β-aminoethyl ether)-N,N,N,N tetraacetic acid (EGTA, tetrasodium salt) for 30 minutes; 20 μM of the calcium channel blocker verapamil for 15 minutes, or 10 mM lidocaine, a general membrane stabilizer that acts on sodium channels, for 30 minutes. Increases in Ca2+ were also measured in cells treated with thapsigargin (5 μM) for 10 minutes to examine the contribution of Ca2+-ATPase to the RDX-induced effect. 23 Additional experiments in serum-free medium examined effects of the calcium channel blocker (nifedipine 20 μM) and the muscarinic blocker (atropine (20 μM) as potential ameliorating agents for RDX-induced increases in intracellular Ca2+. Calcium chloride (1 mM in lieu of carbachol or RDX) was used to determine whether an external source of calcium could increase intracellular calcium concentrations in the system. For measurement of [Ca2+], the growth medium was removed from the cultured SH-SY5Y cells, and 100 μL Fluor-4 dye (Fluo-4 NW calcium assay kit, Molecular Probes Invitrogen Detection Technologies) solution was quickly and carefully added to each well of the 96-well plate. The plate was incubated at 37°C for 30 minutes and then at room temperature for an additional 30 minutes. The plate was measured using a fluorescent plate reader (Fluostar, BMG Labtech, Durham, NC) at wavelengths appropriate for excitation at 485 nm and emission at 520 nm. 22 Basal levels of fluorescence were measured (F 0) for a minimum of 5 cycles (55 seconds) before using an automatic dispenser to add the test agents RDX or carbachol. After stimulation with 200 nM carbachol or 1.5 mM or 7.5 mM RDX, changes in fluorescence were measured at cycles of 11 seconds for a total of 5.5 minutes (30 cycles). DMSO vehicle treatment did not significantly affect baseline. At the end of each experiment, the fluorescence of the maximal intracellular calcium was determined by the addition of 1% Triton X-100 (F max). This was followed by the addition of 10 mM EGTA to obtain the fluorescence of the lowest possible quantity of intracellular calcium (F min). The percentage fluorescence in cells containing RDX or carbachol was determined by the following equation: %F = (F − F 0)/(F max − F min) × 100. 24 Experiments were repeated with different batches of cells at least 3 times to provide sufficient data for statistical analyses.
Data Analyses
For cellular Ca2+ measurements, data analysis focused on the initial RDX titration experiment and on each of the 6 arms of the study constituted by a combination of growth medium (2 levels: with or without FBS) and test compound (3 levels: 1.5 mM RDX, 7.5 mM RDX, and carbachol as the positive control). The primary outcome compared was percentage fluorescence determined as described previously. For the initial RDX titration experiment, 2 mixed-model repeated-measures analysis (ANOVA) models were fitted to the data. The first model aimed to test whether the least squares mean for each combination of cycle (1-30) and dose was significantly different from baseline (mean %F = 0) (Bonferroni’s procedure was applied to correct for multiple comparisons). The second model aimed to compare different doses of RDX over the first 10 cycles (Tukey’s procedure was applied to correct for multiple comparisons). Within each arm of the main study, several agents that modulate intracellular levels of calcium were evaluated. These agents included EGTA, lidocaine, verapamil, and thapsigargin. Nifedipine and atropine were evaluated only for cells grown in medium without FBS. For example, a study arm of carbachol and growth medium with FBS had the following treatments: 200 nM carbachol, 10 mM EGTA, 20 μM verapamil, 5 μM thapsigargin, and 10 mM lidocaine. For each arm of the study, sets of null hypotheses were stated as follows: the test compound in that arm of the study (eg, 200 nM carbachol in the example above) is not significantly different from any of the modulators of intracellular levels of calcium (eg, 10 mM EGTA, 20 μM verapamil, 5 μM thapsigargin, and 10 mM lidocaine in the same example above) for percentage fluorescence. To test the stated hypotheses, percentage fluorescence data were compared between treatments using mixed-model repeated-measures ANOVA followed by the Dunnett’s procedure for multiple comparisons. The model included cycles 1 to 10 and treatment as fixed effects and date of experiment as a random blocking factor. Furthermore, the slice option of the GLIMMIX procedure was used to compare the treatments at each of the cycles. Statistical significance was set at α alpha = .05. All analyses were performed using SAS version 9.1.3 (Cary, NC).
Results
Concentrations of RDX as high as 7.5 mM did not affect the viability of SH-SY5Y human neuroblastoma cells during incubations of durations up to 24 hours. This was true whether determined using Trypan blue, Alamar blue, MTT, or CFDA. Viable cells remained greater than 84% of control with these test agents as well as with 0.1% and 1% concentrations of the vehicle DMSO when compared with cells without either (data not shown). The 10% ethanol treatment included as a positive control had significant effects on viability (eg, viability 11% to 41% of DMSO vehicle after <24 hours of exposure). Examination of effects on mitochondria using the fluorescent dye R123 did not indicate an RDX-induced effect on this organelle (data not shown).
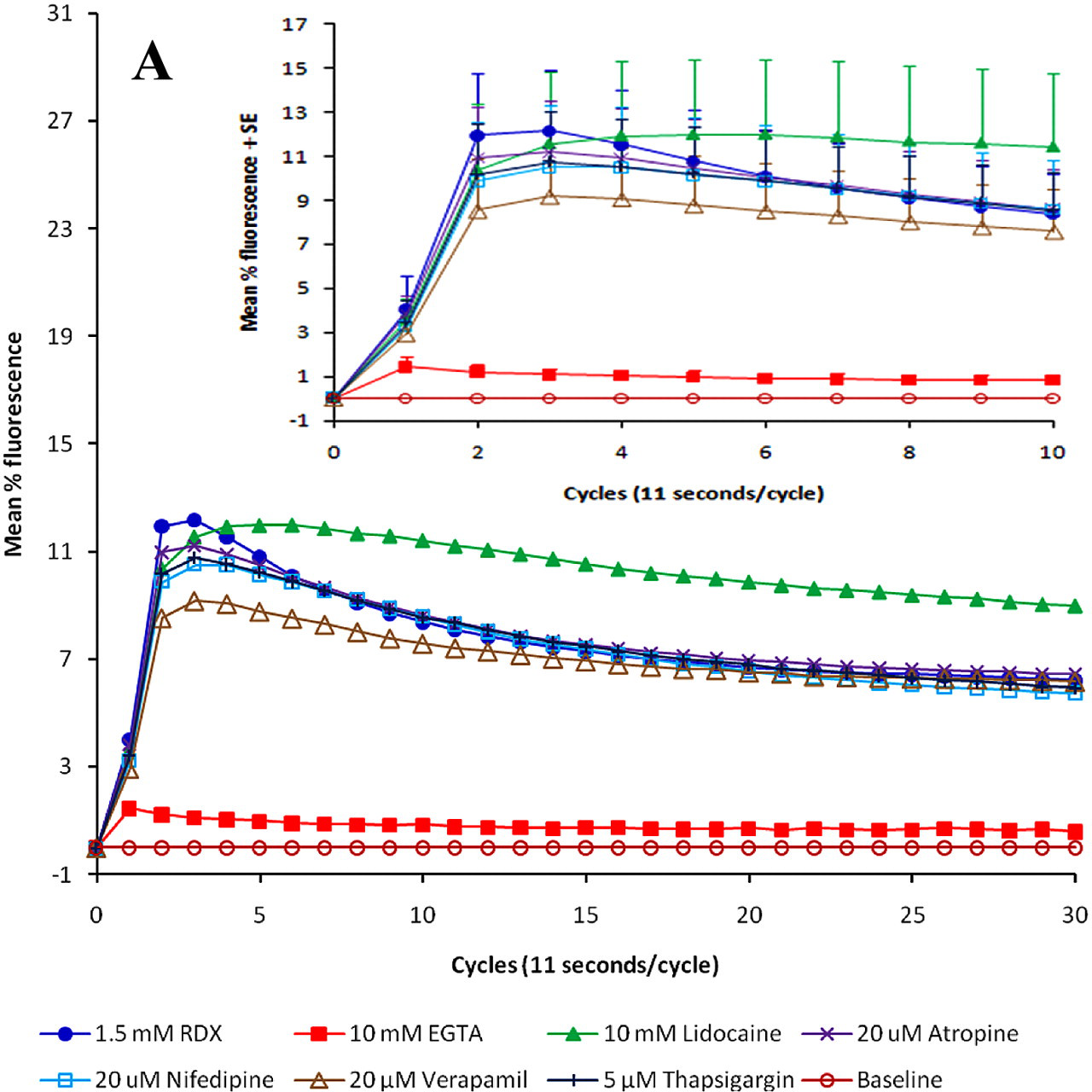
Application of RDX in concentrations from 1.5 µM to 7.5 mM caused an intensive, rapid, and significant increase of intracellular free calcium ions, an effect that diminished with time (Figure 1). Statistically significant changes from baseline were noted as follows: for 1.5 μM RDX, the spike was significant at cycles 2 to 8 (P < .05). For 15 μM RDX, the spike was significant at cycles 2 to 16 (P < .05). For 150 μM RDX, the spike was significant at cycles 2 to 18 (P < .05). For 1.5 mM RDX, the spike was significant at cycles 2 to 30 (P < .05). For 7.5 mM RDX, the spike was significant at cycles 2 to 30 (P < .05). Moreover, this effect was concentration-related. From cycle 4 to cycle 10, calcium increase (as mean percentage fluorescence) in cells treated with RDX 7.5 mM was significantly greater than the calcium increase in cells that were treated with either 1.5 μM, 15 μM, 150 μM, or 1.5 mM of RDX (P < .05). Accordingly, RDX 7.5 mM and RDX 1.5mM were chosen for the later experiments. Carbachol and glutamate, used as positive controls, also immediately increased calcium release from the cells (Figure 2c shows results for 200 nM carbachol; 7.5 mM L-glutamate gave a similar curve; data not shown). External application of calcium chloride had no effect on intracellular calcium levels.
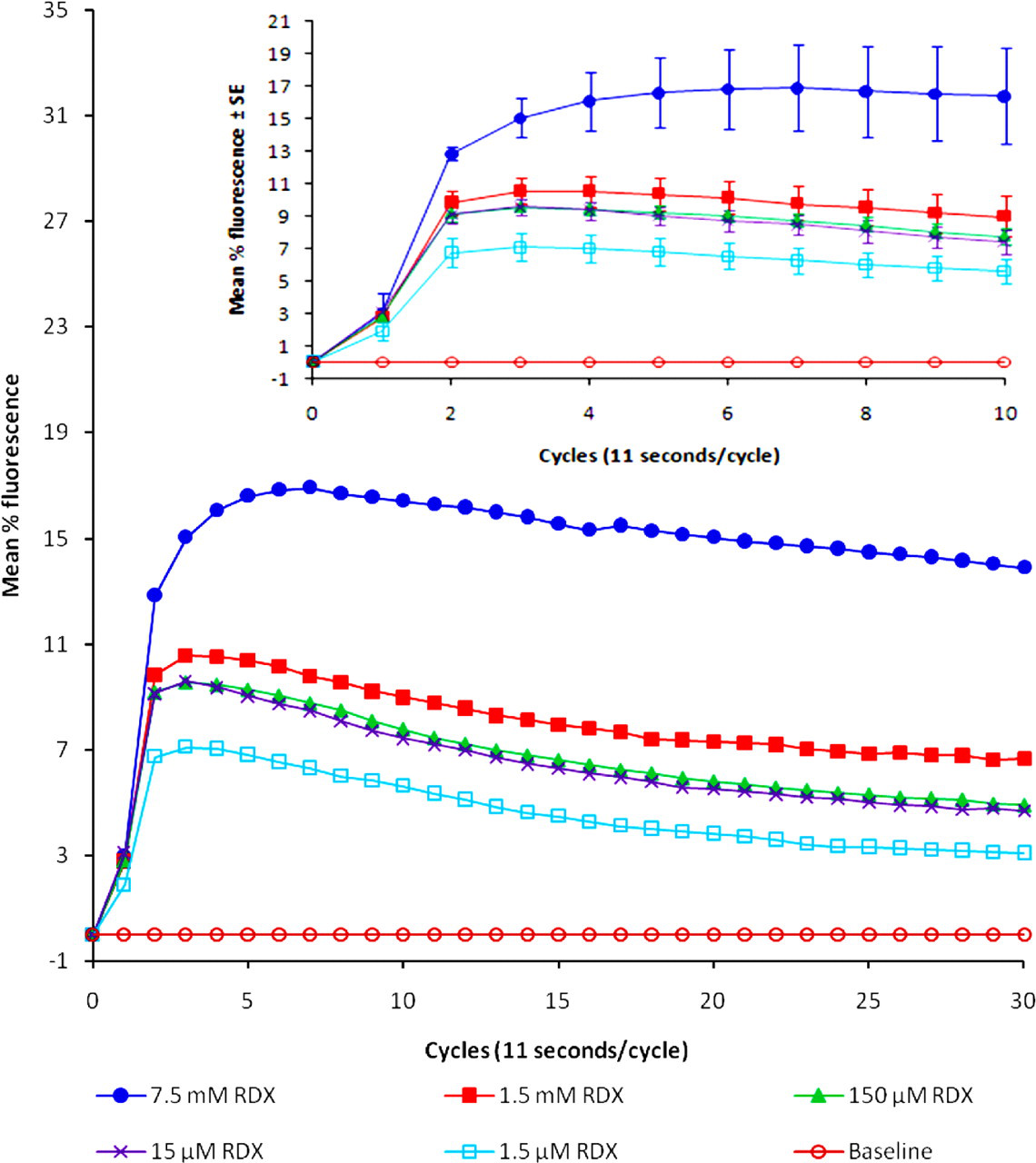
Mean calcium release in SH-SY5Y cells to which RDX had been applied. RDX was applied to cells 1 hour after incubation with Fluoro-4 dye, and calcium levels were measured every 11-second cycle for a total of 30 cycles. The insert illustrates effects in the first 10 cycles (±SE). The mean values are from a minimum of 3 different determinations. Statistically significant changes from baseline (mean %F = 0) were noted as follows: for 1.5 μM RDX, the spike was significant at cycles 2 to 8 (P < .05). For 15 μM RDX, the spike was significant at cycles 2 to 16 (P < .05). For 150 μM RDX, the spike was significant at cycles 2 to 18 (P < .05). For 1.5 mM RDX, the spike was significant at cycles 2 to 30 (P < .05). For 7.5 mM RDX, the spike was significant at cycles 2 to 30 (P < .05).
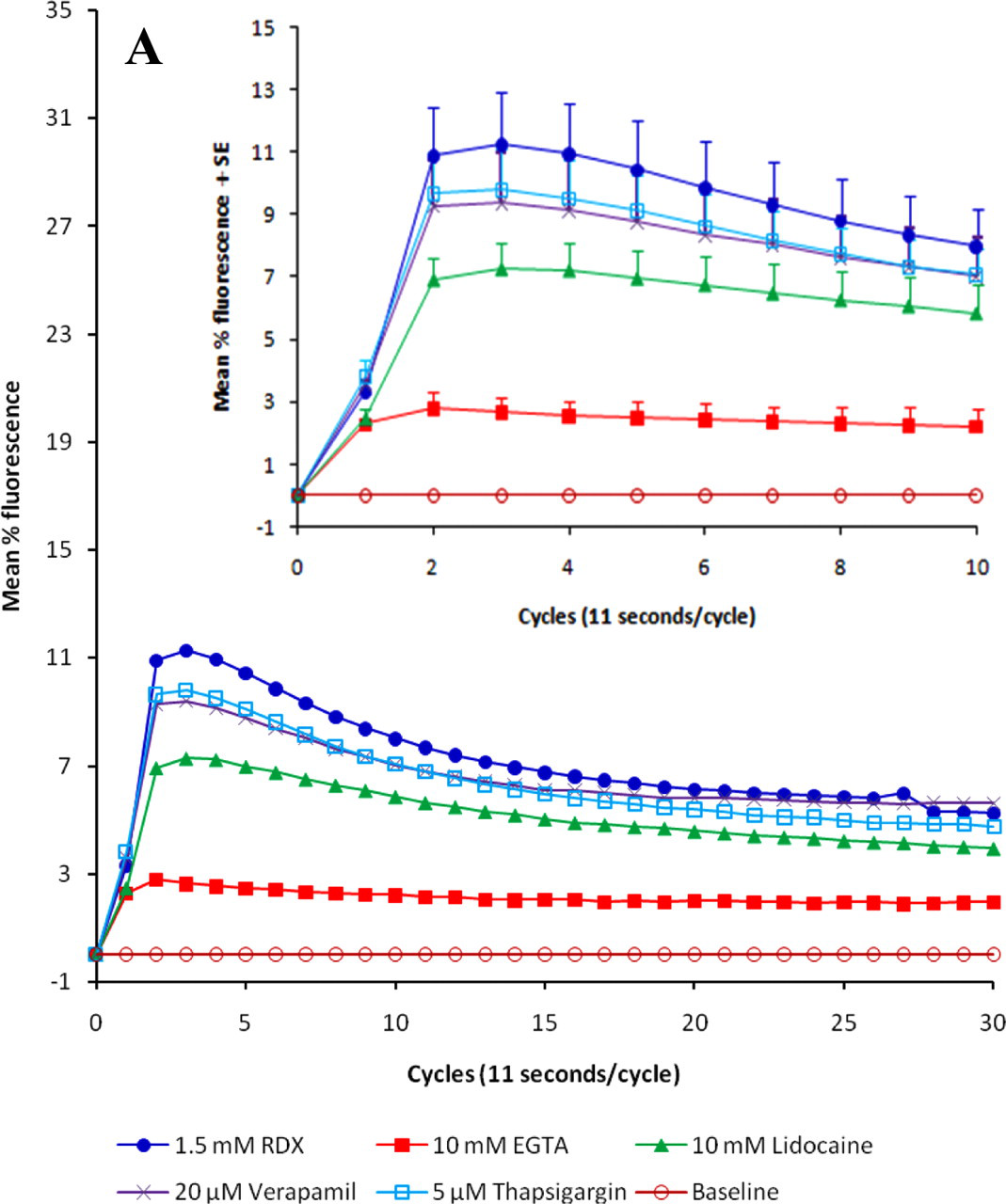
Profile plots for intracellular calcium release induced by RDX and carbachol: (A) 1.5 mM RDX, (B) 7.5 mM RDX, (C) the positive control carbachol. Amelioration was determined in the presence of the calcium chelator EGTA, the membrane stabilizer lidocaine, the calcium channel blocker verapamil, and the Ca2+-ATPase inhibitor thapsigargin. Results are expressed as mean values, n = 4-6; inserts contain values for the first 10 cycles ± SE.
Capability of RDX to increase intracellular free calcium ions was examined and compared with effects of a positive control, carbachol, in the presence of agents used to probe the mechanism of calcium elevation. The first experiments were done in usual cell culture conditions, which included fetal bovine serum. Qualitatively, the peak values for RDX (1.5 and 7.5 mM) were similar to what was observed with 200 nM carbachol (Table 1). The ability to increase intracellular calcium was essentially abolished by preincubation with the calcium chelator EGTA and reduced by preincubation with the calcium channel blocker verapamil, the Ca2+-ATPase inhibitor thapsigargin, and the membrane stabilizer lidocaine. These effects were seen on calcium profiles as well as on peak values (Figure 2).
Peak Values for Calcium Influx in SH-SY5Y Cells Exposed to RDX and Carbachol
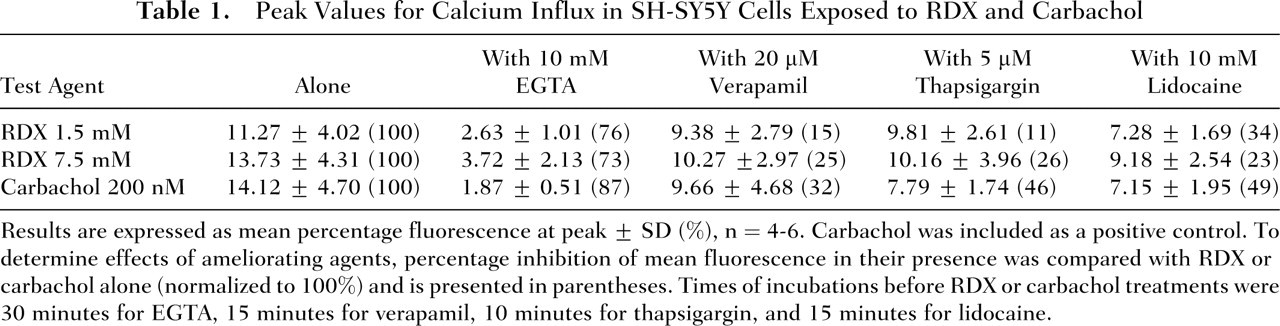
Results are expressed as mean percentage fluorescence at peak ± SD (%), n = 4-6. Carbachol was included as a positive control. To determine effects of ameliorating agents, percentage inhibition of mean fluorescence in their presence was compared with RDX or carbachol alone (normalized to 100%) and is presented in parentheses. Times of incubations before RDX or carbachol treatments were 30 minutes for EGTA, 15 minutes for verapamil, 10 minutes for thapsigargin, and 15 minutes for lidocaine.
When these results were analyzed using a mixed-model repeated-measures ANOVA of cycles 1-10 for arms containing bovine fetal serum, the following observations were made: RDX 1.5 mM: From cycle 2 to cycle 5, calcium increase (or mean percentage fluorescence) in cells treated with RDX 1.5 mM alone was significantly greater than the calcium increase in cells that were pretreated with either 10 mM EGTA or 10 mM lidocaine (P < .05). Furthermore, calcium increase (as mean percentage fluorescence) in cells treated with RDX 1.5 mM alone was significantly greater than the calcium increase in cells that were pretreated with 10 mM EGTA at cycles 6 through 10 (P < .05). All other comparisons were not statistically significant (P > .05). RDX 7.5 mM: From cycle 2 to cycle 7, calcium increase (as mean percentage fluorescence) in cells treated with RDX 7.5 mM alone was significantly greater than the calcium increase in cells that were pretreated with either 10 mM EGTA, 20 μM verapamil, or 10 mM lidocaine (all P < .05). Furthermore, calcium increase (as mean percentage fluorescence) in cells treated with RDX 7.5 mM alone was significantly greater than the calcium increase in cells that were pretreated with 10 mM EGTA at cycles 8 to 10 (P < .05) or 20 μM verapamil at cycles 8 and 9 (P < .05). All other comparisons were not statistically significant (P > .05). Carbachol 200 nM: From cycle 2 to cycle 6, calcium increase (or mean percentage fluorescence) in cells treated with carbachol alone was significantly greater than the calcium increase in cells that were pretreated with either 10 mM EGTA, 20 μM verapamil, 5 μM thapsigargin, or 10 mM lidocaine (all P < .05). Furthermore, calcium increase (as mean percentage fluorescence) in cells treated with carbachol alone was significantly greater than the calcium increase in cells that were pretreated with 10 mM EGTA at cycles 7 to 10 (all P < .05). All other comparisons were not statistically significant (P > .05).
Because increases in intracellular calcium can lead to apoptosis and cell death in SH-SY5Y cells
25
and because albumin in medium has been reported to interfere with (decrease) apoptosis induced by test agents,
25
calcium increases in the absence of serum were determined. Qualitatively, the peak values for RDX (1.5 and 7.5 mM) were similar to those seen when FBS was included in the medium (Table 2 compared with Table 1) but 50% lower than the mean peak value for 200 nM carbachol (Table 2). As observed with media containing serum, the ability to increase intracellular calcium was abolished by preincubation with the calcium chelator EGTA (Table 2). Figure 3 illustrates the curves of RDX-induced intracellular release when FBS was not included in the medium.
Peak Values for Calcium Influx in SH-SY5Y Cells Exposed to RDX and Carbachol in Serum-Free Medium
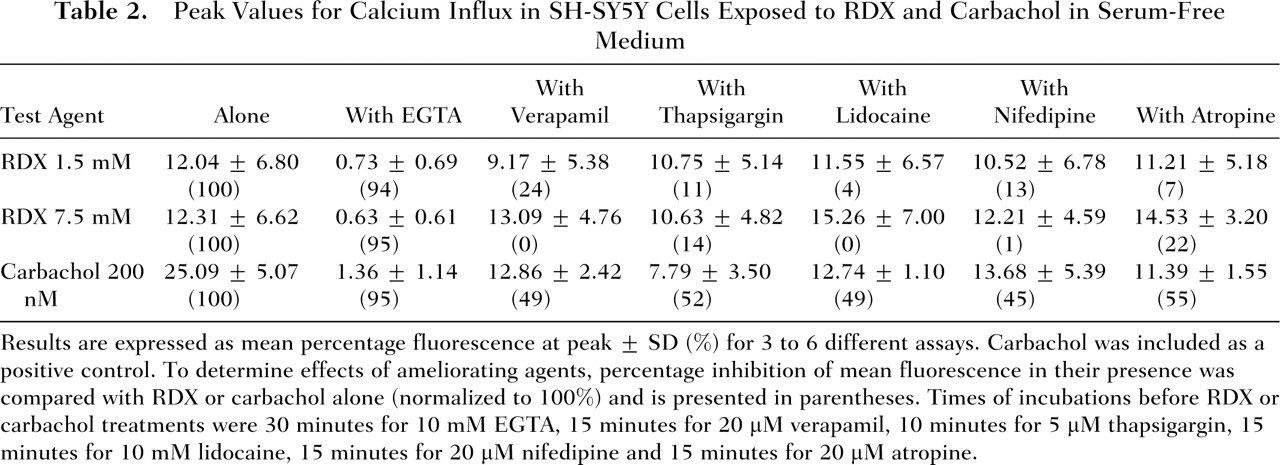
Results are expressed as mean percentage fluorescence at peak ± SD (%) for 3 to 6 different assays. Carbachol was included as a positive control. To determine effects of ameliorating agents, percentage inhibition of mean fluorescence in their presence was compared with RDX or carbachol alone (normalized to 100%) and is presented in parentheses. Times of incubations before RDX or carbachol treatments were 30 minutes for 10 mM EGTA, 15 minutes for 20 μM verapamil, 10 minutes for 5 μM thapsigargin, 15 minutes for 10 mM lidocaine, 15 minutes for 20 μM nifedipine and 15 minutes for 20 μM atropine.
Amelioration of RDX-induced increases in calcium in serum-free cultures: (A) 1.5 mM RDX, (B) 7.5 mM RDX, (C) positive control carbachol. Figures include mean values for 30 cycles (n = 4-6), with inserts containing results for 10 cycles ± SE for 10 cycles. Calcium was measured in the absence and presence of calcium channel blockers (nifedipine, verapamil), membrane stabilizers (thapsigargin, lidocaine), a muscarinic blocker (atropine), and a calcium chelator (EGTA).
When we conducted the analysis using a mixed-model repeated-measures ANOVA of cycles 1 to 10 for arms without bovine fetal serum, the following observations were made: RDX 1.5 mM: From cycle 2 through cycle 6, calcium increase (or mean percentage fluorescence) in cells treated with RDX 1.5 mM alone was significantly greater than the calcium increase in cells that were pretreated with 10 mM EGTA (P < .05). All other comparisons were not statistically significant (P > .05). RDX 7.5 mM: From cycle 2 through cycle 10, calcium increase (as mean percentage fluorescence) in cells treated with RDX 7.5 mM alone was significantly greater than the calcium increase in cells that were pretreated with 10 mM EGTA (P < .05). Additionally, although RDX could increase calcium levels when added to cells in serum-free medium, calcium increase (as mean percentage fluorescence) in cells treated with RDX 7.5 mM alone was significantly less than the calcium increase in RDX-treated cells that were pretreated with 10 mM lidocaine at cycles 6 through 10 (P < .05). All other comparisons were not statistically significant (P > .05). Carbachol 200 nM: From cycle 2 to cycle 8, calcium increase (as mean percentage fluorescence) in cells treated with carbachol alone was significantly greater than the calcium increase in cells that were pretreated with either 10 mM EGTA, 20 μM verapamil, 5 μM thapsigargin, 10 mM lidocaine, 20 μM nifedipine, or 20 μM atropine (all P < .05). Furthermore, calcium increase (as mean percentage fluorescence) in cells treated with carbachol alone was significantly greater than the calcium increase in cells that were pretreated with either 10 mM EGTA, 20 μM verapamil, 5 μM thapsigargin, 20 μM nifedipine, or 20 μM atropine at cycles 9 and 10 (all P < .05). All other comparisons were not statistically significant (P > .05).
These results show the calcium chelator EGTA worked exceedingly well to negate RDX- or carbachol-induced calcium increases in cells living in serum-free medium, even better than it did for cells in the medium that contained 15% FBS. For carbachol, the percentage reductions of the peak calcium ion concentrations with verapamil, thapsigargin, and lidocaine were relatively similar or somewhat enhanced from their efficacy in 15% FBS medium. Nifedipine, a calcium channel blocker with a structure much different from verapamil, and atropine, which blocks the muscarinic site at which carbachol acts, were also effective in serum-free medium but were not tested in 15% FBS.
The calcium chelator EGTA was the most effective agent decreasing RDX-induced increases of intracellular calcium concentrations. None of the proposed mechanistic antidotes showed effects as great in serum-free medium as when they were examined in RDX-treated cells in 15% FBS (although nifepidine and atropine were not tested in the serum-containing medium). These results show that RDX treatment increases intracellular calcium, but its mechanism remains elusive, because neither a Ca2+-ATPase inhibitor (thapsigargin), calcium channel blockers (verapamil, nifedipine), a sodium channel-active agent and general membrane stabilizer (lidocaine), nor a muscarinic antagonist (atropine) could completely ameliorate the RDX-induced calcium increases. Increasing the concentrations of these potential ameliorating agents beyond those used in Figures 2 and 3 did not result in proportional increases in their effectiveness (data not shown).
One unexplained discrepancy between results in serum-containing and serum-free media is the inability of lidocaine to ameliorate RDX-induced effects on increased calcium when exposures were done in serum-free medium. Exaggeration of calcium influx was noted.
Discussion
The present experiments demonstrated that RDX in micromolar to low millimolar concentrations could elevate calcium ion levels in human neuroblastoma cells. Although extrapolation from in vitro experiments to in vivo situations should only be made with great caution, 26 these concentrations are within range of the 0.3 mM measured in brains of rats with seizures after RDX administration. 10 The elevation of cellular calcium was not sustained, which made detection difficult without specialized instrumentation. That the increase was so transient could be a contributing factor in why the events responsible for RDX-induced seizures in animal models have not been determined. The RDX-induced elevation of cellular calcium in neuroblastoma cells was negated in the presence of the calcium chelator EGTA but could only partially be ameliorated by test agents known to affect calcium channels, membrane stability, or active transport within cells. Therefore, specific identification of the molecular mechanisms involved could not be made. That several agents were effective, however, suggests that multiple mechanisms could be involved.
The in vitro experiments presented here suggest that the immediate calcium increases follow release from intracellular calcium stores, because external application of calcium had no effect. Although the changes were not exceedingly large (probably because they involved release from internal stores), they were significant, providing the first evidence of a realistic mechanism for increasing excitotoxicity in neuronal cells exposed to RDX. Because this event occurs within seconds of exposure, detection of micromolar-induced changes required a fluorescence reader with autoinjection. Increases in calcium were more sustained at millimolar concentrations of RDX, as seen both when a fluorescent plate reader was used for detection and when flow cytometry was used. 27 The transient action suggests that a mechanism relating to ion changes contributed to neurotoxic effects of RDX. As noted above, however, blocking of calcium or sodium channels only provided incomplete protection.
The experiments demonstrated that RDX behaved in a manner similar to carbachol in SH-SY5Y neuroblastoma cells, although the effective concentrations of carbachol, an analog of acetylcholine, were much lower. This included the ability of atropine to ameliorate test agent–induced increases in calcium. These cells have muscarinic receptors known to respond to carbachol. 28 It is unknown whether initiation of cellular calcium increases in vivo following RDX would involve this receptor.
Seizures can occur in some RDX-treated rodents. 2,8-10 Although elevation of free intracellular calcium may have a role in the occurrence of seizures, already available therapeutic agents such as verapamil and lidocaine would be expected to have only have limited usefulness in preventing or treating RDX-induced seizures. Although the situation in vivo differs, in vitro experiments suggest that the increase in calcium is transient, complicating selection of appropriate timing for treatment. Difficulty in timing of treatment for seizures in experimental animals may be further complicated by the toxicokinetics of RDX contributing to its presence in the brain. 9 Biotransformation, however, is unlikely to play a role in the RDX-induced effects in the neuroblastoma cells used for the present experiments. 29,30
Seizures result from an imbalance of excitatory and inhibitory transmitters. Glutamate is the excitatory transmitter involved, and calcium increases mediate its release. 13 The association of glutamate and increased intracellular calcium has previously been demonstrated in the cell culture system used in the present study. 14,15 However, attempts to examine potential RDX-induced increases in glutamate concentrations in SH-SY5Y cells exposed to RDX were not successful. 27 In our experiments, the presence of the excitatory neurotransmitter glutamate was determined by a sensitive gas chromatography-mass spectrometry method (EZ:faast, Phenomenex, Torrance, Calif) following exposure of cells to concentrations of RDX up to 1000 μM for 60 minutes, but no significant differences were seen from levels in control cells (~100 pM/μL cell extract). Potassium chloride (KCl, 40-70 mM) serves as a positive control for the induction of glutamate release in other cells 31 but only provided slight but nonsignificant elevation of glutamate release in our SH-SY5Y cells. Although present in this cell line, 14,15 quantities of glutamate are comparatively low 32 ; other cell lines or mixed cultures containing glia, which are involved in glutamate synthesis, 13 may be better suited for studies on this aspect of excitotoxicity.
The RDX-induced effect on calcium concentration, however, even without a preexisting elevation of glutamate, suggests that this ion can induce excitability in SH-SY5Y human neuroblastoma cells. Verification could be done by determining electrical currents, but these cells are too small for this type of patch-clamp experiments (C. Massicotte, J. B. Bloomquist, and M. Ehrich, unpublished observations, October 2000.), even though others have reported patch clamp use of these cells for nicotinic receptor-initiated currents. 33 However, an indication of ion-induced changes (excitability) provides a potential mechanism associated with seizures seen in vivo. One has to take considerable care, however, in extrapolating results seen in vitro with expectations in vivo. 26 Concentrations of RDX, however, in plasma of rats demonstrating seizures after administration of 10 to 12.5 mg/kg RDX have been as high as 240 to 370 μM (5.34-8.28 μg per milliliter of plasma). Brain concentrations have been reported to be 300 μM (7 μg/g). 2 The higher concentrations needed to elicit response in continuous cell lines are not unexpected. 26,30 Because calcium spikes were noted, however, this mechanism deserved closer scrutiny.
These in vitro experiments demonstrated that RDX applied to neuroblastoma cells resulted in a transient increase in free calcium ions. Increases in intracellular calcium are a mechanism that can contribute to excitotoxicity, but one cannot make direct extrapolations from in vitro experiments to in vivo situations.
Footnotes
Acknowledgments
Funding for this research was from the Directorate of Toxicology, US Army Center for Health Promotion and Preventive Medicine, Aberdeen Proving Ground. We thank Drs D. Bannon and C. Cao for critical review of the manuscript, and Dr L. Li for use of his laboratory.
