Abstract
Cadmium, a highly ubiquitous heavy metal, is well known to induce neurotoxicity. However, the underlying mechanism of cadmium-mediated neurotoxicity remains unclear. We have studied cadmium inhibition of neurite outgrowth using human SH-SY5Y neuroblastoma cells induced to differentiate by all-
Introduction
Cadmium is a highly toxic heavy metal that affects human health through environmental and occupational exposure and has been classified as a type I carcinogen by the International Agency for Research on Cancer. 1 Besides its carcinogenic properties, cadmium has been widely reported to cause harmful effects in a variety of organs, including lung, kidney, liver, bladder, and prostate. 2 –5 In addition to peripheral organs, the central nervous system is also subjected to cadmium toxicity. 6 It is shown in experimental studies with animals that cadmium is able to induce neurotoxicity, 7 cerebral edema and hemorrhage, 8 and disruption of blood–brain barrier. 9 Although there are studies showing the neurotoxicity of cadmium on cell culture models like neurons and glial cells, 10 –12 the effects of cadmium on neuronal differentiation remain to be determined.
Cell cultures derived from nervous system tissue have proven to be powerful tools for elucidating cellular and molecular mechanisms of nervous system development and function and have been used to understand the mechanism of action of neurotoxic chemicals. As neurons showing characteristic morphology with many cell processes have the ability of extending neurite, neurite outgrowth in cultured neurons is considered one indication of neuroregenerative potential.
13,14
The human SH-SY5Y neuroblastoma cell line has the capability of undergoing neuronal maturation. It can be differentiated into neuron-like cells with neurites in response to all-
Neuronal differentiation of SH-SY5Y cells in response to RA is coupled with increased expression and activation of transglutaminase (TGase). 18 It was demonstrated that TGase is required for the promotion of neurite outgrowth. 19 Growth-associated protein 43 (GAP-43), a nervous tissue-specific cytoplasmic protein, has been termed a “growth” or “plasticity” protein because it is expressed at high levels in neuronal growth cones during development and axonal regeneration. 20 It is a major protein kinase C (PKC) substrate and is considered to play a key role in formation, regeneration, and plasticity of neurite. 21 A key implication of the GAP hypothesis is that expression of intrinsic determinants of growth competence in neurons may provide a mechanism to regulate neurite outgrowth and structural plasticity in the nervous system. In SH-SY5Y cells, the differentiation was accompanied with upregulation of GAP-43. 22 Therefore, TGase and GAP-43 are widely used as markers for neurite sprouting and neuronal differentiation.
The aim of the present study was to characterize the effects of cadmium on neuronal differentiation. Using human SH-SY5Y neuroblastoma cell line, we assessed the ability of cadmium to interfere with the neurite outgrowth by differentiating neuroblastoma cells. In addition, we evaluated the molecular basis underlying the cadmium-induced morphological effects, with particular emphasis on the expression of GAP-43 which is necessary and sufficient for neuronal differentiation of human SH-SY5Y cells.
Materials and Methods
Chemicals
Cadmium chloride (CdCl2), all-
Cell Culture
The human SH-SY5Y neuroblastoma cells were purchased from Korean Cell Line Bank. Cells were maintained in complete culture medium consisting of Ham F12 and Eagle minimum essential medium (Invitrogen Co, Carlsbad, California) supplemented with 10% heat-inactivated fetal bovine serum (Hyclone, Logan, Utah) plus 100 units/mL penicillin, 100 μg/mL streptomycin, and nonessential amino acids in a humidified incubator with 5% CO2 and 95% air at 37°C. The medium was refreshed every 2 days.
Chemical Treatments
For differentiation studies, the cells were plated in 24-well plates at a density of 1 × 105 cells/mL. After incubation for 2 days, 10 μmol/L all-
Cell Viability
The methylthiazoltetrazolium (MTT) assay was performed to measure cytotoxicity of CdCl2 on differentiating SH-SY5Y cells. Cells were plated in 24-well plates at a density of 1 × 105 cells/mL. After incubation for 2 days, the medium was refreshed, and CdCl2 (0.3 and 3 μmol/L) was administered for 2 days in the presence or absence of 10 μmol/L RA. Following exposure to CdCl2, the cells were incubated for 4 hours with 0.5 mg/mL MTT at 37°C. The reaction was stopped by removing the medium and adding acid isopropanol. The absorbance was measured at 540 nm of wavelength using a microplate reader (Bio-Rad). Laboratories, Hercules, CA.
Cell Morphological Analysis
SH-SY5Y cells were plated in 24-well plates at a density of 1 × 105 cells/mL. After incubation for 2 days, the medium was refreshed, and 10 μmol/L RA and various concentrations of CdCl2 were added simultaneously. After incubation for 2 days, the cells were fixed in 4% paraformaldehyde in phosphate-buffered saline (PBS) for 4 minutes, washed in PBS, stained with Coomassie Brilliant Blue R-250, and washed with PBS. The morphological changes in the cells were observed under a phase-contrast microscope. Those cells whose cell body diameters longer than 2 were considered as neurite-bearing cells. 24 The percentage of the cells with neurites in a particular culture was determined by counting at least 300 cells in each sample.
Measurement of Intracellular Reactive Oxygen Species Generation
Intracellular Reactive oxygen species (ROS) production was measured using 2′,7′-dichlorodihydrofluorescein (DCF-DA; Molecular Probes, Leiden, the Netherlands) as described previously. 25 Briefly, the cells were plated in 24-well plates at a density of 1 × 105 cells/mL. After incubation for 2 days, the medium was refreshed and treated with 10 μmol/L RA and CdCl2 for 2 days. Then, NAC was diluted with distilled water (3, 10, and 30 μmol/L) and added 1 hour prior to treatment with RA (10 μmol/L) and CdCl2 (3 μmol/L). The cells were incubated with 10 μmol/L DCF-DA solutions in the dark at 37°C for 30 minutes and then washed twice with PBS. The fluorescence images were obtained using fluorescence microscopy Olympus BX-50, Olympus Japan; 485-nm excitation and 535-nm emission).
Western Blot Analysis
For Western blot analysis, the SH-SY5Y cells were plated in 100-mm plates at a density of 1 × 105 cells/mL (10 mL). After incubation for 2 days, the medium was refreshed and incubated in the presence and absence of RA and CdCl2. After incubation for indicated times, the cells were washed with ice-cold PBS and lysed with lysis buffer (50 mmol/L 4-(2-Hydroxyethyl)piperazine-1-ethanesulfonic acid pH 7.5, 150 mmol/L NaCl, 10% glycerol, 1% Triton X-100, 1.5 mmol/L MgCl2, 5 mmol/L EDTA) containing protease inhibitor cocktail (Sigma Chemical Co) at 4°C for 30 minutes. Total proteins were obtained after centrifugation at 14 000
Statistical Analysis
Results are expressed as mean ± standard error. Statistical analysis was performed using Student
Results
Cytotoxicity of Cadmium on Differentiating SH-SY5Y Cells
Human SH-SY5Y neuroblastoma cells are used as a well-established cell line for studying neuronal properties and neurotoxicity in culture. To investigate the effects of cadmium on differentiation of SH-SY5Y neuroblastoma cells, we first determined the suitable subacute concentrations of cadmium. After treatment of SH-SY5Y cells with RA (10 μmol/L) for 48 hours in the presence of various concentrations of cadmium, the viability of SH-SY5Y cells was determined by MTT assay. As shown in Figure 1, cadmium concentrations less than 3 μmol/L had no significant effect on the viability of differentiating SH-SY5Y cells.
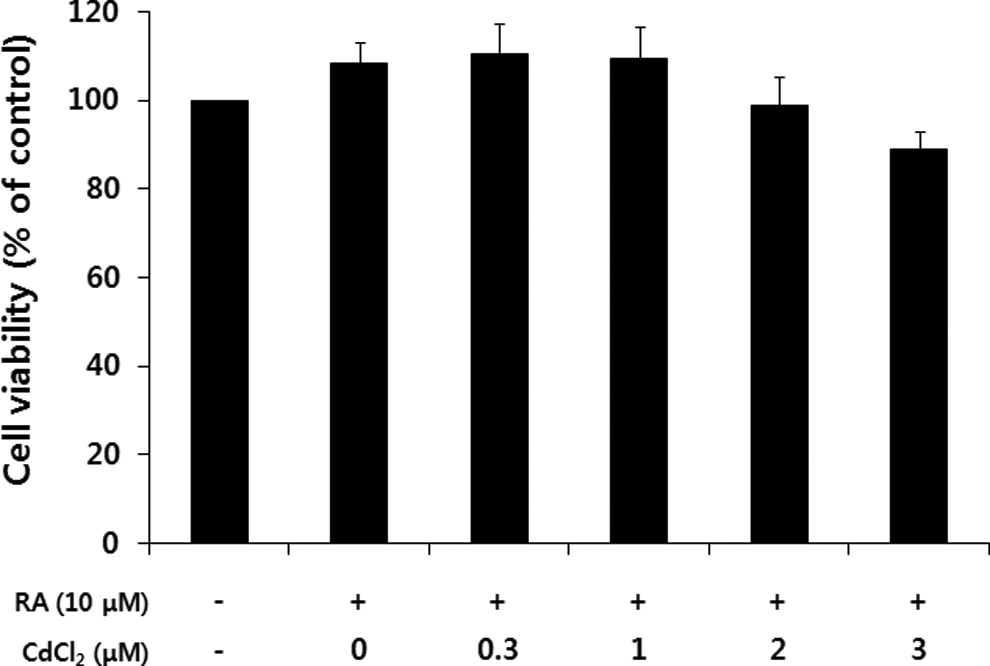
Cell viability of SH-SY5Y after exposure to cadmium for a 48-hour exposure in the methylthiazoltetrazolium (MTT)assay. The cells were treated with cadmium for 48 hours in the presence of retinoic acid (RA; 10 μmol/L). Then, the viability of SH-SY5Y cells was determined by the MTT assay. The percentage of viable cells was calculated by defining the viability of cells without RA and cadmium treatment as 100%. Data are expressed as the mean ± standard error obtained from 4 independent experiments.
Effects of Cadmium on Neurite Outgrowth in Differentiating SH-SY5Y Cells
The neurotoxicity of cadmium on differentiating SH-SY5Y cells was initially assessed by examining its ability to inhibit neurite outgrowth. Cadmium was used at various concentrations up to 3 μmol/L, which had no significant effect on the viability of differentiating SH-SY5Y cells. Although cadmium had no significant effect at low concentrations (<1 μmol/L) on neurite outgrowth of differentiating SH-SY5Y cells, the neurite outgrowth of differentiating SH-SY5Y cells at concentrations of 1 to 3 μmol/L cadmium was significantly inhibited in a dose-dependent manner (Figure 2).
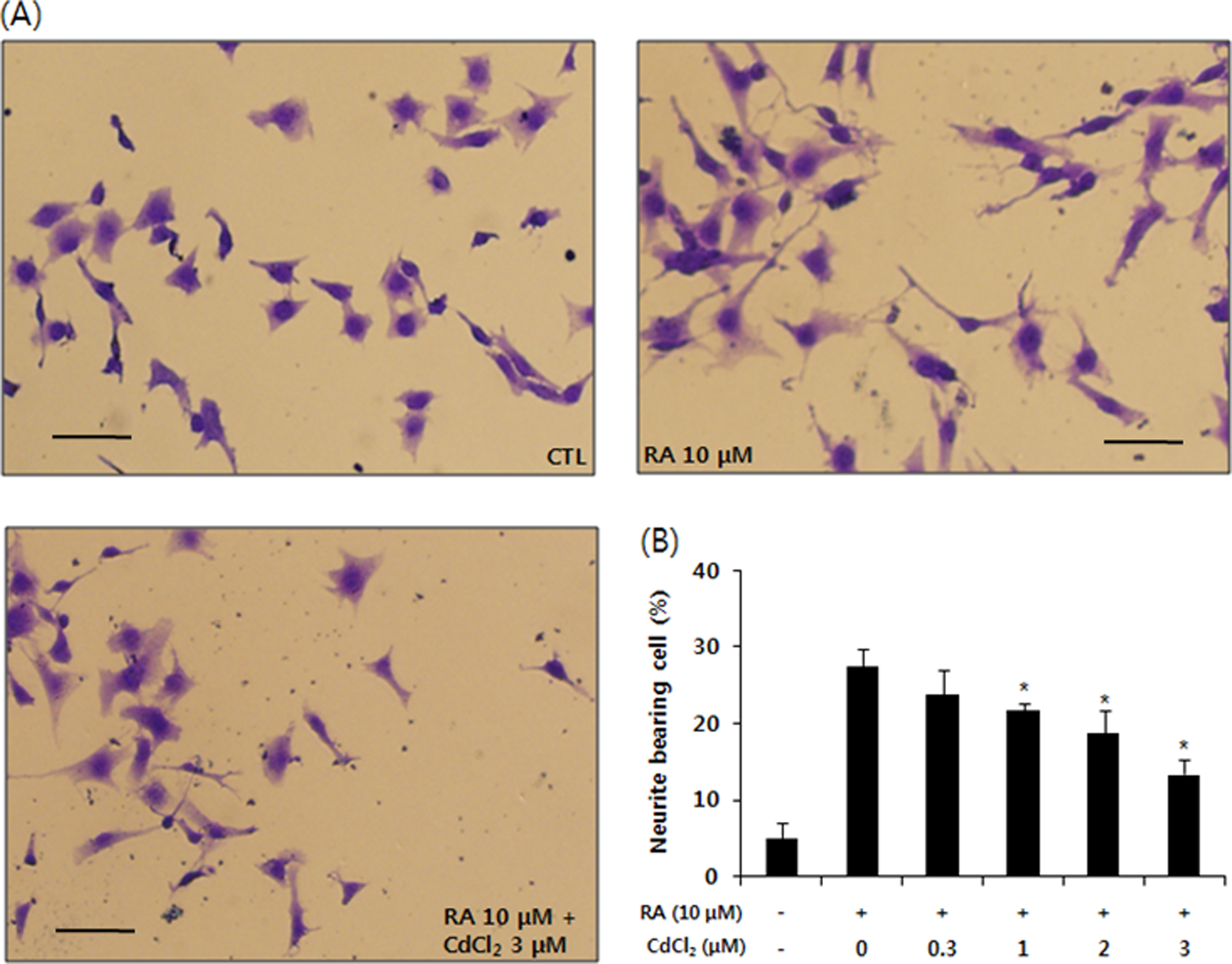
Cadmium inhibits retinoic acid (RA)-induced neurite outgrowth in SH-SY5Y cells. A, The cells were induced to differentiate for 48 hours with RA (10 μmol/L) in the presence or absence of cadmium (0.3 ∼ 3 μmol/L). Cells were fixed in 4% paraformaldehyde and stained with Coomassie Blue. Shown are images of typical fields of cells viewed by inverted light microscope. B, Neurite outgrowth was quantified by counting the number of cells exhibiting neurites that were 1.5 times longer than the cell body diameter in length. The proportion of cells with neurites was expressed as a percentage of the total number of cells. Approximately 200 cells were counted in each medium condition. The data are expressed as the mean ± standard error of 4 independent experiments. *
Effects of Cadmium on the Expression of GAP-43 Protein
Since GAP-43 is a neural-specific protein that is thought to play a role in neurite outgrowth, GAP-43 is used as a common indicator for RA-induced differentiation of SH-SY5Y cells. 26 In order to determine the effects of cadmium on expression of GAP-43 in differentiating SH-SY5Y cells, the cells were treated with 10 μmol/L RA and various concentrations of cadmium for 48 hours. As shown in Figure 3, cadmium-treated cell extracts exhibited decreased cross-reactivities with the GAP-43 antibody when compared to control extracts in a dose-dependent manner (Figure 3).
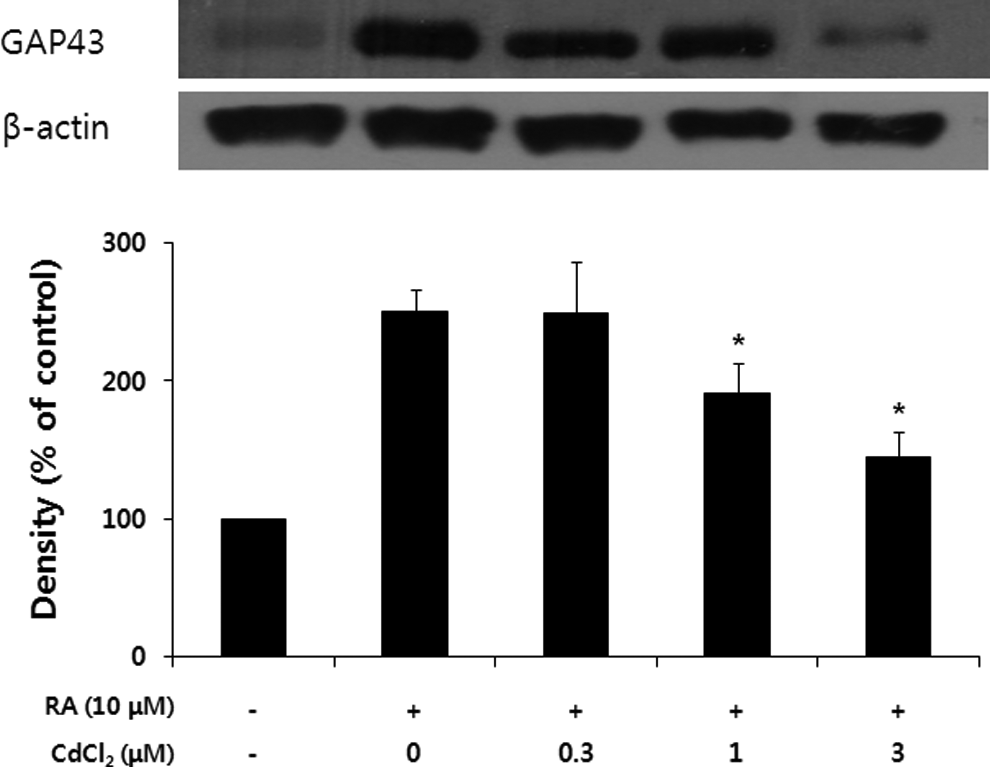
Cadmium inhibits the expression of 43-kDa growth-associated protein (GAP-43) in differentiating SH-SY-5Y cells. The cells were treated with various concentrations of cadmium for 48 hours. The Western blot data represent 1 of three 3 experiments. Densitometric analysis was performed to determine the intensity of GAP43 bands. Values are normalized to actin band and expressed as the percentage of cells treated with retinoic acid (RA) only. Data are mean ± standard error of 3 independent experiments.
Effects of NAC on the Expression of GAP-43 Proteins in Cadmium-Treated Cells
The degree of ROS generation in the cells was measured using the fluorescent dye DCF-DA. Figure 4 shows the fluorescence images obtained using a fluorescent spectrophotometer. An increase in fluorescence was observed in cells treated with cadmium (3 μmol/L), which was reduced in the presence of NAC in a concentration-dependent manner. These results demonstrated that NAC reduced ROS generation using cadmium-treated SH-SY5Y cells as free radical scavenger. As shown in Figure 5, NAC recovered the expression of GAP-43 in cadmium-treated cells.
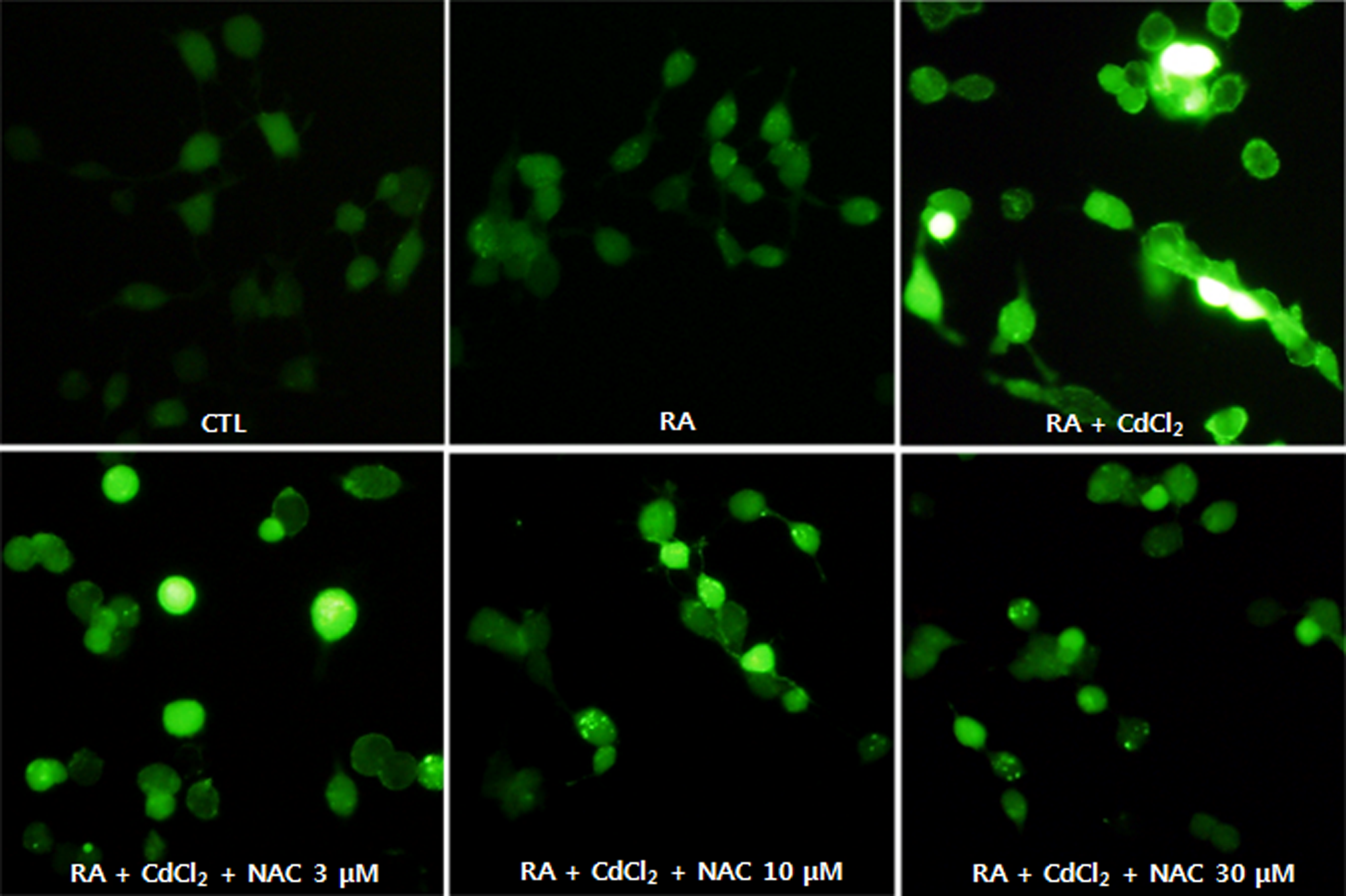
Effect of N-acetyl-
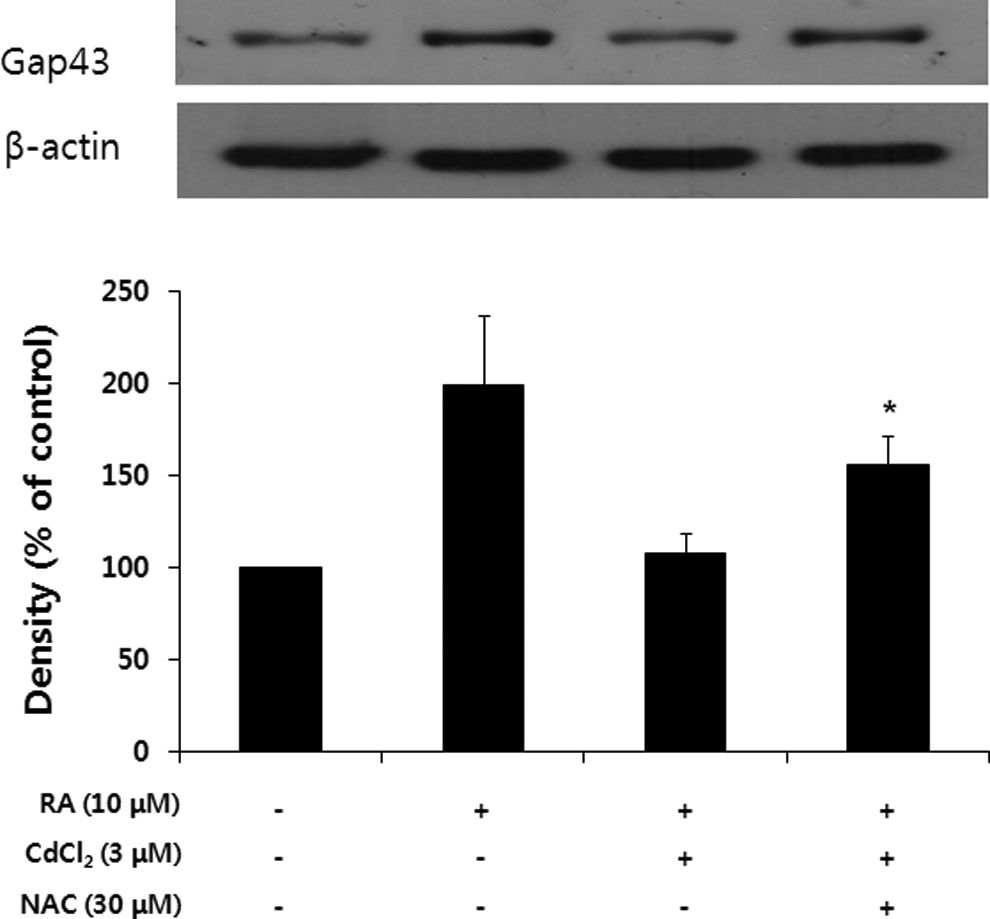
Effect of N-acetyl-
In summary, cadmium inhibited neurite outgrowth in differentiating SH-SY5Y cells at a sublethal concentration (<3 μmol/L). Moreover, Western blot analysis of differentiating SH-SY5Y cells treated with cadmium (1-3 μmol/L) showed a decreased expression level of GAP-43 protein in a dose-dependent manner. Taken together, these discoveries suggested that the reduced level of GAP-43 was associated with the inhibition of neurite outgrowth in differentiating SH-SY5Y cells by cadmium. Also, the involvement of ROS in cadmium-induced inhibition of neurite outgrowth in differentiating SH-SY5Y was suggested.
Discussion
Any changes in neurite outgrowth in differentiating neurons can be used for the elucidation of neuroregenerative potential.
13
A number of human neuroblastoma cell lines including IMR-32, SH-SY5Y, and SK-N-SH cells have been used to evaluate the effects of toxicants on neurite outgrowth. Human SH-SY5Y neuroblastoma cells can be differentiated into neuron-like cells with neurites in response to all-
It is well known that ROS plays a key role in the regulation of signal transduction processes leading to cell proliferation, differentiation, and death. 29,30 The membrane-bound enzymatic complex NADPH oxidase (NOX) generates a superoxide anion, which is the major source of intracellular ROS. The NOX-derived ROS can also be produced by bacterial killing in phagocytic cells, 31 with signaling purpose in cell differentiation. 32,33 It has been demonstrated that NOX-derived ROS are involved in neuronal signaling, contributing to neurite formation. 34,35 The PKC delta and NOX also play important roles in SH-SY5Y neuroblastoma cell differentiation induced by RA, pointing out the role of ROS generation in signal transduction in neuronal cells. 36 However, neurons are known to be particularly susceptible to ROS-induced cell damage due to their high rate of oxygen consumption and oxidative phosphorylation.
It is well documented that the nervous system is also subjected to cadmium toxicity. However, the effect of cadmium on neuronal differentiation is not clear. We have shown that cadmium inhibits neurite outgrowth of differentiating human SH-SY5Y neuroblastoma cells, confirmed by reduced levels of expression of GAP-43 proteins.
The increasing evidence suggests that cadmium disrupts a variety of cellular signaling cascades, which are responsible for cell’s survival, proliferation, and differentiation. Cadmium can induce more or less permanent changes in the levels of second messengers, which disrupt the cell’s physiological functions, affect gene transcription and regulation, and induce cell death. 37 Although cadmium is not considered a Fenton metal, 38 it is capable of increasing the generation of ROS by depleting cellular glutathione and antioxidant enzymes. 39,40 Oxidative stress by cadmium-induced ROS generation is believed to play a major role in apoptosis and aberrant gene expression. 41 Many of the cellular signaling pathways taking place in cells treated with cadmium are mediated directly or indirectly by ROS.
In conclusion, our results showed, for the first time, the ability of cadmium to inhibit neurite outgrowth in differentiating SH-SY5Y cells. The GAP-43 protein, which is thought to play a role in neurite outgrowth, was downregulated in cadmium-treated differentiating SH-SY5Y cells. It was suggested that ROS might be involved in the inhibition of neurite outgrowth in differentiating SH-SY5Y cells by cadmium. Further studies are also required to elucidate the molecular mechanisms for the inhibition of neurite outgrowth in differentiating SH-SY5Y cells.
Footnotes
Author Contribution
Byung Sun Yoo participated in the design and/or interpretation of the reported experiments or results, in drafting and/or revising the manuscript, and provided administrative, technical, or supervisory support. Eun Joo Pak (Figures 3, 4, and 5) and Gi Dong Son (Figures 1 and ![]() ) participated in the acquisition and/or analysis of data.
) participated in the acquisition and/or analysis of data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Kyonggi University Research Grant 2011.
