Abstract
Intraoperative acute pulmonary embolism (PE) is a rare and life-threatening event with symptoms such as pleuritic chest pain, shortness of breath, and anxiety, which are easily masked by general anesthesia. To diagnose PE in a patient under general anesthesia, anesthesiologists must rely on alternative data points such as hypoxia, decreased end-tidal carbon dioxide (ETCO2), tachycardia, electrocardiogram changes, and intraoperative echocardiography. We present a case of acute intraoperative massive PE in a patient undergoing posterior spinal fusion. We discuss the management of acute perioperative PE, focusing on surgical risk, bleeding potential with thrombolytic therapies and anticoagulation, recent advances in catheter-based therapies, and the role of Pulmonary Embolism Response Teams (PERTs) in the assessment and perioperative management of high-risk patients.
Keywords
Introduction
Acute intraoperative pulmonary embolism (PE) is a rare life-threatening event that provides a unique diagnostic challenge for the anesthesiologist, as traditional patient-expressed symptomatology is masked by general anesthesia. Furthermore, its management must balance bleeding potential and ongoing surgical risk in the setting of potential thrombolytic and anti-thrombotic therapies. Pulmonary embolism response teams (PERTs) serve as a multidisciplinary response to streamline management of high-risk patients who have acute hemodynamic compromise secondary to a PE. In the era of increasing treatment complexity, PERTs have been proven to provide the most benefit to those whose disease processes are complicated by conflicting physiology and therefore necessitate comprehensive case review and intricate decision-making. We present a case of intraoperative acute high-risk PE following induction of general anesthesia in a patient undergoing urgent posterior spinal fusion and the successful multidisciplinary management of this case through the utilization of an institutional PERT.
HIPAA authorization was obtained. This manuscript adheres to the EQUATOR Network’s CARE Guidelines.
Case Description
A 47-year-old male with past medical history significant for a prior traumatic brain injury, hypertension, and chronic kidney disease presented with bilateral lower extremity weakness following a ground level fall. After the fall, approximately 48 hours elapsed before the emergency medical team arrived to assist the patient, as he was unable to arise on his own. Upon hospital evaluation, he was diagnosed with an acute vertebral body burst fracture at L1 complicated by spinal canal stenosis. He was urgently taken to the operating room for a multilevel posterior spinal fusion. The patient underwent induction of general anesthesia with lidocaine, propofol, methadone, ketamine, and succinylcholine, followed by endotracheal intubation. Prior to surgical incision, approximately 15 min after induction, the patient unexpectedly required multiple boluses of phenylephrine to maintain an appropriate mean arterial pressure. After a brief episode of ventilator dyssynchrony, the patient experienced acute decompensation with worsening hypoxia and hypotension. An acute decrease in ETCO2 from 28 mmHg to 3 mmHg was noted. The patient failed to respond to additional doses of vasoactive medications and hand ventilation with 100% FiO2, progressing to pulseless electrical activity (PEA). He underwent 8 minutes of cardiopulmonary resuscitation. Once return of spontaneous circulation was achieved, an epinephrine infusion was required to maintain adequate mean arterial pressure. However, due to ongoing hypotension and poor oxygenation saturation, the cardiothoracic anesthesiology team was consulted for transesophageal echocardiography (TEE).
TEE was notable for large clot burden in the main pulmonary artery (Supplemental Video Files 1 and 2) complicated by a severely dilated right atrium resulting in leftward bowing of the interatrial septum (Supplemental Video File 3). Severe right ventricular (RV) dilation, severely reduced RV function and intraventricular septal flattening were also noted (Supplemental File 4). With the support of echocardiography, the diagnosis of acute high-risk PE was made, and the case was canceled. The patient was started on a heparin infusion and taken to the intensive care unit due to the persistent requirement of norepinephrine and epinephrine infusions.
Once in the intensive care unit, the patient was evaluated by our institutional PERT team, which consists of cardiologists, cardiac surgeons, intensivists, and extracorporeal membrane oxygenation (ECMO) specialists. The diagnosis of acute PE was once again demonstrated with a CT angiogram of the chest. Imaging was notable for extensive acute central occlusive and nonocclusive filling defects bilaterally with a large saddle embolus (Figure 1). CT imaging was also notable for right heart dilation and interventricular septum flattening consistent with RV dysfunction. Of note, the patient received lower extremity Doppler studies upon initial presentation to the hospital which were negative for deep vein thrombosis. CT Angiogram Demonstrating a Large Filling Defect in the Main Pulmonary Artery Consistent With a Saddle Embolus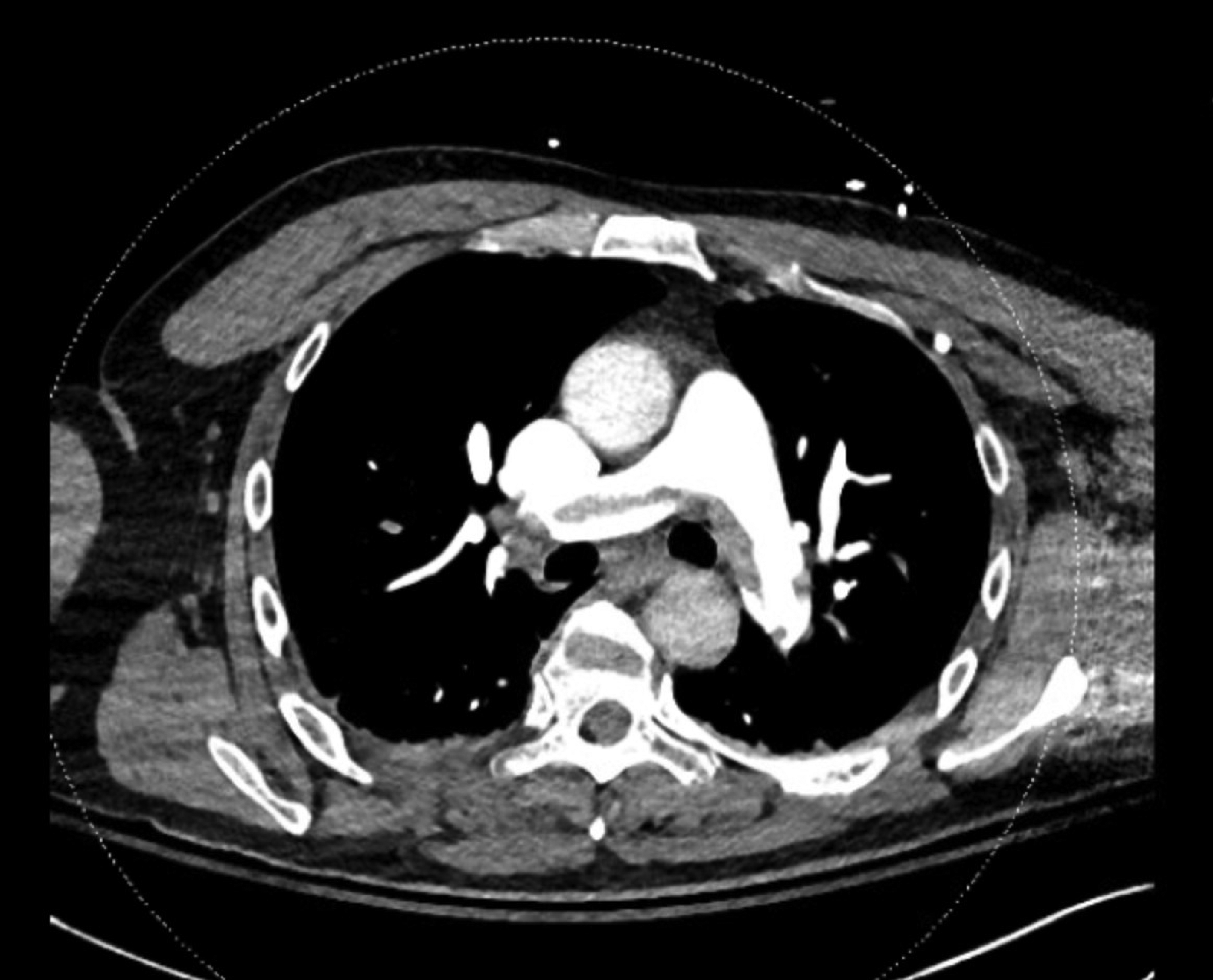
Ongoing discussion between the multidisciplinary PERT team determined that the patient was a candidate for surgical embolectomy and catheter-based therapies (CBT). However, both management techniques were incredibly high risk for clot propagation secondary to mechanical manipulation with CBT or spinal cord hemorrhage secondary to systemic or localized thrombolytics. It was further discussed with the patient’s family that he would be at high risk for a complicated and prolonged postoperative course should he undergo surgical embolectomy. Additionally, at the time of discussion, the patient’s requirement for hemodynamic support with inotropic agents and vasopressors was decreasing, suggesting—however uncertain—that the patient may regain cardiac function with systemic anticoagulation alone. The PERT team addressed the patient’s high surgical risk and risk of paraplegia if he were to undergo cardiopulmonary bypass and/or systemic thrombolytic administration with known traumatic spinal canal stenosis in the setting of his L1 burst fracture. The patient’s family noted that he would prefer to remain as functionally independent as possible and therefore, elected for continued management with a heparin infusion to eliminate surgical risk and mitigate risk of spinal cord hemorrhage.
A transthoracic echocardiogram obtained 2 days following the patient’s initial arrest was notable for a newly reduced ejection fraction of 36%, global left ventricular hypokinesis, and normal right ventricular size and function. The newly reduced left ventricular function was an unexpected clinical finding, especially as RV function had normalized, and the patient did not have a history of coronary artery disease or heart failure. We speculate that this may have been the result of stress-induced cardiomyopathy, as the patient ultimately had full recovery of left ventricular function.
With normalization of his right heart function, and lack of vasopressor and inotropic support, the patient was deemed appropriate by the intensivists and primary surgeon to return to the operating room for a posterior spinal fusion approximately 3 days following his initial procedure. He underwent an uneventful posterior spinal fusion 3 days after his initial arrest. He was successfully extubated on postoperative day 1, 4 days after his initial arrest. The patient was discharged to a skilled nursing facility on postoperative day 27.
Discussion
A high-risk (formerly “massive”) PE is defined by the presence of any of the following: hypotension (systolic blood pressure ≤90 mmHg), cardiovascular collapse, occlusion of >50% of the pulmonary artery, or persistent inotrope requirement. 1 Given the high morbidity and mortality associated with acute high-risk PE, early recognition, diagnosis, and treatment stratification are paramount. In the presented case, a multidisciplinary PERT team was instrumental in the evaluation of the risks and benefits of a multitude of therapeutic options including medical management with anticoagulation, systemic thrombolytics, surgical thrombectomy, or catheter-based therapies.
Catheter-based therapy (CBT) has become the treatment of choice for patients with ongoing hemodynamic instability. A variety of CBTs exist, including aspiration thrombectomy as well as catheter-directed thrombolysis (CDT) with or without ultrasound assistance. Aspiration thrombectomy does not require thrombolysis, whereas CDT utilizes only a fraction of thrombolytics used in systemic administration. The distinct advantage of CBTs is the decreased time to reduction in RV afterload and, therefore, improved RV function compared with heparin infusion alone and without the risk of a major surgical procedure. 2 Although increasing in popularity, CBTs are not without risk. A meta-analysis by Avgerinos and colleagues demonstrated a major bleeding risk (defined as an intracranial bleed, or any bleeding requiring transfusion or intervention) of 6.7% in patients with high-risk PEs who underwent CDT with locally administered thrombolytics. 2 Conclusions on the safety and efficacy of CBT are difficult to draw, as studies are plagued by conflicting outcomes, heterogeneity of patients and PE severity, and limited population size. Multiple studies have demonstrated significant RV size reduction and improved RV to LV ratio at 24 hours and 90 days compared with heparin infusion alone, demonstrating—at minimum—echocardiographic findings that would suggest functional improvement acutely and long term, although this has yet to be correlated with clinical outcomes. 3 However, given a lack of consensus regarding the appropriate patient population in combination with the need for institutional expertise, CBT for PE should be used only under the recommendation of expert consultants.
In contrast, patients may also undergo systemic thrombolysis. However, current clinical evidence does not support the use of thrombolytics in intermediate- to high-risk PE. 4 Moreover, systemic thrombolytics are contraindicated in a plethora of clinical scenarios, which substantially limits their utility in complex cases. In such extreme cases, where the patient exists as an outlier in medical literature, careful consideration of how to apply known contraindications to therapy should be left to a collaborative team of experts.
Surgical embolectomy may serve as a treatment modality for patients with high-risk PEs. Indications for surgical embolectomy include high-risk or intermediate- to high-risk PE with a contraindication to systemic thrombolytic therapy, failed thrombolytic therapy, cardiogenic shock, or hemodynamic instability. Unfortunately, surgical embolectomy is plagued by a high percentage of postoperative complications, with in-hospital mortality rates as high as 11.7%. 1 Additionally, patients who suffer a preoperative cardiac arrest are at even higher risk of mortality—44% compared with 8% in patients who did not present in cardiogenic shock and 24% in patients who presented in cardiogenic shock without requiring cardiopulmonary resuscitation. 5 Finally, the 2019 European Society of Cardiology’s guidelines for management of acute PE mention utilization of ECMO as a bridge to recovery or as an adjunct for surgical embolectomy. 6 However, high-quality evidence such as randomized controlled trials to support the use of ECMO during acute PE is substantially limited, subsequently confining current evidence for this clinical scenario to animal models and small case series.7,8
PERTs are institutionally dependent multidisciplinary teams that function to assess treatment appropriateness, as well as streamline and facilitate management of high-risk acute PE patients. PERTs often consist of an intensivist, a cardiologist/cardiac surgeon, and an interventional radiologist/vascular surgeon.9,10 When a PERT activation occurs at our institution, an additional alert is sent to notify the cardiac anesthesiology and ECMO teams (Figure 2). Schematic Representation of Workflow During Pulmonary Embolism Response Team Activation. PERT Indicates Pulmonary Embolism Response Team; CVICU, Cardiovascular Intensive Care Unit; ECMO, Extracorporeal Membrane Oxygenation; TPA, Tissue Plasminogen Activator (Alteplase)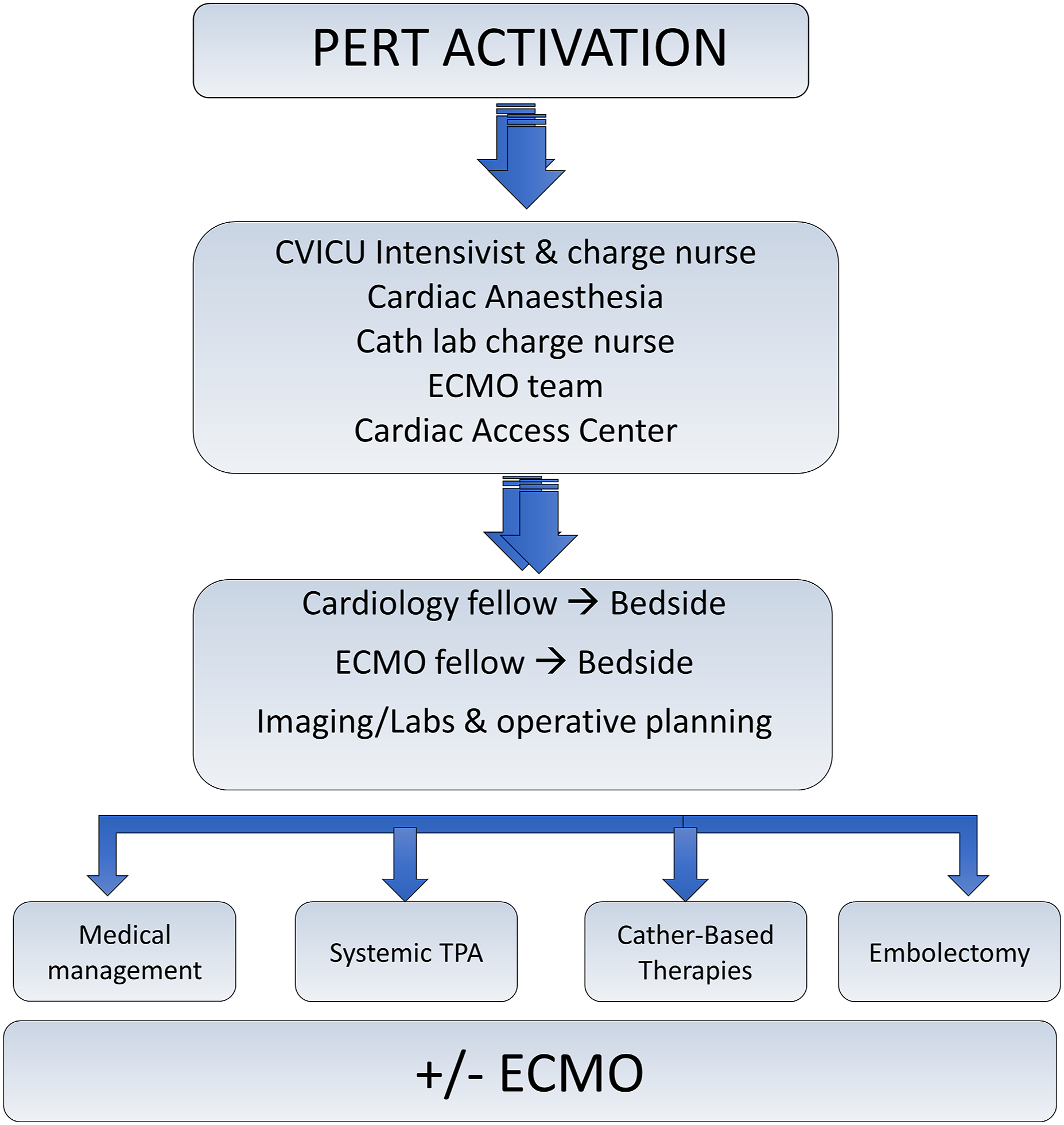
The cardiac anesthesiology team functions to provide cardiac imaging expertise or serve as an avenue for mobilization of operating room resources. PERT implementation has increased in the previous decade as substantial evidence has emerged about their ability to utilize advanced therapies more effectively compared with the pre-PERT era. Patients evaluated by PERTs undergo higher rates of CBTs compared with the pre-PERT era (6.4% vs 3%). 9 This is thought to be a result of improved/increased patient selection and expedited execution of a treatment plan secondary to the expertise provided by PERTs. However, higher utilization of CBT may also coincide not only with the presence of PERT teams but also with growing literature around CBT advancements and the presence of more centers with CBT expertise. As these two variables are highly intertwined, studies have failed to demonstrate the degree to which PERT implementation alone influences patient outcomes. Nonetheless, the literature consistently shows that PERTs benefit patients most by providing a group of highly specialized individuals to manage an acute and rapidly evolving crisis.
To date, there is no literature investigating the conceptualization of perioperative PERTs. Patients who experience an intraoperative PE—while a niche subset—are at profound risk and require specific consideration. While high-risk pulmonary embolisms rarely occur intraoperatively, it is important to recognize that anesthesiologists will continue to provide progressively complex anesthetics for an increasingly sick patient population. The anesthesiologist offers a unique skill set to appropriately lead management of an acute intraoperative PE; however, consideration should be given to what role a PERT activation has perioperatively. Patients who suffer intraoperative PEs may greatly benefit from the detailed and nuanced decision-making of a PERT, given the complex physiologic mechanisms coupled with ongoing surgical risk.
Our patient’s traumatic spinal cord injury represents a direct contraindication to systemic thrombolytic therapy, and his preoperative cardiac arrest would place him at immensely high risk for perioperative complications if surgical embolectomy were pursued. Additionally, in noncardiac operating rooms, special consideration must be given to the number of resources that must be mobilized to care effectively for a patient suffering from an intraoperative PE. Our case demonstrates an expeditious evaluation that may not have occurred if our institutional PERT team did not exist, as the patient was immediately evaluated by cardiology and cardiac surgery once he was moved to an intensive care unit. In some cases, it may be appropriate for consulting teams to evaluate the patient while in the operating room, which further highlights the utility of a perioperative PERT team. Ideally, the team should include TEE-trained anesthesiologists if the appropriate resources are available at the institution. The intraoperative management of an acute PE falls directly on the anesthesiologist. Their role is not limited to managing hemodynamic instability with inotropic and vasopressor support; they can also streamline diagnosis with advanced imaging modalities (such as TEE), facilitate a discussion amongst the primary surgical team and consulting teams such as cardiac surgery and cardiology, and facilitate transport to a higher level of care, such as the catheterization lab for CBT, cardiac operating room for ECMO, or an intensive care unit for further diagnosis and treatment stratification. As such, an anesthesiologist is especially positioned to respond to intraoperative PERT alerts secondary to their mastery of acute hemodynamic compromise, expertise of operating room logistics, and facilitation of expedited transportation, all while the patient is under the direct care of a highly specialized physician.
Finally, our case highlights the role the anesthesiologist plays when managing conflicting medical conditions and notes the potential expansion of that role with the development of a perioperative PERT team, while also calling into focus a niche patient population that may greatly benefit from the multidisciplinary composition of such a team. The availability of multiple specialists within our institution’s PERT allowed the patient’s family to fully explore all available treatment options and ultimately select a treatment plan that was most reflective of the patient’s desires. Moreover, deepening the anesthesiologist’s knowledge of advancements in PE treatments is crucial to maintaining our role as perioperative leaders who can safely, quickly, and effectively manage potentially disastrous perioperative complications.
Supplemental Material
Supplemental Material
Supplemental Material
Supplemental Material
Footnotes
Acknowledgments
The authors thank Martha Tanner and Yvonne Poindexter (Editors, Department of Anesthesiology, Vanderbilt University Medical Center, Nashville, TN), for editorial contributions to the article.
Ethical Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Consent to Participate
The authors have secured a written informed consent statement from the patient. This statement includes consent for publication.
Author Contributions
Kelsei P. Keene, MD: This author helped draft and edit the manuscript and is accountable for the integrity of the published work. Pete P. Fong, MD: This author helped draft and edit the manuscript and is accountable for the integrity of the published work. John M. Trahanas, MD: This author helped draft and edit the manuscript and is accountable for the integrity of the published work. Bantayehu Sileshi, MD: This author helped draft and edit the manuscript and is accountable for the integrity of the published work.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
