Abstract
Neurotransmission mediated by acetylcholine receptors (AChRs) plays an important role in learning and memory functions in the hippocampus. Impairment of the cholinergic system contributes to Alzheimer’s disease (AD), indicating the importance of AChRs as drug targets for AD. To improve the success rates for AD drug development, human cell models that mimic the target brain region are important. Therefore, we characterized the functional expression of nicotinic and muscarinic AChRs (nAChRs and mAChRs, respectively) in human hippocampal neurons differentiated from hippocampal neural stem/progenitor cells (HIP-009 cells). Intracellular calcium flux in 4-week differentiated HIP-009 cells demonstrated that the cells responded to acetylcholine, nicotine, and muscarine in a concentration-dependent manner (EC50 = 13.4 ± 0.5, 6.0 ± 0.4, and 35.0 ± 2.5 µM, respectively). In addition, assays using subtype-selective compounds revealed that major AD therapeutic target AChR subtypes—α7 and α4β2 nAChRs, as well as M1 and M3 mAChRs—were expressed in the cells. Furthermore, neuronal network analysis demonstrated that potentiation of M3 mAChRs inhibits the spontaneous firing of HIP-009 neurons. These results indicate that HIP-009 cells are physiologically relevant for AD drug screening and hence are loadstars for the establishment of in vitro AD models.
Keywords
Introduction
The use of human cell models that mimic physiological conditions, pathophysiological conditions, or both is a key approach to increase success rates in drug development. Following the generation of embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs), as well as the development of neural differentiation methods from these cells, this strategy has been applied to central nervous system (CNS) drug development. 1 However, methods of brain region–specific neural differentiation from human ESCs/iPSCs are not fully developed. In particular, few reports have addressed hippocampal cell differentiation from human ESCs/iPSCs.2,3 In addition, the differentiation protocol includes complicated steps, which must be overcome to apply this method to drug screening.
The hippocampus is an indispensable brain region that plays essential roles in learning and memory functions in the CNS. Acetylcholine (ACh) is an important neurotransmitter implicated in hippocampal functions. Cholinergic neurons in the medial septal nucleus and vertical limb nucleus of the diagonal band project to the hippocampus, 4 where released ACh binds to acetylcholine receptors (AChRs). Because dysfunction of the ACh system is involved in Alzheimer’s disease (AD), AChRs have been targets of AD drug development.5,6 Therefore, an easy-to-handle in vitro human hippocampal cell model expressing functional AChRs would accelerate our understanding of the mechanisms underlying AD and lead to the development of novel therapeutic agents.
AChRs are classified as nicotinic (nAChRs) or muscarinic (mAChRs). Neuronal nAChRs are expressed throughout the CNS and comprise either α (α2–α10) or a combination of α and β (β2–β4) subunits. 7 In the hippocampus, the predominant nAChR subtypes are α7 nAChRs and α4β2 nAChRs. 8 In the case of mAChR subtypes, M1 mAChRs are the most abundant, whereas M5 mAChRs are the least prevalent throughout the brain. 9 In the hippocampus and neocortex, M1 and M3 mAChRs are present at high and moderate levels, respectively. Therefore, in this study, we focused on α7 and α4β2 nAChRs, as well as M1 and M3 mAChRs, all of which are expressed in the hippocampus and are key targets for AD drug development.5,10,11
We previously reported that human hippocampus-derived neural stem/progenitor cells (HIP-009 cells) can be simultaneously differentiated into a population of neurons and astrocytes by use of a simple differentiation protocol and that the gene expression profile of the differentiated HIP-009 population is similar to that of the human hippocampus. 12 We also demonstrated that HIP-009 neurons express multiple functional glutamate receptors and form neuronal networks, indicating that they have the fundamental characteristics of functional neurons.12,13 By exploiting hippocampus-derived features of HIP-009 cells, here we characterized the functional expression of α7 and α4β2 nAChRs as well as M1 and M3 mAChRs in differentiated HIP-009 cells to reveal the potential application of these cells to AD drug screening. In addition, we investigated their roles in neuronal network functions, which are essential to cognition, learning, and memory.
Materials and Methods
Chemicals
ACh, methyllycaconitine citrate (MLA), and 1-(4-methoxybenzyl)-4-oxo-1,4-dihydroquinoline-3-carboxylic acid (BQCA) were purchased from Sigma-Aldrich (St. Louis, MO). (–)-Nicotine, mecamylamine hydrochloride, and tetrodotoxin (TTX) were obtained from Wako (Osaka, Japan). NS 9283, (±)-epibatidine, 4-(4-bromophenyl)-3a,4,5,9b-tetrahydro-3H-cyclopenta[c]quinolone-8-sulfonamide (4BP-TQS), scopolamine hydrobromide, and dihydro-β-erythroidine hydrobromide (DHβE) were purchased from Tocris Bioscience (Bristol, UK). VU 0255035 was obtained from Abcam (Cambridge, UK). (+)-Muscarine iodide was purchased from Toronto Research Chemicals (Toronto, Canada). 4-Diphenylacetoxy-N-methyl-piperidine methiodide (4-DAMP) was obtained from Cayman Chemical (Ann Arbor, MI). ACh, MLA, (–)-nicotine, mecamylamine hydrochloride, DHβE, (+)-muscarine iodide, scopolamine hydrobromide, and TTX were dissolved in distilled water (Thermo Fisher Scientific, Waltham, MA). 4BP-TQS, NS 9283, (±)-epibatidine, BQCA, VU 0255035, and 4-DAMP were dissolved in DMSO (Wako).
Cell Culture
All experiments were approved by the Eisai Research Ethics Committee (approval numbers, 2011-0076, 2012-0076, 2013-0076, and 2014-0076). HIP-009 cells were purchased from GigaCyte (Branford, CT; now PhoenixSongs Biologicals, Branford, CT). The cells were expanded and differentiated according to the manufacturer’s instructions. The 100-mm dishes were coated with 10 µg/mL laminin (Sigma-Aldrich) in phosphate-buffered saline (PBS) without calcium and magnesium (Wako). The cells were seeded and grown on these dishes in Neural StemCell Growth Medium (PhoenixSongs Biologicals) supplemented with growth medium supplement cocktail (Neural StemCell Growth Supplement, 10 ng/mL basic fibroblast growth factor [bFGF], 20 ng/mL epidermal growth factor [EGF], 3 µM CHIR-99021, and 1 µg/mL laminin) and 30 µg/mL gentamicin (Thermo Fisher Scientific). The medium was changed every 2 days, and confluent cells were expanded every 4 or 5 days. HIP-009 cells expanded for eight passages were used in this study. Before neural differentiation, expanded HIP-009 cells were harvested and cultured in Neural Transition Medium (PhoenixSongs Biologicals) supplemented with transition medium supplement cocktail (Neural Transition Supplement, 10 ng/mL bFGF, 20 ng/mL EGF, and 1 µg/mL laminin) and 30 µg/mL gentamicin for 3 days as a predifferentiation process. The HIP-009 cell expansion and predifferentiation processes were carried out at 37 °C in a humidified atmosphere of 2% O2, balanced with N2, and 6% CO2.
For quantitative reverse transcription polymerase chain reaction (qRT-PCR) analysis, HIP-009 cells that had undergone predifferentiation as described above were plated on 24-well plates precoated with 50 µg/mL poly-D-lysine (PDL; Sigma-Aldrich) and 10 µg/mL laminin at 2 × 105 cells/well in Neural Differentiation Medium (PhoenixSongs Biologicals) supplemented with differentiation medium supplement cocktail (Neural Differentiation Supplement, 20 ng/mL brain-derived neurotropic factor, 20 ng/mL glial cell–derived neurotropic factor, and 1 µg/mL laminin) and 30 µg/mL gentamicin (hereafter referred to as differentiation medium). They were then differentiated into neural cells for 1 day or 4 weeks in a humidified incubator with an atmosphere of 2% O2 and 6% CO2 at 37 °C. During the differentiation process, half of the medium was changed twice a week.
Similarly, for fluorescence-based intracellular calcium flux assays, 3.7 × 104 predifferentiated HIP-009 cells were plated on black-walled, clear-bottomed, poly-D-lysine (PDL)–coated 96-well plates (Corning, Corning, NY) in the differentiation medium, and the differentiation process was carried out for 1 day or 4 weeks. During the process, half of the medium was changed twice a week.
For multielectrode array (MEA) recordings, 2 × 105 predifferentiated HIP-009 cells were plated onto the center of MEA chips (Alpha MED Scientific, Osaka, Japan) precoated with polyethyleneimine (0.1%; Sigma-Aldrich) and laminin (10 µg/mL). To form functional neuronal networks, the cells were differentiated for 8 weeks until spontaneous firing was observed throughout the electrodes. During the differentiation process, half of the differentiation medium was changed twice a week.
qRT-PCR Analysis
For qRT-PCR analysis, RNAs were isolated by using an RNeasy Micro Kit (Qiagen, Venlo, Netherlands), treated with DNase (Qiagen), and reverse-transcribed into complementary DNAs (cDNAs) with SuperScript VILO Master Mix (Thermo Fisher Scientific) in accordance with the manufacturer’s instructions. The analyses were performed on an ABI 7900HT System (Thermo Fisher Scientific), using EagleTaq Master Mix with ROX (Roche Diagnostics, Indianapolis, IN) and TaqMan primers and probe sets (Thermo Fisher Scientific). The thermocycling protocol consisted of an initial cycle at 95 °C for 10 min, followed by 40 cycles of PCR at 95 °C for 15 s and 60 °C for 1 min. The messenger RNA (mRNA) level of each sample was normalized against the mRNA expression of glyceraldehyde-3-phosphate dehydrogenase from the same sample, and data were expressed as fold change relative to those of HIP-009 cells differentiated for 1 day.
Fluorescence-Based Intracellular Calcium Flux Assay
After differentiation, the medium was removed from each well, and the differentiated HIP-009 cells were loaded for 1 h at 37 °C with calcium 4 dye (Molecular Devices, Sunnyvale, CA) and probenecid (2.5 mM; Thermo Fisher Scientific) dissolved in an assay buffer. The assay buffer (pH adjusted to 7.4 with NaOH) comprised 20 mM HEPES and Hank’s balanced salt solution with calcium and magnesium without phenol red (Thermo Fisher Scientific). Compounds were diluted in the assay buffer and transferred to compound plates (Thermo Fisher Scientific). After incubation with the dye and probenecid for 1 h, the cells were washed with the assay buffer and loaded with fresh buffer. In the evaluation of each antagonist and positive allosteric modulator (PAM), these compounds were applied to the cells before each stimulation. Intracellular calcium flux was measured with the Functional Drug Screening System 6000 (FDSS 6000; Hamamatsu Photonics, Hamamatsu, Japan), which simultaneously monitors changes in fluorescence in each well of the plate. Baseline fluorescence was recorded for 12 s before the addition of the compound solution, and fluorescent signals were sampled at 0.3-s intervals for 1.75 min in total (excitation wavelength, 480 nm; emission wavelength, 540 nm).
MEA Recordings
Extracellular recordings were performed by using a MED64 System (Alpha MED Scientific). The culture temperature was maintained at 37 °C during the measurements. Signals from all 64 electrodes were sampled simultaneously at 20 kHz and stored in a standard personal computer by using the MED64 Mobius software (Alpha MED Scientific). The threshold for spike detection was 5.5 times the standard deviation of the noise level. To determine the noise level, we applied TTX (100 nM) after evaluation of compounds in each experiment to block the firing of HIP-009 neurons completely.
Statistical Analysis
All data are expressed as means ± standard errors of means (SEMs), and n refers to the number of independent experiments. GraphPad Prism 6.0 (GraphPad Software, San Diego, CA) was used for statistical analysis. EC50 and IC50 values for each compound were determined by a four-parameter logistic curve fitting. Maximum potencies were also calculated by using the same fitting that extrapolated maximum plateaus. Statistical differences between two groups were examined by use of the F test, followed by the two-tailed unpaired Student’s t test (p ≥ 0.05 in the F test) or Welch’s t test (p < 0.05 in the F test). Comparisons of ACh-, nicotine-, or muscarine concentration–induced changes in calcium flux readouts in HIP-009 cells differentiated for 1 day and 4 weeks were made by two-way analysis of variance (ANOVA), followed by Tukey’s multiple-comparison test as a post hoc test. Changes in neuronal network functions induced by the sequential application of compounds were evaluated by a one-way repeated-measures ANOVA. Tukey’s multiple-comparison test was used to examine the differences between two groups. A p value of less than 0.05 was considered statistically significant.
Results
Gene Expression Analysis of Acetylcholine Receptors
To investigate the gene expression profiles of AChRs in HIP-009 cells, we performed qRT-PCR analysis of the cells at differentiation periods of 1 day and 4 weeks. mRNA expression levels of nAChRs (CHRNA4, CHRNA7, and CHRNB2 encoding the α4, α7, and β2 nAChR subunits, respectively; n = 3; Fig. 1A–C ) and mAChRs (CHRM1 and CHRM3 encoding the M1 and M3 mAChR subunits, respectively; n = 3; Fig. 1D , E ) were significantly upregulated in 4-week differentiated HIP-009 cells compared with those of 1-day differentiated cells. RIC3 encodes an acetylcholine receptor chaperone that promotes the functional expression of nAChRs; its mRNA expression levels after 4 weeks of differentiation were also significantly elevated relative to those after 1 day of differentiation (n = 3; Fig. 1F ).
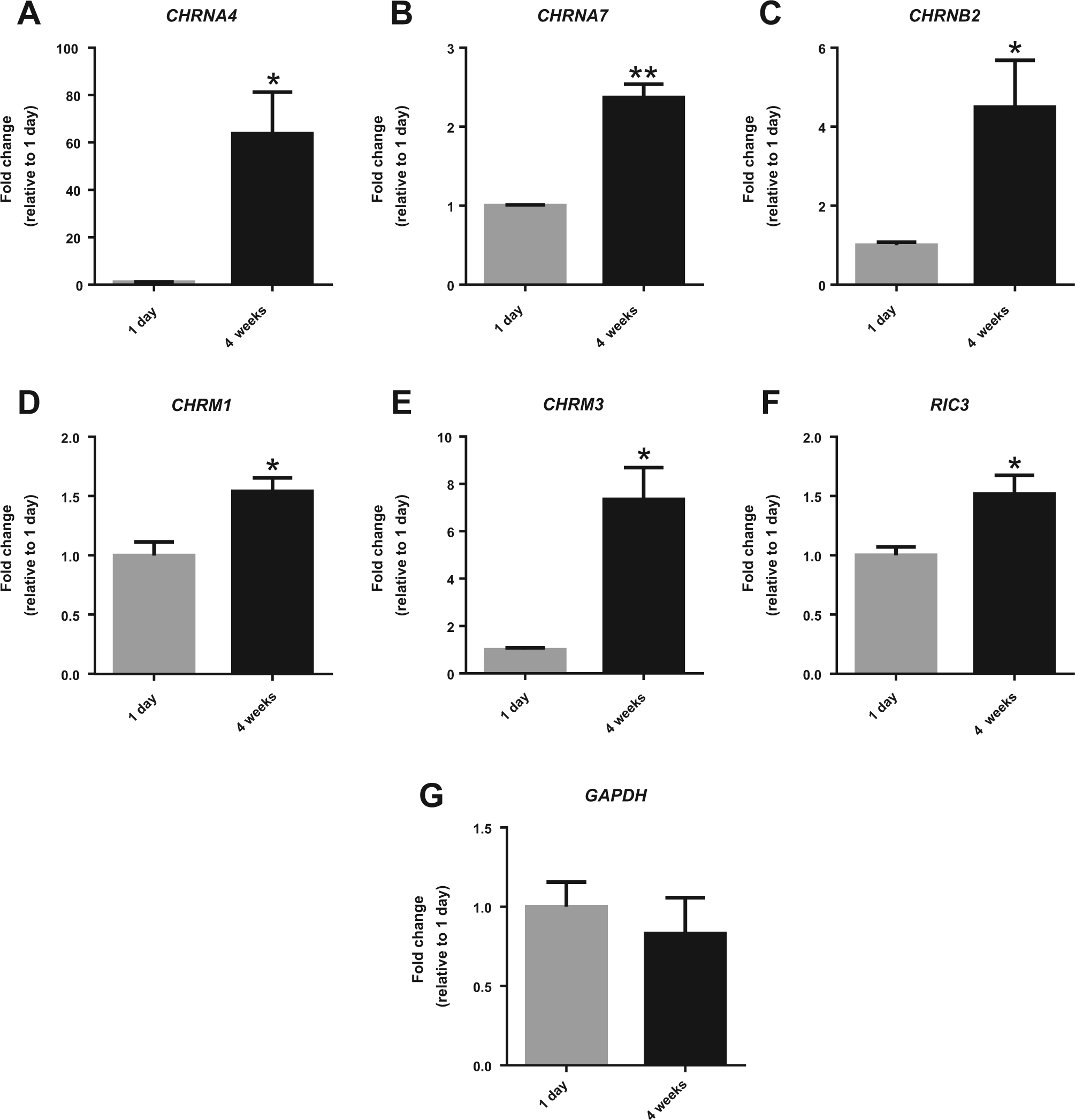
Messenger RNA (mRNA) expression of acetylcholine (ACh) receptors (AChRs) and their chaperone in HIP-009 cells. Quantitative reverse transcription polymerase chain reaction analysis showing the upregulation of mRNA expression of nicotinic AChRs (nAChRs;
Assessment of the Functional Expression of ACh Receptors
To examine the functional expression of AChRs in HIP-009 cells, we performed intracellular calcium flux assays of 1-day and 4-week differentiated HIP-009 cells, based on our previous report demonstrating by using the calcium flux assays that multiple receptors, including N-methyl-D-aspartate and α-amino-3-hydroxy-5-methyl-4-isoxazolepropionate receptors, were expressed functionally in the HIP-009 cells after 4-week differentiation. 12 In cells differentiated for 1 day, stimulation with ACh (n = 3; Fig. 2A ) and muscarine (n = 3; Fig. 2B ) significantly increased fluorescent signals in a concentration-dependent manner, whereas stimulation with nicotine did not (n = 3; Fig. 2C ). In contrast, in the 4-week differentiated HIP-009 cells, stimulation with all three neurotransmitters significantly increased fluorescent signals. Maximum potencies for the 1-day differentiated HIP-009 cells were calculated relative to those of the 4-week differentiated cells as follows: ACh (300 µM), 25.8% ± 1.9% (n = 3); muscarine (300 µM), 47.1% ± 2.3% (n = 3); and nicotine (100 µM), 6.3% ± 0.9% (n = 3). Similarly, these agonists elevated the calcium levels in HIP-009 cells differentiated for 8 weeks in a concentration-dependent fashion ( Fig. 2D–F ).
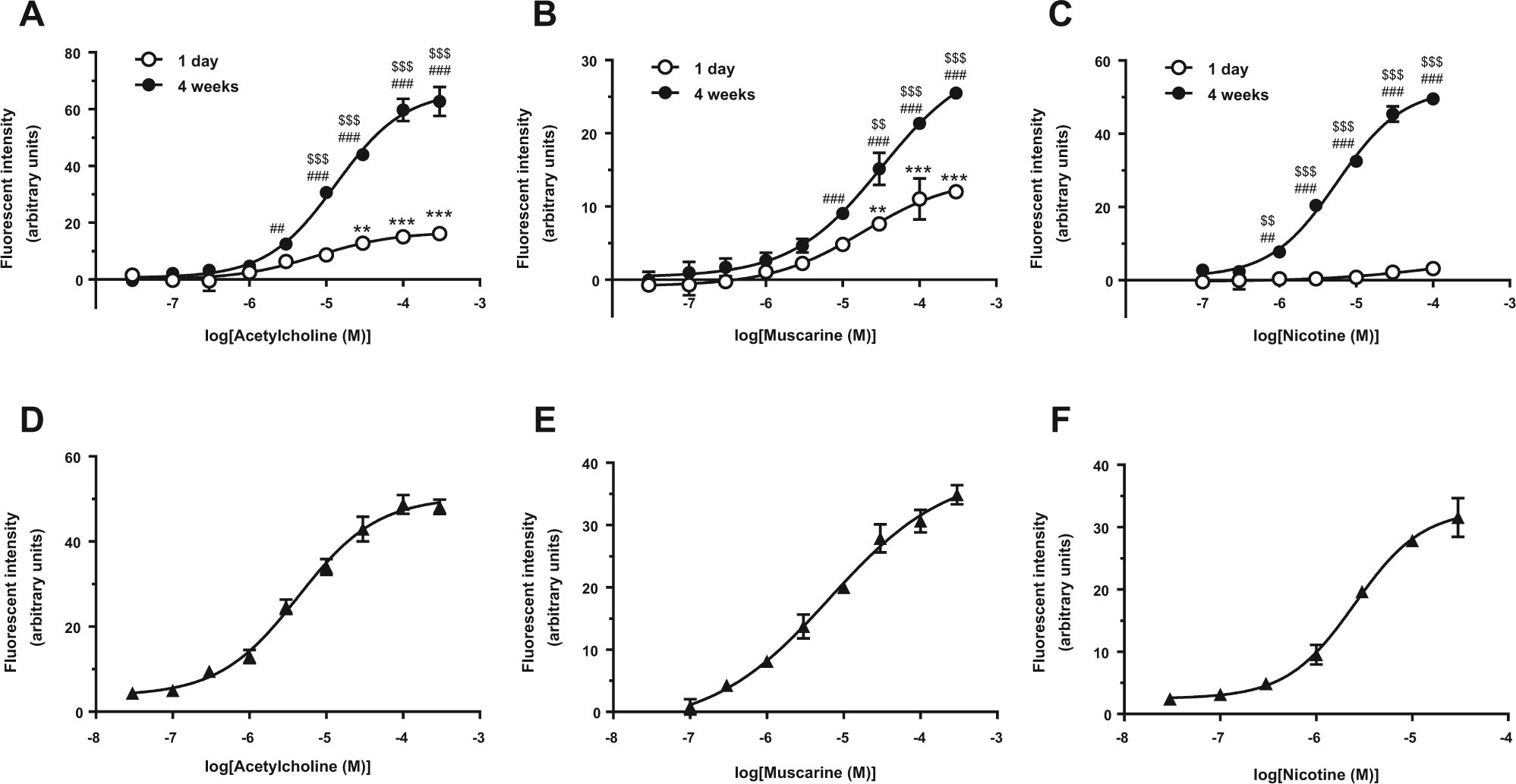
Functional expression of acetylcholine (ACh) receptors (AChRs) in HIP-009 cells. Stimulation with (
Functional Characterization of AChR Subtypes
We used HIP-009 cells that were differentiated for 4 weeks to further investigate which subtypes of AChRs were involved in the intracellular calcium flux. Upon stimulation with a potent allosteric agonist of α7 nAChRs, 4BP-TQS, the intracellular calcium signal increased in the concentration range of ≥ 1 µM in a concentration-dependent manner (n = 3; Fig. 3A ). The 4BP-TQS (20 µM)–induced intracellular calcium flux was completely inhibited by the potent α7 nAChR antagonist MLA, also in a concentration-dependent fashion (n = 3; Fig. 3B ). A concentration-dependent increase in the calcium signal was induced by the α4β2 nAChR agonist epibatidine (n = 3; Fig. 3C ). The epibatidine (12.5 nM)–induced calcium signal was partially suppressed by a competitive antagonist of the α4 receptor subunit, DHβE (n = 3; Fig. 3D ). The epibatidine (1 nM)–induced signal was potentiated by NS 9283, which is a PAM of α4β2 nAChRs (n = 3; Fig. 3E ). The calcium increase stimulated by the coapplication of epibatidine (1 nM) and NS 9283 (1 µM) was blocked by DHβE (n = 3; Fig. 3F ). A PAM for M1 mAChRs, BQCA, elevated the muscarine (10 µM)–induced calcium signal (n = 3; Fig. 3G ). The calcium signal induced by the coapplication of muscarine (10 µM) and BQCA (2.5 µM) was inhibited by the potent M1 mAChR antagonist VU 0255035 (n = 3; Fig. 3H ). An antagonist of M3 mAChRs, 4-DAMP, completely blocked the calcium elevation stimulated by muscarine (100 µM; n = 3; Fig. 3I ). The EC50 and IC50 values of these compounds are shown in Table 1 .
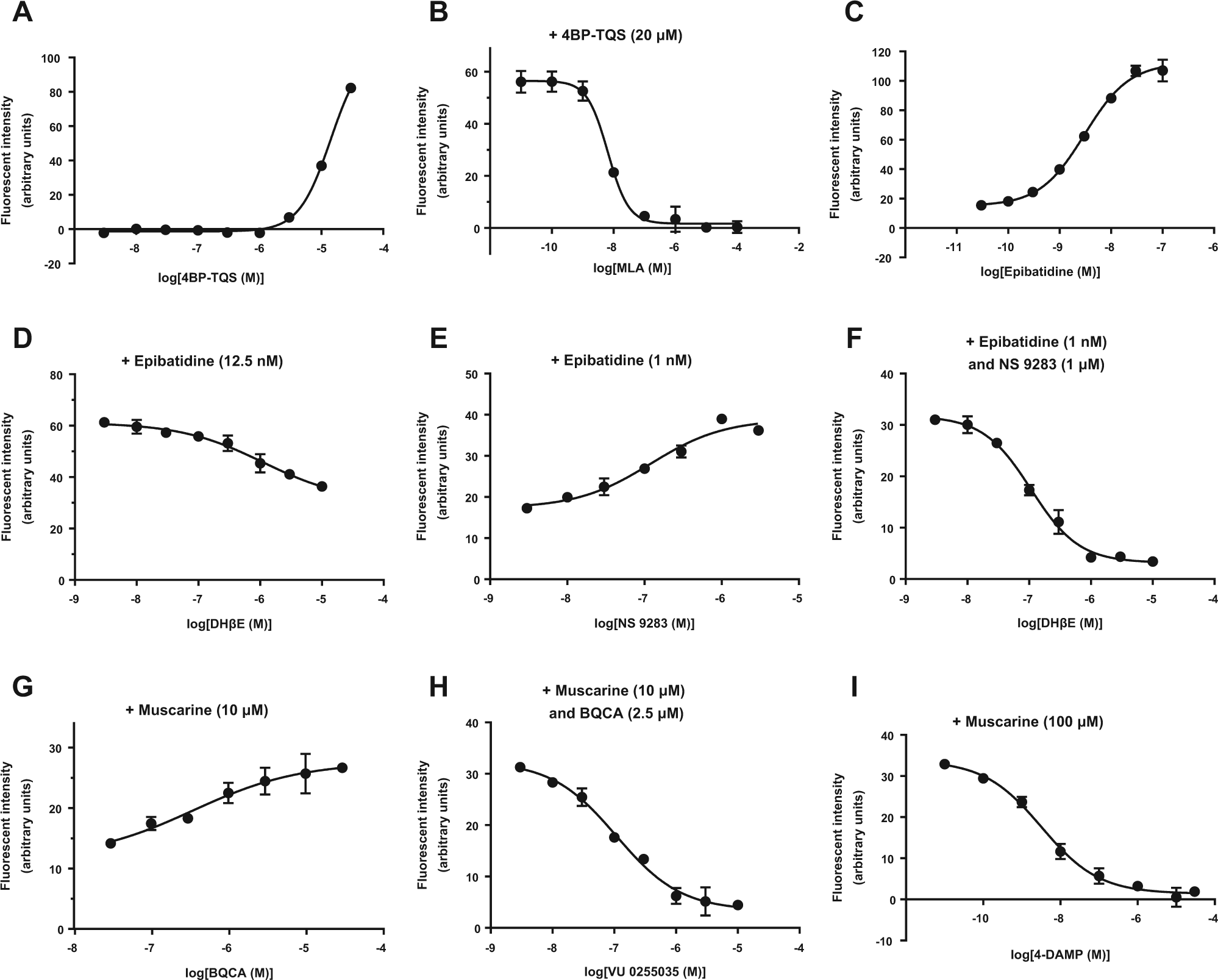
Characterization of the functional expression of each acetylcholine (ACh) receptor (AChR) subtype. (
EC50 and IC50 Values of Compounds Tested.
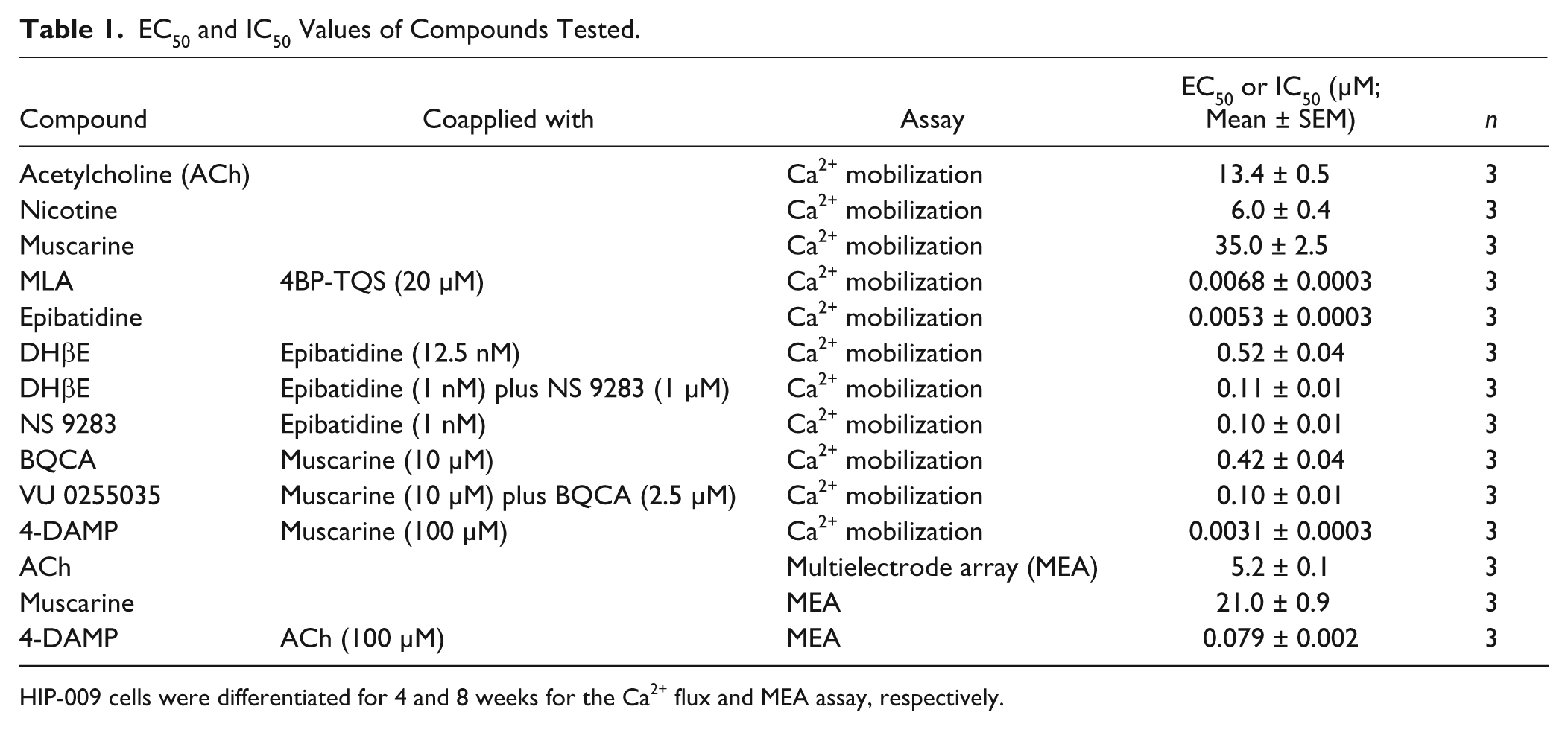
HIP-009 cells were differentiated for 4 and 8 weeks for the Ca2+ flux and MEA assay, respectively.
Involvement of AChRs in Neuronal Network Functions
To explore the role of AChRs in HIP-009–derived neuronal network functions, we performed an MEA analysis by using 8-week differentiated HIP-009 cells, based on our previous report showing that 8-week differentiated cells rather than 4-week differentiated ones were appropriate for pharmacological evaluation of effects of compounds on the neuronal networks. 13 ACh reduced the frequency of spontaneous firing in 8-week differentiated HIP-009 neurons in a concentration-dependent manner (n = 3; Fig. 4A ). The IC50 value was 5.2 ± 0.1 µM (n = 3; Table 1 ). A concentration-dependent decrease in firing was also observed for muscarine (IC50 = 21.0 ± 0.9 µM; n = 3) but not for nicotine (n = 3; Fig. 4A and Table 1 ). The ACh-elicited reduction in firing was reversed by stimulation with the nonselective mAChR antagonist scopolamine (10 µM; n = 3) but not with the nonselective nAChR antagonist mecamylamine (10 µM; n = 3; Fig. 4B ). The reversed firings by scopolamine were completely blocked by TTX (100 nM; n = 3; Fig. 4B ). 4-DAMP reversed the ACh-induced attenuation of firing in a concentration-dependent fashion (EC50 = 79.2 ± 2.1 nM; n = 3), whereas VU 0255035 did not (n = 3; Fig. 4C and Table 1 ).
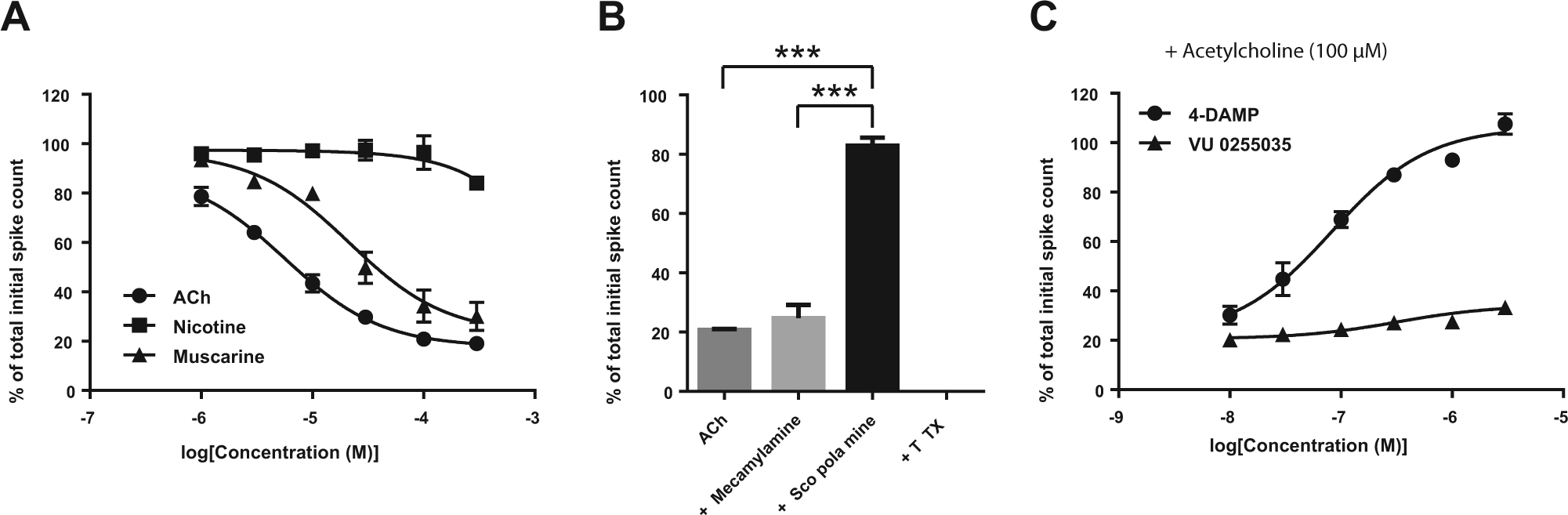
Roles of acetylcholine (ACh) receptors (AChRs) in the neuronal network functions of 8-week differentiated HIP-009 neurons. (
Discussion
In the present study, we demonstrated that HIP-009 cells can be used as a human hippocampal cell-based model and that functional α7 and α4β2 nAChRs as well as M1 and M3 mAChRs are expressed in differentiated HIP-009 cells. In addition, we showed that the activation of M3 mAChRs results in the inhibition of spontaneous firing of HIP-009 neurons.
In our gene expression analysis, we examined changes in the expression of genes that encode nicotinic and muscarinic AChRs in HIP-009 cells, focusing on key AD targets (i.e., α7 and α4β2 nAChRs, as well as M1 and M3 mAChRs). We found that the genes that encode the nAChRs (CHRNA4, CHRNA7, and CHRNB2) and mAChRs (CHRM1 and CHRM3) were upregulated after differentiation for 4 weeks. The RIC3 gene was also upregulated after the 4-week differentiation period. The RIC-3 protein has previously been shown to be a chaperone for homopentameric nAChRs such as α7 nAChRs and is essential for the functional expression of α7 nAChRs on the surface of the mammalian cell line HEK293. 14 Moreover, RIC-3 is expressed in the CA1–CA3 region, which overlaps with the high α7 expression region in the hippocampi of mice. 15 The intracellular calcium flux assay demonstrated that HIP-009 cell stimulation with nicotine did not significantly increase the calcium signal of cells differentiated for 1 day but did significantly increase that of cells differentiated for 4 weeks: the maximum potency for the 1-day differentiated HIP-009 cells relative to those of the 4-week differentiated cells was 6.3% ± 0.9% (n = 3). These results suggest that the functional expression of nAChRs (including α7 nAChRs) on the HIP-009 cell surface was increased after differentiation for 4 weeks and that RIC-3 was involved in the functional expression of nAChRs (particularly α7 nAChRs) in differentiated HIP-009 cells.
The EC50 values of ACh, nicotine, and muscarine in differentiated HIP-009 cells were 13.4 ± 0.5 µM, 6.0 ± 0.4 µM, and 35.0 ± 2.5 µM, respectively. Alkondon and Albuuergue 16 used whole-cell patch-clamp techniques when they reported EC50 values for ACh and nicotine of 129 ± 17 µM and 27 ± 4.8 µM, respectively, in rat hippocampal neurons. Scuvée-Moreau et al. 17 undertook a patch-clamp analysis of hippocampal pyramidal cells in rat brain slices and obtained an EC50 value for muscarine of 0.67 ± 0.15 µM. These differences in EC50 values between differentiated HIP-009 cells and rat hippocampal cells may reflect species differences, although differences in assay systems and/or readouts should also be considered.
It was previously reported that human iPSC-derived neurons, the gene expression profile of which closely resembles that observed in the neonatal prefrontal cortex, functionally express nAChRs.18,19 In these studies, however, the iPSC-derived neurons did not respond to ACh alone up to 1 mM, and a slowly desensitizing current was detected in response to coapplication of ACh and PNU-120596 (a PAM for α7 nAChRs). In contrast, here we demonstrated that differentiated HIP-009 neurons can respond to ACh alone. There are two possible explanations for this difference in reactivity to ACh between HIP-009 neurons and human iPSC-derived cortical neurons. The first is that the expression level of nAChRs in iPSC-derived neurons is low, as stated by Chatzidaki et al. 19 in their report. The second is that hippocampal and cortical neurons show different reactivities to ACh. Evidence in support of this latter explanation comes from a study in adult rats by Odagaki et al., 20 who showed that ACh acted in the hippocampus and cerebral cortex with EC50 values of 71.6 µM and 19.5 µM, respectively, as determined by an antibody-capture scintillation proximity assay.
The selectivity of compounds, such as agonists and antagonists of nAChR and mAChR, is important when determining the functional expression of each subtype in native cells, such as HIP-009 cells, but has less import for engineered cells overexpressing a receptor of interest. However, the selectivity of some agonists or antagonists to a certain subtype becomes decreased (e.g., epibatidine) when they are applied at higher concentrations. 21 Therefore, these compounds were assessed in combinations (e.g., epibatidine and NS 9283 or DHβE) in the calcium flux assay to demonstrate the functional expression of each subtype in differentiated HIP-009 cells. 4BP-TQS, a potent allosteric agonist of α7 nAChRs, increased intracellular calcium; this upregulated signal was blocked by MLA, a potent α7 nAChR antagonist, suggesting the functional expression of α7 nAChRs. Epibatidine is an α4β2 nAChR agonist that also acts on α7 nAChRs with Ki values of 0.02 nM and 233 nM for α4β2 and α7 nAChRs, respectively. 21 We observed an epibatidine concentration-dependent increase in intracellular calcium levels. To clarify the involvement of α4β2 nAChRs in this calcium increase, we examined the effects of DHβE, a competitive antagonist of the α4 receptor subunit (IC50 value = 0.37 µM for α4β2 nAChRs), 22 on the epibatidine-induced calcium mobilization. The epibatidine-induced calcium signal was inhibited by DHβE with an IC50 value of 0.52 ± 0.04 µM. Furthermore, NS 9283 (a PAM of α4β2 nAChRs; EC50 value = 0.42 µM) 23 increased the epibatidine-induced calcium signal (EC50 value = 0.10 ± 0.01 µM). The calcium signal induced by the coapplication of epibatidine and NS 9283 was blocked by DHβE. These results indicate that α4β2 nAChRs function in differentiated HIP-009 cells.
Similarly, we investigated the functional expression of each mAChR subtype in differentiated HIP-009 cells. BQCA is an M1 mAChR-selective PAM, which has greater than 100-fold selectivity over the other subtypes (EC50 value = 0.85 µM). 24 VU 0255035 is an M1 mAChR-selective antagonist with a reported IC50 value of 0.13 ± 0.03 µM. 25 In this study, we observed that BQCA increased the muscarine-induced calcium flux, with an EC50 value of 0.42 ± 0.04 µM, and that the calcium increase generated by the coapplication of muscarine and BQCA was inhibited by VU 0255035, which gave an IC50 value of 0.10 ± 0.01 µM. These results demonstrate the functional expression of M1 mAChRs. We also found that the muscarine-induced calcium flux was blocked by 4-DAMP, with an IC50 value of 3.1 ± 0.3 nM. Araujo et al. 26 reported that 4-DAMP has high affinity (Kd < 1 nM) for M3 mAChRs, whereas it binds to M1 mAChRs with a lower affinity (Kd > 50 nM). Flores et al. 27 also used low concentrations of 4-DAMP (≤500 nM) as an M3 mAChR-selective antagonist. Our result suggests that M3 mAChRs are functionally expressed in differentiated HIP-009 cells.
In our neuronal network analysis of MEA data, we observed that the spontaneous firing of HIP-009 neurons was reduced by ACh in a concentration-dependent manner. This ACh-induced reduction of firing was reversed by scopolamine, a nonselective antagonist of mAChRs, but not by mecamylamine, a nonselective antagonist of nAChRs. These results suggest that mAChRs play an important role in the inhibition of spontaneous firing. To clarify which subtype of mAChRs was responsible for the decrease in firing, we used subtype-selective antagonists. 4-DAMP reversed the ACh-induced firing inhibition in a concentration-dependent fashion, whereas VU 0255035 did not. This result suggests that, among mAChR subtypes, activation of M3 plays a critical role in the inhibition of spontaneous firing. In agreement with these results, de Vin et al. 28 reported that carbachol induced a concentration-dependent depression of field excitatory postsynaptic potentials of rat hippocampal slices on the basis of an MEA analysis. These authors also showed that M3 mAChRs were the major muscarinic subtype mediating the inhibition of excitatory synaptic transmission in the rat hippocampus. In contrast, other researchers found that carbachol produced an increase in the spontaneous firing rate of rat subthalamic neurons and that this excitatory effect occurred via M3 mAChRs. 27 These contradictory outcomes imply that the physiological functions of receptors depend on where they are expressed. Such region-dependent functions have been reported for other subtypes. For example, Foster et al. 29 discovered that activation of somatodendritic M5 mAChRs on substantia nigra pars compacta neurons stimulates their activities, whereas striatal M5 mAChR activation inhibits dopamine release from nigrostriatal synapses. Considering these observations, our results suggest that networks of HIP-009 neurons reflect one aspect of the hippocampus in terms of reactivity to acetylcholine.
In conclusion, we found that α7 and α4β2 nAChRs, as well as M1 and M3 mAChRs, all of which are important targets for AD drug development, are functionally expressed in differentiated HIP-009 cells. We also revealed that the activation of M3 mAChRs mediated inhibition of the spontaneous firing of HIP-009 neurons. To our knowledge, this is the first report to show the functional expression of multiple AChRs in human hippocampal neurons and their roles in neuronal network functions in vitro. This unique cell model having similar responsiveness to acetylcholine in the hippocampus is itself applicable to in vitro AD drug screening targeting AChRs. In addition, this model may be a loadstar to establish a robust method of hippocampal neuron differentiation from human iPSCs, including disease-specific iPSCs, accelerating our understanding of the mechanisms underlying AD pathophysiology. These further applications and investigations are needed to evaluate and improve the clinical predictability of in vitro human cell models, including HIP-009 cells, leading to the development of novel therapeutic agents for AD.
Footnotes
Acknowledgements
We thank Dr. Ken Hatanaka for valuable advice.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
