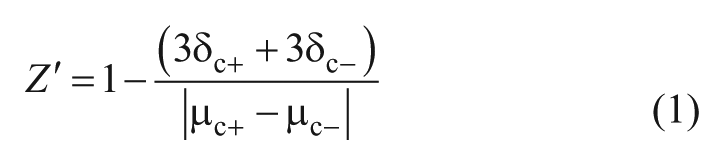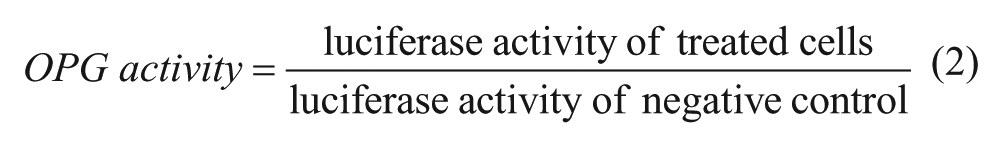Abstract
The ratio between osteoprotegerin (OPG) and the receptor activator of NF-κB ligand (RANKL) in the bone microenvironment indicates the level of osteoclastogenesis, and upregulation of this ratio would improve osteoporosis. In this study, we established a novel high-throughput screening (HTS) system using two stably transfected monoclonal cell lines that either express firefly luciferase under the OPG promoter control or concurrently express firefly and renilla luciferases under control of the OPG and RANKL promoters, respectively. With this system, we can conveniently and rapidly detect the effects of compounds on the expression of OPG and RANKL through changes in firefly and renilla luciferase activities. A total of 8160 compounds were screened using this system, yielding five compounds without previously reported activity. The compound with greatest potential is E05657 with high activity and low effective concentration in the HTS system. It increases the OPG/RANKL ratio and OPG secretion, decreases the NFATc1 expression, and reduces osteoclastogenesis in vitro. These results indicate that this novel HTS system can be used to identify small molecules with potential antiosteoporosis effects, and E05657 is a promising lead compound as a novel antiosteoporosis drug.
Keywords
Introduction
Osteoporosis is a systemic and metabolic skeletal disease characterized by low bone mass and microarchitectural deterioration with a consequential increase in bone fragility and susceptibility to fracture.1,2 Because of the potentially devastating outcomes and high cumulative rate of fractures, osteoporosis has become a serious public health issue.3,4 Osteoporosis is believed to be caused by the imbalance in bone remodeling. Bone loss occurs when the rate of bone resorption mediated by osteoclasts exceeds bone formation. 5 Therefore, a decrease of bone resorption by inhibiting the activity of osteoclasts could improve osteoporosis. The differentiation and maturation of osteoclasts are regulated by cytokines produced by osteoblasts, and the osteoprotegerin (OPG)–receptor activator of NF-κB (RANK)–RANK ligand (RANKL) axis appears to be the most crucial one among the numerous factors. 6 RANK 7 is a signaling molecule located on the cell surface of osteoclasts and their precursors, but the RANK ligand, RANKL,7,8 is expressed in osteoblasts. The binding of RANKL to RANK drives osteoclastic differentiation of hematopoietic progenitor cells into mature osteoclasts. OPG,9–11 which is also expressed in osteoblasts, is a decoy receptor of RANKL. The association of OPG with RANKL blocks the binding of RANKL to RANK. As a result, the differentiation of osteoclasts is inhibited. 12 Therefore, the ratio of OPG/RANKL in the bone microenvironment is crucial for the activities of osteoclasts, and upregulating the ratio will decrease the differentiation and maturation of osteoclasts. 13 Thus, it is considered a strategy for osteoporosis treatment.
To screen small molecules that enhance the endogenous OPG expression is one approach for the development of antiosteoporosis drugs. 14 However, recent studies indicate that some factors enhance the expression levels of both OPG and RANKL, and some of them induce the expression of RANKL more than that of OPG,15,16 which decreases the OPG/RANKL ratio and enhances the differentiation of osteoclast. Therefore, it is necessary to establish a high-throughput screening (HTS) system that is able to concurrently evaluate the effects of compounds on the expression of both OPG and RANKL.
In the present study, we developed a novel HTS system to screen compounds that upregulate the OPG/RANKL expression ratio. For this purpose, the 5.9 kb OPG promoter was cloned upstream of a firefly luciferase reporter gene and then transfected into the human osteosarcoma cell line U-2OS, resulting in a stable clone named UOP. Another plasmid containing a renilla luciferase reporter gene under the control of the 2.0 kb RANKL promoter was integrated into the genome of UOP cells to generate cells termed UORP, which could be used to screen compounds that regulate the OPG/RANKL expression ratio. The HTS includes two steps. First, we use UOP cells to screen upregulators of OPG expression from a library containing thousands of compounds. The resulting candidate compounds were then used to examine their effect on the OPG/RANKL expression ratio in UORP cells, and five candidate compounds were identified from the two-step screening. Among them, E05657 appeared to be the most effective in increasing the OPG/RANKL ratio and OPG secretion in osteoblastic cells and decreasing osteoclastogenesis in vitro at very low concentrations (≤1 μM). Therefore, this study has laid a methodological foundation for the development of novel small-molecule drugs for the treatment of osteoporosis and identified a highly active lead compound.
Materials and Methods
Regents
McCoy’s 5A medium, alpha modified Eagle’s medium (α-MEM), high-glucose Dulbecco’s modified Eagle’s medium (DMEM), and fetal bovine serum (FBS) were purchased from Thermo Fisher Scientific (Waltham, MA). G418 was purchased from Amresco (Solon, OH). Hygromycin was purchased from Roche (Basel, Switzerland). β-Sodium glycerophosphate and
Cell Culture
Human osteosarcoma cell lines U-2OS and the mouse mononuclear macrophage cell line Raw264.7 were obtained from the Cell Center of Basic Medicine, Chinese Academy of Medical Sciences (Beijing, China). The mouse preosteoblast cell line MC3T3-E1 (subclone 14) was obtained from the Cell Center of Chinese Academy of Science (Shanghai, China). U-2OS cells were cultured in McCoy’s 5A medium supplemented with 10% FBS. Raw264.7 cells were grown in high-glucose DMEM supplemented with 10% FBS. MC3T3-E1 cells were grown in α-MEM supplemented with 10% FBS. All cells were incubated at 37 °C in a humidified 5% CO2 incubator.
To induce differentiation of MC3T3-E1 cells into mature osteoblasts, the cells were cultured in osteogenic medium consisting of complete α-MEM supplemented with 10 mM β-sodium glycerophosphate and 50 μg/mL
Reporter Plasmid Construction
A 5.9 kb fragment of the human OPG promoter
17
and 2.0 kb fragment of the human RANKL promoter
18
were amplified by PCR using human genomic DNA as the template and the following primers (sense and antisense): 5′ -TGG
Establishment of Stably Transfected Monoclonal Cell Lines
U-2OS cells were transfected with pGL4.17-OPGp at 80%–90% confluence using Lipofectamine 2000 (Thermo Fisher Scientific) according to the manufacturer’s instructions. The transfected cells were cultured in complete McCoy’s 5A medium for 24 h, followed by treatment with G418 (500 μg/mL) for 1 week. The surviving cells were trypsinized, serially diluted in 96-well plates, and cultured while maintaining G418 selection pressure for 10–14 days. Single cell clones with G418 resistance were chosen, and the firefly luciferase expression activities of the clones were measured by the Luciferase Assay System (Promega). The stably transformed clone with the highest expression level of luciferase was designated as UOP. The UOP cell line was maintained in complete McCoy’s 5A medium containing 400 μg/mL G418.
At 80%–90% confluence, UOP cells were transfected with pGL4.76-RANKLp using Lipofectamine 2000. The transfected cells were grown in UOP maintenance medium for 24 h. Then, hygromycin (300 μg/mL) was added to the medium, and the cells were maintained for 1 week. Single cell clones were obtained from the surviving cells using similar methods for establishment of UOP cells but under dual selective pressure of G418 and hygromycin. The luciferase activity was measured by the Dual-Luciferase Reporter Assay System (Promega). In brief, the substrate of firefly luciferase was added to a cell lysate and the luminescence was detected, followed by quenching the firefly luminescence; meanwhile the substrate of renilla luciferase was added, and then the renilla luminescence was measured. The stably transfected clone with the highest expression levels of both firefly and renilla luciferases was designated as UORP and maintained in complete McCoy’s 5A medium containing 400 μg/mL G418 and 200 μg/mL hygromycin.
Evaluation of HTS System
Genistein was used as a positive control to evaluate UOP cell lines because it upregulates the expression level of OPG.19,20 UOP cells were cultured in 96-well plates to 80%–90% confluence, followed by treatment for 24 h with a series of genistein concentrations from 1 × 10−4 to 100 μM, and the firefly luciferase activity of UOP cells was measured. Then, a dose–response curve was constructed by GraphPad Prism 5 (GraphPad Software, La Jolla, CA). TGF-β1 was used to evaluate the response of UORP cells.14,17,21 The cells were treated with 10 ng/mL TGF-β1 for 24 h, and then firefly and renilla luciferase activities were measured by the Dual-Luciferase Reporter Assay System. The ratio of the two kinds of luciferase activities was calculated.
The Z′ factor is a characteristic parameter to evaluate the quality of an HTS system. 22 As positive controls, genistein and TGF-β1 were used to calculate the Z′ factors of UOP and UORP cells, respectively, to determine whether they can be applied to an HTS assay. UOP and UORP cells were treated with 6.25 μM genistein or 10 ng/mL TGF-β1, respectively, for 24 h. The same number of UOP and UORP cells were treated with 0.1% DMSO or double-distilled (dd) H2O (negative control). Then, the firefly luciferase activity and the ratio of activities of firefly over those of renilla luciferase were detected, and the Z′ factors were calculated according to eq 1:
where δc+ is the standard deviation (SD) of the positive control, δc– is the SD of the negative control, μc+ is the mean of the positive control, and μc– is the mean of the negative control. 22
In addition, the signal-to-background (S/B) ratio, signal-to-noise (S/N) ratio, and signal coefficient of variation (CV%) were calculated according to the methods described by Zhang et al. 22
Two-Step HTS Assay
The HTS assay includes two steps in which upregulators of OPG expression or the OPG/RANKL ratio were screened, respectively. In the first step, UOP cells were seeded in 96-well white plates with a clear bottom at 5 × 104 cells/well. At 80% confluence, the cells were washed once with phosphate-buffered saline (PBS), followed by treatment with 0.1% DMSO (negative control), 6.25 μM genistein (positive control), or the 8160 samples for screening (2 μg/mL of each compound). The cells were incubated at 37 °C for 24 h. Then the cells were lysed and the firefly luciferase activity was detected. The activity of a sample in regulation of OPG expression was defined as eq 2:
A positive compound was defined as the following: the OPG activity of which was more than the mean of the OPG activities of the total samples + 3 × SD of the OPG activities. 22 The second step was performed to determine whether the identified positive compounds had the capacity to upregulate the OPG/RANKL ratio. UORP cells were used in this step. The procedure was similar to the first step except the positive control was changed to 10 ng/mL TGF-β1, and the luciferase activities were measured by the Dual-Luciferase Reporter Assay System as described above. Moreover, the activity of a compound in regulation of the OPG/RANKL ratio was defined as eq 3:
Western Blot Analysis
U-2OS or MC3T3-E1 cells were seeded in six-well plates at 4 × 105 or 1 × 105 cells/well. At 80%–90% confluence, various concentrations of E05657 were added to the cultures. The cells were harvested after 24 h, and the total protein in the cells was extracted using RIPA lysis buffer (PPLYGEN, Beijing, China), followed by separation by 10% sodium dodecyl sulfate–polyacrylamide gel electrophoresis, and then transferred onto a 0.45 μM polyvinylidene fluoride membrane (Millipore, Bedford, MA). The membranes were blocked with 5% (w/v) dry skim milk in Tris-buffered saline containing 0.2% Tween-20 (TBST) for 1 h, followed by incubation with primary antibodies (rabbit polyclonal antibody against OPG [Abcam, Cambridge, MA], rabbit monoclonal antibody against RANKL [Epitomics, Burlingame, CA], or mouse monoclonal antibody against β-actin [Zhongshanjinqiao, Beijing, China]) at 4 °C overnight. Then, the membranes were washed with TBST three times, followed by incubation with horseradish peroxidase (HRP)–conjugated goat anti-rabbit or goat anti-mouse IgG (Zhongshanjinqiao) for 1 h at room temperature. After washing with TBST three times, the protein bands were detected with an Enhanced Chemiluminescence reaction kit (Millipore).
Nuclear factor–activated T cells c1 (NFATc1) were detected using mouse anti-NFATc1 monoclonal antibody (Abcam) in Raw264.7 cells using methods similar to those described above.
Enzyme-Linked Immunosorbent Assay
U-2OS (4 × 105 cells/well) or MC3T3-E1 (1 × 105 cells/well) cells were cultured in six-well plates to 80%–90% confluence, followed by treatment with various concentrations of E05657 for 48 h. The culture supernatants were collected and concentrated by vacuum freeze drying. The quantities of OPG protein in the concentrates were detected using human OPG enzyme-linked immunosorbent assay (ELISA) kit (RayBiotech, Norcross, GA) or mouse OPG ELISA kit (R&D Systems, Minneapolis, MN) according to the manufacturer’s instructions.
Inhibition of Osteoclast Differentiation and TRAP Staining
MC3T3-E1 cells were cultured in osteogenic medium for 3 days and then treated with several concentrations of E05657 for 6 days, followed by collection of the culture supernatant as conditioned medium (CM). Raw264.7 cells were seeded in a 96-well plate at a density of 3 × 104 cells/well and maintained in osteoclastic medium 1 (OM1, complete DMEM supplemented with 30 ng/mL soluble RANKL) for 3 days, and then the CM was mixed with OM1 (1:1, v/v) and added to Raw264.7 cells. After 3 days, Western blotting was performed to analyze changes in NFATc1 expression levels. In addition, Raw264.7 cells with the same density in another 96-well plate were treated with a mixture of CM and OM2 (complete DMEM supplemented with 100 ng/mL soluble RANKL) for 3 days, followed by tartrate-resistant acid phosphatase (TRAP) staining analysis.23,24
TRAP staining was performed using a leukocyte acid phosphatase staining kit (387-A, Sigma-Aldrich, St. Louis, MO) according to the manufacturer’s instructions. In brief, fixed cells were treated with a mixture of fast garnet GBC base and sodium nitrite (1:1, v/v) at room temperature for 3 min. Subsequently, the substrate solution (2.5 mM naphthol–ASBI phosphate, 50 mM tartrate solution, and 100 mM acetate solution, pH 5.0) was added to the cells, followed by incubation for 30 min at 37 °C in the dark. TRAP-stained giant multinucleated cells were examined under a light microscope.
Statistical Analysis
Western blots were scanned and analyzed by Quantity One software (Bio-Rad, Hercules, CA). Data statistics, dose–response curves, and histograms were calculated and constructed with Graphpad Prism 5.0. Data are expressed as the mean ± SD. Results were analyzed by the Student t test. p values of less than 0.05 were considered statistically significant.
Results and Discussion
Establishment, Optimization, and Evaluation of the HTS Assay
Recent studies indicate that the upregulation of RANKL expression stimulated by certain agents is greater than that of OPG. For example, Runx2, an osteogenic transcription factor that enhances OPG expression, also stimulates RANKL expression and accelerates the differentiation of macrophages into osteoclasts. 15 Diercke et al. reported that interleukin-1β not only upregulates OPG expression, but also enhances RANKL expression. In addition, the upregulation of RANKL by these agents is more dramatic than that of OPG, resulting in the decrease of the OPG/RANKL ratio. 16 Small molecular compounds could regulate the expression of OPG and RANKL through their effects on cellular signals involved in various cytokines. Thus, it is not sufficient to evaluate the effect of a compound on osteoporosis by solely examining its regulation of OPG expression. Instead, concurrent examination of the effects of a compound on the expressions of both OPG and RANKL is necessary. By doing so, we can eliminate compounds that may induce the expression of RANKL more than that of OPG.
The luciferase reporter gene is a commonly used tool for drug screening.25,26 In transient transfected cells, constitutively expressed renilla luciferase from plasmids is frequently used as an internal reference to normalize the firefly luciferase, and the ratio of the two kinds of luciferase activities is considered a final result. 27 Because an internal reference is not necessary for stably transfected cells, renilla luciferase can be used to examine the activity of RANKL promoter. Therefore, we established the stably transfected cell line UORP that simultaneously expresses firefly and renilla luciferase from OPG and RANKL promoters, respectively. The ratio change of firefly luciferase to renilla luciferase in UORP cells treated with a compound will indicate the role of this compound in the regulation of OPG and RANKL expression.
Establishment of stably transfected cell lines UOP and UORP
The constructed reporter plasmid pGL4.17-OPGp was transfected into U-2OS osteosarcoma cells, and transfected single cell clones were selected and cultured to obtain the stably transfected cell line UOP, which could be used for screening upregulators of OPG expression ( Fig. 1A ). Furthermore, pGL4.76-RANKLp was transfected into UOP cells and the monoclonal cell line UORP was obtained for screening compounds that can enhance the OPG/RANKL expression ratio ( Fig. 1B ).
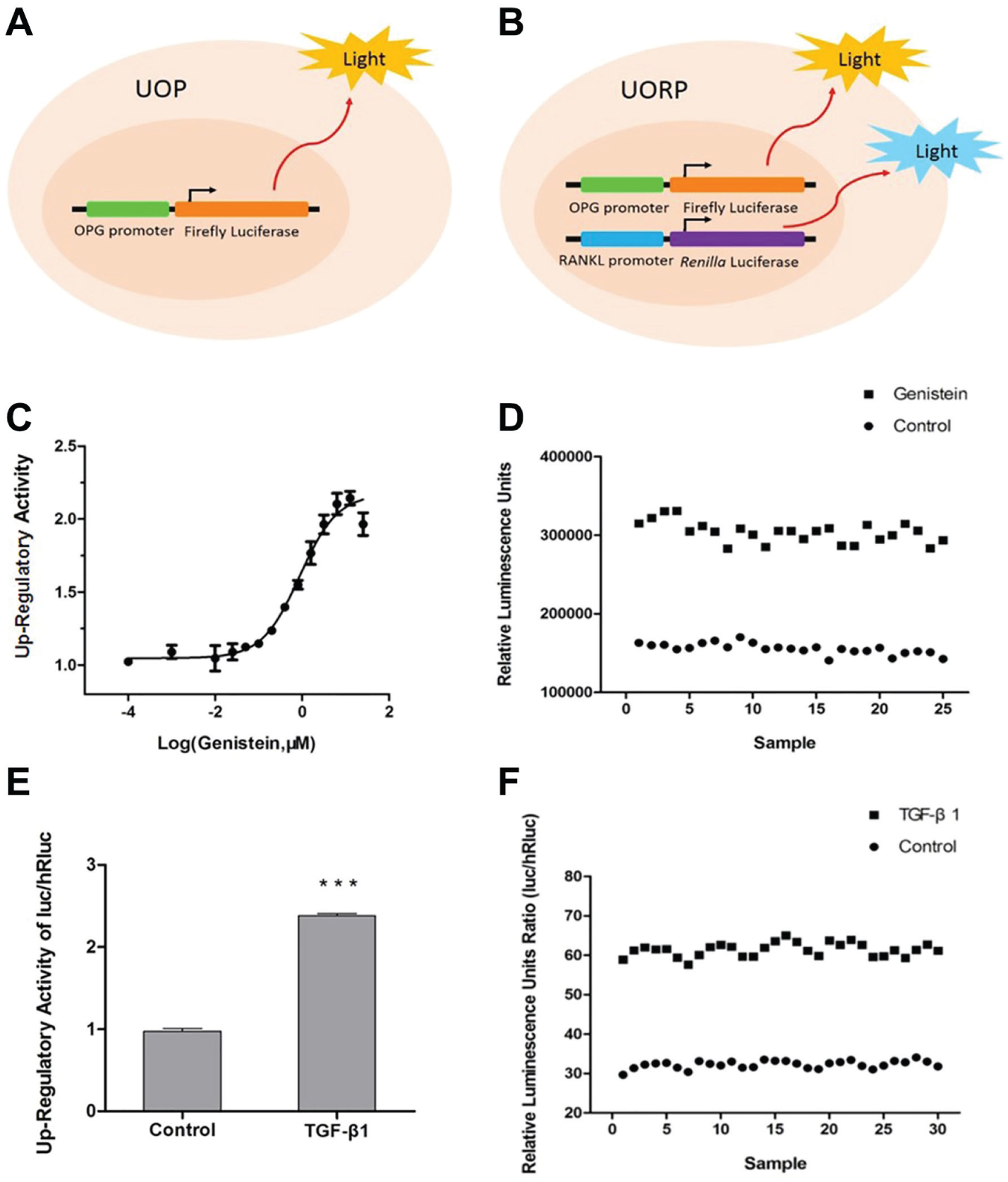
Schematic diagram of the stably transfected cells and evaluation of the HTS system. (
Optimization and evaluation of HTS system
A cell-based HTS system must be optimized for ideal conditions, including the DMSO concentration, cell density, and time for compound treatment. The optimization was performed using UOP cells, and 6.25 μM genistein was used as the positive control. The optimized conditions were as follows: DMSO concentration, 0.1% (
UOP cells were treated with various concentrations of genistein for 24 h. The expression level of firefly luciferase was obviously upregulated as the increase of genistein concentration, and the dose–response curve was calculated. The maximum upregulatory activity was 2.14 (at 6.25 μM), and the half-maximal effective concentration (EC50) was 0.87 μM (
Fig. 1C
). Next, UOP cells were cultured in a 96-well plate, and half of the cells were treated with 6.25 μM genistein, while the rest were treated only with 0.1% DMSO. The firefly luciferase expression levels were detected and the Z′ factor was calculated accordingly (
Fig. 1D
). In addition, the S/B ratio, S/N ratio, and signal CV% were calculated (
UORP cells were also treated with 10 ng/mL TGF-β1 for 24 h, and then the activities of firefly and renilla luciferase were measured. The ratio between the two luciferase activities (firefly/renilla) was increased by 2.4-fold compared with the control cells ( Fig. 1E ). With the same method, the Z′ factor and other indicators were also calculated using 10 ng/mL TGF-β1 as the positive control ( Fig. 1F , Table 1 ).
Indicators of UORP Cells for Use in an HTS Assay.
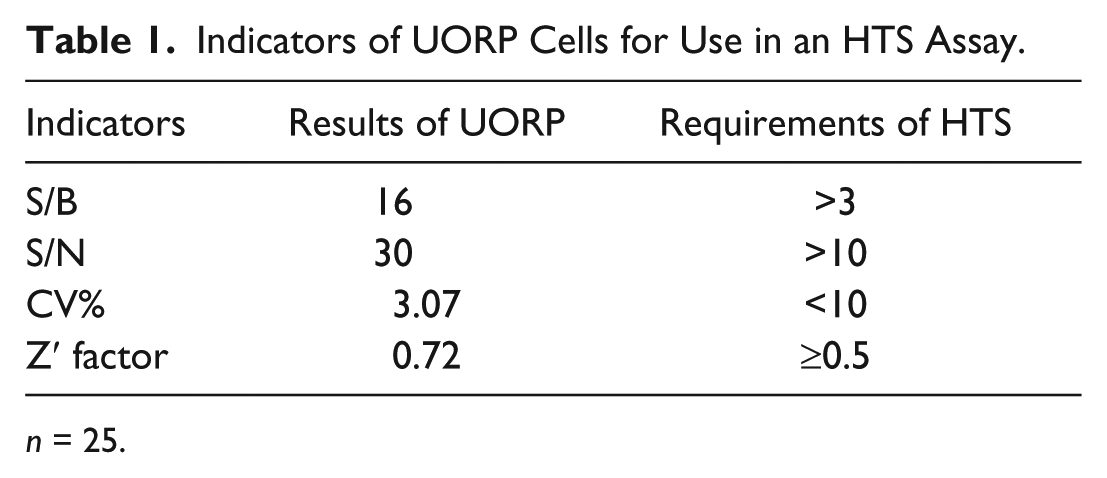
n = 25.
All the evaluation indexes of UOP and UORP cells were confirmed to comply with the requirements of HTS; thereby these cell lines could be used to identify compounds that can enhance OPG expression and the OPG/RANKL expression ratio.
HTS Assay for Screening Compounds That Upregulate the Ratio of OPG/RANKL
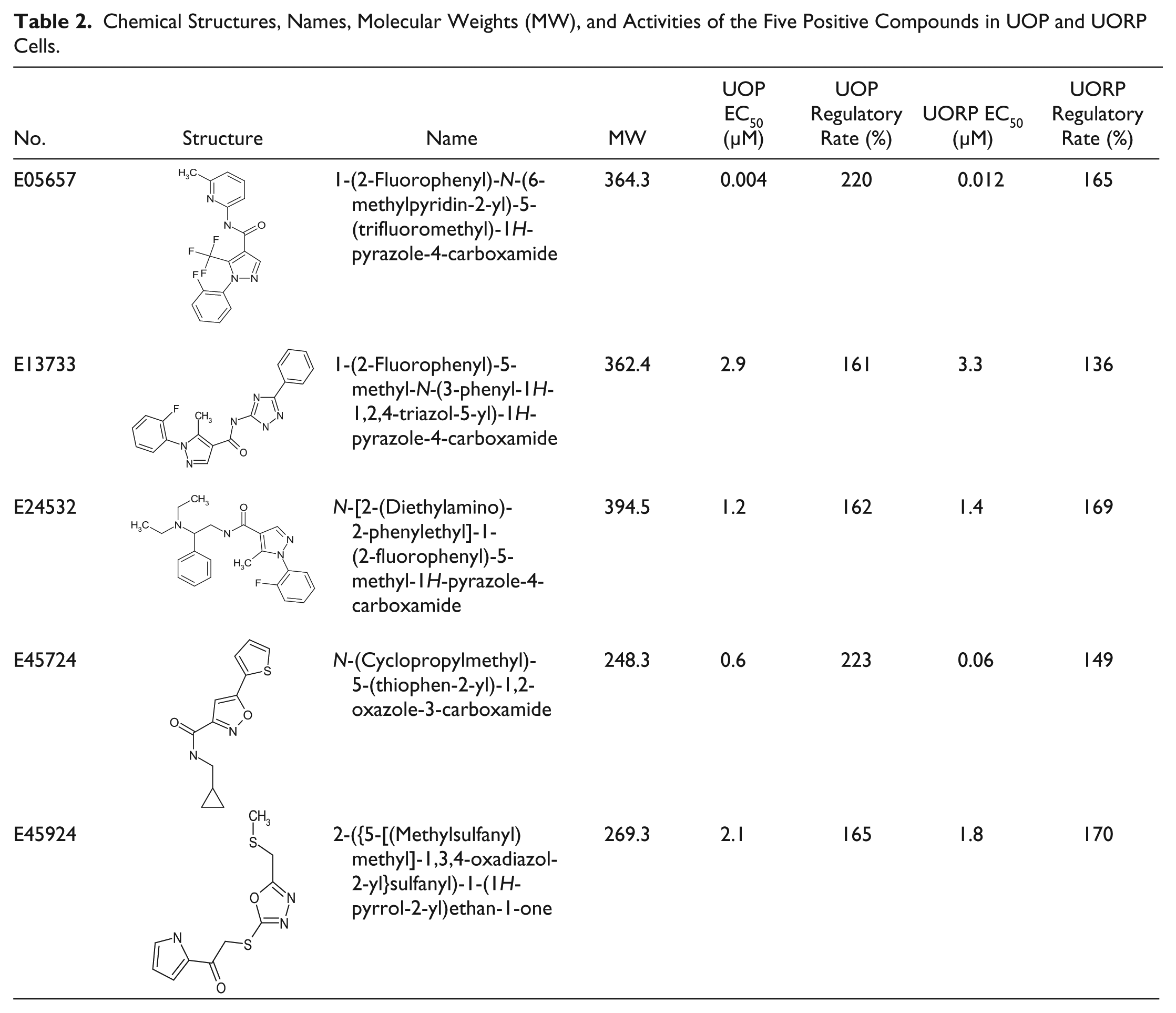
A total of 8160 compounds were added into UOP cell cultures at a final concentration of 2 μg/mL. After 24 h of incubation, the change in the firefly luciferase activity of UOP cells was measured. A total of 56 candidate compounds were selected because their enhancement on OPG activity was ≥1.5 compared with the control, the threshold calculated as mentioned above. These positive compounds were further tested in UORP cells. Finally, five compounds were found to significantly upregulate the ratio of firefly luciferase to renilla luciferase in a dose-dependent manner, indicating that they have the potential to upregulate the ratio of OPG/RANKL expression ( Table 2 , Fig. 2 ). The five compounds identified by HTS that can upregulate the OPG/RANKL expression ratio were searched in Scifinder, revealing no reported activities.
Chemical Structures, Names, Molecular Weights (MW), and Activities of the Five Positive Compounds in UOP and UORP Cells.
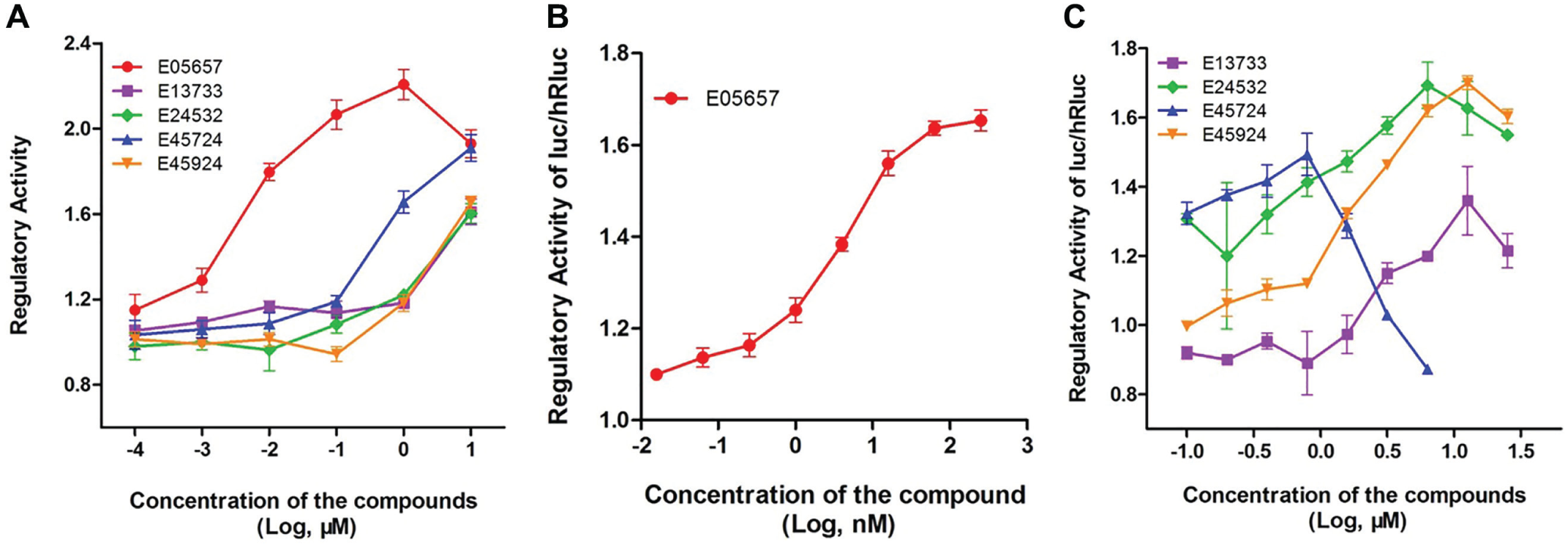
Activities of the five positive compounds in UOP and UORP cells. (
Validation of Active Compound E05657
Compound E05657 is highly active in the upregulation of OPG expression in UOP cells (220%) and in the increase of the OPG/RANKL expression ratio in UORP (165%); meanwhile, the EC50 values for OPG expression and the OPG/RANKL ratio change were as low as 4 and 12 nM, respectively. Furthermore, it exhibited an obvious dose-dependent effect on these cells ( Table 2 , Fig. 3A , B ). Among the five candidate compounds, E05657 appears to have the most potential to upregulate the OPG/RANKL ratio and reduce osteoclastogenesis. Therefore, the activities of E05657 were tested further.
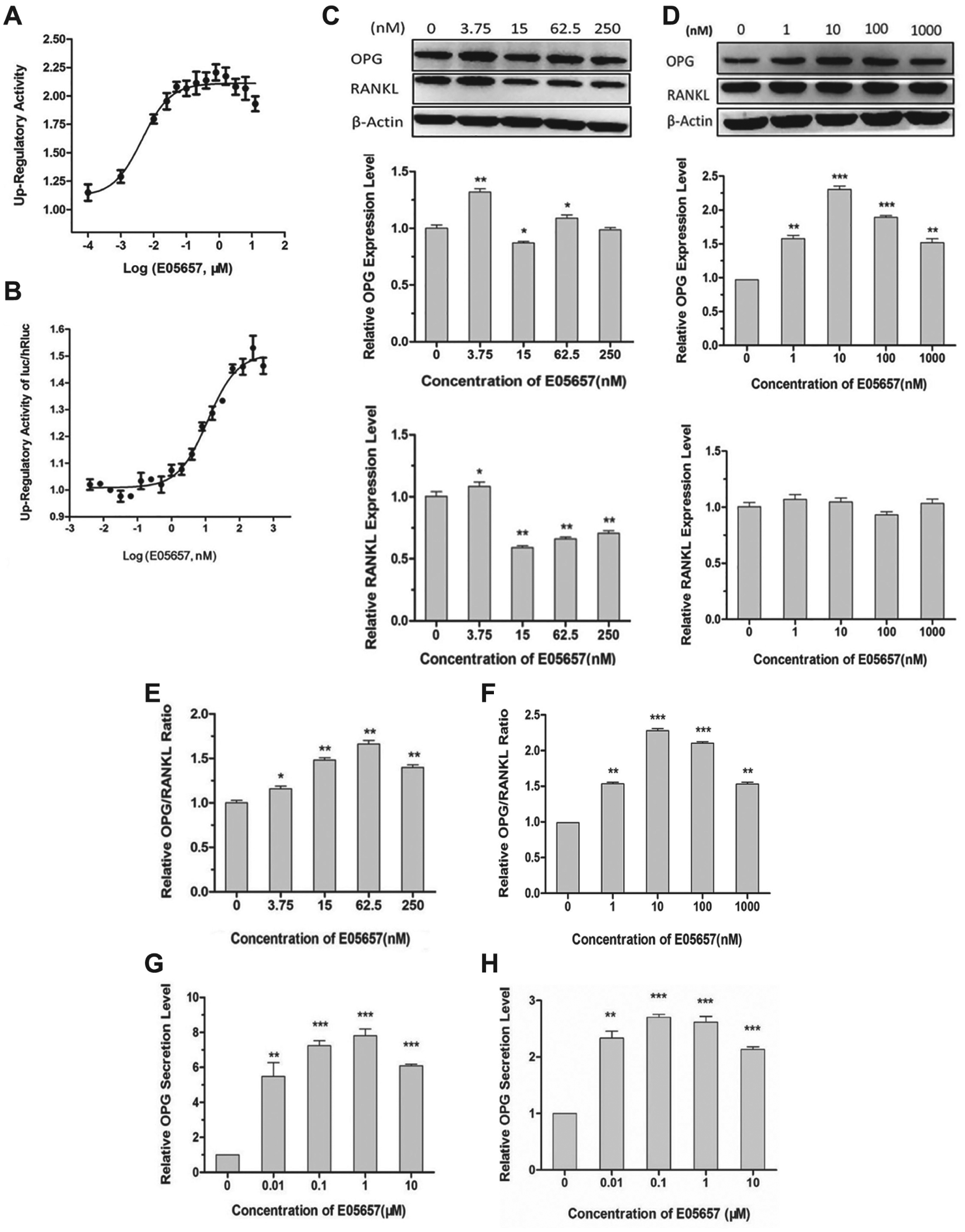
The effects of E05657 on the expression levels of OPG and RANKL, the ratio of OPG/RANKL, and the secretion level of OPG protein in osteosarcoma cells and preosteoblasts. (
E05657 upregulates the ratio of OPG/RANKL in osteoblastic cells
After treatment with different concentrations of E05657, Western blot analysis revealed that the ratios of OPG/RANKL protein levels in both human U-2OS osteosarcomas and mouse MC3T3-E1 preosteoblasts were significantly upregulated in a dose-dependent manner, and the upregulatory rates were up to 1.7-fold or 2.25-fold with 62.5 or 10 nM E05657 treatment, respectively ( Fig. 3E , F ). However, the changes of OPG and RANKL expression levels appeared to be inconsistent between the two types of cells. In U-2OS osteosarcomas, the OPG expression level was significantly increased but RANKL was only slightly enhanced by the treatment with 3.75 nM E05657, and then both OPG and RANKL expression levels were decreased along with the increment of compound concentration, but the decline of RANKL was greater than OPG. In MC3T3-E1 preosteoblasts, the OPG expression level was obviously enhanced by E05657 in a concentration-dependent manner, but there was no statistical difference between the RANKL expression levels ( Fig. 3C , D ).
E05657 induces OPG secretion in osteoblastic cells
OPG is a secreted protein, and only secreted OPG can bind to RANKL to inhibit osteoclastogenesis. To evaluate the activity of a compound, its effect on OPG secretion is as important as that on OPG expression. 28 An ELISA was performed to assess changes of the OPG concentration in supernatants of cultures of human U-2OS osteosarcoma cells and mouse MC3T3-E1 preosteoblasts in the presence of E05657 at concentrations of 0, 0.01, 0.1, 1, and 10 μM for 48 h. OPG secretion in these cells was upregulated remarkably by E05657 treatment in a dose-dependent manner. The maximum increase in U-2OS cells was nearly eightfold, and nearly threefold in MC3T3-E1 cells after treatments with 1 and 0.1 μM E05657, respectively ( Fig. 3G , H ).
In E05657-treated U-2OS osteosarcoma cells, OPG expression was increased first, but then declined along with the increase in compound concentrations. This phenomenon could be explained by ELISA analysis of the supernatants of cell cultures. When treated with E05657 at higher than 10 nM, the secretion of OPG was enhanced by five- to eightfold. Because large amounts of OPG proteins were secreted to the medium, the intracellular OPG levels decreased accordingly. Although the intracellular levels of RANKL also decreased when treated with E05657 at a concentration higher than 10 nM, we did not detect any secreted RANKL in the supernatants of cell cultures (data not shown). These results indicate that E05657 could induce OPG expression and secretion, and reduce the expression level of RANKL in U-2OS osteosarcomas, thereby increasing the OPG/RANKL expression ratio in these cells. In MC3T3-E1 preosteoblasts, both OPG expression and secretion levels were increased by more than twofold, but the intracellular RANKL level was not modulated by E05657. Similarly, we did not detect any secreted RANKL in the supernatant of MC3T3-E1 cell cultures (data not shown). Thus, E05657 increases the OPG/RANKL ratio in MC3T3-E1 preosteoblasts. The above results indicate that E05657 has the potential to inhibit osteoclastogenesis, which was confirmed by TRAP staining.
E05657 inhibits NFATc1 expression and osteoclast differentiation via its effects on osteoblastic cells
After culturing in osteogenic medium for 3 days, we added compound E05657 to MC3T3-E1 cell cultures at concentrations of 0, 15, 62.5, 250, and 1000 nM. After treatment for 6 days, the CMs were collected and mixed with OM1 (DMEM supplemented with 30 ng/mL RANKL) at a 1:1 ratio. The mixed media were added to mouse Raw264.7 mononuclear macrophages, followed by culture for 3 days. The expression levels of NFATc1 in Raw264.7 cells were decreased by about half after the treatment ( Fig. 4A ). The CMs mixed with OM2 (DMEM supplemented with 100 ng/mL RANKL) at a 1:1 ratio were also added to Raw264.7 cells. After 3 days, the osteoclastic differentiation levels were tested by TRAP staining. As a result, the cells treated with CM from control MC3T3-E1 culture supernatant (without E05657) had differentiated into TRAP-positive giant multinuclear osteoclasts, and about 200 osteoclasts were detected per well. However, after addition of CM from MC3T3-E1 supernatants that were treated with 15–250 nM E05657, very few multinuclear cells (less than 20% of control) appeared, and almost no multinuclear cells were observed after addition of CM from MC3T3-E1 culture supernatant treated with 1000 nM E05657 ( Fig. 4B ).
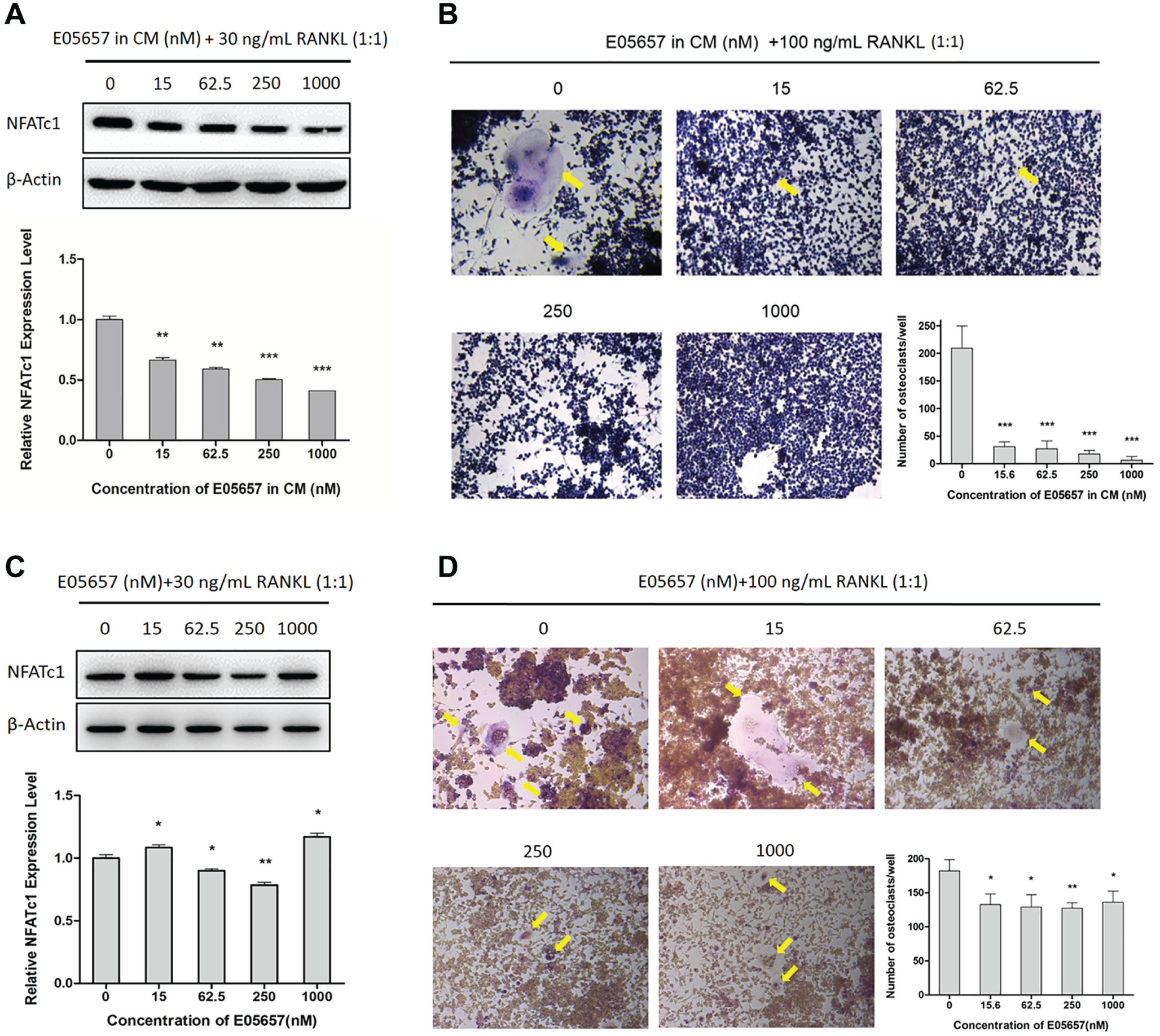
Inhibition of NFATc1 expression and osteoclastic differentiation by CMs from E05657-treated MC3T3-E1 culture supernatants and direct E05657 treatment. (
These results show that the CMs from E05657-treated MC3T3-E1 cells could inhibit osteoclastogenesis in vitro, but the reduction in NFATc1 expression and osteoclast differentiation could be due to the presence of E05657 in the CMs. To investigate if E05657 could directly inhibit osteoclastogenesis, we mixed osteogenic mediums containing 0, 15, 62.5, 250, and 1000 nM E05657 with OM1 or OM2 at 1:1 ratios. Then Raw264.7 cells were exposed to the mixtures for 3 days, and Western blotting and TRAP staining revealed that both NFATc1 expression and osteoclast differentiation seemed to be decreased by E05657 direct treatment. However, the levels of these decline were much lower than that induced by the CMs from E05657-treated MC3T3-E1 cells; the level of NFATc1 expression and the number of osteoclasts were only reduced to no less than 80% or 70% of the controls, respectively ( Fig. 4C , D ).
We have demonstrated that E05657 could stimulate osteoblastic cells to increase the expression and secretion of OPG, but no increase was observed for RANKL expression. Therefore, the OPG/RANKL ratio was enhanced remarkably. Besides, we found that the CMs from MC3T3-E1 cells treated with E05657 could significantly reduce NFATc1 expression in Raw264.7 cells and inhibit their differentiation into osteoclasts. We speculate that E05657 stimulates MC3T3-E1 cells to secret OPG, which inhibits the association of RANKL in OM with RANK on the surface of Raw264.7 cells, causing the reduction of the NFATc1 expression level, because NFATc1 expression is induced by RANKL-RANK interaction. 29 Therefore, osteoclastogenesis was also inhibited because NFATc1 is a master transcription regulator of osteoclastic differentiation. 30 Although direct E05657 treatment could partly reduce NFATc1 expression and osteoclast differentiation, the effect was far less compared with the treatment with the CMs. Thus, we reason that the inhibition of osteoclastogenesis should be mainly attributed to abundant OPG secreted by MC3T3-E1 cells, a result of stimulation by E05657.
In conclusion, we established and evaluated a novel HTS system that can be used to identify upregulators of the OPG/RANKL expression ratio with potential for antiosteoporosis treatment. The established assays can be used to assess the effect of compounds on OPG expression and the OPG/RANKL ratio through changes of firefly and renilla luciferase activities. We screened 8160 compounds and 5 compounds were identified using this two-step HTS system. The candidate with the most potential, E05657, was validated for its effect on the increase of the OPG/RANKL ratio and OPG secretion in human and murine osteoblastic cells. This compound obviously suppresses osteoclastogenesis in vitro. Therefore, E05657 is a lead compound with a superior potential for antiosteoporosis effects and is worthy of further investigation as a novel antiosteoporosis drug.
Footnotes
Acknowledgements
We wish to express our thanks to Professor Yanchang Wang (College of Medicine, Florida State University, Tallahassee) for his help in English revision.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Key New Drug Creation and Manufacturing Program (2012ZX09301002-003) and the National Natural Science Foundation of China (30901811).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.