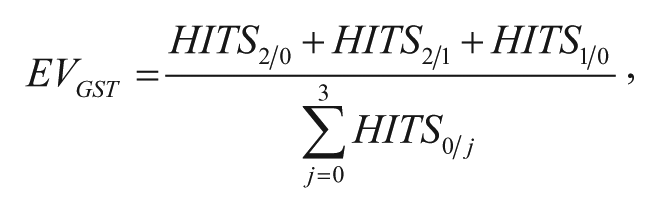Abstract
In high-throughput screening (HTS) campaigns, the binding of glutathione S-transferase (GST) to glutathione (GSH) is used for detection of GST-tagged proteins in protein-protein interactions or enzyme assays. However, many false-positives, so-called frequent hitters (FH), arise that either prevent GST/GSH interaction or interfere with assay signal generation or detection. To identify GST-FH compounds, we analyzed the data of five independent AlphaScreen-based screening campaigns to classify compounds that inhibit the GST/GSH interaction. We identified 53 compounds affecting GST/GSH binding but not influencing His-tag/Ni2+-NTA interaction and general AlphaScreen signals. The structures of these 53 experimentally identified GST-FHs were analyzed in chemoinformatic studies to categorize substructural features that promote interference with GST/GSH binding. Here, we confirmed several existing chemoinformatic filters and more importantly extended them as well as added novel filters that specify compounds with anti–GST/GSH activity. Selected compounds were also tested using different antibody-based GST detection technologies and exhibited no interference clearly demonstrating specificity toward their GST/GSH interaction. Thus, these newly described GST-FH will further contribute to the identification of FH compounds containing promiscuous substructures. The developed filters were uploaded to the OCHEM website (http://ochem.eu) and are publicly accessible for analysis of future HTS results.
Introduction
The glutathione S-transferase (GST), a 211 amino acid protein translating into a 26 kD protein, plays an important role in catalyzing the conjugation of electrophilic substrates to glutathione (GSH) for detoxification of xenobiotics. 1 In biochemical research, this protein is widely used for the production of recombinant proteins from crude bacterial lysates. 2 Therefore, the GST DNA sequence of the parasitic helminth Schistosoma japonicum is frequently integrated into different expression vectors, resulting in a fusion protein that usually carries an N-terminal GST-tag. 3 Using the GST-tag for protein purification has beneficial effects as it not only promotes higher expression levels of the recombinant protein but can also act as a chaperone to improve proper protein folding. Furthermore, it enhances the expression of a soluble protein rather than protein accumulation in inclusion bodies. 2 In addition, the GST fusion protein can be purified without denaturation or use of mild detergents via binding to its natural substrate, the tripeptide glutathione (Glu-Cys-Gly).2,3
GST fusion proteins have been successfully included in a variety of biological applications including immunological studies and vaccine production, 4 structure determinations,5,6 protein-protein and protein-DNA interaction studies, as well as other biochemical analysis. 2 Moreover, GST fusion proteins have been successfully employed in biochemical high-throughput screening (HTS) using the Alpha (Amplified Luminescent Proximity Homogenous Assay) technology (PerkinElmer, Waltham, MA).7–9 The AlphaScreen technology is a homogenous bead-based assay to study biomolecular interactions in vitro. Briefly, appropriate donor and acceptor beads bind to the corresponding tagged protein, and upon interaction of the proteins or other binding partners (e.g., peptides, DNA, RNA), singlet oxygen is transferred from the donor bead to the acceptor bead, thereby producing a luminescent signal. Compared with other technologies, AlphaScreen offers advantages as it allows a large distance of up to 200 nm between donor and acceptor beads and therefore is suitable for the use of larger proteins compared with, for example, Förster resonance energy transfer (FRET) technology, which is limited to 10 nm distance between the donor and acceptor. 9 However, no matter which technological platform is used for conducting HTS campaigns, false-positive Hits may arise that either interfere nonspecifically with one of the assay components or with assay signal generation and/or detection. Therefore, detection of frequent hitter (FH) molecules is one of the most important steps during the hit selection process in order to progress only those compounds that are yielding genuine activity. In the AlphaScreen technology, interfering compounds might either act as singlet oxygen quenchers, color quenchers (also known as inner filters), light scatterers, or acceptor bead competitors, and the AlphaScreen TruHits Kit enables the identification of some of these artifacts. In previous studies, Baell and Holloway 10 published the Pan-Assay-Interference Compounds (PAINS) filters to identify FH classes being promiscuous substructures of chromophores, redox-active, chelating, and protein reactive compounds.10,11 Subsequently, efforts have been made to further extend this knowledge, and chemoinformatic filters that identify FHs, which interfere with the AlphaScreen chemistry (AlphaScreen FHs) or with the His-tag/Ni2+-NTA interaction (His-tag FHs), have been developed and made publicly available. 12
To establish filters that would specifically detect GST-FH compounds, we analyzed data of two different AlphaScreen HTS campaigns based on the GST/GSH interaction and three independent control screenings, with each employing 25,000 small molecules. From these studies, 53 compounds were verified as being specific GST-FHs in a confirmatory AlphaScreen assay that made use of a GST-His fusion protein. These small molecules were further analyzed in counter assays using the TruHits Kit (PerkinElmer) and a Biotin-His peptide based on AlphaScreen experiments demonstrating that these compounds are indeed specific for GST/GSH interaction. These data were the basis for developing novel chemoinformatic filters that would help to identify GST-FH in GSH-coupled matrices. Importantly, the identified GST-FH compounds did not interfere with the anti-GST antibody–based technologies, namely, the time-resolved FRET (TR-FRET) assay and antibody-based pull-down, suggesting a difference in the bias of FHs toward GSH and anti-GST antibody–based detection of GST.
Materials and Methods
Materials
Biological Reagents
General biological reagents not listed elsewhere (e.g., buffers) were purchased from Sigma-Aldrich (St. Louis, MO) and Roth (Germany) and were of the highest available purity.
Library and Compounds
The HTS compound library comprised a diversity set of 25,000 compounds. 12 Reordered GST-FH compounds were purchased (1) from ChemDiv (San Diego, CA): GST-FH.1 (C301-6417), GST-FH.2 (M213-0587), and GST-FH.3 (M213-1079) or (2) from ENAMINE Ltd. (Princeton, NJ): GST-FH.4 (T6730144).
AlphaScreen Reagents
AlphaScreen (PerkinElmer) made use of GSH donor beads (product No. 6765300), Strep-Tactin Alpha donor beads (product No. AS106D), streptavidin donor beads (product No. 6760002), nickel chelate (Ni2+-NTA) donor beads (product No. AS101D), histidine (nickel chelate) detection kit (product No. 6760619C), C-Myc detection kit (product No. 6760611C), and the TruHits Kit (product No. 6760627D).
TR-FRET Reagents
TR-FRET (Cisbio, France) made use of anti–GST-XL665 (product No. 61GSTXLB) and anti–His-K (product No. 61HISKLB).
Methods
Protein Synthesis
For recombinant protein expression of the GST-His fusion protein, the DNA sequence for 6xHis-tag was introduced into the pGEX-4T-1 vector (GE Healthcare Life Sciences, Munich, Germany) and expressed in Escherichia coli strain BL21 RIPL upon induction using 1 mM isopropyl-β-D-thiogalactopyranoside at an OD600 = 0.6 to 0.8. Bacterial pellets were lysed in lysis buffer containing phosphate-buffered saline (PBS) buffer pH 7.4, 0.5 mg/mL lysozyme and protease inhibitor cocktail tablet without EDTA (Roche Diagnostics, Mannheim, Germany). GST-His protein was bound to GSH sepharose 4 Fast Flow (GE Healthcare Life Sciences), followed by washing and elution of GST-His protein using 50 mM reduced GSH (Sigma-Aldrich, Taufkirchen, Germany). Desalting of the eluted protein was accomplished in PBS using HiTrap desalting columns (GE Healthcare Life Sciences) and the ÄKTA purifier system (GE Healthcare Life Sciences) to remove the remaining GSH peptides that would interfere with the assay detection technology.
Hit Selection Process
Of the data of five independent Alpha-Screen HTS campaigns (two assays involving GST-tag for detection and three non–GST-containing assays), all compounds were selected that reduced the AlphaScreen signal by more than 30% in both GST-containing assays. Subsequently, these compounds were analyzed for AlphaScreen-FH and His-FH. 12 Hits positive for AlphaScreen-FH or His-FH were directly excluded. For identification of GST-FH compounds out of the hit list, the mean AlphaScreen signal values were calculated for (1) Hit Set 1 and Hit Set 2 as GST containing screens and for (2) Hit Set 3, Hit Set 4, and Hit Set 5 as non–GST-containing screens. In a first selection process, each single value of (1) needed to be lower than 70%, whereas at the same time, each single value of (2) needed to exceed 80% for the same compounds. In a second criterion, the difference of the means [(2) – (1)] needed to be higher than 30% to define the Hit as a true GST-FH compound.
AlphaScreen Assays (Primary Screening; Confirmatory and Counter Screening)
Screening of 25,000 compounds of five different HTS protein-protein interaction campaigns was performed using the AlphaScreen technology as previously described. 12
Confirmatory AlphaScreen assays of 58 identified GST-FH compounds were accomplished using 30 nM GST-His fusion protein, GSH donor beads, and nickel-chelate acceptor beads in 10-point titration experiments.
For counter screening, the TruHits Kit (PerkinElmer) was conducted in 10-point titrations. 12 As an additional counter screening strategy for AlphaScreen assays, the compounds were tested in 10-point titrations and incubated with 30 nM Biotin-His peptide for 1 h and detected via streptavidin donor beads and nickel-chelate acceptor beads.
All data were processed using Excel (Microsoft Corp., Redmond, WA) and visualized using Prism (Graphpad Software, La Jolla, CA). IC50 calculations were performed in ActivityBase XE (IDBS, Guildford, UK).
TR-FRET Assays
TR-FRET counter assays were performed as previously described 12 using 2 nM of GST-His protein. All data were processed using Excel (Microsoft Corp.) and visualized using Prism (Graphpad Software).
Pull-down Assays
Glutathione Sepharose 4B (GE Healthcare Life Sciences) or Ni Sepharose 6 Fast Flow (GE Healthcare Life Sciences) pull-downs were performed in assay buffer (1× PBS, 5% glycerol, 0.1% Triton-X100, protease inhibitor cocktail tablet without EDTA [Roche Diagnostics]) by preincubation of 250 ng GST-His fusion protein with the individual GST-FH compounds for 30 min. Upon addition of the respective beads (GSH or nickel) and incubation for an additional 2 h, washing was conducted six times using 1× PBS, 5% glyerol, 0.5% Triton-X100, protease inhibitor cocktail tablet without EDTA (Roche Diagnostics). Bound protein was eluted with 2× Rotiload (Roth). GST-Trap_A immunoprecipitation of the GST-His fusion protein was performed according to the protocol provided by the manufacturer (Chromotek, Planegg, Germany). Detection of the pulled GST-His protein was performed by sodium dodecyl sulfate– polyacrylamide gel electrophoresis followed by Western blot analysis using the GST antibody No. 2622 (Cell Signaling Technology, Beverly, MA).
Identification of Additional Experimental Data Using PubChem
For the validation of the developed substructural filters, the PubChem BioAssay database
13
was used. A query line <AlphaScreen gst AND (pcassay_protein_target[filt]) AND (Screening[filt])AND 1000:10000000[Total Sid Count]> retrieved only one primary screening assay implemented using AlphaScreen technology and GST-tagged protein
14
(
Identification of Significant Filters
For development of substructural filters, 53 molecules experimentally identified as GST-FHs were used. Structural analysis involved a common scaffold search, structural similarity grouping, as well as analysis of chemical activity based on the known reactive functional moieties. 15 To estimate the quality of the filters, we used the assessment technique 10 used in our previous publication. 12 Considering that we had a smaller number of assays compared with both previous studies, we calculated the enrichment value (EVGST) with the following equation:
where
Results and Discussion
Detection and Confirmation of GST-FH Compounds from Multiple AlphaScreen HTS Campaigns
To identify GST-FH compounds that interfere with the GST and GSH binding, the data of five previous AlphaScreen-based HTS campaigns, each exploiting different target protein-protein interactions ( Fig. 1 ), were used to perform an in-depth analysis. These screens included two assays, both of which involved the GST/His-tag combination (Hit set 1 and 2). The GSH donor beads bound the GST-tagged protein, and the His-protein was detected via nickel-chelate acceptor beads. The control screens used the combinations of StrepII/His-tag (Hit set 3), biotin/His-tag (Hit set 4), and myc/His-tag (Hit set 5; Fig. 1 ). For identification of GST-FHs within the 25,000 tested small molecules for each assay combination, data were analyzed as follows: In a first selection step, GST-FH had to reduce the AlphaScreen signal by more than 30% in both GST-containing assays, whereas, at the same time, the signals for all non–GST-containing assays needed to be higher than 80% (i.e., GST <70% and non-GST >80% AlphaScreen signal). The second step included a criterion for GST-FHs when the difference of the mean AlphaScreen signal of all non–GST-containing assays and all GST-containing assays exceeded 30% (i.e., non-GST – GST > 30%; Fig. 1 ). Importantly, for flagging the respective compound as a GST-FH, both criteria needed to be fulfilled at the same time. Of 25,000 compounds, we were able to extract 58 small molecules that achieved our predefined two-step criteria. Notably, these were not overlapping with compounds that were already classified as general FHs of the AlphaScreen technology or His-FH interfering with the His-tag/Ni2+-NTA interaction. 12
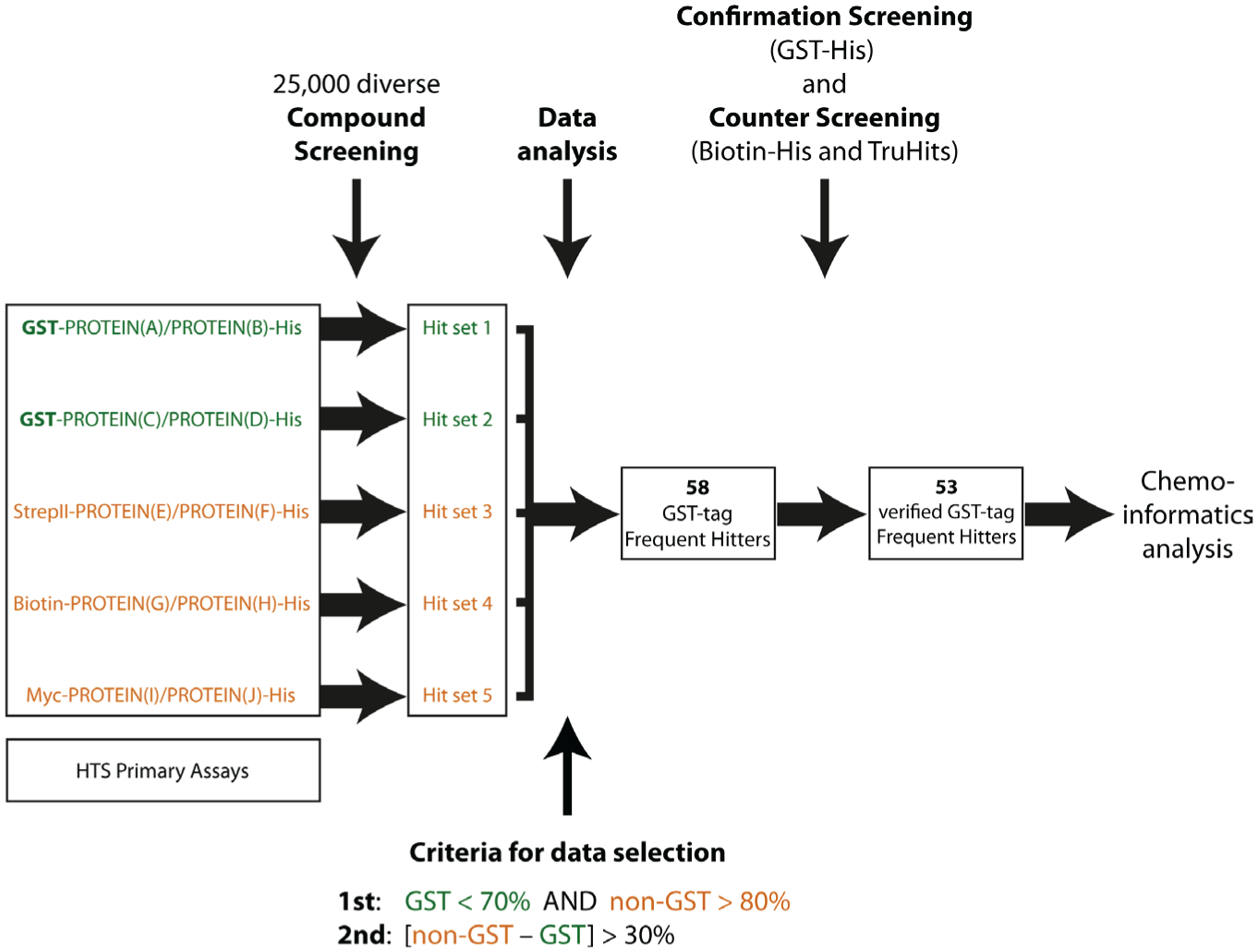
Overall workflow of this study. In five independent high-throughput screening campaigns comprising different protein pairs (A/B, C/D, E/F, G/H, and I/J), 25,000 compounds were tested using the AlphaScreen technology involving various detection systems: glutathione S-transferase (GST)/His (Hit set 1 and 2), StrepII/His (Hit set 3), biotin/His (Hit set 4), and myc/His (Hit set 5). For identification of GST–frequent hitter (FH) compounds, a two-step selection process was taken as a basis: (1) GST-FH had to reduce the signal to less than 70% in Hit sets 1 and 2, whereas signals of Hit sets 3, 4, and 5 needed to exceed 80%; (2) the difference of the signal means of non-GST– and GST–containing assays needed to be greater than 30%. Application of these two criteria led to 58 GST-FH. Fifty-three of these compounds were proven to be true GST-FHs by confirmatory and counter AlphaScreen assays and served as the basis for chemoinformatic analysis.
Subsequently, these 58 compounds were tested in 10-point titration in AlphaScreen assays starting from 50 µM as the highest compound concentration ( Fig. 2A ). Here, the small molecules were examined using a recombinantly expressed GST-His fusion protein, consisting of the GST protein with a C-terminal His-tag that was detected via GSH donor and nickel-chelate acceptor beads. In total, 5 of the 58 tested compounds did not affect the GST-His assay ( Fig. 2A , orange label), and 53 compounds indeed impaired the GST-His AlphaScreen signal with decent activity at least at the maximum concentration of 50 µM ( Fig. 2A , green label). For 37 of 53 active compounds, IC50 values could be calculated, and these values are depicted in Table 1 . Furthermore, biotin-His peptide (streptavidin donor beads and nickel-chelate acceptor beads) as well as the TruHits Kit (streptavidin donor beads and biotinylated acceptor beads) served as controls that should not be affected by the compounds. Although most of the compounds reduced the AlphaScreen signal in the GST-His assay at 25 µM compound concentration ( Fig. 2B ), all 58 tested compounds did not interfere with the biotin-His peptide assay nor affected the TruHits assay ( Fig. 2C , D ). This is in fact very important as it verifies that these compounds do not interfere with the AlphaScreen technology itself, nor do they impair the His-tag/Ni2+-NTA binding.
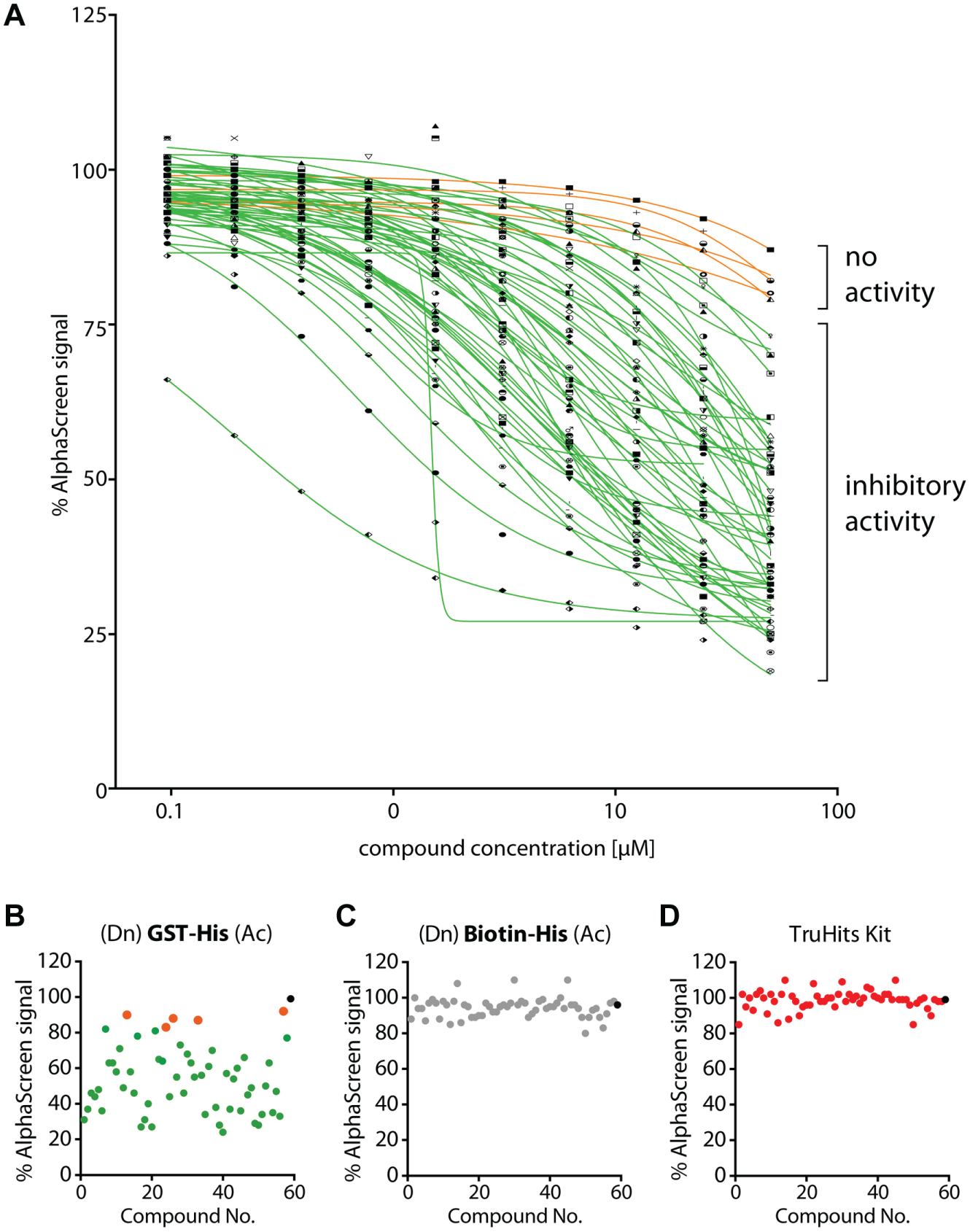
Analysis of glutathione S-transferase (GST)–frequent hitter in confirmatory and counter AlphaScreen assays. (
IC50 values of glutathione S-transferase (GST)–frequent hitter.
The IC50 values and molecular structures are freely available for download at http://ochem.eu/article/93878.
Thus, we provide an experimental proof that 53 compounds certainly interfered with the GST/GSH interaction with different levels of severity and can herewith be classified as GST-FHs. These compounds were then subjected to a chemoinformatic analysis to discover substructures that are responsible for disrupting the GST/GSH interaction.
Preliminary Data Analysis
In the preliminary analysis, 53 GST-FHs were screened against existing substructural filters recognizing promiscuous
16
and PAINS compounds
10
using the ToxAlerts tool
17
of the OCHEM (www.ochem.eu) platform.
18
Eight compounds were recognized as PAINS and three as promiscuous compounds (
Extension of PAINS Filters
1. Two GST-FHs among PAINS-recognized compounds were identified by the “Dhp_bis_amino_CN” filter developed for aminocyanothiopyrans, 10 and this filter was reported to have a very high EV (1400%). Screening of our data has revealed that this filter was also highly specific to GST-FHs (EV = 230%). Because none of the analyzed compounds contained oxa- or aza-analogues of the thiopyran core, we added the corresponding PAINS filter to GST-FH-S without any modifications. One compound with a structurally similar dithione group was also found among GST-FHs. However, because there were no other compounds with this group present either in the Helmholtz Zentrum München (HMGU) or in the PubChem data sets, it was not possible to estimate reliably the contribution of the variance of substituents within this chemical class to its false activity in GST/GSH–employing bioassays (further, we would refer to it as GST promiscuity). Therefore, we created an individual singleton FH filter for this compound.
2. A “Keto_phenone_A” PAINS filter for 9-fluorenone azaderivatives recognized one GST-FH out of three compounds with this structural moiety. This filter was extended to cover all three compounds (
3.Promiscuous o-cyanothiopyridinones were correctly recognized as FHs by the “Cyano_pyridone_A” PAINS filter, which was added to GST-FH-S without modification. The p-cyanopyridinone moiety was also presented as a part of the fused cyclic system for one GST-FH, and this compound provided more than 50% signal reduction in GST-involving AlphaScreen assays and no signal reduction in GST-free assays. However, there were 16 highly structurally similar p-cyanopyridinones among substances that were inactive in GST/GSH-containing bioassays (further referred to as “clean” substances). Thus, a selective filter to identify this FH was created. Another GST-FH containing a pyridinone moiety with an electron-withdrawing substituent was recognized by the PAINS filter “Het_65_E.” One additional GST-FH with a pyridinone fragment was found among GST-FHs. We were not able to extend any of the aforementioned filters for the (thio)pyridinones, and a separate filter was created for this singleton (see “Pyranopyridinones” filter in
4. A PAINS filter “Quinone_A” recognized the GST-FH–containing anthraquinone scaffold. Quinones are well-known electrophilic agents containing an activated double bond, which is capable of binding covalently to nucleophilic moieties. Accordingly, it can be assumed that they bind to the thiol group of the GSH-modifying GST substrate and prevent anchoring of tagged proteins. However, in anthraquinones, the quinoid structure is a part of the aromatic system and does not behave as an activated double bond. Analysis showed that 24 molecules comprised anthraquinone-like structures, which did not provoke any decay in their fluorescent signals. In our opinion, the GST promiscuity of this FH is not solely governed by the presence of a quinone moiety (section “Remarks on the Filters”). Therefore, the corresponding PAINS filter was not incorporated into our GST-FH-S. Instead, a singleton-like filter was created to cover this FH. It can be further extended once more diverse structural information appears.
5. A PAINS filter “Ene_five_het_H” identifies thioazolones with exocyclic double bonds (hidden Michael acceptors). Among the studied GST-FHs, an oxa-analogue also appeared. The filter was augmented to cover the latter and was saved under the shared name “Pyrazolones.”
6. A PAINS filter “Catechol_A” recognized the only GST with a catechol fragment but showed a rather low EV value (17%). We modified the filter by including the molecular scaffold to which the catechol ring belongs (section “Remarks on the Filters”).
7. Although the PAINS filter “Thio_carbonate_A” has not recognized GST-FHs by itself, it describes a molecular fragment that is highly structurally similar to a GST-promiscuous benzoxathiolone. After a slight modification, the filter was added to GST-FH-S as well.
The aforementioned PAINS filters and/or their extensions (seven in total) were included in GTS-FH-S. Although some of these filters were simply duplicates of PAINS filters, their inclusion in the GST-FH collection could provide an important hypothesis to the users about the mode of action of FH and can guide them to design counter screens to validate the hits.
8. The “artifacts” data set contained many aromatic sulfonamides having common fragments depicted in
Fig. 3A
(see also

Chemoinformatic analysis. (
The filters for promiscuous substances
16
shifted out three GST-FHs (
Analysis of FH Not Covered with Existing FH Filters
After applying 14 aforementioned filters, 35 GST-FHs remained undetected. For development of the new filters, we assumed that the GST promiscuity of the studied substances could be due to one of the following reasons:
Occupation of GSH binding site of GST (G-site) due to structural similarity between the GSH substrate and studied substances. This mechanism can be identified by a search of ligands structurally resembling GSH.
Irreversible conjugation of a molecule with Cys-residues of immobilized GSH leading to the inability of recombinant proteins to anchor to bead surfaces. This mechanism applies to reactive species with sufficiently strong electrophilic properties.
Reversible hydrogen bonding of the molecule to sites important for GST-GSH interaction. This mechanism applies to substances with strong hydrogen bond donating/accepting moieties.
Analysis of structural similarity showed that there were no molecules structurally similar (Tanimoto <60%) to GSH among the studied GST-FHs, thus eliminating the first hypothesis.
To identify reactive species with prominent electrophilic features, we used filters to recognize potential electrophilic agents (PEAs)
19
available in ToxAlerts.
17
Application of PEA filters showed that 7 of 35 remaining GST-FHs comprised electrophilic moieties (
Analysis of FH Based on Hydrogen-Bond Formation
The potential to form hydrogen bonds with the receptor was analyzed as the last hypothesis to provide a mechanistic-based development of FHs.
Among the GST-FHs, the most prevalent compounds were 4-heteryl substituted thiazoles comprising the common structure (I) depicted in
Fig. 3B
. Compounds containing this structure were frequently found among the artifacts of AlphaScreen technology as well as among active compounds in the primary screen assay. Detailed structural examination divided them into three main subgroups containing common substructures (
Fig. 3B
,
The GST promiscuity of this type of GST-FHs is provoked by specific noncovalent interactions between FHs and thiol groups of immobilized GSH rather than covalent binding of this group with proteins. We came to this assumption after structural comparison of GST-FHs and “clean” compounds. Indeed, substituent in the fourth position of a thiazole ring likely does not play any role on GST promiscuity because it varies within GST-FHs. A fragment, which is presented in all FHs, includes thiazolyl-substituted aromatic secondary amine ( Fig. 3B , Ic), and it can form noncovalent bonds with the Cys thiol-group. The Cys thiol-group has a pKa = 9.1, 22 and in the experiment condition (pH = 7.4), the neutral form should be dominated. Thus, the neutral form could participate in hydrogen bond formation, as proposed in Figure 3C/i and Figure 3C/ii (potential interactions are shown by the dashed lines).
The first hypothetical mechanism assumes formation of an H-N···H-S hydrogen bond and S···S interactions. The latter are widely distributed in proteins and crystals.23,24 The bond H-N···H-S is formed by secondary amine nitrogen with two aromatic substituents ( Fig. 3C/i ). H-donating properties of this type of the nitrogen atom are decreased due to a steric hindrance of its electron pair, and so this type of interaction may not be sufficiently strong to form the complexes.
The second proposed mechanism is based on N···H-S and S···H-N interactions, which are also observed in crystals of proteins and organic molecules.25–29 However, because of the weak hydrogen bond accepting properties of the S atom in the Cys group, an S···H-N bond is likely to be rather weak. Thus, this hypothesis is also not very likely.
The third hypothesis is based on the assumption that, despite that the pKa value is higher than that of the experiment value, the Cys thiol group can be still ionized. It is known that a local microenvironment created by the presence of a large number of polar groups on bead surfaces (i.e., thiol, carboxyl, and amino groups) can shift the pKa of the Cys thiol group, increasing the concentration of thiolate form.30–32 Hence, the lone electron pair of thiolate-S can participate in an anion hydrogen bond with acidic hydrogen of the amino group, whereas core sulfur electrons form donor-acceptor S···S–specific interactions ( Fig. 3C/iii ). This hypothesis looks the most convincing for us, because anion-hydrogen bonds are strong interactions and, being additionally fortified by specific S···S contacts, should form a stable complex.
The aforementioned three hypotheses are based on the observations that about 60% of the studied GST-FHs contain thio-functionalities in their structure. Indeed, in some cases, corresponding oxo-analogues of the studied hitters do not interfere with GST/GSH–involving assays (
Verification of GST-FH Compounds in TR-FRET and Pull-Down Assays
For further investigation, four compounds were selected using the following criteria: (1) specific inhibition of the GST/GSH interaction, (2) IC50 below 25 µM ( Fig. 2A ; Table 1 ), and (3) belonging to different classes of small-molecule families: GST-FH.1 (M1111171) belonging to the family of the piperazinediones, GST-FH.2 (M1217802) and GST-FH.3 (M3200831) representing the 4-heteryl-thiazoles family, as well as GST-FH.4 (M1231217) as a member of the sulfonylamide_A group ( Fig. 4A ). All four GST-FH were tested in a dose-response study on the GST-His AlphaScreen assay, thereby exhibiting decent inhibition of the AlphaScreen luminescence signal ( Fig. 4B ). In contrast, when adding the GST-FH compounds in the same concentration range to the biotin-His peptide, production of a stable AlphaScreen signal was achieved that was completely unaffected ( Fig. 4C ). This dose-response verification again underlined that the tested compounds are GST-FHs rather than general AlphaScreen or His-FH compounds.
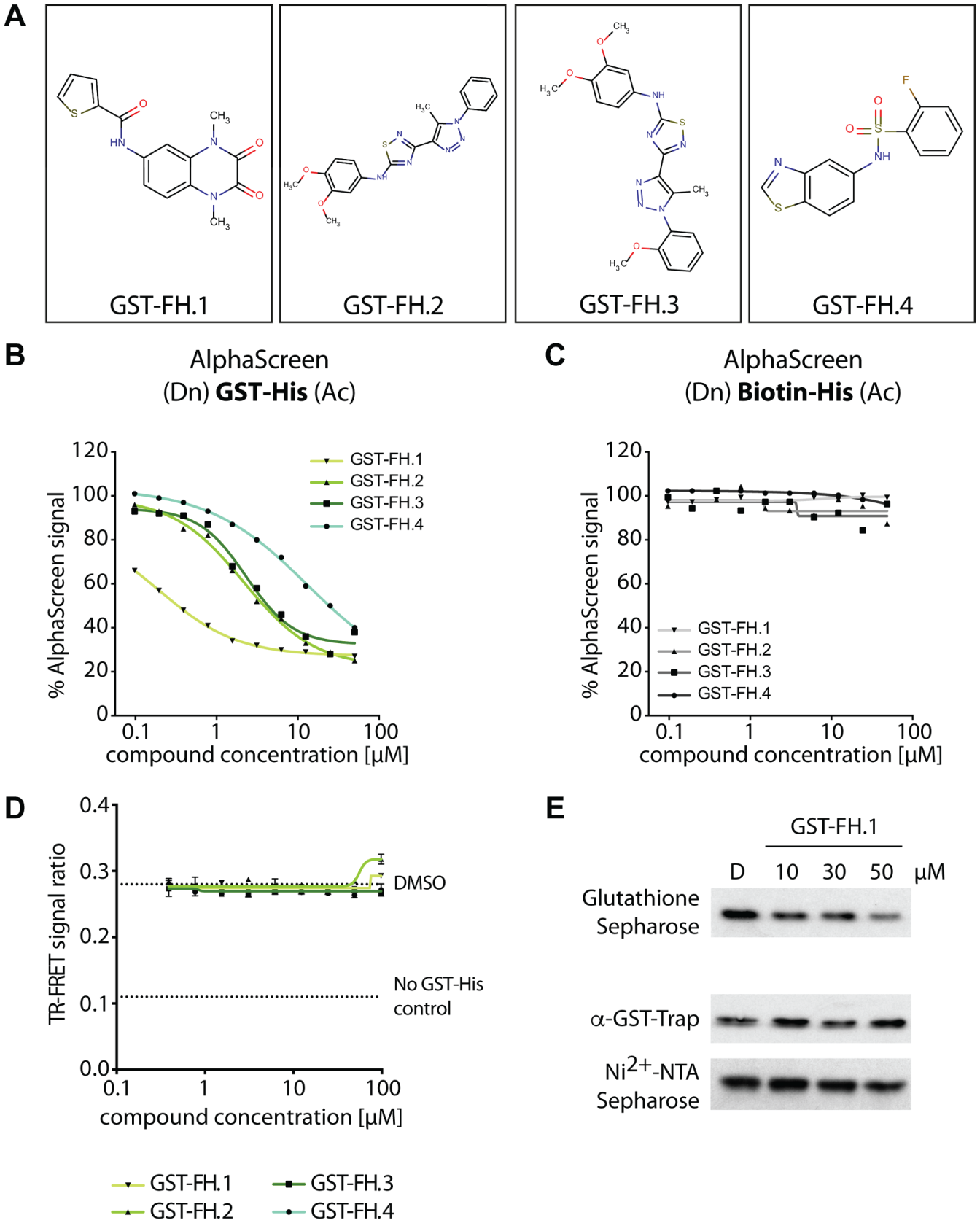
Analysis of selected compounds in the AlphaScreen assays, time-resolved Förster resonance energy transfer (TR-FRET), and pull-down assays. (
To verify the interference of the GST-FH compounds with the GST/GSH interaction, TR-FRET assays that detect the GST-His fusion protein via an anti-GST antibody rather than by GSH-coupled matrix were performed. Therefore, the GST-His fusion protein was incubated with the respective GST-FH compound up to 100 µM and detected via a MAb anti-GST-XL665 (acceptor) and a Mab anti-His-K (donor). In contrast to the AlphaScreen experiments, even at a concentration of 100 µM, all four analyzed compounds did not impair the binding of the anti-GST antibody to the GST-tag ( Fig. 4D ), suggesting a different binding mode of GSH donor beads and anti-GST antibody to the GST-tag. Of note, GST-FH.1 and GST-FH.2 appear to be slightly autofluorescent at the wavelength of 615 nm at high concentrations ( Fig. 4D ).
For further verification, compound GST-FH.1 with the most pronounced inhibition activity on GST/GSH binding in AlphaScreen was selected to prove its inhibitory impact on the GST/GSH interaction in pull-down assays, a very prominent assay type for analyzing protein-protein interactions in low-throughput. Importantly, this assay technology is very distinct from the AlphaScreen technology. To this end, the GST-His fusion protein was incubated with the GST-FH.1 compound and subsequently extracted using GSH-sepharose beads, GST-Traps (antibody-based), or nickel-sepharose beads. The latter two matrices served as controls. Pull-down of GST-His was dose-dependently reduced when applying GST-FH.1 to a GSH-sepharose–based pull-down assay ( Fig. 4E ). Differences in the potency of GST-FH.1 between AlphaScreen and pull-down experiment are due to different amounts of protein and bead input as well as the distinct nature of the assay technologies. In contrast, pull-down of the GST-His by either antibody-based GST-Trap or nickel-sepharose beads was not impaired by GST-FH.1, again proving that this small molecule specifically inhibits the GST-GSH interaction ( Fig. 4E ).
In HTS campaigns, proteins or enzymes are often tagged to have the ability to use different matrices for output detection. Here, various tags such as His, StrepII, or GST are commonly used. The major problem with these tags is that many small-molecule compounds affect the binding of the tag to the corresponding matrix, thereby generating false-positive signals. 12 These compounds are considered to be FHs.10–12 In this study, we wanted to identify and describe novel chemoinformatic filters that detect FHs of the GST/GSH interaction.
In total, 13 GST-FHs contained pyrimidinone fragments. The sensitivity of these compounds to the GST/GSH system was strongly affected by the core structure as well as its substituents. We clustered these compounds into six groups sharing a common scaffold and developed corresponding filters for each cluster (
These and other previously developed filters covered 37 of 53 detected GST-FHs. The remaining 17 compounds showed high diversity. For some of them, even small structural changes resulted in alteration of their promiscuity toward the GST/GSH system. For example, for benzopiperazinediones, a substitution of a benzene hydrogen by methyl group resulted into transformation of GST-FH into a “clean” compound (
Although chemoinformatic filters are very valuable tools for excluding FH compounds, counter assays will always be required to experimentally verify genuine Hit compounds from the list of primary Hits. Ideally, counter screening assays should be based on technologies different from the primary screening assay or need to include appropriate control proteins or reagents. For conducting an HTS involving a GST-tagged protein, one can choose between anti-GST antibody-coated agents or GSH-coated matrix. Here, the affinity of GSH for binding GST (Kd ≈ 5 µM) is weaker compared with anti-GST antibodies. 33 Therefore, GST-FH small molecules might be more efficient in impairing the GST/GSH interaction in comparison with GST/anti-GST antibody binding. Another point to consider is that GSH and anti-GST antibodies most probably bind at distinct binding sites of the GST protein. Thereby, a small molecule might block the GSH-binding pocket, whereas the anti-GST antibody binding site will be still free for interaction.
Altogether, this study has developed highly important GST-FH filters that allow HTS researchers to pinpoint promiscuous GST-tag protein-specific Hits in the primary Hit list. These potential promiscuous compounds could be further tested with another technology or another tag protein to validate the Hits and exclude nonspecific ones. Future studies will help to extent and/or fine-tune these filters in order to optimize their coverage and specificity.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially supported by the FP7 Marie Curie Initial Training Network project Environmental ChemOinformatics (ECO), grant agreement 238701, as well as by the German Research Center for Infection Research (DZIF), TTU 07.802.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.