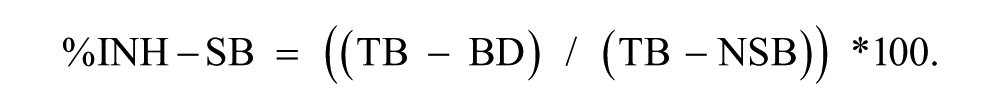Abstract
Mannose-binding lectin (MBL) is a circulating protein that acts as a soluble pattern recognition molecule of the innate immunity. It binds to carbohydrate patterns on the surface of pathogens or of altered self-cells, with activation of the lectin pathway of the complement system. Recent evidence indicates that MBL contributes to the pathophysiology of ischemia-reperfusion injury and other conditions. Thus, MBL inhibitors offer promising therapeutic strategies, since they prevent the interaction of MBL with its target sugar arrays. We developed and characterized a novel assay based on surface plasmon resonance for in vitro screening of these compounds, which may be useful before the more expensive and time-consuming in vivo studies. The assay measures the inhibitor’s ability to interfere with the binding of murine MBL-A or MBL-C, or of human recombinant MBL, to mannose residues immobilized on the sensor chip surface. We have applied the assay to measure the IC50 of synthetic glycodendrimers, two of them with neuroprotective properties in animal models of MBL-mediated injuries.
Keywords
Introduction
Mannose-binding lectin (MBL) is a calcium-dependent (C-type) lectin, belonging to the collectin family, with an important role in innate immunity. 1 MBL circulates in blood complexed with MBL-serine proteases (MASPs) and acts as a soluble pattern recognition receptor, binding specific carbohydrate patterns exposed on the surface of pathogens or of “altered-self” cells through its carbohydrate-recognition domain (CRD). On binding the target cells, MASPs are activated, initiating the lectin pathway (LP) of the complement system.
MBL has been implicated in experimental models of myocardial, 2 renal, 3 and gastrointestinal injury 4 ; cerebral ischemia/reperfusion injury5,6; and traumatic brain injury,7,8 as well as preeclampsia. 9 In all these conditions, MBL appears to contribute to tissue injury, and its deletion or inhibition is protective. MBL inhibitors might therefore be promising therapeutic strategies, and reliable in vitro assays are needed to identify them before more expensive and time-consuming in vivo studies.
MBL-binding properties of compounds can be investigated by a direct surface plasmon resonance (SPR) assay in which unlabeled putative MBL ligands are flowed onto recombinant MBL immobilized on the sensor chip. 5 Binding is revealed in real time as a change of mass at the surface, and the interaction can be characterized in terms of on and off rates (kinetics) and binding strength. 10 Although this approach is fairly straightforward and useful for initial screening purposes, it suffers some limitations: MBL can be bound on a site different from CRD, so it may not result in inhibitory activity; binding affinities can be overestimated due to multivalent interactions; immobilization procedures can alter the binding properties of MBL. We experienced difficulties in immobilizing recombinant murine MBL-C (mMBL-C, one of the two murine isoforms, together with mMBL-A 11 ). Then, last but not least, immobilization of MBL on a surface makes this configuration different from the physiological condition.
We have now developed and characterized a novel SPR-based assay that allows in vitro identification of MBL inhibitors, with a design that closely mimics real conditions, since the compound’s effects are measured by their ability to inhibit MBL binding to mannose residues exposed on the (sensor) surface. Thus, this approach can assess the inhibition of a property of MBL that is relevant to its functional activity. The assay is suited to study the inhibition of native plasmatic mMBL-A and mMBLC and of recombinant human MBL (rhMBL). We used it to measure the inhibitory effects of synthetic polymannosylated glycodendrimers, two of them—Polyman2 and Polyman9—with protective properties in animal models of MBL-mediated injuries.5,8,9
Materials and Methods
Animals
Plasma was obtained from two 8-week-old C57Bl/6 mice (wild-type [WT], 20–25 g; Harlan Laboratories, Indianapolis, IN) and from a C57Bl/6 mouse with a target mutation of both MBL-A and MBL-C genes (MBL−/−, 20–25 g, purchased from Jackson Laboratories [Bar Harbor, ME] and colonized at the Mario Negri Institute). Briefly, blood samples were collected from the vena cava in 10 mM ethylenediaminetetraacetic acid and 0.125% polybrene (Sigma-Aldrich, St. Louis, MO). Plasma was separated by centrifugation at 2000 g at 4 °C for 15 min, immediately stored, and kept at −80 °C until use.
The IRCCS–Istituto di Ricerche Farmacologiche Mario Negri adheres to the principles set out in the following laws, regulations, and policies governing the care and use of laboratory animals: Italian Governing Law (D.lgs 26/2014; Authorisation no. 19/2008-A, issued March 6, 2008, by the Ministry of Health), Mario Negri Institutional Regulations and Policies providing internal authorization for those conducting animal experiments (Quality Management System Certificate–UNI EN ISO 9001:2008–Reg. No. 6121), the National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals (2011 edition), and European Union (EU) directives and guidelines (EEC Council Directive 2010/63/UE). The Statement of Compliance (Assurance) with the Public Health Service (PHS) Policy on Human Care and Use of Laboratory Animals has been recently reviewed (September 9, 2014) and will expire on September 30, 2019 (Animal Welfare Assurance #A5023-01). The studies reported here were within a project specifically authorized by the Italian Ministry of Health (Decree no. 161/2014B).
SPR-Based Assay
SPR is a powerful label-free method widely used to study binding between two macromolecules. 12 Typically, one of the interacting partners is immobilized on a sensor chip surface, and the other is flowed through a microfluidic system in contact with the chip surface. Binding is revealed in real time as a change of mass at the surface. Although the most common application of SPR instruments is to determine affinity parameters for biomolecular interactions, 10 its versatility permits many other uses, including label-free immunoassays.13–15
The novel assay described here involves several steps. Figure 1 summarizes the four steps required for studies with murine plasma (two mMBL isoforms); studies with rhMBL are limited to the first two steps. The first step is the covalent immobilization of mannosylated bovine serum albumin (Man-BSA; Dextra Laboratories, Reading, UK) on the sensor chip (GLC; Bio-Rad, Hercules, CA) using amine coupling chemistry. 16 Thus, Man-BSA molecules are bound to the sensor surface through the protein moieties, leaving the mannose residues exposed. The ProteOn XPR36 Protein Interaction Array system (Bio-Rad) used for these SPR studies has six parallel flow channels that can immobilize up to six ligands on the same sensor chip. In parallel to Man-BSA, we always immobilized BSA (Sigma) as reference. Immobilization levels were about 3000 and 4000 resonance units (RUs, 1 RU 1 pg protein/mm2). After the first immobilization step, the ProteOn XPR36 fluidic system was rotated 90° 17 so that up to six different solutions could be injected simultaneously over the immobilized ligands ( Fig. 1 ). In this second step, we injected MBL-containing solutions, preincubated with or without the inhibitors (see below). We used mouse and human plasma, diluted (as indicated) in 10 mM running buffer (Tris buffer containing 150 mM NaCl and 0.005% Tween-20 [pH 7.4], plus 1.2 mM CaCl2) or running buffer with rhMBL added (R&D Systems, Minneapolis, MN). This second step aims to capture MBL (mMBL isoforms or rhMBL) on the Man-BSA surface. The mannose-dependent SPR binding signal is obtained by subtracting the mannose-independent nonspecific response on the reference surface (BSA only). The following steps are carried out after further rotation of the fluidic system ( Fig. 1 ) and regards the sequential injections of anti-MBL antibodies. After injecting murine plasma, we injected 10 µg/mL anti–mMBL-A and 10 µg/mL anti–mMBL-C antibodies (Hycult Biotech, Uden, the Netherlands) to specifically recognize and quantify the isoform captured during the second step ( Fig. 1 ). Two anti-hMBL antibodies were tested after injection of human plasma: 10 µg/mL anti-hMBL antibody 3E7 (Hycult Biotech) and 10 µg/mL anti-hMBL antibody 3B6 (Abcam, Cambridge, UK). All SPR assays were run at 25 °C. The sensorgrams (time course of the SPR signal in RU) were normalized to a baseline of 0.
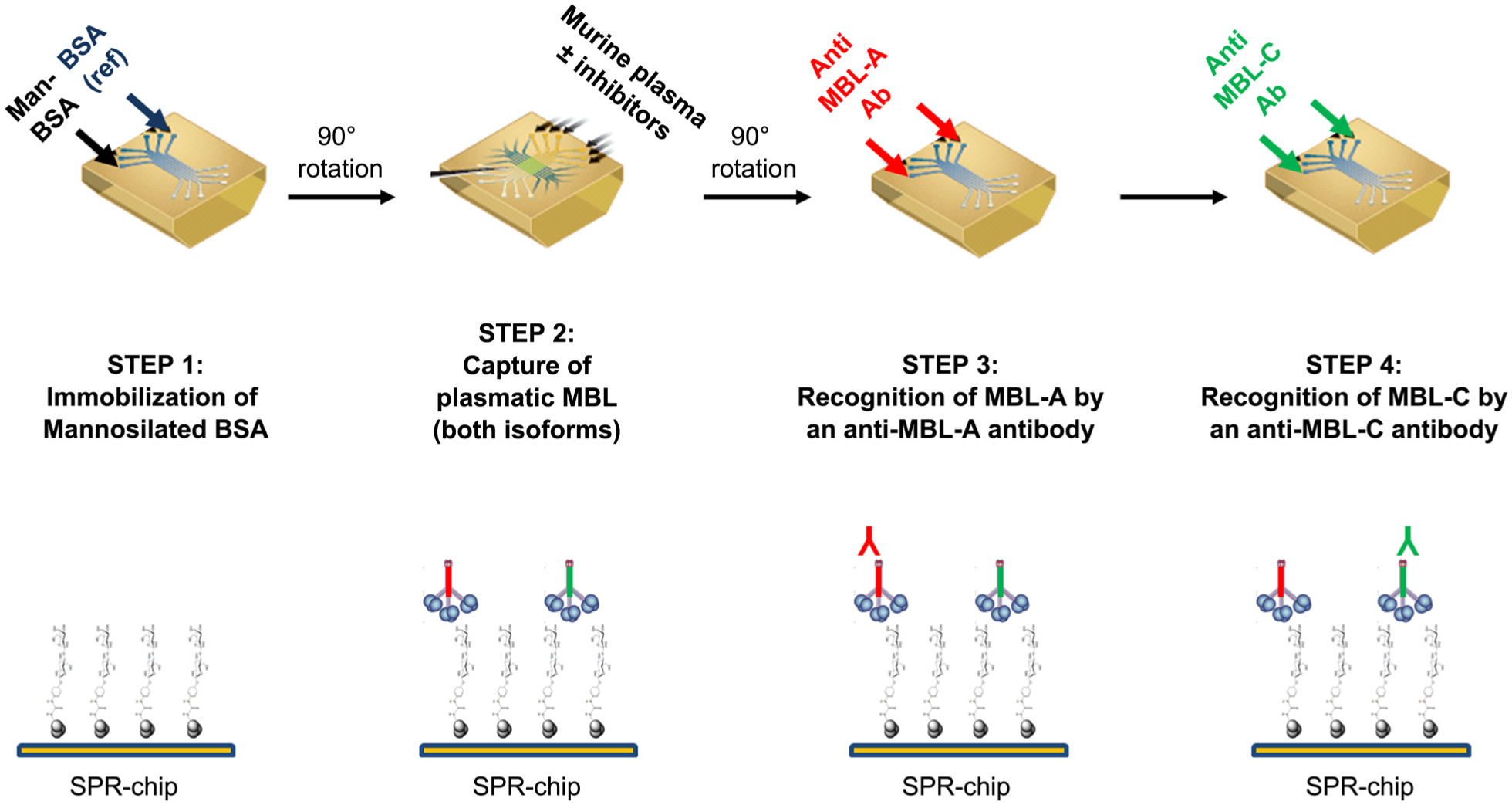
Schematic summary of the surface plasmon resonance assay for inhibitors of murine mannose-binding lectin (MBL) isoforms. Man-BSA, mannosylated bovine serum albumin.
For the analysis of putative MBL inhibitors, diluted plasma or running buffer spiked with rhMBL was preincubated for 30 min at 25 °C, with or without glycodendrimers at different concentrations, and then flowed onto immobilized Man-BSA and BSA ( Fig. 1 , step 2). The six parallel flow channels meant we could analyze the effect of up to four concentrations simultaneously, leaving one for mouse plasma preincubated with vehicle alone (total binding, TB) and one channel for MBL−/− mouse plasma (nonspecific binding, NSB). Since samples are flowed for 10 min at a rate of 30 µL/min, a volume of 400 µL is required for each of them; thus, 2 mL of diluted plasma is needed for the 5-point inhibition curve described above. This means that the plasma from a single mouse (about 200 µL), diluted 100-fold, allows to analyze 10 inhibition curves (e.g., three compounds in triplicate), considering both mMBL-A and mMBL-C.
The SPR binding signal at t = 100 s, in the presence of each drug concentration (binding with drug, BD), was used to calculate the percentage inhibition of specific binding (%INH-SB) as follows:
The aim of these studies was to identify compounds that prevent the MBL interaction with mannose residues and to determine their potency, that is, the concentration inhibiting MBL binding by 50% (IC50).
Synthetic Polymannosylated Glycodendrimers
The glycodendrimers we tested as MBL inhibitors are shown in Figure 2 . They are Polyman2 (also referred to as Dendron 12, 18 which is a tetravalent glycodendron carrying a pseudo-trimannoside headgroup); Polyman9, a hexavalent glycodendrimer with a flexible core, carrying a pseudo-dimannoside headgroup (also referred to as glycodendrimer 13.3 19 ); and Polyman31, a hexavalent glycodendron carrying the same pseudo-dimannoside headgroup on a rigid-core architecture. 20 The synthesis and characterization of these compounds have been described previously.18–20
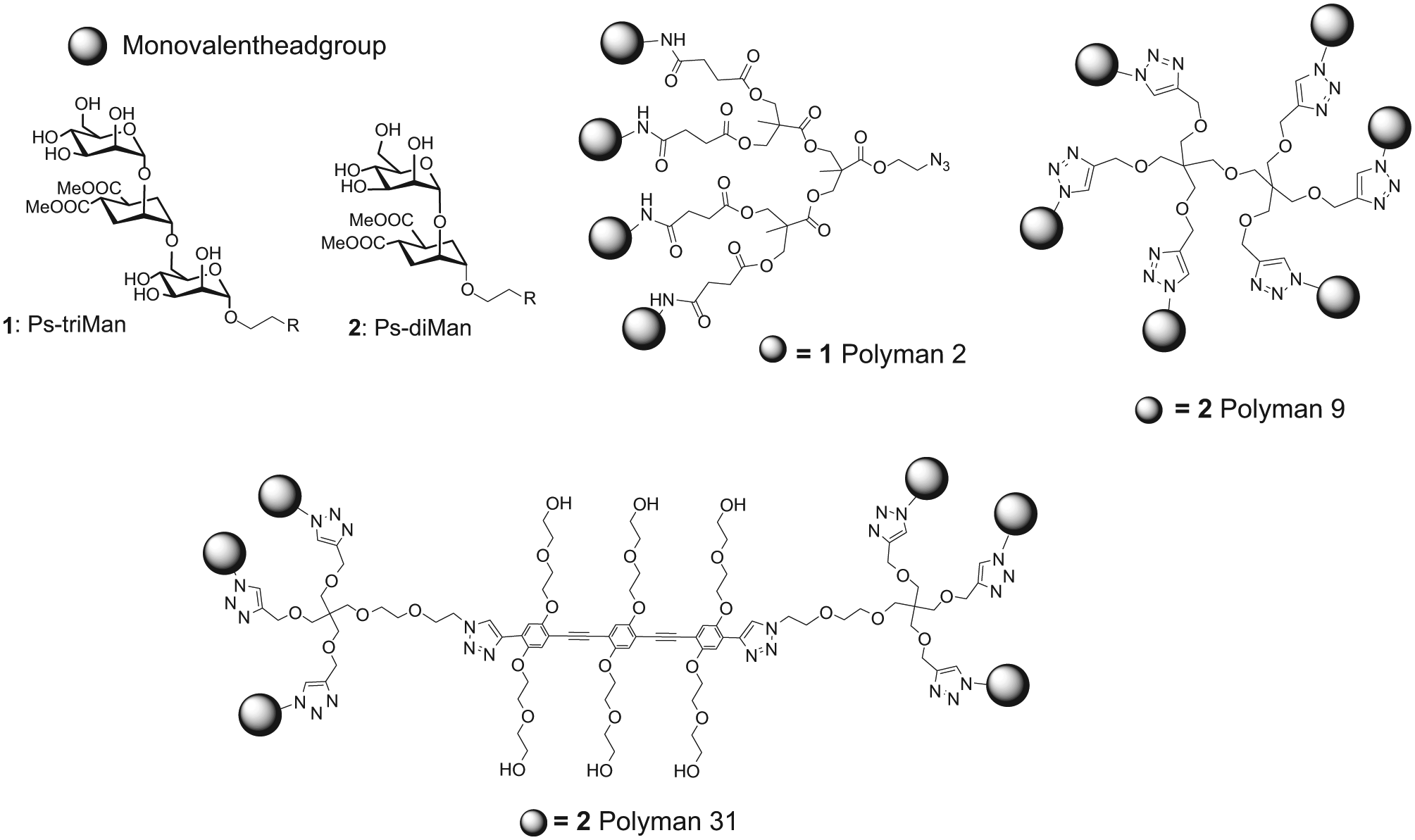
Structures of the glycodendrons used in this study: Polyman2, Polyman9, and Polyman31.
Results
Characterization of the SPR Assay for mMBL-A and mMBL-C
Diluted plasma from WT mice injected over a sensor surface coated with Man-BSA resulted in a marked SPR signal (2550 and 1500 RU, on average, at plasma dilutions of 1:50 and 1:100, respectively), much higher than in a parallel reference surface coated with BSA only (800 RU and 530 RU) (representative sensorgrams in
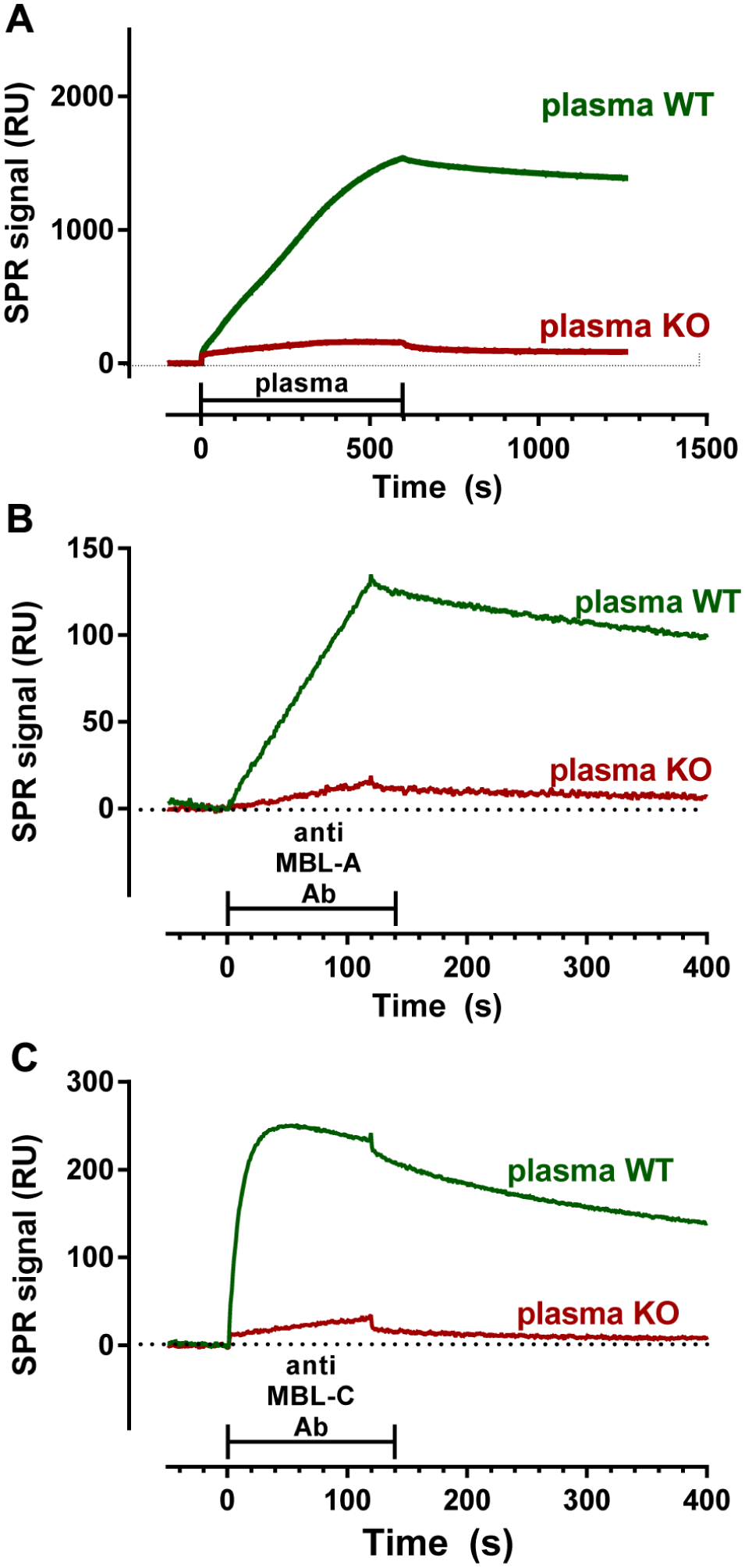
Capture of plasmatic mMBL-A and mMBL-C by immobilized mannose residues. (
The binding of plasmatic mMBL to immobilized mannose residues was very stable, as shown by the low dissociation rates after the injections ( Fig. 3A ), thus allowing sequential injection of the antibodies against mMBL-A and mMBL-C. These injections resulted in clear SPR binding signals ( Fig. 3B , C ), indicating that both murine isoforms were actually captured during the plasma injection; much lower signals were observed in the surfaces where plasma from MBL−/− had been flowed (23% ± 16% and 11% ± 13% with antibodies against mMBL-A and mMBL-C, mean ± SD, n = 18), thus confirming the specificity of the measured signal. The SPR signal with the anti–mMBL-A antibody was linear during the injection period, whereas a plateau was reached during the injection of the anti–mMBL-C antibody. This indicates a higher association rate constant of the anti–mMBL-C than the anti–mMBL-A antibody and that less mMBL-C than mMBL-A was captured by mannose.
As expected, the binding signals due to both mMBL-A and mMBL-C correlated with plasma dilution (
Preincubation of plasma with different concentrations of mannose resulted in concentration-dependent inhibition of both mMBL-A and mMBL-C binding to immobilized mannose, as indicated by the subsequent injections of the corresponding antibodies. The IC50 was similar for the two isoforms, respectively 5.4 and 5.2 mM for mMBL-A and mMBL-C.
Characterization of the Assay for hMBL
Injection of diluted human plasma over immobilized Man-BSA resulted in a marked SPR signal (400 RU on average, at plasma dilutions of 1:50), much higher than that observed in a parallel reference surface coated with BSA only (50 RU) (representative sensorgrams in
Despite the presence of a mannose-dependent binding signal, we were not able to confirm the capture of hMBL with anti-hMBL antibodies. Results were negative with two different anti-hMBL antibodies, at concentrations up to 10 µg/mL, even when plasma was only diluted 1:10 (data not shown). To overcome this problem, we developed a different approach testing the compounds for their ability to inhibit the binding of rhMBL to immobilized mannose. A very similar approach, using BSA-conjugated N-acetyl-D-glucosamine as immobilized sugar, has already been applied.
21
Different concentrations of rhMBL (
Inhibitory Effects of Polymannosylated Glycodendrimers
Polyman2 inhibited the binding of mMBL-A and mMBL-C to immobilized mannose with similar IC50 values (93 and 84 µM; Fig. 4A , B ), whereas Polyman31 was highly selective for the two isoforms, inhibiting mMBL-C with a very low IC50 (0.5 µM) but being much less active on mMBL-A (IC50 >200 µM) ( Fig. 4A , B ). Polyman9 also had a lower IC50 on mMBL-C (65 µM) than on mMBL-A (IC50 >200 µM) ( Fig. 4A , B ). 8
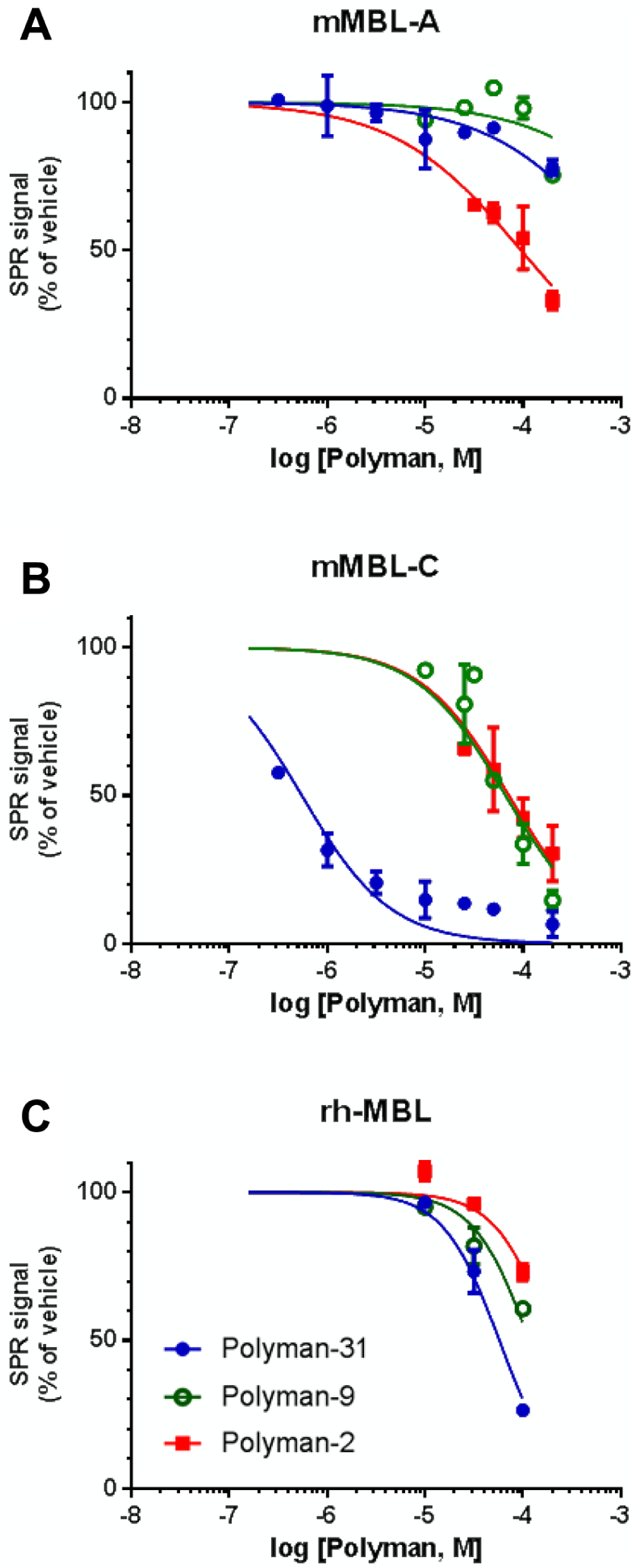
Inhibitory effect of dendrimeric compounds on mMBL-A (
As an example of raw data,
Preincubation of rhMBL (8 nM) with different concentrations of dendrimers resulted in dose-dependent inhibition of rhMBL binding to immobilized mannose ( Fig. 4C ). In this assay, Polyman31 was the most potent inhibitor (IC50 = 62 µM), followed by Polyman9 (~136 µM) and Polyman2 (~270 µM).
Discussion
We report the characterization of a novel SPR assay for in vitro screening of MBL inhibitors, analyzed for their ability to interfere with MBL binding to carbohydrate patterns. This was achieved by flowing MBL-containing solutions, with or without inhibitors, over a sensor chip exposing mannose residues.
The mannose-dependent SPR signal with diluted murine plasma is mostly (80% on average) due to mMBL, as indicated by the results with plasma of MBL−/− mice. This binding has a very low dissociation rate, as expected for multivalent binding, thereby permitting subsequent injections of antibodies against mMBL-A and mMBL-C to specifically recognize and quantify the isoform captured by mannose. In our conditions, 70% to 80% of the mMBL-dependent SPR signal is accounted for by mMBL-A, although its serum levels in C57Bl/6 mice are four to five times lower than those of mMBL-C.22,23 Since the molecular mass of the two isoforms is comparable, 22 this suggests that mMBL-A has greater affinity than mMBL-C for immobilized mannose. To our knowledge, there are no data available in literature comparing the binding of native plasmatic murine MBL isoforms to surface mannose arrays (i.e., using a design mimicking the physiological condition). Consistently with our observation, it was previously reported that the CRD domain of the A isoform of the rat mannose binding protein (MBP-A) had 14-fold higher affinity for mannosylated BSA than the CRD domain of MBP-C. 24 Regarding murine MBL isoforms, the only available data showed that monovalent mannose inhibited the binding of purified mMBL-A and mMBL-C to mannan-coated wells with an IC50 of 1.5 and 4 mM, respectively. 11 Measuring the effect of mannose on the binding of plasmatic mMBL-A and mMBL-C to mannose-coated chips, we could not confirm this slight difference between the two isoforms (IC50 = 5.4 and 5.2 mM, respectively). The physiological relevance of monovalent sugar affinity, however, is limited, since it is well known that the binding of MBL to mannose patterns exposed on a cell surface involves multivalent protein-sugar interactions that significantly increase the overall binding affinity. It might be speculated that the different affinities of plasmatic mMBL-A and mMBL-C for immobilized mannose arrays could be due to a different CRD conformation, with consequences on the avidity effects. The smaller proportion of mMBL-C captured by immobilized mannose was, however, compensated by the higher affinity of the corresponding antibody. Thus, the SPR signal 100 s after injection of the anti–mMBL-C antibody was similar, on average, to that seen 100 s after injection of the anti–mMBL-A antibody.
The SPR data obtained injecting diluted human plasma indicated a mannose-dependent binding signal that, however, could not be definitely associated with the capture of hMBL. In fact, there was no SPR signal on subsequent injection of anti-hMBL antibodies. Possibly, the “mannose-dependent” binding signal after injection of human plasma is either (1) not due to hMBL capture or (2) actually due to hMBL, but the two anti-hMBL antibodies tested did not bind it efficiently enough for detection in our setup.
The mannose-dependent binding signal with human plasma was much lower (20%) than the signal with murine plasma, in agreement with the lower hMBL concentration (1–1.5 µg/mL)25,26 than mMBL-A (8–20 µg/mL) or mMBL-C (30–100 µg/mL).22,23 Insufficient capture of hMBL on the sensor chip might thus contribute to the lack of measurable binding with anti-hMBL antibodies, although results were also negative using plasma or serum diluted 10-fold only. Other factors required for an efficient SPR assay, and possibly not met with native hMBL, are (1) adequate affinity of hMBL for the immobilized mannose residues, particularly an association rate constant permitting the capture of enough hMBL during the short association phase (few minutes), and/or (2) suitable affinity of the anti-hMBL antibodies employed for these studies. In this regard, successful detection of plasmatic mMBL-A is very likely due to its high affinity for immobilized mannose (higher than that of mMBL-C), whereas successful detection of mMBL-C is probably likely due to the high serum concentrations and the high affinity of the anti–mMBL-C antibody (higher than that of anti–mMBL-A antibody).
To evaluate the effects of potential inhibitors on hMBL, we developed an SPR assay using rhMBL. A suitable SPR binding signal was obtained by flowing 8 nM rhMBL, dissolved in buffer (about 2.7 µg/mL; i.e., much higher than the concentration present in diluted human plasma), while lower concentrations gave proportionally lower SPR signals.
We used these assays to study the inhibitory effects of synthetic polymannosylated glycodendrimers. Native MBL is found as oligomers of a trimeric building block, with multiple CRD domains 27 that efficiently bind to carbohydrate patterns through multivalent protein-sugar interactions. Thus, to interfere with these, multivalent inhibitors such as glycodendrimers have been designed and studied over the past 10 years as potential antagonists.28,29 Polyman2 18 is a tetravalent pseudo-trimannoside dendron that showed potent protective activity in mouse models of cerebral ischemia 5 or preeclampsia, 9 where MBL significantly contributed to the injury. Polyman9 is a hexavalent pseudo-dimannoside dendrimer based on a flexible scaffold 19 that showed neuroprotective effects in a murine model of traumatic brain injury. 8 Polyman31 is a new hexavalent pseudo-dimannoside dendrimer based on a rigid-core architecture. 20 In our assay, Polyman2 showed no selectivity between mMBL-A and mMBL-C (IC50 = 93 and 84 µM), whereas Polyman31 had a completely different interaction profile, being very potent on MBL-C (IC50 = 0.5 µM) but almost inactive on mMBL-A up to 200 µM. Polyman9 had a similar profile, inhibiting mMBL-C (IC50 = 65 µM) but not MBL-A (IC50 >>200 µM). 8 These molecules also inhibited rhMBL binding to mannose residues, with an IC50 from 62 to 270 µM. The MBL-C selectivity observed with Polyman31 and Polyman9 is likely due to intrinsic structural features of these dendrimers, since the lack of selectivity of Polyman2 excludes methodological artifacts. The occupation of CRD mannose-binding sites by dendrimers depends on the affinity of the sugar residues and on their spatial arrangement. Polyman31 and Polyman9 carry pseudo-dimannoside headgroups, whereas a pseudo-trimannoside headgroup is present in Polyman2. It could also be envisaged that sterical hindrance could play a role even with a partial CRD occupation, depending on the dendrimer and CRD sizes. In our test, the proteins are present in their native plasmatic form, which is likely to consist of aggregates of CRD trimers.11,22 The distance between binding sites in trimers is estimated to be ~5 nm for hMBL.30,31 The higher affinity of Polyman31 for mMBL-C may reflect a better match between this molecule, where two active ligands can span a distance of 4 to 5 nm, and the MBL-C trimer. The size and composition of the higher order oligomers are less understood, 32 whereas our data exclude the aggregation of the dendrimers (not shown).
The inhibitor concentrations we used (µM range) greatly exceed those of MBL (nM range, in diluted murine plasma and rhMBL). Thus, the percentage inhibition of MBL binding to mannose is indicative of the occupancy of MBL proteins, and the IC50 is a measure of the inhibitor’s affinity for MBL. The affinities of Polyman2 for mMBL-A and rhMBL (IC50 = 93 and ~270 µM) are higher than those measured by direct SPR assay—that is, directly flowing the dendrimer over immobilized recombinant mMBL-A (KD = 4.7 µM, data not shown) or rhMBL (KD = 6 µM5). A similar difference was reported for polymeric inhibitors of the lectin DC-SIGN using analogous SPR approaches 15 and is possibly due to a lower impact of avidity effects when interactions occur in solution. Moreover, since each MBL molecule has multiple CRD domains, it may be envisaged that occupation of most of them is necessary for inhibition of its binding to immobilized mannose, whereas binding to a single CRD is enough to elicit an SPR signal in the direct SPR assay. We consider the IC50 values obtained with the proposed indirect SPR assay more informative since they measure the ability of the compounds to interfere with the main final physiological event to be targeted by MBL inhibitors (i.e., the binding of native mMBL to mannose arrays).
A mannan immunoassay has been described in which murine plasma was incubated on mannan-coated microtiter wells and murine MBL isoforms were then recognized by biotinylated anti-MBL antibodies and europium-labeled streptavidin. 22 This method was used to quantify mMBL-A and mMBL-C concentrations in mouse sera, but to our knowledge, it has never been used to screen MBL inhibitors. In comparison with our SPR assay, immunoassays in microtiter wells are probably more sensitive, due to signal amplification. However, the SPR assay has other worthwhile advantages: (1) it does not need any labeled reagent and takes less of the antibodies than for microtiter wells; (2) the amount of bound anti-MBL antibody—and thus MBL—is immediately determined by SPR, whereas further steps are required when using microtiter wells (e.g., incubation with europium-labeled streptavidin for an immunofluorometric assay) 22 ; and (3) the long incubation of plasma in mannan-coated microtiter wells may reduce the binding between the inhibitor and plasmatic MBL, due to the equilibrium shift induced by the high-affinity binding of free MBL to immobilized sugars; this potential artifact, leading to underestimation of the compound’s inhibitory effect, is probably absent during the very short flow of plasma over the mannose-coated SPR surface.
In conclusion, we have extensively characterized a novel assay, convenient for in vitro screening of putative MBL inhibitors, to select the appropriate compounds for the more expensive and time-consuming in vivo studies. This approach has been successfully used to identify a selective mMBL-C inhibitor, Polyman9, which conferred neurobehavioral protection when given shortly after traumatic brain injury. 8 We also found another glycodendrimer, Polyman31, with even higher affinity and selectivity for mMBL-C.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support, including a fellowship to D. De Blasio, was provided by Fondazione Cariplo (grant number 2012-0590).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.