Abstract
Using human cell models mimicking the central nervous system (CNS) provides a better understanding of the human CNS, and it is a key strategy to improve success rates in CNS drug development. In the CNS, neurons function as networks in which astrocytes play important roles. Thus, an assessment system of neuronal network functions in a co-culture of human neurons and astrocytes has potential to accelerate CNS drug development. We previously demonstrated that human hippocampus-derived neural stem/progenitor cells (HIP-009 cells) were a novel tool to obtain human neurons and astrocytes in the same culture. In this study, we applied HIP-009 cells to a multielectrode array (MEA) system to detect neuronal signals as neuronal network functions. We observed spontaneous firings of HIP-009 neurons, and validated functional formation of neuronal networks pharmacologically. By using this assay system, we investigated effects of several reference compounds, including agonists and antagonists of glutamate and γ-aminobutyric acid receptors, and sodium, potassium, and calcium channels, on neuronal network functions using firing and burst numbers, and synchrony as readouts. These results indicate that the HIP-009/MEA assay system is applicable to the pharmacological assessment of drug candidates affecting synaptic functions for CNS drug development.
Keywords
Introduction
In the central nervous system (CNS), neuronal networks play fundamental and critical roles in cognition, learning, and memory. Functional aberration in the networks is related to neurological diseases, including epilepsy, Alzheimer’s disease, fragile X syndrome, and autism.1−4 To better understand the mechanisms of these diseases and develop novel therapeutic agents, assays constructed based on functional neuronal networks are needed. Under physiological conditions, neurons form networks with other neurons and nonneuronal cells, such as astrocytes. Astrocytes take part in synapse and neuronal circuit formation through both direct contact with neurons and indirect actions (e.g., supplementation of various paracrine factors). 5 Therefore, astrocytes are indispensable in constructing in vitro assays for investigating neuronal network functions.
A multielectrode array (MEA) system is a useful approach that enables us to conduct network-level analyses of neurons in vitro, 6 and it has been applied to the evaluation of efficacy and neurotoxicity of drug candidates to detect readouts as neuronal network functions using rodent neurons. 7 However, such assays using rodent neurons have limitations in predicting the clinical efficacy and neurotoxicity of the candidates due to species differences.8,9 Thus, using human neurons is a solution to overcome the issue. After the advent of human embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs), as well as the development of neural differentiation methods, human neurons derived from these stem cells have been applied to the MEA system for the assessment of human neuronal networks. Odawara et al. 10 observed spontaneous firings of human iPSC-derived neurons when they were co-cultured with rat astrocytes. As described above, astrocytes should be included in neuronal network assay cultures because of their contribution to functional neuronal circuit formation. From a viewpoint of physiological relevancy to human biology, human astrocytes rather than rodent ones should be used in the co-culture, because there are species differences of astrocytes in some aspects, such as morphological complexity and functional responses to cytokines.11,12 As a matter of fact, sources of human astrocytes have been limited to ESCs and iPSCs except postmortem brains. Currently available methods of differentiating ESCs or iPSCs into astrocytes are, however, still cumbersome. 13 This is a critical hurdle in applications of human astrocytes to physiologically relevant MEA assay systems in drug development.
To overcome the issue, we used human neural stem/progenitor cells, HIP-009 cells, in this study. HIP-009 cells have advantages over ESCs/iPSCs: They can differentiate into both neurons and astrocytes simultaneously in the same culture without any complex steps. 14 Here, we report the establishment of the MEA assay system to analyze functions of human neuronal networks in co-culture of human neurons and astrocytes, both of which are simultaneously differentiated from HIP-009 cells as a source. We also applied the HIP-009/MEA assay system to a pharmacological evaluation of effects of reference compounds affecting synaptic receptors or ion channels on neuronal network functions.
Materials and Methods
Chemicals
N-methyl-D-aspartate (NMDA), (+)-MK-801 hydrogen maleate, NBQX hydrate, γ-aminobutyric acid (GABA), picrotoxin, gabazine, and 4-aminopyridine (4-AP) were purchased from Sigma-Aldrich (St. Louis, MO). α-Amino-3-hydroxy-5-methyl-4-isoxazolepropionate (AMPA), kainic acid, and (−)-bicuculline methiodide were obtained from Tocris Bioscience (Bristol, UK). Sodium hydrogen L(+)-glutamate monohydrate, tetrodotoxin (TTX), and carbamazepine were purchased from Wako (Osaka, Japan). ω-Agatoxin IVA and ω-conotoxin GVIA were obtained from Peptide Institute (Osaka, Japan). The following solvents were used according to the manufacturer’s instructions: dimethyl sulfoxide (Wako) for (+)-MK-801 hydrogen maleate, NBQX hydrate, picrotoxin, (−)-bicuculline methiodide, and carbamazepine; and distilled water (Life Technologies, Carlsbad, CA) for NMDA, AMPA, kainic acid, sodium hydrogen L(+)-glutamate monohydrate, GABA, gabazine, TTX, 4-AP, ω-agatoxin IVA, and ω-conotoxin GVIA. To avoid repeated freezing and thawing, they were divided into usage-size aliquots and stored at −20 °C before use.
Cell Culture
All experiments were approved by the Eisai Research Ethics Committee and the Eisai Animal Care and Use Committee. HIP-009 cells were purchased from GigaCyte (Branford, CT; now, PhoenixSongs Biologicals, Branford, CT). Cells were expanded and differentiated as follows. One-hundred-millimeter dishes were coated with 10 µg/mL laminin (Sigma-Aldrich) in Dulbecco’s modified Eagle’s medium (DMEM)/Ham’s F-12 (1:1) (DMEM/F12; Wako). Cells were seeded and grown on these dishes in Neural StemCell Growth Medium (PhoenixSongs Biologicals) added with growth medium supplement cocktail [Neural StemCell Growth Supplement, 10 ng/mL basic fibroblast growth factor (bFGF), 20 ng/mL epidermal growth factor (EGF), 3 µM CHIR-99021, and 1 µg/mL laminin] and 30 µg/mL gentamicin (Life Technologies). The medium was changed every 2 or 3 days, and confluent cells were split for expansion every 4 or 5 days. HIP-009 cells expanded for eight passages were used in this study. Before neural differentiation, expanded HIP-009 cells were harvested and cultured in Neural Transition Medium (PhoenixSongs Biologicals) added with transition medium supplement cocktail (Neural Transition Supplement, 10 ng/mL bFGF, 20 ng/mL EGF, and 1 µg/mL laminin) and 30 µg/mL gentamicin for 3 days. 14 The expansion and transition treatment of HIP-009 cells was carried out at 37 °C in a humidified atmosphere of 2% O2, balanced with N2 and 6% CO2.
For MEA recordings, 2×105 HIP-009 cells that had received the transition treatment as described above were plated onto the center of MEA chips (Alpha MED Scientific, Osaka, Japan) and cultured in Neural Differentiation Medium (PhoenixSongs Biologicals) added with differentiation medium supplement cocktail [Neural Differentiation Supplement, 20 ng/mL brain-derived neurotropic factor (BDNF), 20 ng/mL glial cell–derived neurotropic factor, and 1 µg/mL laminin] and 30 µg/mL geneticin (hereafter referred to as conventional medium). The conventional medium is serum-free. Prior to the plating, the center of MEA chips was precoated with polyethyleneimine (0.1%; Sigma-Aldrich) and laminin (10 μg/mL). In differentiation culture, half of the medium was changed twice a week. Cells were differentiated for 4–8 weeks in a humidified incubator having an atmosphere of 5% CO2 and 95% air at 37 °C.
In this study, two different media were tested: the conventional differentiation medium without or with rat astrocyte-conditioned medium (ACM) (ratio of conventional medium to rat ACM = 4:1). Rat ACM was prepared according to the method of Arthur et al. 15 Briefly, rat primary cerebral cortical cells, which were obtained from rat fetuses on embryonic day 18 from pregnant Wistar rats (Crlj:WI; Charles River Laboratories Japan, Yokohama, Japan), were cultured in DMEM (Wako) supplemented with 10% fetal bovine serum (FBS; Life Technologies) until cells reached 100% confluency. Then, cells were incubated on an orbital shaker overnight at 4 °C and subcultured. After cells grew to 100% confluency, the medium was changed to a fresh one, which was collected after an additional 48-h incubation. In the case of differentiation with rat ACM, the conventional medium without ACM was used in the first 2 weeks and replaced with the rat ACM-supplemented one after that.
MEA Recordings
The extracellular recordings were performed by using a MED64 System (Alpha MED Scientific). Signals from all 64 electrodes were sampled simultaneously at 20 kHz and stored to a standard personal computer by using a MED64 Mobius software (Alpha MED Scientific). The culture temperature was maintained at 37 °C during the measurement. Spike detection was carried out by using the Mobius software with a threshold of 5.5 times the standard deviation of the noise level. To streamline the data analysis with the Mobius, we developed a novel software for spike detection based on the algorithm of the Mobius. To validate the spike detection methodology of our software, we compared the number of spikes detected by the Mobius and ours. Once the comparability of our software was demonstrated, we used it in further analysis. It was also used to make raster plots for visualization of the processed spike data. Burst and synchrony analyses were conducted by using a NeuroExplorer software (Nex Technologies, Madison, AL) according to the report of Li et al. 16
Microarray Analysis
Total RNAs were collected from HIP-009 cells differentiated on MEA chips in the conventional medium, conventional medium supplemented with rat ACM, and conventional medium supplemented with FBS (2%) for 4 or 8 weeks. Twenty-five ng of total RNAs from each MEA culture and human hippocampus (Clontech Laboratories, Mountain View, CA) was reverse-transcribed into double-stranded complementary DNAs (cDNAs) and amplified for 2 h at 40 °C by using a Low Input Quick Amp Labeling Kit (Agilent Technologies, Santa Clara, CA). The cDNAs were then transcribed into antisense complementary RNAs (cRNAs) and labeled with Cy3–cytidine triphosphate fluorescent dyes for 2 h at 40 °C in accordance with the manufacturer’s protocol. Cy3-labeled cRNAs were purified with an RNeasy Mini Kit (Qiagen, Venlo, the Netherlands). Purity and dye incorporation were assessed with a NanoDrop ND-1000 spectrophotometer (Thermo Fisher Scientific, Waltham, MA) and 2100 Bioanalyzer (Agilent Technologies). The minimum acceptable Cy3 labeling was ≥ 6.0 pmol/μg. Next, 600 ng of Cy3-RNAs was hybridized to a SurePrint G3 Human GE Microarray kit 8 × 60K ver. 1.0 by using a Gene Expression Hybridization Kit (Agilent Technologies). After incubation for 17 h at 65 °C and 10 rpm rotation, the microarrays were washed with Gene Expression Wash Buffer 1 (Agilent Technologies) for 1 min at room temperature, and then with Wash Buffer 2 (Agilent Technologies) for 1 min at 37 °C. The microarrays were scanned with a G2505B Microarray Scanner (Agilent Technologies). Data were analyzed with a GeneSpring 13.1 software (Agilent Technologies).
Immunocytochemistry
After MEA recordings, cells were washed with phosphate-buffered saline without calcium and magnesium (PBS; Wako) once, and fixed in 4% paraformaldehyde (Wako) for 30 min at room temperature. Cells were washed with PBS at room temperature three times and permeabilized with 0.2% Triton X-100/PBS for 5 min at room temperature. Nonspecific binding was blocked with Blocking One (Nacalai Tesque, Kyoto, Japan) in PBS (blocking buffer) for 60 min at room temperature. Immunolabeling of cells was carried out with primary antibodies diluted in 0.2% Triton X-100/blocking buffer overnight at 4 °C. After being washed with 0.05% Tween 20/PBS (PBST) three times for 5 min, cells were incubated with secondary antibodies and Hoechst 33342 (1 µg/mL; Sigma-Aldrich) diluted in 0.2% Triton X-100/blocking buffer for 1 h at room temperature. Cells were then washed with PBST three times and rinsed with PBS. The primary antibodies were rabbit anti-MAP2 (1:500; Millipore, Billerica, MA) and rat anti-GFAP (1:500; Life Technologies). The secondary antibodies were Alexa Fluor 488 donkey antirabbit IgG (1:500; Life Technologies) and DyLight 649 donkey antirat IgG (1:500; Jackson ImmunoResearch Laboratories, West Grove, PA). Stained cells were imaged under a fluorescence microscope (BZ-9000; Keyence, Osaka, Japan).
Statistical Analysis
All data are expressed as means ± standard error of the mean (SEM), and n refers to the number of independent experiments. GraphPad Prism 6.0 (GraphPad Software, Inc., San Diego, CA) was used for statistical analysis. Statistical differences between two groups were examined by use of the F-test, followed by the two-tailed unpaired Student’s t-test (p ≥ 0.05 in F-test) or Welch’s t-test (p < 0.05 in F-test). Changes of parameters of neuronal network functions by compound treatment were evaluated by the two-tailed paired Student’s t-test. A probability (p) value < 0.05 was considered statistically significant.
Results
Detection of Spontaneous Firings of HIP-009 Neurons by the MEA
To ascertain whether spontaneous firings were detected in HIP-009 neurons by the MEA, the MEA measurement of neuronal signals of the cells was conducted after 4-week differentiation in the conventional medium. We observed spontaneous firings from about one-third of 64 electrodes, detected by the Mobius software (gray bars in
Fig. 1
). Next, we compared the numbers of spikes detected by the Mobius and in-house developed software to validate the spike detection algorithm of our software. Four representative data sets were shown in
Figure 1
. The numbers of spikes and electrodes recording spikes (active electrodes) detected by the in-house software were shown to be statistically comparable to those by the Mobius (
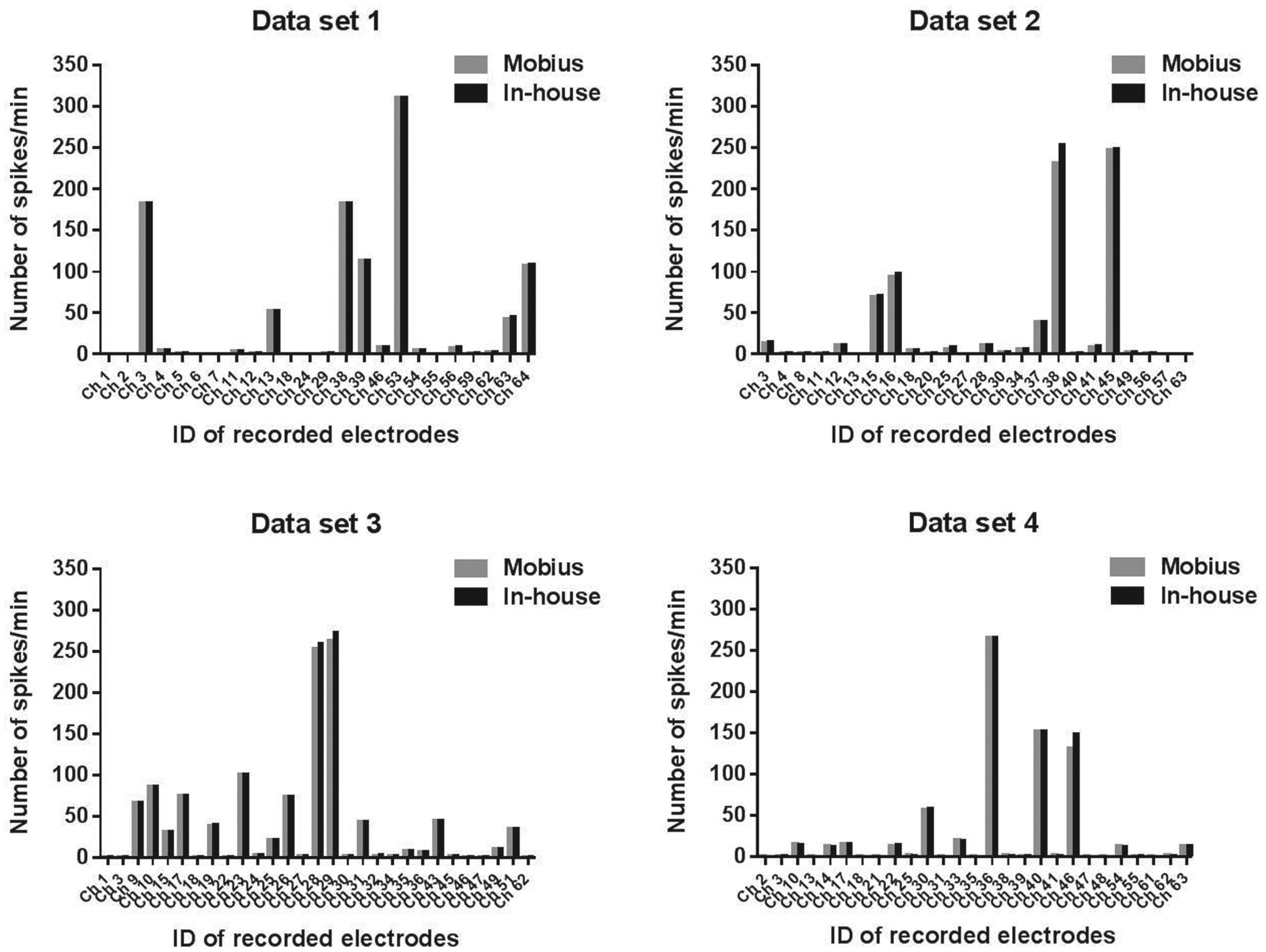
Spontaneous firings of HIP-009 neurons. HIP-009 cells were differentiated in the conventional medium (see Materials and Methods) for 4 weeks before the measurement by using a multielectrode array system (MED64 System, Alpha MED Scientific, Osaka, Japan). The number of spikes per minute detected from each of 64 electrodes was calculated by using Mobius or in-house developed software, and results were compared with each other. Four independent, representative data sets were presented.
Optimization of Differentiation Conditions of HIP-009 Cells
After 4-week differentiation in the conventional medium, spontaneous firings were detected from about one-third of 64 electrodes, and the number of spikes was low (
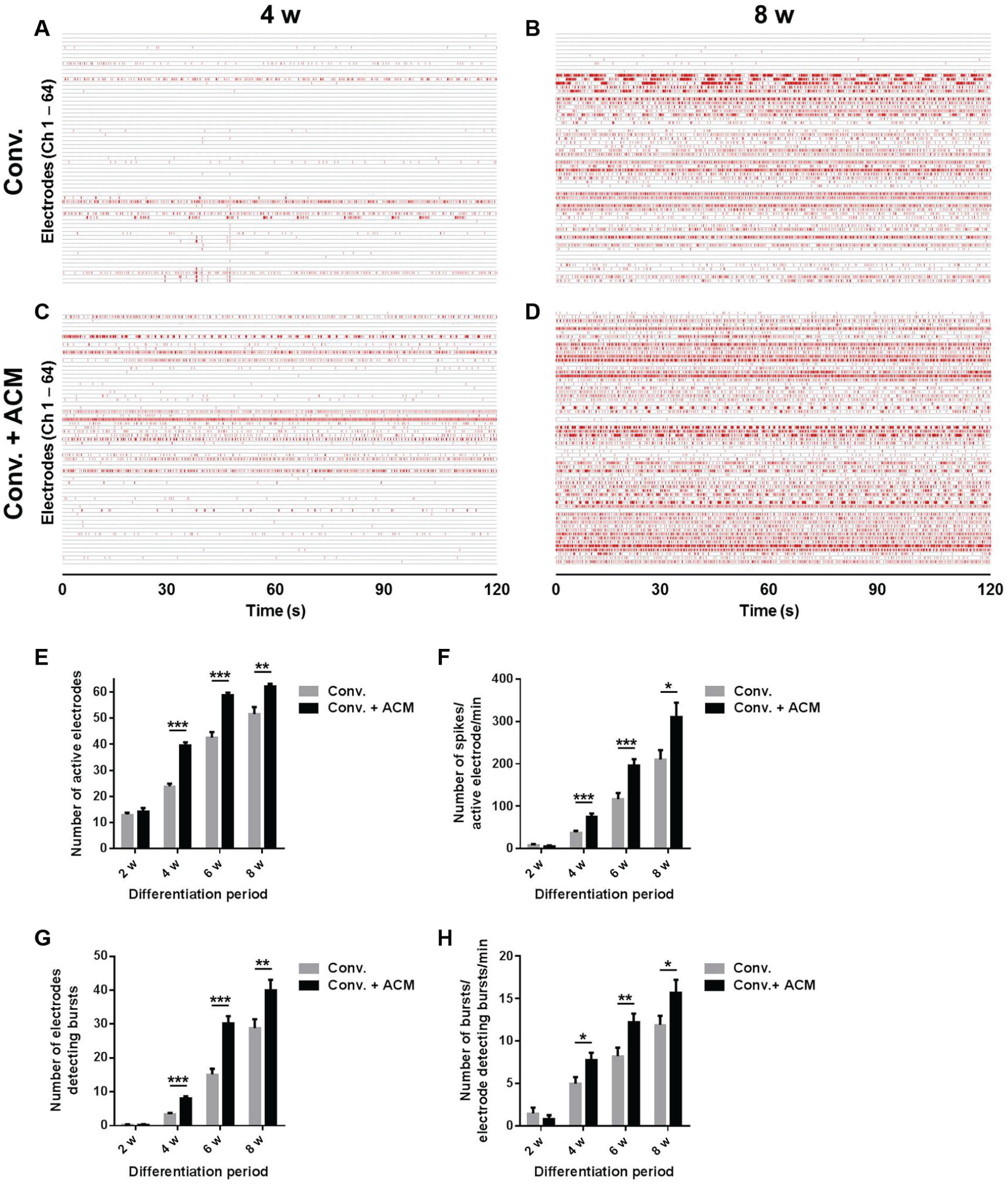
Electrophysiological maturation of HIP-009 neurons. (
Microarray Analysis of HIP-009 Cells Cultured in Different Differentiation Media
The two-dimensional principal component analysis of microarray data was performed to compare comprehensive gene expression of HIP-009 cells differentiated in three types of media (
Immunocytochemical Characterization of Long-Term Differentiated HIP-009 Cells
We previously reported that a neuron-to-astrocyte ratio of 4-week differentiated HIP-009 cells was about 50%/50% in the conventional medium.
14
Likewise, we observed neuronal marker (MAP2)–positive cells and astrocyte marker (GFAP)–positive cells in HIP-009 cells differentiated for 8 weeks in the conventional medium (
Fig. 3
,
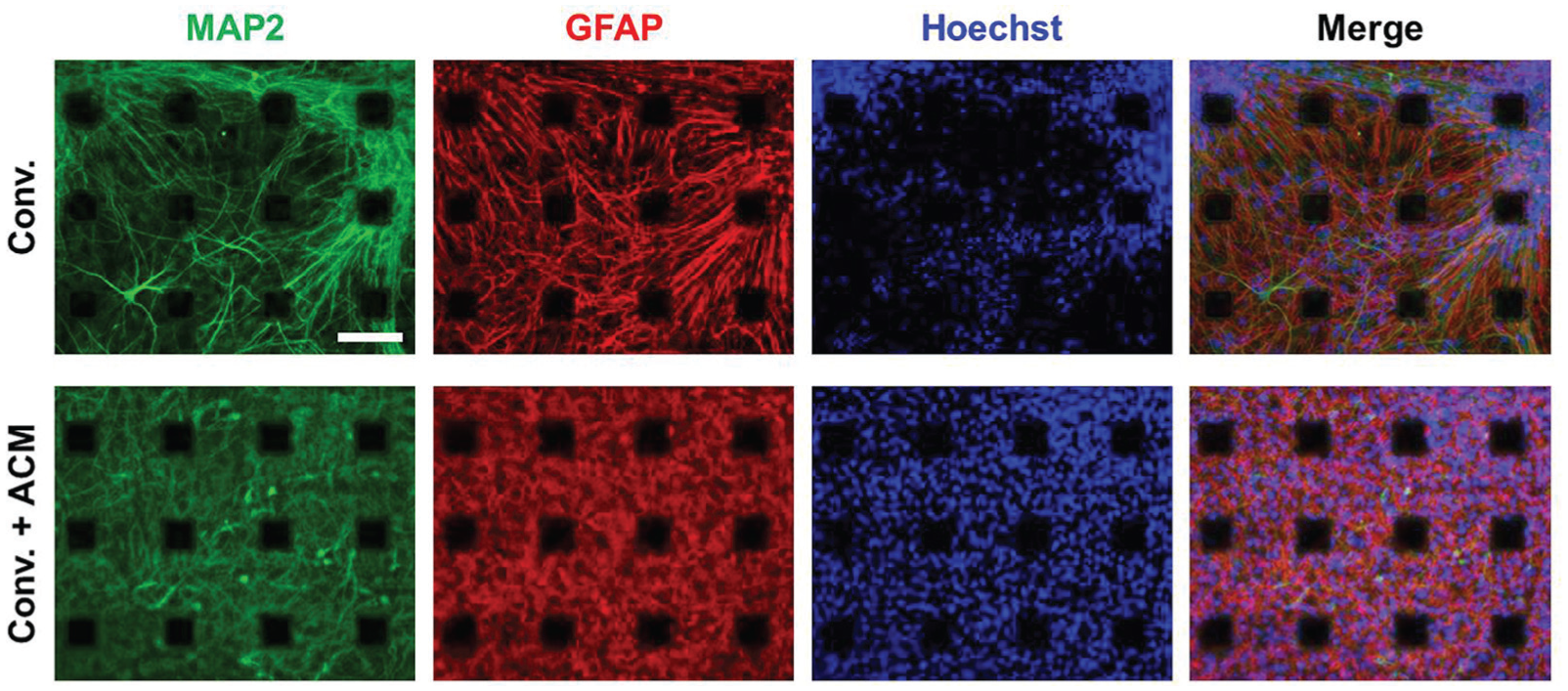
Immunocytochemical characterization of long-term differentiated HIP-009 cells. HIP-009 cells were differentiated on multielectrode array chips in the conventional medium (upper) and the conventional medium supplemented with rat astrocyte-conditioned medium (ACM; lower) for 8 weeks. Then, they were immunostained with neuronal (MAP2) and astrocyte (GFAP) markers. Conv., conventional medium. Scale bar, 100 µm.
Neuronal Network Formation among HIP-009 Neurons
To investigate whether functional neuronal networks were formed among HIP-009 neurons, we assessed the effects of blockers of GABAA, NMDA, and AMPA receptors on firing activities of HIP-009 neurons cultured in the conventional medium supplemented with rat ACM for 8 weeks. A GABAA receptor blocker, gabazine, increased the number of spikes significantly (
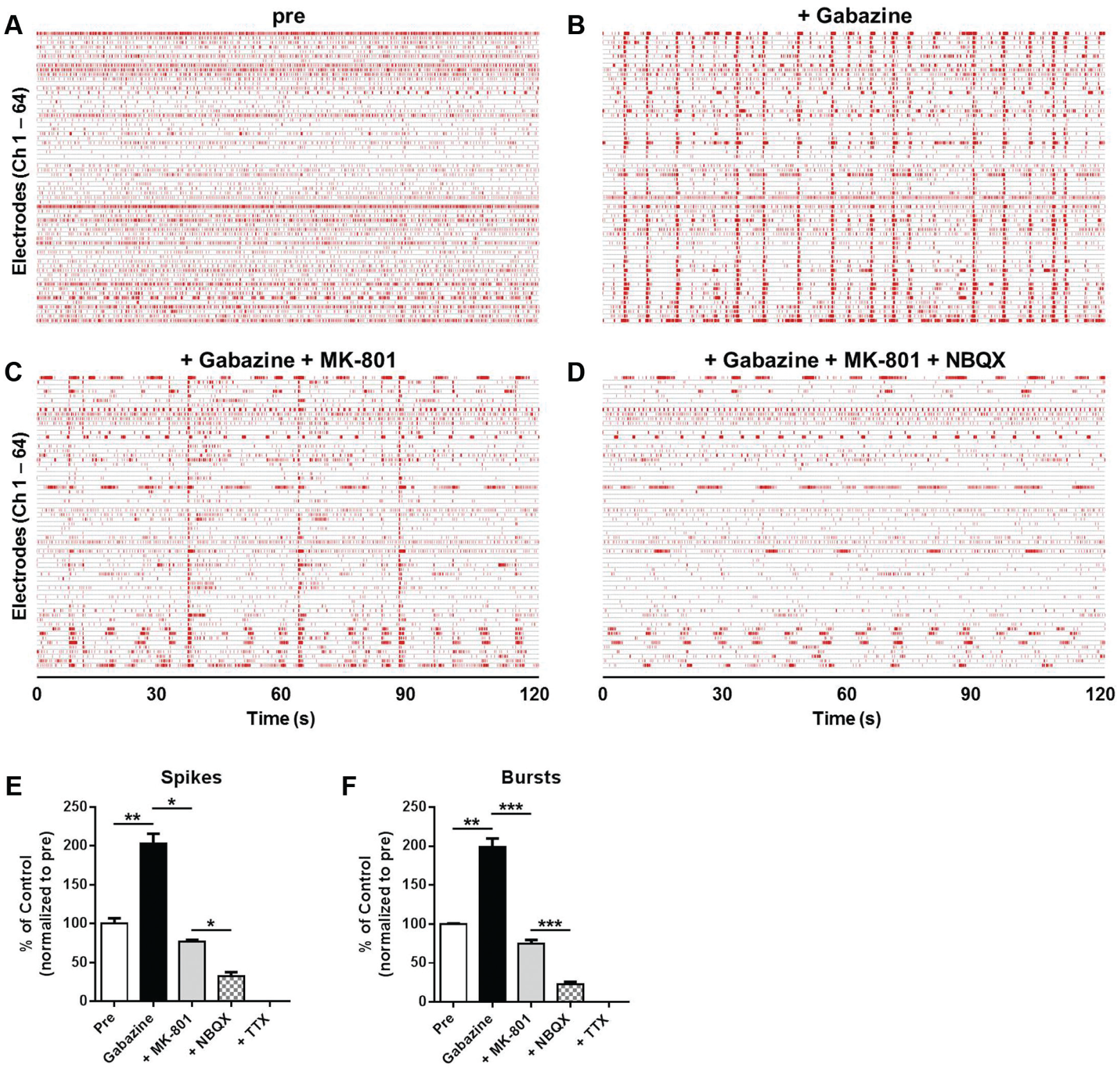
Functional evaluation of neuronal network formation among HIP-009 neurons cultured in the conventional medium supplemented with rat astrocyte-conditioned medium for 8 weeks. Gabazine (30 µM), MK-801 (10 µM), and NBQX (10 µM) were sequentially applied to differentiated HIP-009 cells in this order. Representative raster plots of HIP-009 neurons before the application of gabazine (
Pharmacological Assessments of Reference Compounds Acting on Neuronal Networks
We evaluated the effects of several compounds on human neuronal network functions of HIP-009 neurons. In
Table 1
, effects of the compounds on the number of spikes are summarized. Agonists of glutamate receptors (glutamate), NMDA receptors (NMDA), AMPA receptors (AMPA), and kainate receptors (kainic acid) increased the number of spikes in a concentration-dependent manner; EC50 values were 1.1 ± 0.1, 0.52 ± 0.28, 0.07 ± 0.02, and 0.09 ± 0.02 µM, respectively. In the assays of kainic acid, a decrease of the number of spikes was observed at 10 µM (
Summary of Effects of Compounds on the Number of Spikes of Differentiated HIP-009 Neurons.
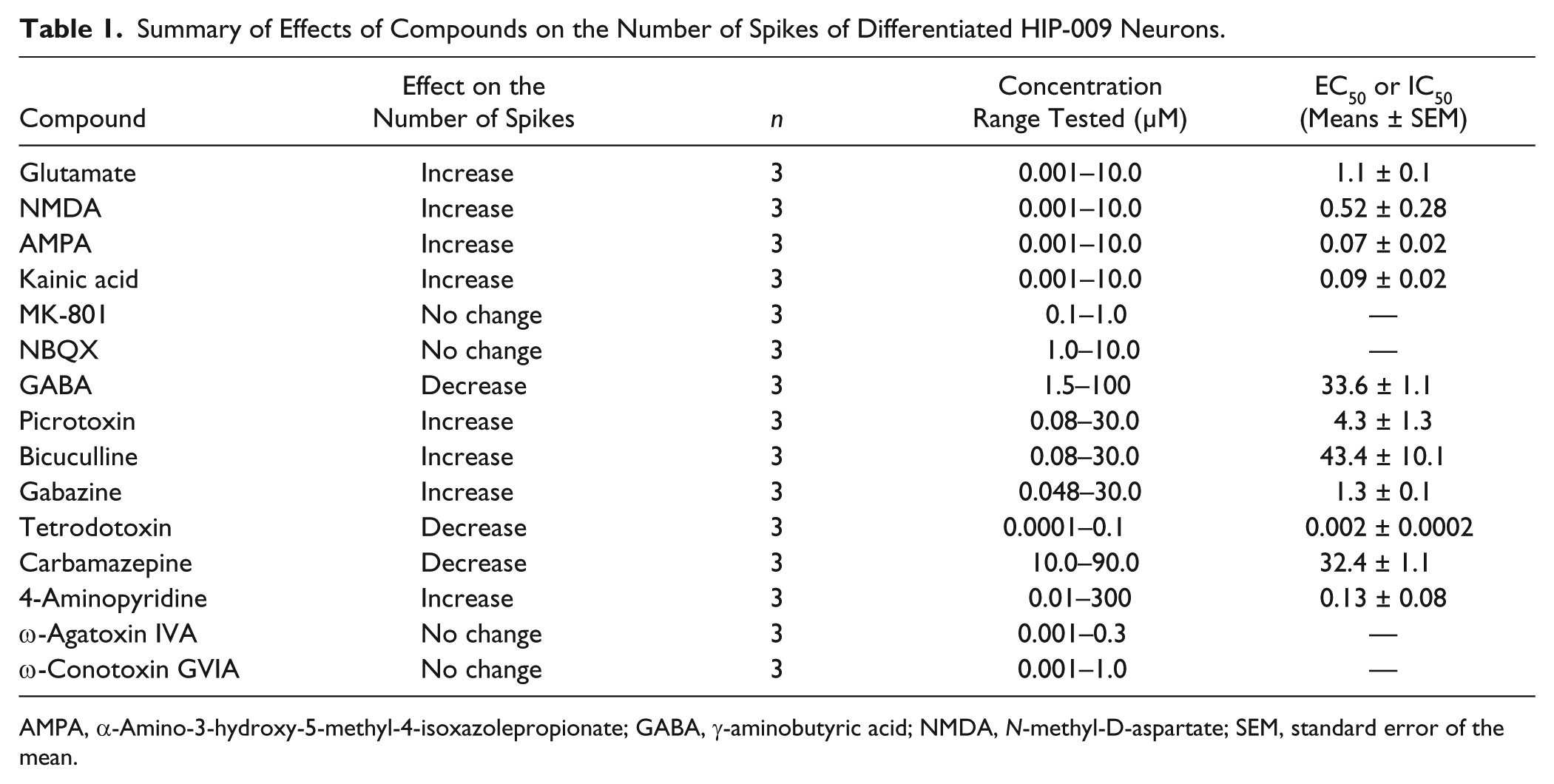
AMPA, α-Amino-3-hydroxy-5-methyl-4-isoxazolepropionate; GABA, γ-aminobutyric acid; NMDA, N-methyl-D-aspartate; SEM, standard error of the mean.
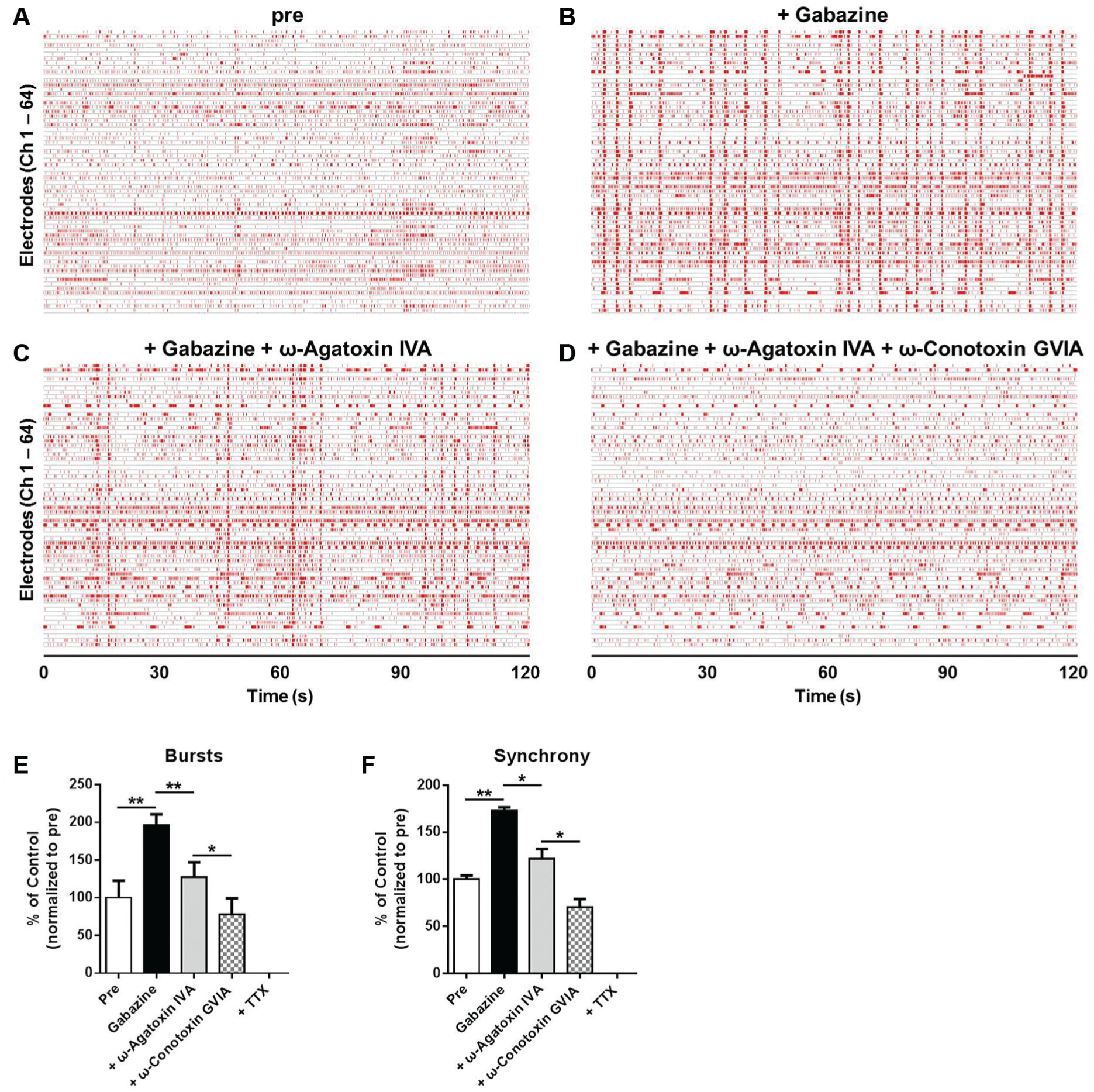
Effects of calcium channel blockers on neuronal network functions of HIP-009 neurons cultured in the conventional medium supplemented with rat astrocyte-conditioned medium for 8 weeks. Gabazine (30 µM), ω-agatoxin IVA (100 nM), and ω-conotoxin GVIA (100 nM) were sequentially applied to differentiated HIP-009 cells in this order. Representative raster plots of HIP-009 neurons before the application of gabazine (
Discussion
In this study, we observed that firing activities of HIP-009 neurons were increased as the differentiation period was longer even in the conventional medium. In contrast, culturing human iPSC-derived neurons alone, Odawara et al. 10 reported that firing activities of the neurons were decreased with the cultivation time extended, and that no firings were seen after a 4-week culture. They demonstrated, however, that firing activities were elevated in prolonged culture when the human neurons were co-cultured with rat astrocytes. Astrocytes play important roles in synapse and neuronal circuit formation through direct contact with neurons. 5 HIP-009 cells are able to differentiate into neurons and astrocytes simultaneously. Thus, our data suggest that human astrocytes differentiated from HIP-009 cells promote synapse formation among HIP-009 neurons.
We also observed that supplementation of rat ACM accelerated the electrophysiological maturation of HIP-009 neurons. Microarray analysis suggests that rat ACM supplementation promoted maturation of HIP-009 neural cells at the gene expression level. Comparing with HIP-009 cells differentiated in the conventional medium plus FBS, it was shown that FBS itself had little effect on the gene expression. It was reported that astrocytes communicated with neurons in an indirect way; calcium signaling in astrocytes is induced by neurotransmitters released in synaptic activity, and, in turn, astrocytic calcium signaling induces the release of gliotransmitters [e.g., glutamate, GABA, D-serine, ATP, tumor necrosis factor α (TNFα), and BDNF] that modulate synaptic activity. 17 Such astrocyte-derived humoral factors are considered to be contained in ACM. Thrombospondins are also astrocyte-secreted proteins that were reported to promote synaptic maturation of rat primary hippocampal neurons through an interaction with the α2δ-1 calcium channel subunit. 18 Beattie et al. 19 showed that astrocyte-derived TNFα enhanced the synaptic strength of rat hippocampal neurons and slices by increasing trafficking of AMPA receptors to the cell surface. Taken together with these reports, we considered that accelerated electrophysiological maturation of HIP-009 neurons by the addition of ACM is caused by concentrated glia-derived neurotrophic factors contained in it.
In addition to the roles in synapse formation, astrocytes are involved in protection of neurons from excitotoxicity by maintaining the extracellular glutamate properly. 20 Scelfo et al. 21 reported by using rat cortical neurons that the number of spikes of the neurons was decreased by kainic acid in a concentration-dependent manner over 50 nM due to excitotoxicity. On the other hand, kainic acid increased the number of spikes of HIP-009 neurons in a concentration-dependent fashion up to 1 μM, and a decrease of the firing rates was just observed at 10 μM. Although species difference should also be taken into account, our data suggest that human astrocytes differentiated from HIP-009 cells protect HIP-009-derived neurons from excitotoxicity of kainic acid.
In view of direct neuron-to-astrocyte contact interactions and paracrine effects, co-culture of human neurons and astrocytes is an essential tool to mimic human physiologically relevant conditions in vitro. An advantage of HIP-009 cells is that human neurons and astrocytes are obtained simultaneously and definitely in the same culture without specific and complicated manipulation. In contrast, it is difficult to differentiate both types of cells from human ESCs and iPSCs in the same culture by using currently available neural differentiation protocols. Another way is differentiating neurons and astrocytes from human ESCs and iPSCs separately and then mixing them to co-culture. However, the methods for differentiating human ESCs and iPSCs into astrocytes are still cumbersome, although their improvement is continued. 13 Therefore, HIP-009 cells are a simple and powerful tool accelerating the establishment of a co-culture system of human neurons and astrocytes.
To verify whether functional neuronal networks were formed among HIP-009 neurons, we examined effects of sequential application of gabazine, MK-801, and NBQX on firing activities of the cells. We observed that gabazine increased the firing activities of HIP-009 neurons, which resulted from a shift of excitatory-inhibitory balance to excitatory dominance on the inhibition of postsynaptic GABAA receptors. Gabazine-induced excited firings were decreased under the level of the baseline by the co-treatment of MK-801 and NBQX, which was attributed to the blockade of postsynaptic NMDA, AMPA, and GABAA receptors. These results suggest that functional synapses and networks are formed among HIP-009 neurons.
By using the HIP-009/MEA assay system, we investigated effects of compounds acting on synaptic receptors or ion channels involved in neuronal network functions. To our knowledge, this is the first report to conduct pharmacological evaluations of various compounds by using the co-culture system of human neurons and astrocytes, whereas there have been reports assessing effects of some compounds on neuronal network functions of rodents by using the MEA system.7,22−24 Johnstone et al. 7 showed that a blocker of NMDA receptors alone (e.g., MK-801 or memantine) caused no changes in the electrophysiological activities of rat hippocampal slices. In accordance with this report, MK-801 did not affect the firing rates of HIP-009 neurons. As a result of GABAA receptor blockers, bicuculline and picrotoxin increased the number of spikes of HIP-009 neurons with the EC50 values of 43.4 ± 10.1 and 4.3 ± 1.3 µM, respectively. Mack et al. 22 showed that bicuculline and picrotoxin elevated the number of firings of rat cortical neurons with EC50 values of 0.41 and 0.15 μM, respectively, by using the MEA system. The differences of the EC50 values between HIP-009 neurons and rat cortical neurons might reflect the species difference, although the difference in brain regions should also be taken into account. Moreover, we tested an antiepileptic drug, carbamazepine, in the HIP-009/MEA assay system. This drug decreased the number of firings with the IC50 value of 32.4 ± 1.1 µM, which corresponds to its clinical serum concentration (15−50 µM). 7
In the assessment of calcium channel blockers, we observed that ω-agatoxin IVA alone or ω-conotoxin GVIA alone had no effect on the number of spikes of HIP-009 neurons, whereas these blockers inhibited gabazine-evoked synchronized bursts. It was previously reported that the blockade of GABAA receptors induced synchronized bursts of rat hippocampal neurons. 23 In the synchronous network activity, calcium influx occurs through presynaptic P/Q- and N-type calcium channels in the CNS. The calcium influx, in turn, triggers synaptic vesicle fusion and neurotransmitter release. These presynaptic calcium channels, together with other presynaptically active regulatory proteins, such as synaptotagmin, syntaxin, SNAP-25, and complexin, are involved in synchronous neurotransmitter release.25−27 By using the MEA, Knaack et al. 24 demonstrated that ω-agatoxin IVA had no change in spontaneous firings of a cluster of mouse cortical neurons, but it inhibited bicuculline-induced signals of the neurons, which is in agreement with our findings. Therefore, it is demonstrated that multiple parameters, including firing, burst rates, and synchrony, are important to properly evaluate the effects of compounds on neuronal network functions.
In conclusion, we established the novel MEA assay system for evaluating functions of human neuronal networks in a co-culture of human neurons and astrocytes derived from fetal neural stem/progenitor cells. This unique assay system relevant to the human CNS is applicable to in vitro pharmacological evaluations of compounds that affect human neuronal network functions. In combination with other technologies, such as gene modification and genome editing of HIP-009 cells, it should help to better understand the mechanisms underlying neurological disorders and neurotoxicity, leading to the development of novel therapeutic agents for the diseases.
Footnotes
Acknowledgements
We thank Dr. Yoichi Imaizumi for technical advice and assistance. We are also grateful to Dr. Ikuro Suzuki for valuable advice.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
