Abstract
Ras homologous (Rho) family GTPases act as molecular switches controlling cell growth, movement, and gene expression by cycling between inactive guanosine diphosphate (GDP)- and active guanosine triphosphate (GTP)-bound conformations. Guanine nucleotide exchange factors (GEFs) positively regulate Rho GTPases by accelerating GDP dissociation to allow formation of the active, GTP-bound complex. Rho proteins are directly involved in cancer pathways, especially cell migration and invasion, and inhibiting GEFs holds potential as a therapeutic strategy to diminish Rho-dependent oncogenesis. Methods for measuring GEF activity suitable for high-throughput screening (HTS) are limited. We developed a simple, generic biochemical assay method for measuring GEF activity based on the fact that GDP dissociation is generally the rate-limiting step in the Rho GTPase catalytic cycle, and thus addition of a GEF causes an increase in steady-state GTPase activity. We used the Transcreener GDP Assay, which relies on selective immunodetection of GDP, to measure the GEF-dependent stimulation of steady-state GTP hydrolysis by small GTPases using Dbs (Dbl’s big sister) as a GEF for Cdc42, RhoA, and RhoB. The assay is well suited for HTS, with a homogenous format and far red fluorescence polarization (FP) readout, and it should be broadly applicable to diverse Rho GEF/GTPase pairs.
Keywords
Introduction
There are more than 150 small monomeric GTPases in the Ras superfamily distributed among the Ras, Rho, Arf, and Rab classes, based on sequence and functional similarities. These proteins are binary switches that transduce signals in diverse processes such as cellular morphology and movement, growth, apoptosis, and gene expression. 1 The GTPase switch is active when guanosine triphosphate (GTP) is bound and inactive following hydrolysis, after which guanosine diphosphate (GDP) remains tightly bound. Return to the active state is positively regulated by guanine nucleotide exchange factors (GEFs), which bind to GTPases and accelerate GDP dissociation. GTPase activating proteins (GAPs) play an opposing role by accelerating GTP hydrolysis. Rho proteins, a 20-member subfamily of Ras GTPases, transduce extracellular cues to intracellular pathways affecting cytoskeletal dynamics. Rho misregulation contributes to tumorigenesis and metastasis in diverse cancers. 2 Though an activating Rac1P29S mutation was recently found in melanoma, 3 oncogenic mutations in Rho proteins are relatively rare 2 and their aberrant activation results indirectly, often via overexpression or receptor-mediated activation of GEFs. 4 Notably, several Rho GEFs were originally classified as oncogenes. 4
GEFs outnumber Rho GTPases by more than three to one and are believed to play a key role in the spatiotemporal and effector control of Rho signaling, as they have been shown to control localized subcellular pools of a given Rho in different pathways contemporaneously.4,5 Of the 80 known GEFs, the majority are found in the Dbl (diffuse B-cell lymphoma) family, consisting of a catalytic Dbl homology (DH) and an adjacent pleckstrin homology (PH) domain. 4 Most GEFs contain multiple protein–protein interaction domains, and evidence suggests they control the specificity of Rho signaling to downstream pathways. 1 Therefore, development of GEF inhibitors that specifically disrupt GEF action on a target Rho GTPase may be a promising approach for blocking pathogenic Rho signaling, potentially without interfering with its normal functions.
The most widely used biochemical assay for GEF activity measures association rates of fluorescent probes such as N-methylanthraniloyl (Mant)-GTP or BODIPY-GTP, which increase in brightness upon binding to GTPases. Though Rho GTPases are especially difficult to target with the environmentally sensitive labels, the method is still used to test compounds identified in virtual screens, especially with Ras subfamily GTPases. However, this method is not suitable for HTS, as it requires high protein concentrations and kinetic reads, leading to complicated plate handling and data analysis, especially when screening large compound libraries. The GTP binding approach was modified for HTS by using endpoint detection and a far red BODIPY-GTPγS probe. 6 However, even with very high concentrations of protein and probe (2 μM RhoA and 500 nM probe), the signal window was relatively narrow (40–50 mP). Most recently, a version of the GTP binding assay was developed using quenching resonance energy transfer. 7 In this format, binding of a GTP-Europium chelate conjugated to GTPase relieves its quenching by a soluble quencher. The method was used to measure activation of RhoA by the Ect2 GEF with a signal-to-background ratio of 3.2.
An alternative to GEF catalytic assays is the use of protein complementation assays (PCAs) to measure protein–protein interactions. For example, a PCA could be constructed to measure GTPase and GEF interactions, each conjugated with complementary halves of fluorescent protein or reporter enzyme domains. The split domains are brought into proximity to generate a signal upon binding of the GTPase and GEF. This approach was originally developed, and has been used extensively, for GEF biosensors used in cellular and in vivo studies. More recently it was applied for a cell-free protein–protein interaction assay to monitor Rho–GEF interactions, using a split luciferase as the reporter. 8 An advantage of the approach is its sensitivity; however, it takes some effort to express, purify, and validate each PCA.
Small-molecule screening efforts have been limited to low-throughput approaches; for example, nuclear magnetic resonance (NMR)–based fragment screening was used to identify inhibitors that disrupt activation of Ras by SOS. 9 A modified yeast two-hybrid system (essentially a protein–protein interaction assay) has been used to find Rho GEF inhibitors. 10 There has been one activity-based biochemical screen for GEF inhibitors, a 10,000-compound screen with LARG/RhoA that used a fluorescence polarization (FP)–based assay for BODIPY-GTP binding. 6
The most potent of the GTPase binding GEF inhibitors identified thus far has an IC50 of 1 μM in cellular assays. 11 There is only one report of an inhibitor that binds to a GEF rather than to a GTPase, and it exhibited an apparent IC50 of 80 nM. 12 Despite their low potency and the limited nature of the screening and design efforts, all of the compounds identified to date exhibit some level of selectivity for both the target GTPase and its interacting GEF. For instance, a Rac1/Vav inhibitor based on the NSC23766 scaffold showed approximately 5- to 10-fold selectivity for Rac1, and the closely related Rac3, over Cdc42. 11 There are no GEF inhibitors in clinical trials; however, a relatively nonspecific GEF-Rac1/Cdc42 inhibitor based on the NSC23766 scaffold suppressed tumor growth and increased survival in a mouse xenograft model for prostate cancer. 13
Though large numbers of compounds have been virtually screened for GEF inhibition, almost all of these efforts have targeted the same site on the GTPase. 11 The low potency of current inhibitors, even after efforts to improve upon first-generation scaffolds, may be due to the nature of the binding site or the fact that the chemical diversity tested in vitro has been quite limited. Applying HTS on large, diverse chemical collections using a mechanistically unbiased assay could result in the identification of different inhibitor classes. These classes might include inhibitors that bind directly to GEFs or to different sites on GTPases, including those acting at a dimer interface. In any event, virtual screening or rational design requires detailed structural information on the GEF/GTPase complex, which is currently available for only 11 of the multitude of potential pairs.
Our goal was to develop a mechanistically unbiased GEF catalytic assay that is well suited for HTS. To achieve this, we used the Transcreener GDP FP Assay, which relies on selective immunodetection of GDP, to measure the GEF-dependent stimulation of steady-state GTP hydrolysis by Rho GTPases. Transcreener is a highly validated HTS assay platform used for large-volume screening of enzymes, including GTPases and GAPs, on automated HTS platforms in 384- or 1536 well formats. 14 The selectivity of the antibody enables detection of GDP in the presence of excess GTP. The GEF assay approach is based on the fact that GDP dissociation is generally the rate-limiting step in the Rho GTPase catalytic cycle. By accelerating the release of GDP, a GEF causes an increase in steady-state GTPase activity, which is detected using the Transcreener GDP Assay. Unlike fluor-GTP binding assays, this method is based on multiple catalytic cycles for both the GEF and the GTPase with the unmodified GTP substrate; thus, it is a measure of functional GEF activity. Here we show proof of concept for this method, using Dbs as a GEF for RhoA, RhoB, and Cdc42. The assay should be applicable to most, if not all, GEF/GTPase pairs, as GDP dissociation is generally the rate-limiting step in catalysis for GTPases in the Ras superfamily.
Materials and Methods
Reagents
The Cdc42 and RhoA proteins were expressed and purified at BellBrook Labs. RhoB was obtained from Abcam (Cambridge, MA; catalog no. ab107139). The active hDbs DH/PH GEF protein domain (residues 629–967) was obtained from Cytoskeleton (Denver, CO; catalog no. GE01). Assays were run in black nonbinding, low-volume 384-well plates (Corning; catalog no. 4514). Transcreener GDP Assay reagents were provided by BellBrook Labs (catalog nos. 3009, 3014, and 3021). The GTPase buffer was comprised of 20 mM Tris at pH7.5, 1 mM EDTA, 10 mM MgCl2, and 0.01% Brij 35.
Enzyme Assays
RhoA and Cdc42 enzyme reactions in the presence and absence of GEF were run in 384-well plates at 30 °C in GTPase buffer and were initiated by the addition of GTP at a final concentration of 1 μM. RhoB in the presence and absence of GEF was run under the same conditions, except the GTP was at a final concentration of 10 µM. For assays run in kinetic mode, Transcreener GDP FP reagents were present and reactions were monitored at timed intervals in a 20 μL reaction volume. For endpoint assays, 10 μL enzyme reactions were quenched at indicated times, generally 2–3 h with 10 μL of Stop & Detect Buffer B (described below), and plates were read. Rho GTPase and Dbs titrations were done in twofold serial dilutions by adding 10 μL enzyme aliquots to wells containing 10 μL of GTPase buffer.
Transcreener GDP FP Assays
The concentration of GDP Antibody determines the dynamic range of the assay. Accordingly, the GDP Antibody was titrated into mock reactions containing tracer, GTP, and GTPase buffer, and EC85 concentrations were determined. The final concentrations of Transcreener GDP Antibody and Alexa633 Tracer present in the kinetic and endpoint assays were 1.04 μg/mL and 2 nM, respectively, except for the reactions containing RhoB, where they were 10 μg/mL and 4 nM, respectively. Ten microliters of Stop & Detect Buffer B (20 mM HEPES [pH 7.5], 40 mM EDTA, and 0.02% Brij 35) was used to quench the endpoint assays, bringing the final volume to 20 μL. Plates were read using 633 nm excitation and 670 nm emission settings to measure polarization values, expressed as millipolarization units (mP). The ΔmP values are the difference between the mP values with and without enzymes, unless otherwise indicated. Values in graphs are means of data from at least two separate experiments, and error bars represent ± standard errors of replicates. The optimized assay conditions were tested on three separate days. EC85 values were calculated by using the EC50 and hillslope values, fitting the equilibrium binding data to a variable slope sigmoidal dose–response curve using the equation EC85= {[85/(100 – 85)]1/hillslope}*EC50. Data were analyzed using GraphPad Prism. Standard curves mimicking the conversion of 1 μM and 10 µM GTP to GDP were prepared as described in the Transcreener GDP FP Assay Technical Manual and used to convert polarization data to GDP formed.
Results and Discussion
Rho GTPases exhibit slow catalytic rates, consistent with their function as binary switches that are on when GTP is bound. The rate-limiting step in the GTPase catalytic cycle is generally the slow dissociation of tightly bound GDP from its guanyl nucleotide binding site. The GEF proteins stimulate the exchange of GTP for GDP on GTPases; therefore, we reasoned that they should increase the steady-state rate of GTPase activity.
To test our hypothesis, we first demonstrated we could detect Rho GTPase activity using the Transcreener GDP FP Assay. The assay relies on detection of GDP using a competitive FP immunoassay ( Fig. 1a ). GDP produced by a GTPase displaces a fluorescent tracer from the antibody, resulting in a decrease in polarization due to increased rotational mobility. The antibody is more than 100-fold specific for GDP versus GTP, which allows sensitive measurement of GTPase initial velocity, that is, detection of GDP in the presence of excess GTP. The assay was calibrated for initial velocity GTPase measurements using 1 μM GTP, and a standard curve was generated for converting raw polarization data to the amount of GDP formed in the reactions ( Fig. 1b ). We titrated two Rho GTPases, RhoA and Cdc42, and observed a robust response with the assay, with both GTPases generating maximal polarization shifts of more than 150 mP ( Fig. 1c ). From these curves, we selected GTPase concentrations that generated approximately 20% of the maximal response to use for detecting GEF effects: 39 nM Cdc42 and 78 nM RhoA.
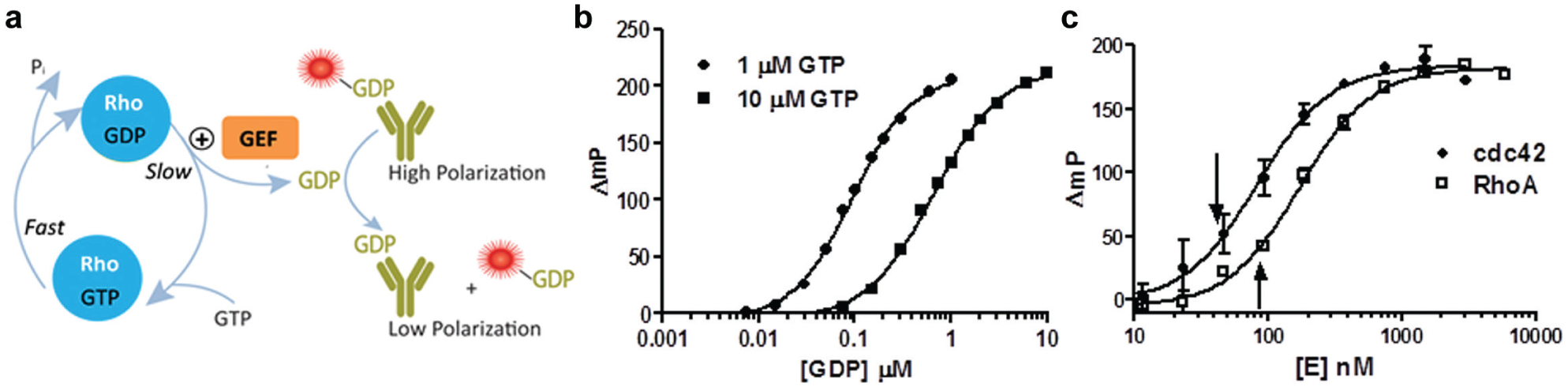
(
When Dbs was added to reactions containing the low concentrations of RhoA and Cdc42, we observed dose-dependent increases in the assay signals ( Fig. 2a ), consistent with our hypothesis that accelerating GDP dissociation would increase the steady-state rate of GTPase activity. We selected 100 nM as an optimal Dbs concentration and ran the combined reactions in kinetic mode ( Fig. 2b,d ) to demonstrate that the GEF is indeed affecting the rate of GDP formation. Note that the very low background level of GDP observed with Dbs alone is not nearly sufficient to account for the increase observed in the presence of RhoA or Cdc42; therefore, we conclude that we are observing a genuine GEF effect. The polarization values from the Rho GTPase/GEF reactions were converted to GDP concentration using a standard curve ( Fig. 2c,e ), and the GEF effect was quantified using the ratio of GTPase activity in the presence and absence of Dbs ( Table 1 ). Dbs enhanced RhoA activity by almost threefold, and Cdc42 was fivefold more active with Dbs present. These GEF effects are based on initial velocity measurements, as the highest amounts of GDP measured represented less than 20% of the GTP present at the start of the reactions ( Fig. 2c,e ). Unlike fluor-GTP binding assays, this method is based on multiple catalytic cycles for both the GEF and the GTPase with the unmodified GTP substrate. For example, the amount of GDP formed during the linear phase of the Dbs/Cdc42 reaction exceeded the Cdc42 concentration by more than fivefold (200 and 39 nM GDP and Cdc42, respectively) ( Fig. 2e ).
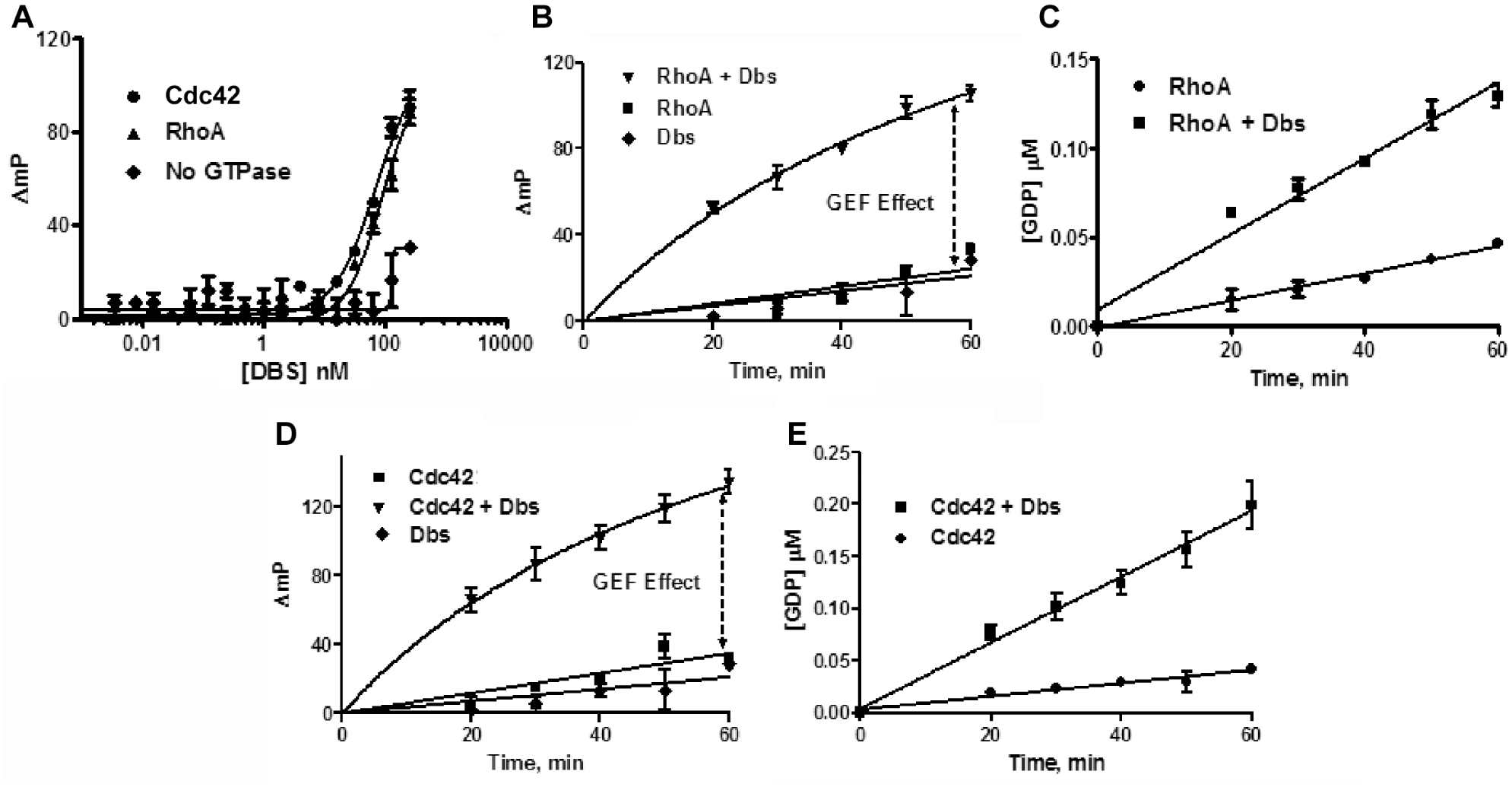
Effects of Dbs on Cdc42 and RhoA. (
Rates of GTPase Activity for RhoA, RhoB, and Cdc42 (min−1) in the Presence and Absence of Dbs.
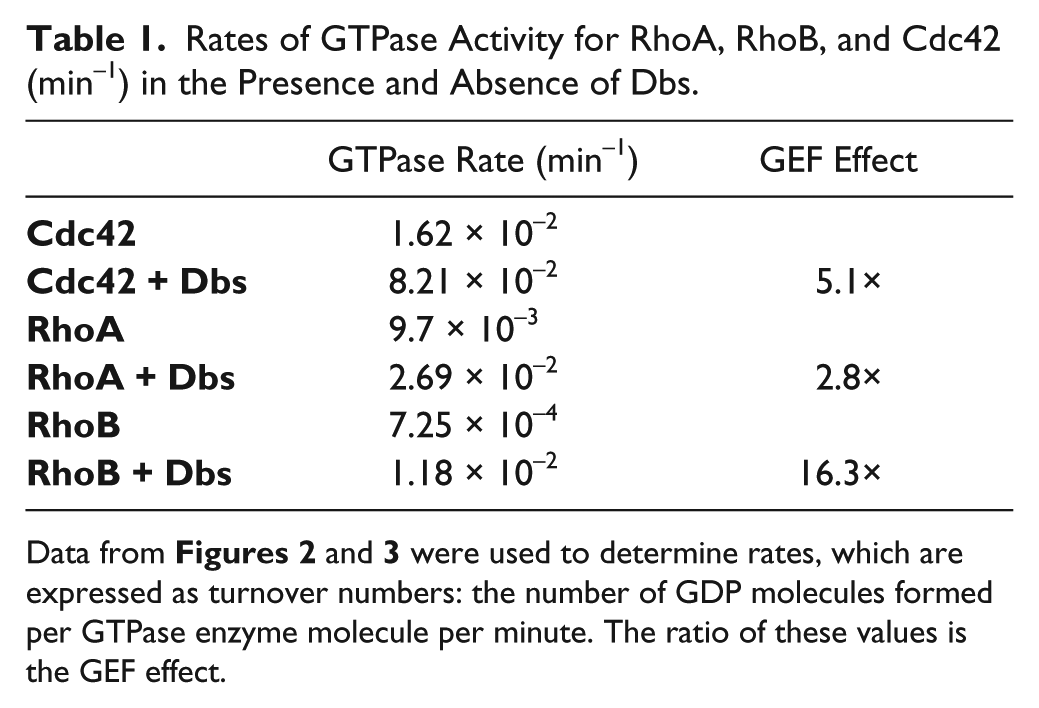
Though not as intensively studied as RhoA and Cdc42, RhoB plays important roles in endosomal trafficking and coordinating cellular signaling by other Rho proteins, particularly in vascular cells in the context of tumor-associated vasculogenesis. 15 RhoA and RhoB share 85% amino acid sequence homology. The RhoA-to-Cdc42 sequence homology is 52%, and RhoB-to-Cdc42 is 54%. In RhoB reactions, all wells contain 10 µM GTP (compared with 1 μM in RhoA and Cdc42 reactions), 400 nM RhoB, and 250 nM Dbs (protein titrations not shown). The ΔmP values obtained at time zero were taken as background and subtracted from the data at each time point to yield apparent activities ( Fig. 3a ). The polarization values from the Rho GTPase/GEF reactions were converted to GDP formation using a standard curve ( Fig. 3b ). Total RhoB-mediated GDP formation in the presence of the GEF was less than 10% of the starting GTP substrate concentration. We observed markedly lower GTPase activity for RhoB alone than with RhoA and Cdc42, even at protein and GTP levels considerably higher than were required to obtain measurable activity for RhoA and Cdc42 ( Table 1 ). However, the addition of Dbs enhanced RhoB GTPase activity 16-fold, a significantly greater GEF effect than for the other two Rho proteins we investigated. Certain Rho GTPases therefore may be more sensitive than others for activation by a GEF protein, with the proviso that activities can vary significantly for different preparations of the same enzyme, and we did not compare GTPases obtained from different sources.
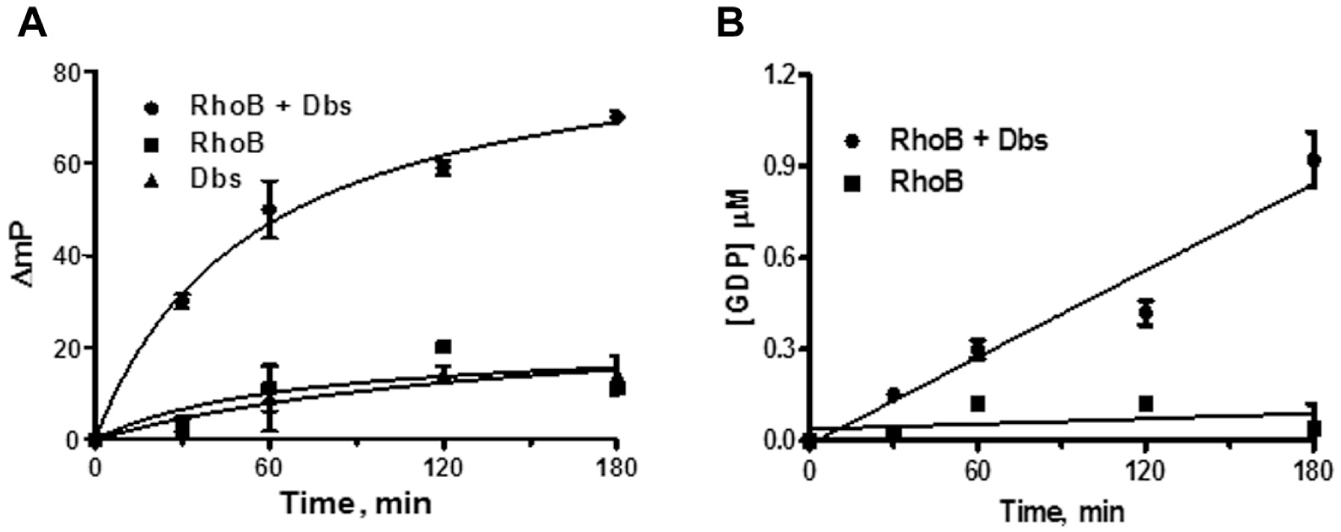
Effects of Dbs on RhoB GDP formation. Reactions were done in the presence of 400 nM RhoB, 250 nM Dbs, and 10 µM GTP and monitored over the course of 180 min. Time course data for RhoB (
We have shown that measuring increases in the rates of GTPase activity with the Transcreener GDP Assay provides a sensitive and convenient method for detecting acceleration of GDP dissociation by a GEF. Importantly, use of an assay based on dynamic catalysis by both the GEF and the GTPase could allow identification of inhibitors with diverse mechanisms, as compared with fluor-GTP binding 6 or protein interaction assays. 10 For example, the assay here is agnostic with respect to the mechanism of inhibition (e.g., competitive or allosteric), or whether the inhibitor binds to the GEF, the GTPase, or their interface. The catalytic nature of the assay also makes it much more sensitive than the current fluor-GTP binding assays, which will reduce by several-fold the amounts of recombinant Rho and GEF proteins required for screens. 6 A limitation of the assay is that it may not be applicable to some mutant GTPases if their GTP hydrolysis and/or nucleotide binding properties are altered, such that GDP dissociation is no longer the rate-limiting step in catalysis.
Though we have not yet validated the GEF-dependent Rho-GTPase assay method described here for compound screening, the Transcreener platform in general is highly validated for HTS, and we used a similar approach to screen for inhibitors of the GAP activity of the regulator of G protein signaling (RGS) proteins with Gα proteins. 14 The Z′ of the screen was 0.82 and the Z factor was 0.73. As in that effort, compounds that inhibit the intrinsic GTPase activity can be triaged by counterscreening, so that only compounds that inhibit the GEF-dependent acceleration of GDP release, that is, GDP:GTP exchange, would be considered hits. Other GTPase assay methods could likely be adapted to this GEF assay approach. Radiometric assays for measurement of GTPase activity are very sensitive; however, they require separation of phosphate from GTP as well as the attendant radioactivity handling and disposal protocols, which are increasingly being phased out of HTS platforms. Measurements of GDP:GTP:Pi ratios can be made using high-performance liquid chromatography (HPLC); however, this method is not amenable to HTS. Colorimetric phosphate detection assays provide a measure of GTPase activity based on the liberation of inorganic phosphate. However, this approach is relatively insensitive for enzymes with low intrinsic enzymatic activity such as RhoB. The use of the Transcreener GDP Assay provides direct GDP detection with no separation steps or coupling enzymes and is more sensitive than other nonradiometric technologies, enabling detection of low-nanomolar amounts of GDP.
Many aspects of GTPase regulation by GEFs are largely undefined. Some GEFs clearly partner with several Rho proteins, whereas others may act only on a single GTPase. 4 Although some GEFs are known to activate certain Rho proteins, the majority of assumed GEF proteins have only been implicated by homology to be active. 4 The GEF assay we have developed and described here will help scientists delineate the selectivity of Rho GEFs for their GTPase substrates and to enable screening for Rho-subtype-specific GEF inhibitors. Such inhibitors could be valuable probes for illuminating the spatiotemporal control of cytoskeletal dynamics regulated by Rho proteins, and may find use as scaffolds for anticancer drugs.
Footnotes
Acknowledgements
We thank George C. Prendergast (Lankenau Institute for Medical Research) for reviewing the draft of this manuscript.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Bellbrook Labs manufactures and markets the Transcreener GDP Assay kits used in the research reported here. The authors, who collaborated in the research, declare no other potential conflicts of interest with respect to the research, authorship, and/or publication of this application note.
Funding
The authors received no financial funding for the research or its authorship.
