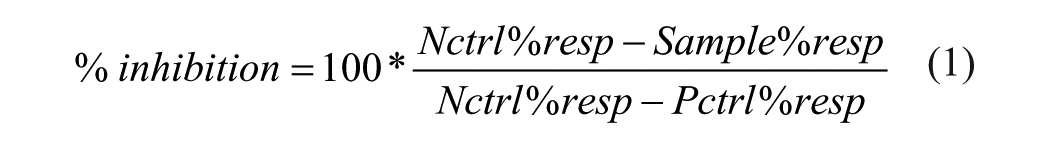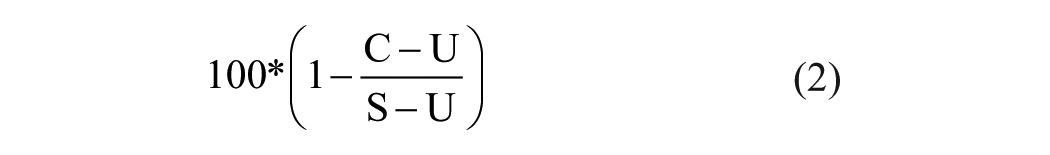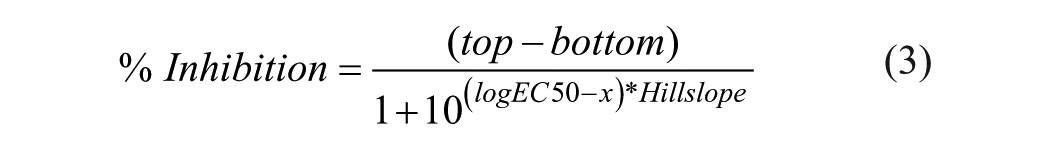Abstract
We screened the National Institutes of Health’s Molecular Libraries Small Molecule Repository for inhibitors of cytotoxic T lymphocyte (CTL) lytic granule exocytosis by measuring binding of an antibody in the extracellular solution to a lysosomal membrane protein (LAMP-1) that is transferred to the plasma membrane by exocytosis. We used TALL-104 human leukemic CTLs stimulated with soluble chemicals. Using high-throughput cluster cytometry to screen 364,202 compounds in a 1536-well plate format, we identified 2404 initial hits: 161 were confirmed on retesting, and dose–response measurements were performed. Seventy-five of those compounds were obtained, and 48 were confirmed active. Experiments were conducted to determine the molecular mechanism of action (MMOA) of the active compounds. Fifteen blocked increases in intracellular calcium >50%. Seven blocked phosphorylation of extracellular signal-regulated kinase (ERK) by upstream mitogen-activated protein kinase kinases >50%. One completely blocked the activity of the calcium-dependent phosphatase calcineurin. None blocked ERK catalytic activity. Eight blocked more than one pathway. For 8 compounds, we were unable to determine an MMOA. The activity of 1 of these compounds was confirmed from powder resupply. We conclude that a screen based on antibody binding to CTLs is a good means of identifying novel candidate immunosuppressants with either known or unknown MMOAs.
Keywords
Introduction
Immune suppression is critical to organ transplantation and treatment of autoimmune diseases. Because of problems with current agents, there is a widely recognized need for novel immune-suppressing compounds that could serve as leads for new drugs. 1 We recently developed an assay for inhibitors of cytotoxic T lymphocyte (CTL) lytic granule exocytosis. 2 CTLs kill cancerous and virus-infected cells and participate in immune processes that lead to graft rejection. One mechanism that CTLs use to kill is the exocytosis of lytic granules that contain cytolytic peptides.3,4 Our assay uses TALL-104 human leukemic CTLs 5 stimulated with soluble agents that bypass the T cell receptor, triggering maximal granule exocytosis. The screen detects the transfer of lysosome-associated membrane protein 1 (LAMP-1) from lytic granules to the plasma membrane via exocytosis.6,7 This results in exposure of an intraluminal epitope on LAMP-1 to antibodies in the external solution, enabling the quantitative fluorescent detection of cells that have responded. High-throughput screening (HTS) is feasible because sampling flow cytometers make it possible to measure cell-associated fluorescence without washing away unbound antibody. The use of a cell line and a maximal—albeit unphysiological—stimulation was chosen to ensure consistent and large enough responses for HTS.
We reasoned that HTS conducted with our method on a large and diverse compound collection could be useful for a variety of reasons. First, because the signals that control lytic granule exocytosis overlap fairly extensively with the signals that control the function of helper T cells,8,9 which synthesize and release cytokines when stimulated, screening for inhibitors of CTL function would be expected to reveal compounds that also inhibit helper T cell function. These would be broadly immunosuppressive and could help fulfill the need for novel immunosuppressants outlined above. Granule exocytosis occurs in minutes, whereas helper T cells take hours to synthesize and release cytokines, so the assay is better suited to large-scale screening than cytokine production or reporter gene expression. Second, screening lytic granule exocytosis could reveal compounds that specifically inhibit CTLs, which would enable a more specific kind of immune modulation than is currently available. Third, we surmised that a screen based on the endpoint of a cellular function controlled by a number of important signaling pathways8,9 would offer a means of identifying compounds working on known targets that could be identified in follow-up experiments, acting as a novel type of high-content screening. Finally, we anticipated that assessing the molecular mechanism of action (MMOA) of hits following screening could lead to the identification of compounds that do not work via any of the known pathways interrogated. Such compounds might involve novel molecular targets. They could serve as leads for new classes of immunosuppressives and could also be used to explore the pathways that control lymphocyte function.
Here, we describe the results of our screening of the National Institutes of Health’s (NIH) Molecular Libraries Small Molecule Repository (MLSMR) of 364,202 compounds with a 1536-well version of the assay, and follow-up experiments to explore the cellular action of confirmed active compounds, with an important goal being the identification of compounds with unknown MMOA. We identified 31 compounds that likely inhibit lytic granule exocytosis by blocking important pathways known to be involved in T cell function, as well as 8 compounds (the actions of 1 of which have been confirmed from powder) for which we could not assign an MMOA. These 8 compounds may be of potential utility in identifying novel pathways that control lymphocyte function and may represent leads to new classes of immunosuppressants.
Materials and Methods
Chemicals, Cells, and Solutions
TALL-104 human leukemic CTLs were maintained as described previously. Experimental saline (ES) was composed of 155 mM NaCl, 4.5 mM KCl, 1 mM MgCl2, 2 mM CaCl2, 5 mM HEPES, and 10 mM glucose. The pH was adjusted to 7.4 with NaOH, and 2% Bovine Serum Albumin (BSA) was added, except when noted. Powder resupply of substance ID (SID) 7977862 was from Chembridge (San Diego, CA).
HTS of the MLSMR
A BioTek MicroFlo Dispenser (BioTek, Winooski, VT) was used to sequentially transfer the following to individual wells of a 1536-well plate (#782101 HiBase; Greiner, Wemmel, Belgium): (1) 4 µL of ES, (2) 4 µL of cells (5000–6000/well), and (3) 2 µL of a mixture of thapsigargin (1 µM final), Phorbol 12-meristate 13-acetate (PMA; 50 nM final), and Alexa647-conjugated anti-LAMP antibodies (1:500 final dilution). Compounds (in 100% DMSO) or DMSO alone was transferred to wells after the RB addition step and prior to the addition of cells using an FX Liquid Handler (Beckman Coulter, Brea, CA) robot equipped with 100 nL pintools (V&B Scientific, San Diego, CA). Final compound and DMSO concentrations were 10 µM and 1%, respectively. Plates were then sealed with aluminum foil sealing film (AlumaSeal; Excel Scientific, Victorville, CA) and continuously rotated end-over-end at 8 rpm to maintain cells in suspension (surface tension retains fluid volumes at the well bottom when plates are inverted) and to facilitate mixing of well contents. Plates were first rotated for 2 h at 24 °C, then transferred to a 4 °C cold room for continuing rotation overnight. On the following day, plates were processed in 4-plate groups in which plates in each group were first allowed to warm to 24 °C (10 min or more while being continuously rotated), then sequentially transferred to a custom HyperCyt HTS platform configured with 4 sampling probes linked to 4 Accuri C6 flow cytometers (BD Biosciences, Franklin Lakes, NJ). Each probe sampled wells from a separate quadrant of the 1536-well plate at a combined rate for all 4 probes of 130–160 wells/min and 10–12 min/plate. This platform has been previously described and operationally validated elsewhere. 10 Custom-modified HyperView software (IntelliCyt, Albuquerque, NM) was used to analyze data. First, flow cytometer data files were parsed by software-based well identification algorithms to segregate data from individual wells. Next, viable cells were identified and gated based on forward and side light scatter profiles elicited by the 488 nm laser. These were then analyzed with respect to Alexa647 fluorescence intensity (FL4, 663–688 nm) excited by the 640 nm laser. A pooled fluorescence intensity histogram of unstimulated cells from all control wells containing DMSO alone (positive controls) was used to establish an intensity threshold above which we detected fewer than 5% of the unstimulated cells. Cells from compound-containing wells with fluorescence intensity exceeding this threshold were considered to be responsive to the thapsigargin–PMA stimulus mixture, and subthreshold cells were considered to be unresponsive. Typically, 80–100% of cells from control wells containing only DMSO and stimulus mixture (negative controls) were responsive.
The response range for the assay was defined as the difference between the averages of % responsive (%Resp) cells in the negative control (NCntrl, stimulated) wells and in the positive control (PCntrl, unstimulated) wells, respectively. Response inhibition mediated by compounds in sample wells was calculated as follows (Equation 1):
Compounds inhibiting 55% or more were considered to be active, a threshold chosen to generate ~2500 active compounds. Wells in which fewer than 10 cells were detected within the viability gate were not analyzed further due to insufficient sample size; 767 of 364,202 tested compounds (0.2%) were labeled undetermined as a result.
LAMP and BLT-Esterase Assays
One hundred microliters of ES-washed TALL-104 cells (2.5×106/ml) was added to each well, mixed, and incubated at 37 °C for 30 min. Five microliters of stimulation solution (ES supplemented with 20 µM thapsigargin and 1 µM PMA) or control solution (ES + 4% DMSO) was added and mixed, and the plate was incubated for an additional 90 min in the dark at room temperature. The plate was centrifuged, and 50 µl of supernatant was transferred to a new plate for BLT-esterase assays, which measure the release of granzyme B by monitoring the cleavage of a synthetic substrate, benzyloxycarbonyl-L-lysine thiobenzyl ester. 11 Meanwhile, 50 µl of ES containing 0.6 µg/ml anti-LAMP antibody was added to the wells containing the pellets, mixed, and incubated for 30 min in the dark at room temperature with constant rotation prior to the addition of 100 µl/well of 2% PFA. The geometric mean value of LAMP-1 fluorescence or the absorbance values at 410 nm were determined from unstimulated (U) or stimulated (S) DMSO-treated or stimulated compound-treated (C) cells, and the percent inhibition of LAMP externalization or BLT-esterase secretion by each compound was calculated as follows (Equation 2):
Fura-2 Measurements
Ninety microliters of ES was added to each well of a black flat-bottomed plate laid out with DMSO- and compound-containing wells, then it was mixed, and the fluorescence excited at 340 and 380 nm measured at 510 nm emission (F340 and F380) was acquired (reading 1, blank B). TALL-104 cells were loaded with 0.5 µM Fura-2 AM for 25 min in the dark at room temperature, then washed with ES and incubated for 20 min. 2.5×107 cells in 10 µl ES were added to each well of the test plate and mixed. The plate was incubated for 30 min at 37 °C and read again (reading 2). Finally, 5 µl of stimulation solution (ES supplemented with 40 µM thapsigargin) or control solution (ES + 4 % DMSO) was added and mixed. After 50 min, the third fluorescence reading was taken (reading 3). U calcium levels were reading 2 from DMSO-treated cells. S calcium levels were reading 3 from DMSO- or compound-treated cells. Blanks for each well were subtracted for F340 and F380 prior to computation of the F340:F380 ratio. Percent inhibition of calcium rise was calculated using Equation 2.
Assays for Protein Kinase C (PKC), and Mitogen-Activated Protein Kinase Kinase (MAPKK) Activity
One hundred microliters of ES-washed TALL-104 cells (2.5×106/ ml) was added to Eppendorf tubes containing compound or DMSO, and incubated at 37 °C for 30 min. Five microliters of stimulation solution or control solution (as above for LAMP and BLT-esterase measurements) was added and mixed, and tubes were incubated for an additional 50 min in the dark at room temperature with constant rotation. After incubation, cells were fixed with 100 µl of 2% PFA and permeabilized by the addition of 1 ml of ice-cold methanol. Cells were washed with FACS buffer, incubated with primary antibodies [rabbit anti-active PKC substrate (Cell Signaling 2261; Cell Signaling, Danvers, MA), 1:100; and mouse anti-p44/p42 MAPK (Cell Signaling 9106), 1:1000] in FACS buffer, then incubated with secondary antibodies (6 µg/ml Alexa488-conjugated donkey anti-rabbit and 7 µg/ml Cy 5-conjugated donkey anti-mouse) and washed. The geometric mean value of Alexa488 and Cy5 fluorescence was determined for U or S DMSO-treated or stimulated C cells. The percentage inhibition of PKC substrate phosphorylation or extracellular signal-regulated kinase (ERK) phosphorylation was calculated using Equation 2.
Immunoblotting to Detect Effects on Calcineurin Activity
TALL-104 cells were nucleofected as described previously with CaNAR1 complementary DNA (cDNA). Five h later, 1×106 ES-washed transfected cells in 400 µl were added to each tube containing 4 µl test compound at an initial concentration of 3 mM or 4 µl DMSO, mixed, and incubated for an additional 30 min at 37 °C. Six hundred microliters of stimulation solution (ES supplemented with 1.67 µM TG and 83.3 nM PMA) or control solution was added and mixed, and the tube was incubated for another 50 min in the dark at room temperature with constant rotation. Cell pellets were lysed in standard radioimmunoprecipitation assay buffer supplemented with 10 mM EGTA, 10 mM EDTA, protease, and phosphatase inhibitor cocktails, and processed for immunoblotting on nitrocellulose membranes.
Immunocytochemical Determination of Compound Effects on ERK Catalytic Activity
TALL-104 cells were nucleofected with ERK activity reporter (EKAR) cDNA. Five h later, 100 µl ES-washed transfected cells (2.5×106/ ml) were treated with test compound or DMSO and stimulated. Cells were fixed and permeabilized, then stained with rabbit anti-phospho Cdc25c antibody (Cell Signaling 9527), followed by a Cy5-conjugated secondary. FL-1 signals [corresponding to yellow fluorescent protein (YFP) expression level] and FL-4 signals (corresponding to anti-phospho-Cdc25C staining intensity) were collected. Percent inhibition was calculated as described above.
Plate Measurements and Flow Cytometry
All plate measurements for follow-up experiments were performed with a BioTek Synergy 2-plate reader (BioTek). Flow cytometry for follow-up experiments was performed on a FACSCalibur (BD Biosciences) at the University of Connecticut Storrs Flow Cytometry Facility. Flow cytometry data were analyzed with FlowJo Software (Treestar, Ashland, OR).
Results
HTS of the MLSMR Results
We performed HTS of the MLSMR in a 1536-well format using clustered high-throughput flow cytometers ( Fig. 1 ). In this implementation, 10 plates are divided into four 384-well quadrants that are analyzed by dedicated cytometers. Representative data from a validation run with half unstimulated and half TG+PMA-stimulated wells are shown in Figure 1A–1C . Using sampling of 900 ms duration (to aspirate ~2 µl of sample) with an intersampling interval of 400 ms (to aspirate an air bubble to separate adjacent samples), it was possible to acquire a plate in ~10.5 min. In validation experiments, this resulted in acquisition of 1150 ± 120 cells per sample (mean ± SD), with Z’ 12 comparing S and U cells >0.8.
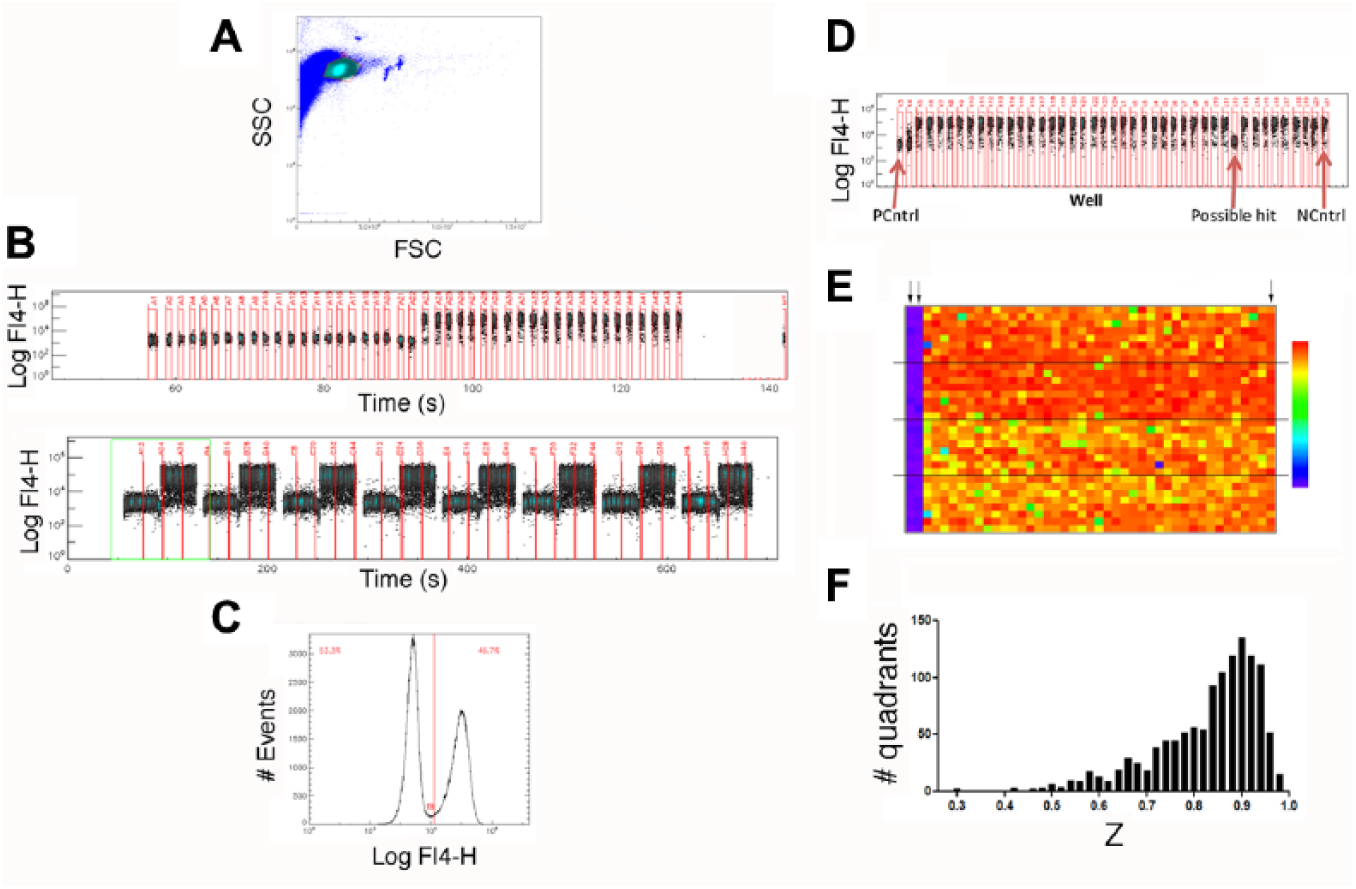
Screening the National Institutes of Health (NIH) Molecular Libraries Small Molecule Repository (MLSMR) in a 1536-well format with high-throughput cluster cytometry. (
We screened 364,202 compounds in 302 plates composed of 1208 quadrants on 21 days throughout 12 weeks. Figure 1D shows a representative row of data, and Figure 1E shows a heat map for a representative screening plate. Responses were analyzed by determining the percentage of responding cells (see below). Horizontal lines indicate the quadrants interrogated by the 4 cytometers. In each plate, 2 columns of DMSO-treated control cells remained unstimulated (left) to serve as positive controls, whereas 1 column of control cells was stimulated (right) to serve as a negative control. Samples were gated on forward and side scatter prior to analysis to exclude dead or dying cells.
We explored 2 different ways to analyze primary data from the plates. The first was to compute the mean fluorescence intensity (MFI) for anti-LAMP fluorescence for cells in the live cell gate. The second involved determining the percentage of responding cells (see Fig. 1C ). Analyzing data by determining the percentage of responding cells was found to give better reproducibility than the MFI analysis, and it was therefore used to analyze primary screening data. Using that analysis, 1195/1208 plate quadrants (98.9%) had Z values 12 > 0.5, consistent with the idea that the assay had appropriate statistical reliability for HTS. Hits from plates that had Z < 0.5 were not excluded. Seven hundred and sixty-seven wells had fewer than 10 cells in the live cell gate and were scored as inconclusive. A low event number may not reflect compound toxicity; a number of issues, such as problems with dispensing cells or acquiring samples, can reduce the event number.
We wanted to select ~2500 compounds for follow-up. Using an arbitrary threshold of 55% inhibition yielded 2404 hits. One thousand four hundred and sixty of these substances that were available and did not have obvious chemical liabilities (reactive groups, dye moieties, or unsuitable properties for chemical optimization) were obtained and retested. One hundred and sixty-one substances were confirmed active, and these were selected for dose–response testing. Dose–response measurements were performed by testing the effects of compounds at 9 concentrations ranging in a threefold dilution series from 7 nM to 15 µM. Data were fitted to Equation 3:
where top is the maximum inhibition; bottom is the response of stimulated cells in the absence of compound; logEC50 is the logarithm of the EC50 (in µM); and Hillslope is the Hill coefficient. Forty-five compounds exhibited acceptable dose-dependent inhibitory curves (monotonic dose dependence with EC50 < 10 µM and least 1 point defining an intermediate region of the curve) when analyzed using the percent positive analysis strategy described above. Additional compounds demonstrated acceptable dose–response behavior, however, when curves were fit to the MFI measurement. Based on these considerations and the availability of compounds, a resupply of 75 substances was obtained for further analysis.
Confirming the Activity of Selected Substances
We first confirmed the activity of the substances using a protocol that combined a repeat of the LAMP assay with BLT-esterase assays,
11
a standard means for measuring granule exocytosis (
Fig. 2
; see also
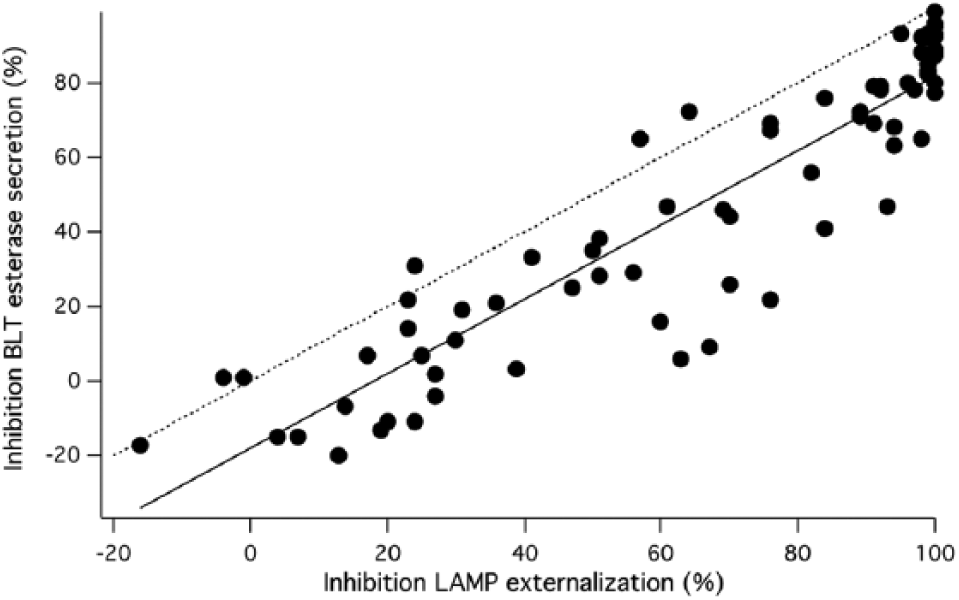
Confirming compound activity. Plot of inhibition measured in the lysosomal membrane protein (LAMP) assay versus inhibition measured in the BLT-esterase assay from the experiment shown in (
Cells were pretreated with compounds or DMSO, then, except for control wells, stimulated with TG+PMA. Fifty minutes after stimulation, plates were centrifuged, and samples of the supernatant were collected for BLT-esterase assays. The pelleted cells were stained with anti-LAMP antibodies for 15–20 min, then fixed and analyzed via flow cytometry. We have shown previously that staining cells after stimulation yields essentially similar results as stimulating them in the presence of the antibody. 13
We found that 48 substances blocked granule exocytosis by >50% as measured by LAMP staining. BLT-esterase measurements reported on average ~20% less inhibition of exocytosis than LAMP staining. Despite this, 41 substances also inhibited lytic granule exocytosis >50% as measured with the BLT-esterase assay. For 7 compounds, there was a sufficient discrepancy between the 2 measures of exocytosis that compounds scored as active on the basis of LAMP externalization were scored as inactive based on BLT-esterase assays. A number of factors, including a modest degree of compound toxicity, could be responsible for this. Those compounds were further investigated.
A Strategy for Identifying MMOA of Active Substances
Follow-up experiments were intended to determine the mechanism by which hit compounds block exocytosis (
Testing for Inhibition of [Ca2+]i Increases
To assess effects on intracellular [Ca2+] ( Fig. 3 ), we measured the fluorescence of substance-containing solutions at 340 and 380 nm excitation prior to adding Fura-2 loaded cells. We then dispensed Fura-2 loaded cells into the wells and incubated them for 15 min with test substances. We measured fluorescence at 340 and 380 nm excitation to estimate resting [Ca2+]i levels, then stimulated cells with TG and measured fluorescence again after 50 min, a time point at which [Ca2+] elevations depend entirely on Ca2+ influx. We found that 8 of the substances that blocked lytic granule exocytosis had sufficiently high fluorescence at 340 and/or 380 nm excitation that we were not able to acquire meaningful Fura-2 signals from cells treated with them. Of the substances with sufficiently low fluorescence that we could acquire Fura-2 signals, 15 inhibited increases in the Fura 340:380 ratio by 50% or more. We retested the highly fluorescent substances with the related dye Fura-Red in flow cytometry, because this dye has the same Kd for calcium as Fura-2 but different spectral characteristics. We loaded cells with Fura-Red, then treated them with substances for 15 min at 37 °C. Half of the substance-treated cells were stimulated with TG+PMA, and half were left unstimulated. Samples were analyzed 50 min later. We found that 6 out of the 8 substances decreased the Fura-Red fluorescence of unstimulated cells in at least 1 of the trials, an effect that could be an artifact related to leakage of the dye from cells or that could reflect elevation of resting calcium levels. Because these possibilities cannot be distinguished with a single wavelength indicator, we were able to measure with confidence only the blocking effects of 2 substances, neither of which inhibited calcium signals >50%. Figure 3 plots the percent inhibition of LAMP-1 responses versus the percent inhibition of Fura signals for substances that decreased exocytosis by >50% (filled squares). The 6 substances for which we could not reliably measure effects on [Ca2+]i with Fura-2 or Fura-Red were not pursued further.
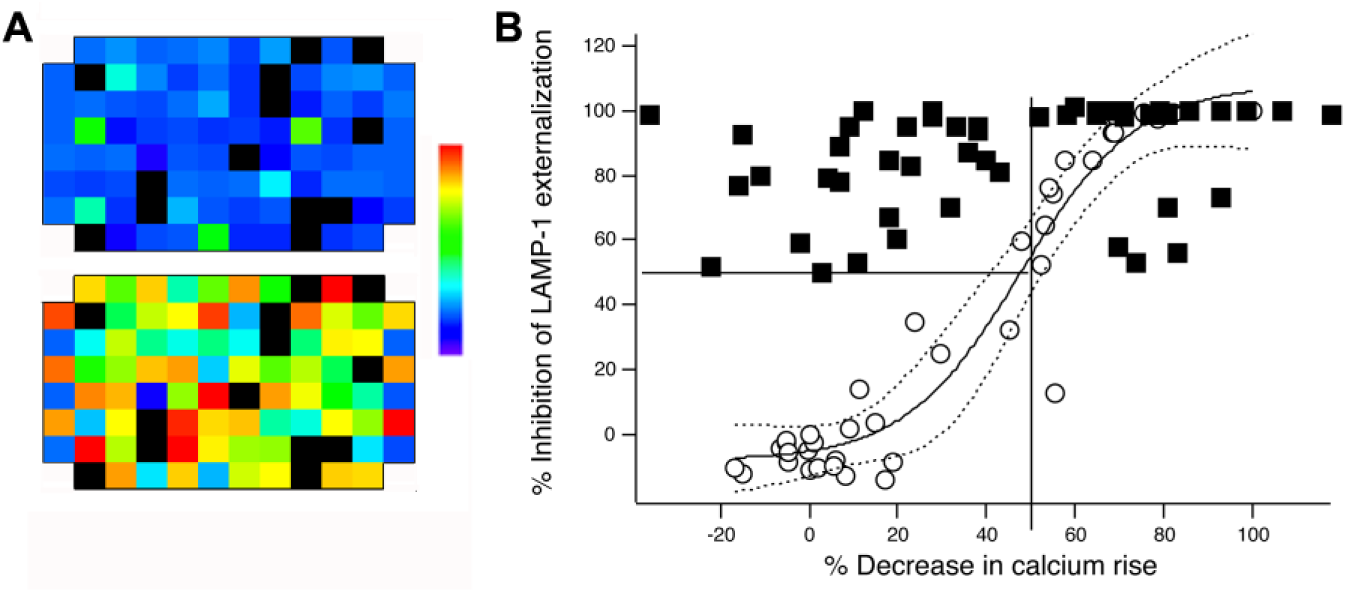
Measuring effects of compounds on calcium influx. (
Overall, 15 of 48 exocytosis-inhibiting substances blocked Fura signals by >50% ( Fig. 3 and Table 1 ), a level that we thought was appropriate to use as a cutoff to discriminate compounds likely to work by blocking Ca2+ increases [all hits are listed by their SIDS, and information about them can be found by searching PubChem Substance: http://www.ncbi.nlm.nih.gov/pcsubstance]. To confirm that this level of inhibition of Ca2+ mobilization is sufficient to block exocytosis, we measured Fura-2 ratios and lytic granule exocytosis while varying extracellular Ca2+ from 0 to 2 mM ( Fig. 3 , open circles). We plotted the relative amount of exocytosis at each concentration of Ca2+ versus the Fura-2 ratio we measured, allowing us to estimate the dependence of granule exocytosis on Fura-reported measures of intracellular [Ca2+]. Inhibiting the increase in Fura ratios by 50% or more reduced lytic granule exocytosis by >50%. Thus, we considered compounds that block exocytosis by >50% but inhibit Ca2+ increases <50% as likely to have an alternate MMOA.
Summary of MMOA Testing.
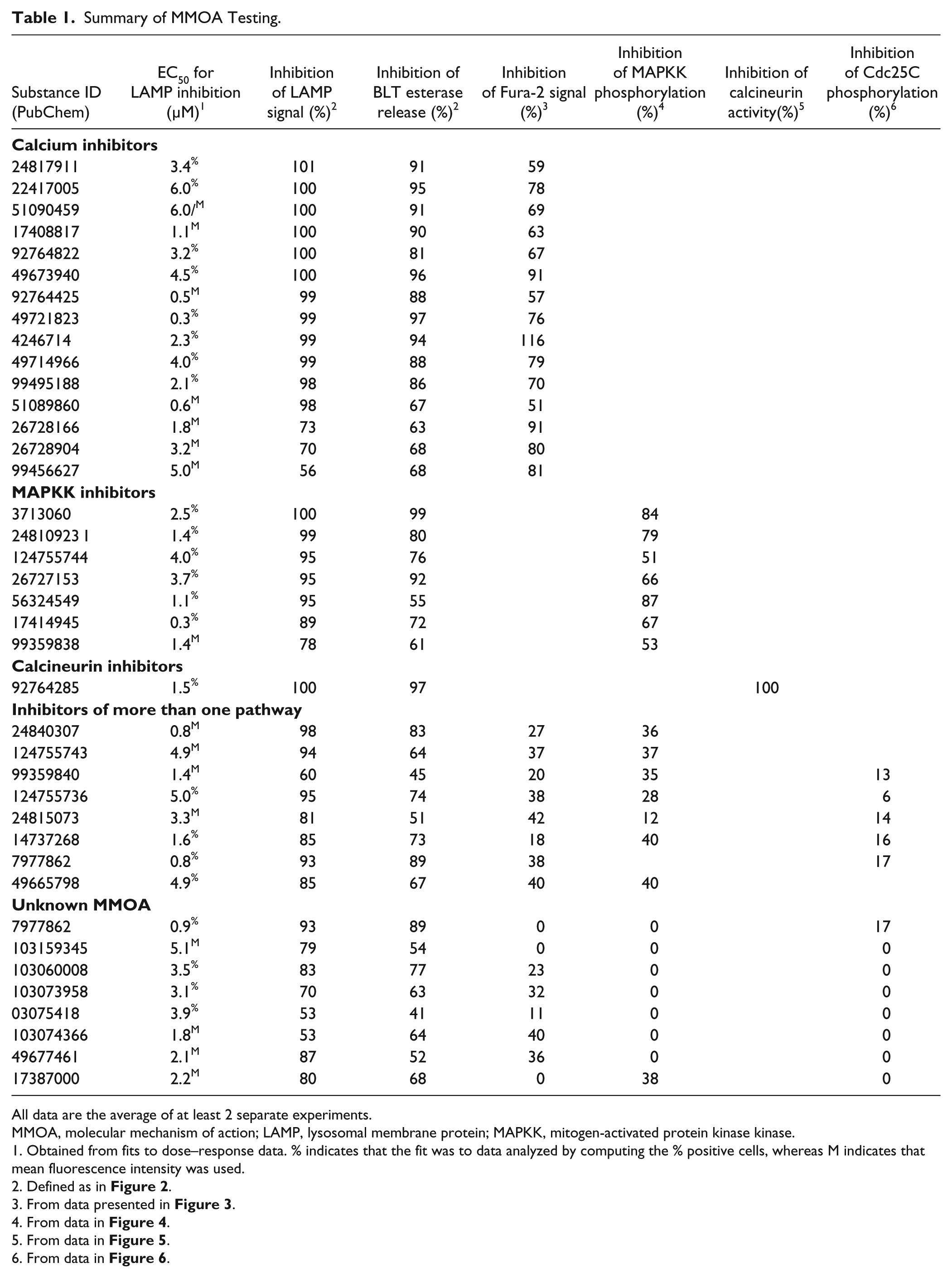
All data are the average of at least 2 separate experiments.
MMOA, molecular mechanism of action; LAMP, lysosomal membrane protein; MAPKK, mitogen-activated protein kinase kinase.
Obtained from fits to dose–response data. % indicates that the fit was to data analyzed by computing the % positive cells, whereas M indicates that mean fluorescence intensity was used.
Defined as in Figure 2 .
From data presented in Figure 3 .
From data in Figure 4 .
From data in Figure 5 .
From data in
Testing for Effects on PKC and Activation of ERK MAP Kinases
We tested the substances whose effects we concluded were not likely due to effects on [Ca2+]i for inhibition of the activity of PKC and activation of the MAPK ERK by upstream MAPKKs ( Fig. 4A ). As a control, we treated cells with the compound Ro31-8220, which blocks PKC and thus PMA-stimulated activation of MAPKKs. 19 Ro31-8220 inhibited PKC substrate phosphorylation by 82% and ERK phosphorylation by 95%. Seven of the exocytosis-blocking compounds tested inhibited ERK phosphorylation by >50%, but none significantly reduced the fluorescence intensity of anti-phospho PKC substrate staining (see Table 1 ). We conclude that 7 compounds likely inhibit granule exocytosis by inhibiting MAPKK activation downstream of PKC.
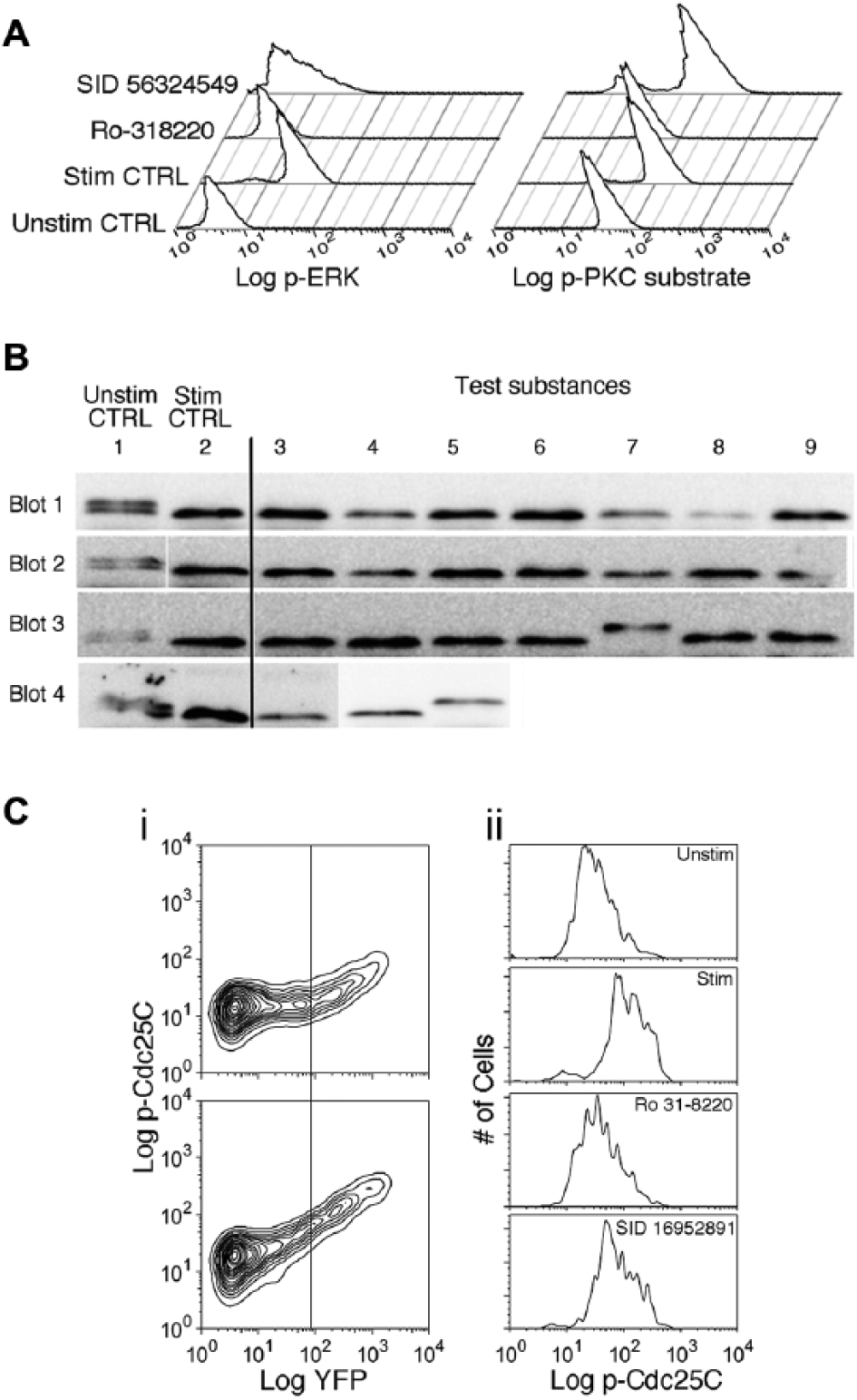
Testing the effects of compounds on protein kinase C (PKC) and mitogen-activated protein kinase (MAPKK) activity, nuclear factor of activated T cells (NFAT) dephosphorylation, and extracellular signal-regulated kinase (ERK) catalytic activity. (
Testing for Effects on Calcineurin Activity
We tested the effects of substances whose effects on exocytosis were not due to effects on Ca2+, PKC, or ERK for their ability to inhibit the activity of the Ca2+-dependent phosphatase calcineurin, which is required for granule exocytosis. To measure calcineurin activity, we used a genetically encoded reporter construct 20 based on the transcription factor NFAT, the best-known calcineurin substrate ( Fig. 4B ). Because NFAT is highly phosphorylated in resting cells, dephosphorylation resulting from calcineurin activity increases the reporter construct’s apparent mobility on SDS-PAGE (sodium dodecyl sulfate polyacrylamide gel electrophoresis) gels from ~125 kD to ~110 kD. Four sets of gels and transfers were required to run all of the samples; Figure 4B is a montage of the results (see legend for details of its assembly). In unstimulated cells (column 1), immunoreactivity was present as 2 bands. As expected, stimulation of cells caused a shift in the pattern of immunoreactivity to a single band of low molecular weight (column 2). Treatment of cells with the calcineurin inhibitor cyclosporin A prior to stimulation caused immunoreactivity to shift exclusively to the high-molecular-weight form (blot 4, column 5). One of the substances we tested, SID 92764285 (blot 3, column 7), completely blocked the shift of the sensor to lower molecular weight, resulting in a single band of high molecular weight, identical to the effect of cyclosporin A. Other substances were without apparent effect.
Testing Effects on ERK Catalytic Activity
Last, we tested whether compounds for which an MMOA had not yet been determined blocked the catalytic activity of ERK ( Fig. 4C ), because this possibility was not excluded by our experiments with the phospho-ERK antibody, which detects activation of ERK by upstream MAPKKs. We used cyto-EKAR, 21 a genetically encoded sensor of ERK activity that contains the phosphorylation site of Cdc25c and an ERK docking motif ( Fig. 4C ). We used an anti-phospho Cdc25c antibody to detect activity. In unstimulated cells, increasing expression of the sensor (as detected by YFP fluorescence) was accompanied by higher levels of anti-pCdc25c staining. Stimulating cells with TG+PMA increased anti-pCdc25c staining specifically in the YFP-positive cells ( Fig. 4Ci ). This increase in anti phospho-Cdc25c staining was inhibited when cells were treated with the PKC inhibitor Ro31-8220 ( Fig. 4Cii ), a result consistent with the involvement of PKC activation in activating ERK. We found that none of the compounds inhibited the increase in anti-pCdc25c staining by >30%, whereas Ro31-8220 blocked by 83%. This suggests that no compound is likely to block lytic granule exocytosis solely by inhibiting ERK catalytic activity.
Identification of a Compound with Unknown MMOA
Sixteen compounds did not inhibit a single pathway tested by >50%. Eight of those, however, exerted effects on multiple pathways that, although separately of insufficient magnitude to account for the block of granule exocytosis, could be imagined to exert summed effects large enough to inhibit release. The compounds with significant multiple inhibitory effects are detailed in Table 1 . Because we know relatively little about how varying the strength of the different signals affects exocytosis, it is difficult to determine whether the effects of these compounds on known MMOAs completely account for their ability to block exocytosis or whether they have additional effects. Our choice to use a relatively high concentration of compounds for MMOA testing may play a role in generating cases of apparent multiple activities. The final 8 compounds had, however, summed inhibitory effects that seemed unlikely to be of sufficient magnitude to account for blocking. We were able to obtain powder resupply of 1 of them, SID 7977862. On retest, this compound blocked exocytosis with an EC50 of ~4 µM, had no effect on cell viability in the short term when tested at 100 µM, and was without inhibitory effect on calcium signals, ERK activation, ERK catalytic activity, or calcineurin activation ( Fig. 5 ).
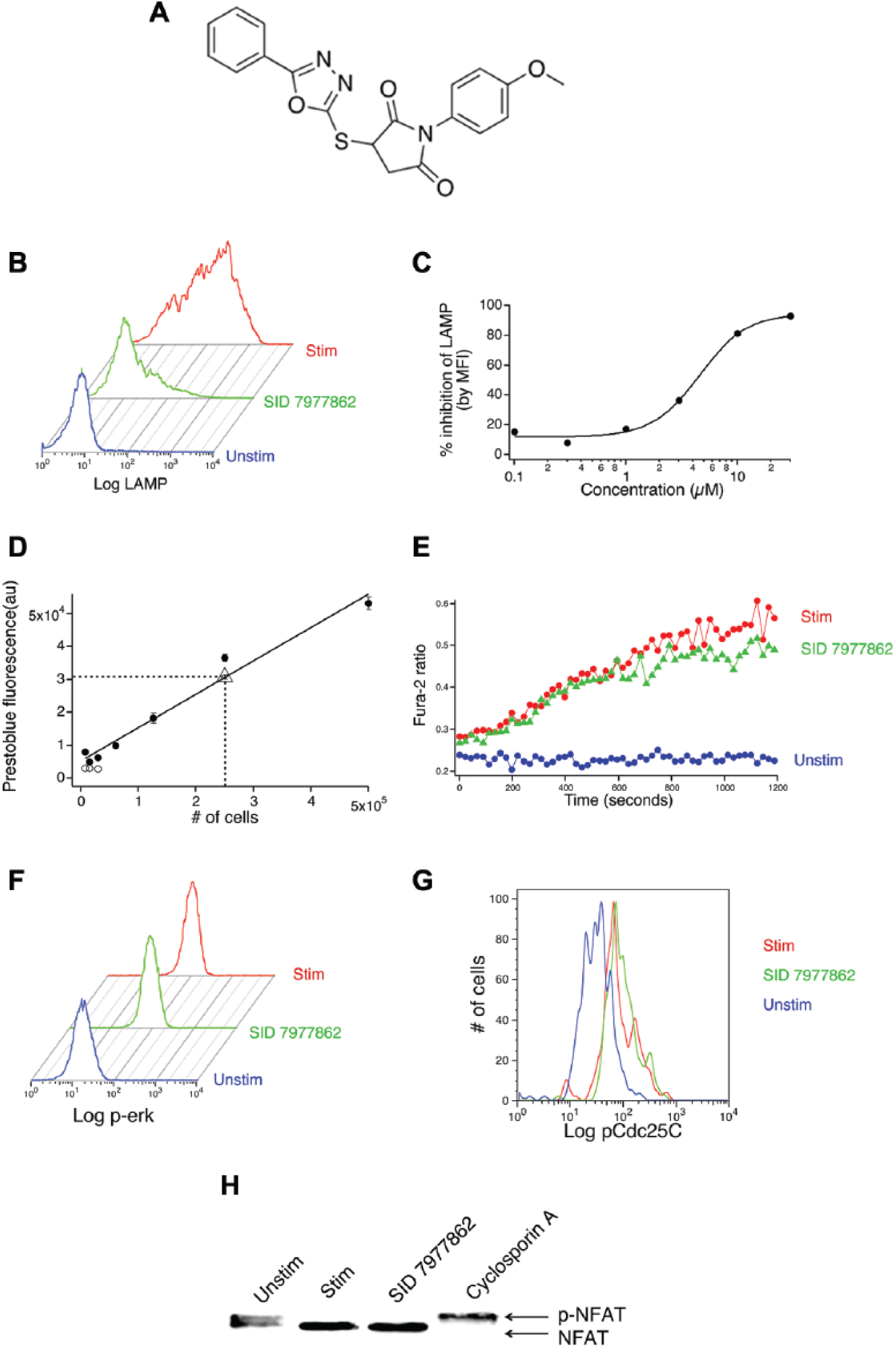
Confirming the effects of substance ID (SID) 7977862 from powder. (
Discussion
Our initial HTS campaign worked essentially as anticipated. Using clustered cytometers, we were able to miniaturize the assay to a 1536-well format, resulting in a substantial decrease in the amount of reagents—primarily, cells and antibody—required. Overall, we had 161 confirmed hits, a number that seemed low to us considering the complexity of the cellular phenotype we were interrogating. This hit rate, however, appears consistent with results from the most directly comparable screening campaign conducted on the MLSMR that we were able to identify. That screen looked for inhibitors of thrombin-stimulated platelet dense granule release using luciferase to detect adenosine triphosphate release accompanying granule exocytosis. Primary screening [assay ID (AID) 1663] of 302,517 compounds identified 661 that were active. Subsequent confirmation (AID 1189) and counterscreening for compounds that inhibit luciferase activity (AID 1891) yielded ~250 active compounds that inhibited platelet granule release. This effort resulted in the discovery of a probe that targeted protease-activated receptor 1. 22 Surprisingly, given what we would anticipate would be many similarities between exocytosis in CTLs and platelets, including the involvement of signaling pathways like PKC and calcium influx, as well as the involvement of SNARE (soluble N-ethylmaleimide-sensitive factor activating protein receptor) proteins (see Li et al. 23 for a recent review of signaling in platelets, and Koseoglu and Flaumenhaft 24 for a discussion of the exocytic machinery), there was only 1 compound active in that screen that we also found to be active. SID 17414945, which we found blocked lytic granule exocytosis and MAPKK activation, inhibited lytic granule exocytosis with an EC50 of ~0.3 µM, but it apparently inhibited platelet granule release much less potently, with an EC50 of 19 µM. We cannot account for these results. It seems unlikely to result from our use of a cell line, because TALL-104 cells recapitulate key features of CTL function. A second potentially comparable phenotypic screening campaign used a luciferase reporter construct to look for inhibitors of NF-κB (nuclear factor kappa-light-chain-enhancer of activated B cells) activation in Jurkat cells stimulated with PMA and ionomycin. That effort identified 735 confirmed active compounds (initial screen in AID 435003, and confirmation in AID 435020). Although the hit rate in that screening campaign was ~5 times higher than ours, the phenotype interrogated includes the entire transcriptional and translational apparatus required to generate the reporter, together with the proteins required for ubiquitination, 25 which may account for the larger number of hits.
One of our hypotheses was that a phenotypic cellular endpoint assay followed by appropriately designed mechanism testing could be used as a high-content screen for important targets. Consistent with this idea, our experiments revealed a number of substances that act on important pathways, including 15 inhibitors of calcium influx, 7 MAPKK inhibitors, and 1 calcineurin inhibitor. Were any of these activities defined in previous screens of the MLSMR? Determining this is, unfortunately, not straightforward and was not facilitated by the structure and query capabilities of the PubChem web interface. To the best of our knowledge, no screen has looked for inhibitors of calcium influx in nonexcitable cells. We were, however, able to identify 3 screens of >100,000 MLSMR compounds conducted on calcium-permeable channels and 5 conducted on potassium channels. Blockers of these channels could be envisioned to block calcium or potassium channels in CTLs, although this is by no means certain. None of the substances we defined as inhibiting calcium signals were reported to block calcium-permeable CAV3 (confirmation in AID 489005), TRPC4 (confirmation in AID 2247), or TRPC6 (confirmation in AID 488961) channels. One substance that blocked Fura-2 signals, SID 49721823, was reported to inhibit KCNK9 potassium channels (confirmation in AID 492992). KCNK9 (also known as TASK-3) channels have been found in lymphocytes, and inhibitors reduce proliferation and cytokine production. 26 SID 49677461 blocked lytic granule exocytosis and KIR2.1 potassium channels (confirmation in AID 2032), but it gave inconclusive results in our Fura assays. Similarly, SID 4964129 blocked lytic granule exocytosis and KCNK3 potassium channels (confirmation in AID 651638), but it also gave inconclusive results in our Fura assays.
There has been a screen of MLSMR compounds on ERK activation by MAPKKs. That effort (AID 1454) used AlphaScreen technology to assess levels of phosphorylated ERK in lysates of cells treated with epidermal growth factor. Of the 8 MAPKK inhibitors we found, only 2 appear to have been tested in that screen, which tested an earlier version of the MLSMR containing fewer compounds. Of the 2 substances both active in our assay and tested in theirs, they found SID 24810923 to be inactive, and SID 26727153 gave inconclusive results. We were unable to identify any screens of the MLSMR that might have been expected to reveal calmodulin or calcineurin inhibitors. The substance that we found to inhibit NFAT dephosphorylation [SID 92764285, 2-chloro-3-(3,5-dimethylmorpholin-4-yl) naphthalene-1,4-dione] was, as expected based on its chemistry, fairly broadly active in deposited assays, but it had no confirmed effects that suggested to us possible effects on either calmodulin or calcineurin.
In addition to revealing important actions of compounds on known targets, we suspected that a screen of lytic granule exocytosis could reveal substances that might work via an unknown MMOA. This appears to be the case because we identified SID 7977862, which blocks lytic granule exocytosis without inhibiting any of the pathways we tested. Inspecting the reported effects of this compound does not lead to well-supported hypotheses about its MMOA. An unconfirmed high-content screen (AID 1381) found that it inhibited dynein motor protein activity, but whereas dynein has been reported to be involved in reorientation of lytic granules to the site of contact with target cells, 27 there is no evidence for dynein involvement in TCR-independent exocytosis. Furthermore, >1000 other compounds were active in AID 1381, but none of them inhibited lytic granule exocytosis in our screen. If dynein were a common target in both assays, we would have expected more extensive overlap. Examination of the confirmed activities reported in the PubChem database for each of the 7 other substances for which we were unable to determine a MMOA also failed to suggest plausible mechanistic hypotheses for any of their actions. In addition to the MMOAs we were able to test, other possible targets include SNARE proteins and associated components of the exocytic machinery (see Schwarz et al. 28 for a recent review). Note that in preliminary experiments, we tested the effects of DMSO stocks of all 8 substances with undefined MMOA (including SID 7977862) on CD3-stimulated interleukin-2 secretion by Jurkat human leukemic T lymphocytes, and we found that all inhibited, suggesting that each is likely to have broad immunosuppressive activity rather than being a specific inhibitor of lytic granule exocytosis (J. Doucette and A. Zweifach, unpublished observations).
Our assay was configured specifically to detect inhibitors. One way we achieved that was the use of very strong artificial stimulation with TG and PMA. We therefore did not—and did not expect to—detect enhancers of exocytosis. A recent study with human NK cells screened the Prestwick Compound Library (PCL) for agents modulating FCGRIII-stimulated lytic granule exocytosis, integrin activation, and protein synthesis. 29 This study found 56 inhibitors and 12 enhancers. Our screen of the PCL2 identified several of the inhibitors they found, including amoxapine, desloratidine, thimerosol, and zotepine. Differences in assay conditions—particularly protein concentration in the buffer used—likely account for incomplete overlap of inhibitors.
Despite an emphasis in recent years on target-based approaches, a recent analysis demonstrates that a majority of first-in-class new drugs released between 1999 and 2008 were discovered by phenotypic screens. 30 Our screen of lytic granule exocytosis identified a number of previously undescribed actions of compounds on known targets that could serve as leads for immunosuppressants, and it also identified several compounds that may work via an unknown MMOA. We determined that at least 1 of these has probe-like properties. This confirms that our phenotypic screen based on lytic granule exocytosis may be a good means of identifying immunosuppressive compounds. Furthermore, we suggest that our basic strategy of screening a complex cellular phenotype that incorporates multiple potential targets and then defining MMOAs for active compounds may be a useful means of identifying interesting biologically active small molecules in compound collections.
Footnotes
Acknowledgements
We would like to thank Amy E. Florian for conjugating the anti-LAMP antibody and for helping grow and ship cells from the University of Connecticut to the University of New Mexico. Christopher K. Lepensky also helped grow and ship cells.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: L. A. Sklar and B. S. Edwards are founders of Intellicyte, Inc., a company that markets a commercial version of the high-throughput sampling cytometer. The contributors, either individually or collectively, have commercial or financial involvements that might present an appearance of a conflict of interest related to the contribution.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by R21 NS066462 to AZ and U54 MH084690 to LAS.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.