Abstract
Niemann–Pick disease type C (NPC) is a rare neurodegenerative disorder caused by recessive mutations in the NPC1 or NPC2 gene that result in lysosomal accumulation of unesterified cholesterol in patient cells. Patient fibroblasts have been used for evaluation of compound efficacy, although neuronal degeneration is the hallmark of NPC disease. Here, we report the application of human NPC1 neural stem cells as a cell-based disease model to evaluate nine compounds that have been reported to be efficacious in the NPC1 fibroblasts and mouse models. These cells are differentiated from NPC1 induced pluripotent stem cells and exhibit a phenotype of lysosomal cholesterol accumulation. Treatment of these cells with hydroxypropyl-β-cyclodextrin, methyl-β-cyclodextrin, and δ-tocopherol significantly ameliorated the lysosomal cholesterol accumulation. Combined treatment with cyclodextrin and δ-tocopherol shows an additive or synergistic effect that otherwise requires 10-fold higher concentration of cyclodextrin alone. In addition, we found that hydroxypropyl-β-cyclodextrin is much more potent and efficacious in the NPC1 neural stem cells compared to the NPC1 fibroblasts. Miglustat, suberoylanilide hydroxamic acid, curcumin, lovastatin, pravastatin, and rapamycin did not, however, have significant effects in these cells. The results demonstrate that patient-derived NPC1 neural stem cells can be used as a model system for evaluation of drug efficacy and study of disease pathogenesis.
Keywords
Introduction
Niemann–Pick disease type C (NPC) is a rare autosomal recessive genetic disease caused by mutations in the NPC1 or NPC2 gene. Deficiency in NPC1 or NPC2 protein results in malfunction of intracellular cholesterol trafficking and lysosomal accumulation of unesterified cholesterols. 1 Clinical manifestations of NPC often include enlargement of the spleen (splenomegaly) and liver (hepatomegaly), but the progressive neurodegeneration is a hallmark of the disease that causes disability and death of NPC patients.
A number of agents have been reported to have therapeutic potential for treatment of NPC. Cyclic oligosaccharides, including hydroxypropyl-β-cyclodextrin (HPBCD) and methyl-β-cyclodextrin (MBCD), are known to reduce brain cholesterol accumulation and increase life span in NPC1 mouse models.2–4 The effect of both compounds on the reduction of lysosomal cholesterol accumulation has been confirmed in the NPC patient-derived fibroblasts2,5 and primary mouse neurons. 6 The benefits of other compounds, including miglustat, 7 curcumin, 8 SAHA, 9 statins, 10 and rapamycin, 11 on some NPC models have also been reported. Miglustat, a substrate reduction drug originally developed for treatment of Gaucher’s disease, has been approved in the European Union for the treatment of NPC disease. HPBCD is currently in clinical trials for NPC treatment. 12 We recently reported that δ-tocopherol significantly reduces lysosomal accumulation of cholesterol and other macromolecules in patient fibroblasts with NPC and other lysosomal storage diseases. 13 The effects of these agents, however, have not been directly evaluated in human NPC neuronal cells, the type of cells more relevant to the disease pathogenesis.
Recent advances in stem cell technology have enabled the generation of disease-specific induced pluripotent stem cells (iPSCs) from patient cells. 14 These iPSCs are able to differentiate into expandable progenitor cells and mature cells, including neurons, cardiomyocytes, and hepatocytes, allowing the establishment of cell-based disease models. Due to their availability in large quantities and their similarities in disease phenotype compared to differentiated mature neurons, neural stem cells (NSCs) and related cells have been used as a cell-based model system for high-throughput screening to evaluate drug efficacy and discover lead compounds.15–19 We recently established a phenotypic screening assay to quantitate the changes of cholesterol levels in normal iPSC-derived neuronal cells 20 and determine the effects of compounds on enlarged lysosomes, a common feature in lysosomal storage diseases. 21 We report here the generation of NPC1 iPSCs from patient dermal fibroblasts and the differentiation of NPC1 iPSCs to NSCs and subsequently neurons for evaluation of drug efficacy.
Materials and Methods
iPSC Generation
Wild-type (WT) fibroblasts (GM05659, Coriell Cell Repository) and NPC1 patient fibroblasts (GM03123, Coriell Cell Repository) were cultured in Dulbecco’s Modified Eagle’s Medium (DMEM) with 10% fetal bovine serum (FBS)/nonessential amino acids (NEAA)/glutamax. The cells were reprogrammed using the nonintegrating CytoTuneTM–Sendai viral vector kit (Life Technologies). 22 Briefly, cells were plated in a six-well plate (5×104/well) for one day and were transduced with the following four transcription factors: Oct4, Sox2, Klf4, and cMyc (multiplicity of infection = 3 for each of the factors). The cells were cultured for another 5 days in fibroblast medium supplemented with 10 µM δ-tocopherol (to reduce the lysosomal cholesterol accumulation) 13 and then passaged onto mouse embryonic fibroblast (MEF) feeder cells (GlobalStem) in stem cell culture medium [knockout DMEM/F12 with 20% knockout serum replacement, 1× NEAA, 1× glutamax, 0.1 mM β-mercaptoethanol, and 8 ng/ml basic fibroblast growth factor (Millipore)] and 10 μM δ-tocopherol. The resulting colonies were expanded on MEF feeder cells and passaged with Dispase. The cells were later adapted to Geltrex-coated plate with Essential 8 medium. The mutation-containing region was PCR amplified from extracted genomic DNA and sequenced.
Immunofluorescence Staining, Karyotyping, and Teratoma Formation
iPSCs were fixed with 4% paraformaldehyde for 15 min at room temperature. The cells were then rinsed with Dulbecco’s Phosphate-Buffered Saline, blocked in blocking buffer (1% BSA and 0.1% Triton X-100 in DPBS) for 30 min and incubated with anti-Oct4 (2840S, Cell Signaling; 1:500 dilution), anti-Sox2 (2748S, Cell Signaling; 1:500), anti-SSEA4 (MAB4304, Millipore; 1:250), or anti-TRA-1-81 (MAB4381, Millipore; 1:250) in blocking buffer at 4 °C overnight. The cells were then incubated with Alexa Fluor 594 conjugated antirabbit immunoglobulin G (IgG) or antimouse IgM (1:1000) in blocking buffer for 1 h at room temperature followed by 4′,6-diamidino-2-phenylindole (DAPI) staining and imaging. Karyotyping was conducted at the Molecular Cytogenetic Core Facility at the Frederick National Laboratory for Cancer Research, and 20 randomly selected metaphases from NPC1 iPSC clones were fully analyzed. For teratoma study, 1×106 cells were resuspended in 200 μl phosphate-buffered saline (PBS) supplemented with 30% Matrigel. The cells were then injected into immunocompromised NSG mice intramuscularly or subcutaneously. Five to eight weeks after injection, the animals were euthanized, and tumors were dissected and fixed with 4% paraformaldehyde, embedded into paraffin, and sectioned for hematoxylin and eosin staining. The animal protocol was approved by the Institutional Animal Care and Use Committee at the University of Maryland School of Medicine
Neural Differentiation from iPSCs
NPC1 iPSCs were differentiated into NSCs by using a kit from Life Technologies. Briefly, iPSCs were digested with Dispase and reseeded onto Geltrex-coated plate at 20% confluence. After cells attached, the medium was changed to Induction Medium containing the Neurobasal Medium plus Neural Induction Supplement (A15640SA, Life Technologies). At day 7 of neural induction, the initial NSCs were dissociated with Accutase and plated for further expansion in Neural Expansion Medium containing Neurobasal Medium and Advanced DMEM/F12 with 1× Neural Induction Supplement. NSCs were characterized by staining with antibodies against nestin and Sox2 that showed 99% positive cells with both neural markers.
For neuronal differentiation, dissociated NSCs were cultured on poly-L-ornithine- and laminin-coated 96-well plates in the Induction Medium with 5 μM Y27632, a Rho-associated protein kinase inhibitor (Y0503, Sigma-Aldrich), for one day. The medium was changed to a differentiation medium containing Neurobasal Medium, 1× B27 (17504-044, Life Technologies), 1× glutamax (35050, Life Technologies), 200 µM L-ascorbic acid (A8960, Sigma-Aldrich), 1 μM cyclic adenosine monophosphate (A6885, Sigma-Aldrich), 10 ng/ml brain-derived neurotrophic factor (10908-010, Life Technologies), and 10 ng/ml glial cell–derived neurotrophic factor (PHC7044, Life Technologies) that was changed every 2 days for 12 days. 20
Immunofluorescence Staining of Neuronal Cells
Cells were fixed in 4% paraformaldehyde for 20 min, rinsed with PBS, and permeabilized with 0.3% Triton X-100 for 15 min, followed by incubation with primary antibody overnight at 4 °C. After washing with PBS, a goat antirabbit secondary antibody conjugated with DyLight 594 (3066-1, Epitomics; 1:200 dilution) was added. Cells were then stained with Hoechst 33342 (H1399, Life Technologies) for 20 min after a wash and imaged using INCell Analyzer 2000 (GE Healthcare) with a 40× objective lens and Texas Red and DAPI filter sets.
Filipin Staining
Filipin dye stains unesterified cholesterol in cells. 13 Cells were cultured at 2000 cells/well in black, clear-bottom, 96-well plates. After washing twice with PBS, cells were fixed with 100 µl/well of 3.2% paraformaldehyde solution for 30 min followed by a cell wash. The cells were stained with 100 µl/well of 50 ng/ml filipin (freshly dissolved in DMSO at 10 mg/ml and then diluted in PBS) at room temperature for 1 h. After washing with PBS, cell nuclei were stained with DRAQ5 (4084, Cell Signaling; 1:1000 dilution in PBS) for 30 min at room temperature. The cells were washed and imaged in INCell Analyzer 2000 with a 20× or 40× objective lens using DAPI and Cy5 filter sets.
Image data analysis was conducted using INCell Analyzer software. With a multitarget analysis protocol, nuclei were segmented using the top-hat segmentation method with a minimum area set at 125 µm and a sensitivity set at 60. Filipin staining was identified as “Organelles” within the analysis software and was segmented using the multiscale top-hat algorithm. Settings for filipin detection involved identification of filipin aggregates ranging in size from 10 to 125 µm (30 to 300 pixels) and a sensitivity setting of 50. Total filipin intensity was calculated from cells exceeding the user-defined threshold for whole-cell intensity.
Amplex Red Cholesterol Assay
An Amplex Red Cholesterol Assay Kit (A12216, Life Technologies) was used to measure the free cholesterol in patient cells. 13 Cells were cultured in black, 96-well plates at a density of 2500 cells/well for 24 h. The medium was removed and replaced with 100 µl/well compound in medium for a 3-day incubation. Cells were then washed with PBS and added with 100 µl/well of reagent mixture from the assay kit. The fluorescence intensity after 1 h incubation at 37 °C was measured in a ViewLux plate reader (Ex = 560±10 nm; Em = 590±10 nm).
Adenosine Triphosphate (ATP) Content Assay
An ATP content assay kit (6016736, PerkinElmer) was used to measure cell viability. 13 The reagent mix from the assay kit was added at a 1:1 ratio to cells for a 10-min incubation followed by luminescence detection in a ViewLux plate reader.
LysoTracker Red Staining
LysoTracker dye stains cellular acidic compartments and visualizes enlarged lysosomes at the proper dye concentration in patient cells. 21 Briefly, 2000 cells/well were seeded in black, clear-bottom, 96-well plates. After washing with PBS, cells were incubated with 100 µl/well 50 nM LysoTracker Red dye (L-7528, Life Technologies) at 37 °C for 1 h. Cells were then fixed in 100 µl/well 3.2% paraformaldehyde solution containing 1 µg/ml Hoechst 33342 (H1399, Life Technologies) in PBS and incubated at room temperature for 30 min, followed by washing and imaging in INCell Analyzer 2000 using DAPI and DsRed filter sets.
Images were analyzed with the multitarget analysis protocol. Nuclei were segmented using the top-hat segmentation method with a minimum area set at 80 µm and a sensitivity set at 50. Stained lysosomes were identified as “Organelles” and were segmented using the multiscale top-hat algorithm. Settings for lysosome detection included identification of granules ranging in size from 0.5 to 45 µm (3 to 13 pixels) and a sensitivity setting of 60. Total organelle intensity was calculated by a user-defined threshold for organelle intensity.
Electron Microscopy
NPC1 NSCs at 150,000 cells/well were seeded in six-well plates with 5 ml medium containing 10% FBS and cultured for 3 days. Cells were fixed in 2% glutaraldehyde, 0.1 M cacodylate buffer, pH 7.2 for 1 h at room temperature, and then they were stored in PBS at 4 °C until transmission electron microscopy analysis was performed. 13 The thin sections were double-stained (uranyl acetate and lead citrate) and examined in a Hitachi H-7650 transmission electron microscope, and images were taken using an advanced-microscopy-technique charge-coupled device camera.
Data Analysis
Concentration–response curves were analyzed and IC50 values calculated using Prism software (GraphPad). The bottom value in the four-parameter fit was fixed at 50% because the concentration–response curves of compounds showed an incomplete inhibition at the bottom plateau due to the limited high concentrations used. Results in the figures are expressed as mean of triplicates ± standard deviation.
Results
Generation of NPC1 iPSCs
Fibroblasts from an NPC1 patient (I1061T/P237S) were reprogrammed to iPSCs by using a nonintegrating Sendai virus reprogramming system that expresses Oct3/4, Sox2, Klf4, and cMyc factors. Immunofluorescence staining showed positive pluripotency markers, including Oct4, Sox2, SSEA4, and TRA-1-81, in the resulting NPC1 iPSC line, similar to WT control cells ( Fig. 1A ). Further characterization of the patient iPSCs showed a normal karyotype ( Fig. 1B ), typical teratoma formation ( Fig. 1C ), and original NPC1 gene mutations ( Fig. 1D ). The results presented here demonstrate the establishment of NPC1 iPSCs from patient-derived dermal fibroblasts.
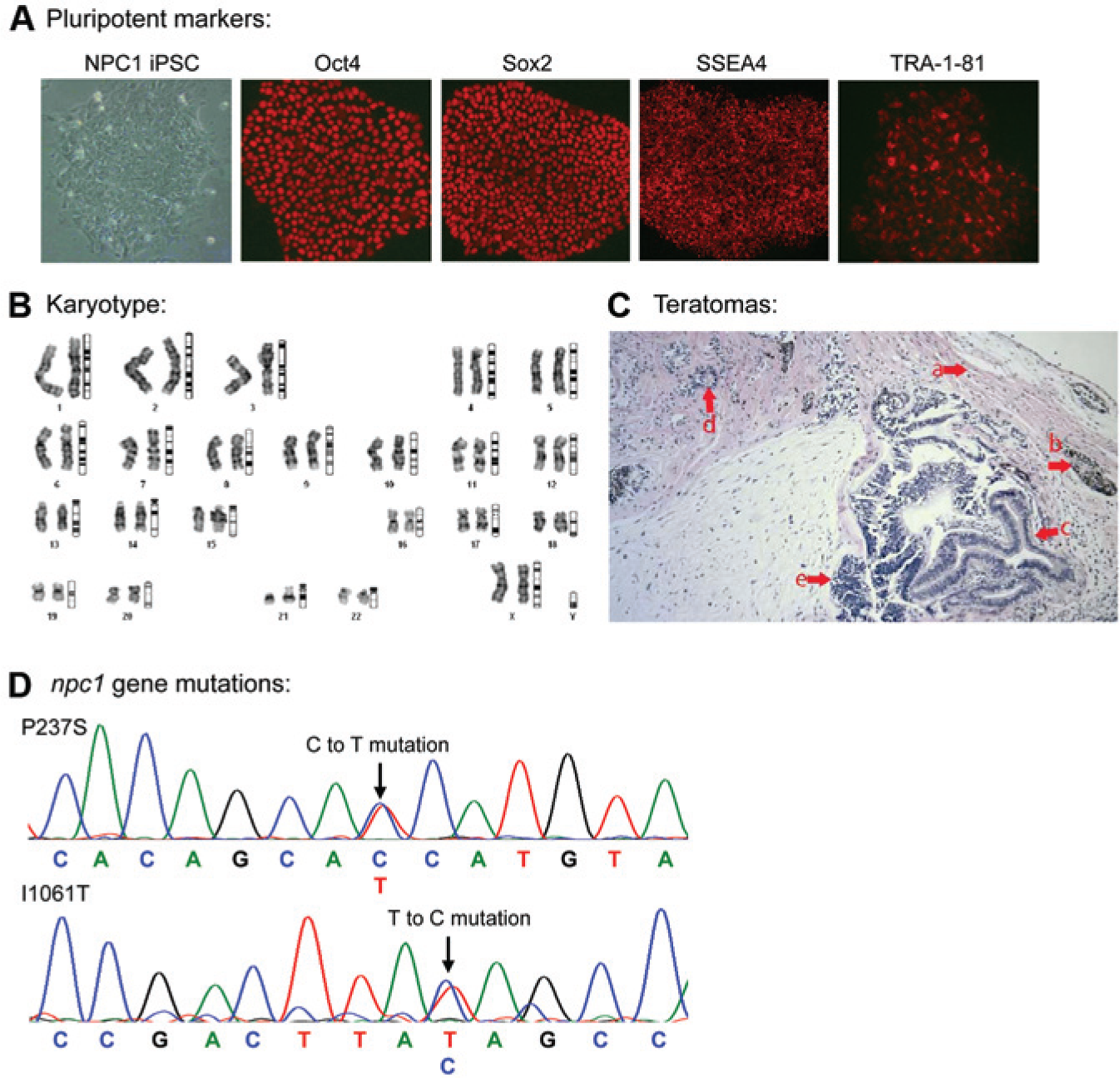
Generation of Niemann–Pick disease type C1 (NPC1) induced pluripotent stem cells (iPSCs). (
NPC1 iPSCs Differentiated to NSCs and Neurons
The NPC1 iPSCs were first differentiated to NSCs with Neural Induction Medium. We found the time required for NSC induction was similar to that of control cells, and the normal NSC markers, nestin, and Sox2, were present in NSCs ( Fig. 2A ).
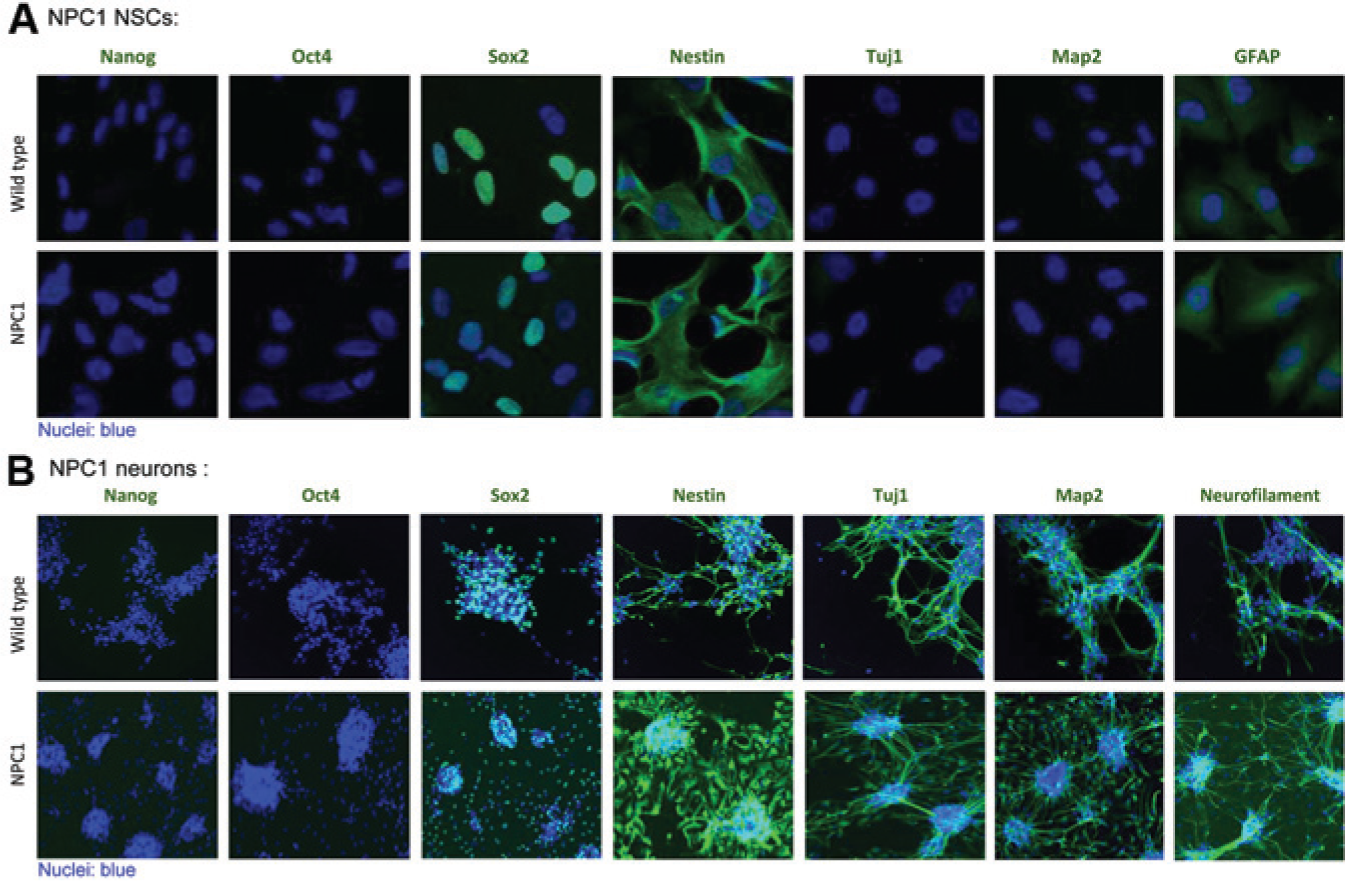
(
The NPC1 neurons were then differentiated from the NSCs for 14 days 20 when cells exhibited typical neuronal cell morphology with a dense network of neurites and positive neural markers, including β-tubulin III, microtubule-associated protein 2 (Map2), and neurofilament-1 ( Fig. 2B ). The time required for NPC1 neuron maturation, cell morphology, and expression of neural markers was similar to that of control cells. Together, the results indicate that the NPC1 iPSCs can be differentiated to NSCs and then to neurons with apparent normal morphology and neural markers.
Cholesterol Accumulation Phenotype Present in NPC1 iPSC Differentiated Neuronal Cells
Because lysosomal accumulation of unesterified cholesterol is a major phenotype of the disease, we first examined the filipin cholesterol staining in NSCs differentiated from the NPC1 iPSCs. We found slightly increased filipin cholesterol staining in NPC1 NSCs cultured in serum-free medium compared to the control cells ( Fig. 3A ). This lower cholesterol accumulation might be due to the lack of cholesterol in the culture medium for NSCs. The addition of 10% serum that contains cholesterol to the culture medium further increased cholesterol accumulation in lysosomes of NPC1 NSCs compared to the WT control cells ( Fig. 3A ). Increase in the LysoTracker dye staining was also observed in the NPC1 NSCs, which is indicative of enlarged lysosomes due to cholesterol accumulation ( Fig. 3A ). Thus, the results demonstrated the presence of the cholesterol accumulation phenotype in these NPC1 NSCs.
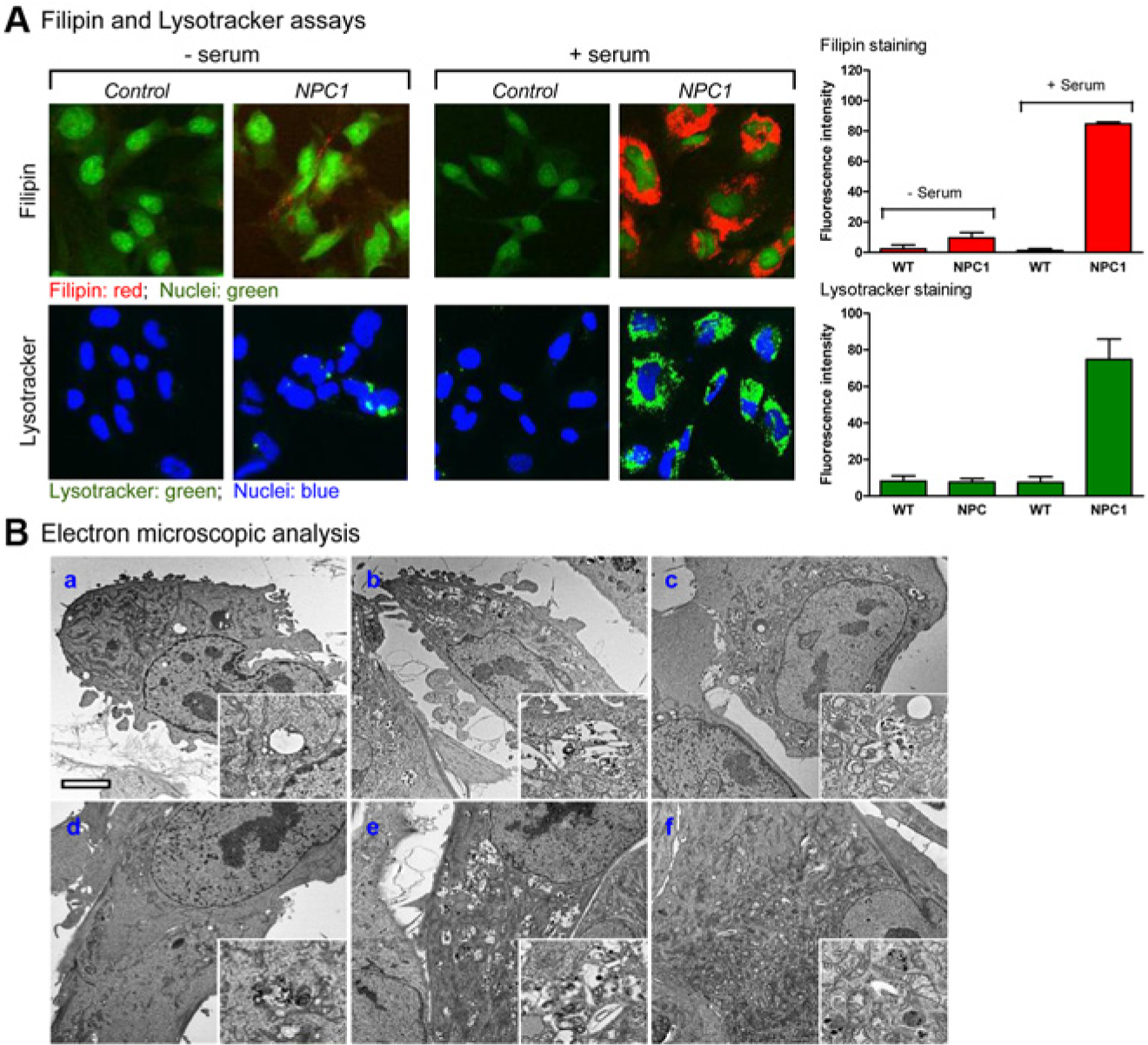
Lysosomal cholesterol accumulation in differentiated Niemann–Pick disease type C1 (NPC1) neuronal cells derived from the NPC1 induced pluripotent stem cell (iPSC) line. (
Thin-sectioning electron microscopy analysis shows the clear hallmark of NPC in the NPC1 NSC.
21
We further looked at the ultrastructural features in the newly developed NPC1 NSCs and observed a significant amount of lysosomes with lamellated, osmophilic structures in a clear matrix (
Fig. 3B
), but not in healthy WT control cells. Similar amounts of these structures were observed in NPC1 NSC treated with 20 µM δ-tocopherol or 300 µM MBCD, albeit they appeared to be slightly reduced in the latter treatment. In NPC1 NSC cells treated with 500 µM HPBCD or the combination of 50 µM HPBCD and 10 µM δ-tocopherol, the amount of these hallmark structures in the cytosol was essentially reduced to the WT level (
Fig. 3Bd
Amelioration of Cholesterol Accumulation in NPC1 Neuronal Cells by Cyclodextrins and δ-Tocopherol among Nine Agents Tested
We found that HPBCD, MBCD, and δ-tocopherol concentration-dependently reduced the cholesterol accumulation in NPC1 NSCs measured by a biochemical cholesterol assay with EC50 values of 26.3, 14.3, and 19.4 µM, respectively ( Fig. 4A ). The cholesterol reduction effect of these three compounds was also confirmed by filipin staining that specifically stains the unesterified cholesterol in NPC1 neurons ( Fig. 4B ). Similarly, these three compounds also reduced filipin staining and LysoTracker staining in NPC1 NSCs ( Fig. 4C ), which is indicative of reduction of cholesterol accumulation and enlarged lysosomes. Together, the results confirmed the effect of these three compounds on reduction of cholesterol accumulation in both NPC1 NSCs and neurons.
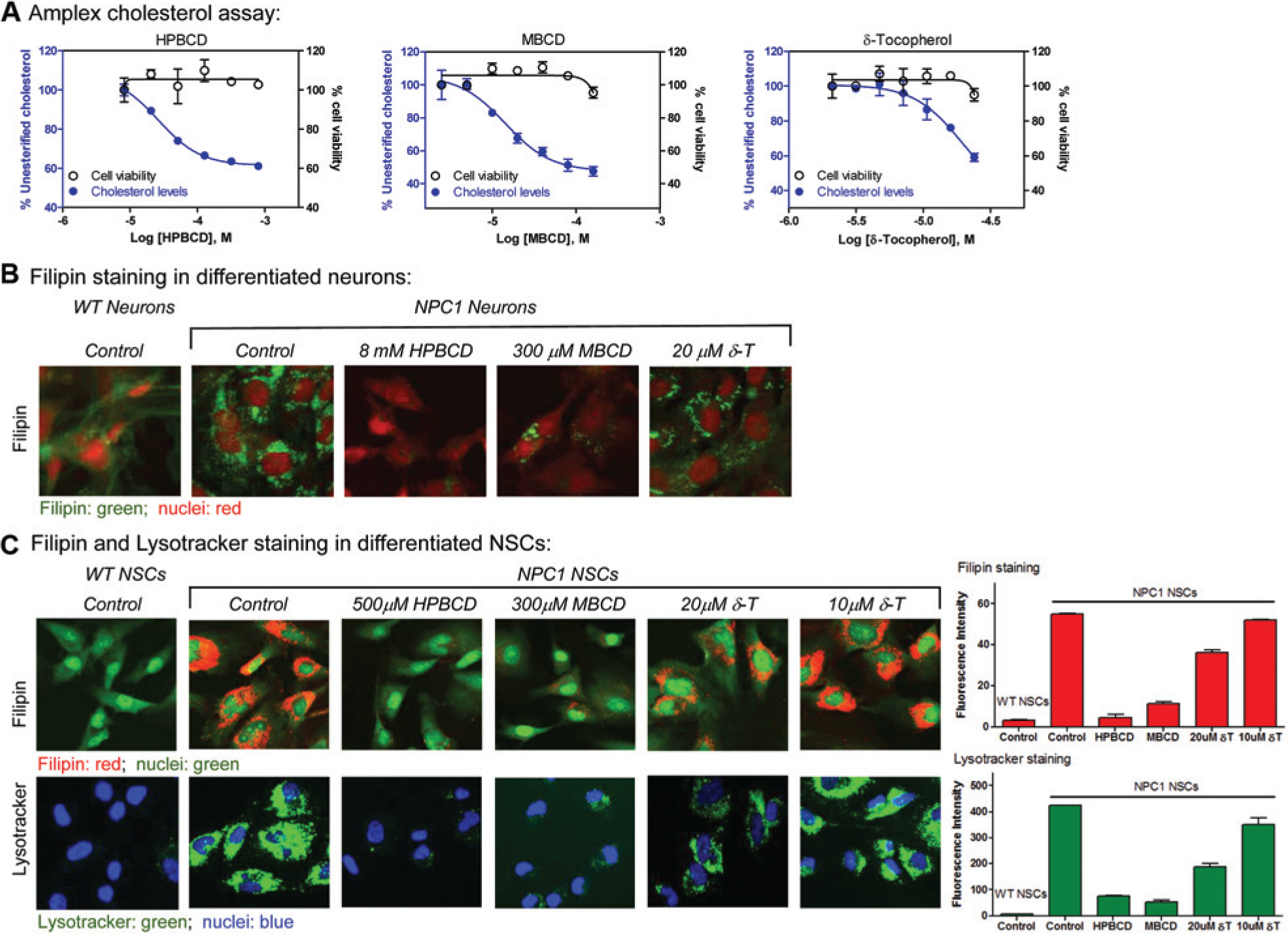
Effects of cyclodextrins and δ-tocopherol (δ-T) on reducing cholesterol accumulation and enlarged lysosomes in Niemann–Pick disease type C1 (NPC1) neural stem cells (NSCs). (
Compared to the compound potencies determined in NPC1 fibroblasts from the same patient (GM03123), HPBCD was more potent in the NPC1 neuronal cells compared to the NPC1 fibroblasts. MBCD showed similar activity in both NSCs and fibroblasts derived from the same NPC1 patient. Both cyclodextrins almost completely reversed cholesterol accumulation in NPC1 NSCs at higher concentrations. However, the maximal effect of δ-tocopherol on reduction of lysosomal cholesterol accumulation was weaker than that of cyclodextrins in human NPC1 NSCs, and that in NPC1 fibroblasts. 13 The results revealed a significant difference in drug responses to HPBCD and δ-tocopherol between NPC1 neuronal cells and dermal fibroblasts even though they were derived from the same NPC1 patient, whereas the potency of MBCD remained similar in both cell types.
We also examined the effects of miglustat, SAHA, curcumin, lovastatin, pravastatin, and rapamycin on reduction of lysosomal cholesterol accumulation in human NPC1 NSCs and found that they did not significantly reduce filipin staining in human NPC1 NSCs, indicating the lack of cholesterol-reducing effect in these cells ( Fig. 5 ). Thus, the results further demonstrated the cell type–dependent drug response in NPC cells. As a result, cyclodextrins and δ-tocopherol are the only agents that exhibited cholesterol-reducing effect in the human NPC1 NSCs among the nine compounds examined.
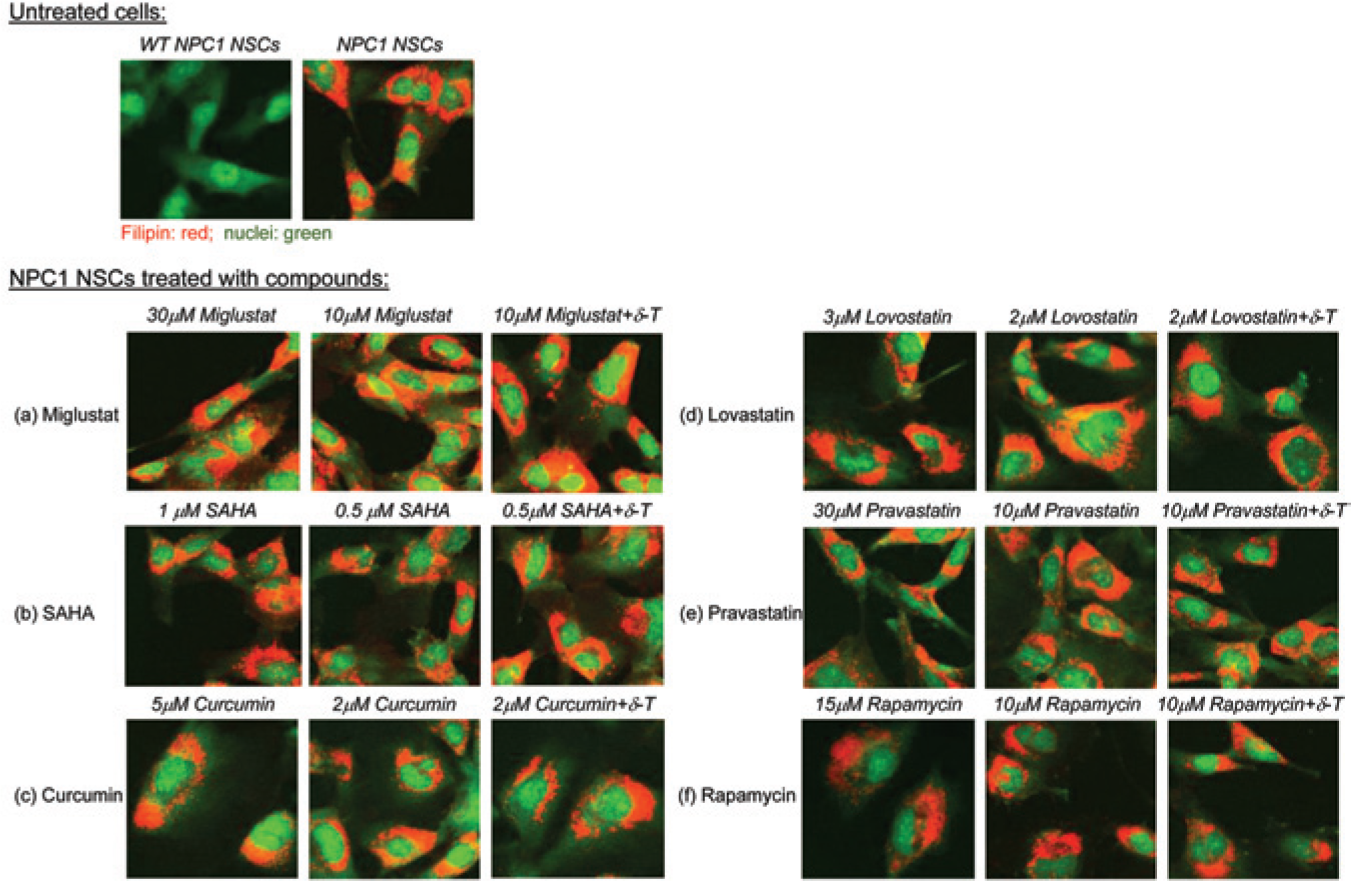
Effects of the other six drugs on the filipin staining in Niemann–Pick disease type C1 (NPC1) neural stem cells (NSCs). No significant effect on reduction of lysosomal cholesterol accumulation was observed after treatment with (
Synergistic Effects of Cyclodextrin and δ-Tocopherol on Reduction of Lysosomal Cholesterol Accumulation in NPC1 Neuronal Cells
A combination of two drugs with different mechanisms of action may result in the synergistic or additive effect that improves the therapeutic efficacy. Distinctive chemical structures of cyclodextrin and δ-tocopherol indicate a mechanistic difference in drug action. The therapeutic effect of cyclodextrins in NPC cells requires a high concentration 12 that is clinically difficult to achieve due to the limitation of drug pharmacokinetics and potential drug toxicity. Thus, we determined the potential synergistic effect of a cyclodextrin and δ-tocopherol combination in human NPC1 NSCs. We found that a combination of 50 µM HPBCD and 10 µM δ-tocopherol almost completely reduced cholesterol accumulation in the NPC1 NSCs, comparable to 500 µM HPBCD used alone ( Fig. 6A ). Similarly, 20 µM MBCD plus 10 µM δ-tocopherol achieved a similar cholesterol reduction effect in the NPC1 NSCs as 300 µM MBCD used alone ( Fig. 6B ). A parallel reduction of LysoTracker staining was also observed by the same combination treatment ( Fig. 6A and 6B ). Taken together, the results demonstrate a synergistic effect with combination treatment of HPBCD or MBCD with δ-tocopherol on reduction of lysosomal cholesterol accumulation in human NPC1 NSCs. Importantly, the concentration of cyclodextrins in combination was reduced by 10-fold for a similar cholesterol reduction effect as seen when used as a single agent.
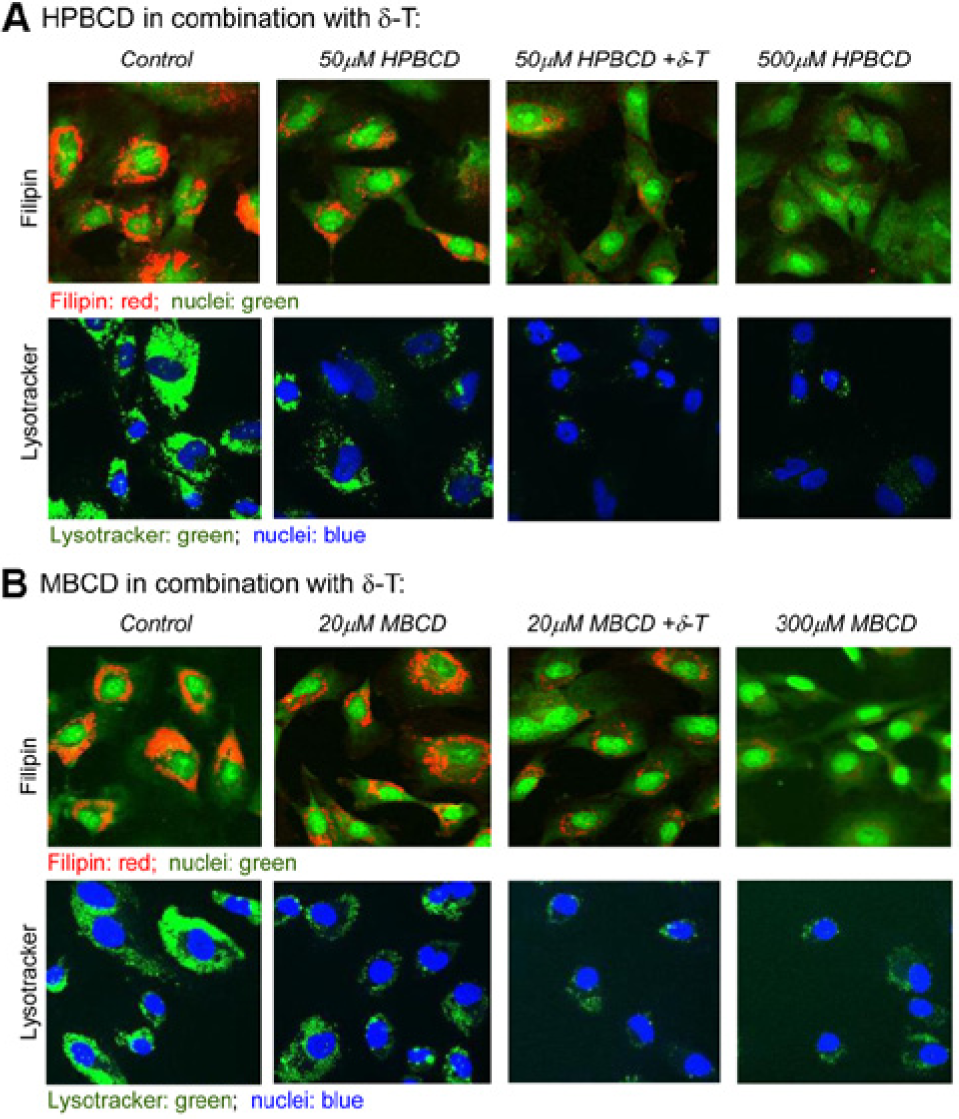
Combination treatment of cyclodextrin and δ-tocopherol (δ-T) in Niemann–Pick disease type C1 (NPC1) neural stem cells (NSCs). (
Discussion
In this study, we describe a successful generation of iPSCs from dermal fibroblasts derived from a NPC1 patient with I1061T/P237S mutations (GM03123, Coriell Cell Repository). Lysosomal cholesterol accumulation is present in the NPC1 neuronal cells differentiated from the NPC1 iPSCs, recapitulating the cellular hallmark of NPC disease. We demonstrate that cyclodextrins and δ-tocopherol are able to reduce lysosomal cholesterol accumulation in differentiated NPC1 neuronal cells, providing a human neuronal cell–based disease model for evaluation of drug efficacy and study of disease pathophysiology.
Recent advances in iPSC technology have enabled generation of cell-based disease models using human cells differentiated from patient iPSCs. 14 Since the generation of the first patient-derived iPSCs in 2009, 23 more than 40 human iPSC cell lines have been produced from patient cells with various diseases. 14 Generation of iPSCs from patient somatic cells is still a challenge despite the advancement of iPSC technologies. Altered health condition and/or disease phenotypic change in the patient cells as well as high passage numbers of patient fibroblasts may hamper iPSC formation.24–26 For example, mutations in ALK2 kinase caused repression of iPSC generation from patient dermal fibroblasts with fibrodysplasia ossificans progressiva, which was overcome by treatment with an ALK2 inhibitor. 27 To date, only four human iPSC lines were reported for 50 lysosomal storage diseases, including Gaucher,26,28 mucopolysaccharidosis (MPS) type 1, 29 MPS IIIB, 30 and Pompe 25 diseases, suggesting that disease phenotypes in the patient fibroblast cell lines may suppress the iPSC generation. Delayed appearance of iPSC colonies from fibroblasts with Gaucher disease was found. 26 The MPS IIIB iPSC-like colonies from the patient fibroblasts proliferated poorly due to deficiency in α-N-acetylglucosaminidase (NAGLU), which was overcome by expressing NAGLU in engineered iMEF that released the recombinant enzyme into culture medium. 30 Deficiency in acid-α-glucosidase also suppressed iPSC generation in Pompe fibroblasts, which was rescued by overexpression of WT GAA in the patient fibroblasts. 25
We initially found that only a few iPSC-like colonies appeared during the iPSC derivation process, and all of them disappeared after five to seven passages. The difficulty in NPC1 iPSC generation might be due to cholesterol accumulation phenotype in NPC1 fibroblasts, 13 as well as the higher passage number of commercially obtained fibroblasts. After the treatment of NPC1 fibroblasts with δ-tocopherol, a compound that reduces cholesterol accumulation in NPC1 fibroblasts, 13 we have successfully established NPC1 iPSCs that have been cultured for more than 20 passages. The neuronal cells differentiated from NPC1 iPSCs exhibited cholesterol accumulation phenotypes, and they also displayed normal neuronal cell morphology. During our study, another group reported the generation of NPC1 iPSCs from a patient fibroblast cell line (GM18436, 1628delC, and GLU612ASP) that have different mutations compared to the NPC1 cell line we used (GM03123, p.I1061T, and p.P237S), and a retroviral transfection method was used to transform the fibroblasts. 31 Cholesterol accumulation phenotype was also exhibited in the differentiated NPC1 neuronal cells, although the efficacy of known compounds has not been evaluated. 31 The result is consistent with our finding of lysosomal accumulation of unesterified cholesterol.
Although NPC1 dermal fibroblasts with cholesterol accumulation phenotype have been used in various experiments, skin damage does not occur in NPC patients. Because neuronal degeneration is responsible for the morbidity and mortality of NPC, the NPC neuronal cells are the disease-relevant model system that is more suitable for evaluation of compound efficacy. Recently, three human NPC1 neuronal models have been reported, including neurons directly differentiated from NPC patient dermal fibroblasts, npc1 silenced human embryonic stem cells, and SH-SY5Y cells.32–34 Unlike most NPC patient cells, which have single amino acid missense mutations, silencing npc1 gene may not recapitulate the real disease pathophysiology, and the experiment may have off-target effects as well. Although the isolated dermal stem cells possess the same mutations from NPC1 patients, low efficiency of neuronal differentiation and less maturity of differentiated neurons from this cell type limit their applications as a disease model system. Therefore, our NPC1 iPSCs and the differentiated neuronal cells are a desirable disease model system that has retained both disease genotype and phenotype. These cells are useful as a model system to study the disease pathophysiology and to evaluate compounds using a phenotypic screen for drug development. 35
NPC1 iPSC-derived NSCs were used for evaluating a total of nine agents that have been previously reported to have potential therapeutic effects on NPC disease. Cyclodextrins (HPBCD and MBCD) and δ-tocopherol are the only three agents that show positive effects on reduction of lysosomal cholesterol accumulation in human neuronal cells. Our results confirmed the efficacy of these three agents in human patient neuronal cells. We also found the additive or synergistic effect of combination treatment of cyclodextrin and δ-tocopherol in the differentiated NPC1 human neuronal cells. Importantly, the concentration of the cyclodextrins can be reduced by 10-fold when combined with δ-tocopherol. A high dose of HPBCD (2–2.5 g/kg body weight) is reportedly needed for the treatment of NPC patients. 12 This is partly due to the low potency of HPBCD, which was confirmed in the human NPC1 neuronal cells in this study. Thus, a 10-fold reduction in the HPBCD dose will be more applicable for its clinical use.
The cholesterol-reducing effect of miglustat, SAHA, curcumin, lovastatin, pravastatin, and rapamycin was not observed in the differentiated NPC1 human neuronal cells in this study. The mechanisms of action for these six agents may be different from those of cyclodextrin and δ-tocopherol, which enhance lysosomal exocytosis.2,13 For example, miglustat was initially approved as a substrate suppression therapy drug for Gaucher’s disease and the mechanism of action for NPC is still not clear. Lovastatin and pravastatin are cholesterol-lowering drugs that inhibit a critical enzyme in the cholesterol synthesis pathway. Our results indicated an existence of differential drug responses in different NPC cell types. Because our iPSC differentiated human neuronal cells are a disease-relevant cell type with the same NPC1 mutations in the NPC patients, they offer an orthogonal approach for evaluation of drug efficacy. Our results indicate that these neural stem cells can be produced in large quantities, and the assay results obtained from these cells are reproducible. Thus, these cells can be used for low or medium-throughput compound screens, such as those recently reported.17–19
In summary, we have generated an NPC NSC cell-based disease model from an NPC1 iPSC line that exhibits cholesterol accumulation phenotype. The therapeutic effects of three agents, HPBCD, MBCD, and δ-tocopherol, have been observed in the differentiated human NPC1 neuronal cells, whereas the effects of the other six agents—including miglustat, SAHA, curcumin, lovastatin, pravastatin, and rapamycin—were not seen. Our data demonstrated that these human NPC1 neuronal cells are a new disease model system for determination of drug efficacy as well as study of disease pathophysiology. Our results also implicated that the high HPBCD dose needed for treatment of NPC in the clinical trial 12 may be reduced significantly if it is combined with δ-tocopherol through direct application in the central nervous system.
Footnotes
Acknowledgements
We thank Sandra Burkett of Frederick National Laboratory for Cancer Research for iPSC karyotyping and Dr. Dan Ory of Washington University for providing HPBCD.
Abbreviations
HPBCD, hydroxypropyl-β-cyclodextrin; iPSCs, induced pluripotent stem cells; MBCD, methyl-β-cyclodextrin; NPC, Niemann–Pick disease type C; NSCs, neural stem cells
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by grants from Maryland Stem Cell Research Fund, Maryland Clinical Nutrition Research Unit (DK072488), the Baltimore Diabetes Research and Training Center (P60-DK-079637), and NIH Center for Regenerative Medicine. This work is also supported by the Intramural Research Programs of the National Center for Advancing Translational Sciences and federal funds from the National Cancer Institute, National Institutes of Health, under contract HHSN26120080001E.
