Abstract
In the ongoing search for new therapeutic compounds, lignans and neolignans, which are widely distributed in plants, deserve special attention because of their interactions with several biological targets. Searching for potential antiangiogenic agents related to natural lignans/neolignans, we were attracted by a previously studied synthetic dihydrobenzofuran neolignan. We synthesized the compound by means of an eco-friendly, enzyme-mediated biomimetic coupling of the methyl ester of ferulic acid, and the present study was aimed to deeply investigate its effect in angiogenesis bioassays validated in our laboratory. In addition, a previously well-defined granulosa cell model was employed to evaluate the effect of dihydrobenzofuran neolignan on cell viability, steroidogenesis, and redox status. Present data support the antiangiogenic effect of this neolignan. Moreover, we demonstrate that, at least at the highest concentrations tested, dihydrobenzofuran neolignan affects granulosa cell viability and steroidogenesis. In addition, the compound inhibits generation of free radicals and stimulates scavenger enzyme activities. The present data, which are a further deepening of the evaluation of the biological activities of the dihydrobenzofuran lignan in well-defined cell models, are of interest and worthy of special attention.
Introduction
In recent years, a renewed attention has been devoted to natural products as leading compounds in drug discovery. Many natural products found in plants are potential therapeutic agents to be used in a wide range of pathologies.
1
Phytochemicals include compounds with various biological properties, which have possibly evolved to allow plants to cope with environmental challenges, including exposure to radiation and toxins and defense against pests and infectious agents. Among phytochemicals, phenolic compounds, and in particular those found in food plants, have received particular attention in past years, in particular as antioxidant and cancer chemopreventive agents.
2
However, because their bioavailability is generally poor, this observation has stimulated the search for synthetic analogues, to be employed as potential antitumor therapeutic agents.3,4 Within the large family of polyphenols, lignans and neolignans are a distinct group; in fact, their biogenesis, based on oxidative coupling of radical monomeric species, leads to an impressive structural variety paralleled by an array of promising biological properties. In addition, analogues of the natural lignans or neolignans can be prepared by simple metal- or enzyme-mediated biomimetic synthesis.4,5 In the frame of our interest in natural-derived compounds with antiproliferative and antiangiogenic properties, we have recently obtained the unusual benzoxanthene lignan
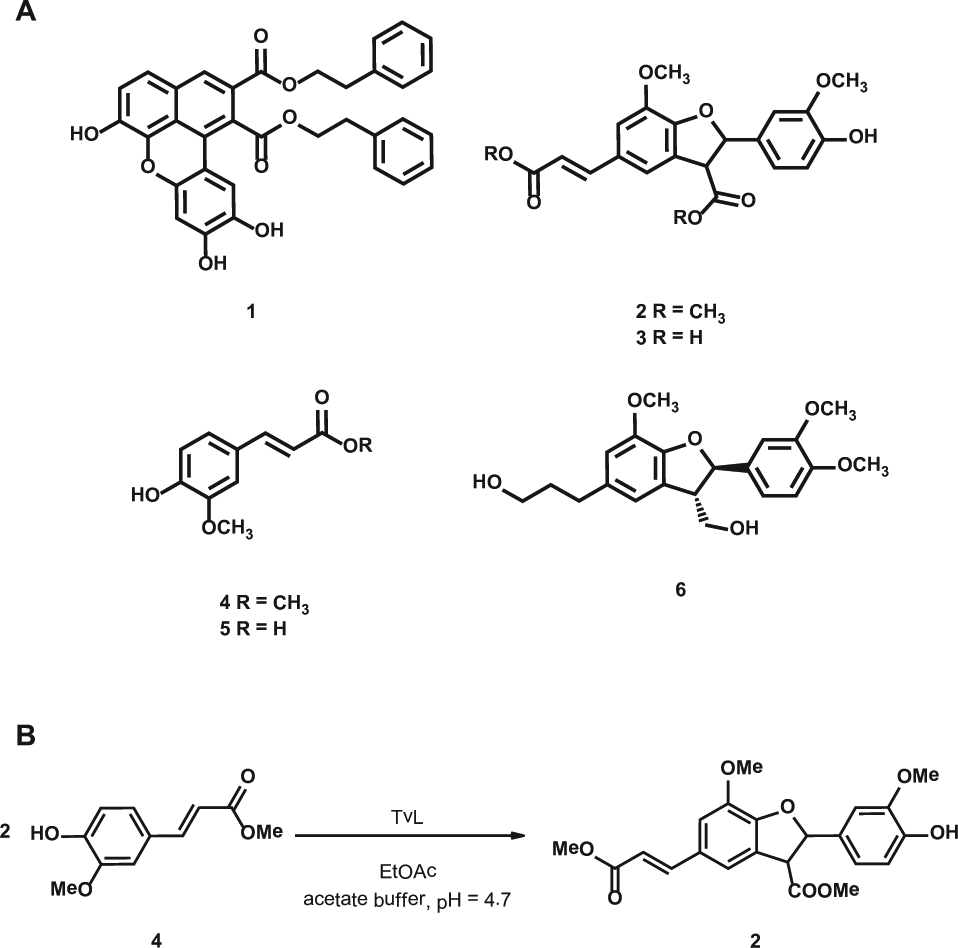
(
Therefore, the present study was conducted to deeply assess the effect of compound
Materials and Methods
Chemistry: Synthesis of Compounds 4 and 2
Laccase from Trametes versicolor laccase (TvL) and all reagents used in this study were obtained from Sigma (Milan, Italy, and St. Louis, MO) unless otherwise specified; ferulic acid was purchased from Fluka (Milan, Italy).
Synthesis of methyl ferulate (4 )
To a solution of ferulic acid (322 mg, 1.66 mmol) in MeOH (100 mL), H2SO4 conc. (2 mL) was added; the resulting mixture was stirred for 2 h under reflux. After cooling at room temperature, 200 mL of ethyl acetate was added to the mixture. The organic phase was washed with an NaHCO3 solution and saturated brine, dried over anhydrous Na2SO4, and the solvent evaporated under vacuum to yield the methyl ferulate
Synthesis of compound 2
The methyl ferulate (
Evaluation of the Effects of Compound 2 on Angiogenesis
Angiogenesis bioassay
Endothelial cell culture
An immortalized porcine aortic endothelial cell (AOC) line 20 was generously provided by José Yelamos (Hospital Universitario Virgen de la Arrixaca, El Palmar, Murcia, Spain). In all experiments, AOCs at the 19th passage were used and seeded in culture medium (CM) composed of M199 supplemented with sodium bicarbonate (2.2 mg/mL), bovine serum albumin (BSA; 0.1%), penicillin (100 IU/mL), streptomycin (100 µg/mL), amphotericin B (2.5 µg/mL), selenium (5 ng/mL), and transferrin (5 µg/mL).
Three-dimensional endothelial cell culture on a fibrin gel support
The microcarrier-based fibrin gel angiogenesis assay was performed as described by Basini et al.
21
Briefly, 12.5 mg of gelatin-coated cytodex-3 microcarriers in 1 mL phosphate-buffered saline (PBS) was incubated for 3 h to hydrate. After two washings in PBS and one in CM, the microcarriers were put in flasks containg 5 mL CM; AOCs (5 × 105) were added and cultured for 24 h to let the endothelial cells coat the microcarriers. For the fibrin gel preparation, 40 µL microcarriers covered by AOCs were pipetted into six-well plates containing a solution of fibrinogen (1 mg/mL PBS, pH 7.6), added to 1250 IU thrombine (250 µL). Fibrin gels were allowed to polymerize for 30 min at 37 °C and then equilibrated for 60 min with 2 mL M199. After a change of the medium, AOCs were treated with VEGF (100 ng/mL; PeproTech EC Ltd, London, UK) in the presence or absence of 0.1, 1, 10, and 100 µM of compound
Quantification of AOC growth on the fibrin gel matrix
Endothelial cell proliferation in the fibrin gel matrix was evaluated by means of the public domain National Institutes of Health Program Scion Image Beta 4.02 (Scion Corporation, MA, http://rsb.info.nih.gov/nih-image/). Ten pictures were taken for each gel at 48 and 96 h; images were converted into gray scale, resized to 50% (Paintbrush Software, MS Office), and saved as 24-bit Bitmap format compatible with Scion. The modified images were then imported into the program, and measurements were made by drawing the perimeter of the area occupied by the AOCs expressed as number of pixels. To validate the measurement of the area covered by AOCs in fibrin gels as a reliable method to evaluate cell proliferation, fibrin gels were stained by the nuclear dye bis-benzimide (Hoechst 33258, 20 µg/mL in PBS for 60 min) and examined by the fluorescence microscope. 22 This procedure was performed 20 times; for each experiment, the number of nuclei was counted under fluorescence, and pictures of the aera covered by AOCs were taken to measure the surface covered in the fibrin gel. A strong correlation was observed between the area covered by AOCs and the number of nuclei found in the same area (r = 0.96).
VEGF production
Granulosa cell collection
Swine ovaries were collected at a local slaughterhouse from Large White cross-bred gilts, parity = 0. The stage of the cycle was unknown. Follicles were classified on a dimension-based fashion.
15
The ovaries were placed into cold PBS (4 °C) supplemented with penicillin (500 IU/mL), streptomycin (500 µg/mL), and amphotericin B (3.75 µg/mL); maintained in a freezer bag; and transported to the laboratory within 1 h. After a series of washings with PBS and ethanol (70%), granulosa cells were aseptically harvested by aspiration of large follicles (>5 mm) with a 26-gauge needle and released in medium containing heparin (50 IU/mL), centrifuged for pelleting and then treated with 0.9% prewarmed ammonium chloride at 37 °C for 1 min to remove red blood cells. Cell number and viability were estimated using a hemocytometer under a phase contrast microscope after vital staining with trypan blue (0.4%) of an aliquot of the cell suspension. Cells were seeded in CM M199 supplemented with sodium bicarbonate (2.2 mg/mL), BSA (0.1%), penicillin (100 IU/mL), streptomycin (100 µg/mL), amphotericin B (2.5 µg/mL), selenium (5 ng/mL), and transferrin (5 µg/mL). Once seeded, cells were incubated in the presence or absence of compound
Granulosa cell VEGF production
Granulosa cells (106/1 mL CM + 1% fetal calf serum) were seeded in 24-well plates and incubated for 48 h, as indicated above. VEGF in culture media was quantified by an enzyme-linked immunosorbent assay (ELISA; Quantikine, R&D System, Minneapolis, MN). This assay, developed for human VEGF detection, has been validated for pig VEGF. 23 The assay sensitivity was 8.74 pg/mL, and the inter- and intra-assay coefficients of variation were always less than 7%. A Victor Reader (Perkin Elmer, Boston, MA) set to read at a wavelength of 450 nm emission was used to quantify the reaction product.
Evaluation of the Effects of Compound 2 on Granulosa Cell Function
Granulosa cell viability
Granulosa cells (2 × 105/200 µL CM) were seeded in 96-well plates. Cell viability was assayed using a bioluminescent assay (ATP-lites; Packard Bioscience, Groningen, the Netherlands), which measures intracellular adenosine triphosphate (ATP) levels as an indicator of cell numbers. ATP is a cell viability marker because it is present in all metabolically active cells, and the concentration declines very rapidly when the cells undergo necrosis or apoptosis. The ATP lite-M assay system is based on the production of light caused by the reaction of ATP with added luciferase and D-luciferin. This is illustrated in the following reaction scheme: ATP + D-luciferin + O2 → luciferase, Mg2+ → oxyluciferin + AMP + PPi + CO2 + light. The emitted light is proportional to the ATP concentration. Briefly, 50 µL of mammalian cell lysis solution was added to 100 µL of cell suspension, and the plate was shaken for 5 min in an orbital shaker at 700 rpm to lyse the cells and stabilize ATP. Then, 50 µL of substrate solution was added to the wells, and the microplate was shaken for 5 min in an orbital shaker at 700 rpm. The plate was placed in the dark for 10 min, and the luminescence was measured in a luminometer Victor Reader (Perkin Elmer). The results were recorded in counts per second, and the percentage of cell viability was calculated with reference to the negative control (cells without compound
Granulosa cell steroid production
Granulosa cells (104/200 µL CM) were seeded in 96-well plates and supplemented with androstenedione (28 ng/mL). Culture media were then collected, frozen, and stored at −20 °C until progesterone (P4) and estradiol 17β (E2) determination by validated radioimmunoassays. 24 P4 assay sensitivity and ED50 were 0.24 and 1 nmol/L, respectively; E2 assay sensitivity and ED50 were 0.05 and 0.2 nmol/L. The intra- and interassay coefficients of variation were less than 12% for both assays.
Evaluation of the Effects of Compound 2 on Granulosa Cell Redox Status
Superoxide (O2–) assay
O2– production was evaluated by WST-1 (4-[3-(4-iodophenyl) – 2-(4-nitrophenyl)-2H – 5-tetrazolium]-1,3-benzene disulfonate) test (Roche, Mannheim, Germany). The assay is based on the cleavage of the water-soluble tetrazolium salt, WST-1, to a yellow-orange, water-soluble formazan. Evidence exists that tetrazolium salts can be used as a reliable measure of intracellular O2– production. 25 A total of 104 cells/200 µL CM were seeded in 96-well plates and incubated for 48 h. During the last 4 h of incubation, 20 µL of WST-1 was added to cells, and absorbance was then determined using the Victor Reader (Perkin Elmer) at a wavelength of 450 nm against 620 nm. The coefficients of variation were less than 3%.
Hydrogen peroxide (H2O2) assay
H2O2 production was measured by an Amplex Red Hydrogen Peroxide Assay Kit (Molecular Probes, PoortGebouw, the Netherlands); the Amplex Red reagent reacts with H2O2 to produce resorufin, an oxidation product. Briefly, 2 × 105 cells/200 µL CM were seeded in 96-well plates and incubated for 48 h. After centrifugation for 10 min at 400 × g, the supernatants were discarded, and cells were lysed adding cold Triton 1% in TRIS HCl (100 µL/well) and incubating on ice for 30 min. Undiluted cell lysates were used to perform the test and read against a standard curve of H2O2 ranging from 0.195 to 12.5 µM. The absorbance was determined with the Victor Reader (Perkin Elmer) using a 540-nm filter. The coefficients of variation were less than 5%.
Scavenging enzyme assays
Granulosa cells (2 × 105/200 µL CM) were seeded in 96-well plates and incubated for 48 h. After centrifugation for 10 min at 400 × g, the supernatants were discarded, and cells were lysed adding cold Triton 1% in TRIS HCl (100 µL/105 cells) and incubating on ice for 30 min. Superoxide dismutase (SOD), catalase, and peroxidase activities were assessed in cell lysates as described below.
SOD activity was determined by a SOD Assay Kit (Dojindo Molecular Technologies, Kumamoto, Japan). Cell lysates were tested without dilution, and a standard curve of catalase ranging from 0.156 to 20 U/mL was prepared. The colorimetric assay was performed by measuring formazan produced by the reaction between tetrazolium salt (WST-1) and superoxide anion (O2–), produced by the reaction of an exogenous xantine oxidase. The remaining O2– is an indirect hint of the endogenous SOD activity. The absorbance was determined with Victor Reader (Perkin Elmer) reading at 450 nm against 620 nm. The coefficients of variation were less than 3%.
Catalase activity was measured by an Amplex Red Catalase Assay Kit (Molecular Probes) based on the formation of an oxidation product (resorufin) derived from the reaction between H2O2 given in excess and the Amplex Red reagent in the presence of horseradish peroxidase. Cell lysates were diluted 1:10 to perform the test and read against a standard curve of catalase ranging from 62.5 to 1000 mU/mL. The absorbance was determined with Victor Reader (Perkin Elmer) using a 540 nm filter. The coefficients of variation were less than 5%.
Peroxidase activity was measured by an Amplex Red Peroxidase Assay Kit (Molecular Probes) based on the formation of an oxidation product (resorufin) derived from the reaction between H2O2 given in excess and the Amplex Red reagent. Cell lysates were used undiluted to perform the test and read against a standard curve of peroxidase ranging from 0.039 to 5 mU/mL. The absorbance was determined with Victor Reader (Perkin Elmer) using a 540 nm filter. The coefficients of variation were less than 5%.
Statistical Analysis
The experiments were repeated at least five times (six replicates/treatment). Experimental data are presented as mean ± SEM; statistical differences between treatments were calculated with analysis of variance using Statgraphics package (STSC Inc., Rockville, MD). When significant differences were found, means were compared by Scheffè’s F test; p values less than 0.05 were considered statistically significant.
Results
Chemistry: Synthesis of Compounds 4 and 2
We have synthesized compound
Evaluation of the Effects of Compound 2 on Angiogenesis
Effect in angiogenesis bioassay
Compound
In particular, 1 and 10 µM exerted a dose-dependent effect (p < 0.001), which was significantly stronger (p < 0.001) after the 96 h culture. On the contrary, the highest concentration tested appeared to be significantly (p < 0.001) more effective after the 48 h incubation ( Fig. 2 ).
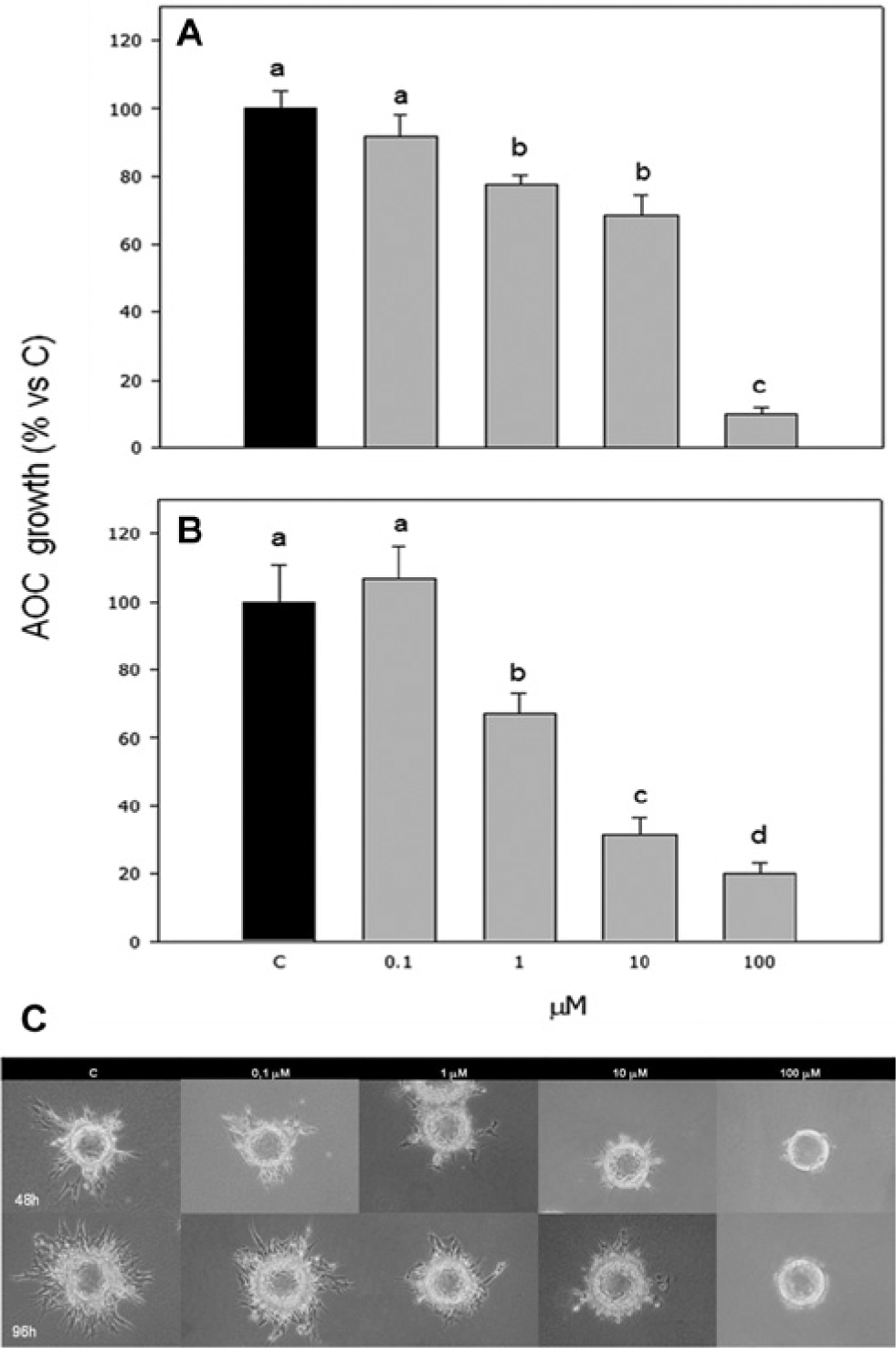
Effect of the treatment with compound
Effect on granulosa cell VEGF production
Basal VEGF production from granulosa cells amounted to 904 ± 72 pg/mL (mean ± SEM). Compound
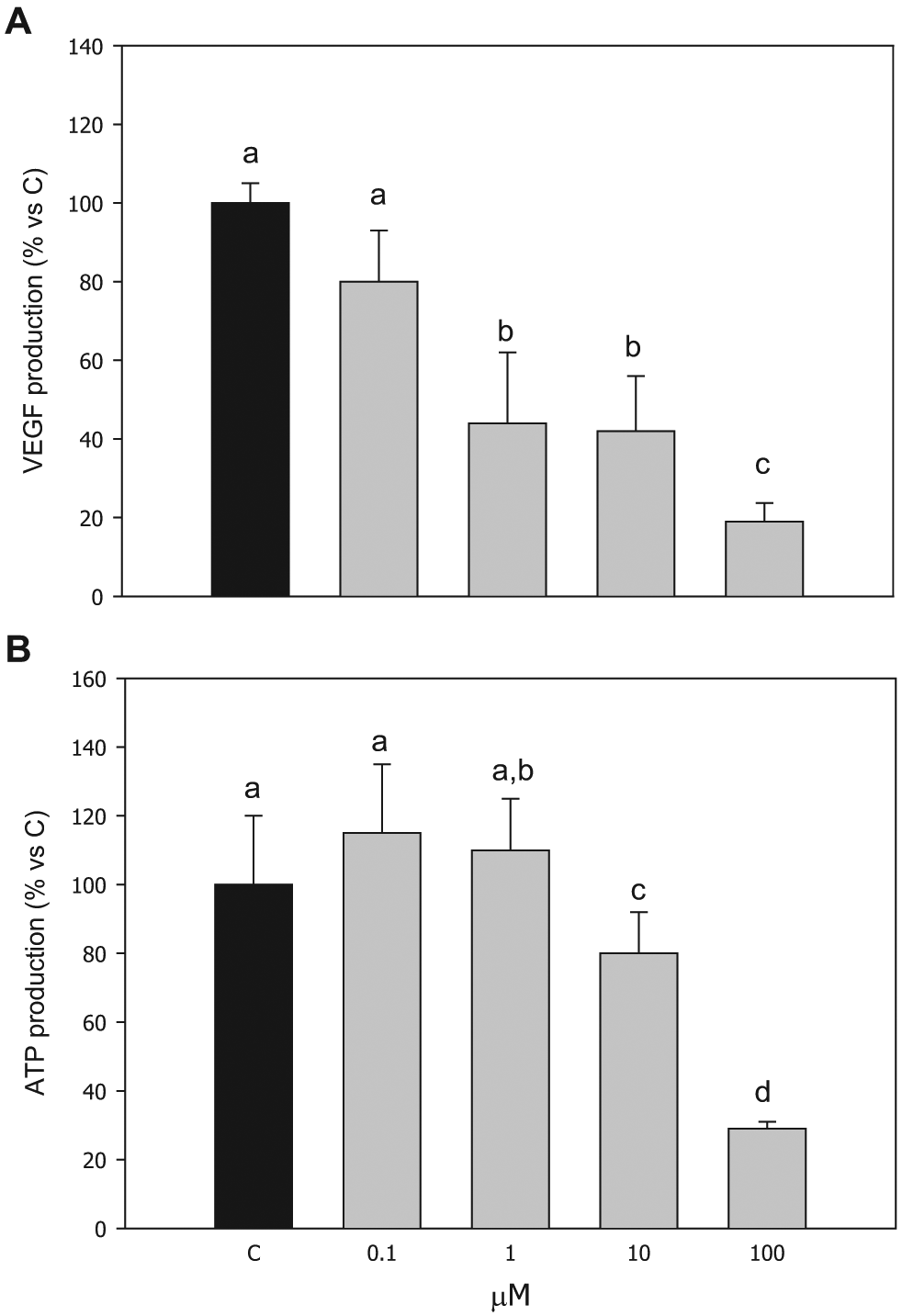
Effect of the treatment with compound
Evaluation of the Effects of Compound 2 on Granulosa Cell Function
Granulosa cell viability
Granulosa cell viability was negatively affected by the highest concentrations (10 and 100 µM) of compound
Granulosa cell steroid production
E2 and P4 basal production by granulosa cells was 5.3 ± 0.9 and 86.1 ± 11.2 ng/mL, respectively. Both steroids were significantly (p < 0.001) stimulated by 100 µM of compound
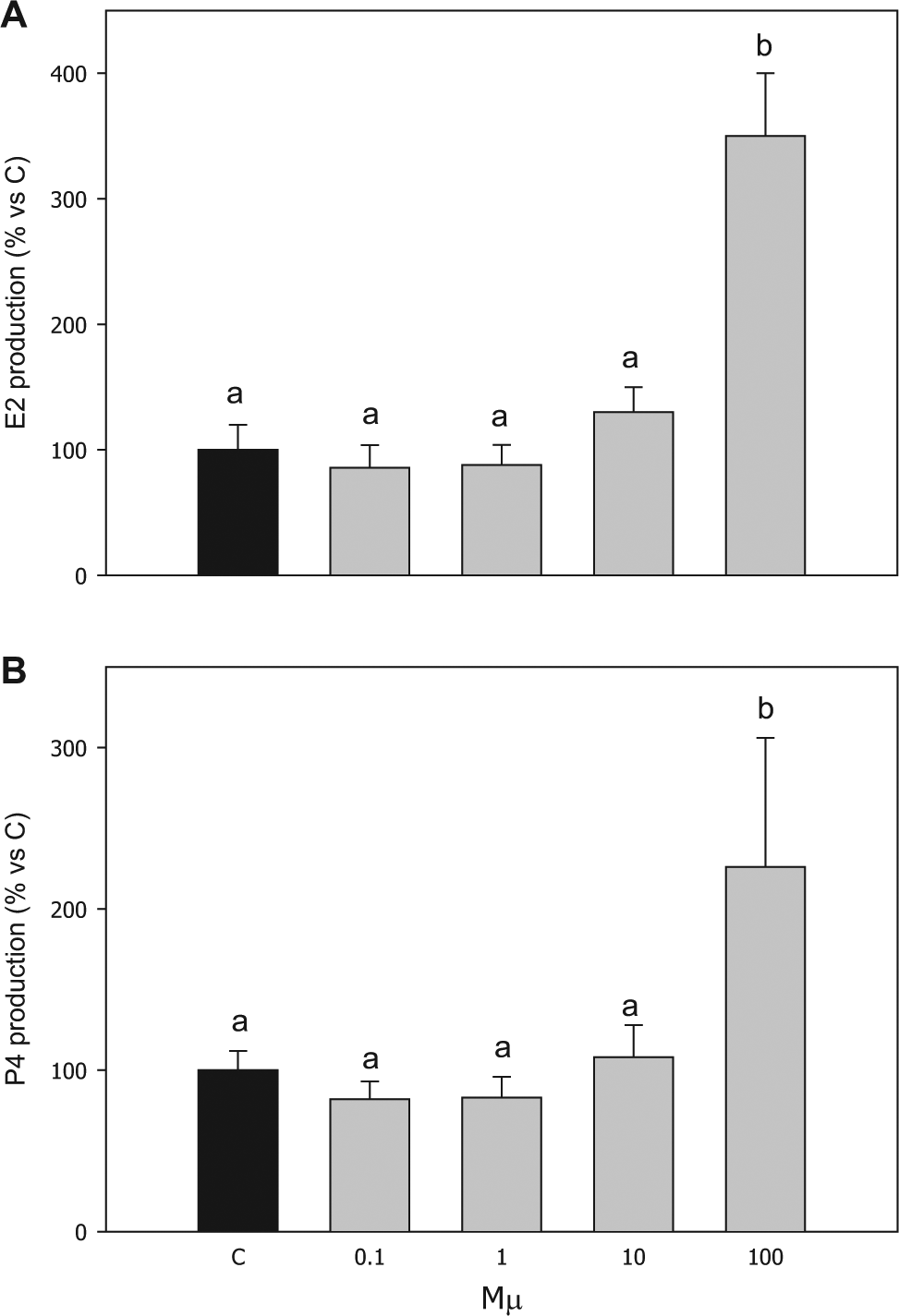
Effect of the treatment with compound
Evaluation of the Effects of Compound 2 on Granulosa Cell Redox Status
Superoxide (O2–) production
The effect induced by compound
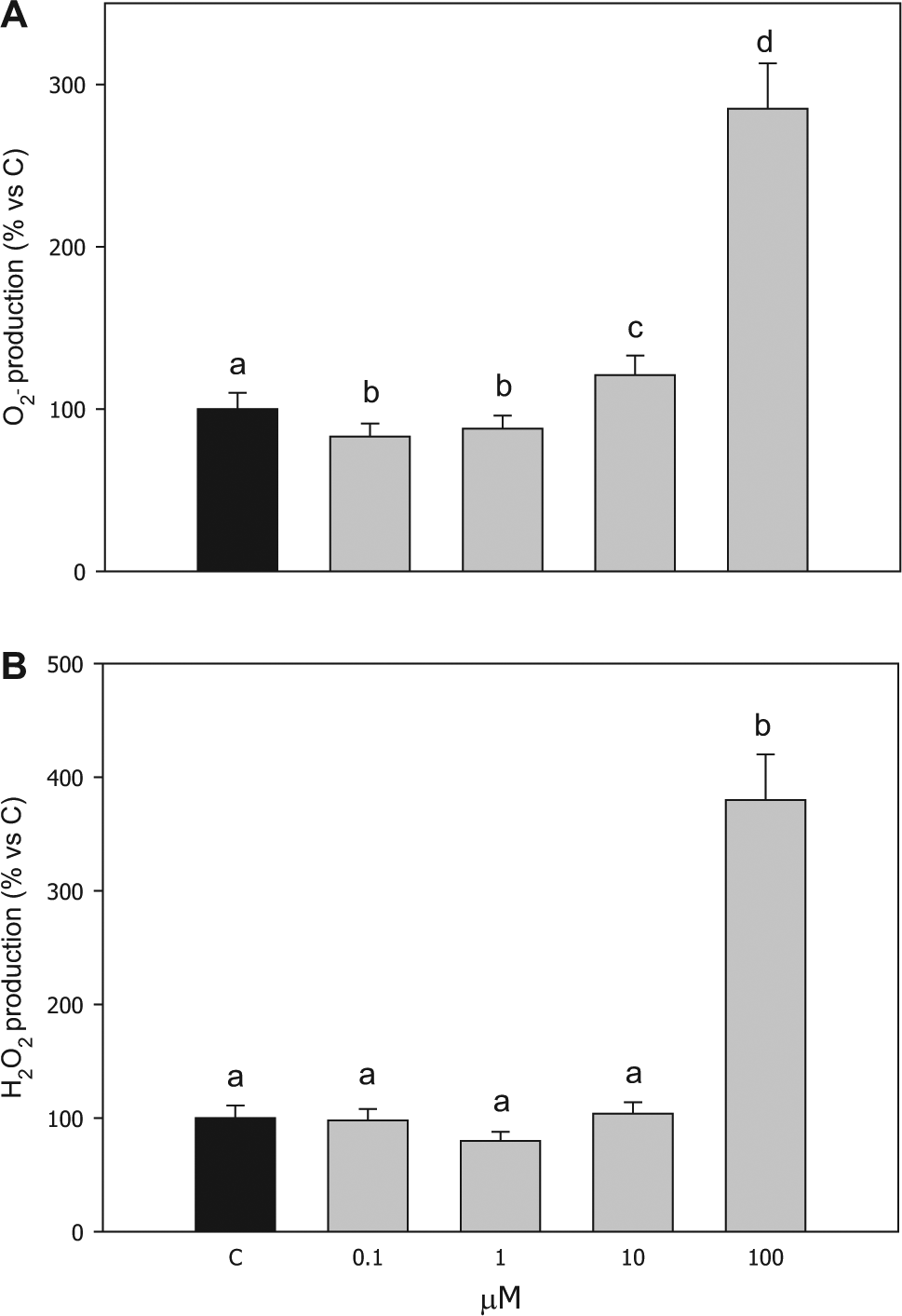
Effect of the treatment with compound
Hydrogen peroxide (H2O2) production
Compound
Scavenging enzyme activities
The activity of all the enzymatic scavengers examined was significantly increased by 100 µM of compound
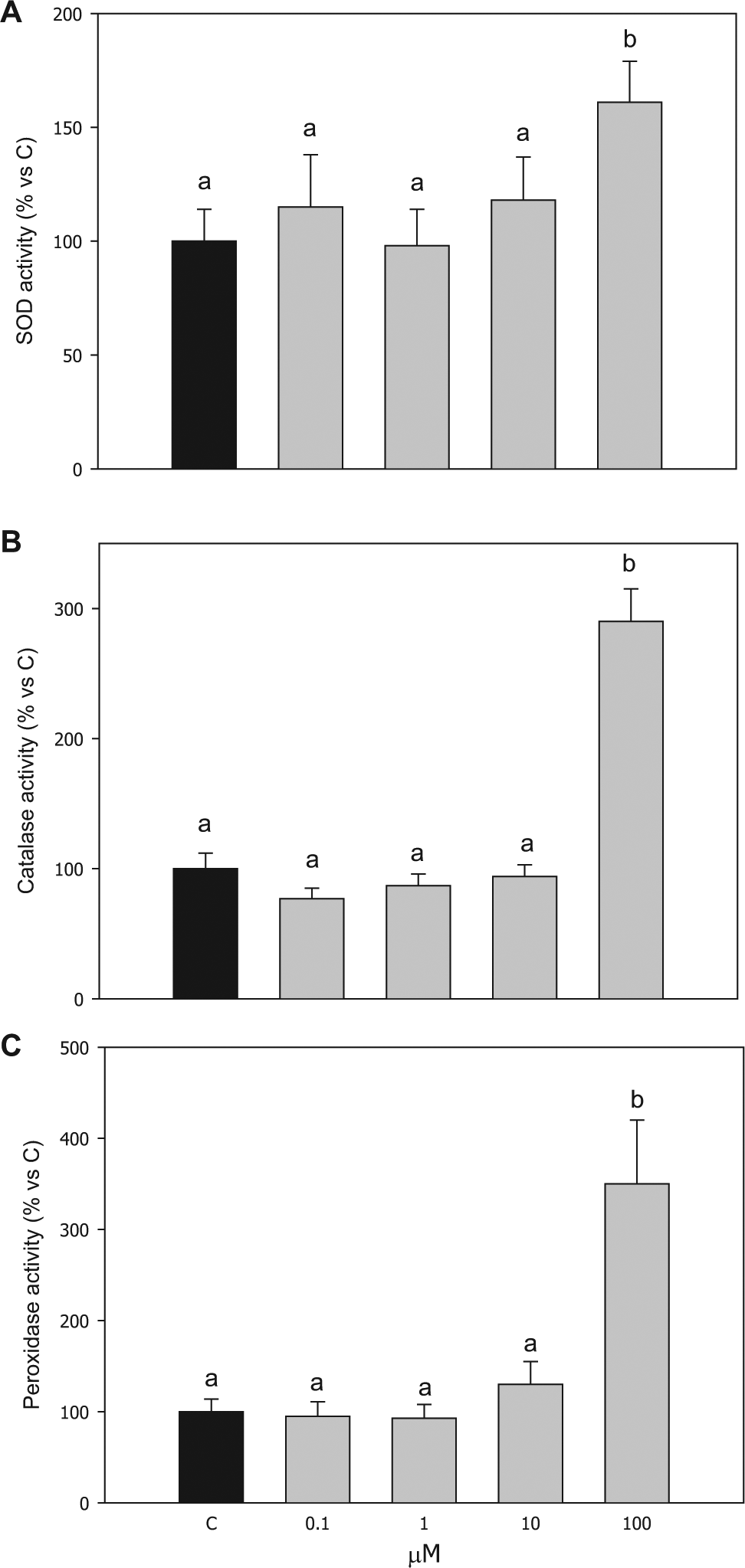
Effect of the treatment with compound
Discussion
Angiogenesis is a feature of a limited number of physiological processes, whereas pathological angiogenesis occurs in diseases such as cancer, rheumatoid arthritis, endometriosis, and diabetic retinopathy. Notably, because of the critical dependence of solid cancers on neoangiogenesis for growth, progression, and metastasis, it is not surprising that, in the past two decades, worldwide academic and industrial research has been focused on looking for angiotherapeutic agents to fight cancer. However, clinical trials with antiangiogenic modalities targeting VEGF/VEGFR2 signaling have shown limited efficacy and occasional toxic side effects. Therefore, screening strategies for herbal phytochemicals based on other signaling pathways have been explored.
19
Among these molecules, ferulic acid (
Moreover, our present research was aimed to better investigate the effects of compound
Interestingly, compound
Taken together, the results of this study represent a further deepening in the evaluation of the biological activities of the dihydrobenzofuran lignan compound
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by grants of the Università degli Studi di Parma (FIL) and Università degli Studi di Catania (Progetti di Ricerca di Ateneo, Catania, Italy) and by MIUR, Ministero dell’Università e della Ricerca (PRIN 2009, Rome, Italy).
