Abstract
Disruption of protein processing in the secretory pathway is a measurable hallmark of endoplasmic reticulum (ER) stress. Activation of ER stress–mediated pathways has been implicated in numerous diseases, including cancer. To identify agents that induce ER stress, we established a screen for compounds that reduce secretion of the reporter protein Gaussia luciferase (GLUC). Given the clinically validated importance of targeting ER stress–mediated pathways in the treatment of multiple myeloma (MM), we used this hematological malignancy as a model for validating our screening system. From a screen of 2000 marketed drugs and natural compounds in KMS11 and ARP1 MM cells, we identified 97 agents that reduced GLUC secretion in both cell lines by at least 30%. To confirm inducers of ER stress, we applied a secondary screen that assessed splicing of the unfolded protein response (UPR) transcription factor XBP1. One agent, theaflavin-3,3′-digallate (TF-3), was chosen based on its history of safe human consumption and further validated through studies of ER stress–related pathways, including the UPR and apoptosis. Given these promising results, this screen could be a useful tool to identify agents targeting ER stress–related mechanisms in other cellular systems wherein ER stress plays a role in disease etiology.
Introduction
The endoplasmic reticulum (ER) stress response constitutes a cellular process triggered by a variety of conditions that cause an imbalance in intracellular homeostasis and threaten proper cell function. 1 The stress response initiated upon recognition of an overabundance of misfolded or damaged proteins activates an adaptive effort aimed at neutralizing the deleterious effects of excessive protein accumulation and restoring proteostasis. 2 Severe and/or persistent levels of ER stress aggravate these systems, causing cells to abandon prosurvival efforts and instead initiate a proapoptotic signaling cascade involving activation of caspase 4.3,4 Recently, this dual-stress response system has been implicated in the pathogenesis of several human diseases, including diabetes, cancer, and neurodegenerative disorders. As such, exploitation of ER stress mechanisms is an area of intense therapeutic interest. 5
Blocking or reduction of protein processing in the secretory pathway is a hallmark of ER stress. 6 We previously reported the use of Gaussia luciferase (GLUC) secretion as a method for assessing the ER stress–inducing capability of naturally occurring isothiocyanates and arsenic trioxide. 7 Recognizing the clinically validated importance of targeting ER stress–mediated pathways in the treatment of a variety of diseases, we sought to employ this assay as a screening tool to identify and characterize novel agents that specifically target this critical pathway.
Multiple myeloma (MM) is a malignancy of terminally differentiated B cells accounting for approximately 10% of all hematological malignancies and affecting more than 20,000 patients each year in the United States. 8 Despite recent advances in targeted therapies and use of high-dose chemotherapy with autologous stem cell transplant, there is still no curative treatment. Relapse of disease and development of resistance are major obstacles to overcome for improving treatment response and patient survival. 9
A distinguishing characteristic of myeloma plasma cells is the large quantity of monoclonal paraprotein they synthesize and secrete, rendering them especially sensitive to the effects of ER stress. After synthesis, immunoglobulin is folded in the ER, where the unfolded protein response (UPR) maintains the balance between protein production and folding capacity. 10 The proteasome inhibitor bortezomib (BTZ) disrupts protein equilibrium in the ER by preventing misfolded proteins from being properly degraded. As such, it is a potent inducer of the UPR and ultimately of apoptosis. 11 Consistent with this notion, BTZ has demonstrated clinical efficacy as first-line treatment in patients with MM. However, BTZ is administered by subcutaneous injection, and approximately one-third of those receiving BTZ may have serious side effects such as peripheral neuropathy. 12 Thus, the need for the development of novel agents targeting ER stress–mediated pathways in the treatment of MM is of great clinical importance.
Protein folding is a complex process that requires chaperone proteins, glycosylating enzymes, and the proper oxidizing environment. ER stressors impair this process and cause accumulation of unfolded or misfolded proteins, leading to activation of the UPR, comprising three pathways. Activation of IRE1 (inositol-requiring protein-1) by ER stress signaling causes sequence-specific cleavage and subsequent splicing of messenger RNA (mRNA) encoding the transcription factor XBP1. XBP1s, the spliced form of XBP1, induces expression of the majority of UPR-related genes. A second branch of the UPR is initiated by PERK (protein kinase RNA-like ER kinase), which, upon activation, phosphorylates the α-subunit of the translation initiation factor eIF2, culminating in the attenuation of global translation initiation. In the third pathway, the transcription factor ATF6 (activating transcription factor 6) is activated through proteolytic cleavage after translocation to the Golgi upon conditions of ER stress. 13
Therefore, using the naturally secreted GLUC as a quantifiable indicator of protein secretion, 14 we developed an assay that allowed the effects of 2000 natural compounds and marketed drugs on GLUC secretion to be tested. From this screen, we identified 97 compounds that potentially perturbed protein secretion as a potential readout of ER stress. Based on its long and safe history of human consumption, one compound, the black tea polyphenol theaflavin-3,3′-digallate (TF-3), was further characterized with regard to growth inhibition and induction of ER stress in MM.
Materials and Methods
Compound Library Screening
The Spectrum library (Microsource Discovery Systems, Gaylordsville, CT), comprising 2000 marketed drugs and naturally occurring compounds, was used to screen for inhibitors of GLUC secretion in ARP1 and KMS11 MM cell lines.
Cell Lines
The human MM cell lines ARP1 and KMS11 were kindly provided by Dr. Hearn Cho (New York University School of Medicine, New York, NY).15,16 Cells were cultured in RPMI-1640 medium supplemented with 10% fetal bovine serum (FBS), 2 mM L-glutamine, and 1% penicillin-streptomycin at 37 °C in a 5% CO2 humidified atmosphere.
Gaussia Luciferase Secretion Assay
Commercially available lentiviral particles obtained from GenTarget (San Diego, CA) expressing GLUC under the control of a cytomegalovirus (CMV) promoter were introduced into KMS11 and ARP1 MM cells by infection. This lentivirus vector (GenTarget) carries an expression cassette for humanized GLUC, red fluorescent protein (RFP) for microscopic visualization, and puromycin resistance for selection of stable clones. For infection, cells were cultured in 6-well tissue culture plates to a density of 3 × 106 cells/mL and then diluted to 1 × 106 cells/mL in complete media. Lentiviral particle was added at a ratio of 100 µL of viral particles to 1 mL of cells. After 24 h, equal parts of fresh media containing puromycin selection were added. After 72 h, efficacy of transduction was assessed by RFP fluorescence. After infection, cells were maintained in media containing 3 µg/mL puromycin to establish stable clones. For secretion assays, 10,000 cells/well were plated in 96-well culture plates. Cells were immediately treated with 5 µM of compounds from the Spectrum library. Following 24-h incubation, expression and secretion of GLUC was monitored using the BioLux Gaussia Luciferase Assay Kit (New England Biolabs, Ipswich, MA) according to the manufacturer’s instructions through measurements of luciferase activity, as indicated by relative light units (RLUs) on a microplate luminometer (Molecular Devices, Sunnyvale, CA). Percent reduction in GLUC secretion was determined by the following equation: 1 – (RLU of treated cells/RLU ratio of the vehicle-treated cells). Any compound that inhibited GLUC secretion by >30% was considered a “hit.”
Cell Proliferation Assay
All cells were seeded in 96-well plates at a density of 5000 cells per well. After overnight incubation, cells were treated with candidate compound at concentrations ranging from 100 to 5000 nM. After a 72-h incubation period, proliferation was assessed using a tetrazolium dye reduction assay (CellTiter 96 AQueous Non-Radioactive Cell Proliferation Assay; Promega, Madison, WI). Absorbance was recorded on a microplate reader at 490 nm. Cellular proliferation was expressed as a percentage with vehicle-treated cells set at 100%. Each assay was performed in triplicate with mean values reported.
Western Blotting
KMS11 cells were harvested after treatment with 0 or 1 µM of candidate compounds for 24 h. Cells were then collected, washed in 1× in phosphate-buffered saline (PBS), and harvested in extraction buffer (1% Triton X-100, 50 mmol/L Tris, 2 mmol/L EDTA, 150 mmol/L NaCl, pH 7.5) containing protease inhibitor cocktail (Roche Applied Science, Indianapolis, IN) and phosphatase inhibitor cocktail (Sigma, St. Louis, MO). The lysates were centrifuged at 14,000 g, 4 °C for 20 min in a microcentrifuge. The protein concentrations of supernatants were measured with a protein assay kit (Bio-Rad, Hercules, CA). Proteins were separated by 12% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and transferred onto polyvinylidene difluoride membranes (Polyscreen; PerkinElmer, Waltham, MA). Antibodies used in these experiments include phospho-eIF2α for UPR studies, HSP 90 as a general marker of ER stress, and cleaved poly (ADP-ribose) polymerase (PARP) and cleaved caspase 4 for apoptosis studies (Cell Signaling Technology, Danvers, MA). For all blots, antiactin (Sigma) was used as a loading control. Immunoreactive bands were visualized using enhanced chemiluminescence detection reagent (Perkin-Elmer) and X-OMAT processing.
XBP1 Splicing
For the secondary screen, ARP1 and KMS11 cells were treated with 5 µM of indicated compounds for 24 h. KMS11 cells were also treated with indicated concentrations of TF-3, BTZ, or tunicamycin (TM) for 24 h. After indicated treatments, total RNA was isolated from lysed cells with the RNeasy Mini kit (Qiagen, Germantown, MD). XBP1 splicing was assessed by semi-quantitative reverse transcription PCR (RT-PCR) as described previously.17,18 Complementary DNA (cDNA) was produced from total RNA preps using the ImProm-II Reverse Transcription System (Promega). Primers spanning the fragment of XBP1 containing the intron targeted by Ire1α were used: 5′-TACGGGAGAAAACTCACGGC-3′ and 5′-GGGTCCAACTTGTCCAGAATGC-3′. The thermal PCR cycling conditions are as follows: 95 °C for 5 min, 95 °C for 1 min, 58 °C for 30 s, 72 °C for 30 s, and 72 °C for 5 min with 35 cycles of amplification. To properly distinguish the PCR products, samples were digested with Pst1 (New England Biolabs) that only cuts the unspliced cDNA. 19 Products were then separated on a 2.0% agarose/1× TAE gel and visualized by ethidium bromide.
Statistics
Unless otherwise noted, experiments were performed in triplicate. All data are presented as the mean ± standard error of the mean (SEM). 20 The p values were determined by a two-sided Student t test with unequal variance, with p < 0.05 considered significant.
Results
GLUC as a Reporter of Proteostasis in Human Myeloma Cells
GLUC is a naturally secreted luciferase that can be easily monitored through extracellular release of luciferase activity in real time. 14 Using the established MM cell lines KMS11 and ARP1, we created clones stably expressing GLUC under the control of a CMV promoter. Transduction efficiency of ARP1 and KMS11 MM cells expressing GLUC immediately after lentiviral transduction was determined by RFP expression by fluorescence microscope ( Fig. 1 ). Growth of uninfected cells was inhibited by addition of 3 µg/mL puromycin to the medium. Stable clones generated from this selection were subsequently used in our screening assay.
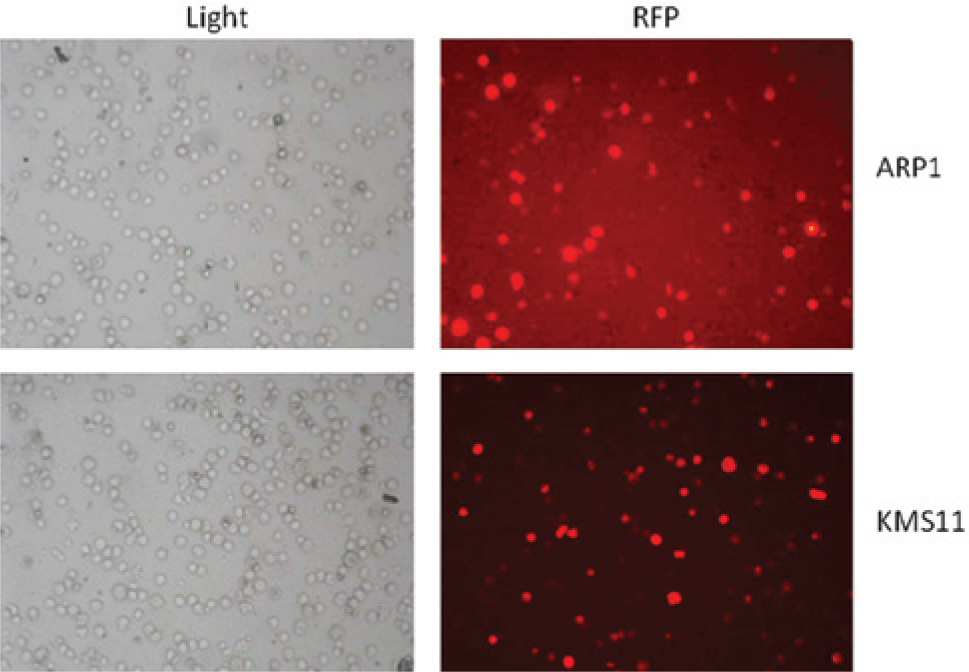
Transduction efficiency of KMS11 and ARP1 cells. Phase contrast (light) and fluorescent microscopy (red fluorescent protein [RFP]) imaging of ARP1 and KMS11 cells immediately after retroviral transduction. Cells expressing RFP have the GLUC expression construct stably integrated into the genome.
Linearity of the GLUC Assay
To ensure that the parameters of our assay system were within linear range, we assessed GLUC secretion in KMS11 and ARP1 with respect to time and cell density. Bioluminescence as a function of the secretion of GLUC by MM cell lines into the medium is linear with respect to time based on assessments from 9 to 30 h as values beyond the 30-h time point reach a plateau ( Fig. 2A , B ). In addition, linearity of GLUC secretion was also observed at cellular concentrations up to 60,000 cells/well or 80,000 cells/well in ARP1 and KMS11 cells, respectively, at 24 h ( Fig. 2C , D ). Based on these data, we decided to perform our screen using 40,000 cells/well with luciferase measurements taken at 24 h.
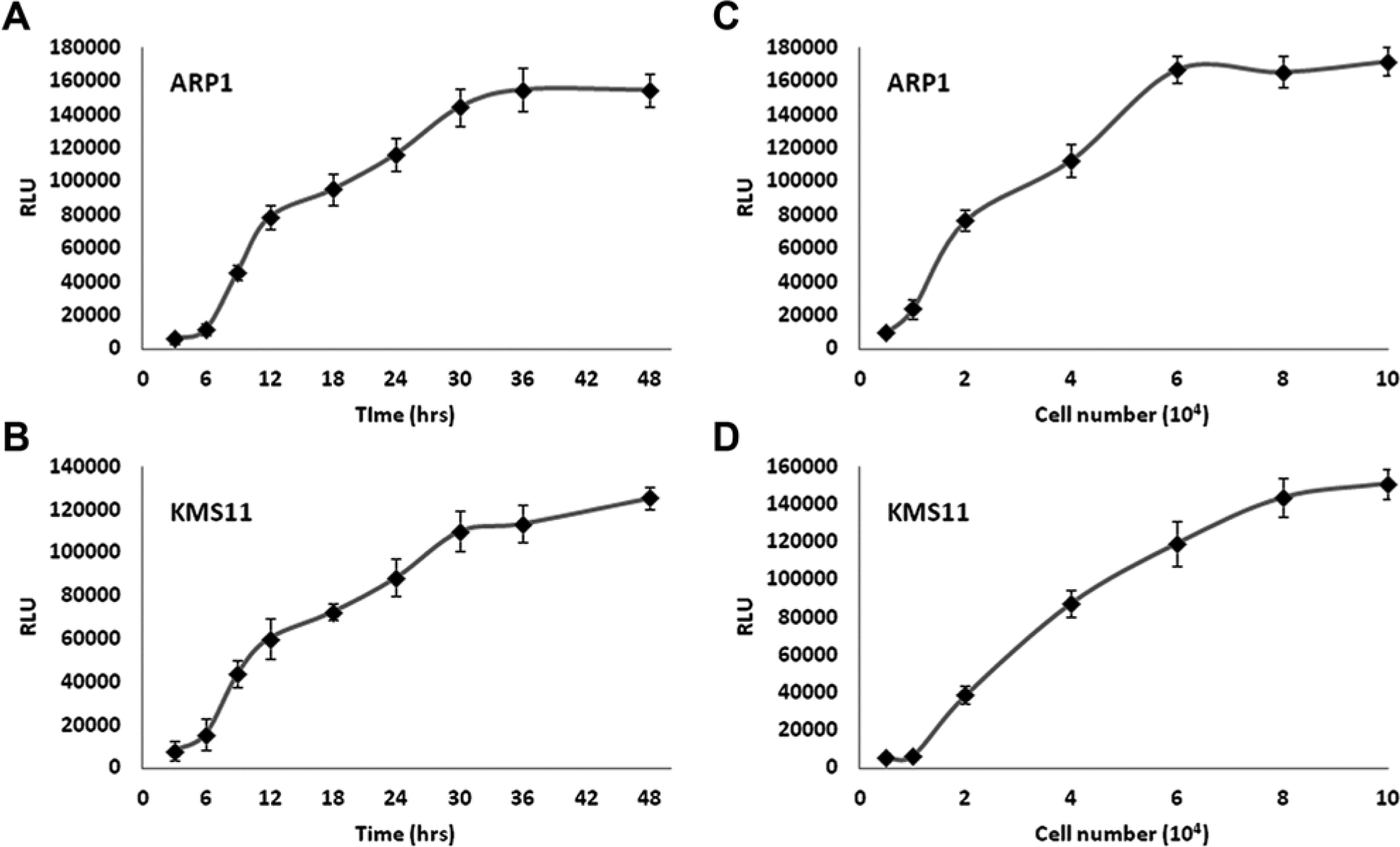
GLUC assay linearity with regard to time and cell concentration. Stable clones of ARP1 and KMS11 cells expressing GLUC were assessed for GLUC secretion at varying time points (
Proof of Principle of GLUC Screen to Identify ER Stress–Producing Agents
To determine whether GLUC secretion is a useful gauge of MM proteostasis, we tested the proteasome inhibitor BTZ and the well-characterized ER stress inducer TM as positive controls. BTZ is used clinically, whereas TM is a laboratory reagent. In addition, the DNA crosslinker cisplatin was used as negative control because it has been shown to induce apoptosis without interfering with proteasomal activities. 21 As shown in Figure 3 , 5 µM BTZ and 10 µg/mL TM similarly inhibit GLUC secretion in both KMS11 and ARP1 MM cells after 24 h of treatment. In contrast, 10 µM cisplatin has no effect on GLUC secretion in these cell lines ( Fig. 3 ). Moreover, Figure 4A shows that BTZ inhibits GLUC secretion in a dose-response fashion up to concentrations of 5 µM, where the inhibitory effect appears to reach saturation. These data suggest that our assay system is able to identify agents that disrupt protein secretion via ER stress–mediated pathways. Moreover, these important controls were used throughout the screening process to ensure the reliability and robustness of the screen.
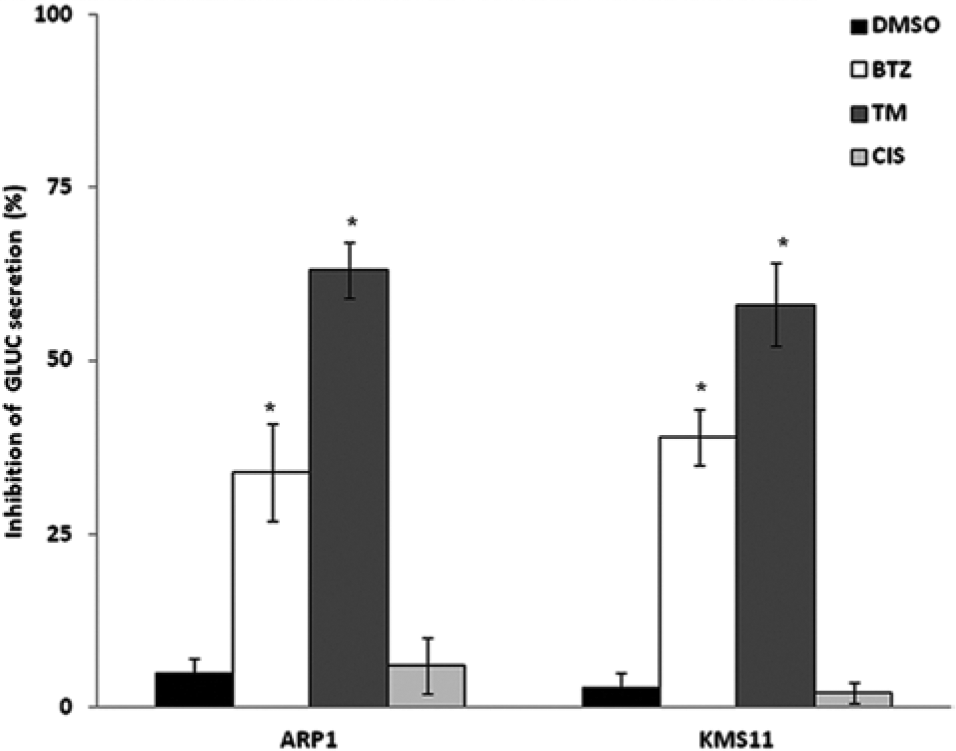
GLUC secretion as an indicator of proteostasis. The effect of 5 µM bortezomib (BTZ), 10 µM cisplatin (CIS), or 10 µg/mL tunicamycin (TM) on GLUC secretion is displayed. Inhibition of GLUC secretion is expressed as a percentage based on the following equation: 1 – (RLU of treated cells/RLU ratio of untreated cells). Cells treated for 24 h. All experiments were performed in triplicate, and error bars were calculated using standard error of the mean. *p < 0.05 as compared with control. RLU, relative light units.
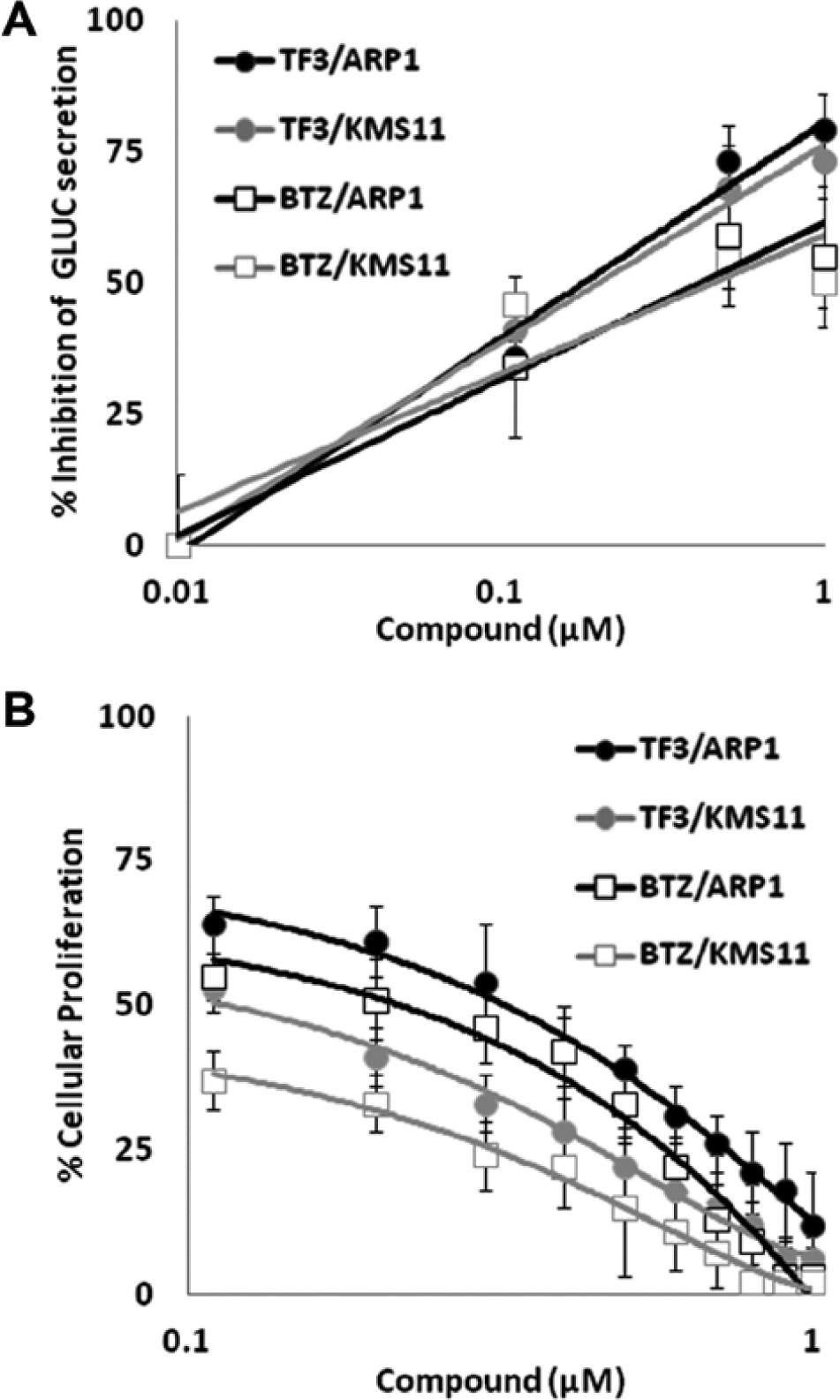
TF-3 inhibits cellular proliferation and GLUC secretion as effectively as bortezomib (BTZ) in KMS11 and ARP1 cells. (
Identification and Prioritization of Compounds
For screening, KMS11 and ARP1 cells were seeded in 96-well plates and treated with 5 µM of compounds from the Spectrum library (Microsource Discoveries) for 24 h. Conditioned medium from each compound treatment was harvested to assay GLUC secretion alongside samples treated with DMSO alone, BTZ, and TM as controls. Compounds of interest were considered to be those that inhibited GLUC secretion in both cell lines at least 30% after 24 h of treatment. We chose >30% inhibition of GLUC secretion in both cell lines as our threshold for determining initial hits based on the activity of BTZ, which served as our pharmacological benchmark. As shown in Figure 3 , 5 µM BTZ inhibited GLUC secretion by 34% and 39% in ARP1 and KMS11 cell lines, respectively.
From our screen of 2000 compounds, we identified 97 initial “hit” compounds. Of these compounds, 62 are human drugs, 31 are natural products, and 4 are traditionally used in veterinary medicine. Given that the library we screened is composed of marketed drugs and well-characterized bioactive compounds and natural products, we use previously published information as a means of prioritizing our list of hits. Because we are ultimately interested in bringing these compounds to the clinic, we focus our efforts on compounds that are active at clinically achievable concentrations. Furthermore, ease of drug administration (e.g., oral route and/or favorable pharmacokinetics) and safety profile are also considered in the prioritization of hits.
After narrowing our list of candidate compounds to those that may be viewed as clinically active (i.e., history of safety, achievable bioavailability), we employed a secondary screen to specifically identify and confirm inducers of ER stress. Cleavage of the unfolded protein response transcription factor XBP1 was assessed after treatment with 5 µM candidate compound for 24 h ( Table 1 ). Of the candidate compounds assessed, 14 compounds induced XBP1 cleavage in both MM cell lines. Therefore, the overall hit rate for this comprehensive screening technique is 0.7% (n = 2000).
GLUC Secretion, Growth Inhibitory, and XBP1 Splicing Activity of the Top Candidates from Screening.
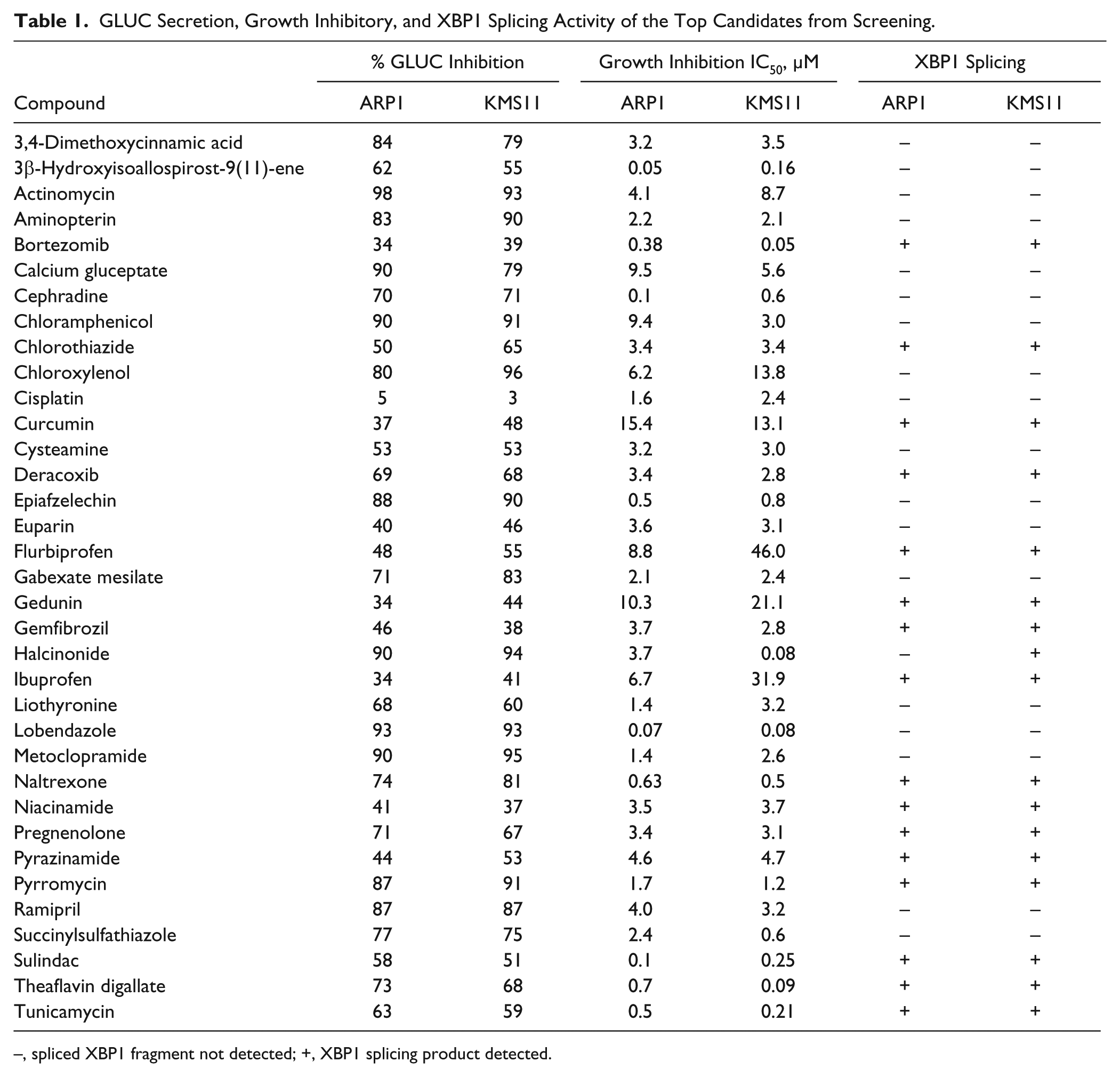
–, spliced XBP1 fragment not detected; +, XBP1 splicing product detected.
Of these compounds, we selected the black tea polyphenol theaflavin digallate (TF-3) for use in validation studies of mechanism of action based on its long and safe history of human consumption and an absence of prior reports of its ability to induce ER stress, as well as its remarkable potency as an inhibitor of GLUC secretion and proliferation of MM cell lines at physiologically achievable concentrations ( Table 1 ).
TF-3 as a Modulator of Proteostasis and Inducer of Apoptosis
As shown in Figure 4A , 0.5 µM TF-3 inhibited GLUC secretion by 73% and 68% in ARP1 and KMS11 cells, respectively. The IC50 values of TF-3 as an inhibitor of GLUC secretion in ARP1 and KMS11 MM cell lines are 0.22 µM and 0.35 µM, respectively ( Fig. 4A ). In comparison, the IC50 values of BTZ as an inhibitor of GLUC secretion in ARP1 and KMS11 MM cell lines are 0.56 µM and 0.64 µM, respectively ( Fig. 4A ). Moreover, TF-3 is also a potent inhibitor of MM cell line growth, with an activity pattern comparable to that of BTZ ( Fig. 4B ). The IC50 values of TF-3 as an inhibitor of growth in ARP1 and KMS11 MM cell lines are 0.59 µM and 0.27 µM, respectively, close to the potency exhibited by BTZ (0.38 µM and < 0.1 µM; Fig. 4B ).
To determine the mechanism of TF-3–induced death, we assessed induction of apoptosis as measured by cleavage of PARP. Cleavage of full-length PARP protein into a stable fragment is a well-documented marker of apoptosis. 22 As shown in Figure 5A , TF-3 is a potent inducer of cleavage of both PARP and the ER stress–specific caspase 4 in KMS11 cells. Altogether, these results indicate that TF-3 is a potent inducer of ER stress and apoptosis in KMS11 MM cells.
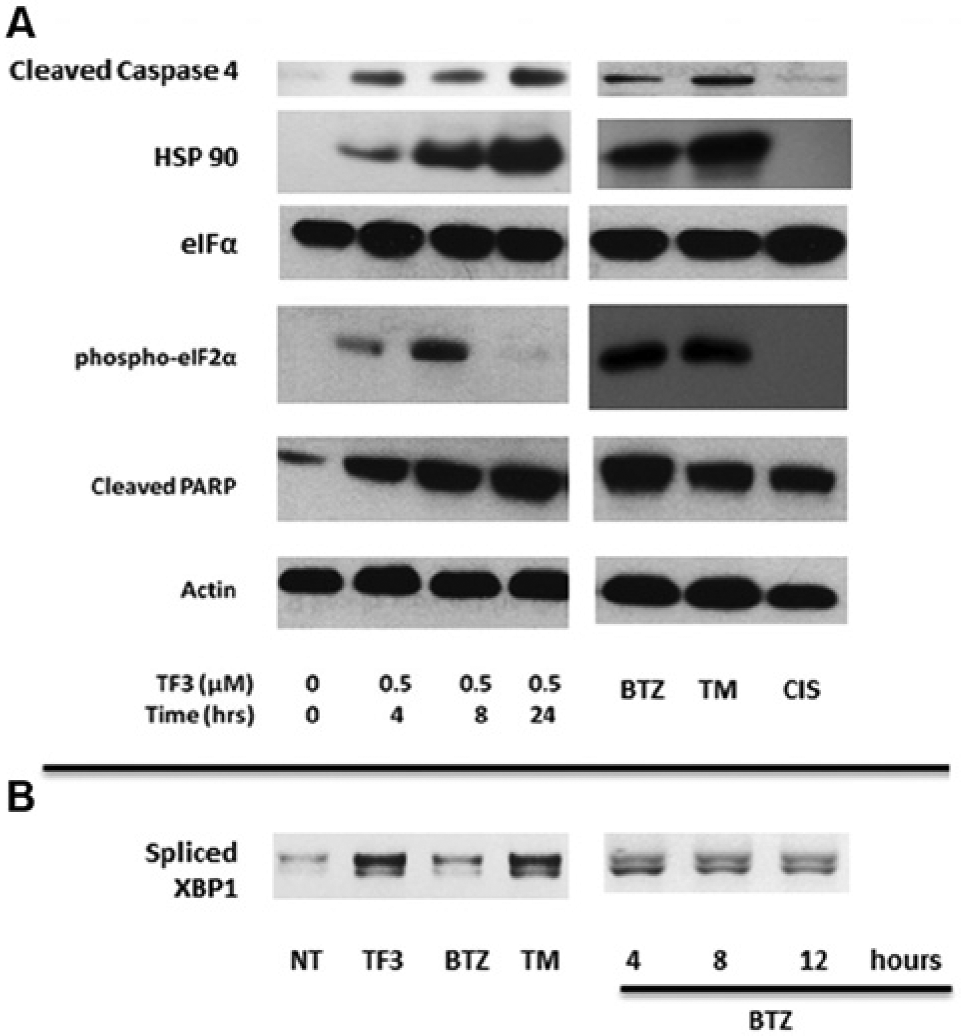
Treatment with TF-3 enhances endoplasmic reticulum (ER) stress and activates the unfolded protein response. (
To further elucidate the mechanism of theaflavin-mediated ER stress response, we evaluated the key UPR marker phospho-eIF2 as well as a general marker for ER stress (HSP90) by Western blotting. 23 As shown in Figure 5A , our data are consistent with the involvement of the UPR in the cellular response to TF-3, as indicated by enhanced phosphorylation of eIF2-α early in the time course. It is important to note that total eIF2-α levels were not affected by treatment. By 24 h, phosphorylation of this key regulator of protein synthesis is no longer apparent and possibly indicates a cellular shift to induction of the apoptotic machinery. In addition, HSP90 expression was also upregulated upon treatment with TF-3. Our positive controls BTZ and TM also effectively induced these markers of ER stress, whereas our negative control cisplatin had no effect. Activation of the IRE1 arm of the UPR response was observed through enhanced splicing of the UPR transcription factor XBP1 upon treatment with TF-3 ( Fig. 5B ). The level of spliced XBP1 observed upon treatment with TF-3 is similar to cells treated with the positive control TM. Interestingly, the effect of BTZ on XBP1 cleavage at 24 h is much lower. Therefore, we tested BTZ at earlier time points, and notable XBP1 splicing was observed ( Fig. 5B ).
Discussion
In the present study, we developed a screen to identify agents that inhibit GLUC secretion as a readout of MM cell proteostasis and used it to screen 2000 natural products and marketed drugs. The GLUC secretion system has previously been used as a reporter assay in mammalian cells to monitor GLUC secretion and ER stress after treatment with the ER stress inducer thapsigargin. 14 Here we employed a similar system in MM cells to screen for agents potentially effective at targeting proteostasis systems critical to MM cell survival. Our GLUC secretion screen was conducted at 24 h with 40,000 cells/well as these values were well within the linear range of our assay ( Fig. 2 ). Additional characterization of one candidate compound, TF-3, shows that it is a potent inducer of ER stress and apoptosis ( Fig. 5 ), thereby further supporting the notion that our screening system is effective at identifying agents that target ER stress–mediated pathways.
Because our screen was conducted with a panel of marketed drugs and natural products, a significant amount of information regarding these compounds is already known. Therefore, we used an informatics approach as an alternative, cost-efficient means to prioritize our list of initial hits. Because our ultimate goal is to bring these compounds into a clinical setting, we were specifically interested in focusing on compounds that are active at clinically achievable concentrations with a well-documented safety profile in humans.
We applied an initial filter to our candidate compounds based on previously published mechanistic information of candidate compounds. Previously reported inducers of ER stress identified by our screen include ibuprofen, sulindac, gedunin, and curcumin.24–27 Identification of these known ER stress–inducing agents serves as an additional validation to our assay system. In addition, based on the available information, other “hits” were secondarily eliminated based on their known mechanisms of action, which could easily explain their detection as a hit in our screen. For example, actinomycin is a well-known inhibitor of DNA transcription; therefore, it likely inhibits GLUC secretion by acting upstream of the protein secretion pathway. Thus, we are able to eliminate hits that target pathways upstream of protein secretion like the DNA transcription suppressor actinomycin ( Table 1 ).
Using this approach, we narrowed down our list of hits to 32 compounds that met our criteria of safety and achievable bioavailability. As a secondary screen, we assessed the XBP1 splicing-inducing capabilities of the candidate compounds in both ARP1 and KMS11 MM cells. Because XBP1 splicing is specifically indicative of activation of the UPR as a result of ER stress–related mechanisms, the application of this secondary screen is necessary to confirm that our hits are inducers of ER stress. The secondary screen further narrowed our list of hits to 14 compounds, thereby resulting in an overall screen hit rate of 0.7%.
With our goal of ER stress specificity and clinical utility in mind, we narrowed our focus to compounds that were most effective at inhibiting GLUC secretion and MM cell growth as well as inducing XBP1 splicing ( Table 1 ). We ultimately decided to focus our assay validation studies on TF-3 as it was one of the most effective inhibitors of GLUC secretion and MM cellular proliferation without a previously documented role in promoting ER stress ( Table 1 ). Indeed, TF-3 closely mirrors BTZ’s GLUC secretion and MM growth inhibitory profile ( Fig. 4 ).
Additional characterization of TF-3 showed that it is a potent inducer of ER stress and apoptosis ( Fig. 5 ), thereby further supporting the notion that our screening system is effective at identifying agents that target ER stress–mediated pathways. Indeed, our data show that exposure of KMS11 cells to TF-3 causes an early upregulation of the key chaperone protein HSP90 and phosphorylation of eIF2α ( Fig. 5A ). Phosphorylation of eIF2α inhibits the initial step of polypeptide synthesis. 28 If the cell is unable to reinstate proteostasis through inhibition of protein synthesis, the focus of the UPR signaling pathway shifts to induction of apoptotic cascades. Our data may potentially reflect the transition to apoptotic initiation after 24 h of exposure to TF-3 since at this time, eIF2α is no longer phosphorylated, and markers of cellular apoptosis such as cleavage of caspase 4 and PARP are most prominent at 24 h ( Fig. 5A ).
GLUC is a reporter luciferase from the marine copepod Gaussia princeps that can be easily expressed in mammalian cells using commercially available reporter plasmids. 29 It is the basis for a well-established and highly sensitive assay for monitoring the secretory pathway and ER stress. 14 Although this assay has been used previously to identify cellular genes critical to recombinant protein production, 30 our screening system described herein is the first to our knowledge to employ this cost-effective assay as a means for identifying potential therapeutic agents from compound libraries that target ER stress–related pathways in MM cells. Given our promising results using MM as a model system and the ability to express GLUC in a variety of mammalian cells, this screening system may serve as a potentially useful tool to identify agents that promote ER stress in other disease models.
Disruption in ER stress–related signaling pathways has been implicated in a variety of diseases, including cancer, diabetes, and neurodegenerative disorders such as Alzheimer and Parkinson diseases. With specific regard to cancer, because uncontrolled cell growth is a hallmark of malignant tumors, which tends to create stressful conditions like hypoglycemia, hypoxia, or accumulation of misfolded proteins during upregulated protein synthesis, the adaptive prosurvival components of the ER stress response are often found chronically activated in tumor cells. 31 This ER stress adaptive phenotype distinguishes tumor cells from normal cells and has been proposed as opportunity for therapeutically targeting cancer cells. 32 Indeed, the anticancer activities of a number of ER stress–inducing agents like celecoxib and nelfinavir are currently being assessed in clinical trials.5,31 Interestingly, our screen identified three traditional nonsteroidal anti-inflammatory drugs (NSAIDs) as initial hits: sulindac, flurbiprofen, and ibuprofen ( Table 1 ). The traditional NSAIDs inhibit both COX-1 and COX-2 enzymes, whereas new-generation drugs, such as celecoxib, primarily inhibit COX-2. 33
Tea is a popular beverage worldwide. Green tea and black tea are both derived from the plant Camellia sinensis. Black tea is subject to an additional fermentation step in the manufacturing process. This additional step results in the oxidation of tea flavanols and generates the theaflavins that are specific to black tea. Theaflavins comprise 10% of black tea extract and are responsible for black tea’s unique taste and distinguishing color. 34 Theaflavins have a benzotropolone ring structure and are collectively composed of a mixture of theaflavin (TF-1), theaflavin-3-gallate (TF-2a), theaflavin-3′-gallate (TF-2b), and TF-3. Over the past two decades, a significant amount of research has been devoted to extensively explore the molecular mechanism of the polyphenolic constituents of black tea as chemopreventative agents. This research has created a body of literature attributing various antitoxin and antimutagenic properties to black tea polyphenols.35–37 In addition, theaflavins have been reported to inhibit proliferation and induce apoptosis in a variety of cancer cell lines by modulation of various targets. Although no specific compound within the extract they studied was tested or identified, a recent publication by Mujtaba and Dou 38 showed that black tea extract inhibits tumor proteasome activity. This is consistent with our screening results, suggesting that TF-3 possesses the ability to disrupt protein secretion via the induction of ER stress. However, the relative contribution of theaflavins—and specifically which theaflavin—to the disruption of proteostasis is a subject of further study.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project described was supported in part by PHS grant AR041880 from NIAMS.
