Abstract
The development of genetically encoded redox biosensors has paved the way toward chemically specific, quantitative, dynamic, and compartment-specific redox measurements in cells and organisms. In particular, redox-sensitive green fluorescent proteins (roGFPs) have attracted major interest as tools to monitor biological redox changes in real time and in vivo. Most recently, the engineering of a redox relay that combines glutaredoxin (Grx) with roGFP2 as a translational fusion (Grx1-roGFP2) led to a biosensor for the glutathione redox potential (EGSH). The expression of this probe in mitochondria is of particular interest as mitochondria are the major source of oxidants, and their redox status is closely connected to cell fate decisions. While Grx1-roGFP2 can be expressed in mammalian mitochondria, it fails to enter mitochondria in various nonmammalian model organisms. Here we report that inversion of domain order from Grx1-roGFP2 to roGFP2-Grx1 yields a biosensor with perfect mitochondrial targeting while fully maintaining its biosensor capabilities. The redesigned probe thus allows extending in vivo observations of mitochondrial redox homeostasis to important nonmammalian model organisms, particularly plants and insects.
Introduction
Previously developed genetically encoded redox biosensors enable compartment-specific real-time measurements of redox changes in living cells. In particular, redox probes based on redox-sensitive green fluorescent protein (roGFP) 1 have found an increasing number of applications in plant and animal systems.2,3 RoGFPs exhibit a pair of cysteines engineered onto their surface in proximity to the fluorophore. Disulfide bond formation leads to ratiometric changes in the roGFP excitation spectrum. In contrast to cpYFP-based redox probes, ratio measurements based on roGFP2 are insensitive to pH within the physiological range. 4
In the presence of glutaredoxins, roGFPs are able to equilibrate with the glutathione (GSH)/glutathione disulfide (GSSG) redox pair. Reduced glutathione is one of the major antioxidants and is present in millimolar concentrations in cells. Its oxidized counterpart, GSSG, is generated by glutathione peroxidases during the reduction of peroxides. RoGFP2 has a midpoint potential of approximately −280 mV and is capable of measuring the glutathione redox potential (EGSH) in the range between −320 and −240 mV. As unstressed cells exhibit a steady-state EGSH between −300 mV and −320 mV, roGFP2 is well suited to monitor pro-oxidative changes in most subcellular compartments, with the exception of the secretory pathway. More detailed information about the properties of roGFP-based biosensors is given in Meyer and Dick. 4
Recently, a composite biosensor with improved sensing properties was created by combining the roGFP2 domain with human glutaredoxin-1 (Grx1). 5 The translationally fused Grx domain further increases the specificity of roGFP2 for the GSH/GSSG redox couple and abolishes the dependency of the biosensor on endogenous Grx. In addition, the close proximity of Grx1 and roGFP2 promotes rapid equilibration between the dithiol-disulfide pairs of roGFP2 and glutathione. The improved reaction kinetics facilitates measurements of EGSH with high temporal resolution. 5
Like roGFP2, Grx1-roGFP2 can be targeted to a variety of cellular compartments, and its expression in mitochondria has been found to be unproblematic in human and murine cells. However, we now find that Grx1-roGFP2 is not targetable to mitochondria in all organisms. In important models such as Drosophila melanogaster and Arabidopsis thaliana, the Grx domain interferes with mitochondrial targeting and/or import. Here we show that this limitation can be removed by inverting the domain order of the probe. Importantly, the rearrangement does not compromise biosensor functionality in any way.
Material and Methods
Cloning of roGFP2-Grx1
RoGFP2-Grx1 was generated by replacing Gpx4 in the vector roGFP2-[GGSGG]6-Gpx46 by Grx1. Initially, the coding sequence of Grx1 was amplified by PCR from a Grx1-roGFP2 plasmid with primers 5′-AAGAATTCGCTC AAGAGTTTGTGAACTGCAAAATCCAG-3′ and 5′-CC ATCGATTTACTGCAGAGCTCCAATCTGCTTTAG-3′ and inserted into the target vector pBluescript II KS(+) containing roGFP2-[GGSGG]6-Gpx4 with EcoRI and ClaI to replace Gpx4 behind the linker sequence. Subsequently, the entire roGFP2-Grx1-cassette was amplified by PCR and ligated into the respective expression vectors. For biosensor expression in Drosophila, the pUAST vector was used. Mitochondrial targeting sequences were from Neurospora crassa adenosine triphosphate (ATP) synthase (mito-Grx1-roGFP2 with NC targeting peptide) and human cytochrome c oxidase 8 (mito-Grx1-roGFP2 with COX8 targeting peptide), as used previously for mitochondrial targeting of enhanced green fluorescent protein (EGFP) in Drosophila. 7 The inverted construct made use of the COX8 targeting peptide (mito-roGFP2-Grx1). For production of recombinant roGFP2-GRX1, the coding sequence was cloned into pQE30 with BamHI and HindIII, leading to an N-terminal fusion to 6xHis. Recombinant proteins were produced and purified as described earlier. 5
Transformation of Arabidopsis
For expression of Grx1-roGFP25 and roGFP2-Grx1 in A. thaliana, both constructs where amplified by PCR and inserted into the binary vector pBinAR-SHMT with BamHI and SalI behind the mitochondrial targeting peptide of Arabidopsis serine hydroxymethyltransferase (SHMT; GenBank AJ271726.1). After electroporation of binary vectors into Agrobacterium tumefaciens strain AGL-1, the Agrobacteria were incubated in 30 mL of selective LB medium (50 µg/mL rifampicin, 100 µg/mL carbenicillin, and 50 µg/mL kanamycin) for 24 h on a shaker at 28 °C and 220 rpm as preculture. Subsequently, 300 mL of selective LB medium was inoculated with this preculture and incubated for a further 24 h under the same conditions. Wild-type Arabidopsis plants were transformed through floral dip as described. 8 Transformed plants were grown under constant light conditions until seed maturation. RoGFP-expressing plant lines were selected through observation of roGFP fluorescence at seedling stage on a fluorescence stereomicroscope (MZ FLIII; Leica, Mannheim, Germany).
Culture and Transient Transfection of Schneider and HeLa Cells
Schneider cells were grown in Schneider’s Drosophila Medium (Invitrogen, Karlsruhe, Germany) supplemented with 10% heat-inactivated fetal calf serum (FCS), 2 mM L-glutamine, 100 U/mL penicillin, and 100 mg/mL streptomycin (Invitrogen). Transient expression was achieved by cotransfection with an actinGal4 plasmid, using CellFectin (Invitrogen) according to the manufacturer’s protocol. HeLa cells 9 were grown in Dulbecco’s modified Eagle’s medium (DMEM) (Gibco, Karlsruhe, Germany) supplemented with 10% heat-inactivated FCS, 2 mM glutamine, 100 U/mL penicillin, and 100 µg/mL streptomycin (Gibco). Cells were transfected using polyethylenimine (PEI) as described. 10 We found the optimal transfection conditions to be 3 µg PEI per 1 µg DNA with change of the transfection medium after 3 h. Twenty-four hours after transfection, cells were seeded into eight-chambered coverglasses (Lab-Tek; Nunc GmbH, Langenselbold, Germany) in full medium without phenol red and grown 24 h until imaging.
Generation and Husbandry of Transgenic Flies
The pUAST-COX8-roGFP2-Grx1 expression construct was injected into an yw strain and recipient flies selected. pUAST-COX8-roGFP2-Grx1 flies were crossed with tubulinGal4 flies. Larval wing discs of biosensor-expressing flies were dissected in phosphate-buffered saline (PBS) and imaged immediately. Flies were raised at 25 °C on a cornmeal–soy flour–malt–beet syrup medium (cornmeal, 80 g/L−1; dry yeast, 25 g/L−1; soy flour, 10 g/L−1; malt, 80 g/L−1; beet syrup, 22 g/L−1; agar, 8 g/L−1; nipagin, 1.5 g/L−1; ethanol, 11.25 g/L−1; and propionic acid, 12.5 g/L−1) supplemented with dry yeast.
Microscopy
Ratiometric imaging of Arabidopsis lines expressing roGFP constructs was performed on an LSM 510 META laser scanning microscope with a 63× C-Apochromat 1.2 NA water immersion lens (Zeiss, Jena, Germany). RoGFP fluorescence was excited at 405 and 488 nm in multitracking mode with line switching. Fluorescence was collected using a band-pass filter at 505 to 530 nm, and mean intensities from four scans were recorded. Time course data were analyzed as described previously. 11 Mitochondria were stained with 500 nM tetramethylrhodamine methyl ester (TMRM) for 20 min. TMRM staining was recorded with excitation at 543 nm and a 560- to 615-nm band-pass emission filter. For imaging of transiently transfected Drosophila Schneider cells, a PerkinElmer Ultra View ERS spinning disk confocal microscope was used, equipped with a PlanApo VC 60×, 1.2 NA water immersion lens (PerkinElmer, Waltham, MA). Fluorescence was excited sequentially with 405-nm and 488-nm laser lines and detected through a 500- to 554-nm band-pass filter on an electron-multiplying charge-coupled device (EM-CCD; C9100-02; Hamamatsu Photonics, Herrsching, Germany). TMRM was visualized by excitation at 568 nm and a 580- to 650-nm emission filter. Freshly dissected larval wing discs were imaged with an LSM 710 ConfoCor 3 equipped with a PlanApo DICII 63×, 1.4 NA oil immersion lens (Zeiss). Fluorescence was excited sequentially at 405 and 488 nm (line by line) and detected at 500 to 530 nm. Mitochondrial targeting was verified with TMRM applied at a concentration of 100 nM. TMRM staining was visualized by excitation at 561 nm and detected with a 580- to 650-nm filter. Data were exported as 16-bit Tiff and processed in ImageJ (National Institutes of Health, Bethesda, MD). HeLa cells were imaged on the Zeiss LSM 710 described above with a stage top incubator that maintained temperature (37 °C) and humidity (100%). The microscope was equipped with an EC Plan-Neofluar 40×/1.30 NA oil objective. The biosensor was excited sequentially frame by frame with the 408- and 488-nm laser line and detection set to 500 to 530 nm. Image processing was performed as previously described. 5
In Vitro Analysis of Recombinant roGFP2-Grx1
Recombinant proteins (Grx1-roGFP2 and roGFP2-Grx1) were expressed in Escherichia coli strain BL21 (Agilent Technologies, Waldbronn, Germany) and purified via immobilized metal ion adsorption chromatography (HiTrap columns; GE Healthcare, Freiburg, Germany). The purified recombinant proteins were desalted and diluted into a standard reaction buffer (100 mM potassium phosphate, 1 mM EDTA [pH 7.0]) to a final concentration of 1 µM. The emission of roGFP2 (505–515 nm) was measured after excitation at 390 and 480 nm in a plate reader (FLUOstar Optima; BMG Labtech, Offenburg, Germany) equipped with built-in injectors. The ratio of the emission at the two respective excitation wavelengths (390/480 nm) was calculated and plotted against time. To ensure full reduction by addition of 2 mM GSH glutathione reductase and 125 mM NADPH were included in the roGFP reduction assays. For oxidation experiments, isolated proteins were first reduced with 20 mM dithiothreitol (DTT) for 20 min on ice and then desalted with Zeba Spin Desalting columns (Thermo Fisher Scientific, Bonn, Germany).
Results
Mito-Grx1-roGFP2 Fails to Enter Fly and Plant Mitochondria
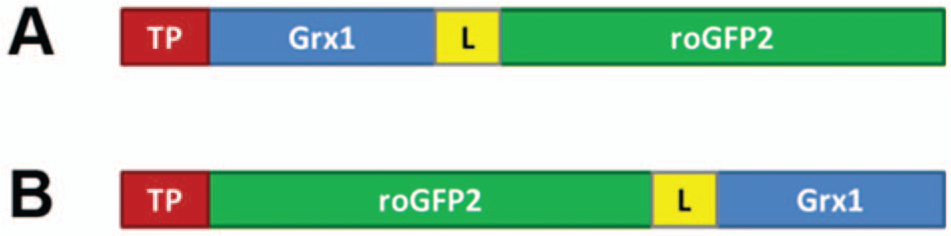
Previously, Grx1-roGFP2 has been expressed successfully in both human and murine mitochondria. However, to our surprise, mitochondrial targeting of Grx1-roGFP2 ( Fig. 1A ) does not occur as expected in various nonmammalian eukaryotes, despite the use of established targeting peptides and multiple transformation events. For example, mito-Grx1-roGFP2 with the targeting peptide of Arabidopsis mitochondrial serine hydroxymethyltransferase (SHMT) accumulates in the cytosol of Arabidopsis cells and does not efficiently enter mitochondria ( Fig. 2A – D ). Likewise, Drosophila Schneider cells with either of two previously established targeting peptides ( Fig. 3B , C ) show an expression pattern indistinguishable from cytosolic Grx1-roGFP2 ( Fig. 3A ). This mislocalization was observed consistently in hundreds of cells from several transformation events. Thus, Grx1-roGFP2 is not properly targeted or imported into mitochondria in at least two widely used nonmammalian model organisms. As mitochondrial targeting of conventional GFPs is well known to succeed in both model organisms,7,12 we reasoned that the presence of the Grx domain between the targeting peptide and the roGFP2 domain ( Fig. 1A ) interferes with either targeting or import.
RoGFP2-Grx1, but not Grx1-roGFP2, can be targeted to the mitochondrial matrix in nonmammalian eukaryotes. (
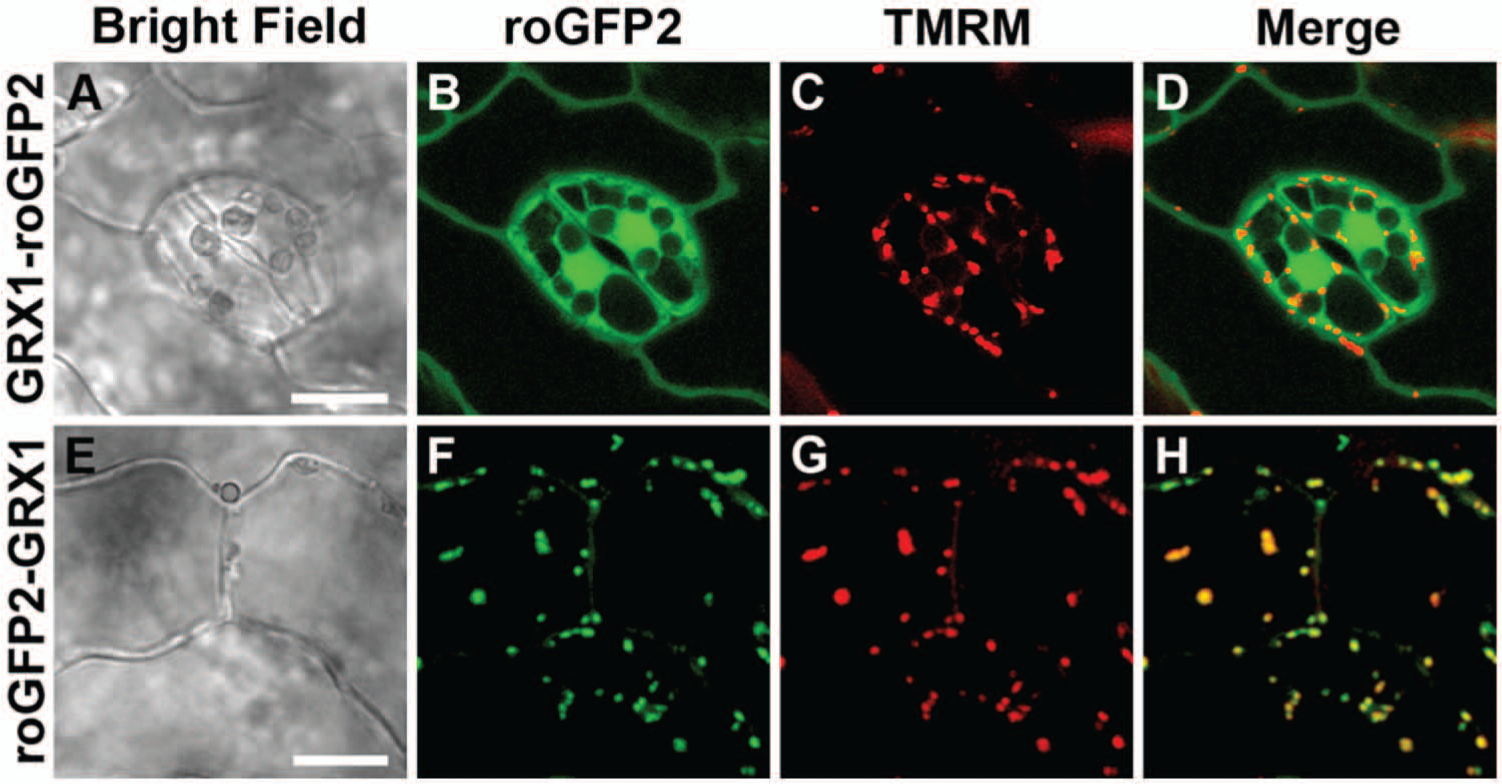
RoGFP2-Grx1, but not Grx1-roGFP2, can be targeted to the mitochondrial matrix in Arabidopsis thaliana. (
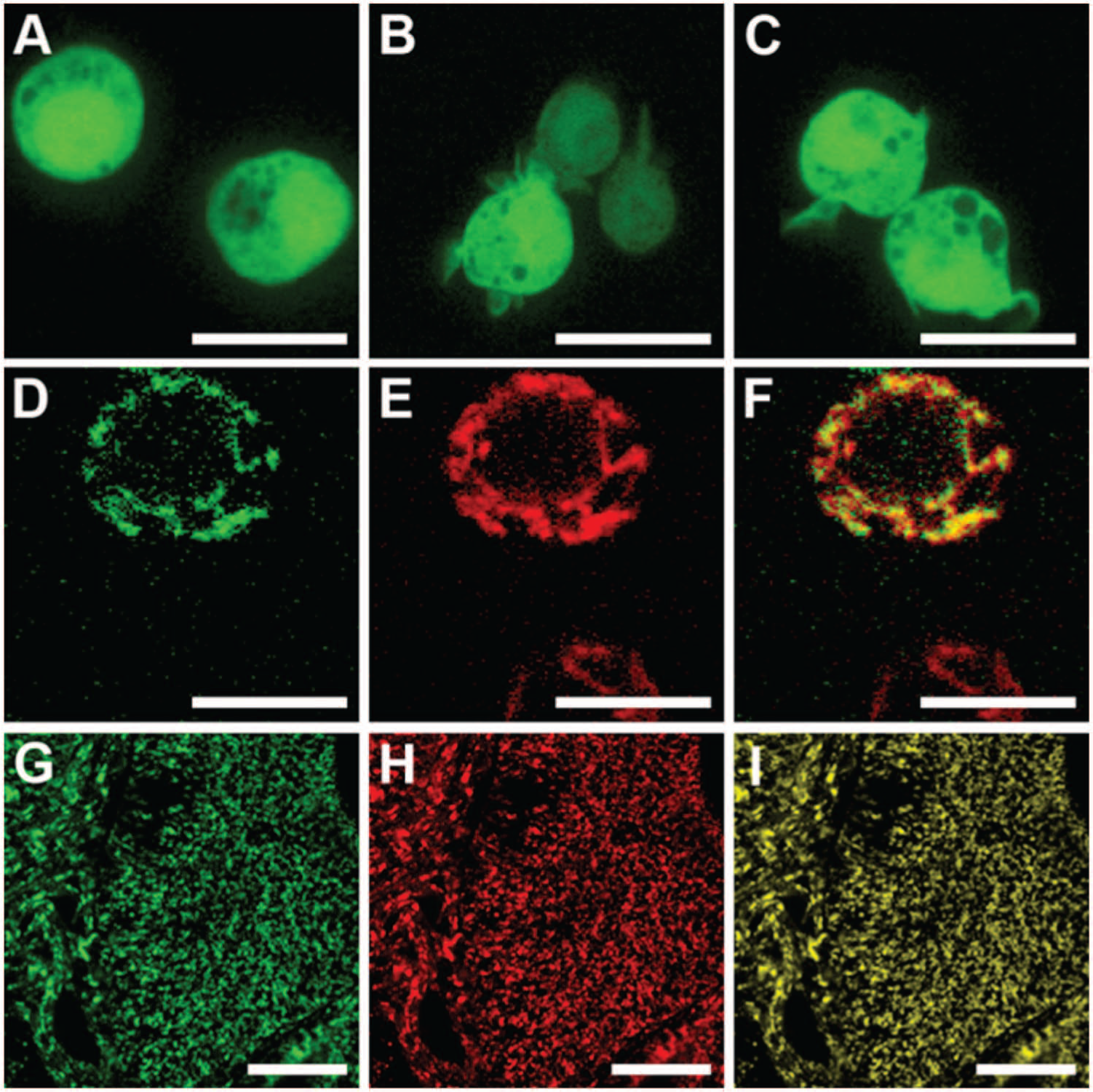
RoGFP2-Grx1, but not Grx1-roGFP2, can be targeted to the mitochondrial matrix in Drosophila melanogaster. (
The Domain-Inverted Biosensor, Mito-roGFP2-Grx1, Is Properly Targeted to Fly and Plant Mitochondria
In an attempt to solve the targeting problem, we therefore relocated the Grx1 domain by translationally fusing it to the C-terminus of roGFP2 ( Fig. 1B ) and asked whether the inverted chimeric construct, mito-roGFP2-Grx1, is properly targeted to plant and fly mitochondria. This approach was successful in all transformation attempts, as shown for transgenic Arabidopsis ( Fig. 2E – H ), Drosophila Schneider cells ( Fig. 3D – F ), and transgenic Drosophila flies ( Fig. 3G – I ). Only in exceptionally rare cases with very high expression of SHMT-roGFP2-Grx1 in Arabidopsis was incomplete mitochondrial targeting with residual fluorescence in the cytosol and the nucleoplasm observed. The successful targeting of the inverted probes naturally led to the question of whether Grx1-roGFP2 and roGFP2-Grx1 are equivalent probes with identical redox properties.
The Live Imaging Response of Grx1-roGFP2 and roGFP2-Grx1 Is Indistinguishable
To compare the intracellular responsiveness of Grx1-roGFP2 and roGFP2-Grx1 in real time, we used HeLa cells as a well-established model environment for genetically encoded redox probes. We expressed both probes in the cytosol and monitored their response to exogenously added hydrogen peroxide by live imaging. The comparison of the response to the oxidant of Grx1-roGFP2 and roGFP2-Grx1 did not show any significant difference ( Fig. 4A , B ). Thus, we concluded that sensitivity and kinetics are well comparable and that the domain-order inversion is not disadvantageous for the responsiveness of the probe.
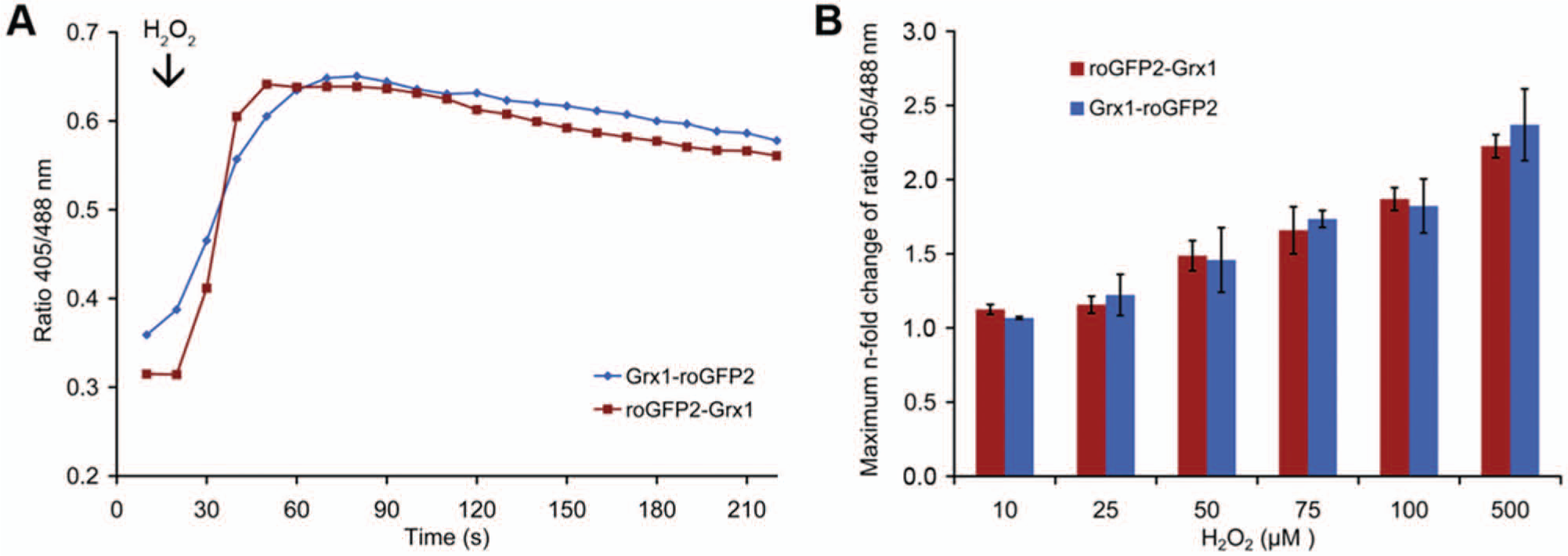
RoGFP2-Grx1 and Grx1-roGFP2 show the same properties in live-cell imaging. (
Having established successful targeting of roGFP2-Grx1 to mitochondria, we further tested the dynamic response of the probe in transgenic Arabidopsis seedlings. The probe responded rapidly to redox changes imposed through perfusion of roots with 1 mM H2O2, reaching almost 100% oxidation ( Fig. 5 ). Subsequent washout of H2O2 caused a rapid reduction of the probe reflected by a decreasing fluorescence ratio. To further test for the maximum response of the novel probe, roots were also perfused with 10 mM H2O2 and 10 mM DTT, respectively. These measurements confirmed that the probe was almost fully reduced in untreated resting mitochondria and that 1 mM H2O2 was sufficient to induce full oxidation.
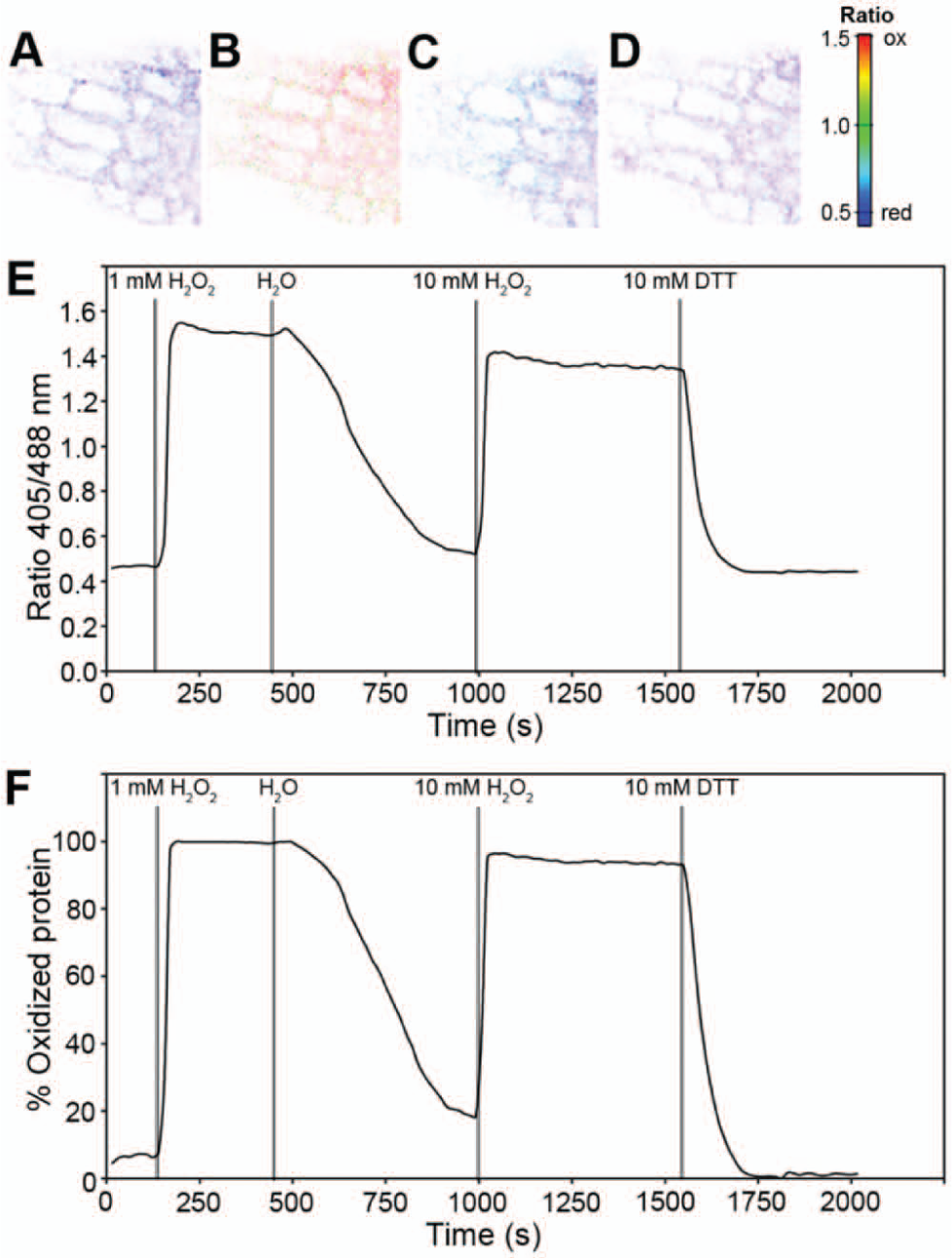
Dynamic response of roGFP2-Grx1 targeted to Arabidopsis mitochondria. (
In Vitro Characterization Indicates Functional Superiority of the Inverted Probe
To obtain a more detailed functional comparison of the two biosensors, the corresponding recombinant proteins were investigated in vitro. The dynamic range of roGFP2-Grx1, defined by full oxidation with 10 mM H2O2 and full reduction with 10 mM DTT, respectively, was about 12-fold and thus only marginally lower than the ~13-fold range of Grx1-roGFP2 ( Fig. 6A , B ). Reduction of the oxidized probes with 2 mM GSH in the presence of glutathione reductase and NADPH, and thus in the absence of residual GSSG, revealed that the reversed domain order leads to a faster probe response ( Fig. 6A , B ). While subsequent oxidation by addition of 200 µM GSSG did not reveal any significant difference between the two probes, more subtle differences became apparent when the prereduced probes were oxidized with lower concentrations of GSSG. Using GSSG concentrations between 0.1 and 10 µM, oxidation of roGFP2-Grx1 was consistently faster than oxidation of Grx1-roGFP2 ( Fig. 6C ). In conclusion, we find that Grx1-roGFP2 can be functionally replaced by roGFP2-Grx1, which may even be superior in terms of its response kinetics. The latter biosensor, because of its superior targetability, thus allows mitochondrial redox measurements in the broadest possible range of model eukaryotes.
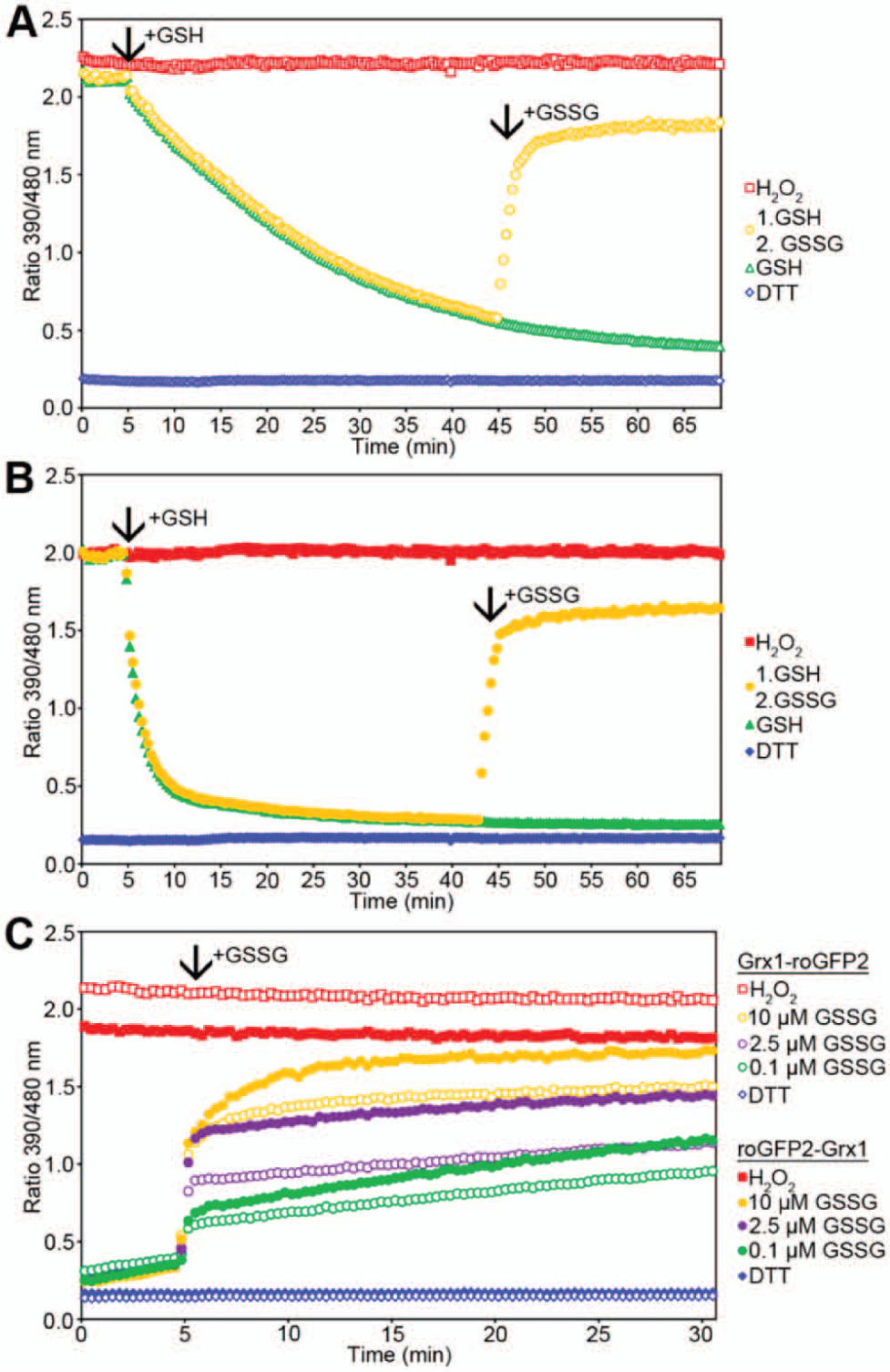
In vitro roGFP2-Grx1 displays faster kinetics for reduction and oxidation than Grx1-roGFP2. (
Discussion
We have found that the two fusion proteins Grx1-roGFP2 and roGFP2-Grx1 differ fundamentally in their ability to be expressed in plant and fly mitochondria. While Grx1-roGFP2 can be expressed in mammalian mitochondria, it does not express in mitochondria of plants and flies, despite the use of established species-specific targeting peptides. Whereas nonfused EGFP can be targeted successfully into fly and plant mitochondria,7,12 the N-terminal fusion of human Grx1 to roGFP2 seems to interfere with the mitochondrial import machinery ( Figs. 2 and 3 ). While similar problems with organellar targeting of GFP-based biosensors, to our knowledge, have not been documented in the literature, it has been discussed previously that differences in two proteomic data sets for yeast mitochondrial proteins generated through expression of GFP fusions and mass spectrometry may result from experimental conditions that affect the ratio between the GFP folding and protein transport kinetics and thus essentially block mitochondrial import. 13 Here we report that fusion of Grx1 to the C-terminus of roGFP2 solves the problem of mitochondrial import in flies and plants. Importantly, we also show that roGFP2-Grx1 is functionally on par with Grx1-roGFP2 and even exhibits improved kinetic response properties. Thus, both probes are interchangeable biosensors for all practical purposes with a possible advantage for the inverted probe for fast, dynamic measurements. This functional equivalence of the two probes is not unexpected for the following reasons: first, other redox probes with C-terminally attached domains are known to be fully functional, including roGFP2-Orp16 and rxYFP-Grx1p. 14 Second, the electron transfer mechanism depends on collisions between the active site of glutaredoxin and the dithiol-disulfide pair on the roGFP2 surface. Given the length and flexibility of the linker domain (30 amino acids), it is easily conceivable that the domain rearrangement does not restrict these collisions. In fact, the slight advantage in the oxidation kinetics of roGFP2-Grx1 that is apparent ( Fig. 6C ) may be explained by the fact that the C-terminus of roGFP2 is closer to its dithiol-disulfide pair, which might allow additional steric flexibility for interdomain collisions in the inverted construct.
The fusion of oxidoreductases to roGFP2 can improve chemical specificity and significantly enhance its kinetic properties. The domain-order inversion (i.e., fusion of Grx1 to the C- instead of the N-terminus of roGFP2) solves the problem of mitochondrial targeting without compromising biosensor function. The redesigned biosensor, roGFP2-Grx1, was successfully targeted to mitochondria in different eukaryotic model species. As a result, mitochondrial redox homeostasis can now be studied in nonmammalian model organisms as it has been shown recently in Drosophila during development and aging. 15 Thus, we generally recommend using roGFP2-Grx1 over Grx1-roGFP2 in experiments aiming at dynamic live-cell imaging in mitochondria. Our findings have important practical relevance for the generation of redox biosensor-transgenic model organisms and for the design of future redox probes.
Footnotes
Acknowledgements
We thank Dr. Bruce Morgan and Dr. Markus Schwarzländer for critical reading of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Deutsche Forschungsgemeinschaft (DFG) to A.J.M. (grant ME1567/6-1), the Baden-Württemberg Foundation to T.P.D., the State of Baden-Württemberg to A.J.M., and a scholarship from the Boehringer Ingelheim Fonds to M.C.S.
