Abstract
Surrogate readouts of G-protein–coupled receptor signaling pathways using highly engineered systems are often employed in the drug discovery process. However, accumulating data have demonstrated the importance of selecting relevant biological activity rather than technically facile assays to support high-throughout screening and subsequent structure-activity relationship studies. Here we report a case study using sphingosine-1-phosphate receptor 1 (S1P1) as the model system to compare compound activity in six different in vitro assays with their ability to predict in vivo efficacy. S1P1 has long been validated as a therapeutic target for autoimmune diseases. In this article, in vivo and in vitro studies on 19 S1P1 agonists are reported. In vitro activities of these S1P1 agonists, together with S1P and FTY720p, on Ca2+ mobilization, adenylyl cyclase inhibition, extracellular signal-related kinase (ERK) phosphorylation, β-arrestin recruitment, and receptor internalization, were determined. The in vitro potency of these compounds was correlated with their ability to induce peripheral lymphocyte reduction. The results revealed that inhibition of adenylyl cyclase and induction of β-arrestin recruitment and receptor internalization are good indicators to predict in vivo efficacy, whereas induction of Ca2+ mobilization through Gqi/5 coupling and ERK phosphorylation is irrelevant. This study demonstrated the importance of identifying an appropriate in vitro assay to predict in vivo activity based on the biological relevance in the drug discovery setting.
Keywords
Introduction
Assay development is a critical step in the drug discovery process. The choice of assays selected for high-throughput screening (HTS) and structure-activity relationship (SAR) activity is often dictated not only by their relevance to the desired effect of the drug on a certain pathway but also by the feasibility, robustness, and cost of the assay. Thus, robust assays such as calcium mobilization through recombinant G-protein coupling have been a common default HTS assay for G-protein–coupled receptor (GPCR) drug discovery for some time. Over the past few years, increasing evidence has demonstrated the importance of understanding pathways preferentially activated or inhibited by biased agonists or antagonists on a particular GPCR,1–3 since they can lead to different biological outcomes. This presents a need to design and develop assays to triage pathway-specific as well as receptor-specific compounds. Therefore, when choosing assays for drug candidate screens and optimization, their biological relevance should outweigh the ease of assay development and execution. In this report, we used sphingosine-1-phosphate receptor 1 (S1P1) as an example to demonstrate the necessity of evaluating various achievable in vitro assays and determine their predictability for in vivo efficacy.
S1P, a product of the ceramide pathway,4,5 is produced mainly by platelets, 6 vascular endothelial cells, 7 or red blood cells. 8 It regulates a wide range of biological functions4,9 such as cardiovascular10,11 and pulmonary function, 12 hearing, 13 and immune cell trafficking, 14 either as an intracellular lipid secondary messenger 15 or as an extracellular ligand. 16 As a ligand, S1P exerts its biological function through a family of five GPCRs 16 to which it binds with high affinity. 14 Among these five GPCRs, S1P1 has been validated as a therapeutic target for multiple sclerosis (MS). FTY720, 17 a pan-S1P receptor agonist5,14 that binds to four of the S1P receptors except for S1P2, has been demonstrated to exert its beneficiary effect by sequestering lymphocytes in MS patients through agonism of the S1P1 receptor.18,19 In addition, several other S1P1 selective agonists have been reported to have similar therapeutic potential.20,21 On the other hand, compelling evidence suggests S1P1 agonists can also exert their function by tightening the stromal portal of the lymph node, therefore restricting lymphocyte efflux. 22 These different opinions of the mechanism underlining the function of existing S1P1 agonists warrant the need for thorough understanding of the relationship of desired in vivo activity and potencies in different in vitro assays. As such, multiple in vitro assays have been employed to interrogate this target; these include GTP binding,11,14,23 Ca2+ mobilization,11,14 adenylyl cyclase inhibition, 15 extracellular signal-related kinase (ERK) phosphorylation, 23 β-arrestin recruitment, 24 and receptor internalization. 23 It is intriguing to understand whether these assays have similar power to predict the in vivo impact on peripheral lymphocyte sequestration in rats. In this study, we profiled 19 S1P1 compounds ( Table 1 ) from three different chemical classes for their ability to affect adenylyl cyclase inhibition, Ca2+ mobilization, pERK generation, β-arrestin recruitment, and receptor internalization. The potency from each in vitro assay was correlated with their potency on in vivo peripheral lymphocyte reduction. The result demonstrated Ca2+ mobilization and ERK phosphorylation had little predictability of the in vivo activity. Thus, they should not be chosen for an HTS or SAR study, even though Ca2+ mobilization was one of the most facile and robust assays we developed. On the other hand, adenylyl cyclase inhibition, β-arrestin recruitment, and receptor internalization showed good correlation with in vivo activity. Therefore, β-arrestin recruitment and receptor internalization were employed for the HTS and SAR study, respectively. Even though there were overlapping hits between HTS campaigns using Ca2+ mobilization versus β-arrestin recruitment (PathHunter; DiscoveRx, Fremont, CA), compounds with better potency on Ca2+ mobilization did not necessarily show increased potency in the in vivo study. Our systematic approach demonstrated the necessity of determining the predictability of highly engineered in vitro assays for in vivo impact by developing a set of assays targeting different signaling pathways and identification of a small set of tool compounds with wide spectrum of in vitro activity in these assays prior to the launch of HTS. The effort spent on carefully selecting the best assay by balancing biological effect relevance, robustness, throughput, and cost for HTS and SAR can be rewarded by confidently rank ordering the compounds using in vitro assay results and significantly reducing the requirement for the labor-intensive and costly in vivo assays.
Structure of Test Compounds.
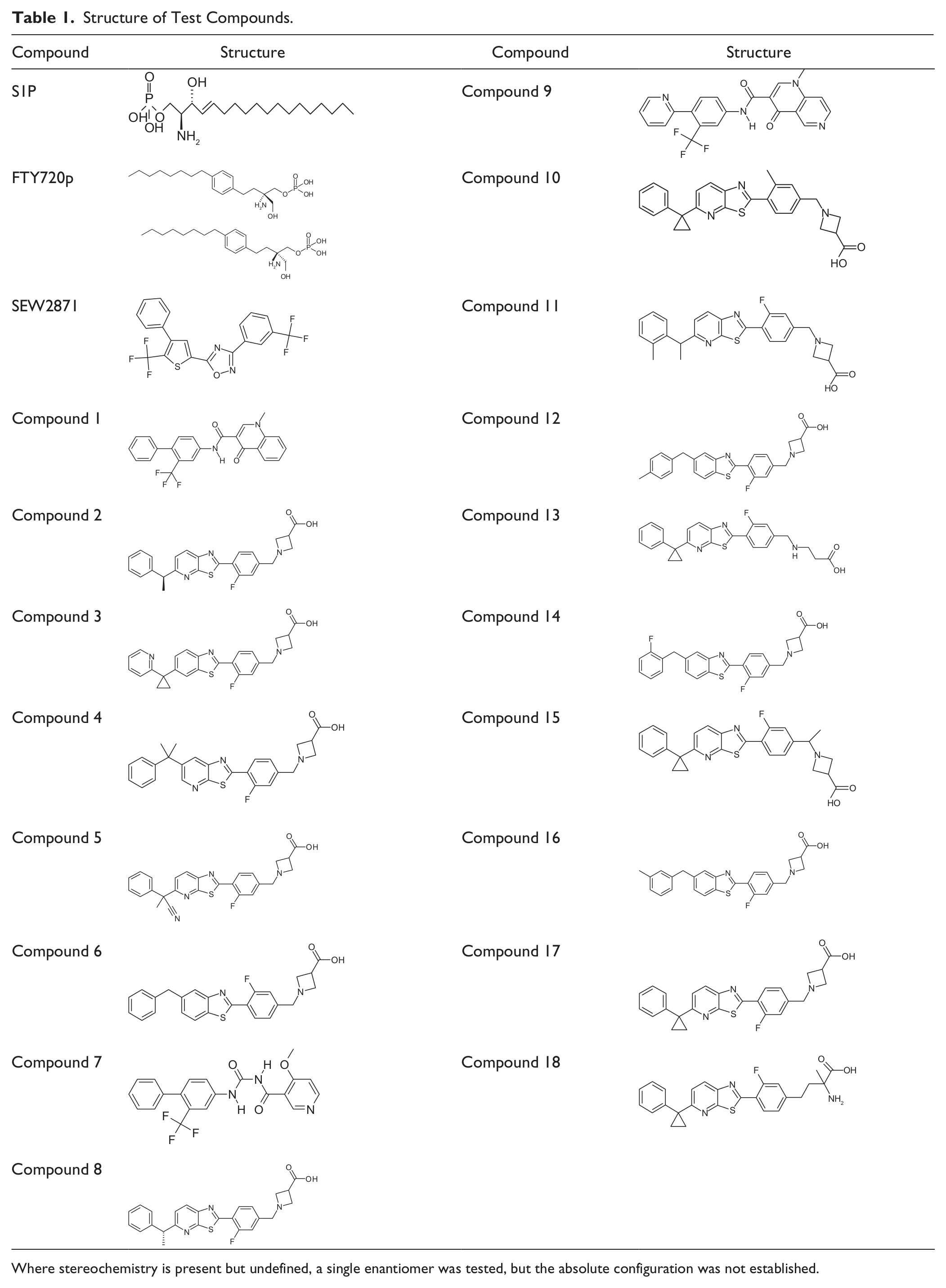
Where stereochemistry is present but undefined, a single enantiomer was tested, but the absolute configuration was not established.
Materials and Methods
S1P Receptor Agonists and Other Reagents
S1P was purchased from EMD Chemicals (Gibbstown, NJ). SEW2871 and forskolin were purchased from Sigma (St. Louis, MO). FTY720p and compounds 1 to 18 were synthesized in house as described previously.25–31 Pertussis toxin was purchased from List Biological Laboratories (Campbell, CA). Calcium indicator was purchased from BD Biosciences (Franklin Lakes, NJ). Hoechst was purchased from Invitrogen (Carlsbad, CA). LANCE cAMP detection kit, SureFire phosphor-ERK, and total ERK detection kits were purchased from PerkinElmer (Waltham, MA).
Recombinant Cell Lines and Plasmids
Human S1P1 (GenBank ID: 1901) was cloned into the expression vector pCDNA3.1 (Invitrogen) and stably expressed in CHO-K1 cells coexpressing a chimeric G-protein Gq/i5 (Molecular Devices, Mountain View, CA). This construct was also stably expressed in Jurkat cells. Human S1P1-PK was constructed in pCMV-ProLink vector and stably expressed in the β-arrestin PathHunter CHO cell line (DiscoveRx). U2OS cells stably expressing an S1P1 receptor–enhanced green fluorescent protein (eGFP) fusion protein were purchased from BioImage (Soeborg, Denmark). These cell lines were maintained in their growth medium containing 100 U penicillin, 1 µg/mL streptomycin, and 2 mM L-glutamine, if not otherwise specified.
Ca2+ Mobilization Assay
CHO-K1 cells coexpressing Gq/i5 and human S1P1 were maintained in the cell growth medium containing F-12/Ham’s, 10% fetal bovine serum (FBS), 500 µg/mL Geneticin, and 300 µg/mL hygromycin B. CHO-K1 cells only expressing Gq/i5 were maintained in the same medium without Geneticin. Cells were seeded at 25,000 cells/well in a 384-well plate in the same medium containing 10% charcoal/dextran-treated FBS replacing FBS with or without 100 ng/mL pertussis toxin and incubated overnight at 37 °C. Cells were then incubated with Ca2+ indicator in Hank’s balanced salt solution (HBSS) buffer containing 20 mM HEPES and 0.1% bovine serum albumin (BSA). Change in intracellular fluorescence intensity due to Ca2+ mobilization was monitored using FLIPRtetra (Molecular Devices) by addition of serially diluted compound (concentrations ranging from 0.3 nM–25 µM) onto calcium indicator–loaded cells, and the plate was imaged for 90 seconds. S1P at 200 nM was used as the positive control to calculate percentage of control (POC).
Cyclic AMP Assay
Parental and S1P1-expressing Jurkat cells were maintained in the medium containing RPMI1640, 10% charcoal/dextran-treated FBS, and 600 µg/mL hygromycin. Parental Jurkat cells were maintained in the same medium without antibiotics. When needed, cells were treated with 100 ng/mL pertussis toxin as described above. Then, 10,000 cells/well in HBSS buffer containing 5 mM HEPES and 0.1% BSA were dispensed into a 384-well plate and incubated with serially diluted compounds in the presence of 25 µM forskolin at room temperature for 30 min. Intracellular cAMP was quantified using the LANCE cAMP kit (PerkinElmer) according to the instructions. Cyclic AMP concentration-dependent fluorescence signal change was recorded using EnVision (PerkinElmer). S1P at 400 pM was used as the positive control. For assays using parental Jurkat cells or Jurkat cells treated with pertussis toxin, the signal from cells treated with 25 µM forskolin was used as the positive control, and the signal from cells without forskolin treatment was used as the negative control.
Phospho-ERK and Total ERK Assay
Parental and S1P1-expressing Jurkat cells were incubated in media containing 10% charcoal/dextran-treated FBS in high-glucose Dulbecco’s modified Eagle’s medium (DMEM) at 4 million cells/mL with or without 100 ng/mL pertussis toxin at 37 °C overnight. For kinetic studies, compounds at the indicated concentration (S1P at 1 µM; FTY720p at 1 µM; SEW2871 and compounds 2, 3, and 4 at 25 µM; and compounds at 2.5 µM) were incubated with 100,000 cells/well in a 384-well plate for 5, 10, 15, 30, 45, and 60 min. The amount of pERK or total ERK was quantified using SureFire AlphaScreen pERK or total ERK kits (PerkinElmer) according to the instructions. AlphaScreen signal was recorded using an EnVision spectrometer. Cells without compound treatment were used as time 0. For compound impact, serially diluted compounds were incubated with 100,000 cells/well at 37 °C for 5 min. The amount of pERK generated was quantified in the same way as in the kinetic measurement. In this assay, 1 µM S1P was used as the positive control to calculate POC.
Receptor Internalization
U2OS cells expressing human S1P1-eGFP fusion protein were maintained in the medium containing DMEM high glucose, 10% FBS, and 500 µg/mL Geneticin. Eight thousand cells/well were seeded in PerkinElmer 96-well View plates in the same medium containing 1% FBS. Following overnight incubation at 37 °C, cells were further starved in the same medium containing 0.1% fatty acid free BSA without FBS at 37 °C for 2 h. Cells were then incubated with serially diluted compounds at 37 °C for 1 h. After compound treatment, cells were fixed and nuclei stained using 1 µg/mL Hoechst in phosphate-buffered saline (PBS) and imaged using the ArrayScan VTI (Cellomics, Pittsburgh, PA). The degree of S1P1 receptor internalization was estimated by counting the number of fluorescent endosome spots formed per cell using the Spot detector algorithm in the ArrayScan software package. Spot count per cell (spotcountperobject) was calculated as output and plotted as a function of compound concentration. POC was calculated using 1 µM of an Amgen (Thousand Oaks, CA) internal S1P1 compound, 1-(4-(6-benzylbenzofuran-2-yl)-3-fluorobenzyl)azetidine-3-carboxylic acid, 26 as the positive control. This molecule yielded a similar but more consistent level of S1P1 internalization than S1P (data not shown).
Alternatively, a decrease of S1P1 receptor on the cell surface was determined in S1P1-expressing Jurkat cells by flow cytometry using an antibody against the V5-tag expressed at the N-terminus of the S1P1 receptor. Cells were treated with serially diluted compounds at 37 °C for 16 h and then stained with 15 µg/mL anti–V5 IgG2a antibody (Life Technologies, Carlsbad, CA) on ice, followed by 5 µg/mL Alexa Fluor 488 goat anti–mouse IgG2a antibody and 7-AAD staining in Dulbecco’s PBS (DPBS) buffer containing 3% charcoal/dextran-treated FBS and 0.1% sodium azide. Fluorescence intensity was measured using the LSRII FACS machine (Becton Dickinson, Franklin Lakes, NJ). A previously published compound, 1-(3-methyl-4-((4-pheyl-5-(trifluoromethyl)thiophen-2-yl)methoxy)benzyl)zaetidine-3-carboxylic acid, 32 at 100 nM was used as the low control (internalized S1P1), and the value of DMSO only was used as the high control (S1P1 on surface). Internalization results were captured as the geometric mean of the mean fluorescence intensity.
β-Arrestin Recruitment Assay
Human S1P1 PathHunter (DiscoveRx) cells were maintained in the medium containing F-12/Ham’s, 10% FBS, 600 µg/mL Geneticin, and 300 µg/mL hygromycin B. In total, 10,000 cells were seeded in each well of a 384-well plate in the medium containing 10% charcoal/dextran-treated FBS in F-12/Ham’s medium and incubated at 37 °C overnight. Cell medium was replaced with compound serially diluted in the assay buffer containing 0.1% BSA in F-12/DMEM, and the mixture was incubated at 37 °C for 90 min. Then, the PathHunter assay was performed according to the instructions, and luminescence signal was measured in Leadseeker (General Electric, Fairfield, CT). In this assay, 200 nM S1P was used as positive control to calculate POC.
Peripheral Lymphocyte Reduction Assay and Pharmacokinetics Determination
Peripheral lymphocyte reduction measurements were conducted in female Lewis rats (n = 5/group; 175–200 g). Compounds or vehicle (20% hydroxypropyl β-cyclodextrin, 1% hydroxypropyl methylcellulose, and 1% Pluronic F68) were dosed orally, and 24 h later, blood samples were collected via cardiac puncture after rats were euthanized via CO2 asphyxiation or anesthetized under isoflurane. Blood samples were used for complete blood count analysis to delineate the number of lymphocytes (Advia Hematology System; Bayer Corp., Tarrytown, NY). Plasma concentration of each test compound was determined by liquid chromatography/tandem mass spectrometry.
Data Analysis and Correlation Study of In Vitro and In Vivo Assay Results
In each in vitro assay, primary results were normalized against the positive and the negative controls, as indicated in the Materials and Methods section, to calculate POC value of each test compound at a given concentration. POC values were then fitted by a four-parameter logistic nonlinear regression model to calculate EC50. Two-tailed Spearman rank-order correlation was employed to assess the monotonic relationship between the in vivo and in vitro activities. Two-tailed Pearson correlation was employed to evaluate the linear correlation between in vivo and in vitro activities.
Results
In Vitro Assessment of Downstream Signaling Pathways Mediated by S1P1 Agonists
Activation of Gαi-coupled GPCR S1P1 triggers multiple cell signaling pathways. Upon receptor activation, Gαi dissociates from the trimeric G-protein and interacts with adenylyl cyclase and, in turn, inhibits its function. This activity can be monitored by measuring cAMP level in the presence of forskolin upon agonist treatment. Concomitantly, Gβγ activates the mitogen-activated protein kinase signaling cascade, including ERK phosphorylation. In addition, activity on Ca2+ mobilization is often used as surrogate readout for Gαi-coupled receptors by coexpressing the receptor and a chimeric G-protein such as Gqi/5. Following receptor activation, attenuation of the signal is quickly initiated through receptor internalization by receptor phosphorylation and β-arrestin recruitment. Since receptor downregulation has been postulated to be the mechanism by which S1P1 agonism affects lymphocyte egress from the lymph nodes, β-arrestin activation and S1P1 receptor internalization were also evaluated.
We established assays to study G-protein coupling and β-arrestin binding/receptor internalization using S1P, FTY720p, and SEW 2871 (
Figs. 1
and
2
). Inhibition of adenylyl cyclase and stimulation of ERK phosphorylation activities were monitored in Jurkat cells expressing recombinant hS1P1 receptor. Ca2+ mobilization activity was monitored in CHO-K1 cells coexpressing hS1P1 and Gqi/5. Receptor-specific signaling was demonstrated by the absence of signal on parental cells and receptor-expressing cells in the presence of pertussis toxin (PTX) for Ca2+ mobilization (
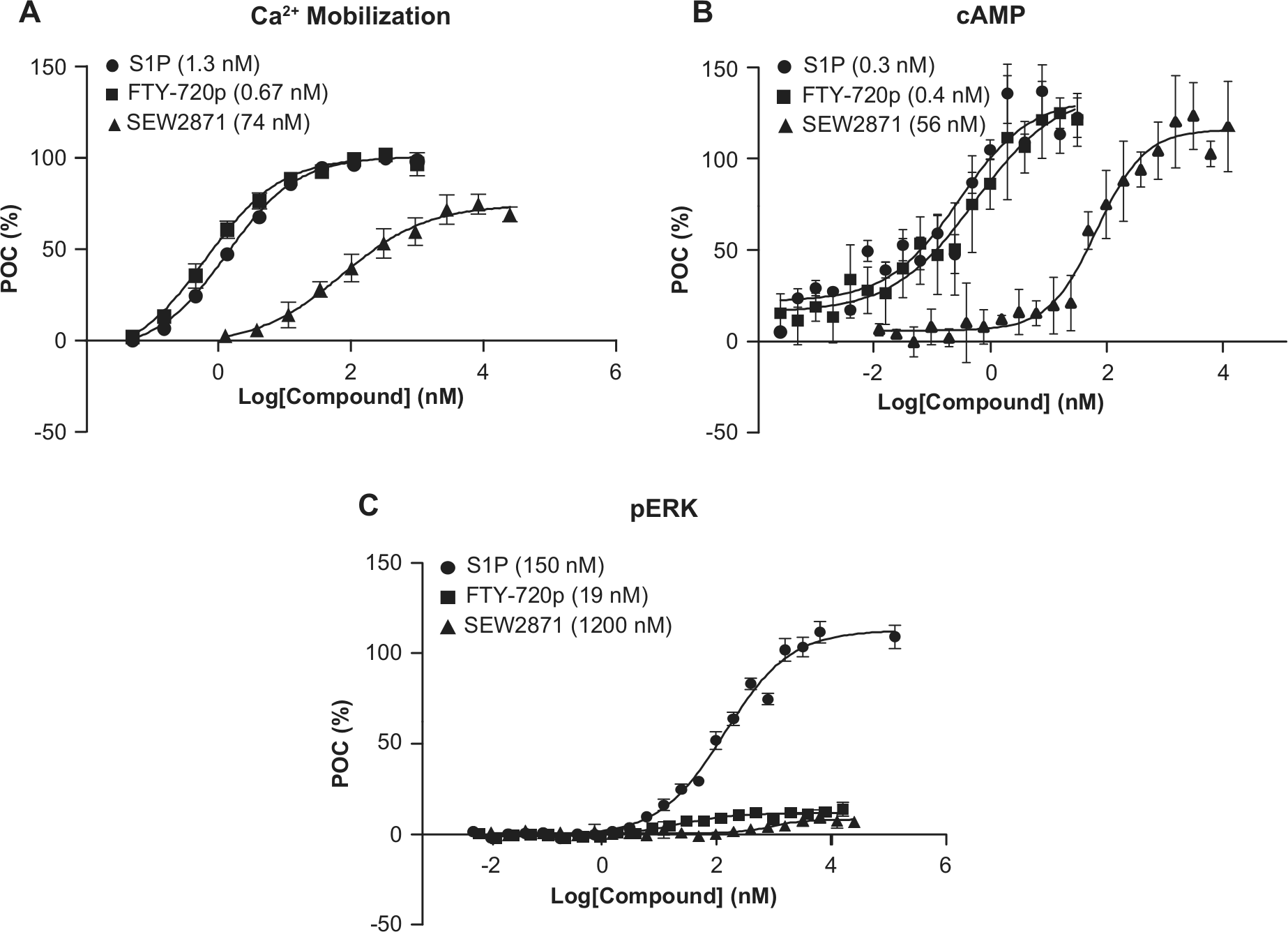
Assays assessing G-protein–coupled signaling were validated by the activity of sphingosine-1-phosphate (S1P), FTY720p, and SEW2871 inducing Ca2+ mobilization, cAMP accumulation, and extracellular signal-related kinase (ERK) phosphorylation. (
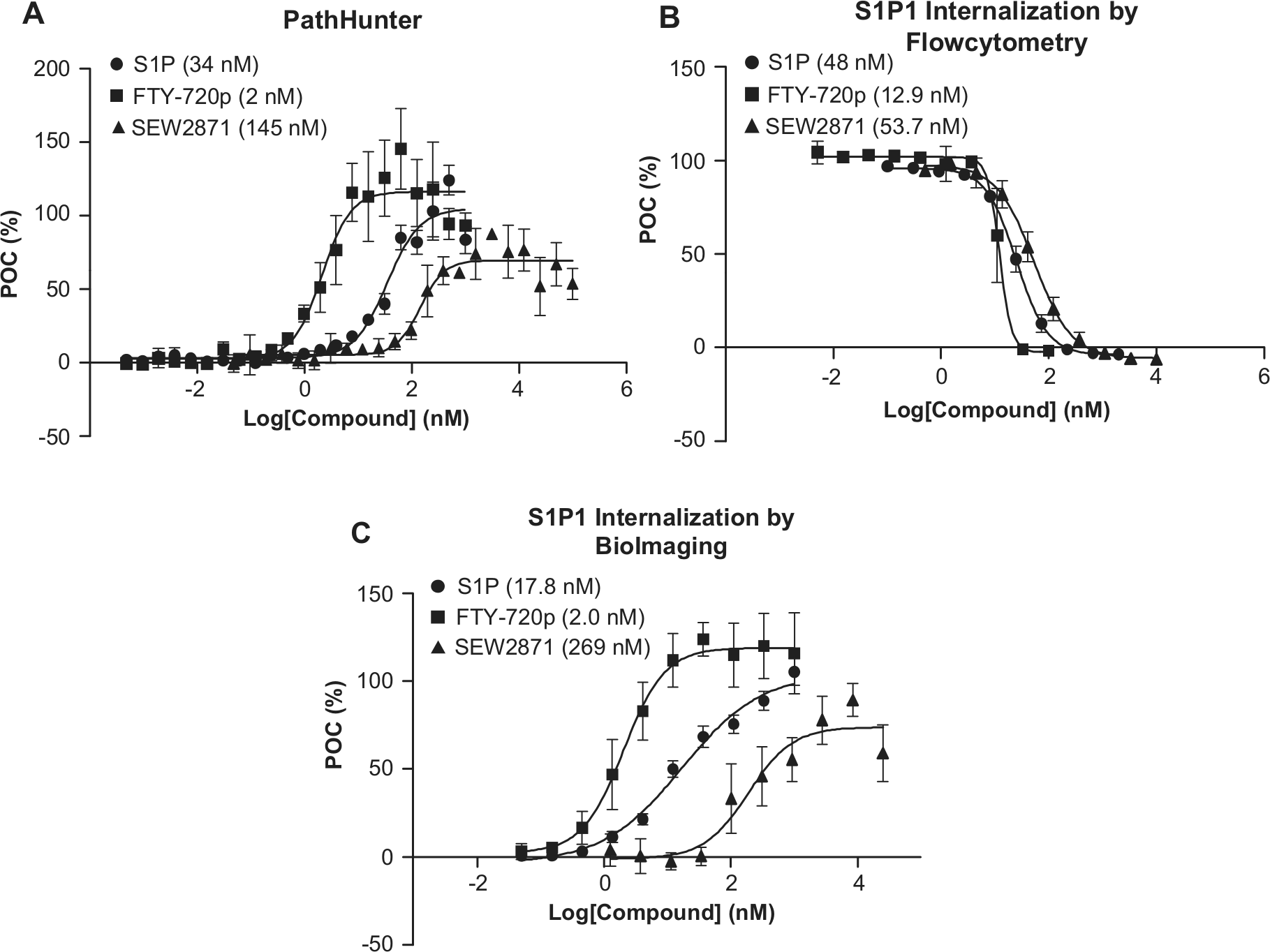
β-Arrestin binding and sphingosine-1-phosphate receptor 1 (S1P1) receptor internalization assays were validated using S1P, FTY720p, and SEW2871. (
Potencies of S1P, FTY720p, and SEW2871 in these in vitro assays are consistent with published results, if available.11,14 There are a few noteworthy points: we delineated, through time course analysis, that our compounds affected rapid ERK phosphorylation. All test compounds caused transient ERK phosphorylation, which peaked at ~5 min, followed by a decline to baseline around 30 min (
pEC50s of Test Compounds in All Assays.
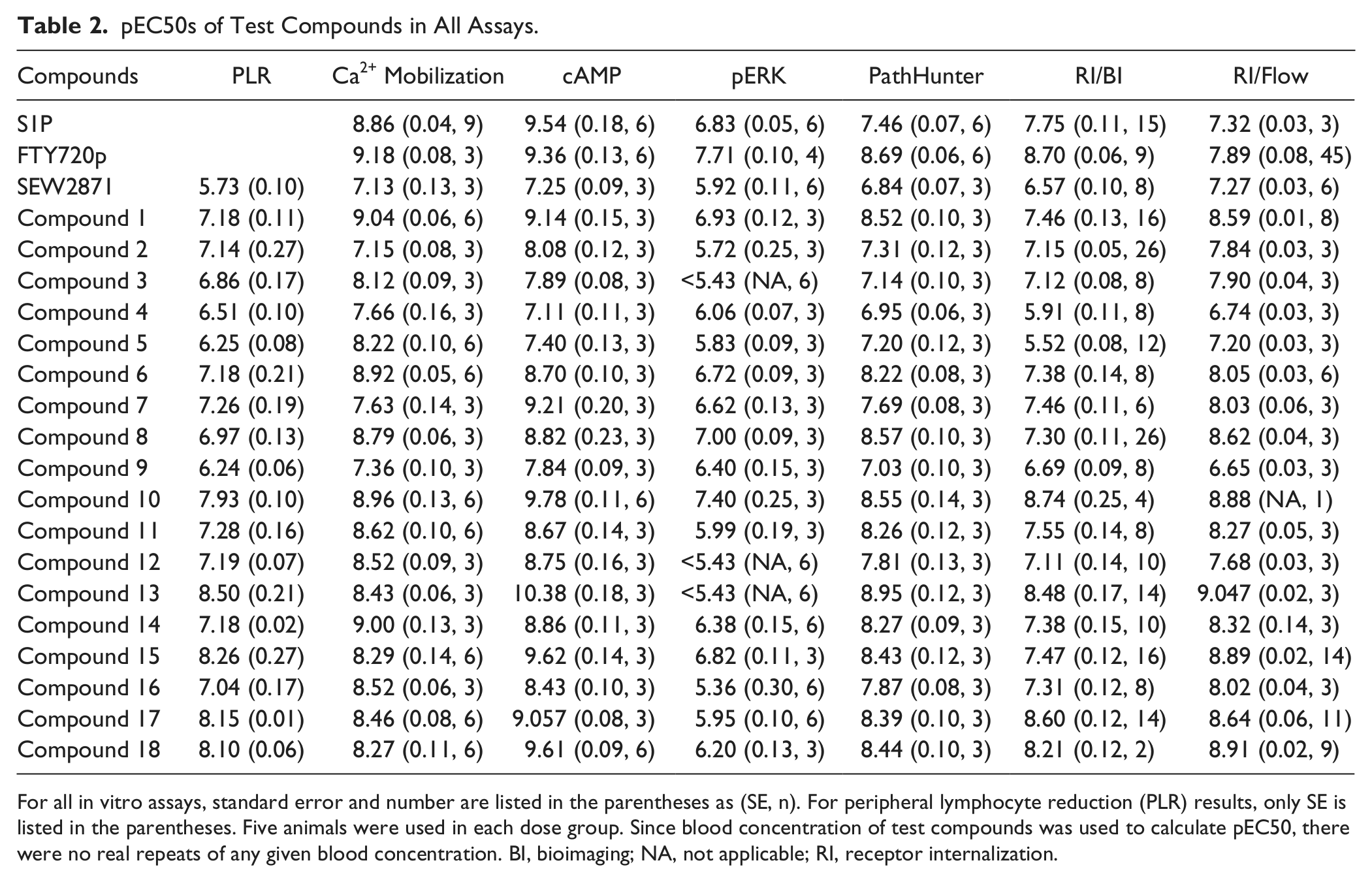
For all in vitro assays, standard error and number are listed in the parentheses as (SE, n). For peripheral lymphocyte reduction (PLR) results, only SE is listed in the parentheses. Five animals were used in each dose group. Since blood concentration of test compounds was used to calculate pEC50, there were no real repeats of any given blood concentration. BI, bioimaging; NA, not applicable; RI, receptor internalization.
Correlation of S1P1-Mediated G-Protein–Dependent Signaling and β-Arrestin–Dependent Receptor Internalization Activities vs. PLR
It is believed that sequestration of naive lymphocytes in the secondary lymph nodes due to downregulation of the S1P1 receptor11,14 is a major contributor to the efficacy of FTY720 in preclinical autoimmune models as well as in MS in humans. Peripheral lymphocyte depletion seen in conjunction with efficacy in preclinical models as well as in MS patients indicates that reduction in circulating peripheral lymphocytes can be used as a pharmacodynamic measure of impact and a surrogate for efficacy. Compounds 1 to 18 and SEW2871 were tested in a peripheral lymphocyte reduction (PLR) assay as described in the Materials and Methods. All compounds demonstrated linear blood exposure (data not shown). Lymphocyte count of each test animal taken 24 h posttreatment was plotted against its corresponding total plasma compound concentration, taken at the same time, and a four-parameter model was used to calculate the pEC50 value of each compound ( Table 2 ).
We examined the correlation of in vivo potency of these compounds against their in vitro activity on S1P1 G-protein– and β-arrestin–dependent activities. Although the faction unbound in rat plasma has been determined by ultracentrifugation for most of the compounds (
Correlation of pEC50s of an In Vivo PLR Assay and Six Different In Vitro Assays Analyzed by Two-Tailed Pearson and Spearman Correlation Analysis.
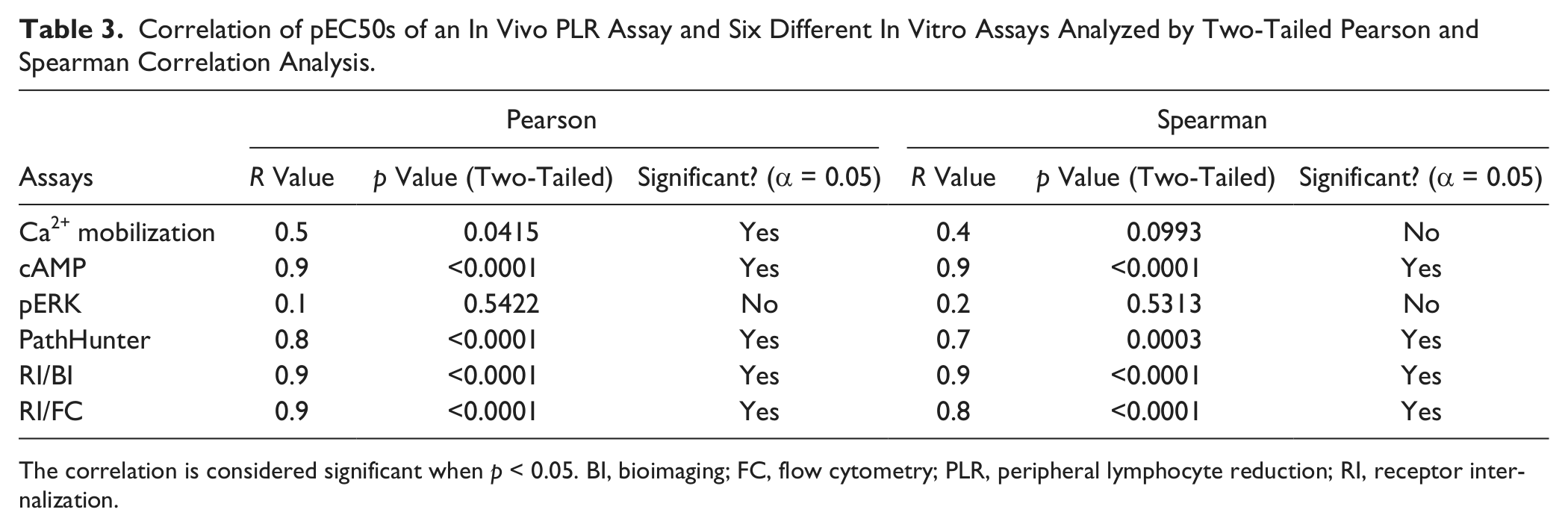
The correlation is considered significant when p < 0.05. BI, bioimaging; FC, flow cytometry; PLR, peripheral lymphocyte reduction; RI, receptor internalization.
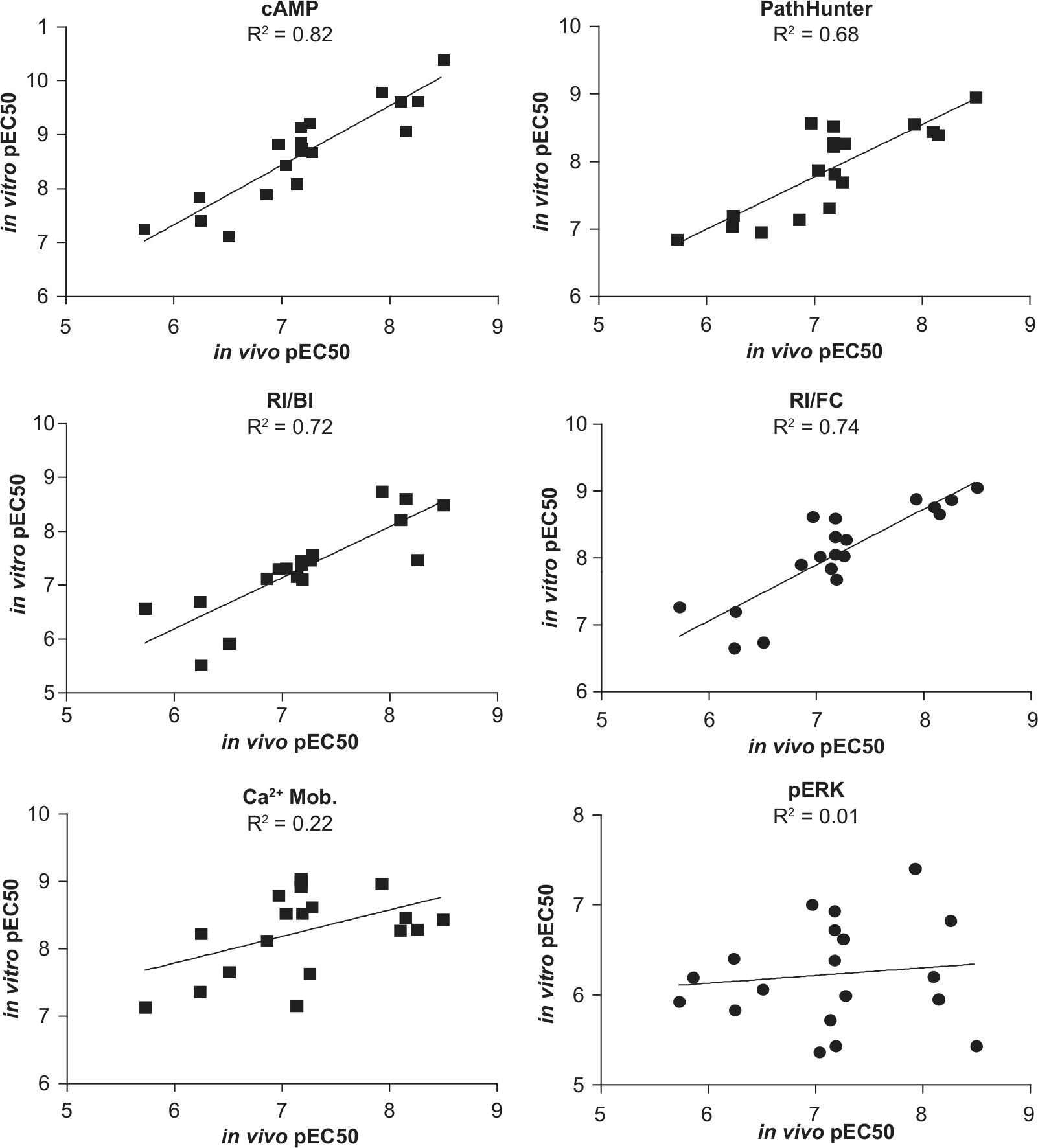
Linear fit of pEC50 of in vitro assays vs. in vivo peripheral lymphocyte reduction. Activities from cAMP, PathHunter, and two receptor internalization assays showed reasonable linear correlation to the in vivo activity. However, Ca2+ mobilization and extracellular signal-related kinase (ERK) phosphorylation showed poor linear correlation, and therefore these two assays should not be used to predict in vivo activity. BI, bioimaging; FC, flow cytometry; RI, receptor internalization.
Discussion
The approval of FTY720 for first-line MS treatment has demonstrated that affecting lymphocyte trafficking by S1P1 agonism is a valid approach to modulate autoimmune diseases. The binding of FTY720p to S1P1 on lymphocytes within lymph nodes results in receptor downregulation and internalization. It is believed that downregulation of this receptor results in the inability of the lymphocytes to sense the S1P gradient that is important for egress from the lymph nodes. Although FTY720p is an agonist of several S1P GPCRs, it has been shown that an S1P1-specific agonist, SEW2871, also induced peripheral lymphocyte sequestion in rodents, 11 demonstrating that agonism of S1P1 alone is sufficient for peripheral lymphocyte reduction. A significant amount of effort has been invested to identify efficacious S1P1-specific candidates across the drug discovery industry. In our quest to identify S1P1-selective agonists, we established six different in vitro assays to measure compound activity on human S1P1 receptor. In addition, a PLR assay was established in rat for compound prioritization and selection for further advancement. We report the correlation between in vivo impact and in vitro potency of 19 S1P1 agonists representing three chemical classes, including SEW2871, across several assays established to measure different downstream events after receptor binding. Ideally, the correlation should be performed in single species for both in vivo and in vitro assays; however, high homology between human and rat S1P1 (92.8% identity), as well as similar efficacy exhibited by FTY720p in human patients and rat models,17,35 boosts the confidence that correlation between in vivo activity in rat and in vitro activity on cells expressing human receptor should reasonably reflect the correlation of in vivo and in vitro activity in a single species.
It has been demonstrated that S1P1 is the major receptor mediating the peripheral lymphocyte reduction effect in vivo. 23 Even though a small set of compounds representing only three chemical classes was included in this study, our correlation analysis indicates that receptor coupling to different signaling pathways is not equal since our small molecules appear to differentiate among them, as shown by the differences in rank order potency in some of these assays. The in vitro assays that best predicted in vivo pharmacodynamic effect were adenylyl cyclase inhibition, the two receptor internalization assays, and β-arrestin recruitment. ERK phosphorylation and Ca2+ mobilization measurements were poorly correlating and would not be good choices for optimizing efficacy in the S1P1 program. Caution must be made in extrapolating this to the signaling systems used in primary cells since these assays were developed to provide robust, reproducible reads by receptor overexpression or use of chimeric G-proteins. However, it is interesting that the cAMP assay was most correlative and was performed in the same cell line as ERK phosphorylation, which was least correlative. This demonstrates that subtleties in the small-molecule binding can lead to different functional consequences. Another interesting observation is that the actual EC50 values in the PathHunter β-arrestin binding assay as well as two different receptor internalization assays were in the same range as the EC50 value of the PLR assay while there was a potency shift from the in vitro cAMP assay to the in vivo assay. Even though the rank order of inhibitory activity of cAMP production tracks well with in vivo potency, the in vivo potencies were almost 100-fold weaker than inhibition of adenylyl cyclase for all compounds. One possible explanation is that inhibition of cAMP production is required but not sufficient to induce PLR, and indeed activities along β-arrestin binding and receptor internalization pathway were critical for in vivo activity.
In GPCR drug discovery, surrogate readout using nonendogenous coupling through chimeric or overexpressed G-protein is commonly employed, especially for Gαι-coupled receptors. We demonstrated that compound activity using surrogate readout such as Ca2+ mobilization can diverge significantly from its activity by native G-protein coupling. Furthermore, we showed that agonists of the same receptor, such as S1P1, may not be identical in the way they induce receptor coupling to activate different downstream pathways, resulting in distinct rank order and intrinsic potency among different signaling pathways. Although the availability of small molecules to test the predictability of the in vitro assays to an in vivo pharmacodynamic effect is rare, this article highlights the importance of monitoring the relationship between in vitro assays depicting different signaling pathways and the in vivo biological effect as soon as a reasonable set of tool compounds becomes available.
Footnotes
Acknowledgements
We thank Amgen scientists Laura Scott for S1P1 HTS using the PathHunter assay; David Brankow, Marrisa van der Valk, and Dr. Sylvia Hu for molecular cloning and cell line generation; and Dr. Michael Eschenberg and Jessie Gu for critical review on statistical analysis. We also thank Dr. Allan Atkinson from PerkinElmer for assistance on the ERK phosphorylation assay.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
