Abstract
Testing small molecules for their ability to modify cysteine residues of proteins in the early stages of drug discovery is expected to accelerate our ability to develop more selective drugs with lesser side effects. In addition, this approach also enables the rapid evaluation of the mode of binding of new drug candidates with respect to thiol reactivity and metabolism by glutathione. Herein, we describe the development of a fluorescence-based high-throughput assay that allows the identification of thiol-reactive compounds. A thiol-containing fluorescent probe, MSTI, was synthesized and used to evaluate small molecules from the Library of Pharmacologically Active Compounds (LOPAC) collection of bioactive molecules. LOPAC compounds that are known to react with sulfur nucleophiles were identified with this assay, for example, irreversible protease inhibitors, nitric oxide–releasing compounds, and proton-pump inhibitors. The results confirm that both electrophilic and redox reactive compounds can be quickly identified in a high-throughput manner, enabling the assessment of screening libraries with respect to thiol-reactive compounds.
Keywords
Introduction
Confirmation of activity and selectivity of hit molecules identified by high-throughput screening (HTS) is an essential part of drug discovery. Especially for inhibitor screens, this often results in hundreds to thousands of hit molecules. The characterization of these molecules by secondary screens, which are not always amendable to a higher throughput format, leads to a bottleneck in the discovery pipeline. Frequently, these hit molecule selections contain a large number of promiscuous inhibitors that have a very poor outcome in the following steps of drug discovery. Multiple underlying nonspecific mechanisms have been identified for these inhibitors, such as aggregation, 1 redox activity, 2 protein modification, 3 and compound interference with the assay signal. 4 High-throughput assays have been developed to detect compound aggregation 5 and redox active compounds. 6 The application of alternative detection methods such as different fluorescent dyes or luminescence can be used as a secondary assay to rule out false-positive HTS hits based on signal interference. Finally, cheminformatics with promiscuity/reactive functionality filters are often applied to identify reactive molecules among hit compounds.7–12 Importantly, some Food and Drug Administration (FDA)–approved drugs would be filtered out by these filters such as irreversible H+, K+-ATPases, inhibitors for duodenal and gastric ulcers.
Currently, HTS assays that determine the electrophilic properties of small molecules—and thus the ability to react with naturally occurring thiols—have yet to be fully developed. One of the few approaches is a competitive binding assay using glutathione and fluorescein-5-maleimide. 13 Although, this assay could be adapted to high throughput, it does not allow for the differentiation between electrophilic compounds that react with glutathione and nucleophilic compounds that react with fluorescein-5-maleimide. In addition, many screening compounds interfere with the yellow/green fluorescence detection at 480/520 nm. 4 More recently, another lower throughput method has been developed using heteronuclear single quantum coherence (HSQC) nuclear magnetic resonance (NMR) by monitoring the 13C shift of small molecules that bind to the thiols in a La antigen protein. 14 As such, there is still a great need for a simple high-throughput method that can accurately assess the thiol-binding abilities of small molecules.
Herein we present the development of a fluorescence-based (E)-2-(4-mercaptostyryl)-1,3,3-trimethyl-3H-indol-1-ium (MSTI) assay that enables the identification of thiol-reactive small molecules in a high-throughput manner. In contrast to the very low-throughput detection of small molecule–glutathione adducts using high-performance liquid chromatography (HPLC), we have developed a nucleophilic fluorescent probe, with discreet fluorescence at 510/650 nm. The MSTI assay enables the detection of thiol-reactive compounds in a multiwell plate format in contrast to many reported fluorescent sulfur-based probes that have been developed for the detection of thiol-disulfide equilibria in biological samples.15–17 To our knowledge, the MSTI assay enables researchers to detect thiol-reactive compounds within large screening libraries for the first time. Strong electrophilic drug candidates can represent a liability in drug discovery because of their elevated toxicity in cell-based assays and in vivo studies. These compounds can form nonspecific protein interactions, cause allosteric structural protein changes, and deplete glutathione levels, which are essential for the redox chemistry of the cell. 18 Using the MSTI assay, these promiscuous inhibitors can be identified in a high-throughput manner and eliminated at an early discovery stage of drug discovery.
Material and Methods
Materials
All materials were used as they were received, with no further purification (J. T. Baker, Phillipsburg, NJ). Phosphate-buffered saline (PBS) was prepared in 1-L batches using 18 MΩ water with 3.23 mM K2HPO4·7H2O (J. T. Baker), 7.84 mM KH2PO4 (J. T. Baker), 5 mM KCl (Fisher), and 150 mM NaCl (Fisher) and adjusted to pH 7.0 with HCl (Mallinckrodt, St. Louis, MO) and NaOH (Fisher, Fair Lawn, NJ). The absorbance readings were completed in a 384-well UV plate (781801; Greiner Bio-One, Monroe, NC). The assay was performed in a 384 flat-bottom well black assay plate (3573; Corning, Corning, NY), which was sealed with an aluminum cover (6570; Corning) during incubation and mixing.
Instrumentation
All of the absorbance and fluorescence readings were performed on a Tecan Infinite M1000 plate reader (Tecan, Männedorf, Switzerland). Small-volume transfers were performed on the Tecan Freedon EVO liquid-handling system with a 100-nL pin tool (V&P Scientific, San Diego, CA). Chromatograms and mass spectra of molecules and reaction products were collected using Thermo Surveyor MSQ (Thermo Fisher Scientific, Billerica, MA) liquid chromatography/mass spectrometry (LC/MS) with an APCI probe with 10 µA corona or ESI probe with 3 kV capillary, 350 °C probe temperature, and a Waters XBridge C18 (Waters Corp., Milford, MA), 5-µm, 4.6 × 30-mm column. A Biotage (Uppsala, Sweden) SP1 flash chromatography system and Gilson (Middleton, WI) preparative LC (Prep. LC) (215 Liquid Handler, 306 Pump, 112 UV Detector) with a Waters XTerra Prep MS C18 OBD column (5 µm, 19 × 50 mm) were used for the purification. A Bio-Tek (Winooski, VT) MicroFlo Select instrument was used for the addition of the 30 µM MSTI solution to the assay plate. LC/MS was performed for all compounds using a water/methanol gradient to confirm a purity of >99%.
Synthesis
4-(S-acetylthio)benzaldehyde 19
4-(Methylthio) benzaldehyde (1.35 mL, 8.92 mmol) and tBuSNa (2 g, 17.85 mmol, 2 eq) were suspended in dimethylformamide (DMF) (30 mL), and the reaction mixture was heated with stirring at 160 °C for 4 h. The resulting brown suspension was cooled, and acetyl chloride (2.2 mL, 24.6 mmol) was added. After 2 h, the resulting suspension was poured into water, and diethyl ether was added. The ethereal layer was extracted with water three times, dried, and evaporated. Next, flash chromatography was performed (SP1.0 [Biotage], CH2Cl2/hexanes, 1:1); 1H NMR (300 MHz, CDCl3, ppm) δ 2.49 (s, 3H), 7.61 (m, 2H), 7.92 (m, 2H), 10.06 (s, 1H); 13C NMR δ 31.2, 130.6, 135.2, 136.1, 137.1, 192.1, 192.9. The yield was 40% (642 mg).
(E)-2-(4-(acetylthio)styryl)-1,3,3-trimethyl-3H-indol-1-ium (acetyl-MSTI)
(15) 4-(S-Acetylthio)benzaldehyde (0.200 g, 1.11 mmol) was dissolved in 10 mL acetic anhydride, and then 1,2,3,3-tetramethyl-3H-indolium iodide (0.330 g, 1.11 mmol) and sodium acetate (0.09 g, 1.11 mmol) was added. Stirring was continued at room temperature for 12 h, and then solvent was removed by vacuum. The solid was washed with diethyl ether (20 mL), then toluene (20 ml). Drying the precipitate in vacuum afforded orange product (55%, 0.210 g); 1H NMR: (300 MHz, CDCl3, ppm) δ 8.25 (d, J = 16.2 Hz, 1H, -CH = CH-), 8.20 (d, J = 7.9 Hz, 1H), 7.90(d, J = 16.4 Hz, 1H, -CH = CH-), 7.69–7.57 (m, 6H), 4.51 (s, 3H, -CH3), 2.49 (s, 3H, -COCH3), 1.89 (s, 6H, -CH3). 13C NMR: (300 MHz, CDCl3, ppm) δ 192.44, 182.56, 153.31, 143.05, 141.47, 134.78, 129.92, 122.68, 115.39, 114.55, 99.87, 53.62, 37.56, 30.67, 26.58; MS: (C21H22NOS+): 336.1 m/z.
(E)-2-(4-mercaptostyryl)-1,3,3-trimethyl-3H-indol-1-ium (MSTI)
Acetyl-MSTI (20 mg, 0.05 mmol) was dissolved in 2 mL tetrahydrofuran at 25 °C, and 25% NH4OH(aq) (20 mg, 0.5 mmol) was added in one portion. After 20 min of stirring at this temperature, the reaction mixture was acidified with 3 M HCl and was then extracted three times with 15 mL dichloromethane. After evaporation of the solvent, a yellow solid was obtained (10 mg, 44% yield); 1H NMR: (300 MHz, CDCl3, ppm) δ 8.25 (d, J = 16.20 Hz, 1H, -CH = CH-), 8.20 (d, J = 7.9, 1H), 7.90(d, J = 16.4 Hz, 1H, -CH = CH-), 7.69–7.57 (m, 6H), 4.51 (s, 3H, -CH3), 1.89 (s, 6H, -CH3); MS: (C19H20NS+): 294.13 m/z; NB: 13C NMR characterization of the reduced thiol was problematic even at high concentration due to oxidation and precipitation and the longer acquisition time needed for 13C NMR. 16
MSTI Assay
In the preparation of the “compound plate,” 15 µL of the 10-mM solution of small molecules in DMSO were dispensed in a 384-well polystyrene plate filling rows 1 to 18. A second 384-well polystyrene plate, which can be used as “control plate,” had rows 19 to 24 filled with 15 µL DMSO. Acetyl-MSTI was dissolved in methanol as a 10 mM solution and added to a PBS buffered solution at pH 12 with 50% methanol in a ratio of 1:10. After stirring for 2 min, the solution was diluted with PBS at pH 7.4 with 2% DMSO, 0.01% NP40, and 5% methanol to form a 30-µM solution of MSTI at pH 7.4. Next, 20 µL of the 30-µM solution was dispensed in rows 1 to 23 (black polystyrene “assay plate”). A 30-µM solution of aceyl-MSTI (positive control) was made in the same buffer, and 20 µL of this solution was dispensed to the assay plate (row 24). With the Tecan liquid-handling system, 100 nL from the compound plate and 100 nL from the control plate were transferred into the assay plate using the pin transfer tool. The assay plate was then centrifuged for 2 min at 1000 rpm, covered with the aluminum cover, and put on the plate shaker for agitation during the incubation period. After 30 min of incubation, the assay plate was once again centrifuged for 2 min at 2000 rpm to ensure a uniform liquid surface during the reading. The assay plate was then read for fluorescent intensity using the Tecan M1000 plate reader. An excitation wavelength of 510 nm and emission wavelength of 650 nm with a bandwidth of 20 nm and 10 nm, respectively; 100 flashes; 25-µs integration time; optimized gain; and z-position (optimized to the 30-µM MSTI solution) were used for the quantification of the fluorescence signal. The Z′ value for the assay was then calculated using MSTI as the negative control (0% binding) and acetyl-MSTI as the positive control (100% binding). 20 The percent binding of the small molecules at a concentration of 100 µM was reported as normalized response.
Results
To detect thiol-reactive compounds, and thus electrophiles, we designed a fluorescent probe that exhibits different spectroscopic properties in the nucleophilic state and as a conjugate with electrophiles. Therefore, an aromatic nucleophilic thiol functionality was connected to a conjugated π-system of a fluorophore. We chose an indolium dye and thiophenol to compose (E)-2-(4-mercaptostyryl)-1,3,3-trimethyl-3H-indol-1-ium (MSTI; Fig. 1A ). At a pH of 6.0 and higher, MSTI exists as an anion, as shown by the unchanged absorbance signal over a pH range ( Fig. 2B ). After the addition of an electrophile, such as 2-iodoacetamide, MSTI forms a covalent adduct ( Fig. 1A ). We isolated the conjugate of MSTI and 2-iodoacetamide and characterized it by 1H-NMR, MS, and spectroscopic analysis (see supplementary material). Importantly, the conjugate has significantly different spectroscopic properties that allow the differentiation of MSTI and MSTI-conjugate using fluorescence.
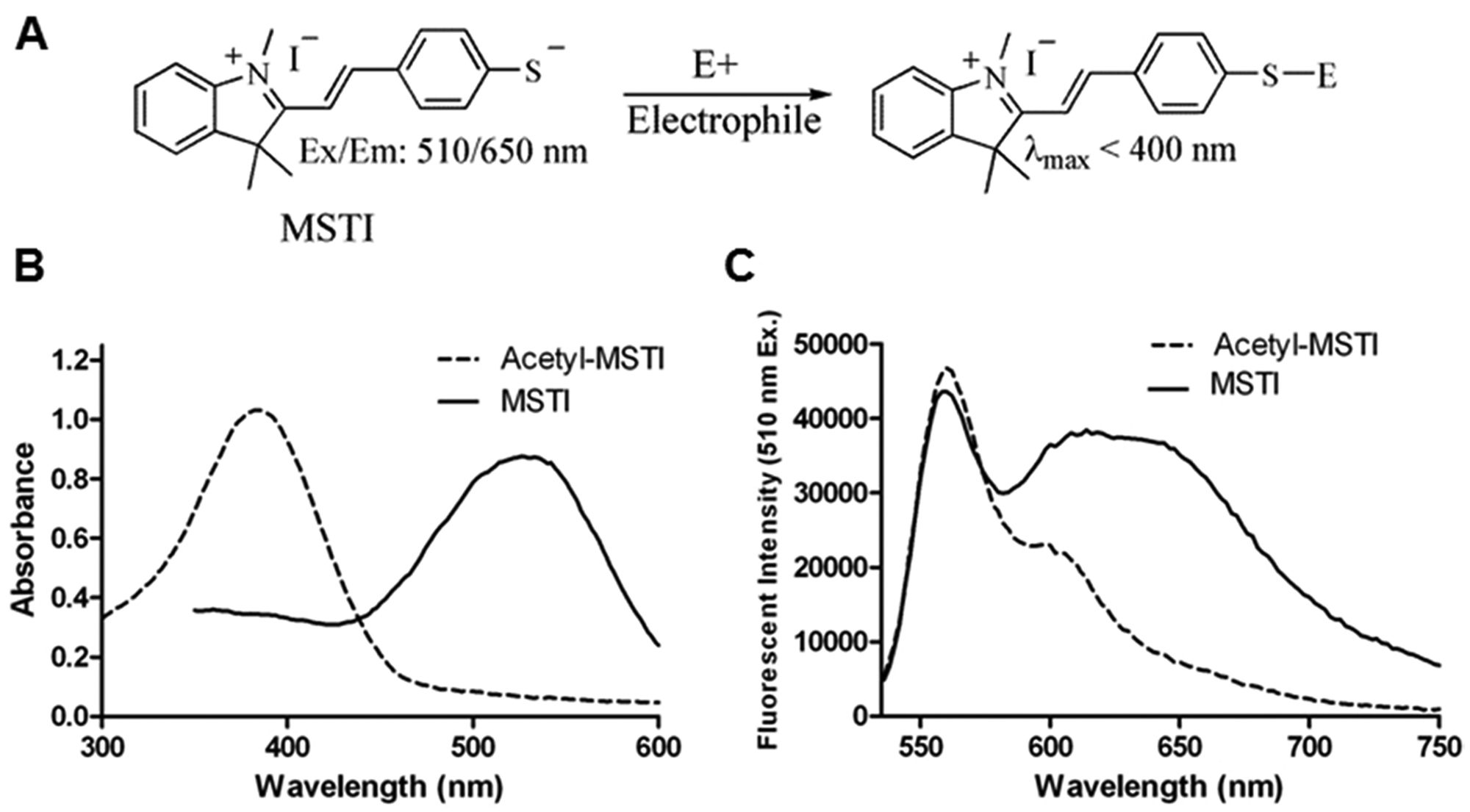
Spectroscopic properties of the (E)-2-(4-mercaptostyryl)-1,3,3-trimethyl-3H-indol-1-ium (MSTI) assay. (
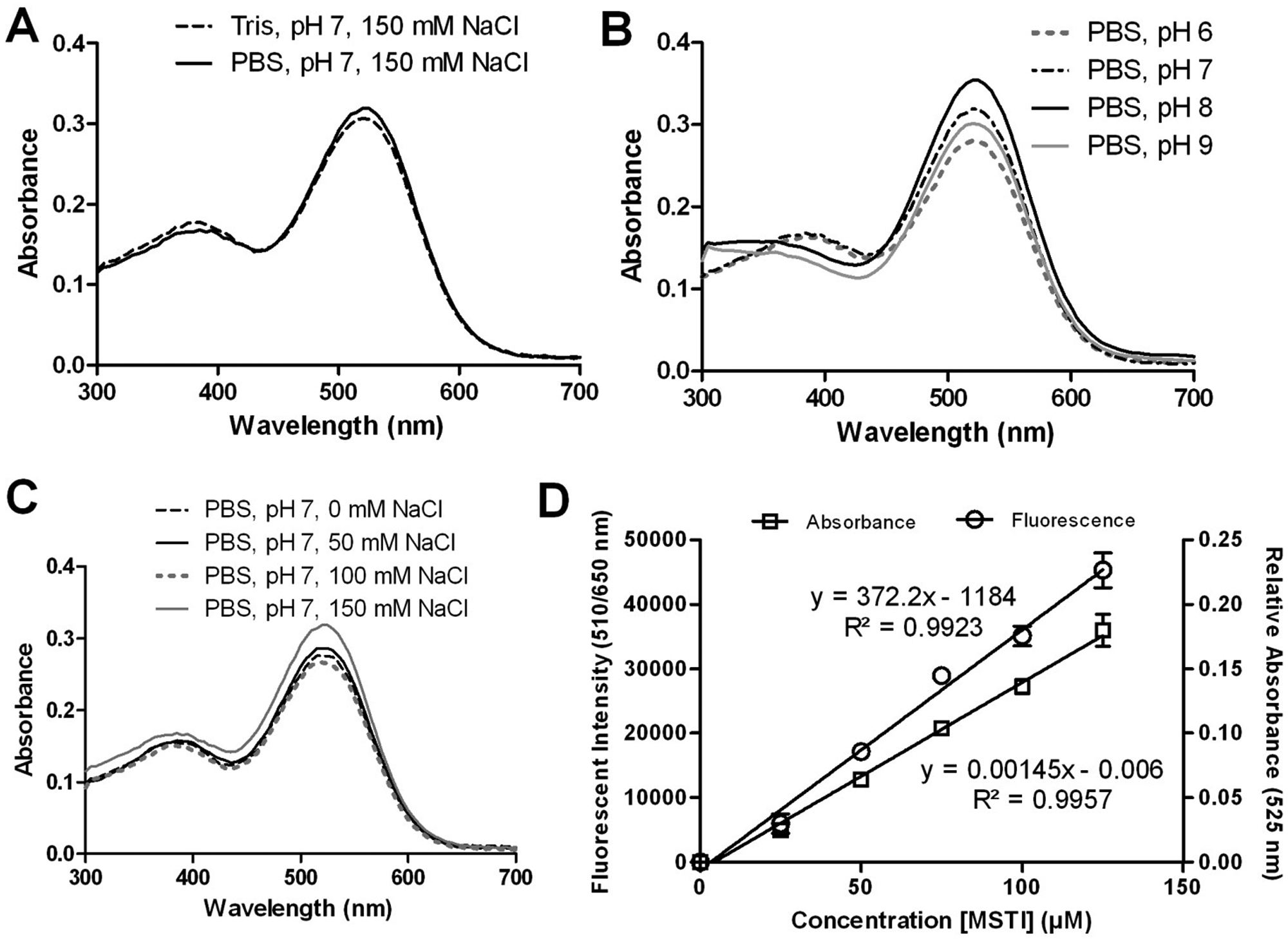
Buffer optimization for the (E)-2-(4-mercaptostyryl)-1,3,3-trimethyl-3H-indol-1-ium (MSTI) assay. (
MSTI, like all thiophenols, is oxygen sensitive and rapidly forms disulfides in non-degassed solvents. Our attempts to store MSTI for a prolonged time in the reduced form as a solid or in solution were not successful. Similar difficulties have been reported for fluorescent thiophenols. 16 Nevertheless, we characterized MSTI by mass spectrometry and 1H-NMR. MSTI was synthesized from acetyl-MSTI under alkaline condition, and acetyl-MSTI was in turn synthesized from 4-formylbenzyl thioacetate and 1,2,3,3-tetramethyl-3H indolium iodide (see supplementary material for the reaction scheme).
Acetyl-MSTI is stable as a solid as well as in solution and represents an excellent precursor for MSTI. Therefore, MSTI was generated in situ from acetyl-MSTI for screening purposes. A 10-mM methanol solution of acetyl-MSTI was diluted in degassed PBS with 50% by volume methanol solution at pH 12 and stirred for 2 min. After that time, the solution became purple in coloration, and more than 80% of acetyl-MSTI was converted into MSTI as determined by absorbance, fluorescence ( Fig. 1B , C ), and LC/MS (see supplementary material).
At a concentration of 500 µM in PBS, MSTI at pH 12.0 and acetyl-MSTI at pH 7.4 showed a strong and different absorbance spectrum. After changing the pH of the MSTI solution from 12.0 to 7.4, no change of the absorbance spectra was observed. The maximal absorbance (λmax) of acetyl-MSTI was measured at 384 nm, whereas MSTI λmax was 526 nm ( Fig. 1B ). This absorbance shift was very likely responsible for the appearance of a pink color for the MSTI solution. The fluorescence emission range, between 530 and 750 nm for both compounds at an excitation wavelength of 510 nm, was measured for 100 µM MSTI and 200 µM acetyl-MSTI, respectively. The gain optimization function of the instrument (Tecan M1000) automatically adjusts the highest fluorescence value between 40 000 and 50 000 units. The λmax fluorescent emission was 562 nm for acetyl-MSTI, and a broad emission peak between 550 and 700 nm was observed for MSTI ( Fig. 1C ). Because MSTI was generated in situ from acetyl-MSTI, around 20% of acetyl-MSTI remained, which was responsible for the first emission peak at 562 nm in the emission spectra of MSTI. However, a large fluorescence emission difference for MSTI and acetyl-MSTI was observed beyond 600 nm.
To optimize the fluorescence signal, the composition of the buffer was studied by monitoring the absorbance while varying the buffer reagent, pH, ionic strength (concentration of sodium chloride), or the concentration of MSTI ( Fig. 2 ). A 200-µM MSTI solution was used for these experiments. A similar absorbance in PBS (50 mM phosphate, 150 mM NaCl, pH 7.0) and Tris buffer (10 mM Tris base, 150 mM NaCl, pH 7.0) was observed for MSTI ( Fig. 2A ). At different pH values (pH 6–9) in PBS, MSTI showed only a marginal difference in absorbance with the highest values between pH 7 and 8 ( Fig. 2B ). The optimal NaCl concentration was 150 mM, giving the highest absorbance for MSTI ( Fig. 2C ). Finally, we measured the absorbance at 525 nm and fluorescence at 510/650 nm for MSTI in PBS (pH 7, 150 mM NaCl) at different concentrations ( Fig. 2D ). Both absorbance and fluorescence intensity were linear between 0.01 and 125 µM MSTI. A relative standard deviation of <5% for the absorbance and fluorescence was observed for all concentrations used. From this study, it was determined that an acceptable Z′ value 20 of more than 0.6 can be achieved with as little as 30 µM MSTI in the presence of phosphate buffer (50 mM), pH 7, and 150 mM NaCl.
Many organic compounds have a limited solubility in water and are usually predissolved in DMSO before addition to a biochemical or cell-based assay. Increasing amounts of DMSO can greatly enhance the solubility of small molecules but may also influence the performance of an assay. The influence of DMSO in the presence of a subset of small molecules ( Fig. 3B ) at a concentration of 100 µM in phosphate buffer (50 mM, pH 7.4, 150 mM NaCl) and MSTI (30 µM) is illustrated in Figure 3 .
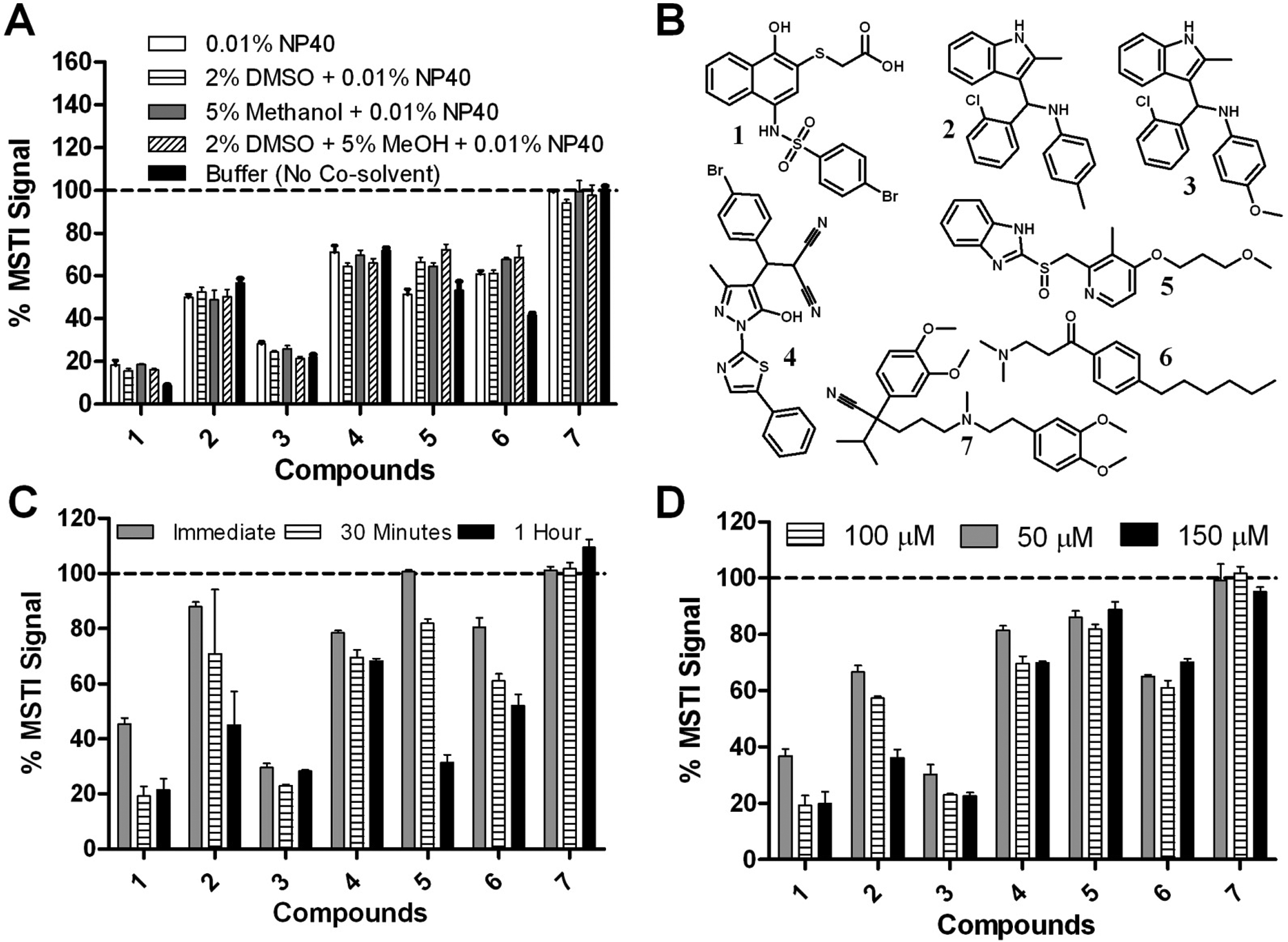
Evaluation of the (E)-2-(4-mercaptostyryl)-1,3,3-trimethyl-3H-indol-1-ium (MSTI) assay in the presence of small molecules was performed in phosphate-buffered saline (50 mM phosphate, pH 7.4, 150 mM NaCl) and MSTI (30 µM) with an excitation and emission wavelength of 510 nm and 650 nm, respectively. All measurements were carried out in triplicate. (
Compounds
Three different additives (methanol, DMSO, and NP40) and a combination of additives were investigated (
Fig. 3A
). In this study, 30 µM MSTI and compounds
With the optimized MSTI assay conditions—PBS (50 mM, pH 7.4, 150 mM NaCl), MSTI (30 µM), compound (100 µM), 5% methanol, 2% DMSO, and 0.01% NP40—we then screened a library of small molecules. The Library of 1280 Pharmacologically Active Compounds (LOPAC1280; Sigma Aldrich, St. Louis, MO) was used to determine the quality of the assay and the ability to identify compounds that are reactive toward nucleophiles mimicked by MSTI. Each LOPAC compound was measured at a concentration of 100 µM in triplicate ( Fig. 4 ).
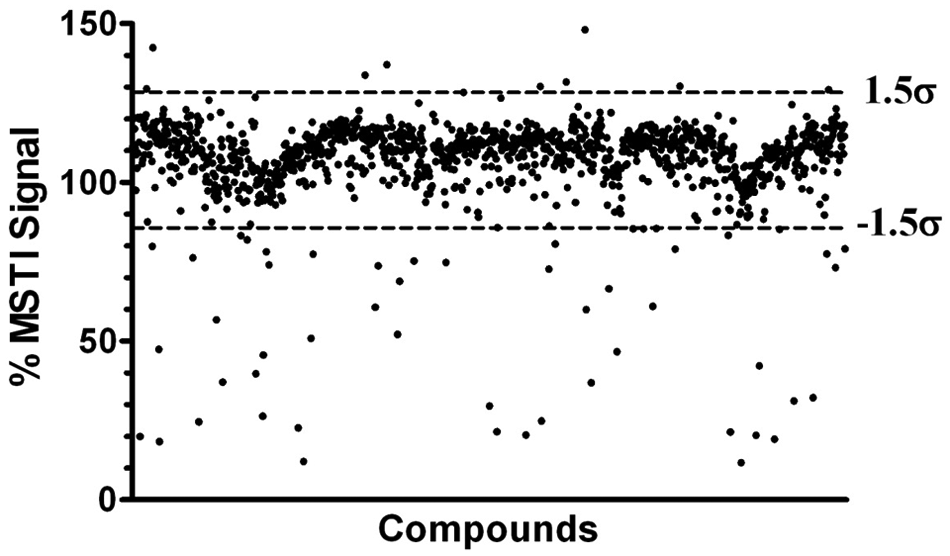
Results of MSTI-LOPAC screen (1280 compounds). LOPAC, Library of 1280 Pharmacologically Active Compounds; MSTI, (E)-2-(4-mercaptostyryl)-1,3,3-trimethyl-3H-indol-1-ium.
The Z′ values of this screen ranged between 0.62 and 0.88 with a mean of 0.75 (see supplementary material). The mean fluorescence intensity of all compounds in the presence of MSTI was 107.0% of the MSTI signal with a standard deviation (σ) of 14.2%. To safely distinguish between active and inactive molecules, a cutoff of σ = 1.5 from the mean was chosen as indicated with dotted lines in Figure 4 . Observing a subset of random 224 LOPAC compounds more closely, the cutoff of 1.5σ represents an acceptable distinction between both populations (see supplementary material). In addition, the background fluorescence was determined for each LOPAC molecule (100 µM) in the absence of MSTI at a 510-nm excitation and a 650-nm emission wavelength. The vast majority of LOPAC compounds exhibited no significant fluorescence intensity in comparison with the assay media. However, 11 compounds exhibited an intrinsic fluorescence of more than 1.5σ of the mean fluorescence signal (21.3%) of MSTI (see supplementary material).
The MSTI-LOPAC screen identified 9 compounds that exhibit a fluorescence signal of more than 129% of the average MSTI fluorescence intensity and 55 compounds with less than 85% of the average MSTI signal. The summary of these compounds is provided as supplementary material. Based on the ±1.5 σ cutoff, the hit rate was 5%. The majority of the hits identified, as predicted, were electrophilic compounds (
Fig. 5A
). These include transition metal complexes bearing ions such as Au2+ and Pt2+, α-haloketones, quinones, nitric oxide (NO)–releasing compounds, halo-alkenes, and unsaturated carbonyl compounds. Several MSTI conjugates have been characterized by 1H-NMR and MS, such as the 2-iodoacetamide-MSTI conjugate (see supplementary material). Other compounds identified are those that can undergo a conversion to an electrophilic compound such as β-aminoketones (conversion into unsaturated ketones) and 2-chloroamine derivatives (conversion into aziridines) (
Fig. 5B
). Interestingly, we also identified lansoprazole, a proton-pump inhibitor that forms disulfide bonds with cysteine residues. The same mode of action has been reported for positive control compound
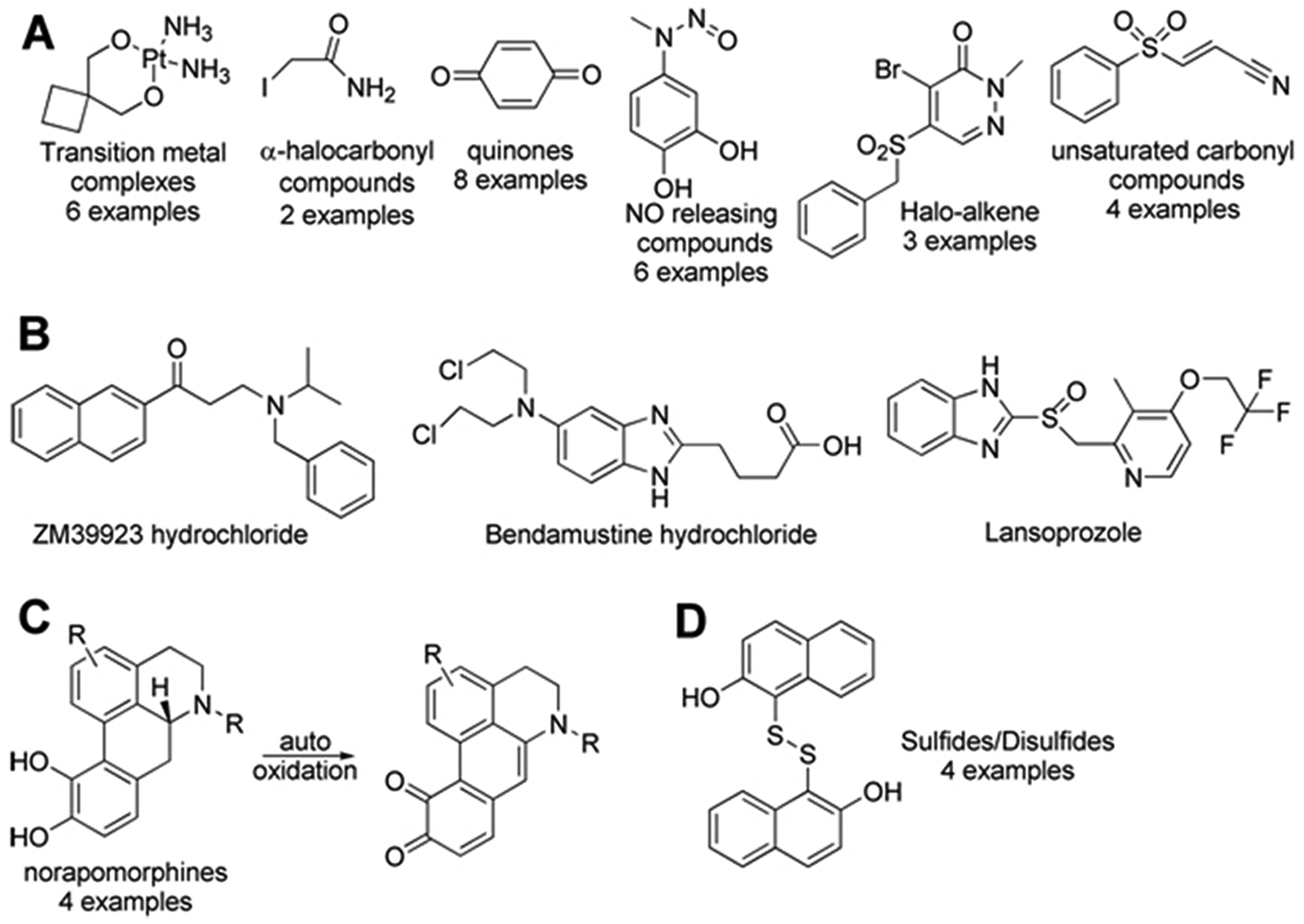
Thiol-reactive compound classes identified by the (E)-2-(4-mercaptostyryl)-1,3,3-trimethyl-3H-indol-1-ium (MSTI) assay. NO, nitric oxide.
This assay also identified the compound class of apomorphines ( Fig. 5C ). A possible explanation could be the formation of the corresponding diketone during the storage in DMSO, which has been reported. 27 The diketones, in contrast to the norapomorphines, have a red-shifted absorbance and appear with a blue-green discoloration, which might be the underlying mechanism for these false-positive hits. Finally, disulfides within the LOPAC screening collection were identified as hit compounds ( Fig. 5D ). These compounds are likely to be reduced by MSTI followed by the formation of oxidized MSTI species with reduced fluorescence intensity.
Discussion
Molecules that react with thiols such as cysteine have the potential to nonselectively modulate proteins and alter their modes of action, which is one of the hallmarks of promiscuous inhibition. The identification of such compounds is now possible with the MSTI assay. The MSTI molecule bears a nucleophilic thiol group that can easily react with electrophiles to form MSTI adducts. The covalent bond formation has a dramatic influence on the fluorescent properties of MSTI, significantly reducing its fluorescence at 650 nm. The MSTI assay only requires an incubation time of 30 min, and although optimized for a 384-well format, it is easily convertible to a 1536-well format, including a single centrifugation step of the assay plates (Adam Yasgar, unpublished observations). The application of a precursor, acetyl-MSTI, has the advantage that the reactive MSTI probe can be reliably produced in situ, thus circumventing any challenging storage regimes for MSTI. The MSTI assay is the first HTS assay that identifies thiol-reactive small molecules among screening library compounds. This assay also confirms the mode of action of irreversible inhibitors among LOPAC library compounds as shown for a selection of active molecules and their corresponding MSTI adducts (supplementary material). The reaction between MSTI and thiol-reactive compounds is dose dependent with a similar affinity but different efficacy, which could represent the difference between weak and strong electrophiles. The far-red detection of MSTI limits the number of molecules interfering with the assay, which was 0.85% for the LOPAC screening library. These molecules represent the largest group of false positives. The assay has an excellent reproducibility (Z′ > 0.6) and standard deviation of <5% for each compound. We anticipate that the MSTI assay will be a helpful tool to quickly identify potential promiscuous inhibitors among screening hits and enable the fast identification of the mode of action of hit compounds with regard to their ability to react with nucleophilic protein residues.
Supporting Information
The supplementary material includes the mass spectra, 1H-NMR, and fluorescence spectra of products formed between MSTI and other small molecules; quantification of the conversion of acetyl-MSTI to MSTI; dose-response analysis of active compounds; time dependency of Z′; evaluation of MSTI-LOPAC screening results; and MSTI-LOPAC hit compounds with their % MSTI values.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported in part by the University of Wisconsin Milwaukee, the UWM Research Growth Initiative (RGI), National Institute of Drug Abuse (R03DA031090), the UWM Research Foundation, the Lynde and Harry Bradley Foundation, the Richard and Ethel Herzfeld Foundation, and the Molecular Libraries Initiative of the National Institutes of Health Roadmap for Medical Research (U54MH084681).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
