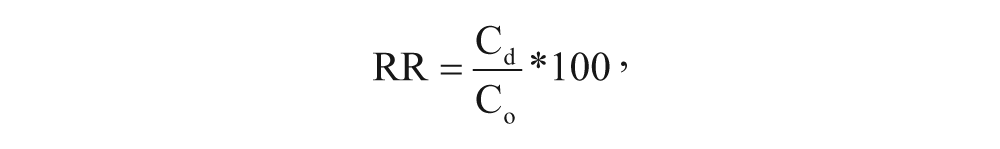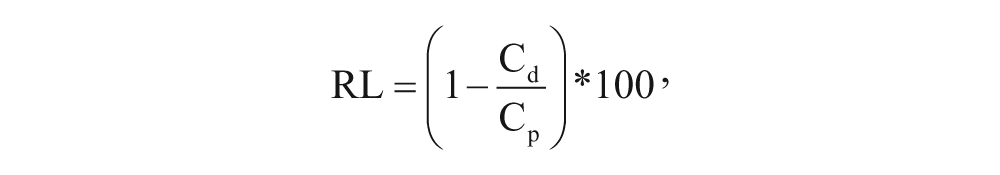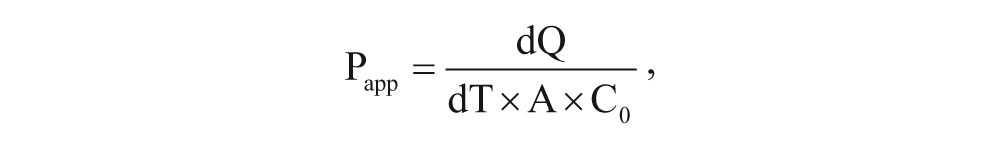Abstract
An integrated assay system involving dual/triple-probe microdialysis techniques in rats was developed earlier for testing interactions with P-glycoprotein (P-gp) at the blood-brain barrier using quinidine/PSC-833 as a P-gp substrate/inhibitor combination. The aim of the present study was to expand our assay system to mice using microdialysis with simultaneous sampling of blood and brain and to compare the result with a primary mouse brain endothelial cell monolayer (pMBMEC) assay. Brain penetration of quinidine was dose dependent in both anesthetized and awake mice after intraperitoneal drug administration. PSC-833 pretreatment caused a 2.5- to 3.4-fold increase in quinidine levels of brain dialysate samples in anesthetized or awake animals, after single or repeated administration of PSC-833. In pMBMEC, a 2.0- to 2.5-fold efflux ratio was observed in the transcellular transport of quinidine. The P-gp–mediated vectorial transport of quinidine was eliminated by PSC-833. These results indicate that quinidine with PSC-833 is a good probe substrate-reference inhibitor combination for testing drug-drug interactions with P-gp in the in vivo and in vitro mouse systems. With increasing number of humanized transgenic mice, a test system with mouse microdialysis experimentation becomes more important to predict drug-drug interactions in humans.
Introduction
Efflux transporters provide defense barrier mechanism of the main sanctuaries of tissues of the body. P-glycoprotein (P-gp) and breast cancer resistance protein (BCRP) are the most important efflux transporters at the blood-brain barrier (BBB) at the apical membrane of brain microvascular endothelial cells. 1 P-gp is encoded by the MDR1 gene in humans and mdr1a (primarily expressed in the brain) and mdr1b genes in rodents. 2 It is important to distinguish if poor brain penetration of an investigational drug is due to poor passive permeability or to efflux by an apically located transporter. Monolayers of immortalized cell lines (CaCo-2, LLC-PK1, and MDCKII) expressing human transporters and primary cell cultures (brain microvascular endothelial cells) serve as an efficient tool for screening transporter activities at the BBB.2–4 Several compounds interact with the efflux transporters in vitro but play a minor role in substrate efflux at the BBB in vivo. 5 On the contrary, some high passive permeability substrates such as verapamil, itraconazole, and ketoconazole are negative in the MDCKIIMDR1 assay but show a P-gp–dependent brain exposure in vivo.6–8 Therefore, in vivo testing of transporter interactions at the BBB is essential.
The brain entry of quinidine (QND) appears to be restricted. 9 P-gp mediates the efflux of QND across the BBB, as demonstrated by Kusuhara et al, 10 using the MDR1 inhibitor (PSC-833) and mdr1a knockout mice. In our previous study, 11 an in vitro–in vivo assay system with elements of different complexity (membrane-based assays, cell-based assays, in vivo assays) was validated for testing drug–P-gp interactions at the BBB using QND. In this study, QND and PSC-833 were identified as a specific P-gp probe substrate/inhibitor combination. In dual- and triple-probe rat microdialysis experiments, systemically or locally administered PSC-833 increased the brain penetration of QND, suggesting that the microdialysis techniques can be used for in vivo assessment of drugs for P-gp interactions of drugs at the BBB. 11 The Food and Drug Administration recommends digoxin as a P-gp probe substrate in vitro 12 ; however, its in vivo application in preclinical studies is limited because of its low LD50 value in rodents. 13 In the ATPase assay, QND was superior compared with digoxin (IC50 values were 7.8 and 68.5 µM, respectively), and in MDCKII and rat brain endothelial cell (RBEC) monolayers, QND also outperformed digoxin. 11
In vivo microdialysis is a gold-standard technique for determination of unbound, pharmacologically active drug levels in the target tissue. This technique, which involves the surgical implantation of a microdialysis probe into brain or peripheral tissues, is technically very demanding but has many advantages. 14 It allows the in vivo measurement of drug transport into specific brain region(s) and monitoring of the time course of drug-drug interactions. Furthermore, more than one compound in more than one target tissue can be measured simultaneously using dual/triple-probe approaches.
There is an increasing interest in creating “genomically” humanized mouse models, in which human genomic loci are transferred into the mouse genome. To study human efflux transporter interactions in vivo, transporter humanized mice may be the best preclinical tools. Therefore, a further advantage of in vivo microdialysis is that it can be used to characterize the effects of mdr1a gene knockouts/or knock-ins on the integrity of BBB in mice.2,15,16
Some studies described P-gp and substrate interactions at the BBB in mice using single–brain probe17–19 or dual–brain probe (parenchymal and ventricular) microdialysis.20–22 However, only very few studies have been published in which brain penetration of a drug is tested using one vascular and one brain microdialysis probe simultaneously in mice. 23 In the blood, many drugs are present in the unbound and bound form. The free drug concentration in blood is readily available for most distribution processes. Determination of the free drug concentrations in dialysates from blood and brain is a reliable direct method to monitor brain penetration of molecules and to test drug-drug interactions at the BBB.
The aim of the present study was to validate QND and PSC-833 as an appropriate P-gp substrate/reference inhibitor combination to mice. The further aim was to correlate vectorial transport of QND in vitro using a primary mouse brain microvascular endothelial cell (pMBMEC) monolayer assay.
Materials and Methods
Chemicals
Sodium chloride, potassium chloride, magnesium chloride hexahydrate, calcium chloride dihydrate, digoxin, and chloral hydrate were purchased from Fluka (Sigma-Aldrich Chemie GmbH, Steinheim, Germany). Quinidine, collagenase type 2, bovine serum albumin (BSA), Percoll, fetal calf serum, hydrocortisone, fibronectin, and puromycin were purchased from Sigma (Sigma-Hungary Kft., Budapest, Hungary). Dulbecco’s modified Eagle’s medium (DMEM)/F12 and Hank’s balanced salt solution (HBSS) from Invitrogen (Csertex Kft, Budapest, Hungary), collagenase/dispase and basic fibroblast growth factor (bFGF) from Roche (Roche Hungary Kft, Budapest, Hungary), plasma-derived serum (PDS) from First Link (Birmingham, UK), and Transwell filters from Costar Corning (Zenon Bio Kft, Szeged, Hungary) were used. Quinidine (9-3H) was purchased from American Radiolabeled Chemicals (St. Louis, MO). PSC-833 was supplied by Solvo Biotechnology (Szeged, Hungary).
Animals
Male NMRI mice (ToxiCoop, Budapest, Hungary) weighing 28 to 32 g, with free access to food and water, were used throughout this study. All animal experiments were performed in full compliance with the guidelines of the Association for Assessment and Accreditation of Laboratory Animal Care International’s expectations for animal use, per the spirit of the license issued by the directorate for the Safety of the Food Chain and Animal Health, Budapest and Pest County Agricultural Administrative Authority, Hungary.
Microdialysis Studies
Selection of Peripheral Probe and In Vitro Feasibility Study
Peripheral probes from several vendors were tested to determine if they could be used for sampling blood in the jugular vein. After this evaluation, a slightly modified version of peripheral probes from Microbiotech (Stockholm, Sweden) was chosen and used for the in vitro and in vivo microdialysis experiments.
Before conducting in vivo microdialysis experiments, the mouse brain and blood microdialysis probes were characterized for relative recovery (RR) and relative loss (RL) in vitro. Microdialysis probes (MAB1.4.3, MAB1.4.4, MAB8.4.2, MAB8.4.3) were placed in a CMA (9-oxo-10-acridine acetic acid) in vitro stand and connected to a CMA/102 microdialysis pump (input) and to a plastic collection vials (output). Artificial cerebrospinal fluid (CSF) was perfused through the brain probes and peripheral perfusion fluid (PPF) through the peripheral probes. The chemical composition of CSF was 147 mM NaCl, 2.7 mM KCl, 1.2 mM CaCl2 × 2 H2O, and 0.85 mM MgCl2 × 6 H2O, and that of PPF was 147 mM NaCl, 4 mM KCl, and 2.3 mM CaCl2 × 2 H2O.
In the first set of in vitro feasibility studies, probes with different membrane surfaces and various perfusion flow rates (1.0 or 0.5 µL/min) were compared for mouse brain (MAB8.4.3, MAB8.4.2) and peripheral (MAB1.4.4, MAB1.4.3) probes using a “gain”-type experimental arrangement. 24 The probes were perfused with PPF or CSF for 45 min (during this time, the probes were kept in the air) for equilibration, and samples were collected every 15 min. The probes were then immersed in QND solution, and the samples were collected for 90 min. After 90 min, the probes were removed from QND solution and perfused further with CSF or PPF for 60 to 75 min. (Considering the smaller membrane surface of the mouse microdialysis probes, an approximately three times higher quinidine concentration [1 µM] was used in the present study than in a previous study on in vitro recovery of CMA 12/2-mm probes for quinidine. 25 )
In the second part of the in vitro feasibility studies, the previously selected microdialysis probes (MAB1.4.3. and MAB8.4.3.) were tested in “gain” and “loss” experiments 24 at a lower perfusion flow rate (0.5 µL/min). In the RR study, the probes were perfused with CSF or PPF for 90 min and then immersed in QND solution for 150 min. Subsequently, the probes were perfused for 120 min with the perfusion fluids (CSF, PPF). Samples were collected every 30 min during the experiment. RR was determined from the QND concentrations according to the following equation:
where Cd is the QND concentration in the dialysate samples and Co is the QND concentration in the original solution.
In the RL study, the probes (MAB1.4.3 and MAB8.4.3) were perfused with QND solution (1 µM) and, after an equilibration period (90 min), immersed in CSF or PPF for 150 min. The subsequent washout period was 120 min. RL was determined from the QND concentration using the following formula:
where Cd is the concentration of QND in the dialysate samples and Cp is the concentration of QND in the perfused solution.
Dual-probe microdialysis in anesthetized mice
Animals were anesthetized with chloral hydrate (450 mg/kg intraperitoneally [IP]). The right jugular vein was exposed and the MAB1.4.4 microdialysis probe was inserted into the vein through the pectoral muscle. After checking the flow through the peripheral probe, the tubings of the probe were exteriorized between the scapulae. Then the animals were placed in a Stoelting stereotaxic instrument, and the brain probe (MAB8.4.3) was implanted into the right frontal cortex using the following coordinates with respect to the bregma: anterior-posterior (AP), +2.2 mm; from midline (ML), 1 mm; and dorsal-ventral (DV), −3 mm. 26 Microdialysis probes were connected to a CMA/102 microdialysis pump and perfused with CSF (brain probe) or PPF (peripheral probe) at a flow rate of 0.5 µL/min. After a 1-h equilibration, the animals were treated with QND (20 or 40 mg/kg IP), and then the sample collection was continued for 5 h. Control mice received vehicle (1.2% cc ethanol, 8.0% Tween-80, 2.2% PEG 300 in saline) IP 20 min prior to QND administration. In the inhibitor-treated group, PSC-833 was injected 20 min prior to QND treatment at a dose of 10 mg/kg IP. The microdialysate samples were collected every 60 min and placed on dry ice immediately. The frozen samples were stored at −70 °C in a freezer until transferring to a bioanalytical laboratory.
Single-Probe Brain Microdialysis in Awake Mice
Under chloral hydrate anesthesia, the animals were placed in a stereotaxic frame, and a CMA/7 guide cannula (CMA, Solna, Sweden) was implanted above the target region of the brain (frontal cortex) at the following coordinates: AP, 2.2 mm; L, −0.7 mm; and DV, −1.25 mm. Then it was fixed with dental cement and anchor screws to the skull, and the animals received 1 mL saline subcutaneously to avoid dehydration. During a recovery period of 4 to 5 days, the operated animals were kept individually in their cages on a heating pad at 28 to 30 °C.
On the afternoon prior to microdialysis experiments, the brain probes (CMA/7.2) were introduced through the guide cannulae, and the animals (one control and one inhibitor treated simultaneously) were placed into containers of the awake mouse system (Instech Laboratories, Inc., Plymouth, PA). The perfusion of CSF was adjusted to a 0.2-µL/min flow rate for the night. The next morning, the flow rate was set back to 0.5 µL/min for the entire time of the experiment. The brain microdialysis samples were collected every 60 min from 1 h prior to QND administration (20 or 40 mg/kg IP) until 5 h after the treatment. Control mice received vehicle at 20 min prior to QND treatment, and the P-gp inhibitor-treated groups received PSC-833 (10 mg/kg IP). After collection, the samples were frozen and stored at −70 °C until bioanalysis.
In some experiments, the animals were left in the container of the awake mouse system after collecting the last dialysate samples for the first day. The administration of QND and PSC-833 or vehicle was repeated the next day at the same time using the same experimental protocol, including the same perfusion conditions.
Dual-Probe Microdialysis in Awake Mice
The guide cannula implantation was performed similarly to the above description. On the day of microdialysis experiments, the animals were mildly reanesthetized, and the venous probe (MAB1.4.3) and brain probe (CMA7/2) were introduced into the target tissues. The animals were placed into a movement-responsive animal system (developed by Solvo Biotechnology), and probes were connected to microdialysis pumps. CSF and PPF were perfused at a flow rate of 0.5 mL/min, and the samples were taken every 60 min. After 2 h equilibration, QND (40 mg/kg) was administered IP (zero time). One group of animals was treated with vehicle and another group received PSC-833 (10 mg/kg) IP 20 min prior to QND treatment. The sample collection was continued for a further 5 h.
Isolation of Mouse Brain Microvascular Endothelial Cells (MBMECs)
Primary mouse brain microvascular endothelial cells were isolated from 4- to 6-week-old BALB/c mice (for reference, see Sziráki et al. 11 ). Briefly, after removal of meninges, cerebral cortices were cut into small pieces and digested with 1 mg/mL collagenase type 2 for 75 min at 37 °C. After separation of myelin by centrifugation in 20% BSA, a second digestion was performed with 1 mg/mL collagenase/dispase for 50 min at 37 °C. Microvessel fragments were collected after centrifugation on Percoll gradient (10 min, 1000 g) and plated onto fibronectin/collagen-coated dishes. Endothelial cells growing out of the microvessels were cultured in DMEM/F12, 10% plasma-derived serum, and growth factors. During the first 2 days, 4 µg/mL puromycin was added to remove contaminating cells. Glial cultures were prepared from newborn rats and cultured on poly-L-lysine–coated surfaces. After reaching confluency, the endothelial monolayer was supplemented with 550 nM hydrocortisone, 250 µM CPT-cAMP, and 17.5 µM RO-201724 and placed into dishes containing glial cultures for 24 h.
Mouse Brain Microvascular Endothelial Cell (pMBMEC) Monolayer Assay
Transwell filters containing endothelial cells were removed from the plates containing the glial culture. Filters were washed with Ringer-HEPES solution (pH 7.4). QND or digoxin was added at final concentrations of 0.1 µM and 10 µM, respectively. Radiolabeled QND and digoxin as tracers also resulted in a radioactive concentration of 1 µCi/mL. The inhibitor (PSC-833) was added together with the test compound at final concentrations of 1 µM. Samples were taken from the basolateral or apical side at 15 min, and transcellular transport of QND or digoxin was determined by measuring the radioactivity using a liquid scintillation counter (MicroBeta; PerkinElmer, Waltham, MA).
Analytical Determination of QND in Dialysate Samples
QND levels of dialysate samples were measured by high-performance liquid chromatography (HPLC) with fluorescence detection. The HPLC system was a Merck-Hitachi LaChrom System (Hitachi High-Technologies Corporation, Tokyo, Japan), consisting of a D 7000 interface, L 7100 pump, L 7200 autosampler, and L 2480 fluorescence detector. Separation of QND was achieved using a C18-RP column (Phenomenex [Torrance, CA] Luna C18, 5 µ, 100 Angstrom, 150 × 2 mm RP) with an isocratic elution profile, consisting of 68% water (0.05% v/v H2SO4, 0.01 mol/L sodium octane-1-sulfonate monohydrate) and 32% acetonitrile with an excitation wavelength of 248 nm, an emission wavelength of 460 nm, and a flow rate of 0.4 mL/min. The 10-µL aliquots of samples were injected into the HPLC module. Concentration-time profiles of QND levels of blood and brain dialysates were generated with Microsoft Excel (Microsoft Corp., Redmond, WA) and with MicroCal Origin (GE Healthcare, Piscataway, NJ), including determination of area under the curves (AUCs).
Data Analysis of In Vivo Experiments
A paired sample t-test was used for comparing blood and brain means of AUCs and Cmax values in the different treatment groups.
Data Analysis of Monolayer Efflux Experiments
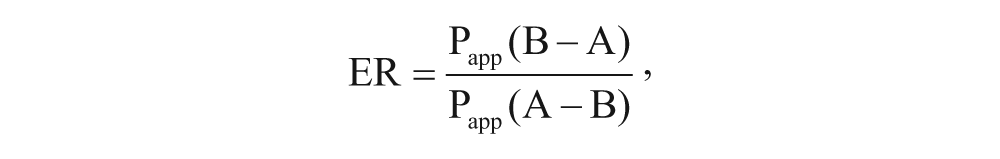
Permeability coefficients and efflux ratios were calculated using the following formulas:
where dQ is the transported amount, dT is the incubation time, A is the surface of filter, and C0 is the initial concentration.
Results
In Vitro Feasibility Study with Microbiotech Mouse Microdialysis Probes
Effects of membrane length and flow rate on RR of Microbiotech probes for QND
Recoveries of brain (MAB8.4.2) and peripheral (MAB1.4.4) probes were compared first at a flow rate of 1 µL/min. The 2-fold difference in the membrane length of the two probes resulted in more than a 2-fold difference in the RRs (27.4% and 12.6%, respectively) (data not shown). The recoveries of the brain probes with different membrane length (MAB8.4.2 and MAB8.4.3) at a flow rate of 0.5 µL/min were also compared (data not shown). The RR was proportional with the membrane length (3-mm membrane: 33.4%; 2-mm membrane: 23.1%). The RRs of MAB8.4.2 probes perfused with CSF at a flow rate of 1 µL/min or 0.5 µL/min showed a negative correlation (1 µL/min: 12.6%; 0.5 µL/min: 23.1%).
Determination of bidirectional diffusion of QND through MAB1.4.3. and MAB8.4.3 mouse probes in vitro
As a second part of the in vitro feasibility study, MAB1.4.3 and MAB8.4.3 microdialysis probes were further tested using a 1-µM QND test solution and a 0.5-µL/min flow rate. RR and RL were 38.38% ± 2.59% and 41.75% ± 2.50% for MAB1.4.3 ( Fig. 1A , B ) and 34.55% ± 2.60% and 35.88% ± 2.86% for MAB8.4.3 ( Fig. 1C , D ), respectively. These results indicate that the diffusion of QND through the membrane of both the peripheral and the brain probes is symmetrical. In addition, the values are close to each other for the two types of probes; therefore, this pair of probes is suitable for in vivo microdialysis experiments to study the brain penetration of QND with simultaneous sampling of blood and brain.
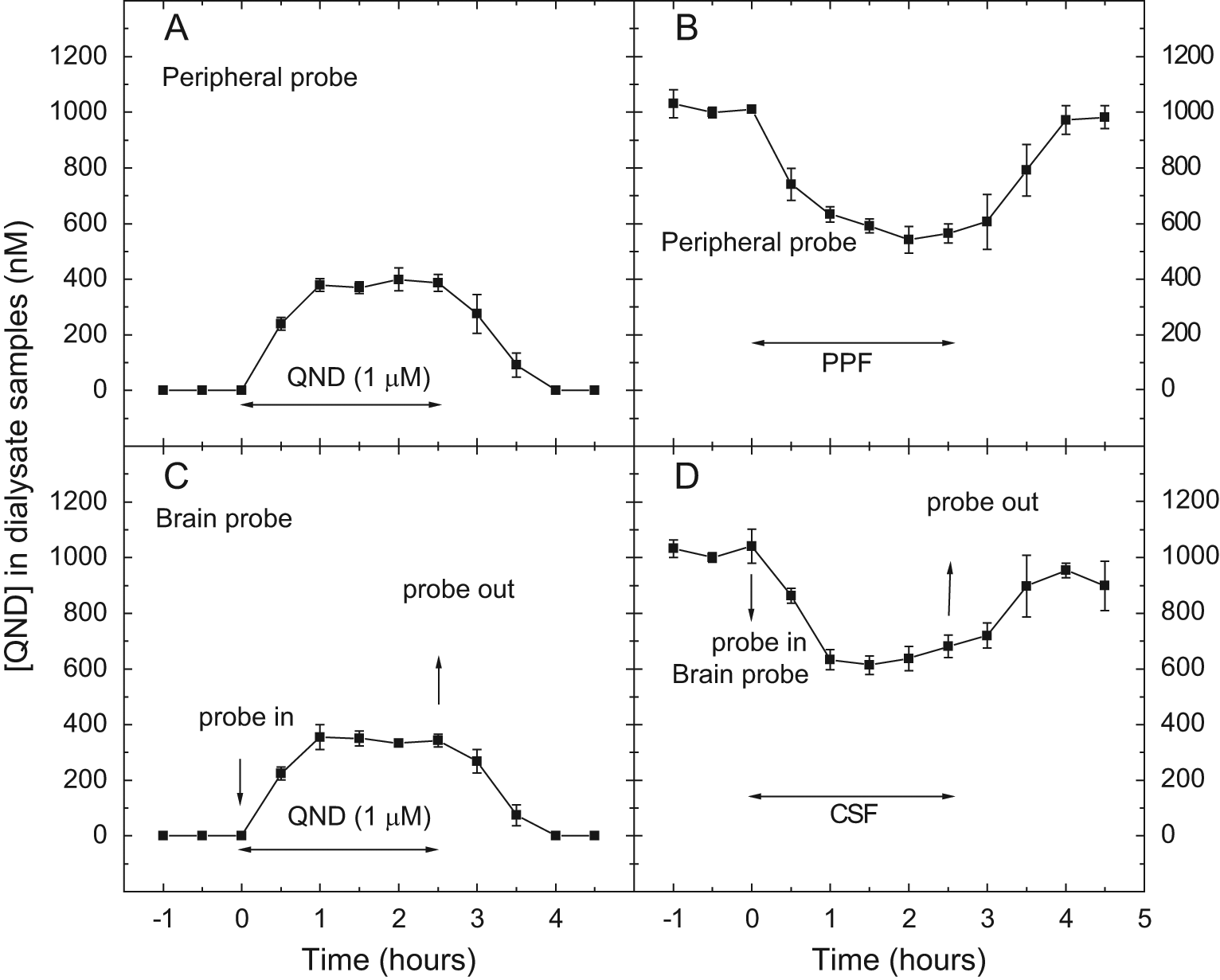
Concentration-time profiles of quinidine (QND) in dialysate samples from the in vitro gain (
Determination of P-gp Substrate-Inhibitor Interactions at the BBB In Vivo
Brain Penetration of QND and QND–PSC-833 Interaction in Anesthetized Mice
Time-concentration curves of QND in dialysate samples are presented in Figure 2A , B . In the control animals, the brain penetration of QND was dose dependent, and the differences between the brain AUCs and blood AUCs for the two doses were statistically significant (p < 0.003 for brain and p < 0.001 for blood, respectively). The brain penetration of QND is characterized by the brain-to-blood ratio; the AUCbrain/AUCblood ratios in animals treated with QND (20 and 40 mg/kg IP) were 0.042 and 0.069, respectively ( Fig. 2A ). Pretreatment with PSC-833 (10 mg/kg IP) resulted in a 4-fold increase (to 0.17; p < 0.035) at the lower QND dose and a 2.6-fold increase (to 0.18; p < 0.009) at the higher QND dose, respectively ( Fig. 2B ).
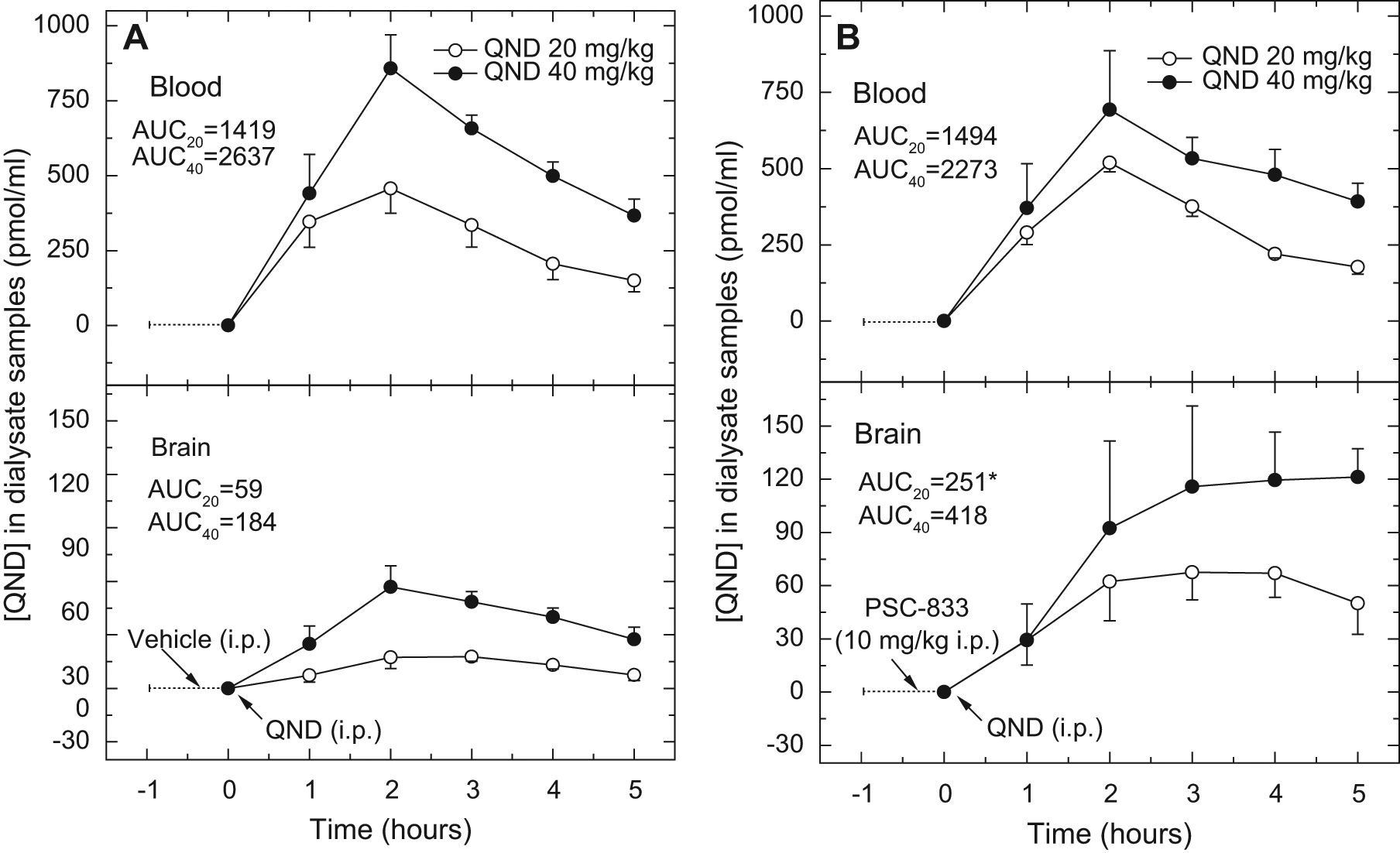
Concentration-time profiles of quinidine (QND) in dialysate samples from in vivo microdialysis experiments in anesthetized mice. The dialysate samples were collected from blood (upper panels) and brain (lower panels) every 60 min at a flow rate of 0.5 µL/min. Animals were treated with QND (20 or 40 mg/kg intraperitoneally [IP]) at zero time. Vehicle (1.2% cc ethanol, 8.0% Tween-80, and 2.2% PEG-300 in saline) (
Brain Penetration of QND and QND–PSC-833 Interaction in Awake Mice
Single-probe approach
Four groups of awake mice were implanted with a single brain probe into the frontal cortex without using the peripheral probe. One pair of these animals, one QND-treated mouse (20 or 40 mg/kg IP) and one mouse receiving both PSC-833 and QND, were simultaneously tested on each experimental day. Similar to anesthetized mice, the brain penetration of QND was dose dependent both in the control group and in the group receiving the combination of QND and PSC-833 ( Fig. 3A , B ). PSC-833 pretreatment enhanced the brain penetration of QND apparently due to the inhibition of P-gp at the BBB; there was a 6-fold increase in AUCbrain in animals treated with 20 mg/kg QND and a 4.5-fold increase in animals treated with 40 mg/kg QND ( Fig. 3B ). The effect of the P-gp inhibitor on the brain penetration of QND was statistically significant at both doses (p < 0.05 for AUCbrain and p < 0.01 for Cmax).
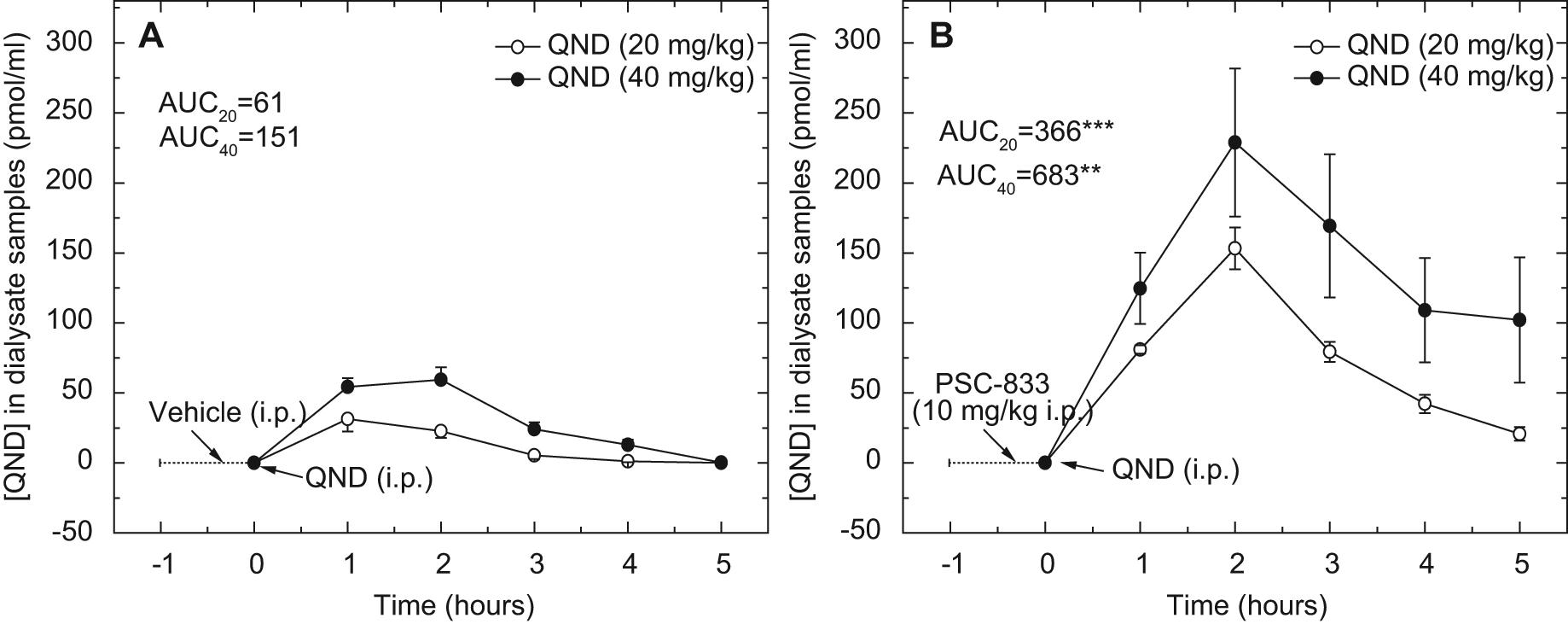
Concentration-time profiles of quinidine (QND) in dialysate samples from in vivo microdialysis experiments in awake mice with a single-probe approach. The dialysate samples were collected from brains every 60 min at a flow rate of 0.5 µL/min. Animals were treated with QND (20 or 40 mg/kg intraperitoneally [IP]) at zero time. Vehicle (1.2% cc ethanol, 8.0% Tween-80, and 2.2% PEG-300 in saline) (
In some animals from both the control and PSC-833–treated groups, the first day treatments were repeated on the following day in the single-probe experimental setup. The concentration-time profiles were determined in brain dialysate samples obtained both on the first and second days ( Fig. 4 ). The effect of PSC-833 on the brain penetration of quinidine was statistically significant on both days (p < 0.01 for the first day and p < 0.05 for the second day for Cmax values). The brain penetration of QND on the second day was higher than on the first day both in the control and the PSC-833–treated groups (AUC = 70 and 113 in controls and 442 and 611 in PSC-833–treated animals, respectively). However, the difference between the 2 days was not statistically significant.
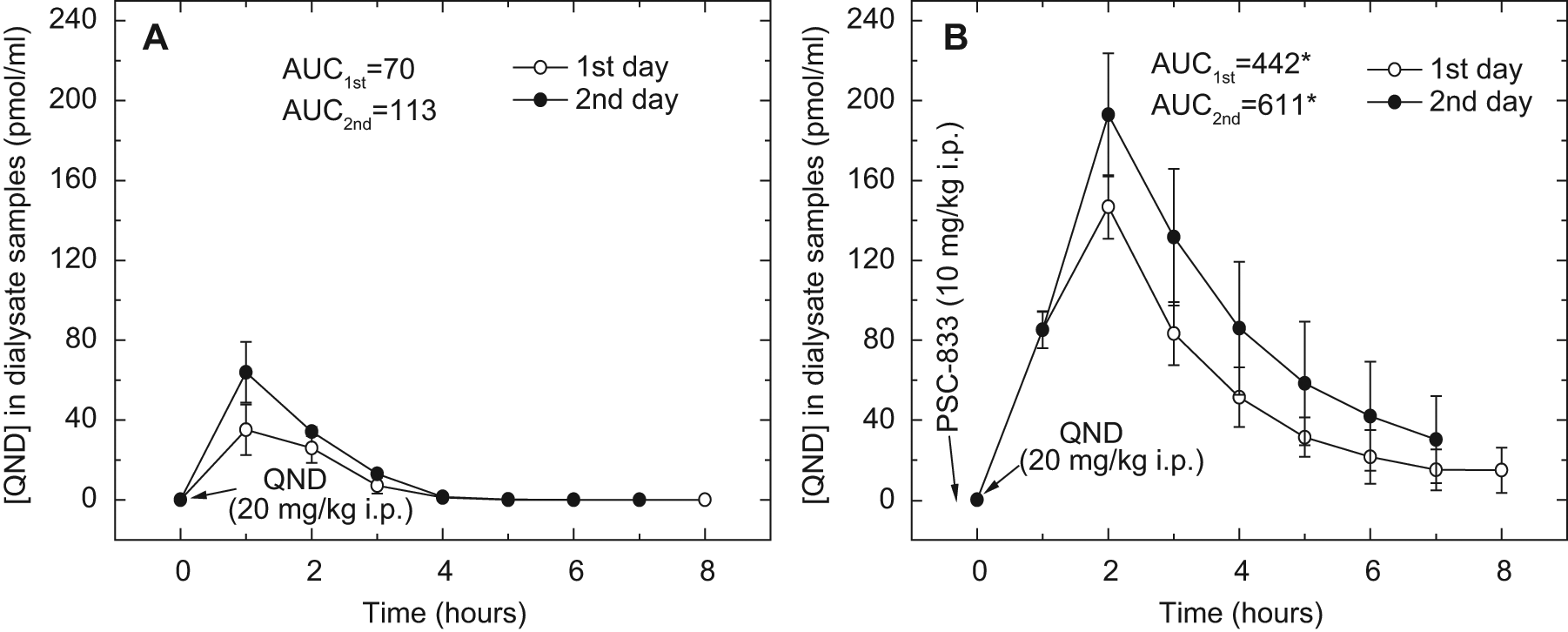
Concentration-time profiles of quinidine (QND) in dialysate samples from in vivo microdialysis experiments in awake mice with repeated drug administration. The dialysate samples were collected from brains every 60 min at a flow rate of 0.5 µL/min. Animals were treated with QND (20 mg/kg intraperitoneally [IP]) at zero time. Vehicle (1.2% cc ethanol, 8.0% Tween-80, and 2.2% PEG-300 in saline) (
Dual-probe approach
Awake mice implanted with one brain and one vascular probe, to simultaneously sample brain and blood, were also divided into two treatment groups: a PSC-833– and QND-treated group and a control group with only QND. The concentration-time profiles are presented in Figure 5 . The AUCbrain/AUCblood ratio in control mice was 0.224 ( Fig. 5A ), similar to that determined earlier in awake rats (unpublished data). Brain penetration of QND increased in a statistically significant manner (AUCbrain/AUCblood ratio = 0.997, p < 0.01) after PSC-833 treatment ( Fig. 5B ).
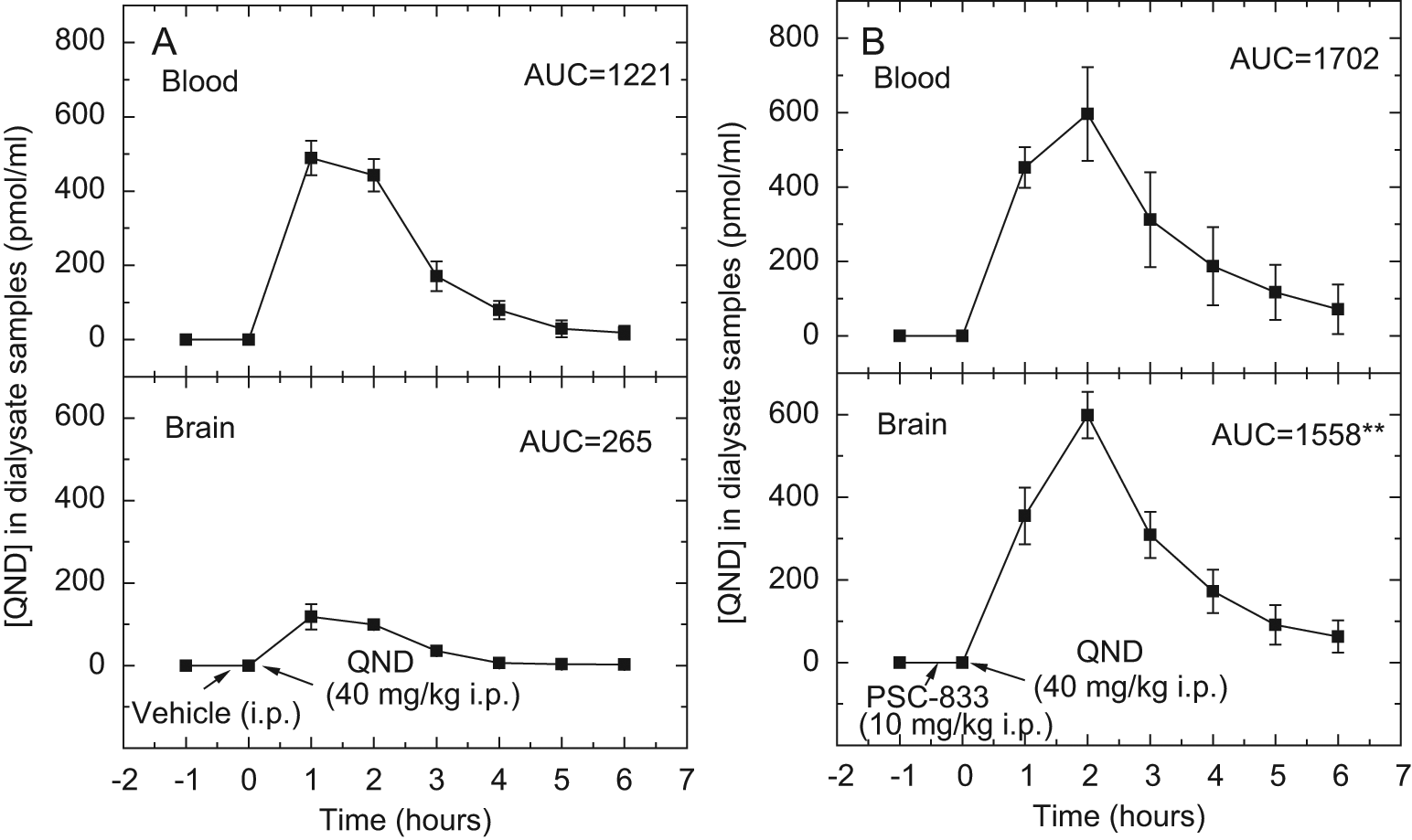
Concentration-time profiles of quinidine (QND) in dialysate samples from in vivo microdialysis experiments in awake mice with the dual-probe approach. The dialysate samples were collected from brains every 60 min at a flow rate of 0.5 µL/min. Animals were treated with QND (40 mg/kg intraperitoneally [IP]) at zero time. Vehicle (1.2% cc ethanol, 8.0% Tween-80, and 2.2% PEG-300 in saline) (
P-gp Substrate-inhibitor Interactions at the BBB In Vitro
Primary mouse brain microvascular endothelial cell monolayer assay
The QND transcellular transport (0.1 µM) efflux ratio in the pMBMEC monolayer using solo-cultured and astrocyte co-cultured endothelial cells with a 15-min incubation was 2.23- and 2.5-fold ( Fig. 6 ). (Although the resistance of the endothelial cells was increased in the presence of rat astrocytes [70 vs. 106 Ω*cm2], the efflux ratio did not increase significantly.) Similar efflux ratios were found using a 30- and 60-min incubation (data not shown). In the presence of P-gp inhibitor, PSC-833 (10 µM) apical (“A”) to basolateral (“B”) transport was increased, and B to A was decreased, thus diminishing the efflux ratio (0.99 and 1.01) ( Fig. 6 ). In some experiments using a somewhat different isolation protocol, the transcellular transport of digoxin and interaction of digoxin and PSC-833 were also investigated. In these experiments, however, the efflux ratio was below 1.0, and therefore, the results could not be interpreted (data not shown).
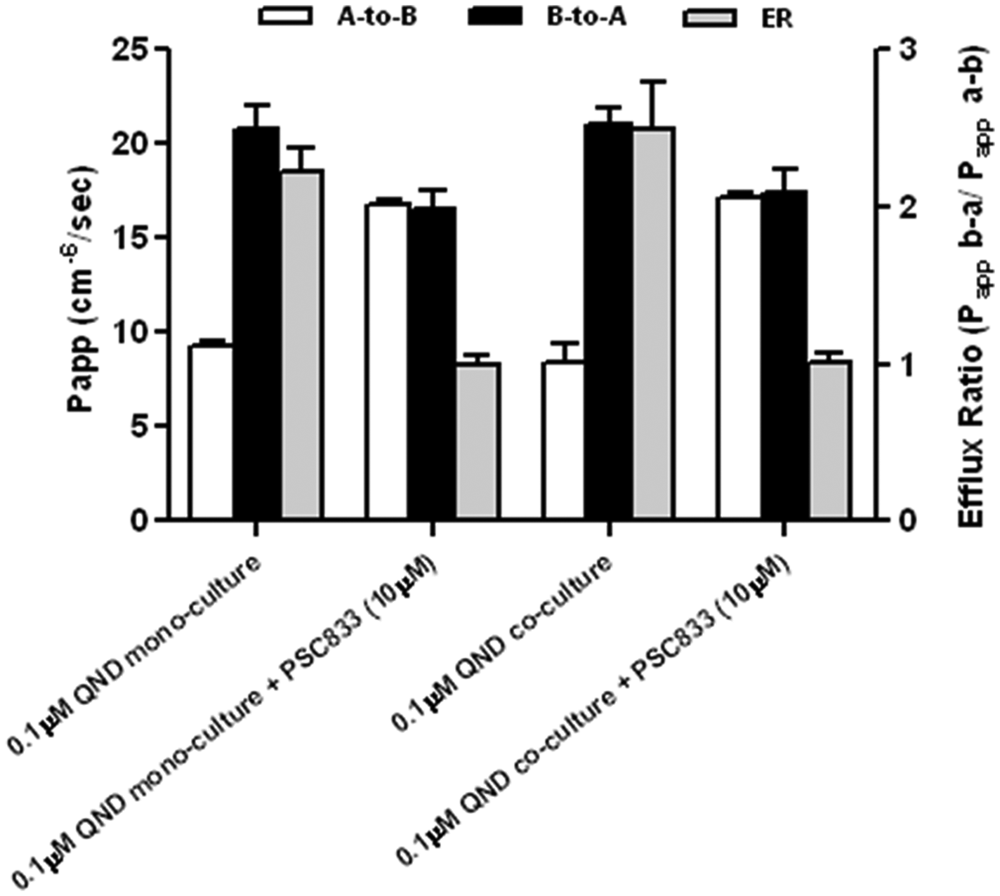
Modulation of P-gp–mediated vectorial transport of quinidine (0.1 µM) in the solo-cultured primary mouse brain endothelial cell monolayer (pMBMEC) (solo-cultured EC) and in pMBMEC co-cultured with astrocytes (co-cultured EC) monolayer by PSC-833 (10 µM).
Discussion
Previously, we have reported 11 that the dual-probe microdialysis approach is an effective tool to assess investigational drugs for P-gp interaction at the BBB in rats. Moreover, we provided evidence that QND–PSC-833 is a P-gp–specific probe substrate/reference inhibitor combination applicable in several assay systems in vitro and in vivo.
In the present study, we extended the application of simultaneous sampling of blood and brain by microdialysis and the validation of QND–PSC-833 as P-gp probe substrate/reference inhibitor combination in mice.
As there was no commercially available peripheral microdialysis probe for properly sampling blood in the mouse jugular vein, consultations with different vendors and in-house pilot studies were required. Finally, an appropriate peripheral/brain microdialysis probe combination was identified in collaboration with Microbiotech/se AB (Stockholm, Sweden). The in vitro feasibility study demonstrated ( Fig. 1 ) that this pair of probes can be used for determining levels of QND in dialysate samples from blood and brain in mice. Our results show that the diffusion of QND is symmetrical both in the brain and in the peripheral probe with a 3-mm membrane. The in vitro recoveries of Microbiotech probes for QND are comparable with the in vitro recovery of CMA12/2-mm probes for QND. 25
In the present study, microdialysis experiments were performed in both anesthetized and awake mice. Each has advantages and disadvantages that need to be considered when planning experiments or interpreting the data. Experiments are easier to perform and less uncomfortable in anesthetized mice. It may be beneficial to eliminate sensory or behavioral influences during the experiments. On the other hand, microdialysis experimentation in awake mice eliminates the influence of anesthetic drugs, allows for a longer sample collection period, and even allows for repeated drug administration. However, the awake animal experiments are more challenging because the moving animal can damage the probe membrane inserted into the jugular vein. Several preliminary experiments and small surgical modifications were needed to exclude the factors that endangered the integrity of the membrane of the vascular probe.
Simultaneous sampling of brain and blood by microdialysis techniques in mice applied in this study is a novel approach for investigating drug-drug interactions (DDIs) at the BBB. In our previous study, 11 the brain penetration of QND was characterized by a brain-to-blood ratio generated from the AUC values of concentration-time profiles of microdialysate samples from the brain and blood of anesthetized rats. This value was ~0.3 for the controls and ~1.0 for the PSC-833–pretreated rats. 11 In the present study in anesthetized control mice, the brain-to-blood ratios were much lower than in rats. The AUCbrain to AUCblood ratio was significantly increased (from 0.04–0.17) in the PSC-833–treated mice at the lower QND dose ( Fig. 2 ). In awake mice, the increase of the AUCbrain to AUCblood ratio in PSC-833–treated animals (from 0.22–0.99) was similar ( Fig. 5 ) to the increase observed in rats. 11
Kusuhara and coworkers 10 described that in mdr1a knockout mice, the brain-to-plasma concentration ratio of QND was more than 27-fold higher than in wild-type mice. This value is greater than the value observed in our study. Nevertheless, it indicates the essential role of P-gp (mdr1a) in the transport of QND at the blood-brain interface. Species differences such as the difference observed in brain-to-blood levels of quinidine between mouse and rat have been reported for other P-gp substrates. The brain-to-blood ratio of colchicine for control and PSC-833–treated freely moving rats (measured in the extracellular fluid of the frontal cortex and in a peripheral vein) were 0.04 and 0.15, respectively. 27 In contrast, in another study, the brain-to-blood ratio of AUCs for colchicine in hippocampal extracellular fluid was 0.14 in awake mice, 23 indicating again a possible species difference in the function of P-gp. At a quantitative level, excellent correlation was found between CsA-induced elevation of 11C-verapamil brain levels between rats and humans. 28 Similarly, an excellent correlation was reported between the CsA-induced increase of 11C-loperamide levels in the two species. 29
Similar to experimentation in rats, for testing in vivo–in vitro correlation, an in vitro mouse BBB model (pMBMEC monolayer) was also observed. Astrocyte co-culture was included to mimic the brain microenvironment. Rat astrocytes were chosen in preference to mouse astrocytes due to ease of culture and higher yields. QND transport through the monolayer in the presence and absence of PSC-833 was compared in the mouse endothelial cell monolayer and in co-culture with astrocytes. A PSC-833–dependent vectorial transport of quinidine was clearly shown. Vectorial transport of digoxin in this pMBMEC system could not be observed, similar to the RBEC monolayer assay in our previous study. 11
Reports on DDI at the blood-brain barrier leading to modulation of brain exposure are scarce. 30 Based on data generated in rodents, it is likely that there are more drug-drug interactions leading to significantly elevated brain levels than DDIs at the plasma level, since in genetic or chemical P-gp knockouts, brain levels go up sometimes a magnitude higher than plasma levels do.16,27 But brain levels are not routinely measured at the clinics. The only tool applicable to humans is measurement of CSF levels. However, P-gp is oppositely oriented in the blood-CSF barrier (pumping substrates toward CSF), and hence data are controversial (reviewed in Eyal et al. 30 ). The only relevant method is imaging and, indeed, P-gp–mediated 11C-verapamil–CsA 28 and 11C-loperamide–CsA 29 interactions at the BBB have been shown in humans.
In summary, an in vivo–in vitro assay system previously used in rats was extended to mice. Here we provided further evidence that QND and PSC-833 are a good substrate/reference inhibitor combination in testing candidate drugs for DDIs at the BBB. Considering the increasing number of humanized transgenic mice, a test system with mouse microdialysis experimentation becomes more important to predict drug-drug interactions in humans.
Footnotes
Acknowledgements
Expert help by Gyuláné Berekhelyi in the experimental procedures, as well as help from William Johnson, PhD, and Timea Rosta, MSc, in the preparation of the manuscript is acknowledged.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research leading to these results has received funding from the European Union’s Seventh Framework Programme (FP7/2007-2013) under grant agreements no. 202213 (European Stroke Network), as well as grant GOP1.3.2-09-2010-0008 (supported by the Hungarian National Development Agency under the Economic Development Operational Programme, industrial R&D capacity development priority) and Xenobiotic Transporter Technology Platform Therapeutic and Toxicological Applications (XTTPSRT1, OM-00230/2005).