Abstract
Translation initiation is a fine-tuned process that plays a critical role in tumorigenesis. The use of small molecules that modulate mRNA translation provides tool compounds to explore the mechanism of translational initiation and to further validate protein synthesis as a potential pharmaceutical target for cancer therapeutics. This report describes the development and use of a click beetle, dual luciferase cell-based assay multiplexed with a measure of compound toxicity using resazurin to evaluate the differential effect of natural products on cap-dependent or internal ribosome entry site (IRES)–mediated translation initiation and cell viability. This screen identified a series of cardiac glycosides as inhibitors of IRES-mediated translation using, in particular, the oncogene mRNA c-Myc IRES. Treatment of c-Myc–dependent cancer cells with these compounds showed a decrease in c-Myc protein associated with a significant modulation of cell viability. These findings suggest that inhibition of IRES-mediated translation initiation may be a strategy to inhibit c-Myc–driven tumorigenesis.
Introduction
Protein synthesis is a tightly regulated process that enables posttranscriptional control of gene expression and represents a critical point allowing cells to rapidly and efficiently respond to changes in their cellular environment. 1 Deregulation of this process is associated with the development, the progression, and even the chemoresistance of cancers. 1 The expression of key factors of translational machinery has been shown to be deregulated in many tumors (e.g., eIF4E). 2 These highlight the possibility that protein synthesis represents an area with potential as a target for drug discovery. 3 The discovery of small-molecule regulators of protein synthesis, often initially used as anti-infective agents, has provided important tool compounds helping to explore the translation pathway in both prokaryotes and eukaryotes. The identification of protein synthesis inhibitors, such as cycloheximide 4 and, more recently, hippuristanol 5 or silvestrol, 6 has been used to get further insight into the functions of translation factors and to isolate intermediate complexes of the translation process. A number of experimental cancer therapeutics modulating translation are already in clinical trials (e.g., inhibitors of the eIF4F complex, the mTOR pathway, or the MNK proteins). 3
In eukaryotic cells, translation of mRNAs into protein is a complex process that can be divided into three stages: initiation, elongation, and termination. 7 Translational initiation, a rate-limiting step, represents a critical point allowing the cell to respond to changes in the environment, 1 thus regulating cellular homeostasis, proliferation, differentiation, or even malignant transformation. Translation initiation can occur by two different mechanisms: a cap-dependent or a cap-independent mechanism. Under normal physiological conditions, the translation machinery (40S ribosomal subunits and translation initiation factors) is recruited to the mRNAs via the 5′ cap structure and scans the 5′ untranslated region (5′ UTR) of the mRNA until an appropriate AUG initiation codon is encountered. 7 The recruitment of the 60S ribosomal subunit enables translation elongation to start. Some cellular mRNAs recruit ribosomes via an alternative cap-independent mechanism directed by internal ribosome entry site (IRES) elements in their mRNAs’ 5′ UTRs. 1 IRES elements have been found in several cellular mRNAs, where they are thought to have a role in maintaining translation under conditions where global translation is impaired (e.g., mitosis or under stress conditions such as hypoxia or radiation). 1 These IRES elements are present in mRNAs containing long 5′ UTR that can fold to form a functional RNA secondary structure that drives translation initiation by directly recruiting the ribosome in close proximity to the initiation codons. Such cellular IRES elements are known to require canonical translation factors as well as specific trans-acting factors for their function. 8 The cellular IRESs identified so far tend to be in key regulatory genes whose protein products are required during pathophysiological processes (e.g., in cancer cells), where the regulation of cap-dependent translation is impaired, for example, during genotoxic stress. 9 Overexpression of eIF4E enhances translation of several IRES-containing mRNAs, such as vascular endothelial growth factor (VEGF), fibroblast growth factor 2 (FGF2), and c-Myc, which have been shown to have a direct role in cancer. 10 Mutations in IRES sites may also have a role in driving cancer progression. For example, a point mutation in the c-Myc IRES, shown to increase IRES-directed translation by enhancing the binding of IRES trans-acting factors (ITAFs), has been found in patients with multiple myeloma. 11 These findings indicate that secondary structures in the mRNA 5′ UTR play a critical role in translation regulation in normal physiology and pathophysiological diseases. 12
Many previous studies of IRES elements were conducted using bicistronic constructions, containing the IRES cloned in between two reporter genes. 13 These vectors allow the expression of two reporter proteins: the first one under the control of the 5′ cap and second one under the control the IRES element. Such constructs isolate the 5′ UTR RNA secondary structure from the 5′ cap and so facilitate the study of its function in directing translation initiation. The expression level of both proteins enables one to compare the effect of cell culture conditions or treatments on both cap-dependent and cap-independent translation from the same sample. Moreover, such a bicistronic reporter gene system was used in a high-throughput screening (HTS) campaign performed to identify compound regulating the cap-dependent or the viral IRES hepatitis C virus (HCV)–dependent translation. 14
This report describes a cell-based HTS campaign implemented to identify novel tool compounds that act to specifically modulate, either positively or negatively, the activity of an IRES element. The screening assay was designed to report on three different assay readouts from one sample, 15 monitoring compound cytotoxicity as well as compound effects against cap-dependent and IRES-mediated translation. Ultimately, this study identified cardiac glycosides as impairing IRES-mediated translation from the IRES element present in the c-Myc 5′ UTR. This inhibition was observed when monitoring IRES-directed translation initiation in both bicistronic and monocistronic reporter constructs. Further testing of these compounds in c-Myc–dependent and c-Myc– independent cells lines showed reduced synthesis of the c-Myc protein, which appears associated with reduced proliferation/survival and increased apoptosis level of these c-Myc–dependent cancer cells. This discovery provides another link between cancer cell proliferation, IRES-mediated translation, and protein synthesis regulation by cardiac glycosides.
Materials and Methods
Plasmid
Amplification of genes and target IRES by PCR and construction of all plasmids are described in detail under the Supplementary Methods. Click beetle red (CBRluc) and green (CBGluc) luciferases and IRES elements were subcloned in the pCR II-TOPO plasmids (#K4510-20; Invitrogen, Carlsbad, CA). The pCBRluc-IRES-CBGluc plasmids contain target IRES were cloned between the two reporter genes click beetle red (CBRluc) and click beetle green (CBGluc) luciferases in the pcDNA5-FRT plasmid. The pCBRluc-IRES-CBGluc plasmids contain the IRES element being studied cloned between the two click beetle reporter genes in the pcDNA5-FRT plasmid which was obtained from Invitrogen.
Cell Culture
All cell culture reagents were provided by Invitrogen unless otherwise stated. Flp-In HEK293 cells (Invitrogen) were cultured in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% heat-inactivated fetal bovine serum, 2 mM L-glutamine, 4.5 g/L glucose, 50 µg/mL hygromycin, 50 U/mL penicillin, and 50 µg/mL streptomycin. c-Myc–dependent (A2780) or c-Myc–independent (Caov3 and COV318) cancer cell lines were culture respectively in RPMI or DMEM each supplemented with GlutaMAX, 10% heat-inactivated fetal bovine serum, 4.5 g/L glucose, 50 U/mL penicillin, and 50 µg/mL streptomycin. Cells were incubated in a 100% humidified atmosphere of 5% CO2 at 37 °C.
Compound Treatment
The compound collection used in the high-throughput screen consisted of 14 000 natural products from a proprietary library (Novartis Pharma AG, Basel, Switzerland). Rapamycin and silvestrol were provided by the Novartis Compound Archive as powder and diluted in 90% DMSO and 10% H2O. Emetine (219282; Sigma, St. Louis, MO), cycloheximide (C104450; Sigma), and benzalkonium chloride (B6295; Sigma) powders were diluted in 90% DMSO and 10% H2O. Cells were seeded 16 h before treatment, as described in the Results, in 30 µL of medium in white 384-well microplates (354661; BD, Franklin Lakes, NJ) or 5 µL of medium in white 1536-well microplates (789183-A; Greiner Bio-One, Frickenhausen, Germany) using a Multidrop dispenser (Thermo Scientific, Waltham, MA). Compounds, dissolved in 90% DMSO and 10% H2O, were added in culture medium at a required final concentration by acoustic energy using the Echo 550 liquid transfer station (Labcyte, Sunnyvale, CA) as described in the Results. Cells were incubated with compounds for up to 24 h. A control dilution of 90% DMSO and 10% H2O was used as neutral compound (i.e., without any effect on the assay system), and benzalkonium chloride was used as the positive control for compound effects on cell viability and inhibition of the expression of both reporter genes.
Luciferase Reporter Monitoring
Click beetle red (CBR) and chick beetle green (CBG) luciferase expression monitoring was performed using the Chroma-Glo Assay System (E4920; Promega, Madison, WI) following the manufacturer’s instructions. In all experiments, the luminescence was measured using the Wallac ViewLux microplate imager (1430-0010; PerkinElmer, Waltham, MA) using 530-nm and 610-nm emission wavelength filters for respective monitoring of CBG and CBR luminescence.
To eliminate nonspecific inhibitors of luciferase enzyme activity, or light quenchers, compounds validated in a concentration response were tested again using the same cell line at concentrations from 0 to 10 µM. However, the compounds were added 5 min after cell lysis using the Chroma-Glo reagent. The plates were incubated for 30 min at 37 °C and the luminescence was monitored as described previously. Since compounds are added after the luciferase substrate, luciferase inhibitors and light quenchers should have an immediate effect on the luminescence, whereas compounds having an effect on the cell physiology should not.
Cell Viability, Apoptosis, and Cytotoxicity Assays
A stock solution of 4.4 mM resazurin was prepared in 100% DMSO. For each experiment, fresh dilutions of 7.5 µM resazurin were prepared in 5 mM Tris-HCl (pH 5.5). Cells were incubated with a final concentration of 1.5 µM resazurin for between 30 min and 1 h. Resofurin fluorescence was measured on the EnVision Multilabel Reader (PerkinElmer) using 560-nm excitation and 590-nm emission wavelengths.
Cell apoptosis, cytotoxicity, and viability were performed in 1536-well assay plates using the triplex ApoTox Glo kit (G6320; Promega) following the manufacturer’s instructions.
Screening Assay Procedure
A set of ~14 000 natural product compounds was tested against the luciferase expression in a 1536-well format in “triplicate” as described in
Screening Data Analysis
Assay quality was defined using the Z′ factor with two control compounds: DMSO as the neutral compound and benzalkonium chloride as the active compound, which kills the cells, using the following equation: 1 − [(3 × SD positive control) − (3 × SD negative control)/(mean positive control − mean negative control)], where SD is the standard deviation. 16 Screening results were analyzed using an in-house developed software, and data were compared with OriginPro 8.1 (OriginLab Corporation, Northampton, MA) and Spotfire (TIBCO, Somerville, MA) software. The luminescence value for each sample was corrected using the neutral and active controls as 0 and −100% residual activity. Potential hits were considered active by the following criteria: samples showing CBG luminescence less than −50% residual activity (for inhibitors) or greater than 50% residual activity (for activators) with a CBR luminescence and a compound toxicity remaining between −25% and 25% activity.
IC50 Calculations
To determine the relative IC50s of candidate hit compounds, an 8-point titration of each compound was assessed in quadruplicate with a 10-µM compound concentration as the upper limit. Percent activation or inhibition of luciferase synthesis at each concentration was calculated relative to assay plate control wells: DMSO as the neutral compound and benzalkonium chloride as the active compound killing the cells. Averages from quadruplicate measurements were fitted to generate the dose-response curve and determine the relative IC50 concentration for each compound using the OriginPro 8.1 software.
Ligation-Mediated Microsphere Flow Cytometery Profiling
LMF, a method very similar to that described previously by Peck et al., 17 was used to profile the transcriptional signature of a focused set of 100 responsive genes, implicated in the regulation of a number of major cell pathways to characterize the cellular response of compound treatment. The transcription profile monitored in response to hit treatment was then compared with a database of profiles that have been generated using a set of known reference/control compounds. This method then suggests compounds with known targets that cause a similar transcription profile.
Results
Rationale, Design, and Development of the Screening Assay
The objective of this project was to identify compounds that act specifically on the regulation of IRES-mediated translation (
The reporter gene assay used for the primary screen was multiplexed to monitor compound cytotoxicity as well as compound activity against both cap-dependent and IRES-directed translation. The monitoring of three data points from a single sample should increase the probabilities to select a nontoxic active series of compounds. This should facilitate the selection and testing of additional compounds from the Novartis compound collection. The assay multiplexing was possible by using first resazurin, assessing the cell viability to monitor the compound toxicity,
18
and second the dual luciferase expression system, which uses two variants of the click beetle luciferase that emit light at different wavelengths (
To investigate assay reliability for screening, different tests were performed using the pCBRluc-cMyc-CBGluc cells. First, the monitoring of the influence of the IRES element on translation showed that, under normal cell culture conditions, all the IRES elements increased the expression of CBGluc above background (i.e., a construct containing no IRES insert), with the largest signal being observed for EMCV IRES ( Fig. 1A ) as previously shown. 19 Then, the effect of known translation inhibitors was tested. For the bicistronic construct containing the c-Myc IRES element, emetine or cycloheximide inhibited the expression of both reporter genes (cap dependent and IRES mediated), with an equal potency associated with a minor effect on cell viability after the test period of 24 h ( Fig. 1B , C and Table 1 ). Rapamycin was found to inhibit expression of both reporter genes with a similar potency but with incomplete inhibition, suggesting that mTOR influences both cap-dependent and cap-independent translation with similar but weak efficiency ( Fig. 1D and Table 1 ). Finally, silvestrol showed inhibition of expression for both reporter genes but with almost a 10-fold higher potency for inhibition of the cellular IRES-mediated translation than for inhibition of the cap-mediated translation ( Fig. 1E and Table 1 ). These results are consistent with the explanation that the reduction of an available and functional eIF4F complex would affect translation from mRNA with highly structured 5′ UTR. 20 Similar data were obtained when testing these compounds on the expression of reporter genes from the bicistronic construct containing the VEGF or the EMCV IRES elements (data not shown).
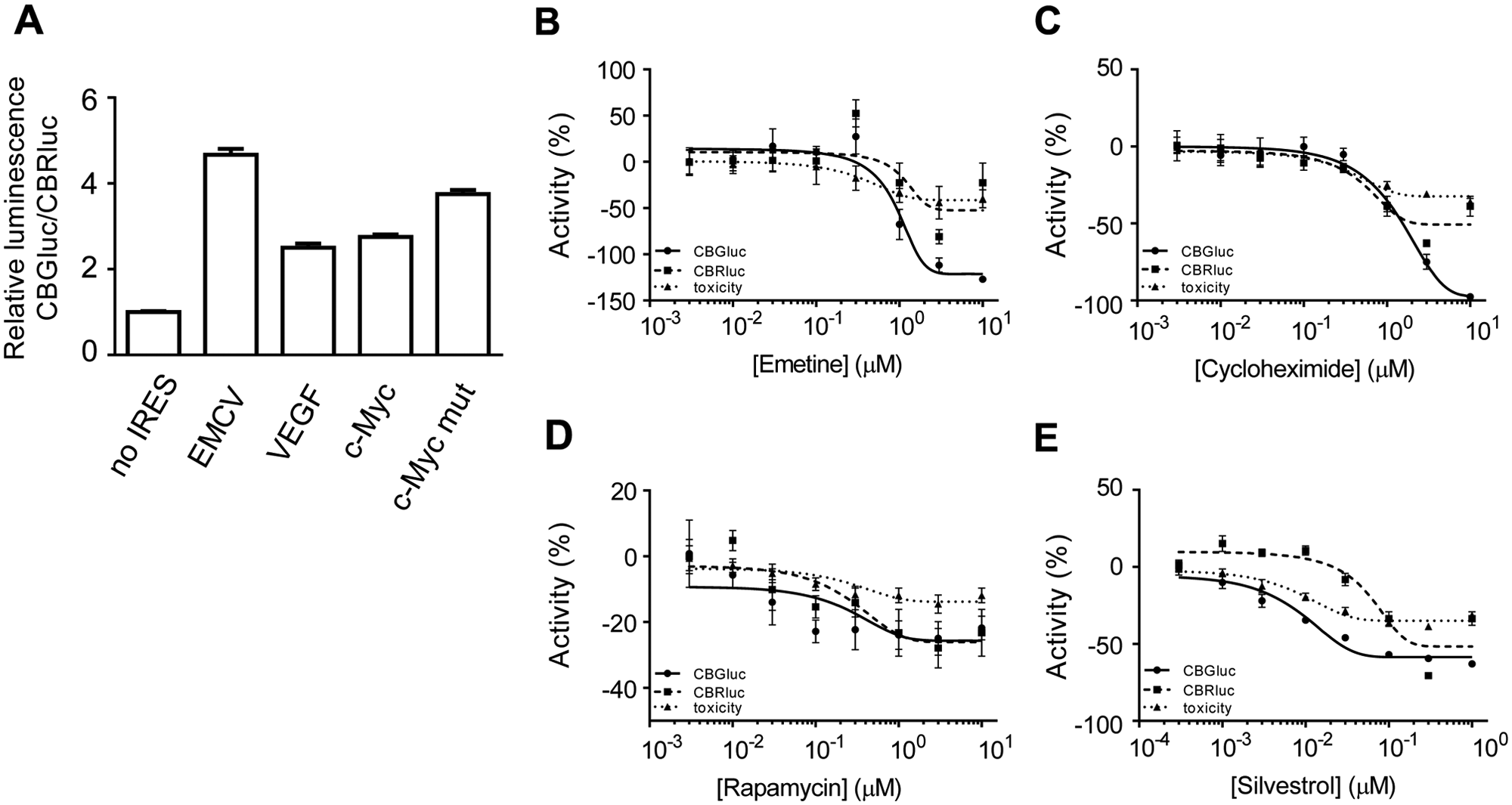
Screening assay development controls. (
Calculated Half-Maximum Inhibitory Concentration (IC50) for All Four Cardiac Glycosides’ Toxicity and Activity on Cap-Dependent and IRES-Mediated Translation.

Values are presented as mean ± SD. An estimate of experimental error is given by calculating the standard deviation of the mean with n = 3. IRES, internal ribosome entry site.
Compound Screen Identifies c-Myc and VEGF IRES-Mediated Translation Modulators
To identify positive or negative modulators of the expression of IRES-directed translation, a set of ~14 000 natural compounds was tested at a single concentration of 5 µM as described in
The primary screening data showed that, as expected, the vast majority of the compounds were inactive (
In total, 175 compounds were identified to modulate the c-Myc–driven CBGluc expression and 118 compounds identified to modulate the VEGF-CBGluc expression with an overlap of 58 compounds; 235 compounds identified as either activators or inhibitors of CBGluc expression, driven by the c-Myc or VEGF IRES elements, were then selected for validation. These hits were evaluated in more detail against all three readouts by measuring their activity in concentration dose response ranging from 3.16 nM to 10 µM. Of these initial hits, 123 were confirmed as active in a concentration-response assay. The EC50 (for modulation of IRES activity) values for these confirmed hits ranged from 0.2 nM to more than 17 µM. This validation was performed with the same bicistronic cell lines used for primary screening, showing that only a minor fraction of the confirmed hits displayed toxicity or modulated the CBRluc reporter by more than 50%. Most of the hits showed activity only against the CBGluc reporter gene expression, either by activation or inhibition, without showing any effect on CBRluc activity or cell viability. Nonspecific inhibitors of luciferase enzyme activity, or light quenchers, were excluded from further analysis.
To further characterize the compound selectivity for the cellular IRES, hits confirmed to modulate the cellular IRES activity were tested by monitoring their effect on CBGluc expression under the control of the viral EMCV IRES cloned in a bicistronic context. Interestingly, we observed that several compounds were not specific for just the cellular IRES but regulated both the EMCV and the cellular IRES with almost similar potency (data not shown). This might reflect that the targets of these compounds are cellular factors required for expression of both viral and cellular IRES elements. A number of the hits did show a higher potency or even a clear selectivity for inhibition of the cellular IRES compared with inhibition of EMCV IRES element activity (
Cardiac Glycosides Specifically Modulate IRES-Mediated Translation
To confirm the compounds’ effect on translation, further experiments were performed in cell lines containing the monocistronic reporter constructs driven by the IRES element cloned in a more physiological context (
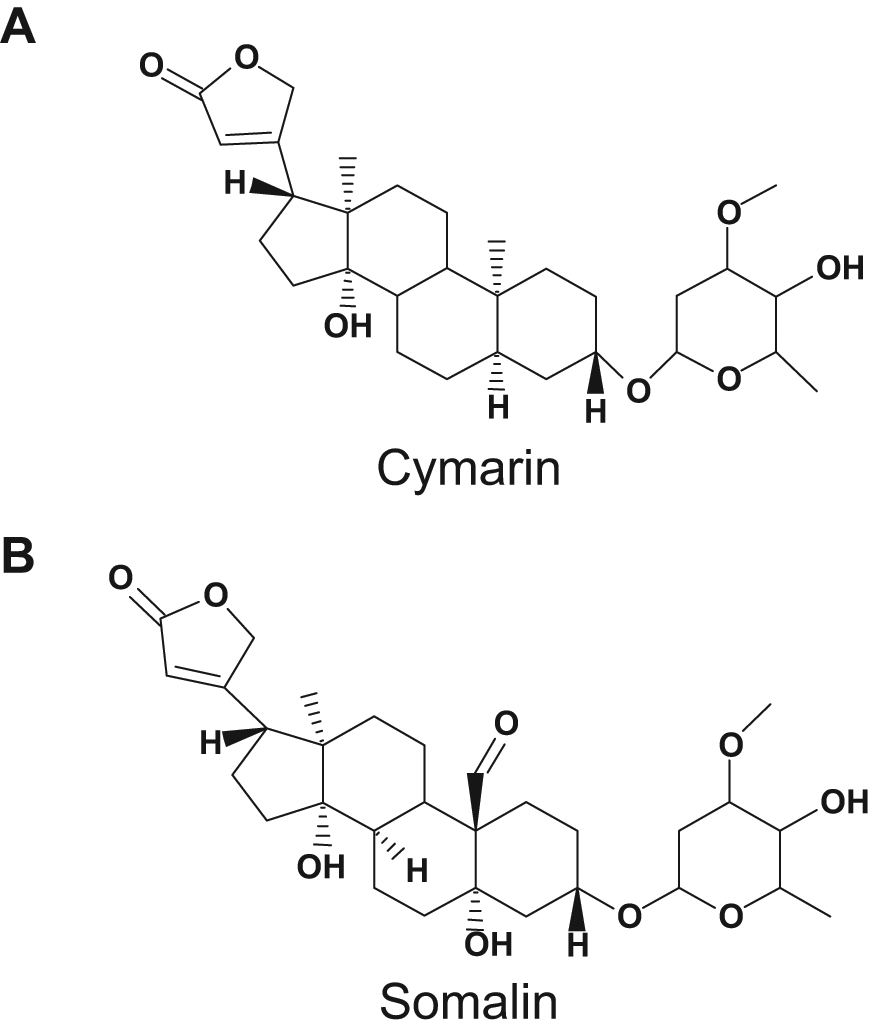
Cardiac glycoside inhibits c-Myc internal ribosome entry site (IRES)–mediated translation. Chemical structure of cymarin (
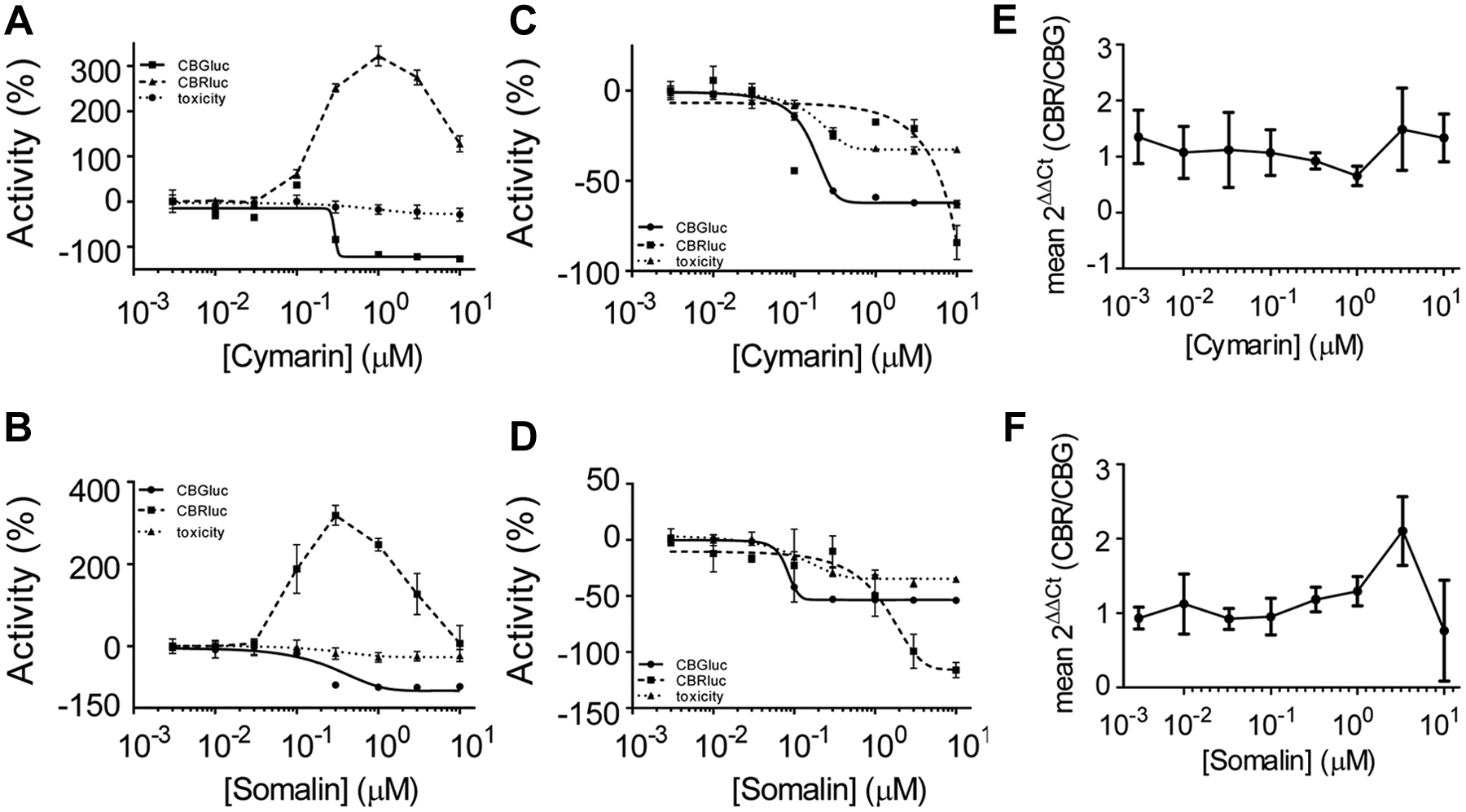
Cymarin and somalin inhibit the reporter gene expression at the level of translation. (
Calculated Half-Maximum Inhibitory Concentration (IC50) in the Various Cell-Based Assays Detailed for Cymarin and Somalin Toxicity and Activity on Cap-Dependent and IRES-Mediated Translation.
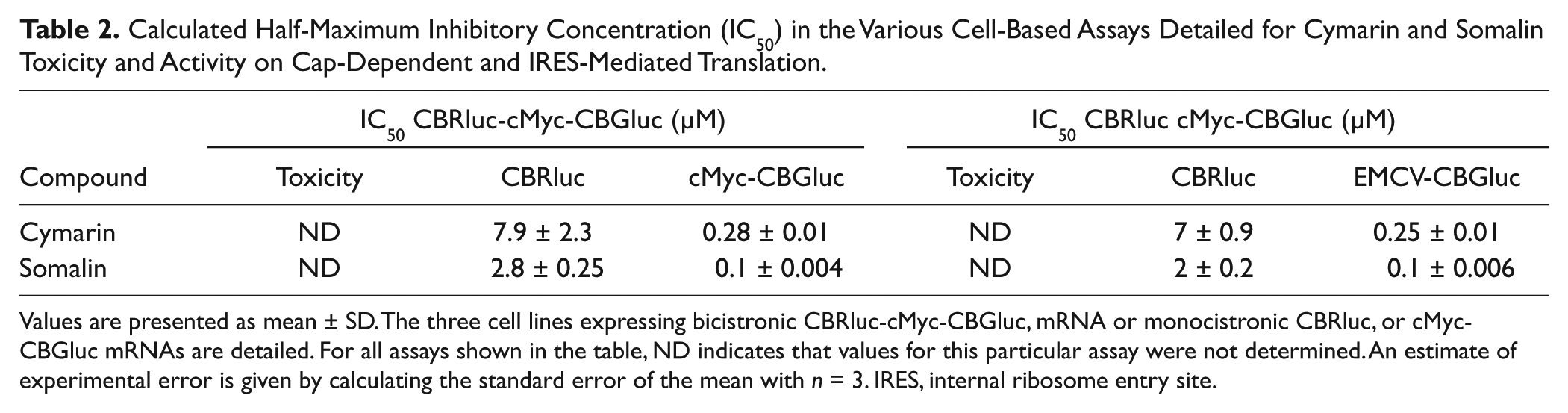
Values are presented as mean ± SD. The three cell lines expressing bicistronic CBRluc-cMyc-CBGluc, mRNA or monocistronic CBRluc, or cMyc-CBGluc mRNAs are detailed. For all assays shown in the table, ND indicates that values for this particular assay were not determined. An estimate of experimental error is given by calculating the standard error of the mean with n = 3. IRES, internal ribosome entry site.
To analyze the effect of cardiac glycosides on transcription, an experiment monitoring the effect of compound on a panel of 100 different gene transcript levels, implicated in major cell pathways, using an assay protocol similar to that of Peck et al.,
17
was performed (
Cardiac Glycosides Modulate IRES-Mediated Translation during Genotoxic Stress
Previous studies have reported that under conditions of cellular stress (such as UV exposure, hypoxia, genotoxic damages, or heat shock), cap-dependent translation is impaired while the IRES-mediated translation is less affected. 1 Thus, stress conditions induce a relative increase of IRES-directed compared with the cap-directed translation. In particular, after treatment of cells with DNA-damaging agent mitomycin C (MMC), cMyc IRES-driven translation still occurs. 9 To investigate whether the translational response of IRES activity to stress might be influenced by the hits, the compound effect on the CBRluc and cMyc-CBGluc reporter genes was tested under stress conditions ( Fig. 4 ). First, the cell lines were treated with 10 µM MMC, and the time courses of cell viability and luciferase expression, monitoring at regular time points over 24 h, were performed ( Fig. 4A ). Under such stress conditions, there was a greater inhibition of the cap-dependent CBRluc expression than c-Myc IRES-driven translation ( Fig. 4A ). After a 14-h incubation, the CBRluc signal was reduced by about 60%, whereas the CBGluc signal was only decreased by 15% ( Fig. 4A ). After a 24-h incubation, the differential expression of the reporter gene tended to be reduced with a CBG luminescence decrease of about 50% and the CBRluc by about 70%, without an effect on cell viability as monitored using resazurin ( Fig. 4B ). Similar results were obtained using a 20-µM MMC treatment (results not shown). The effect of the cardiac glycosides on the IRES-directed translation was then tested under stress conditions. Following treatment with compounds for about 6 h, 10 µM MMC was added to the cell media between 16 and 18 h prior to monitoring luminescence. Pretreatment of cells with cardiac glycosides decreased cMyc-CBGluc expression and thus abolished the apparent increase in IRES-dependent translation even in the presence of the DNA-damaging agent ( Fig. 4C , D ). Similar results were obtained for the VEGF-CBGluc–directed gene expression (data not shown). Similar to CBGluc expression, the cell viability was affected by cotreatment of cells with MMC and cymarin or somalin, as monitored by resazurin, indicating cardiac glycosides might sensitize cells to stress treatment. These results indicate that the cardiac glycosides inhibit IRES-mediated translation under conditions of stress.
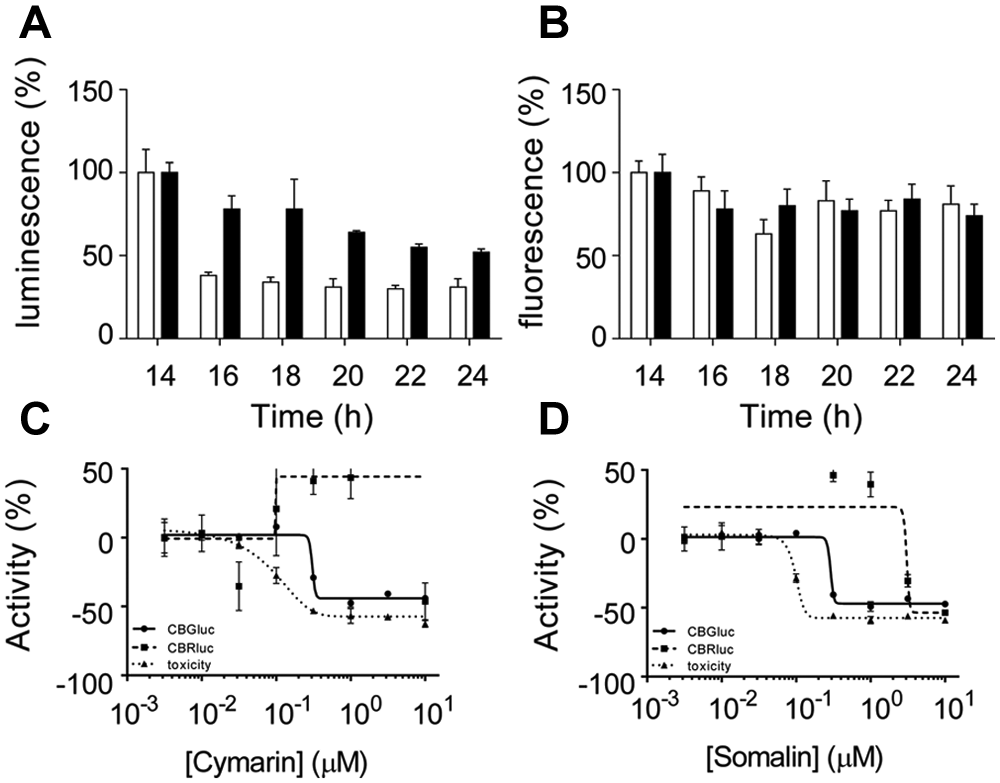
Cymarin and somalin abolished internal ribosome entry site (IRES)–mediated translation under stress conditions. (
Cardiac Glycosides Inhibit the Proliferation of c-Myc–Dependent Cancer Cell Lines
The hits shown to modulate the reporter gene IRES-CBGluc expression under the control of the c-Myc IRES element were then tested in the context of MYC dependency. Ovarian cancer cells A2780 and Caov3 were characterized for their dependence on c-Myc for proliferation rate (
To confirm that the cardiac glycosides have an effect on c-Myc expression, c-Myc mRNA and protein levels were monitored in these cancer cell lines ( Fig. 5 ). A2780 and Caov3 cancer cells were treated with DMSO or cardiac glycosides for 24 h. Total protein was extracted, and an immunoblot of A2780 cells lysates revealed that a decrease of c-Myc expression correlated with the increase of cymarin concentration, whereas the tubulin expression was not affected ( Fig. 5A ). Similar effects were obtained with an increasing concentration of somalin ( Fig. 5B ). Moreover, compared with the c-Myc protein basal level observed in the presence of DMSO, a decrease of c-Myc protein was observed in the presence of 100 nM cardiac glycosides (i.e., the determined IC50 concentration) ( Fig. 5C ), whereas levels of the c-Myc mRNA remained unchanged ( Fig. 5D ). Culture of Caov3 cells in similar conditions showed a high increase of c-Myc mRNA level in Caov3 cells, whereas the c-Myc protein level remained undetectable (data not shown). These results demonstrate that cardiac glycosides seem to preferentially inhibit c-Myc expression, by blocking IRES-directed translation, in A2780 cells.
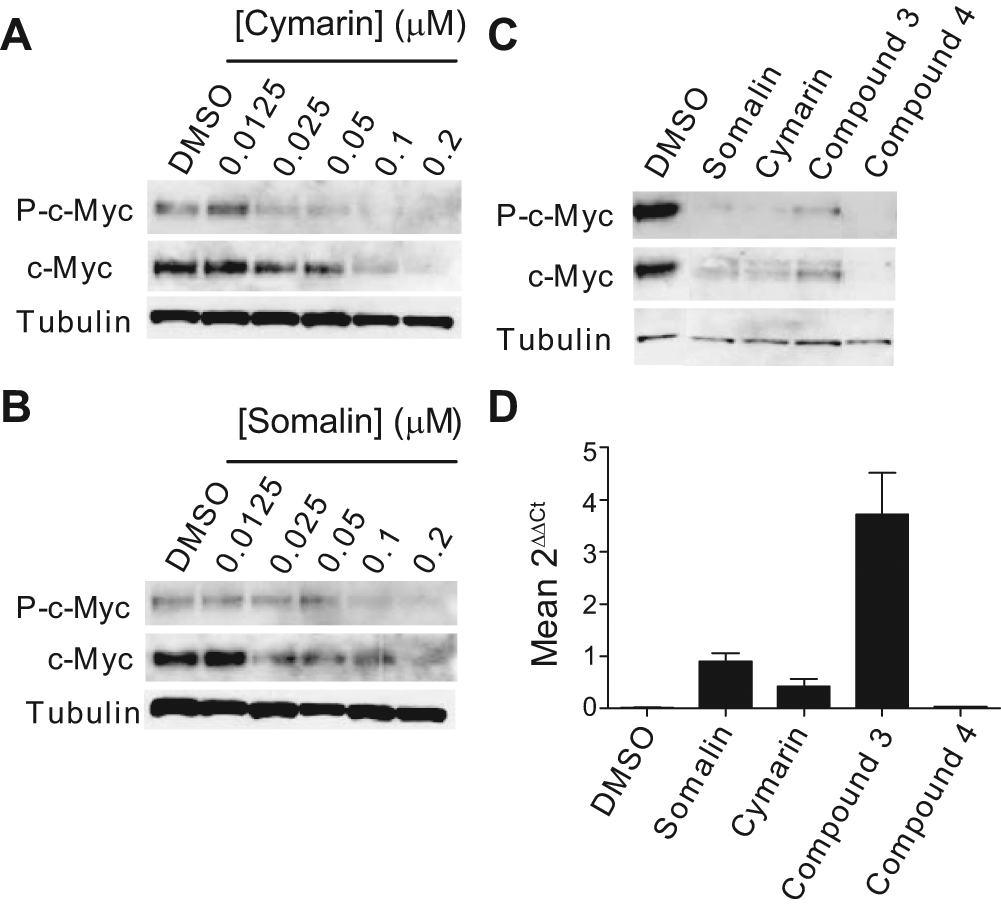
Cardiac glycosides affect c-Myc–dependent ovarian cancer cell proliferation/survival. (
The proliferation of these ovarian cancer cells lines was then tested in the presence of cardiac glycosides ( Fig. 6 ). A2780 and Caov3 cancer cells were treated with increasing concentrations of cymarin in a time course experiment; cell proliferation was then monitored after a 24-h, 48-h, and 72-h incubation with compounds. After 24-h incubation, 100nM cardiac glycosides triggered an induction of apoptotic caspase-3 ( Fig. 6A ). This induction was only associated with a slight effect on cell viability ( Fig. 6B ) or the number of intact cells as monitored by the vital dyes GF-AFC ( Fig. 6C ) or with resazurin ( Fig. 6D ). In all these assays, the Caov3 cells were less affected than the A2780 cells ( Fig. 6 A - D ). After a 48-h incubation, caspase 3 induction was induced with 100 nM compound in A2780 cells, whereas caspase was only weakly induced at 1 µM for Caov3 cells ( Fig. 6E ). This caspase 3 induction in A2780 cells was associated with a cytotoxic effect ( Fig. 6F ) and viability decrease ( Fig. 6G , H ), whereas Caov3 cells were less affected. After a 72-h exposure, the effects of cymarin were similar to those observed after a 48-h incubation for both cell lines, indicating a weak effect of cymarin on the proliferation/survival of c-Myc–independent cells in a longer time period ( Fig. 6I – K ).
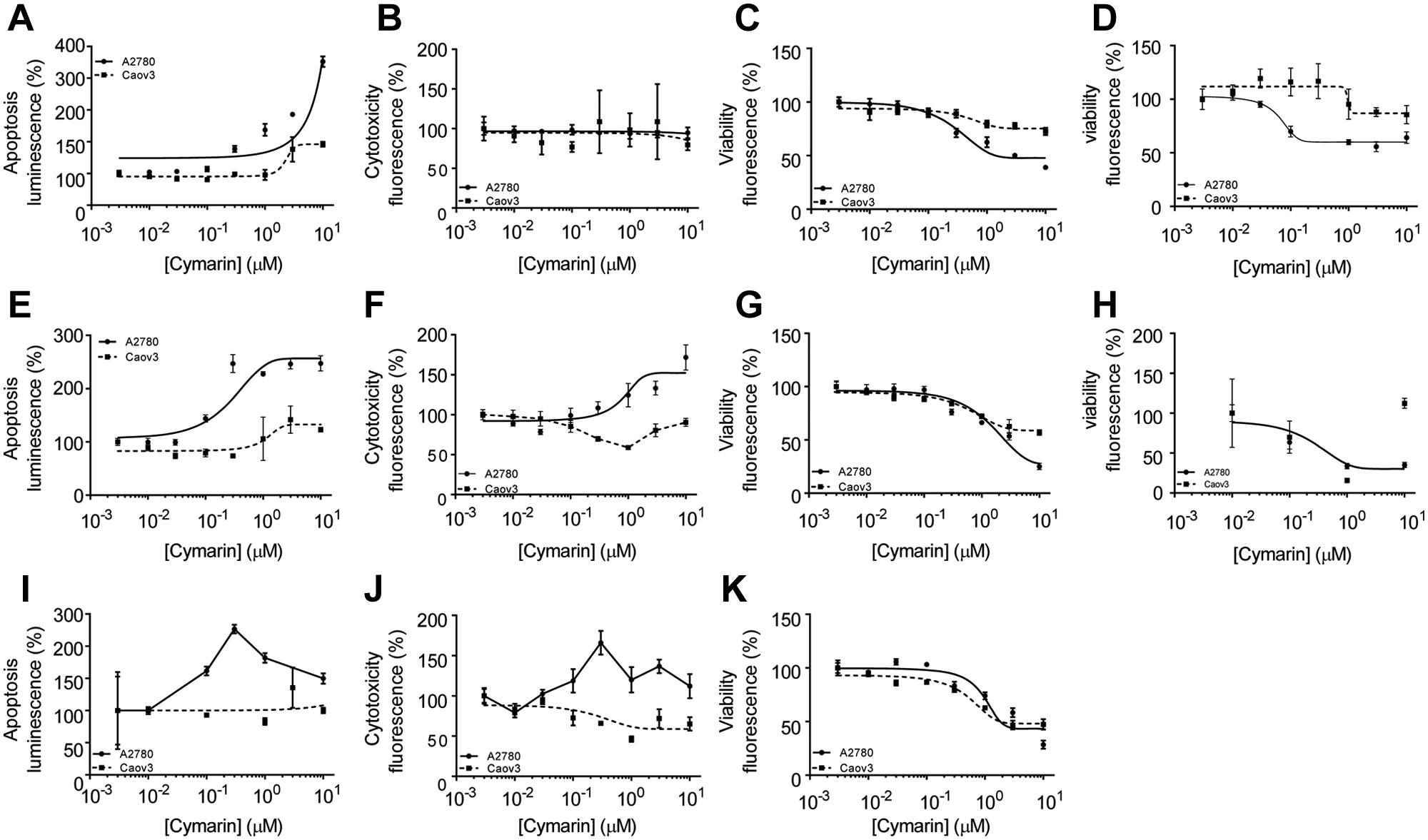
Cymarin cardiac glycoside induces apoptosis and affects the cell viability of c-Myc–dependent cancer cells A2780. A2780 and Caov3 cells were seeded in a 1536-assay plate and treated with an indicated concentration of cymarin for 24 h (
These results demonstrate that inhibition of c-Myc protein expression is possibly a critical mechanism by which cardiac glycosides affect the viability of A2780 cancer cells. Similar results on apoptosis, cytotoxicity, and cell viability were obtained for both cell lines with somalin, as well as the related cardiac glycoside compounds 3 and 4 (data not shown).
Discussion
The aims of this project were to screen a focused library of natural product compounds to study the regulation of the translational initiation from cellular IRES elements. To accomplish this, a cell-based, dual-luciferase HTS assay was designed and implemented. This cell-based, high-throughput reporter gene assay proved to be robust and performed well enough to allow screening in a miniaturized 1536-well format. The assay system potentially offers a number of advantages in addition to being able to monitor multiple assay readouts in a high-throughput format. By using two variants of the click beetle luciferase that differ in only one amino acid and still use the same chemical reaction for the generation of light, it is possible that artifacts due to luciferase/compound interactions (as previously described for firefly luciferase 21 ) will be reduced. In addition, the Flip-In cell lines used in this study contained a DNA-FRT site to allow integration of the expression plasmid at a unique genomic site, reducing the potential for clonal differences due to the plasmid integrating at different genomic sites.
The initial primary screen performed on HEK293-expressing bicistronic reporter genes was set up to allow the multiplexing of three different readouts for the simultaneous identification from one sample of (1) molecules affecting the cell viability, compounds that apparently modulate, by activation or inhibition, (2) cap-dependent or (3) IRES-mediated translation. Inhibitors of cap-dependent translation and toxic molecules were not analyzed further. However, a number of confirmation assays were conducted to validate specific modulators of IRES-mediated translation. The screen identified known cardiac glycoside compounds, such as cymarin and somalin, and two other similar compounds as potent inhibitors of IRES-mediated protein synthesis. Cardiac glycosides are a class of drugs derived from plants (e.g., the best known being Digitalis) that have been used to treat congestive heart failure and arrhythmias. Studies have suggested that cardiac glycosides have activity as anticancer agents. 22
Cardiac glycoside treatment of cells lines expressing a monocistronic version of the reporter genes, used to validate the compound effect on luciferase expression in more physiologic conditions, showed in a time-dependent and a concentration-dependent manner specific inhibition of IRES-mediated translation. Induction of genotoxic stress by mitomycin C treatment of monocistronic cells, showing a notable difference between cap-mediated and IRES-mediated reporter gene translation, was observed. This relative increase of IRES activity, induced by MMC, was abolished under pretreatment of cells with cardiac glycosides, suggesting that the affected target is specifically implicated in IRES-mediated translation.
Previous reports have described additive and synergistic antitumor effects of rapamycin in combination with other molecules. Here, combination of cardiac glycoside treatment with 1 µM rapamycin did not significantly potentiate the effect of cardiac glycosides on monocistronic reporter gene expression, cap dependent or IRES mediated (data not shown).
Another observation reported here is that these cardiac glycosides are potent cytotoxic agents in a c-Myc–dependent cancer cell line. Treatment of c-Myc–dependent ovarian tumor cell lines with cardiac glycosides induced a clear decrease of c-Myc protein, at the level of translation, associated with a cell viability defect, whereas a c-Myc–independent cancer cell line (Caov3) was less affected.
Although inhibition by cardiac glycosides of both IRES-mediated translation in general and c-Myc synthesis in particular is clearly a compound class effect, the mechanism of action of these drugs on IRES-mediated translation remains unclear. It has been previously shown that a mutant c-Myc IRES, identified in multiple myeloma cells, exhibits an increased c-Myc IRES activity associated with an increased c-Myc protein synthesis.
11
A recent study reports that the increase of mutant c-Myc IRES activity was found to be correlated with the expression of YB-1 and PTB, two proteins shown to be overexpressed in cancer cells, and to contribute to cell proliferation and tumorigenesis.
23
In this study, the c-Myc mutant IRES, regulating the CBGluc expression, did not modulate the cardiac glycoside compounds’ efficiency, except for compound 4, suggesting that the YB-1 and PTB proteins might not be targeted by cardiac glycosides under these assay conditions (
Cardiac glycosides have been shown previously to be potent inhibitors, at the level of translation, of the expression of the tumor suppressor p53 as well as the hypoxia-inducible transcription factor 1 (HIF-1) in hypoxic tumor cells.25,26 It was shown that digoxin very potently inhibits HIF-1 mRNA translation and affects mouse xenograft tumor growth associated with modest effects on global protein synthesis. It was also reported that p53 and HIF-1 mRNAs contain IRES elements.27,28 However, whether cap-dependent or cap-independent translation of p53 or HIF-1 is actually inhibited by cardiac glycosides is not known. As p53 and HIF-1 mRNAs contain an IRES element, cardiac glycosides might affect the HIF-1 as well as the p53 IRES-mediated translation. Because p53 is an important factor in triggering cell response to stress, understanding the mechanisms behind p53 translation regulation will lead to further advances in cancer research. Moreover, identification of mutations within the p53 IRES sequence may highlight new insights into the process of tumorigenesis and provide a new tool to assess clinical responses to chemotherapeutic agents and prognosis.
Cardiac glycosides represent starting pharmacophores upon which to improve biological activity with respect to both selectivity and potency. They also offer a pharmacological approach by which to regulate signaling nodes deregulated in cancer and provide a chemical genetic tool with which to explore translation and to regulate IRES element activity. A remaining challenge would be to identify the compound target and characterize their mode of action. mRNA translation is found at a point of convergence of oncogenic pathways and is a tightly regulated process with many components representing an attractive target for drug discovery.
In conclusion, it was shown that the alteration of the cellular functions caused by cardiac glycosides can have a profound effect on intracellular signaling pathways, including those regulating IRES-mediated translation. This effect may provide an exciting new approach to the treatment of human cancers, showing a dependency of cap-independent translation.
Footnotes
Acknowledgements
We gratefully acknowledge Gilles Weinzaepflen and Hervé Mees from the Novartis Compound Archive for compound plate preparation, Karoline Boesch and Jérôme Giovannoni for liquid chromatography/mass spectrometry analysis, the Cell-Based and Medium-Throughput Screening groups, and the High-Throughput Biology group at Novartis for helpful discussions and experimental help. We also thank Anne Willis for providing the pGL3- cMyc-mutant plasmid and Christian Thoma and Claudia Casanova for helpful discussions and suggestions to improve the study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Novartis Institutes of Biomedical Research and MCD is funded as a postdoctoral fellow by the NIBR Education Office.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
