Abstract
A shift from conventional cytology to a molecular approach could improve cervical cancer screening. This proof-of-concept study aims to develop a high-content imaging platform for the simultaneous detection of multiple biomarkers for cervical disease. Liquid-based cytology (LBC) samples were used to optimize a dual ProExC/Ki-67 immunofluorescence staining protocol for SurePath-fixed cells. The simultaneous and automated detection of these biomarkers was performed using the BD Pathway 435 system. The ability of high-content imaging to detect dysplastic cervical cells was assessed using keratinocytes spiked with immunopositive SiHa cells and a high-grade squamous intraepithelial lesion (HSIL) LBC sample. The percentages of Ki-67- and ProExC-immunopositive objects correlated significantly with the percentages of spiked SiHa cells. The dysplastic cells of the HSIL sample could be detected using high-content cell analysis. In conclusion, high-content imaging allows the simultaneous and automated detection of Ki-67- and ProExC-immunopositive dysplastic cells in LBC specimens.
Introduction
Cervical carcinoma is the third most prevalent cancer in women worldwide. In Western Europe, it is only the 15th most common cause of cancer death in women as a result of cytology-based cervical screening. 1 However, the efficacy of cytological screening is hampered by a high interobserver variability and high false-positive and false-negative rates.2,3 The recognition of the strong causal relationship between a persistent infection with high-risk human papillomavirus (HR-HPV) and cervical cancer has resulted in the introduction of HPV DNA testing in clinical practice.4–10 However, because of the high prevalence of transient infections, HPV detection shows a poor positive predictive value (PPV) for cervical disease.
Because both cytology and HPV DNA detection show limitations as standalone screening tests, disease-specific biomarkers of HPV-associated transformation are being developed and validated. Multiple viral and/or cellular markers could be detected in series or in parallel to prevent the unnecessary follow-up of transient HPV infections. In the routine screening setting, liquid-based cytology (LBC) samples would be well suited to test for a panel of biomarkers, but observer- independent marker detection would be required.
This proof-of-concept study aims to develop a technological platform for the simultaneous detection of multiple markers for cervical dysplasia. The approach is based on high-content imaging and uses multiwell plates for automated image acquisition and analysis. The biomarkers ProExC and Ki-67 were used to identify dysplastic cells in SurePath-fixed LBC specimens. ProExC is an antibody cocktail against topoisomerase II alpha (TOP2A) and minichromosome maintenance protein 2 (MCM2) proteins, two markers of aberrant S-phase induction. Ki-67 is a nuclear marker for cellular proliferation.
Materials and Methods
Cell Lines
The SiHa cervical carcinoma cell line, containing one or two copies of HPV16 per cell, was purchased from the American Type Culture Collection (Manassas, VA). Low-passage cultures (5–10 passages after purchase) were used. Cells were harvested at 80% confluence and seeded in imaging plates or fixed in 10 mL SurePath (BD Diagnostics, Burlington, NC) before seeding.
Patient Samples
LBC samples with normal cytology or high-grade squamous intraepithelial lesions (HSIL), fixed in SurePath preservative fluid for 1 to 3 days, were randomly selected in the Department of Cytopathology and Molecular Diagnostics (Riatol, Sonic Healthcare Benelux, Antwerp, Belgium). To remove debris, mucous, and inflammatory cells, the clinical samples were subjected to the BD SurePath Cell Enrichment process. The enriched cellular fraction was resuspended in 24% ethanol.
The study was performed in accordance with the guidelines of the ethical committee of the University of Antwerp (study identification number 11/41/290). Study-specific patient identification codes were assigned to each sample in accordance with patient confidentiality standards.
Dual ProExC/Ki-67 Staining
SiHa cells were used as positive controls for the dual stain with BD ProExC and Ki-67. Live or SurePath-fixed cells were plated in BD Falcon 96-well poly-L-lysine–coated black/clear imaging plates (BD Labware, Bedford, UK) and used to obtain a functional staining protocol.
Plating and staining protocol for live SiHa cells
Live cervical carcinoma cells were seeded in the imaging plate with a density of 30 000 cells per well and cultured overnight. The immunofluorescent protocol was optimized and carried out as follows. Paraformaldehyde (4%, 10 min) was used for fixation and ice-cold methanol (−20 °C, 5 min) for permeabilization. A specific antibody binding was blocked (blocking buffer: 10% goat serum in phosphate-buffered saline [PBS], 30 min; Jackson ImmunoResearch, West Grove, PA). The cells were incubated with BD ProExC antibody cocktail (undiluted; BD Diagnostics) for 1 h. A goat anti-mouse secondary antibody (1/1000 dilution in blocking buffer; Invitrogen, Paisley, UK) labeled with an Alexa Fluor 546 fluorochrome was added for 30 min. To prevent aspecific binding of the mouse anti–Ki-67 antibody to the goat anti-mouse secondary antibody, mouse serum was used (blocking buffer: 10% mouse serum in PBS, 30 min). The cells were incubated with a primary Alexa Fluor 647–labeled Ki-67 antibody (1/10 dilution in blocking buffer) (BD Biosciences, San Diego, CA) for 1 h. Nuclear counterstaining was performed with DAPI (5 µg/mL in PBS, 15 min). All steps were performed at room temperature (RT), and Tris-buffered saline with Tween-20 (TBST) was used as washing buffer. Negative staining controls were implemented by the substitution of the primary antibodies with PBS.
Plating and staining protocol for SurePath-fixed cells
To maximize the attachment of the SurePath-fixed cells to the imaging plate, several parameters had to be optimized: the harvesting of the cervical carcinoma cells (cell trypsinization vs scraping), the seeding procedure (seeding cells in SurePath vs seeding cells in PBS: 10-min centrifugation at 3000 g and resuspension of the cell pellet in PBS), the cell sedimentation (range sedimentation time 10 min to 2 h), the fixation step (no fixation step vs 10 min ethanol fixation), and the drying step (no drying vs drying at RT or 37 °C, range drying time 30 min to 2 h).
The protocol that was used for the live cells did not enable staining of the SurePath-fixed cells. Therefore, additional steps were optimized to develop a functional staining procedure: rehydration after drying (no rehydration vs rehydration with PBS), an antigen retrieval step (antigen retrieval buffer: SureDetect Slide Preparation buffer [BD Diagnostics] vs Target Retrieval Solution, Citrate pH 6 [Dako, Glostrup, Denmark]; heating step: warm water bath vs heating block [range, 95 °C to 99 °C, range heating time 15 to 40 min]), and a permeabilization step (no permeabilization vs permeabilization with methanol or Triton X-100).
The final protocol for the SurePath-fixed cells was carried out as follows. SurePath preservative fluid containing scraped cervical carcinoma cells was added to the imaging plate with a density of 30 000 cells per well. After 20 min of sedimentation, the fixative was removed and the cells were dried for 1 h at RT. A volume of 300 µL SureDetect Slide Preparation buffer (1×) was added to the wells for overnight incubation at RT to perform antigen retrieval. The plate was placed on a 99 °C heating block for 40 min. After a 20-min cool-down, the SurePath-fixed cells were subjected to the staining protocol as previously described for the live SiHa cells.
High-Content Image Acquisition and Analysis
The BD Pathway 435 system and Attovision software (BD Biosciences, San Diego, CA) were used for high-content imaging. An Olympus (Tokyo, Japan) 20× 0.75 NA objective was used in combination with filters for Alexa Fluor 546 (excitation/emission maxima: 556/573 nm, exposure time 0.2 s, gain 0), Alexa Fluor 647 (excitation/emission maxima: 650/668 nm, exposure time 0.5 s, gain 25), and DAPI (excitation/emission maxima: 358/461 nm, exposure time 0.01 s, gain 0) filters. The laser autofocus was applied, and 16 image fields (4 × 4 montage, field of view: 0.00528 cm2) were acquired per well. DAPI fluorescence was used to define the nuclear area, and subobject measurements determined the number of Alexa Fluor 546– and Alexa Fluor 647–positive objects within the DAPI-defined boundaries. A three-step polygon segmentation algorithm was set to perform measurements in the nuclear area. Each step determined the dye-specific segmentation settings (DAPI image preprocessing: open 5 × 5, sharpen hat, RB 50 × 50, shading, threshold: automatic level 1+0%, split: watershed with erosion factor 6, scrap: 80–1000, dilation width: 1; Alexa Fluor 546 image preprocessing: open 5 × 5, RB 50 × 50, shading, threshold: 1092–4095 gray values, split: watershed with erosion factor 7, scrap: 80–1000, dilation width: 1; Alexa Fluor 647 image preprocessing: close 5 × 5, RB 50 × 50, shading, threshold: 290–4095 gray values, split: watershed with erosion factor 0, scrap: 20–1000, dilation width: 3). A sufficiently long segmentation time-out was defined to ensure that every well was completely segmented.
Data analysis was performed using the Image Data Explorer software (BD Biosciences).
High-content imaging of spiked cell samples
To assess the analytical sensitivity of high-content imaging for the detection of dysplastic cells, a spiking experiment was performed using SiHa cells and cells from LBC samples with normal cytology. The keratinocytes have been spiked with 0%, 0.2%, 1%, 10%, 50%, and 100% SiHa cells, which express the molecular markers. Each spiked sample was plated in 6-fold and subjected to dual ProExC/Ki-67 staining and high-content imaging.
To assess the correlation between the percentages of spiked cells and the percentages of Ki-67– or ProExC-immunopositive objects, a Pearson’s correlation was performed using PASW Statistics 18.0 (SPSS, Inc, an IBM Company, Chicago, IL).
High-content analysis of a patient HSIL LBC sample
To assess the feasibility of high-content imaging for the detection of dysplastic cells in clinical samples, five replicates of an HSIL LBC sample were plated and subjected to dual ProExC/Ki-67 staining and high-content imaging.
Results and Discussion
Dual Staining of SiHa Cervical Carcinoma Cells
SiHa cells were subjected to dual ProExC/Ki-67 staining. Using a standard protocol, the live (paraformaldehyde-fixed) cells showed a bright and specific fluorescent signal for both nuclear markers ( Fig. 1A ), whereas the SurePath-fixed cells were immunonegative. Also, the SurePath-fixed cells attached poorly to the imaging plate. The protocol was modified, and several steps appeared to be crucial to obtain good plate attachment and enhance antigenicity. The cells should be harvested by scraping and dried after sedimentation. An antigen retrieval step (i.e., heating of the cells in antigen retrieval buffer) brought about the ProExC/Ki-67 immunofluorescence. The cell culture passage number, the confluency of the culture when harvested, and the fixation time affected the immunogenicity of the cells. The importance of the latter factor was emphasized by an experiment that compared the immunofluorescence of live cells, cells fixed for 2 days, and cells fixed for 17 days ( Fig. 1 ). The live and recently fixed cells showed bright fluorescence, whereas prolonged fixation in SurePath significantly reduced immunoreactivity.
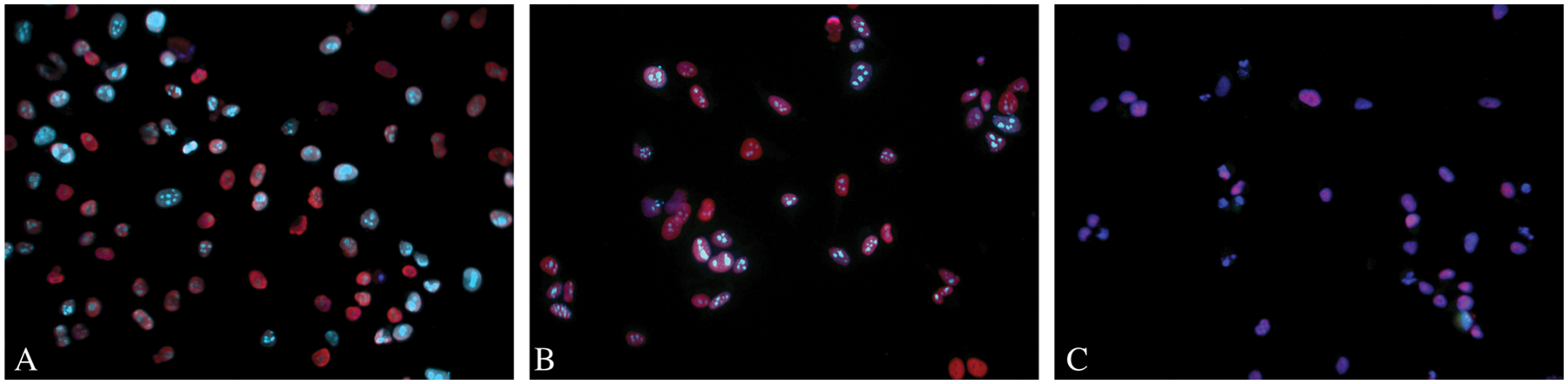
Effect of prolonged fixation on the immunogenicity of the SiHa cells. Pictures from left to right: (
High-Content Imaging of Spiked Cell Samples
Patient keratinocytes with normal cytology were spiked with 0%, 0.2%, 1%, 10%, 50%, and 100% SiHa. Each spiked sample was plated in 6-fold and subjected to dual ProExC/Ki-67 staining and high-content imaging for subobject analysis. The number of ProExC- and Ki-67–positive objects within the DAPI-defined boundaries was determined. Figure 2 shows the results of the ProExC and Ki-67 subobject measurements in the nucleus as the percentage of immunopositive cells over the spiking range. The subobject count correlated significantly with the spiking percentage (Pearson’s correlation coefficient r for ProExC = 0.980 with p < 0.001 and r for Ki-67 = 0.978 with p < 0.001). For example, the spiked samples containing 1%, 10%, 50%, or 100% SiHa cells contained 0.65% ± 0.55%, 7.28% ± 3.95%, 29.26% ± 10.77%, and 80.94% ± 5.97% dual immunopositive cells, respectively. The sample with 100% normal keratinocytes (0% SiHa cells) showed 0.02% ± 0.05% dual-stained ProExC and Ki-67 regions of interest (ROIs).
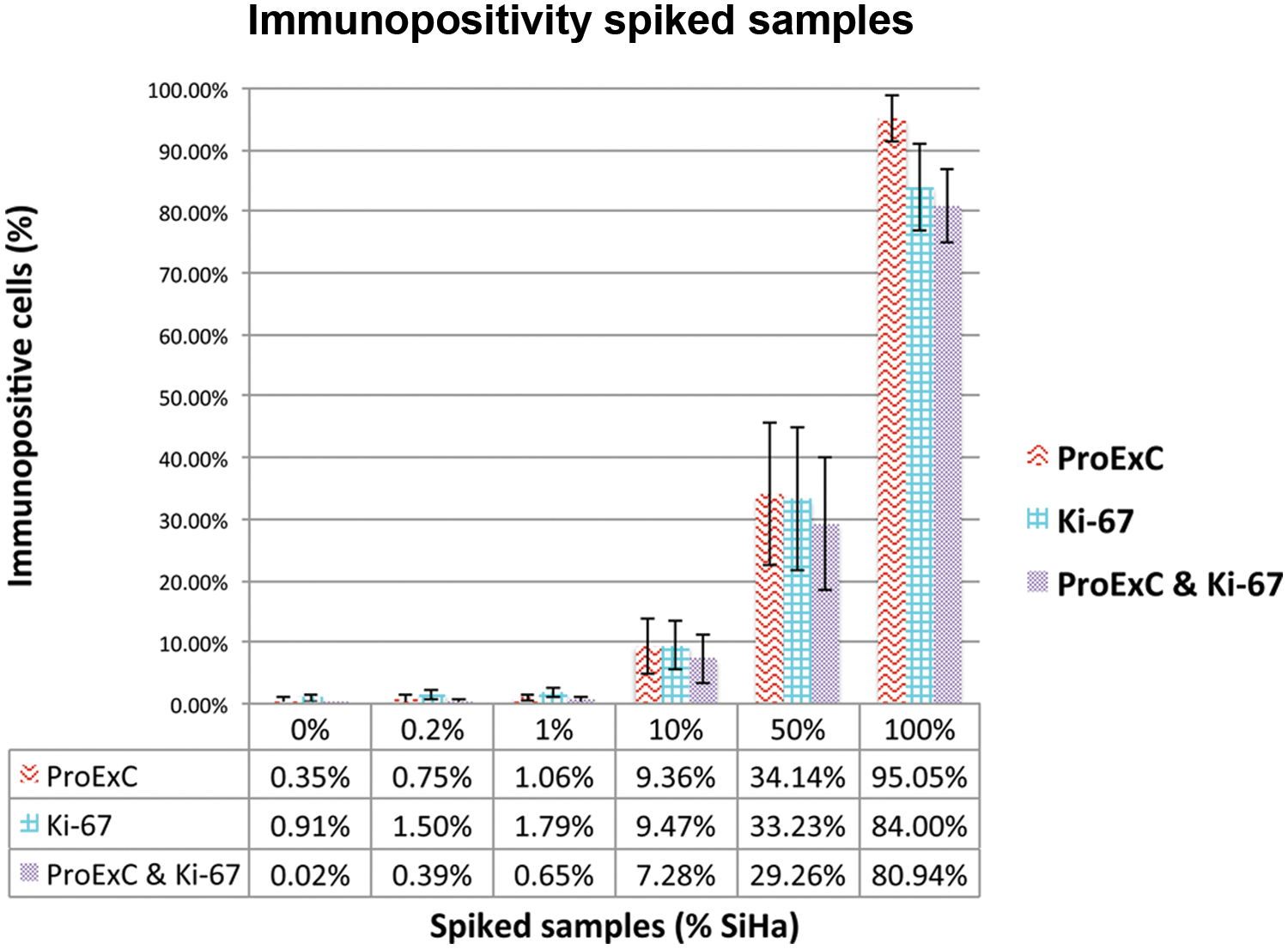
Mean percentages and standard deviation of ProExC-, Ki-67–immunopositive cells, and double-immunoreactive cells for each of the spiked samples. The subobject count correlates with the percentage of spiked SiHa cells.
High-Content Analysis of a Patient HSIL LBC Sample
An HSIL LBC sample was plated in 5-fold and subjected to dual ProExC/Ki-67 staining and high-content imaging for subobject analysis. The patient cells attached excellently to the imaging plate but were often present as clusters. Many showed bright ProExC and Ki-67 immunofluorescence ( Fig. 3 ). The number of ProExC- and Ki-67–positive objects within the DAPI-defined boundaries was determined. Figure 4 shows the results of the ProExC and Ki-67 subobject measurements in the nucleus as the percentage of immunopositive cells in each replicate of the HSIL LBC sample. The coefficient variation percentage (CV%) for the ProExC immunopositivity was 17.57%, whereas it was only 5.96% for Ki-67 and 13.52% for the dual stain. Overall, the HSIL LBC sample contained 17.15% ± 3.01% monostained ProExC, 21.07% ± 1.26% monostained Ki-67, and 6.70% ± 0.91% double-stained cells.
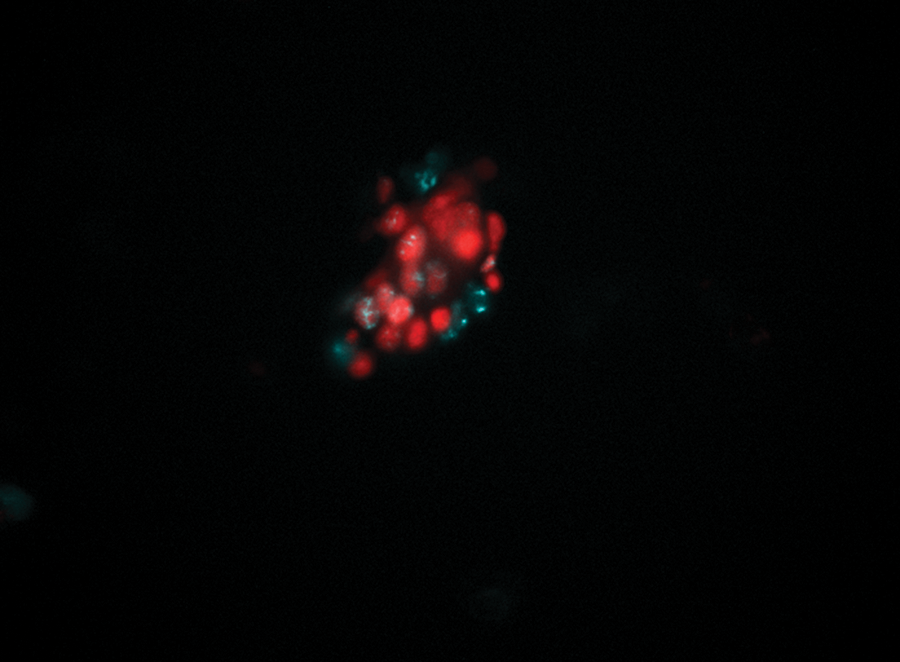
Dual ProExC/Ki-67 staining of high-grade squamous intraepithelial lesion (HSIL) liquid-based cytology (LBC) sample cells. Dysplastic cell clusters showed ProExC and Ki-67 immunofluorescence. ProExC: red; Ki-67: cyan.
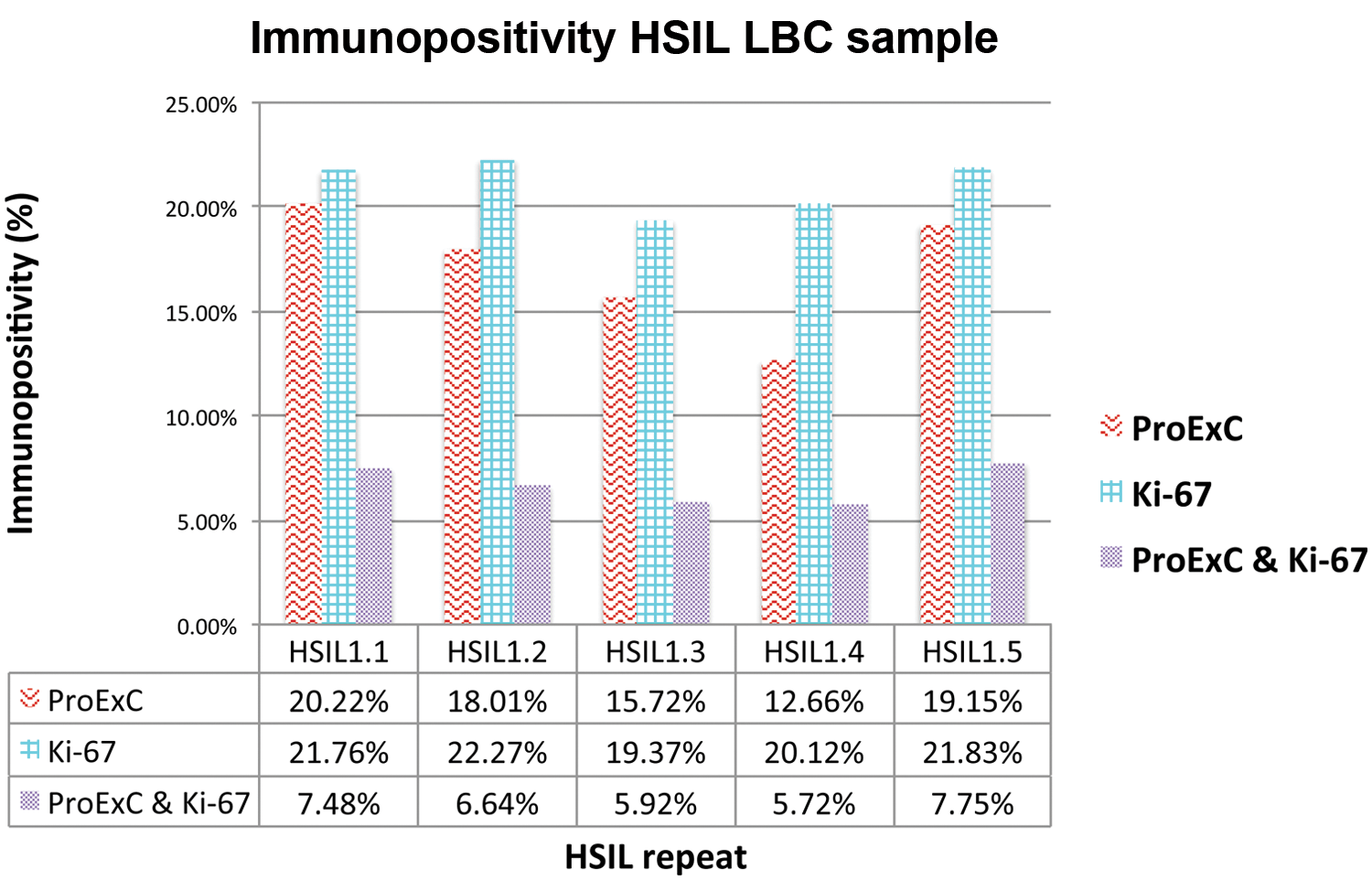
Percentages of ProExC- and Ki-67–immunopositive cells and double-immunoreactive cells for each replicate of the high-grade squamous intraepithelial lesion (HSIL) liquid-based cytology (LBC) sample.
Cervical cancer screening is evolving from cytology-based programs to HPV-based prevention. On one hand, the highly sensitive HPV tests have excellent negative predictive values and support the extension of the screening interval for HPV-negative women.11,12 On the other hand, due to its low PPV, standalone HPV testing could result in the unnecessary follow-up or overtreatment of transient cervical abnormalities. Especially in the vaccination era, in which the PPV of the existing screening tests will be further reduced, 13 the detection of additional molecular markers seems essential to guide the clinical management of HPV-positive women. Biomarkers for transforming HPV infections, cell cycle deregulation, dysplasia, and/or cancer could be combined to identify clinically significant lesions.
The concept of the combined detection of multiple protein markers has already been commercialized in the field of cervical screening in the form of the CINtec Plus Kit (Roche mtm laboratories AG, Heidelberg, Germany). This assay combines the qualitative immunocytochemical detection of Ki-67 and p16, a validated marker for the HPV E7-induced cellular transformation. 14 The CINtec Plus dual stain is based on an enzyme-linked antibody chromogen method in which the brown cellular staining indicates p16 overexpression, whereas the red nuclear staining indicates Ki-67 expression. Petry et al. 15 reported a sensitivity of 91.9% and a specificity of 82.1% for the identification of cervical intraepithelial neoplasia (CIN) 2+ in women with cytology-negative and HPV-positive results, which suggests the clinical potential of multimarker detection strategies. However, the clinical applicability may be hampered by the well-known shortcomings of immunocytochemistry, such as interobserver variation and the absence of systems for automated detection and analysis. The evaluation of the double-stained cells is time-consuming and technically challenging. Nondysplastic endocervical and metaplastic cells tend to be positive for Ki-67 or p16, which contributes substantially to the complexity of the interpretation. 16 Moreover, immunocytochemistry allows the simultaneous detection of only two markers. Especially for routine high-throughput purposes, assays that allow observer-independent automated detection of biomarkers could present a valuable alternative for the subjective interpretation of the results. Innovative technological platforms that allow the combined detection of more than two fluorescently labeled targets might support the development of effective biomarker-based cancer diagnostics or assist the screening for candidate markers.
This proof-of-concept study aims to evaluate a high- content imaging platform for the simultaneous detection of multiple biomarkers for cervical disease. The technology combines fluorescence microscopy in a multiwell format with automated image acquisition and quantitative analysis. Multiple HPV and cellular targets could be simultaneously imaged to assess numerous viral and cellular features, including cell morphology (e.g., cell size, shape, clustering), in a single assay. The increased scale and speed of image acquisition and analysis required to perform high-content imaging demand a sophisticated instrumentation platform, which is provided by the BD Pathway high- content cell analyzer.
In this study, antibodies against Ki-67, TOP2A, and MCM2 proteins (BD ProExC) were used for high-content cell analysis. These biomarkers indicate aberrant S-phase induction and have been validated for cervical cancer screening purposes. 17 TOP2A is an enzyme that unknots and decatenates DNA in preparation for DNA replication, transcription, chromosome segregation, and cell cycle progression, whereas MCM2 is a member of the DNA licensing factor family. 18 The ProExC test is based on antibodies directed against TOP2A and MCM2 and has proven to correlate excellently with HSIL diagnosis and HPV infection in cytology samples.19–21 In cervical smears with atypical squamous cells of undetermined significance (ASCUS) cytology, ProExC proved to be a more sensitive and specific biomarker for CIN2+ than HR-HPV status. 22 Ki-67 is a nuclear marker of active cell proliferation, which is highly associated with the presence of high-grade lesions.20,23
As the study aimed to evaluate a high-content imaging platform for cervical screening purposes, cell line–based LBC preparations and a clinical LBC sample were used.
The cells had been fixed in SurePath preservative fluid for about 2 days, before they were subjected to Ki-67 and ProExC immunofluorescence staining. SurePath and many other fixatives contain formaldehyde, which induces reversible protein cross-linking, leading to the masking of antigen epitopes and the loss of immunoreactivity. An antigen retrieval step was crucial to obtain immunopositivity, and the protocol was modified accordingly. The influence of the fixation time on the immunoreactivity was demonstrated by a comparison of the staining intensity between fresh cells and cells fixed for 2 or 17 days ( Fig. 1 ). The fresh cells and the cells fixed for 2 days showed intense Ki-67 and ProExC staining, whereas the cells fixed for 17 days remained immunonegative. This problem could be mediated in the clinical setting through resuspension of the cells in an alcohol-based medium after arrival in the lab. The clinical samples used in this study were resuspended in 24% ethanol after preprocessing to preserve the immunoreactivity.
This study shows that high-content imaging allows the simultaneous detection of Ki-67 and ProExC immunofluorescence in LBC specimens. The automated detection of these markers can be achieved using the segmentation and subobject measurement tool of the Attovision software. Figure 2 shows that the subobject count of Ki-67 and ProExC in the nucleus significantly correlated with the percentage of spiked SiHa cells. For example, the spiked samples containing 1%, 10%, 50%, or 100% SiHa cells contained 0.65% ± 0.55%, 7.28% ± 3.95%, 29.26% ± 10.77%, and 80.94% ± 5.97% dual immunopositive cells, respectively. This correlation implies that the system is able to quantitatively detect the fraction of spiked SiHa cells, even in the presence of an overload of normal keratinocytes. The proportion of dysplastic cells in HSIL and biopsy-proven squamous cell carcinoma LBC samples ranges from 0.9% to 55.9%. 24 Our results indicate that this range of dysplastic cells can be detected by high-content cell analysis. The sample with 100% normal keratinocytes (0% SiHa cells) showed 0.02% ± 0.05% dual-stained ProExC and Ki-67 ROIs. The detected fluorescence could be derived from autofluorescent cell debris, the presence of endocervical cells,16,22 or insufficient background subtraction.
To assess the potential of high-content imaging to detect dysplastic cells in clinical samples, five replicates of an HSIL LBC sample were subjected to dual ProExC/Ki-67 staining and high-content imaging. The HSIL LBC sample contained a mean percentage of 17.15% ± 3.01% ProExC-, 21.07% ± 1.26% Ki-67–, and 6.70% ± 0.91% double-immunopositive cells. The high CV% of the ProExC staining (17.57%) might be due to the occurrence of cell clusters. The segmentation process identifies, separates, and defines objects of interest and involves a scrapping step to remove fluorescent artifacts or cell debris from subsequent analyses. Clustered cells cannot always be separated as individual ROIs. When the whole cell cluster is identified as a single object that exceeds the defined maximum scrap size, it will also be excluded from the analyses. The number of cell clusters and the way in which they are scrapped can differ between the replicates, which could explain the observed CV% for ProExC.
In contrast, the CV% for Ki-67 was not that high (5.96%). This may be related to the differences between the Ki-67 and ProExC staining and the applied laser-based autofocus settings. Ki-67 shows a punctuated pattern in most phases of the cell cycle and was stained with a directly labeled monoclonal antibody, resulting in a highly focal fluorescent signal. The ProExC antibody cocktail was visualized using a fluorescently labeled secondary antibody and intensely stained the entire nucleus. Consequently, the optimal focal plain slightly differed for ProExC and Ki-67. The laser autofocus offset (i.e., the difference between the z-position found by laser autofocus and the z-position of the image in focus) was based on the Ki-67 immunofluorescence to accurately image the Ki-67 punctuated pattern. As a result, Ki-67 segmentation might have been performed more precisely, leading to the lower CV%, compared with ProExC.
The staining percentages, as shown in Figure 4 , indicate that not all ProExC-immunopositive cells express Ki-67 and vice versa. Different expression patterns of ProExC and Ki-67 in cervical biopsies have already been described. 25 A small subset of cases with discordant ProExC/Ki-67 immunostaining raised the possibility that ProExC might be better than Ki-67 as an indicator of CIN persistence/progression. However, large prospective studies of cases with discordant ProExC/Ki-67 staining are needed.
Overall, the HSIL patient sample contained 6.70% double-stained ROIs, whereas the patient sample with normal cytology showed dual fluorescence in 0.02% of the ROIs. This means that high-content imaging can discriminate normal from dysplastic samples and could play a role in clinical cervical cancer screening. These results should be verified in a larger group of clinical samples, which was beyond the scope of this proof-of-concept study. Moreover, the high-content imaging platform could be used to evaluate or validate new biomarkers or combinations for cervical cancer screening. Biomarker evaluation and validation requires the analysis of a large number of clinical samples. 26 The BD Pathway high-content cell analyzer allows such high-throughput analysis for biomarker research and development in an automated and standardized way. Because of the flexibility of the system toward the detection of fluorescently labeled targets, protein markers could also be combined with nucleic acid–based markers, such as viral DNA or chromosomal abnormalities.
In conclusion, this study proves the concept that high-content imaging automatically detects and analyzes different fluorescently labeled biomarkers for cervical dysplasia.
Footnotes
Acknowledgements
We thank D. De Rijck for his assistance with the figures.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Johannes Bogers is the promoter of this research project for which the University of Antwerp obtains a financial contribution from Becton Dickinson. Frans Nauwelaers and Martin Ryser are employed by Becton Dickinson. All other authors have no conflicts of interest to declare.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by Becton Dickinson. Maria Isabel Micalessi is supported by the Industrial Research Fund of the University of Antwerp and by Euregional PACTII. Gaëlle Boulet and Johannes Bogers are supported by Euregional PACTII, a project funded by the European Union through the Interreg IV program of Grensregio Vlaanderen-Nederland. Isabel Pintelon is supported by the Core Facility for Biomedical Microscopic Imaging of the University of Antwerp. Peter Verstraelen has a PhD fellowship from the Flemish government agency for Innovation by Science and Technology (IWT).
