Abstract
Chronic inflammation is at least partially mediated by the chemokine-mediated attraction and by the adhesion molecule–directed binding of leukocytes to the activated endothelium. Therefore, it is therapeutically important to identify anti-inflammatory compounds able to control the interaction between leukocytes and the endothelial compartments of the micro- and macrocirculation. When testing novel drug candidates, it is, however, of the utmost importance to detect side effects, such as potential cytotoxic and barrier-disruptive activities. Indeed, minor changes in the endothelial monolayer integrity may increase the permeability of small blood vessels and capillaries, which, in extreme cases, can lead to edema development. Here, we describe the development of a high-throughput screening (HTS) platform, based on AlphaLISA technology, able to identify anti-inflammatory nontoxic natural or synthetic compounds capable of reducing tumor necrosis factor (TNF)–induced chemokine (interleukin [IL]–8) and adhesion molecule (ICAM-1) expression in human lung microvascular endothelial cells. Quantification of cell membrane–expressed ICAM-1 and of cell culture supernatant–associated levels of IL-8 was analyzed in HTS. In parallel, we monitored monolayer integrity and endothelial cell viability using the electrical cell substrate impedance sensing method. This platform allowed us to identify natural secondary metabolites from cyanobacteria, capable of reducing ICAM-1 and IL-8 levels in TNF-activated human microvascular endothelial cells in the absence of endothelial monolayer barrier disruption.
Keywords
Introduction
Cardiovascular diseases represent the major cause of morbidity and mortality in industrialized countries. The vascular bed, with endothelial cells as a central component, plays a critical role in various diseases such as arteriosclerosis, sepsis, and thrombosis. The endothelium becomes activated by stimuli from cellular damage or infection, resulting in the subsequent recruitment of leukocytes. The proinflammatory mediators tumor necrosis factor (TNF), interleukin (IL)–1β and lipopolysaccharide are well-studied factors providing inflammatory signals to endothelial cells.1,2 These mediators activate signaling pathways such as NF-κB and p38 MAPK 3 that in turn induce the production of inflammatory mediators, such as chemokines and adhesion molecules. Chemokines induce leukocyte chemotaxis via G-protein–coupled receptors. Among the many chemokine receptor ligands, IL-8 was demonstrated to play a prominent role in attracting neutrophils and monocytes. Therefore, a deregulation in their expression or function can contribute to the initiation and perpetuation of chronic inflammation. 4 Endothelial cells play an early and rate-limiting step in most inflammatory processes. They are activated in response to bacterial products (such as endo- and exotoxins5,6), cytokines, and inflammatory mediators, which leads to leukocyte recruitment from the circulation. 7 Leukocyte adhesion to activated endothelium is a multistep process with sequential and overlapping stages mediating the transendothelial migration from the vascular lumen to the extracellular tissue. 8 This cascade involves leukocyte rolling, followed by firm attachment to endothelial cells, and finally migration through the paracellular space of the endothelial lining. Compounds that can reduce the secretion of chemokines and the expression of adhesion molecules in activated endothelium could thus serve as potential anti-inflammatory drug candidates.
When screening molecular libraries targeting the vascular endothelium for the reduction of the proinflammatory response, one has to ensure that the reduced inflammatory signal is not due to cytotoxicity. Moreover, other undesired side effects, such as the loss of endothelial barrier integrity, should also be discovered early on in the development of therapeutically promising natural compounds. Soluble forms of adhesion molecules have been detected in the circulation under conditions reflecting endothelial damage or dysfunction. It has also been shown that soluble intercellular adhesion molecule 1 (ICAM-1) may serve as a prognostic marker for stage II colorectal cancer patients and as a marker in a number of other pathological conditions.9,10
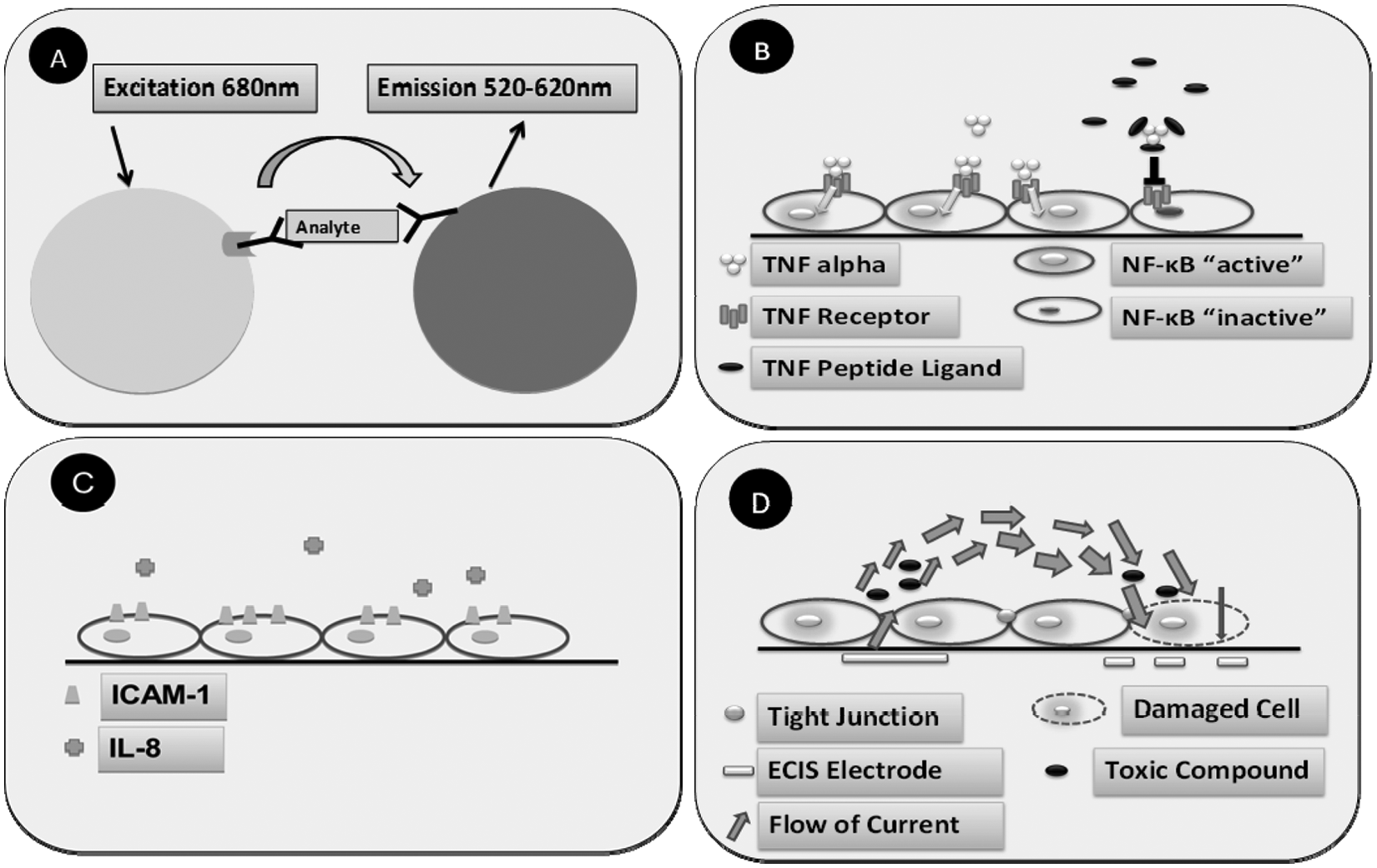
We describe here the development of a high-throughput screening (HTS) platform for the identification of natural and synthetic molecules able to reduce the activation of the endothelium induced by the proinflammatory cytokine TNF. The Amplified Luminescence Proximity Homogenous Assay (AlphaScreen) detects membrane-expressed ICAM-1 as well as soluble IL-8 in cell culture supernatants of TNF-stimulated human lung microvascular endothelial cells (HL-MVEC). We could first demonstrate the specificity and usability of the assay for the identification of molecules capable to reduce ICAM-1 expression as well as IL-8 secretion in unstimulated HL-MVEC. Second, we used electrical cell substrate impedances sensing (ECIS) technology to monitor effects of positively tested secondary metabolites from cyanobacteria on endothelial monolayer integrity and viability in real time. As such, we could separate false-positive but toxic and monolayer-disrupting compounds from truly anti-inflammatory candidates. By using the above described platform, we have identified natural secondary metabolites as potential anti-inflammatory substances ( Fig. 1 ). Because the above described assays were performed in 384-well plates, they have the potential to be adapted for higher throughput and automation.
Materials and Methods
AlphaLISA assay and cell-based AlphaLISA for the quantification of proinflammatory cytokines and adhesion molecules, respectively
For the AlphaLISA quantification of cytokines and adhesion molecules, the following reagents, cells, plastic material, and medium were used: AlphaLISA ICAM-1 Immunoassay Research kit (PerkinElmer, AL282C); AlphaLISA IL-8 Immunoassay Research kit (PerkinElmer, AL224C); BRANDplates, 384-well white (Brand, 781981); HL-MVEC (Provitro, 121 0144); endothelial cell medium (PAA, U15-002); human tumor necrosis factor (NIBSC, 88/786); and SB203580 (Sigma, S8307-1MG).
HL-MVECs were plated at a concentration of 5000 cells/well in a white 384-well plate in 50 µL of endothelial medium (1% penicillin/streptomycin, 0% fetal calf serum [FCS]) and incubated at 37 °C in a CO2 incubator with 5% CO2 for 4 h. Then the cells were stimulated with different TNF concentrations and incubated at 37 °C in a CO2 incubator with 5% CO2 for 18 h. The supernatants were transferred to an empty 384-well plate and stored at −20 °C. For the AlphaLISA ICAM-1 assay, HL-MVECs were grown in white 384-well plates and incubated with a mixture of a biotinylated anti–ICAM-1 antibodies (1 nM final concentration in the well), the AlphaLISA acceptor beads (10 µg/mL final concentration in the well), and the assay buffer. The HL-MVECs were incubated at room temperature for 1 h in the dark. Afterward, the AlphaLISA streptavidin donor bead solution was added (40 µg/mL final concentration in the well), and the plate was incubated again for 30 min at room temperature in the dark. After the incubation, the plate was measured with the SpectraMax Paradigm Modular Multi-Mode Reader (Molecular Devices) and an AlphaScreen detection cartridge (excitation at 680 nm and emission at 570 nm). For the AlphaLISA IL-8 immunoassay, the supernatants were taken and mixed with the biotinylated anti–IL-8 antibody (1 nM final concentration in the well) in combination with the AlphaLISA acceptor beads (10 µg/mL final concentration in the well). The further procedure of the assay was performed in the same way as with the AlphaLISA ICAM-1 assay described above.
Isolation of Bioactive Compounds from Cyanobacteria
Culture conditions
The cyanobacterial strain Nostoc sp. Lukešová 30/93 was isolated from forest soil in Krušné Hory in the Czech Republic. The cyanobacteria were cultivated in 350 mL glass tubes on liquid Allen-Arnon medium11,12 and bubbled with 2% CO2-enriched air at a constant temperature of 28 °C, under low continuous illumination with banks of cool-white fluorescent lights of 50 Wm–2 (Philips, Osram Dulux L, 55W/12-950). After 5 to 7 d of cultivation, the culture was harvested by centrifugation (Hettich 320 centrifuge) at 4500 rpm at 10 min, stored at −70 °C, and lyophilized (Lyovac GT3, Leybold-Hereus). To obtain sufficient amount of biomass, mass cultivation in a 100 L glass panel under the same conditions was performed.
Extraction and purification of the active compounds
The freeze-dried biomass (4 g) was extracted twice with 200 mL of 50% methanol by mortar and pestle for 1 h at room temperature. Subsequently, the extract was centrifuged at 4500 rpm for 15 min (Hettich 320 centrifuge). The supernatant was transferred into a 1000 mL separatory funnel and subjected to liquid-liquid partitioning by adding ethyl acetate and water in the final ratio of 1:1:4 (methanol:ethyl acetate:water). After the 2 phases separated, the lower one was collected and evaporated at 40 °C under vacuum (Büchi Rotavapor R-114); the upper one, containing pigments, was discarded. The dry residue was dissolved in 600 mL of 20% methanol and concentrated on C8 HLB cartridges (Waters Oasis). The bioactive compounds bounded to the stationary phase were eluted by 4 mL of 70% methanol and subjected to the subsequent analysis.
Preparative HPLC
The preparative system consists of 2 dual-piston preparative HPLC pumps Prep 100 (DeltaChrom), column thermostat, and gradient controller LabAlliance (Watrex). The concentrated extract (0.5 mL) was injected into the semipreparative HPLC column (Reprosil 100 C18, 250 × 8 mm, 5 µm, dr. Maisch GmbH) and eluted by methanol/water gradient (50%−80% methanol for 50 min, 80%−100% methanol for 1 min, 100% methanol for 6 min) with a flow rate of 2.6 mL/min. Detection of samples was performed both by UV-Vis detector in 210 nm (Knauer) and mass spectrometer (HP 1100 MSD SL-Ion Trap, Agilent) by splitting of 5 vol% of eluent coming out from the column, which allowed continuous monitoring and precise collection of the eluted fractions. The retention times of nostopeptolide A1 and A3 were 36.8 and 39.1 min, respectively. Subsequently, nostopeptolide A1 was subjected to another purification step. The sample (0.5 mL, 1 mg/mL) was injected into the semipreparative HPLC column (Reprosil 100 Phenyl, 250 × 8 mm, 5 µm; Watrex) and eluted with the flow rate of 2.0 mL/min by acetonitrile/water gradient (40%−45% acetonitrile in 5 min, isocratic elution with 45% acetonitrile for 5 min, 45%−60% acetonitrile for 10 min, 60%−100% acetonitrile for 2 min, and 100% acetonitrile for 5 min.). The detection of eluted compounds was performed as described above. The retention time of nostopeptolide A1 was 11.4 min.
The collected fractions were evaporated under vacuum and used in the analytical LC-MS experiments and in the bioassays.
LC-MS analysis
The purified cyanobacterial metabolites were analyzed using the Agilent 1100 Series connected to mass spectrometer HP 1100 MSD SL-Ion Trap. The samples were analyzed on a reverse-phase column (Zorbax XDB-C8, 4.6 × 150 mm, 5 µm) at 30 °C and eluted with the flow rate of 0.6 mL/min by methanol/water gradient (30%−100% methanol for 30 min, 100% methanol for 5 min), modified by 0.1% formic acid to improve ionization. The ion trap mass spectrometer and PDA detector were used to monitor the eluted compounds. The operating parameters of the mass spectrometer were as follows: the spray needle voltage was set at 4.5 kV, and the drying temperature was 325 °C. As the nebulizing and drying gas nitrogen was used, a flow rate of 50 mL/min and 10 mL/min was used, respectively. The ESI capillary voltage was 230 V, and helium was used as auxiliary gas (15 mL/min). In the MS/MS experiments, the selected precursors were targeted to 900 m/z, and the collision energy 100% of 5 V was applied. All MS and MS/MS spectra were acquired in the positive and negative ion mode, in a broad mass range of 50 to 2200 m/z and scanning speed of 13 000 m/z per second.
ECIS Method
Materials used were ECIS plates (Applied Biophysics, 96W10E+), fibronectin (Sigma, F2006-1MG), HL-MVEC (Provitro, 121 0144), endothelial cell medium (PAA, U15-002), human TNF (NIBSC, 88/786).
The ECIS technology can be used to analyze the barrier integrity of cell types forming monolayers. ECIS measures the change in impedance of a small electrode to alternating current (AC) flow. The heart of the measurement device is a specialized microplate that has 96 individual wells for cell culturing. Each well of the ECIS plate contains ten 250 µm diameter gold electrodes and a much larger gold counter electrode that connects to the ECIS electronics. The ECIS plate was precoated with fibronectin (10 µg/mL) for 1 h at 37 °C in a CO2 incubator with 5% CO2. Then 60 000 HL-MVECs per well were plated in 200 µL endothelial cell medium (1% penicillin/streptomycin, 10% FCS) and incubated overnight. On the next day, the medium was changed, and 200 µL endothelial medium (1% penicillin/streptomycin and 0% FCS) was added. The ECIS plate was measured with an AC at 4000 Hz. Afterward, the impedance (Ohm) was normalized. After 1 h, the cells were stimulated with human TNF (10 ng/mL) or several test substances (1 µg/mL). Six replicates were performed for each sample.
Results and Discussion
Establishment and Characterization of an AlphaLISA-Based Assay for the Detection of Adhesion Molecules and Chemokines: Responsiveness of Endothelial Cell Lines to TNF
Most cell-based enzyme-linked immunosorbent assays require fixation of cells and several washing steps for removing unbound primary and secondary antibodies. These manipulation steps can potentially lead to high variations in cell numbers in individual wells of the microplate, which can hinder the evaluation of the results. To avoid these problems, we used AlphaLISA as a homogenous detection technology, which allows the analysis of surface-bound proteins as well as of soluble factors secreted in the cell culture medium, by means of sequential addition of the detection reagents. AlphaLISA is a bead-based method ( Fig. 1a ), which relies on the proximity of donor and acceptor beads conjugated with antibodies specific for the analyte of interest (e.g., ICAM-1 or IL-8). Upon an excitation at 680 nm, the photosensitizers inside the donor beads convert ambient oxygen to the singlet oxygen state. Only when the donor and acceptor beads are in close proximity (200 nm) can the single oxygen molecules diffuse to the acceptor bead, which results in a bright emission at 520 to 620 nm. We have used AlphaLISA to test the responsiveness of HL-MVEC to the proinflammatory cytokine TNF by comparing the ICAM-1 or IL-8 levels. At the same time, we determined the optimal concentration of TNF, which could cause a significant upregulation of ICAM-1 and IL-8. Interestingly, a clear upregulation of ICAM-1 and IL-8 was observed already at a low dose of TNF stimulation (100 pg/mL). Although all experiments were done by manual pipetting in 384-well plates, only small standard deviations were observed ( Fig. 2 ). From these data, we conclude that the assay has a good potential to be performed in a fully automated HTS environment.
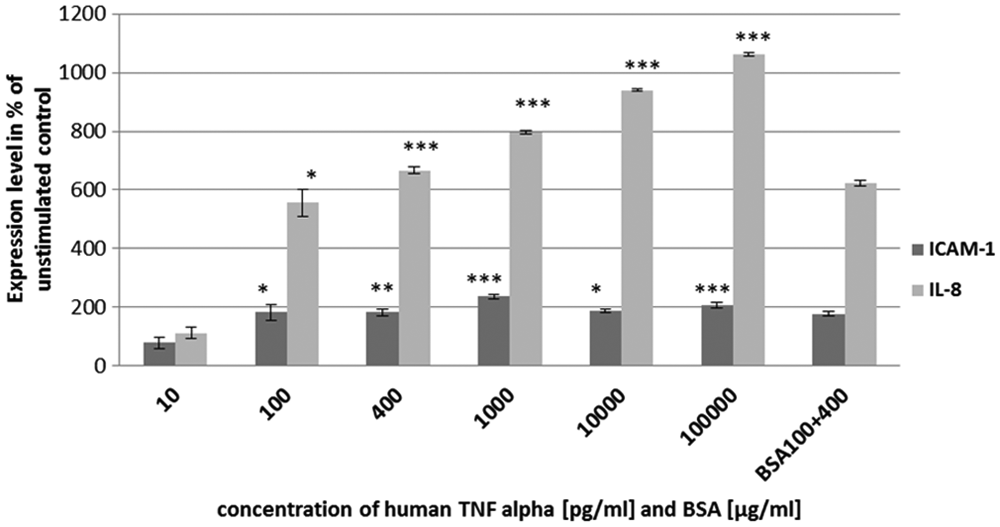
Responsiveness of endothelial cell lines to tumor necrosis factor (TNF). To determine the optimal concentration of TNF for the stimulation of human lung microvascular endothelial cells, they were treated with various concentrations of the cytokine. In addition, a bovine serum albumin (BSA) control was used to exclude unspecific binding of proteins to beads. There are no significant changes of the BSA control compared with TNF 400 pg/mL. *p < 0.05; **p < 0.001; ***p < 0.0001.
Robustness of the AlphaScreen Assay
Upon optimization of the AlphaLISA assay above, we evaluated the reproducibility and robustness of the assay (
Kinetic Measurement of ICAM-1 and IL-8 after hTNF Stimulation
After stimulating the cells with 10 pg/mL, 100 pg/mL, 1000 pg/mL, or 10 000 pg/mL of hTNF, the expression of ICAM-1 and the secretion of IL-8 were measured. The time points for the measurement were 4 h, 12 h, 18 h, and 24 h. As demonstrated in Figure 3 , we could clearly observe an increased production of marker proteins in correlation with time.
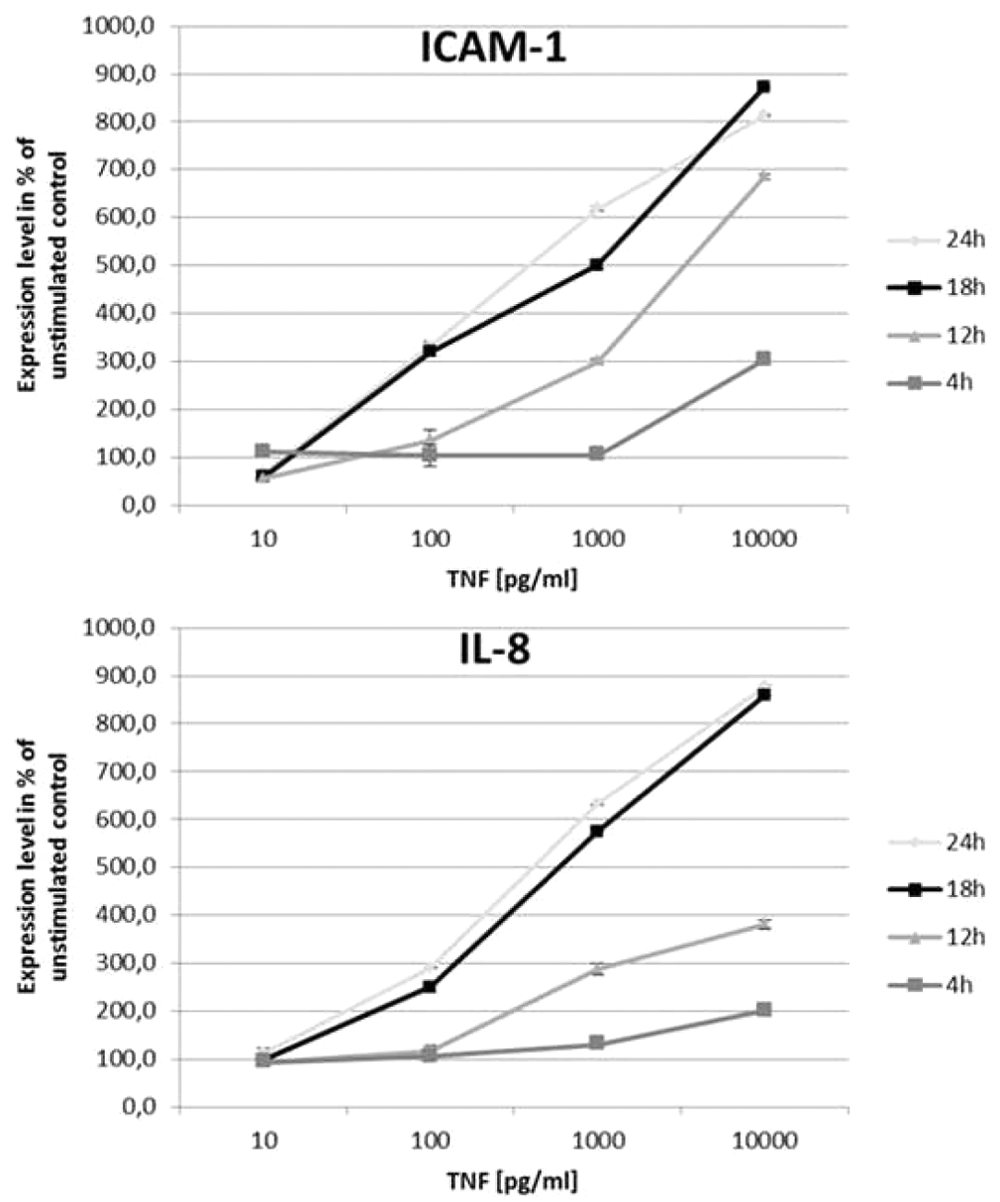
Kinetic measurements of ICAM-1 and IL-8. Human lung microvascular endothelial cells were treated with several concentrations of hTNF alpha and then incubated for 4 h, 12 h, 18 h, and 24 h.
Dose-Response Curve of ICAM-1 and IL-8 Expression in the Presence of the p38 MAPK Inhibitor SB203580
As shown in Figure 4 , we have been using the p38 MAPK inhibitor SB203580 for assay validation and to generate a dose-response curve. SB203580 belongs to the group of pyridinyl imidazole inhibitors and is known to possess anti-inflammatory effects. 13 The TNF concentration used in these experiments was 100 pg/mL, which enabled us to generate a dose-response curve for IL-8 and ICAM-1 ( Fig. 4 ). For ICAM-1, an IC50 value of 82 µM SB203580 could be calculated.
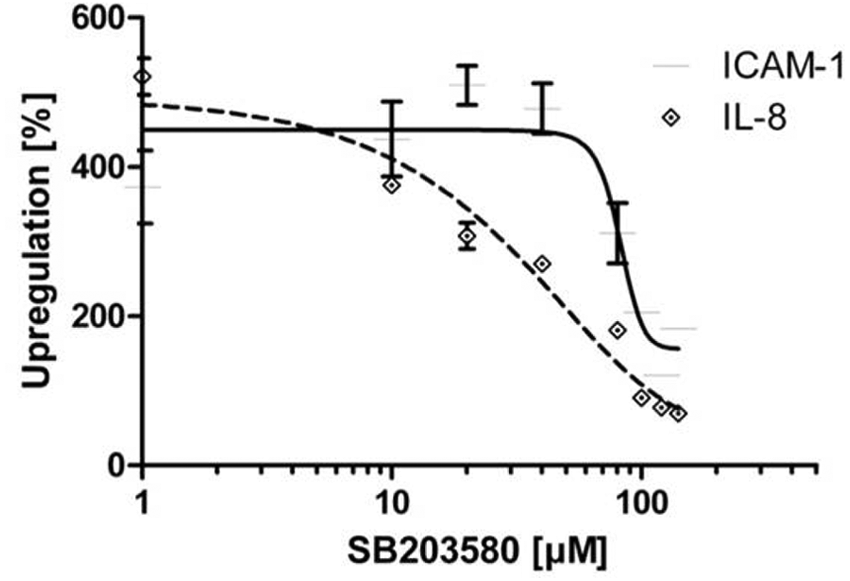
AlphaLISA detected inhibition of TNF alpha stimulation of endothelial cells with SB203580. Human lung microvascular endothelial cells were treated with several concentrations of SB203580 for 30 min, and the cells were then stimulated with 100 pg/mL of TNF alpha.
Identification of Cyanobacterial Metabolites
The nostopeptolides as a group of natural toxins characterized by a cyclic peptide core structure in the mobile phase consisting of methanol water provides protonated molecule [M+H]+ at m/z 1081 as the relatively intensive ion in positive mode electrospray mass spectrometry (ESI-MS). The most abundant ion corresponds to the pseudomolecular sodiated adduct [M+Na]+ at m/z 1103 and additional low intensive ion is potassium adduct [M+K]+ at m/z 1119. The molecular mass of the nostopeptolide was confirmed by negative ion mode shown as a deprotonated [M-H]– ion peak at m/z 1079 as the main ion in the full scan spectrum mode.
The collision-induced dissociation of the molecular ion [M+H]+ in positive mode ESI leads to the structurally significant diagnostic ion peak at m/z 898 as a dominant fragment that corresponds to the loss of the Ileu/Val side chain (–183 Da, C10H17NO2) of the nostopeptolide core structure (
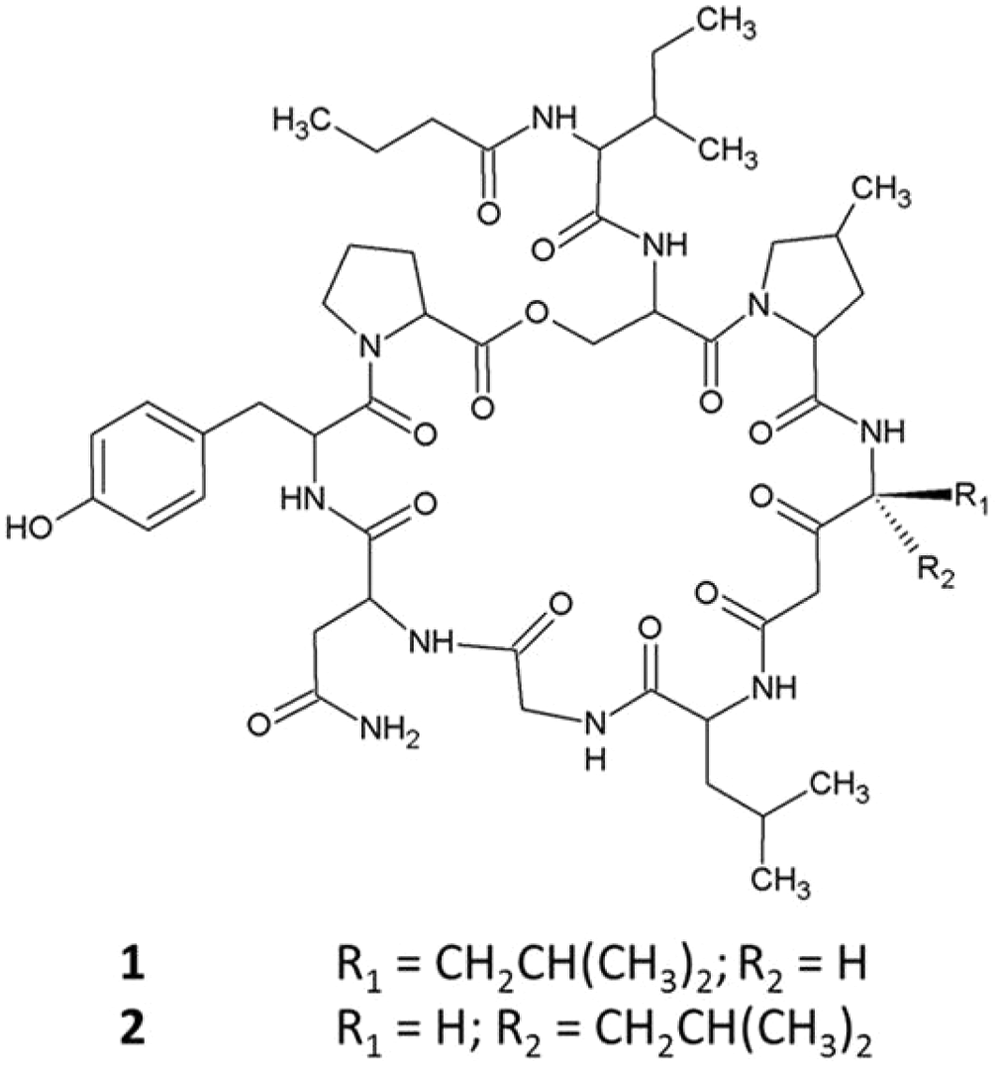
Structure of nostopeptolide A1 (
Testing of Bioactive Compounds Isolated from Cyanobacteria
Considering the reproducibility and robustness of the AlphaLISA reported above, we can conclude that the assay is suitable for HTS. Therefore, this platform was used to determine the effect of bioactive compounds isolated from cyanobacteria. The compounds were isolated at the Institute of Microbiology, Academy of Science, in the Czech Republic and processed as described earlier. We have tested 78 different cyanobacterial extracts and purified metabolites in various concentrations. Initially, 7 samples were found to be able to reduce ICAM-1 and IL-8 levels in 100 pg/mL TNF-treated HL-MVECs. However as described below, 5 of them showed cytotoxicity and barrier disruptive activity when applied to ECIS. Therefore, we ended up with 2 compounds, which significantly blocked the upregulation of ICAM-1 and IL-8 after stimulation of HL-MVECs with 100 pg/mL hTNF ( Fig. 6 ). This activity indicates the anti-inflammatory potential of the novel cyanobacterial substances. Two nostopeptolides, A1 and A3, isolated from the Nostoc sp. strain, were able to reduce IL-8 levels from 600% to 300% and ICAM-1 levels in supernatants from 300% to 100% ( Fig. 6 ). However, the activity has not been affected by a difference in stereochemistry of the samples. Cells were stimulated with a relatively low concentration of TNF (100 pg/mL TNF), in view of the rather modest inhibitory activity of the compounds. Using various approaches, the possibility of finding weak and strong inhibitors is given, which can then be further optimized using, for instance, lead optimization, which is performed in drug discovery. Nevertheless, screening conditions can be set to a higher stringency, using, for example, higher concentrations of TNF, allowing for the detection of more potent inflammation inhibitors.
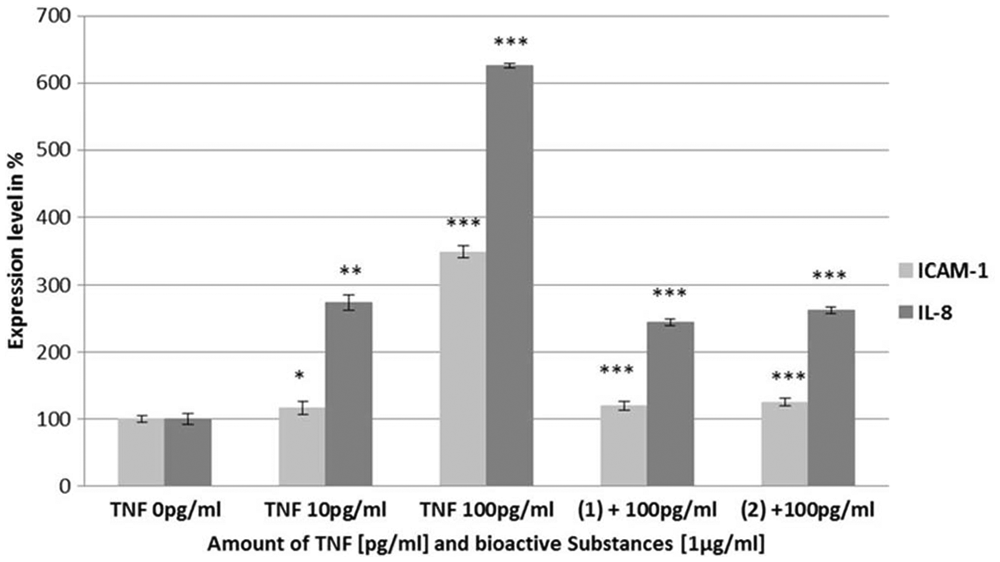
Screening of bioactive compounds. Human lung microvascular endothelial cells were pretreated with nostopeptolide A1 (1) and A3 (2) for 30 min and then stimulated with 100 pg/mL of TNF. Both isomers were able to reduce TNF alpha–induced expression levels of ICAM-1 and IL-8 on endothelial cells significantly, compared with TNF 100 pg/mL. *p < 0.05; **p < 0.001; p < 0.0001.
Assessment of the Barrier-Disruptive Activities of the Bioactive Cyanobacterial Compounds in Endothelial Monolayers by ECIS
To determine whether the effects observed in the AlphaLISA tests are due to a possible cytotoxic effect of the compound, substances were subjected to ECIS analysis of HL-MVEC monolayer integrity. In a first approach, we have established a dose-response curve by using different concentrations of Triton X-100 ( Fig. 7A ), which is toxic to HL-MVECs. When adding decreasing concentrations of Triton X-100 to the cells, we found that the change in impedance could clearly monitor the level of toxicity in real time. In a second approach, we applied the positively tested cyanobacterial compounds to HL-MVEC monolayers to assess their cytotoxic potential ( Fig. 7B ). Interestingly, we found a barrier integrity–supporting effect, when applying the mixture of testing compounds to the monolayers of lung endothelial cells, as compared with the control.
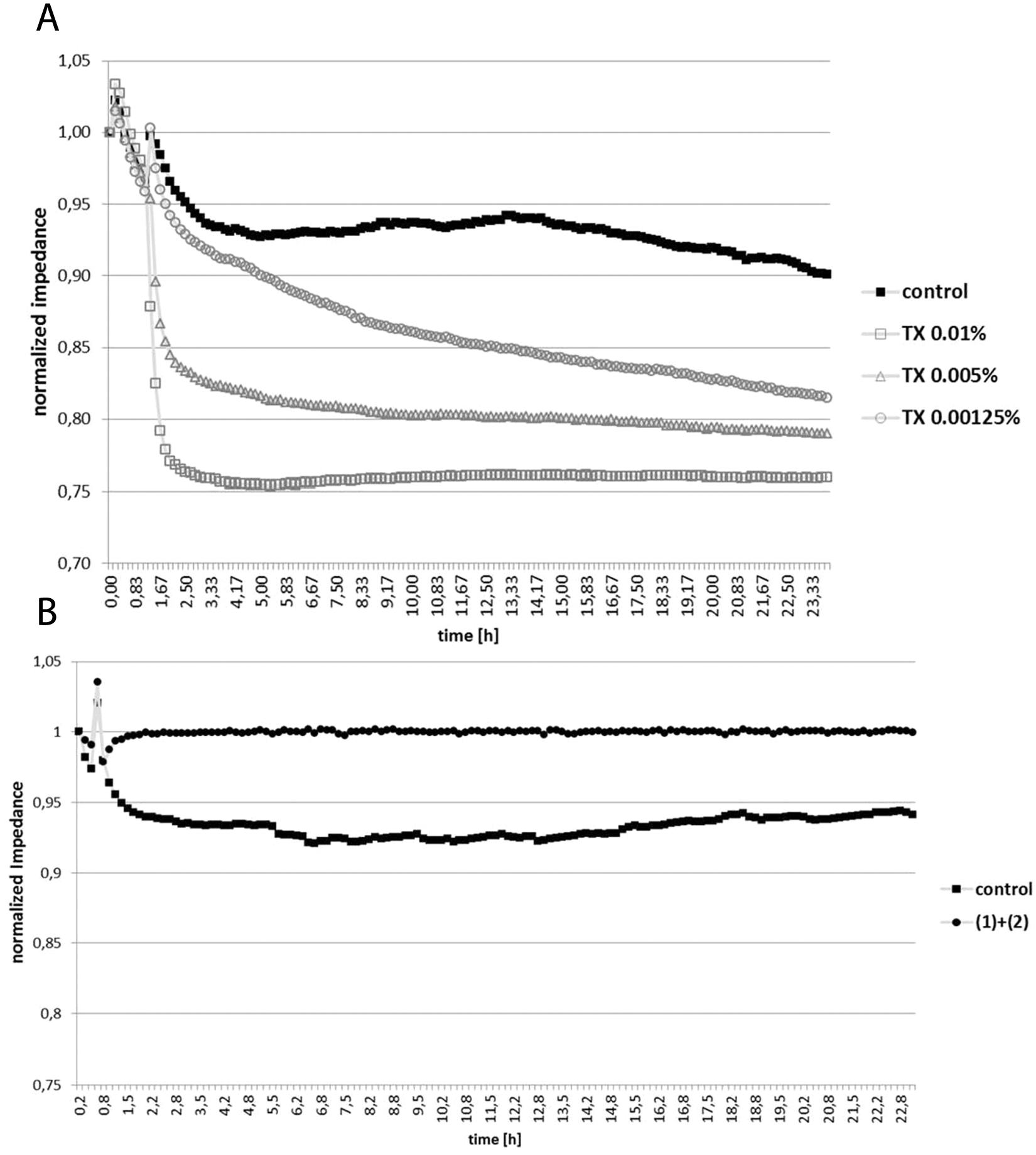
(
The destruction of the capillary endothelium is a hallmark of many inflammatory diseases, and a combination of anti-inflammatory and barrier integrity–supporting properties would be an ideal therapeutic intervention. We could moreover show that previously identified cytotoxic cyanobacterial compounds induced cytotoxicity and barrier integrity disruption (data not shown).
Discussion
We have established a novel combination of assays for the identification and toxicity profiling of potentially anti-inflammatory compounds using AlphaLISA and ECIS. The assay can distinguish true inhibitors of inflammation from false-positive cytotoxic inhibitors. The average Z-factor value of the assay was greater than 0.7 (for AlphaLISA), indicating it could be further adapted from the 384-well to a 1536-well format. Furthermore, the assay is capable of screening for both weak and strong inhibitors by adapting the stimulation dose of the proinflammatory cytokine TNF. Using the platform described, we have screened more than 78 samples of cyanobacterial metabolites and identified 2 weak inhibitors for the expression and production of proinflammatory mediators in response to TNF treatment in human lung microvascular cells. Taken together, the combination of the 2 methods presented in this article allows for the identification of promising anti-inflammatory substances, which could further be tested in in vivo models. Because compounds are tested early and in real time in pathologically relevant primary human cells, cytotoxic and barrier-disintegrating side effects can be detected at an early phase of preclinical research.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the European Territorial Co-operation grant (M00140) and the Österreichische Forschungsförderungsgesellschaft (FFG, 821021, 822710).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
