Abstract
Bone marrow-derived mesenchymal stem cells (BMSCs) are a good cell source for regeneration of cartilage as they can migrate directly to the site of cartilage injury and differentiate into articular chondrocytes. Articular cartilage defects do not heal completely due to the lack of chondrocytes or BMSCs at the site of injury. In this study, the chemotaxis of BMSCs toward chemokines, which may give rise to a complete regeneration of the articular cartilage, was investigated. CCR2, CCR4, CCR6, CXCR1, and CXCR2 were expressed in normal BMSCs and were increased significantly upon treatment with proinflammatory cytokines. BMSC migration was increased by MIP-3α and IL-8 more than by MCP-1 or SDF-1α. IL-8 and MIP-3α significantly enhanced the chemotaxis of BMSCs compared with MCP-1, SDF-1α, or PBS. Human BMSC recruitment to transplanted scaffolds containing either IL-8 or MIP-3α significantly increased in vivo compared to scaffolds containing PBS. Furthermore, IL-8- and MIP-3α-containing scaffolds enhanced tissue regeneration of an osteochondral defect site in beagle knee articular cartilage. Therefore, this study suggests that IL-8 and MIP-3α are the candidates that induce the regeneration of damaged articular cartilage.
Introduction
An articular cartilage injury causes severe chronic pain and decreased mobility. Several surgical techniques have been widely used to repair injured articular cartilage. Subchondral drilling is a simple method, but the articular cartilage defects are filled with fibrocartilage. Osteochondral autograft transplantation is a promising method to treat articular cartilage injuries, but donors are limited. Although autologous chondrocyte transplantation is another good treatment modality, ex vivo cell expansion is required, and the treatment can be both expensive and difficult (6,7,39). To overcome these problems, tissue engineering using biodegradable materials has been attempted to successfully repair damaged articular cartilage (41). Autologous chondrocyte or mesenchymal stem cell (MSC) seeding on implanted scaffolds is necessary for the successful regeneration of damaged articular cartilage because tissue engineering using scaffolds alone is insufficient to avoid the current problems (1).
Cartilage regeneration using autologous cells can be accomplished by the natural progression of cartilage development (17). Autologous MSCs have been highly regarded as an alternative cell source for the repair of articular cartilage defects as autologous MSCs can differentiate into chondrocytes and osteocytes under tissue-specific culture conditions (42). The ex vivo expansion of MSCs is required for cartilage regeneration. However, the loss of osteogenic or chondrogenic potential of primary MSCs and the secondary surgery for cultured cell implantation remain as problems in this therapeutic strategy (34). Therefore, a new strategy should be developed to overcome the current problems and successfully repair articular cartilage using autologous MSCs.
A chemokine is a chemotactic cytokine that plays a crucial role in cell recruitment. Recent studies have demonstrated that chemotaxis of bone marrow-derived MSCs (BMSCs) is induced by chemokines and repairs the wounded tissue (29,37). If this concept is applied to cartilage repair, the in situ recruitment of BMSCs to the site of defect using chemokines may be an effective method for inducing cartilage tissue regeneration. Unfortunately, few reports of articular cartilage tissue regeneration using chemokine-recruited BMSCs exist. Therefore, our purpose was to identify the chemokines that trigger autologous BMSCs to be sent from a bone marrow niche to the site of articular cartilage damage for cartilage regeneration.
Materials and Methods
Cell Culture
Human bone marrow was aspirated from the posterior iliac crest of eight healthy male and female donors aged 13—60 years with approval of the Institutional Review Board of our institution, and written informed consent was obtained from all patients. BMSCs were isolated based on their capacity to adhere to tissue culture plastic, and the isolated cells from each donor were cultured separately. BMSCs were cultured in DMEM-low glucose (DMEM-LG; Gibco, Grand Island, NY, USA) containing 10% FBS (Gibco), and a 1× antibiotic—antimycotic solution (Gibco). BMSCs (passage 2—4) were used in all experiments.
In Vitro Mimicking of Articular Cartilage Injury
After serum starvation of BMSCs [serum-free DMEM-LG containing 1× antibiotic—antimycotic solution, 1% (v/v) insulin, transferrin, and selenium supplement (ITS; Gibco)] for 24 h, cells were stimulated by adding 10 ng/ml of interleukin-1β (IL-1β; R&D Systems Inc., Minneapolis, MN, USA) or 10 ng/ml of tumor necrosis factor-α (TNF-α; R&D Systems Inc.) to the media for 4, 24, or 48 h. The stimulated cells were washed with phosphate-buffered saline (PBS) solution (Gibco) twice and then used for our experiments.
Reverse Dot Blot Hybridization
Sample DNA of 19 chemokine receptors, TATA box-binding protein (TBP), 18S rRNA (as a positive control), and rabbit GAPDH (as a nonspecific negative control) were amplified using RT-PCR (Table 1). Probe cDNA was synthesized from IL-1β or TNF-α-stimulated human BMSCs. Sample DNA was spotted onto Hybond-N+ nylon membranes (Amersham Pharmacia Biotech, Uppsala, Sweden). The probe cDNA was labeled using γ32P-dCTP (NEN Life Sciences, Boston, MA, USA). The membranes were hybridized with the probe cDNA for 15 h and then washed in 2× saline-sodium citrate (Sigma-Aldrich, St. Louis, MO, USA)/0.1% sodium dodecyl sulfate (Sigma-Aldrich). Hybridized probes were acquired with an integrated imaging plate (Fujifilm, Tokyo, Japan) and detected using a fluorescent image analyzer (FLA-7000; Fujifilm). Images of hybridized samples were quantified by densitometry (TINA pixel analyzer version 2.10; Raytest Isotopenme gerate GmbH, Straubenhardt, Germany).
Primer Sequences of Sample DNA for Reverse Dot-Blotting

S, sense primer; AS, antisense primer.
Wound Healing Assay
BMSCs were plated at 2,000 cells per well on silicon culture inserts (Ibidi, Integrated BioDiagnostics, Munich, Germany) attached to 24-well tissue culture plates (SPL Life Sciences, Pocheon-Si, South Korea) in DMEM-LG containing 10% FBS. The cells were incubated until they reached ~80% confluence, and the culture inserts were then removed. The cells were gently washed in PBS. BMSCs were treated with 500 ng/ml of IL-8 (R&D Systems Inc.), stromal cell-derived factor-1α (SDF-1α; R&D Systems Inc.), monocyte chemotactic protein-1 (MCP-1; R&D Systems Inc.), and macrophage inflammatory protein-3α (MIP-3α; R&D Systems Inc.) in serum-free media. Cells that migrated to the wounded area (diameter = 4 mm) were photographed and counted before and after chemokine treatment for 1, 3, 5, 7, 9, and 12 h. Cells were counted in triplicate by three independent observers.
Real-Time In Vitro Cell Tracking Assay
BMSCs were grown to ~80% confluence in silicon culture inserts on glass side disks [Live Cell Instrument Inc. (LCI), Nowon-gu, Seoul, Korea] in DMEM-LG containing 10% FBS. After serum starvation of BMSCs for 24 h, the culture inserts were removed from the glass disks. The cells were gently washed in PBS, and 500 ng/ml of chemokine (IL-8, SDF-1a, MCP-1, and MIP-3α) was added to BMSCs in each well containing serum-free media. To track BMSCs moving into the gap (diameter = 4 mm), cell-attached glass slides were incubated in 35-mm dish magnetic chambers (Chamlide™; LCI), and the migration of BMSCs was recorded using a microscope (CK-41; Olympus, Tokyo, Japan) equipped with a CCD camera (KP-D20BU; Hitachi, Tokyo, Japan) and capture software (TOMORO version FG 2.9.19; Olympus) every 5 min for 48 h. The total distance of cell migration and the average cell migration velocity were calculated using ImageJ software (version 1.44; NIH, Bethesda, MD, USA). The experiment was performed in triplicate.
In Vitro Chemotaxis Assay
BMSCs were serum starved for 24 h and inoculated at 10,000 cells per 200 μl of serum-free media into each 24-well culture insert system (8-μm pores) (Falcon; Becton, Dickinson and Company, Heidelberg, Germany). A volume of 400 μl of serum-free media containing 0.1% bovine serum albumin (BSA) (Sigma) or 500 ng/ml of IL-8 or MIP-3α was added into the outside of each insert, and the cells were incubated for 6 h. After incubation, the cells were washed in PBS and stained with crystal violet solution (Merck KGaA, Darmstadt, Germany). The nonmigratory cells were removed with a cotton-tipped swab (Millipore, Billerica, MA, USA), and stained cells (attached on the outside of the inserts) were counted using a microscope (Olympus). Cell counting was performed in triplicate by three observers.
Osteogenic or Chondrogenic Differentiation of Human BMSCs
After BMSCs were grown to confluence in growth medium, osteogenic differentiation was achieved with cultures in a basic medium containing 100 nM of dexamethasone (Sigma-Aldrich), 50 μM of ascorbic acid-2-phosphate (Sigma-Aldrich), and 10 mM of β-glycerophosphate (Sigma-Aldrich), and 10 ng/ml of IL-8 and MIP-3α each for 14 days. Ascorbic acid and the two chemokines were added daily, and the osteogenic differentiation medium was changed every 2—3 days. The osteoblast differentiation was confirmed by alkaline phosphatase (ALP) and von Kossa stainings. For ALP staining, the cells were stained with an alkaline dye solution (Sigma-Aldrich) for 30 min in the dark and counterstained with Mayer's hematoxylin solution (Sigma-Aldrich). For von Kossa staining, the cells were incubated in fresh 3% silver nitrate (Sigma-Aldrich). The experiment was performed in triplicate.
For chondrogenesis, BMSCs were dotted for 4 h at 8,000 cells per 10 μl of growth medium in each culture well. Chondrogenic differentiation was induced with cultures in DMEM-high glucose containing 50 μM of ascorbic acid-2-phosphate, 10 ng/ml of transforming growth factor-β3 (TGF-β3; R&D Systems Inc.), and 10 ng/ml of IL-8 and MIP-3α each for 21 days. Ascorbic acid, TGF-β3, and the two chemokines were added daily. Chondrogenesis was confirmed by hematoxylin and eosin (H&E) staining, Masson's trichrome staining, and RT-PCR for type II collagen A1 and aggrecan (Table 1). Differentiated micromasses were embedded in paraffin and stained with H&E and with Masson's trichrome solution (Sigma-Aldrich) to examine collagen fiber formation. The integrated optical density of total collagen in the micromass was measured using a microscope equipped with a CCD camera (BX-51; Olympus) and analyzed using MetaMorph® Microscopy Automation & Image Analysis Software (version 7.7.1.0; Molecular Devices Inc., Sunnyvale, CA, USA). The experiment was performed in triplicate.
In Vivo Chemotaxis Assay
An in vivo chemotaxis assay was performed in male nude mice by injecting human BMSCs and transplanted poly-lactide-co-glycolide (PLGA) scaffolds containing either chemokines or PBS. This experiment was conducted in accordance with the ethical guidelines for the Care and Use of Laboratory Animals of the Yonsei Biomedical Research Institute Committee.
Complexation of the Scaffolds and the Chemokines
PLGA scaffolds (porosity = 70%, pore size = 300 ~ 500 μm, diameter = 5 mm, thickness = 4 mm, disk type) were used as a chemokine carrier (21). Briefly, The PLGA (poly D, L-lactide-co-glycolic acid, MW = 97,000, Resomer; Sigma-Aldrich) scaffold was prepared by the novel sintering method. PLGA and NaCl (300—500 μm, 70% w/w) were homogeneously mixed and put into disk mold; the mixture was pressed at 150 MPa for 2 min and baked at 180°C for 35 min. For the salt removing, it was leached in deionized water with shaking for 7 days. Next, the scaffolds (diameter = 5 mm, thickness = 4 mm) were freeze dried for 48 h and sterilized by ethylene oxide gas. The scaffolds were soaked by repeated pipetting in PBS containing 500 ng/ml of IL-8 or 500 ng/ml of MIP-3α for 24 h at 4°C. PBS- or chemokine-containing scaffolds were then subcutaneously transplanted into the backs of nude mice.
Cell Labeling with NIR Nanoparticles
Near-infrared (NIR) nanoparticle labeling [NEO-LIVE Magnoxide 675, Extension/Emission (Ex/Em) = 675/700 nm; Biterials Co. Ltd., Itaewon-dong, Seoul, Korea] was performed according to the manufacturer's instructions. Briefly, human BMSCs were incubated until they reached a confluence of ~80%. NIR nanoparticles were collected by centrifugation at 13,000 × g for 10 min and washed with PBS twice and added to cells in DMEM-LG containing 10% FBS at a ratio of 4 mg/ml of growth medium. Then cells were incubated at 37°C, 5% CO2 for 48 h. After labeling, cells were washed with PBS twice and collected by trypsinization (Invitrogen, Carlsbad, CA, USA). Cell labeling was confirmed using a multispectrum in vivo fluorescence imaging system (Maestro 2; Cambridge Research & Instrumentation Inc., Cambridge, MA, USA).
Real-Time In Vivo Fluorescence Imaging Assay
Carrier free chemokine-containing scaffolds and NIR-labeled human BMSCs were implanted into 20 balb/c male nude mice (Orient Bio Inc., Seongnam, South Korea). A total of 2 × 106 BMSCs were injected into the tail veins, and scaffolds containing IL-8 or MIP-3α were subcutaneously transplanted into the backs of the mice. After scaffold transplantation, the skin was closed using black nylon sutures (Ethicon Inc., Somerville, NJ, USA). The movement of injected cells was tracked in real time every 2—3 days using a multispectrum in vivo fluorescence imaging system (Cambridge Research & Instrumentation, Inc.). The images were quantified using Maestro 2 software (Maestro2 Work Area; Cambridge Research & Instrumentation, Inc.). All mice were euthanized, and all scaffolds were harvested at 6 weeks. At necropsy, inserted scaffolds were closely examined, photographed, and harvested at 6 weeks. The scaffolds and adjacent tissues were evaluated for subdermal tissue formation and vascular infiltration. This experiment was performed at least in triplicate.
Confocal Laser Scanning Microscopy
Detailed observation using a laser scanning microscope (LSM 700; Carl Zeiss MicroImaging GmbH, Jena, Germany) was performed to confirm the presence of the injected NIR-labeled human BMSCs in the chemokine/scaffold complexes. All 5-mm-thick sections on the glass slides were stained with eosin solution (Sigma-Aldrich) and mounted to identify the cell nucleus. Eosin-stained sections were observed using laser scanning microscope with a laser beam of Ex = 630 nm/Em = 700 nm (Carl Zeiss).
Immunohistofluorescence Analysis
Immunohistochemical reactions using mouse anti-CD11b—FITC and CD45 (Abcam, Cambridge, UK) were performed. All sections were treated with 3% H2O2 (Duksan Pure Chemicals, Ansan, South Korea) for 10 min to inactivate endogenous peroxidase. Then, the sections were incubated with anti-CD11b—FITC (dilution factor = 1:100) or CD45 (dilution factor = 1:50) primary antibodies overnight. The sections were incubated with FITC-conjugated secondary antibody (dilution factor = 1:5,000) for 30 min at room temperature. After counterstaining with hematoxylin (Sigma-Aldrich), the sections were mounted, and the images were acquired using an inverted fluorescence microscope (IX-71, Olympus) and a digital camera (DP-71, Olympus).
Preparation of Biodegradable Biphasic Scaffold for Osteochondral Defect Model of Beagle
The poly lactic acid (PLA)/poly-L-lactide/β-tricalcium phosphate (β-TCP) scaffolds for the bone part were prepared with PLA powder (MW = 245,000; CM Research, Seoul, Korea) and β-TCP (100~300 μm; Berkeley Advanced Biomaterials, Berkeley, CA, USA) mixed with NaCl (diameter = 300~500 μm; Sigma-Aldrich) by the novel sintering method. Poly-l-lactide-co-ε-caprolactone (PLCL; MW = 328,000) was synthesized and used in a gelpressing method to fabricate scaffolds for the cartilage part. After PLA, β-TCP, and NaCl particles (300~500 μm, 70% w/w) were homogeneously mixed and placed in a cylindrical mold (diameter = 5.85 mm), the mixture was pressed at 150 MPa for 2 min at room temperature using a hydraulic press to yield a solid block that was further treated at 210°C for 30 min. PLCL synthesized in our laboratory was dissolved in tetrahydrofuran (THF) (5% w/v) and then mixed with NaCl particles (300~500 μm, 85% w/w). The THF was then evaporated in air to form the PLCL gels, which were subsequently pressed on the PLA/β-TCP blocks prepared above to combine two parts in a cylindrical mold (diameter = 6 mm). Next, the residual THF was evaporated for 24 h at room temperature, after which it was completely removed under vacuum for 72 h. The salts were then leached out by placing the gels in distilled, deionized water with constant shaking for 7 days. The resulting scaffolds were then freeze dried for 48 h, after which they were sterilized with ethylene oxide gas. The final product was 8 mm in length (PLA/β-TCP =7 mm, PLCL = 1 mm) and 6 mm in diameter.
Surgical Procedures and Histological Analysis in Osteochondral Beagle Defect Model
Male beagles (Orient Bio Inc.) were used as experimental animal models (n = 12) having a weight of constant 14~15 kg the same week of age to overcome individual variations. This experiment was also conducted in accordance with the ethical guidelines for the Care and Use of Laboratory Animals of the Yonsei Biomedical Research Institute Committee. Under general anesthesia using 2% isoflurane (Sigma-Aldrich), a medial parapatellar incision was made over the right knee, and the patella was everted. The 6-mm-diameter and 8-mm-deep cylindrical osteochondral defect, identical to the dimension of a biphasic scaffold, was made using an Osteochondral Autograft Transfer System (Arthrex, Naples, FL, USA) in the weight-bearing articular surface of the femoral medial condyle. The biodegradable biphasic scaffold composed of poly-l-lactide acid/β-TCP was press fitted into the defect of the knee and the surface of repaired defect flushed smoothly with the native articular surface. Each scaffold containing 5 μg of IL-8 or MIP-3α was inserted into the osteochondral defect site to evaluate the regeneration potential of the damaged bone and cartilage.
Statistical Analysis
All data are represented as mean ± standard deviation (SD) (n ≥ 3). The experimental groups were compared with the control group in triplicate. Statistical significance was determined using paired Student's t-tests, two-way ANOVA, and post hoc tests (pairwise comparisons) with a Bonferroni adjustment to control for errors according to experimental methods. Significance was defined as values of p < 0.05.
Results
Proinflammatory Cytokines Increased the Expression of Chemokine Receptors in BMSCs
As an articular cartilage is injured, the expression of IL-1β or TNF-α increases. These proinflammatory cytokines induce the release of chemokines, which can recruit various types of cells including BMSCs (26). Thus, to examine the changes in the expression of chemokine receptors by proinflammatory cytokines, an in vitro cartilage injury condition was induced using recombinant human IL-1β or TNF-α, and a reverse dot blot assay was performed on human BMSCs. The expressions of CCR2, CCR4, CCR6, CXCR1, and CXCR2 were significantly increased in stimulated BMSCs from more than two donors compared with those of the control group (p < 0.05) (Fig. 1 and Table 2). RT-PCR detected CCR7 and CCR10 in normal BMSCs from five donors. CCR2, CCR4, CCR6, CCR8, CCR9, CXCR1, CXCR4, CXCR6, and CXCR7 were expressed in human BMSCs obtained from three donors (data not shown). CCR2, CCR4, CCR6, CXCR1, and CXCR2 were expressed in normal human BMSCs. The expression of these chemokine receptors was also increased in human BMSCs stimulated by proinflammatory cytokines that are released into osteoarthritis (OA) cartilage (Table 2). The ligands of these receptors were screened based on the “Protein Knowledge base” (UniProtKB/Swiss-Prot, http://www.uniprot.org). The chemokines MCP-1 (a ligand of CCR2 and CCR4), MIP-3α (a ligand of CCR6), and IL-8 (a ligand of CXCR1 and CXCR2) were selected as the candidates inducing BMSC recruitment. We added SDF-1α in this study because SDF-1α/CXCR4 is the best known pathway that induces the recruitment of hematopoietic stem cells and MSCs within the bone marrow niche (25). Therefore, the following experiments were performed with MCP-1, MIP-3α, IL-8, and SDF-1α.

Increased expression of chemokine receptors by proinflammatory cytokines in human BMSCs. Black arrows indicate the increased chemokine receptors by proinflammatory cytokines compare with control. Reverse dot blotting was performed with serum-starved BMSCs that were stimulated by ether IL-1β or TNF-α. TBP and 18S rRNA were used as positive controls, and NC is a nonspecific negative control using rabbit GAPDH DNA for reverse dot blotting. Control: a group of probes from normal BMSCs; IL-4h, IL-24h, and IL-48h: a group of probes from BMSCs stimulated by IL-1β for 4, 24, and 48 h, respectively; TNF-4h, TNF-24h, and TNF-48h: a group of probes from BMSCs stimulated by TNF-α for 4, 24, and 48 h, respectively.
The List of Chemokine Receptors Expressed by RT-PCR in Normal BMSCs and Stimulated BMSCs in Reverse Dot Blots

The maximum value of increased fold change compared with that of the control group is shown in parentheses.
Chemokines Enhanced Cell Migration
The migratory effects of four chemokines were investigated using the wound healing assay. The number of cells that migrated into the gap increased significantly by treating all the chemokines tested; chemokine efficacy increased over the range of chemokines tested from MCP-1, SDF-1α, and MIP-3α to IL-8 (Fig. 2A) compared with that of the control group. Live-cell tracking of the wound healing assay was performed to determine the total distance and velocity of migrating BMSCs induced by the four chemokines. All chemokines (500 ng/ml) not only increased the total distance by 2.5- to threefold in all our test groups compared with the control group, but also enhanced the migration velocity of BMSCs. In particular, the velocity induced by MIP-3α was greater than that induced by the other chemokines when compared with the velocity of the control group (Fig. 2B). Cell proliferation was unaffected by the four chemokines tested, and no cytotoxicity was observed by the chemokines at 0—500 ng/ml (data not shown). A Transwell assay was performed to examine the chemotactic effect of chemokines on BMSCs in vitro. The chemotaxis of BMSCs was significantly induced by 500 ng/ml of MIP-3α (3.4-fold), 500 ng/ml of IL-8 (3.3-fold), 500 ng/ml of MCP-1 (2.1-fold), and 500 ng/ml of SDF-1α (1.3-fold) compared with that of the control group (Fig. 2C, D). MIP-3α (3.4-fold) and IL-8 (3.3-fold) enhanced the chemotaxis of BMSCs more than the other chemokines tested. Thus, IL-8 and MIP-3α were selected as the most potent candidates for recruiting BMSCs. The chemotaxis of BMSCs toward IL-8 or MIP-3α was inhibited by the removal of the chemokine gradient (Fig. 2E).
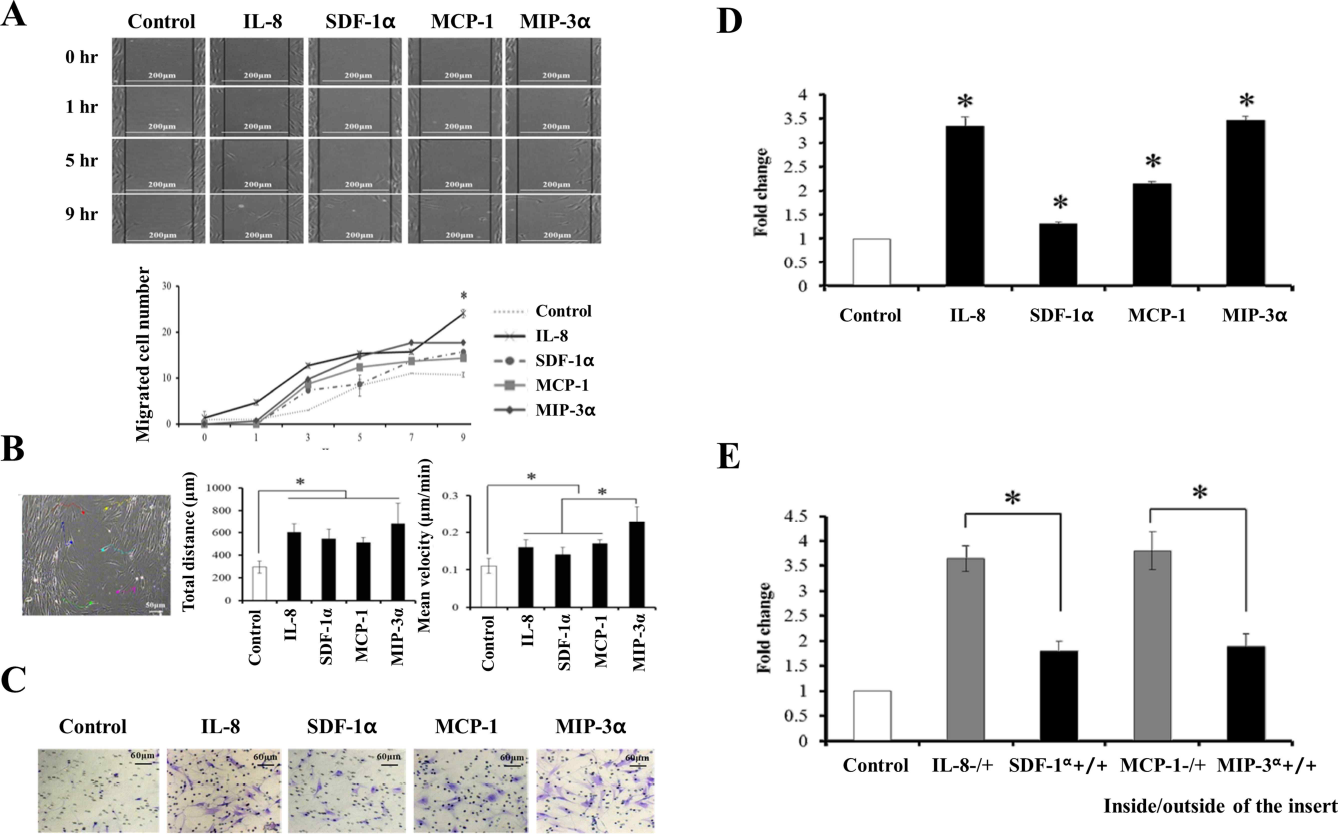
The effect of chemokines on BMSC migration and in vitro chemotaxis. After treating BMSCs with 500 ng/ml of chemokines, the wound healing assay was performed (A, left), and the cells that migrated into the gap were counted (A, right). Live-cell tracking was performed using the wound healing model for 48 h. Migrating BMSCs were pointed and analyzed (color point, B, left), and their total migrated distance (B, middle) and mean migrating velocity (B, right) were calculated. Serum-starved BMSCs were inoculated into the culture inserts (8-μm-pore membranes), and the chemokines were added into the inserts. Cells that migrated to the outside of the membrane were stained (C) and counted (D). The BSA or chemokine gradients were removed by adding BSA or chemokine to both the inside and outside of the inserts. Cellular migration was counted (E). The control was 500 ng/ml of BSA.
Chemokines Did Not Affect Osteogenic or Chondrogenic BMSC Differentiation
ALP, an early stage osteogenic marker, was increased in the osteogenic differentiated groups treated with osteogenic media with or without either IL-8 or MIP-3α at days 3 and 7 compared with the undifferentiated control (Fig. 3A). The von Kossa staining revealed that calcium deposition, a late osteogenic marker, was also increased with progression of the osteoblast differentiation in the groups treated with an osteogenic medium alone or with an osteogenic medium containing chemokines at days 7 and 14 compared with that of the control group. However, in the groups treated with both chemokines and osteogenic media, early or late osteogenic markers were not increased (Fig. 3B). The expression of total collagen fibers increased during chondrogenesis in the micromass cultures exposed to a chondrogenic medium, but there was no additional effect of the chemokines at days 7 and 14 (Fig. 3C, D). The expression of chondrogenic marker genes, such as glycosaminoglycan (GAG) and type II collagen, was examined by RT-PCR. GAG and type II collagen expressions increased during chondrogenesis induced by a chondrogenic medium, but there was no significant effect of the two chemokines on chondrogenesis at days 7 and 14 (Fig. 3E).

Osteo/chondrogenesis of BMSCs using two candidate chemokines. Osteogenesis was induced using an osteogenic medium with or without the indicated chemokines. At days 3 and 7, ALP levels were checked by ALP staining assay (A). At days 7 and 14, the calcium content was checked by von Kossa staining (B). Chondrogenesis was induced using a chondrogenic medium containing TGF-β3 with or without the indicated chemokines. At days 7 and 14, collagen fiber expression was checked by Masson's trichrome staining (C, blue-colored area). The total area of trichrome-stained collagen fibers (blue-colored area) was measured with a Meta-morph image analyzer (D). At days 3, 7, and 14, the expression of aggrecan and type II collagen was determined by RT-PCR (E).
Two Candidates Recruited BMSCs In Vivo
To confirm the chemotactic effect of the two candidates (IL-8, MIP-3α) on BMSCs, we observed in vivo chemotaxis in nude mice. From day 1 to 1 week following injection, we found a significant number of human BMSCs in the lung. At 2 weeks later, BMSCs were located in the liver and spleen. At week 3, we found that human BMSCs had moved into the scaffolds containing PBS, IL-8, or MIP-3α. Interestingly, the NIR signal of labeled BMSCs at chemokine-containing scaffolds increased at 4 to 6 weeks postinjection; IL-8 was a more effective chemokine than MIP-3α (Fig. 4A, arrows). The BMSC signal was not increased at the PBS-containing scaffold (Fig. 4A, triangle). There was no inflammatory reaction at the transplanted sites of all mice through 6 weeks postoperation. To confirm that the chemotaxis of BMSCs toward IL-8 or MIP-3α is unrelated to the position of the transplanted chemokine—scaffold complexes, the transplantation position of the chemokine—scaffold complexes was altered, as shown in Figure 4B. Injected human BMSCs moved toward IL-8- and MIP-3α-containing scaffolds independent of location (Fig. 4B, arrows). The total detected signals of human BMSCs (total counts/exposure time) at the scaffolds were quantified at week 6. The total signal of BMSCs at the IL-8-containing scaffold increased by 5.3-fold (p < 0.05) and that at the MIP-3α-containing scaffold increased by 4.1-fold (p < 0.05) compared with the PBS control (Fig. 4C). Signal quantification using other factors such as the average signal (scaled counts/s) and the max signal (counts) showed similar results to the total signals at the IL-8- or MIP-3α-located sites (data not shown). At week 6, all transplanted scaffolds were harvested and examined. The BMSC signals in IL-8- or MIP-3α-containing scaffolds increased compared with both PBS-containing scaffolds and scaffolds that were obtained from non-cell-injected mice. The spleens were used as a positive or negative control for the NIR signal (Fig. 4D). In the gross morphological analysis, chemokine-containing scaffolds were more compactly fused with subcutaneous tissues, and there were more blood vessels in the scaffolds compared with the PBS-containing control scaffolds. However, there was no significant difference between the IL-8- and MIP-3α-containing groups (Fig. 4E).
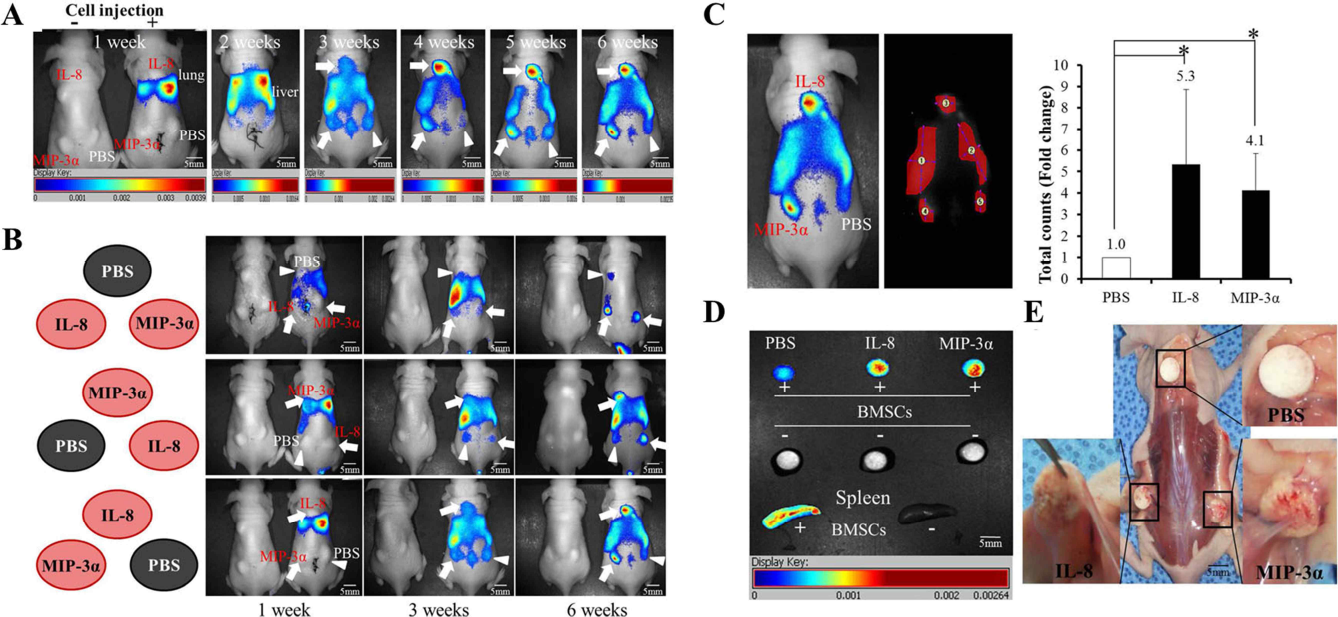
Real-time tracking and detected signal quantification of BMSCs using an in vivo imaging system. The movement of labeled human BMSCs toward chemokine-containing scaffolds (arrows) or a PBS-containing scaffold (triangle) in nude mice was detected for 6 weeks (A). The location of the transplanted scaffolds containing chemokines or PBS was altered (B, left panel), and the movement of BMSCs toward chemokine-containing scaffolds (arrows) or a PBS-containing scaffold (triangle) was tracked in real time for 6 weeks (B, right panel). At 6 weeks postinjection, all mice were euthanized. The signals generated by the recruited BMSCs toward the chemokine-containing scaffolds were detected (C, left panel) and quantified (C, right panel). Transplanted scaffolds were harvested and examined (D). All scaffolds were photographed and morphologically analyzed (E).
Chemokine-Recruited BMSCs Induced Tissue Formation
At 6 weeks postoperation, all the scaffolds were harvested from euthanized mice. H&E staining was performed to examine inflammation, tissue formation, and the recruitment of inflammatory cells and BMSCs to the PBS-or chemokine-containing scaffolds. Vascular infiltration was occasionally seen on the edge of the PBS-containing scaffolds, and various cell types including fibroblastic cells and macrophages around these blood vessels were also found. There was no cell infiltration or blood vessel formation on the inner part of the PBS-containing scaffolds. Vascular infiltration was found in both the edge and the interior of IL-8- or MIP-3α-soaked scaffolds; a large number of fibroblast cells were found surrounding blood vessels and inside the pores of the scaffolds. Additionally, active extracellular matrix (ECM) formation by these fibroblast cells, potentially recruited human BMSCs, was found in the interior of the scaffolds containing chemokines [Fig. 5, red (C) and green (D) squares]. There were only a few monocytes or macrophages in either the IL-8- or MIP-3α-containing scaffolds, which was similar to that observed with the PBS control. Thus, no significant inflammatory reaction was observed in any group, and tissue formation was significantly increased by fibroblast cells, which may represent the chemokine-recruited BMSCs (Fig. 5).

Histological analysis using H&E staining of harvested scaffolds. At 6 weeks, the harvested samples were sectioned and analyzed by H&E staining. Squares in black (A), blue (B), red (C), or green (D) indicate the magnified regions.
Human BMSCs and Immune Cells Were Present in the Scaffolds Containing Chemokines
To determine whether the recruited fibroblast cells in the scaffolds are injected human BMSCs, all the scaffolds were observed using confocal microscopy. The NIR signal of the injected cells was strongly detected at the edge of the scaffolds containing chemokines, but the NIR signal was not observed in the scaffolds soaked in PBS (Fig. 6A). NIR-labeled human BMSCs were found within the chemokine-containing scaffold pore surfaces, which were filled with fibrous ECM. No NIR signal was detected on the inside of the scaffolds containing PBS (Fig. 6B). These data suggest that injected human BMSCs moved toward either IL-8-or MIP-3α-containing scaffolds and then secreted local ECM components. According to UniProtKB/Swiss-Prot, IL-8 attracts neutrophils, basophils, and T-cells, but not monocytes. In addition, MIP-3α attracts macrophages, lymphocytes, and neutrophils. Thus, to examine the influx of immunocytes into the scaffolds containing either IL-8 or MIP-3α, immunofluorescence was performed using a CD11b-specific antibody to detect macrophages and neutrophils, and a CD45-specific antibody was used to detect Bor T-cells. CD11b-expressing cells, such as macrophages and neutrophils, were rarely found around the blood vessels in any group (Fig. 6C). More CD45-positive B- or T-cells were found around the vessels in scaffolds containing MIP-3α than in those containing PBS or IL-8 (Fig. 6D). However, there was no significant difference among all the groups.

Confirmation of recruited human BMSCs and immune cells in scaffolds. At 6 weeks, the harvested samples were sectioned and analyzed by confocal microscopy. Black arrows indicate NIR-labeled human BMSCs at the edge (A) or in the interior (B) of the scaffolds. The harvested samples were sectioned and analyzed by immunostaining with CD11b- (C) or CD45 (D)-specific antibodies. BF, bright field.
Repair of Articular Osteochondral Defects Using Chemokine-Containing Scaffolds in a Beagle Model
Animal studies were performed to evaluate the effects of IL-8 and MIP-3α on the regeneration of damaged cartilage in a beagle model. We observed the gross morphology of the regenerated articular cartilage in osteochondral defects of a beagle model at 12 weeks postinjection of chemokine-containing scaffold. In the IL-8-containing scaffolds, the osteochondral defect was almost entirely filled with cartilage-like tissues to the surrounding cartilage compared to that of the PBS-containing scaffolds. The PBS-containing scaffolds showed a slight depression in the center of the regenerated surface. MIP-3α-containing scaffolds showed entirely filled tissues with a smooth surface, but the regenerated tissues were shown to be bloody in color (Fig. 7A, upper panel). In histological analysis, PBS-containing scaffolds showed fibrous tissue formation and irregular surface in the osteochondral defect site, whereas IL-8-containing scaffolds showed cartilage-like tissue formation with the presence of lacunae and a smooth surface in the osteochondral defect site. However, there was no lacunae formation in the MIP-3α-containing scaffold, although the defect site was entirely filled and showed a smooth surface in morphology. We also observed that there were no inflammatory changes in synovium membrane and scaffold-implanted sites (Fig. 7A, lower panel). Masson's trichrome staining showed that the regenerated cartilage in IL-8-containing scaffolds was entirely filled in morphology and higher in collagen contents than that of PBS-containing scaffolds. However, MIP-3α-containing scaffolds did not show collagen synthesis at the regenerated cartilage site although the site was entirely filled with tissues (Fig. 7B). μCT results showed that PBS-containing scaffolds (21.58 ± 3.25) did not induce massive bone regeneration. In contrast, the IL-8- (36.23 ± 2.47) and MIP3-α-containing scaffolds (29.99 ± 1.75) showed superior bone regeneration compared to the PBS group (Fig. 7C). Consequently, these results indicate that IL-8 and MIP3-α could enhance the bone regeneration by recruiting BMSCs into the osteochondral defect site.

Effects of IL-8 and MIP-3α on cartilage regeneration using an osteochondral defect in a beagle model. (A) At 12 weeks postinjection of chemokine-containing scaffolds, all beagles were euthanized. The photographs show osteochondral defect sites of the PBS group, IL-8 group, and MIP-3α group (upper panel). The histological appearance of the osteochondral defects was observed using H&E staining (lower panel, 100×). Black arrows indicate the osteochondral defect site. (B) Masson's trichrome staining was performed to check collagen synthesis of the cartilage defect site. Collagen fibers are stained blue (upper panel, 8×; lower panel 40×). (C) μCT results show bone regeneration of osteochondral defect sites at 12 weeks postimplantation of PBS-, IL-8-, or MIP-3α-containing scaffold (depth: 10 mm; width: 6 mm). The above data have been confirmed on three beagles tested per each group, while the data shown are representative. BS, biphasic scaffold.
Discussion
Although several techniques are used for cartilage repair, there is currently no effective treatment for complete repair of injured articular cartilage. A sufficient supply of chondrogenic progenitor cells is necessary for the successful regeneration of an articular cartilage defect. BMSCs have higher proliferative ability than articular chondrocytes. In addition, they are well characterized, easily available, and can meet the cellular demand for cartilage tissue regeneration (4). Unfortunately, the in vitro expansion of primary cells, including articular chondrocytes or BMSCs, has several disadvantages. In vitro expansion to obtain adequate cell numbers is known to result in dedifferentiation of articular chondrocytes. Similarly, upon repeated subculture in vitro, primary BMSCs lose their multipotency and self-renewal capacity, leading to senescence (5,23). It has been reported that intravenously injected MSCs can migrate to the injured site of various tissues, such as bone fractures and myocardial infarctions (9,38). However, the homing ability of MSCs can be decreased by in vitro culture and long-term culture necessary for the preparation of these cells for clinical use (34). In this regard, if autologous BMSCs with osteo/chondrogenic potential could be recruited to sites of damage without ex vivo expansion, complete regeneration of damaged cartilage is possible.
In the cartilage defect model, secretion of proinflammatory cytokines, such as IL-1β and TNF-α, increases with increasing degrees of damage. Secretion of these cytokines results in the secretion of chemokines or matrix metalloproteinases (MMPs) from synovium and chondrocytes as an early repair response (27,31). It has also been reported that five CC receptors, including CCR1, CCR2, CCR3, CCR4, and CCR5, and four CXC receptors, including CXCR1, CXCR2, CXCR4, and CXCR5 were expressed in human BMSCs. Thus, we focused on chemokine recruiting of autologous BMSCs from OA patients for articular cartilage repair. In this study, expressions of the CCR2, CCR4, CCR6, CXCR1, and CXCR2 genes were significantly increased by stimulation with IL-1β and TNF-α. Some chemokines are secreted after tissue injury, followed by MSC expression of chemokine receptors, leading to migration of cells to specific inflammatory sites (11). Thus, MCP-1, MIP-3α, and IL-8, which are the main ligands of CCR2, CCR4, CCR6, CXCR1, and CXCR2, were selected as recruiting factors for BMSCs.
When MSCs are transplanted, the possibility of overgrowth is a concern, as overgrowth remains poorly understood and controversial. The long-term in vitro culture of human MSCs can alter their biological properties, leading to tumorigenesis. These MSCs showed an increased proliferation rate. Moreover, long-term cultured MSCs induced tumor formation in mice (35). It has also been reported that primary MSCs did not form teratomas when they were transplanted (15). It has been reported that chemokines, including regulated on activation, normal T expressed and secreted (RANTES) and interferon-γ inducible protein 10 (IP-10), induce human MSC proliferation and migration (32). However, MIP-3α and IL-8 did not affect human BMSC proliferation in this study. Therefore, we suggest that these chemokines can trigger direct in vivo homing of fresh BMSCs to the cartilage defect site without the concern of malignant tumor formation.
The mechanism of leukocyte chemotaxis toward multiple chemokines released from inflammatory sites is well known (28). Until now, however, the homing mechanism of BMSCs was only partially understood. The chemokine receptor is a family of seven-transmembrane, G-protein-coupled receptors (GPCRs). There are two major chemokine/chemokine receptor signaling pathways in various cell types including BMSCs. The first is the Gαi subunit of GPCR, which induces tyrosine kinase cascades including the FAK-Src pathway. The second is the Gbg subunit, which induces the PI3K-AKT signaling pathway (12,40). These signaling pathways trigger the rolling and homing of BMSCs by expression of adherent molecules, such as β1-integrin, P-selectin, vascular cell adhesion molecules, and certain MMPs (14,19). In this study, in vitro migration or chemotaxis of human BMSCs was significantly induced by MCP-1, MIP-3α, IL-8, and SDF-1α. IL-8 and MIP-3α were particularly effective in enhancing migration capacity, such as migration velocity and distance, as well as chemotaxis in vitro. Although a detailed mechanism was not examined in this study, the in vitro chemotaxis of BMSCs toward either IL-8 or MIP-3α was reduced by focal adhesion kinase (FAK) silencing using siRNAs (data not shown). Thus, it appears that the chemotactic effect on BMSCs is related to previously identified signaling pathways, including FAK.
It has been reported that BMSC homing is induced by several growth factors and chemokines (10,16). Although these growth factors can recruit BMSCs, the expectation of complete healing of hyaline cartilage damage and a successful clinical result are very difficult to achieve as growth factors have multiple functions. These functions include effects on cell proliferation or multilineage differentiation and enhancing neovascularization. There is a limited understanding of their effects in vivo (13,30). As shown in the results, neither IL-8 nor MIP-3α affected the osteoblast differentiation or chondrogenesis of human BMSCs. Multipotent BMSCs can initiate niche-specific differentiation and local tissue formation when they are stimulated by environmental signals, including cytokines and the surrounding ECM in vivo (3,20). It has been reported that BMSCs migrated toward cutaneous T-cell-attracting chemokine from damaged skin and then regenerated skin tissues following differentiation to keratinocytes (18). Therefore, if IL-8 or MIP-3α is injected into cartilage defect sites, BMSCs can be recruited and differentiate into chondrocytes by cartilage niche-specific stimulation. In this regard, it appears that IL-8 and MIP-3α recruit BMSCs for articular cartilage repair, and neither of these chemokines has any effect on cell differentiation or proliferation.
Previous reports show that autologous primary BMSCs move toward chemoattractants released in various damaged tissues in vivo and then enhanced repair of injured heart, bone, and muscle (5,24,29). BMSCs are also potent chondrogenic progenitors for hyaline cartilage repair (33). Using a real-time in vivo imaging system, the in vitro chemotactic effect of either IL-8 or MIP-3α on human BMSCs was verified in nude mice. Human BMSCs injected into the tail veins moved via systemic circulation to the heart, lung, liver, and spleen (in that order), followed by BMSC attraction into IL-8- or MIP-3α-containing scaffolds under the skin. The injected BMSCs were recruited to the chemokinesoaked scaffolds more than to the PBS-infused scaffolds independent of implantation site. These results suggest that systemically circulating human BMSCs were attracted by either IL-8- or MIP-3α-induced signaling. Unlike the PBS-containing scaffolds, the chemokine-containing scaffolds were compactly unionized with infiltrations of host subcutaneous tissues and blood vessels. The formations of the subcutaneous tissue and keratin-like tissue were also induced by either IL-8 or MIP-3α-recruited BMSCs within the scaffolds. In addition, few BMSCs moved to the suture sites to heal the incision wound. In the results of an animal study using beagles with the osteochondral defects at a site of articular cartilage, we confirmed that IL-8 and MIP-3α can be used as good candidates with therapeutic potential for regeneration of damaged cartilage. These results suggest that IL-8- or MIP-3α-recruited BMSCs move to and differentiate into a local niche-specific cell type and then regenerate damaged tissues such as OA cartilage.
The wound healing process is complex and consists of inflammation, cell proliferation, and ECM remodeling. An excessive inflammatory response induces the fibrillation of the articular cartilage leading to cartilage degradation (8). Articular cartilage damage in OA is caused by phenotypic alterations of articular chondrocytes (36). Although OA does not fit the classic definition of inflammatory disease, it has been reported that OA-stimulated chondrocytes show similar behavior to active macrophages (2). BMSCs can inhibit the production of proinflammatory cytokines and promote the survival of damaged cells. BMSCs can also interact with immune cells to regulate the immune response (22). In our results, immune cells such as macrophages, neutrophils, and lymphocytes were not observed in the interior of either IL-8- or MIP-3α-containing scaffolds.
Conclusion
Cartilage regeneration requires a large number of fresh articular chondrocytes or BMSCs with chondrogenic potential. However, ex vivo expansion to obtain the required number of cells causes the dedifferentiation of chondrocytes or the loss of the native properties of BMSCs. Therefore, the in situ recruitment of BMSCs using IL-8 or MIP-3α may allow us to obtain a large number of primary BMSCs for articular cartilage repair. Physically and structurally adequate biodegradable scaffolds enable the adherence and chondrogenic differentiation of BMSCs as well as the hyaline cartilage formation necessary to regenerate damaged cartilage. Thus, both IL-8 and MIP-3α may help overcome the difficulties presented by the current methods including the fibrillation of regenerated cartilage, the loss of function of in vitro expanded cells, and the limitation of donor sites for transplantation.
Footnotes
Acknowledgments
This work was supported by a faculty research grant of Yonsei University College of Medicine (6-2009-0153) and by a grant (Code: A110328) from the Korea Health Technology R&D Project, Ministry of Health & Welfare, Republic of Korea. The authors declare no conflicts of interest.
